Abstract
Sulfur dyes are water insoluble and require reduction as well as solubilization before application. These dyes are used for dyeing cotton with heavy shades, primarily because of good light and wash fastness at low cost. Sodium sulfide used for reduction is highly toxic, releasing sulfur products in drained out liquor, including hydrogen sulfide gas. Enzymes capable of reducing sulfur dyes were studied to replace sodium sulfide in dyebath formulations. It was found that a few enzymes viz. protease, pectinase, lipase, and catalase can effectively reduce all sulfur dyes. Dyebath potential, color strength (K/S), reduction bath stability, and colorfastness of dyeing were comparable among the reducing systems, thus demonstrating the feasibility of enzymatic cotton dyeing with sulfur dyes.
Introduction
Sulfur dyes are characterized by disulfide (S-S) or oligo sulfide [(S-S)n] bonds between aromatic residues1–7and are used to inexpensively produce deep shades on cotton. Application of these dyes on cotton yields overall good to excellent colorfastness, except that against chlorine. Because of their insolubility in water, sulfur dyes need to be first reduced and solubilized at boil with sodium sulfide to form alkali soluble thiol groups, which show substantivity for cotton.8–14 Subsequent oxidation reverts the reduced and solubilized dye to its parent insoluble form, leading to the regeneration of di- and poly-sulfide linkages in situ on cotton.11,14–16 Unused sulfur dyes are reverted back to a water insoluble state after oxidation and may be eliminated by adsorption onto activated sludge in wastewater treatment plants. 17 However, sodium sulfide, used for reduction and solubilization of sulfur dyes, is toxic; it releases hydrogen sulfide in air and dissociates to several toxic byproducts. Therefore, replacement of sulfide with an alternative, eco-friendly reducing system is highly desirable.
Application of enzymes in chemical processing of textiles is rapidly gaining wide recognition because of their non-toxic and eco-friendly characteristics.18–23 Several oxidoreduc-tase and hydrolase type enzymes might provide the desired reduction potential (around –450 mV) for reducing sulfur dyes in alkaline media.15,20,23–27 Protease, pectinase, and lipase carry out nucleophilic attacks on substrates containing structurally analogous Asp-His-Ser triads.18,27–30 In contrast, catalase completes its catalytic cycle in two two-electron reductive and oxidative processes.18,19,30–33
Protease, pectinase, lipase, and catalase were applied separately, along with sodium hydroxide in place of sodium sulfide, to verify their effectiveness on the reduction of sulfur dyes prior to cotton dyeing. Surface color strength and colorfastness of this enzymatically-dyed cotton gave similar results to conventional dyeings obtained using sodium sulfide.34–37
Laccase, an oxidoreductase type enzyme, is an oxidizing agent. Potassium dichromate, used in combination with acetic acid for oxidation of reduced sulfur dyes on cotton, results in deposition of chromium on the fabric, giving it a harsh handle; hexavalent chromium remaining in the resulting wastewater causes problems for aquatic organisms and is carcinogenic. Laccase in acidic medium can effectively oxidize sulfur dyes on cotton fabric and in the bath; the resulting precipitated sulfur dyes can then be reused for dyeing. 38
In the present work, cotton was dyed with sulfur dyes, reduced and solubilized separately with sodium sulfide or protease, pectinase, lipase, or catalase under alkaline conditions. The reduction potential of each reduction bath at various stages of dyeing was measured. Color strength of dyeings, as well as colorfastness of the dyed cotton, were assessed and compared. Amylase, cellulase, and laccase were also studied, but these did not have the required reduction potential and were excluded from this report.
Experimental
Materials and Equipment
Thoroughly pretreated cotton fabric (ends/dm: 362, picks/ dm: 284, warp: 20s, weft: 30s, and GSM: 156) from Auro Textile Mills was used. Ten granular and non-reduced sulfur dyes with their Colour Index (C.I.) name and numbers as well as respective λmax values, viz. Black 1 (C.I. 53185, 620 nm), Blue 4 (C.I. 53235, 600 nm), Green 1 (C.I. 53166, 400 nm), Red 10 (C.I. 53228, 500 nm), Yellow 9 (C.I. 53010, 400 nm), Blue 7 (C.I. 53440, 620 nm), Green 22 (C.I. 53451, 400 nm), Green 8 (C.I. 53175, 400 nm), Brown 8 (450 nm) and Green 12 (C.I. 53045, 400 nm), were procured from Sulfast. Sodium sulfide and sodium hydroxide were procured from SDFCL. Enzymes, viz. alkaline protease (Maps India Ltd.), alkaline pectinase (Clariant), alkaline lipase (Noor Creations), and catalase (Rossary Biotech) were used in this study. Equipment, including a water bath, computer color matching software (Datacolor Check), lightfastness tester (Paresh Engineering Works), wash-fastness tester (R B Electronics), and crockfastness tester (Prolific Engineers) were used. A reduction (redox) potential and pH meter (Century Industries Ltd.) was used to assess redox potential and pH of the reduction baths. The redox potential was measured using an Ag/AgCl reference electrode and a Pt-Ag/AgCl combined electrode filled with saturated KCl solution.
Procedures
Cotton was dyed with various sulfur dyes to give 5 wt % shades. Reduction baths were prepared separately using sodium sulfide alone (2× the dye weight), as well as with various enzymes at 0.25, 0.5, 0.75, 1, 1.25, 1.5, 1.75, and 2 wt % concentrations along with sodium hydroxide (1 wt %), and the baths were heated to 90 °C. A sulfur dye (5 wt % with respect to cotton) was added to each of these baths and stirred thoroughly until completely reduced and solubilized. Cotton fabric was dyed in these baths for 2 h at 90 °C. The dyed cotton samples were thoroughly washed, followed by oxidation with potassium dichromate and acetic acid (1 wt % each) at 50–60 °C for 30 min. Soaping was then performed with nonionic detergent and soda ash (0.5 wt % each) at a liquor ratio (LR) of 30:1 for 30 min at 90 °C. Finally, samples were thoroughly washed and dried. Redox potentials and pH of baths were assessed at different stages of dyeing (i.e., before dye addition and after dye reduction, as well as at the end of dyeing) to maintain the required reduction potential, allowing the dye to remain reduced and solubilized until dyeing was complete. Stability of the reduction baths were also studied in presence and absence of dye.
The dyed cotton was tested for lightfastness, washfastness, and crockfastness. Cotton samples (3 × 1 cm), dyed with various sulfur dyes using various reducing systems, were placed in the lightfastness tester along with eight blue-dyed control samples (SDC) and their fading characteristics were studied along with the controls against xenon arc exposure. The samples were then compared to the change in color gray scale and graded from 1 to 8.
Washfastness grades were evaluated according to the ISO 3 method. Half of the dyed samples (10 × 4 cm) were sandwiched between white cotton and wool fabrics (5 × 4 cm each) followed by stitching all three fabrics around their edges. Treatment baths were prepared by dissolving laundry soap (0.5 g) along with soda ash (0.2 g) in 100 mL water. The sandwiched samples were treated in these baths for 30 min at 60 ± 2 °C, and finally washed and dried. The exposed part was used for change in color, while the stitches were opened and compared for staining of cotton and wool using the gray scale.
Crockfastness was evaluated using the crockfastness tester. Dyed cotton samples (5 × 5 cm) were prepared, and mounted and gripped on the tester. They were then rubbed for 10 cycles against white cotton fabric in dry and wet states. The extent of fading and staining (graded 1–5) was evaluated using the gray scale.
Results and Discussion
Sulfide-based reduction baths showed overall reduction potentials around –560 mV before addition of dye, –535 mV after reduction, and –515 mV at the end of dyeing. Initial studies showed that the effective concentration of sulfide was twice that of the dye to achieve optimal color strength on cotton at pH ∼ 11 when dyeing for 2 h at 90 °C (Table I). 34
Influence of Sodium Sulfide on Dyebath Status and K/S with Green 1
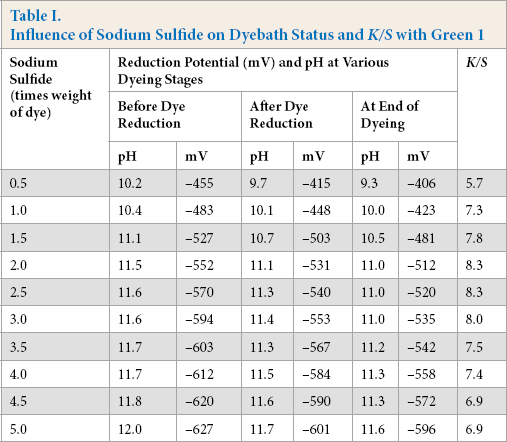
To study the ability of various enzymes to reduce sulfur dyes, reduction baths were prepared with enzyme (1×, same weight as dye) and sodium hydroxide (1 wt %), and the fabric was dyed for 2 h at 90 °C. Five sulfur dyes were initially tried in the enzymatic reduction system. The reduction potentials of baths at different stages of dyeing and the color strength of cotton were assessed (Table II).
Overall Dyebath Status and K/S of Cotton in Enzymatic Reduction Baths a
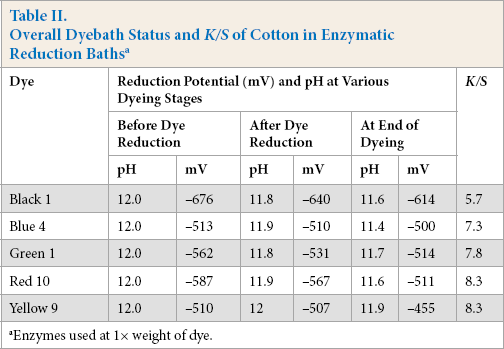
Enzymes used at 1× weight of dye.
The enzymes studied generated the required reduction potential in alkaline media comparable to that of using sulfide to reduce and solubilize sulfur dyes. The initial trials of sulfur dye reduction with these four alkaline enzymes and five dyes gave very similar average K/S values (Table II). Subsequently, dyeing parameters with each enzyme, including concentration, time of dyeing, and temperature, were optimized and were found to be the same as that of sulfide, except for concentration. While sulfide showed optimal color strength on cotton at twice the dye concentration, optimal concentrations of protease, pectinase, lipase, and catalase at alkaline pH were 0.25×, 34 0.5×, 35 0.5×, 36 and 0.5× 37 the dye concentration used, respectively.
Comparative Performance of Sodium Sulfide and Enzymes
Reduction Potential and pH
Reduction potential and pH of reduction baths prepared with optimized reducing agents, sodium sulfide (2× the dye concentration) and alkaline enzyme formulations—protease (0.25×), pectinase (0.5×), lipase (0.5×), and catalase (0.5×)— at different stages of dyeing with Green 1 dye are detailed in Table III.
Reduction Potential at Various Dyeing Stages with Green 1
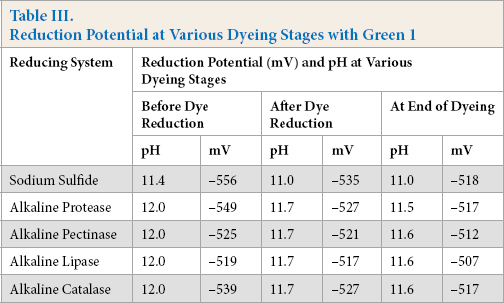
Use of optimized sulfide, as well as the alkaline enzyme formulations, developed nearly identical reduction potential in the dyebath, adequate for complete reduction of sulfur dyes. The reduction potential remained quite high even at the end of dyeing, confirming that the reduced status of the dye was maintained throughout the dyeing process. Tough there were some differences in reduction potential developed between these two systems, they were not substantially different. The other sulfur dyes tested gave similar results.
Comparative Color Strength
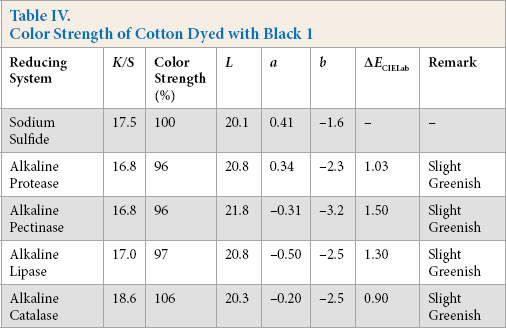
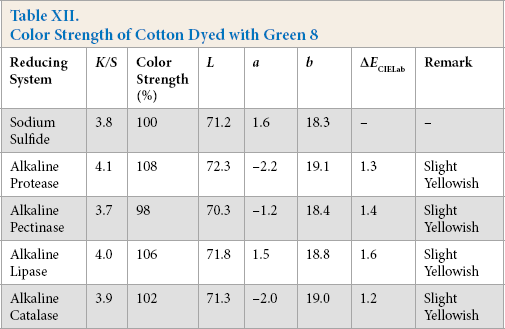
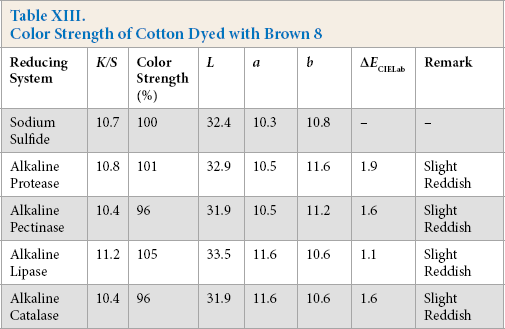
Comparative color strength (CS) of cotton dyed with ten sulfur dyes using sulfide and alkaline enzyme systems were studied and are shown in Tables IV–XIII. Although the color strength of cotton dyed using these reducing systems remained comparable, the hue of the dyed cotton using the enzymatic systems showed marginal shifts in hue (ΔECIELab) for the dyes used, probably due to over-reduction of the dyes. This shift in hue was not due to over-oxidation as oxidation was carried out with acidic potassium dichromate for both sulfide and alkaline enzyme reducing system treatments. However, the shifts, observed instrumentally, were not prominent on visual examination (Scheme 1).

Comparative color strength on cotton after various enzymatic reduction treatments.
Color Strength of Cotton Dyed with Black 1
Color Strength of Cotton Dyed with Blue 4
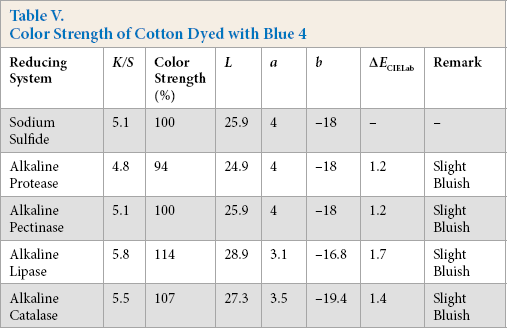
Color Strength of Cotton Dyed with Green 1
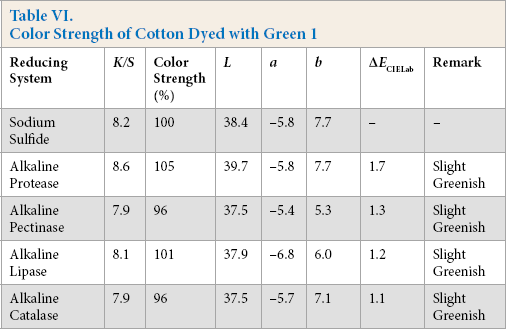
Color Strength of Cotton Dyed with Red 10
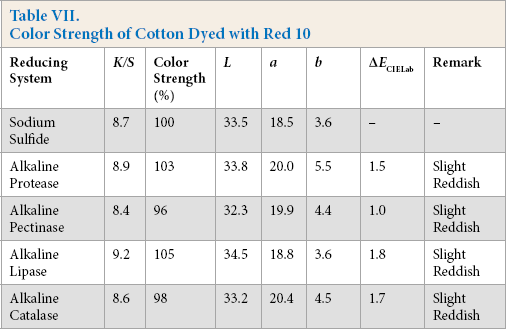
Color Strength of Cotton Dyed with Yellow 9
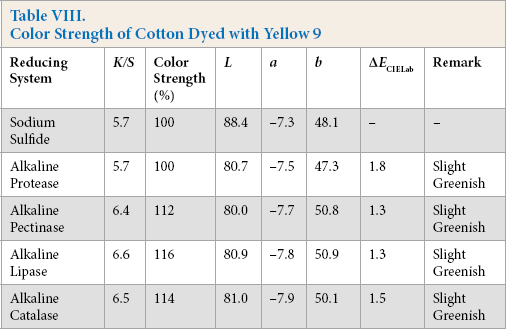
Color Strength of Cotton Dyed with Green 12
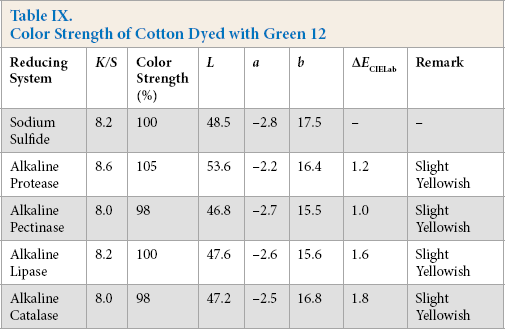
Color Strength of Cotton Dyed with Blue 7
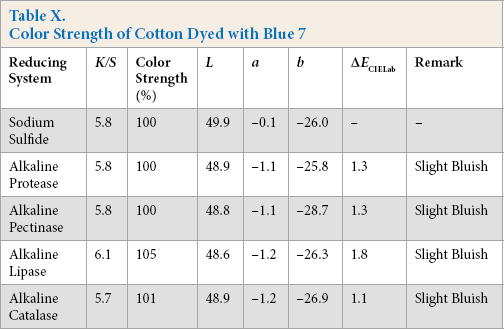
Color Strength of Cotton Dyed with Green 22
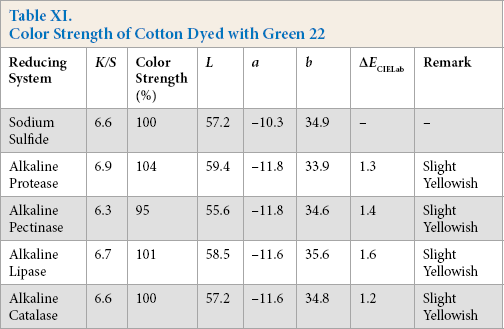
Color Strength of Cotton Dyed with Green 8
Color Strength of Cotton Dyed with Brown 8
Stability of Reduction Baths
Without Dye
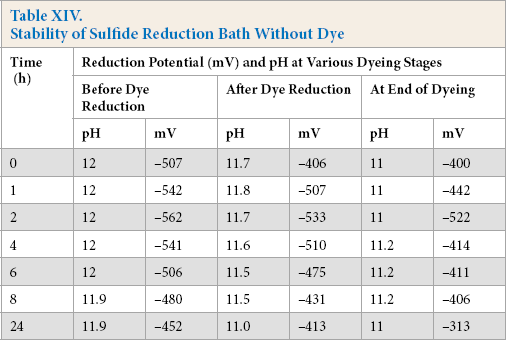
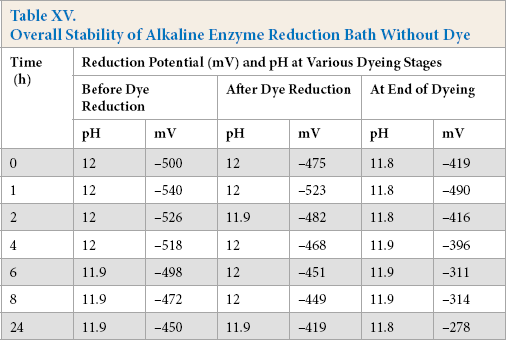
Reduction baths were prepared and stored for varying times in the absence of dye. Afterwards, Green 1 dye was added to these baths followed by heating to 90 °C, and the cotton fabric was dyed for a given amount of time. The reduction potential for sulfide and various enzymatic systems are shown in Tables XIV and XV, respectively.
Stability of Sulfide Reduction Bath Without Dye
Overall Stability of Alkaline Enzyme Reduction Bath Without Dye
The color strength of dyed cotton was found to be nearly the same in sulfide and alkaline enzyme systems after a storage time of 1 h prior to dyeing; the color strength was better with sulfide and catalase after 2 h, while use of the other enzyme systems showed a loss of color strength beyond 1 h of storage. A consistent fall in color strength beyond storage time of 2 h was found in all the reducing systems (Fig. 1a).
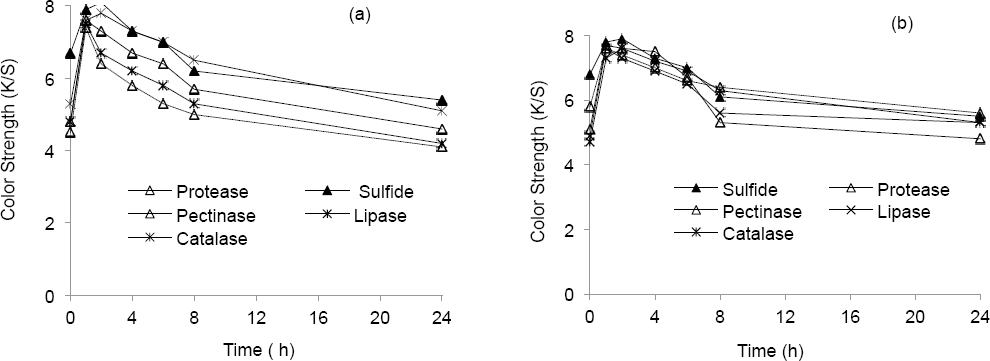
Color strength of cotton dyed from stored reduction baths (a) in absence of dye and (b) in presence of dye.
With Dye
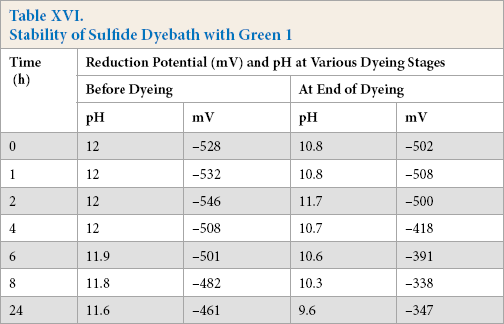
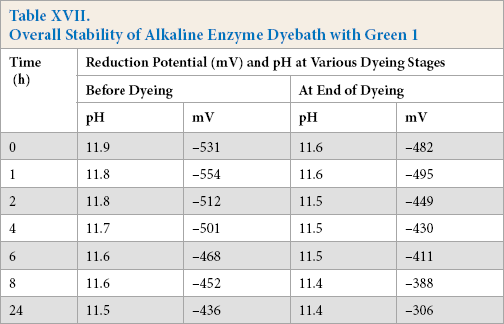
Dye (Green 1) was added to the reduction baths at 90 °C and was stored over varying times. The reduction potential after storage for the sulfide and enzyme systems tested are shown in Tables XVI and XVII, while the color strength values are shown in Fig. 1b. The results were very similar to those obtained from the identical experiments without dye.
Stability of Sulfide Dyebath with Green 1
Overall Stability of Alkaline Enzyme Dyebath with Green 1
Colorfastness of Dyed Cotton Using Various Reducing Systems
Lightfastness
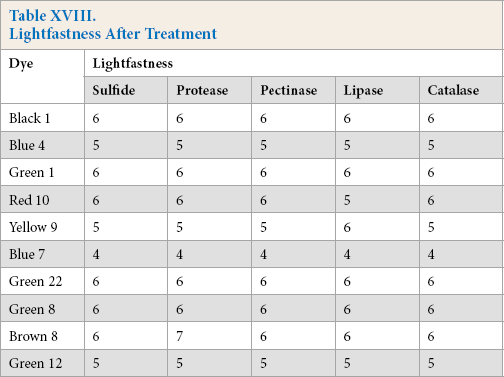
Lightfastness of dyed cotton using the five reducing systems resulted in very good to excellent lightfastness, although there were nominal differences in a few cases (Table XVIII).
Lightfastness After Treatment
Washfastness
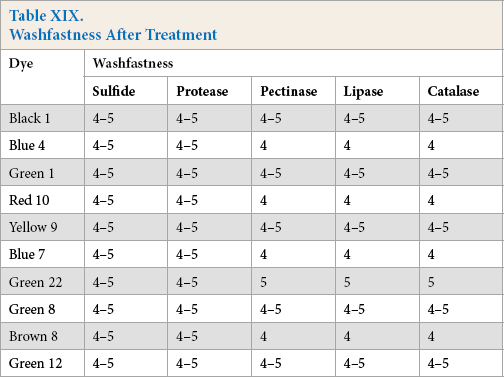
Washfastness of dyed cotton using the five reducing systems were in the very good to excellent grade range (Table XIX).
Washfastness After Treatment
Crockfastness
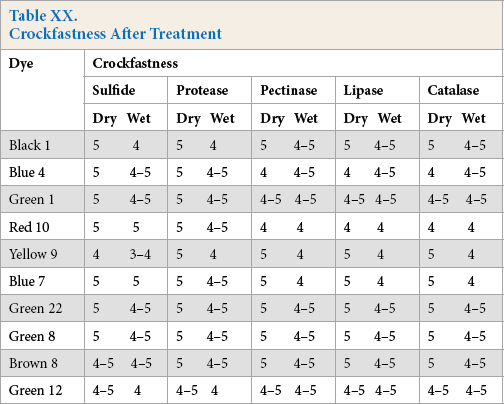
All crockfastness data showed that the fastness grades in dry crocking were better than compared to the wet crocking results. Crockfastness grades were very good to excellent for all reducing systems, except when Yellow 9 was used, where cotton dyed with Yellow 9 using the sodium sulfide system showed moderate wet crockfastness. The detailed report is shown in Table XX.
Crockfastness After Treatment
Conclusions
Alkaline protease, pectinase, lipase, and catalase were found to be equally effective in reduction and solubilization of sulfur dyes with no substantial difference in color strength as compared with sodium sulfide reduction; tonal changes were observed in a few cases, but these were within acceptable limits and undetectable by visual assessment. This may be due to their differences in structure, favorable mechanism, catalytic conditions, and so forth. Reduction bath stability was also substantially good. Overall colorfastness was very good, except for poor lightfastness for Blue 7 and poor wet crockfastness for Yellow 9.
Alkaline protease, pectinase, lipase, and catalase can be used instead of sodium sulfide in dyeing cotton with sulfur dyes. The enzymatic reductions tested achieved the color strength, reduction bath stability, and colorfastness comparable to that of sodium sulfide. This provides an eco-friendly technology for cotton dyeing with sulfur dyes, and thus eliminates the use of toxic sodium sulfide.
