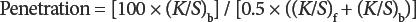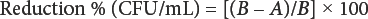Abstract
The main objective of this study is to investigate the effect of eugenol group substitutions (tetra and octa) on fastness and antibacterial properties of metallophthalocyanines printed on cotton fabric. Eugenol-substituted and non-substituted metallophthalocyanines (Zn, Co, Ni, and Cu) were synthesized. Cotton fabrics were then colored using a pigment printing process. Color strength (K/S values) were calculated using the Kubelka–Munk equation. Antibacterial properties of the fabrics were investigated according to ASTM E2149-01. All synthesized phthalocyanine pigments exhibited various levels of antibacterial activity against Staphylococcus aureus and Klebsiella pneumoniae bacteria on cotton fabric. Antibacterial protection was achieved by coloration without any additional antibacterial finishing. Crockfastness, lightfastness, washfastness, and perspiration and water fastness values of the fabrics were measured according to ISO standards.
Introduction
Phthalocyanines (Pcs), macrocyclic compounds containing four pyrrole units, are structurally similar to porphyrins and tetraazaporphins. The two hydrogen atoms attached to the two isoindole group can be replaced by metal atoms from every group of the periodic table to form metal phthalocyanines (MPc). For example, eugenol-substituted and non-substituted metallophthalocyanines (Zn, Co, Ni, and Cu) were synthesized.1–3 Also, each of the sixteen peripheral hydrogen atoms on the four benzene rings can be substituted by various atoms and groups.3,4
Several reports have shown the antimicrobial effect of metal-lophthalocyanines against several species of bacteria.5–10Pcs are effective photosensitive substances. Following their irradiation, singlet oxygen is generated that can kill bacteria. Water-soluble phthalocyanine compounds have, in the presence of oxygen, water, and with photoactivation by irradiation with visible and/or infrared light, good activity against microorganisms.6–8
Eugenol, 4-allyl-2-methoxyphenol, is a pale yellow, essential oil component found in certain plants—basil, cinnamon, lemon balm, and nutmeg—but is primarily extracted from clove plants. The name is derived from the scientific name for clove, Eugenia aromaticum or Eugenia caryophyllata. Eugenol has wide medicinal applications due to its effect on the central nervous and cardiovascular systems. It is used as an analgesic, antipyretic, antimicrobial, antiseptic, and anti-depressant agent, especially in dentistry. It is commonly used as an additive in products such as perfumes or clove favored cigarettes.11,12 However, it can have both local irritative and cytotoxic effects, as well as induce hypersensitivity reactions such as contact urticaria or chronic urticaria.13,14 Tis compound is active against many pathogenic bacteria, fungi and viruses.15–17 Several reports have shown the antimicrobial effect of eugenol against several species of bacteria including Listeria monocytogenes, Streptococcus pyogenes, Proteus vul-garis, Escherichia coli, Staphylococcus aureus, Campylobacter jejuni, Bacillus cereus, Enterobacter aerogenes, Aeromonas hydrophila, Salmonella enterica, Salmonella typhimurium, Helicobacter pylori, and Enterococcus faecalis.15,18–20
Many reports on the antibacterial properties of metal ions and their complexes have appeared in the literature.21–26Yasuyuki et al. stated that pure metals such as Co, Ni, Cu, Zn, Zr, Mo, and Pb had antibacterial properties against both Gram-positive (e.g., S. aureus) and Gram-negative bacteria (e.g., E. coli). 22 The antibacterial properties varied significantly with different metals and the effectiveness of metals to resist bacterial attachment varied with the bacterial strain. Commonly-used metal ions are Co, Cu, Ni, and Zn due to the formation of low molecular-weight complexes. 23 Certain metal ions penetrate into bacteria and inactivate their enzymes, and others can generate hydrogen peroxide, thus killing bacteria. 23
Heavy metal ions, such as Ni, Cu, and Co, have allergenic, mutagenic, or carcinogenic effects at higher exposure levels. But, in phthalocyanine dyes, the metal is an integral part of the molecule. It is bound to the fiber with the dye and normally can only be released together with the dye. 27
In our previous study, it was found that metallophthalo-cyanine (M: Zn) with tetra peripheral eugenol asubstituents had antibacterial activity against S. aureus on cotton fabric according to AATCC Test Method (TM) 147-1998. 28
In this research, the previous study was enhanced by examining the effect of eugenol group substitutions (tetra and octa) on fastness, color shade, and antibacterial properties of metallophthalocyanines (M: Zn, Co, Ni, and Cu) printed on cotton fabric. Variations in antibacterial properties of Pcs, resulting from different central metal ions and the number of eugenol groups present against different bacterial species (S. aureus and Klebsiella pneumoniae), were investigated.
Experimental
Materials
Materials used in pigment suspension and printing are given in Table I. For metallophthalocyanine synthesis, all reagents and solvents used were analytical grade. The bleached, alkaline scoured, 100% cotton fiber fabric (İzmir Basma Factory) was a satin weave (120 g/m2).
Materials used for Pigment Suspension Preparation and Pigment Printing 28
Pigment grinding was carried out with a planetary ball mill (Retsch PM 100). Pigment particle size distributions were determined using a laser diffraction particle size analyzer (Mas-tersizer 2000, Malvern Ins. Ltd.). Pigment printing processes were carried out in a Zimmer Mini-Md color sampling device.
Synthesis
Eugenol-substituted and non-substituted metallophthalocya-nines were synthesized according to the literature procedure.1–3Suspensions of the pigments were prepared with the assistance of a dispersant and salt using the zirconia ball milling method described in our previous study. 28 The particle size distributions were then characterized. After pigment printing, antibacterial and fastness properties of the fabrics were investigated.
Non-Substituted Metallophthalocyanines
Metallophthalocyanines (M: Zn, Co, Ni, and Cu) without substituents were synthesized as described in the literature. 3 Phthalonitrile and an appropriate metal salt (Zn(CH3COO)2, CoCl2, NiCl2, or CuCl2), in a 4:1 molar ratio, was heated to 200 °C with a small amount of 1,8-diazabicyclo[5.4.0]undec-7-ene base in dimethylformamide (DMF) for 24 h. The resulting cyclotetramerization gave the metal phthalocyanines (MPc: ZnPc, CoPc, NiPc, and CuPc) shown in Scheme 1.
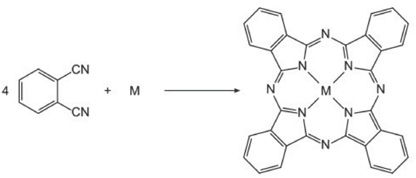
Synthesis of metallophthalocyanines (MPc: ZnPc, CoPc, NiPc, CuPc.).
Tetra Peripheral Eugenol-Substituted Metallophthalocyanines
Synthesis of metallophthalocyanines (M: Zn, Co, Ni, and Cu) with tetra peripheral eugenol groups was performed according to Şaşmaz et al. 1 Phthalonitrile (1,2-dicyanobenzene) derivatives containing the eugenoxy group (4-allyl-2-me-thoxyphenoxy) were first prepared (Scheme 2). Next, cyclotetramerization of the substituted phthalonitriles in the presence of the appropriate metal salt, as described previously, gave the corresponding tetra-substituted metal phthalocyanine (MPc-t-eugenol, Scheme 3). Analytical data for the products obtained were comparable with those in the literature. 1
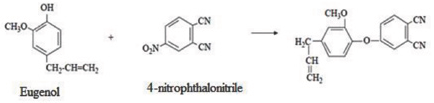
4-(4-Allyl-2-methoxyphenoxy)-1,2-dicyanobenzene.
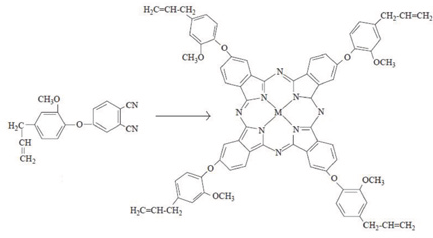
Structure of the metallophthalocyanine pigments containing four eugenol groups (ZnPc-t-eugenol, CoPc-t-eugenol, NiPc-t-eugenol, and CuPc-t-eugenol).
Octa Peripheral Eugenol-Substituted Metallophthalocyanines
Synthesis of metallophthalocyanines (M: Zn, Co, Ni, and Cu) with octa peripheral eugenol groups were performed according to Şaşmaz et al. 2 First, 1,2-bis(4-allyl-2-methoxyphenoxy)-4,5-dicyanobenzene was synthesized (Scheme 4). Next, cyclotetramerization of the substituted phthalonitriles in the presence of the appropriate metal salt, as described previously, gave the corresponding octa-substi-tuted metal phthalocyanines (MPc-op-eugenol, Scheme 5). Analytical data for the products obtained were comparable with those in the literature. 2
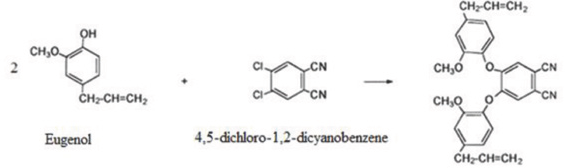
1,2-Bis(4-allyl-2-methoxyphenoxy)-4,5-dicyanobenzene.
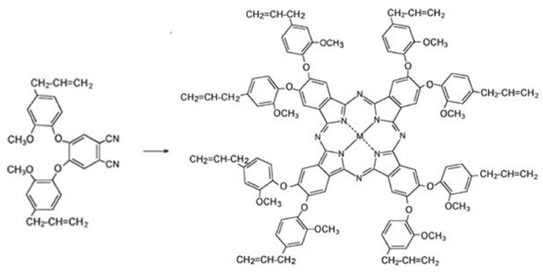
Structure of the metallophthalocyanine pigments containing eight eugenol groups (ZnPc-op-eugenol, CoPc-op-eugenol, NiPc-op-eugenol, and CuPc-op-eugenol).
Printing
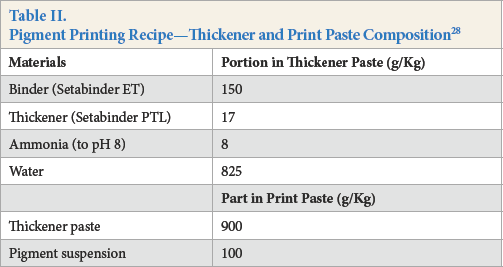
After grinding, the pigment suspensions were used for preparing printing pastes. The pigment printing recipe is given in Table II. The pigment pastes were printed on cotton fabrics and then sequentially dried and cured at 150 °C for 5 min in a stenter.
Pigment Printing Recipe—Thickener and Print Paste Composition 28
Color Measurement
The samples were measured using Easy Match QC software on a Hunterlab Ultrascan Pro color spectro-photometer attached to a personal computer. The color strength (K/S) of the sample was calculated using the Kubelka–Munk equation.
The penetration of the pastes through the printed, dried, and cured fabric samples (Eq. 1) was based on the difference in reflectance between the face and back of the printed area, and used the derived K/S values for the front and the back of the fabric. 29
The (K/S)b and (K/S)f values are for the back and front of the fabric, respectively.
Antibacterial and Fastness Test Methods
Antibacterial properties of the fabrics were investigated according to ASTM E2149-01 against Gram-positive S. aureus and Gram-negative K. pneumoniae bacteria. ASTM E2149-01 is designed to evaluate the resistance of non-leaching antimicrobial-treated specimens to the growth of microbes under dynamic contact conditions. 30 The antimicrobial activity is expressed in percent reduction of the living organisms after contact with the test specimen compared to the number of bacterial cells surviving after contact with the control. The percentage reduction is calculated using Eq. 2.
A is the surviving cells (CFU/mL) for the flasks containing the treated substrate after the specified contact time and B is “0” CFU/mL contact time before the addition of the treated substrate.
Fabrics were tested according to ISO 105-X12 for crock-fastness, ISO 105-B02 for lightfastness, ISO 105-CO6 for washfastness, ISO 105-E04 for perspiration fastness, and ISO 105-E01 for water fastness.
Results and Discussions
Synthesis
All spectroscopic data and elemental analyses of the synthesized pigments gave good agreement with the literature values.1,2
Pigment Grinding
Dispersion quality of the pigment can significantly influence its application properties, such as shade, color strength, and dispersion stability, as well as adsorption on the substrate. To achieve the optimal benefits of a pigment, it is necessary to obtain as full a reduction as possible to the primary particle size. 31
The synthesis of Pcs at higher temperatures led to the most thermostable β-phase with particle sizes between 5 and 100 μm. Dissolving the β-phase in concentrated sulfuric acid and precipitation with water or grinding in a ball mill in the presence of a salt resulted in α-phase Pcs, with particle sizes ranging from 30 to 50 nm. 32 Particle size distribution analyses of the microscale pigments in suspensions are shown in Figs. 1–12. The greatest number of particles ranged in size from 0.1 to 1 μm. The CoPc-t-eugenol suspension had the most uniform particle size distribution, as well as the least amount of aggregation (Fig. 5). Peaks occurring from 10 μm to 100 μm in Figs. 1–12 were due to particle aggregation in the suspension; the greater the peak areas were in this range, the greater the aggregation.
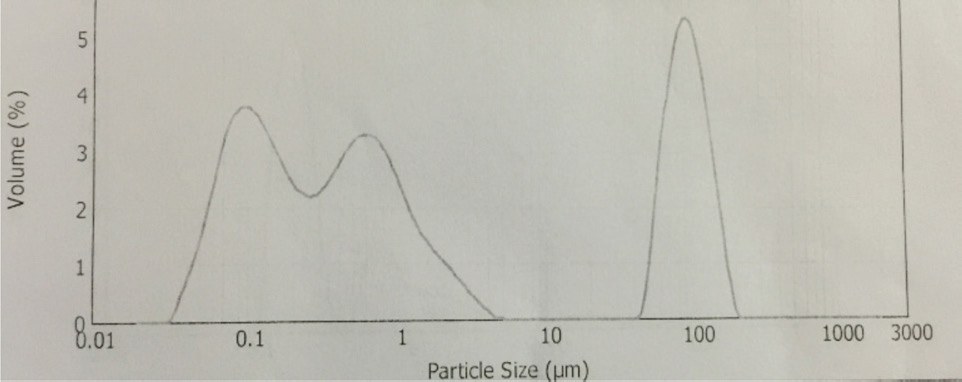
ZnPc particle size distribution.
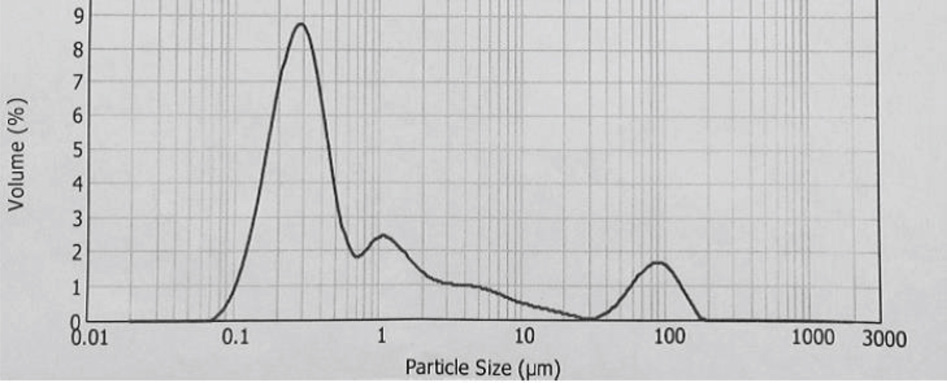
ZnPc-t-eugenol particle size distribution.
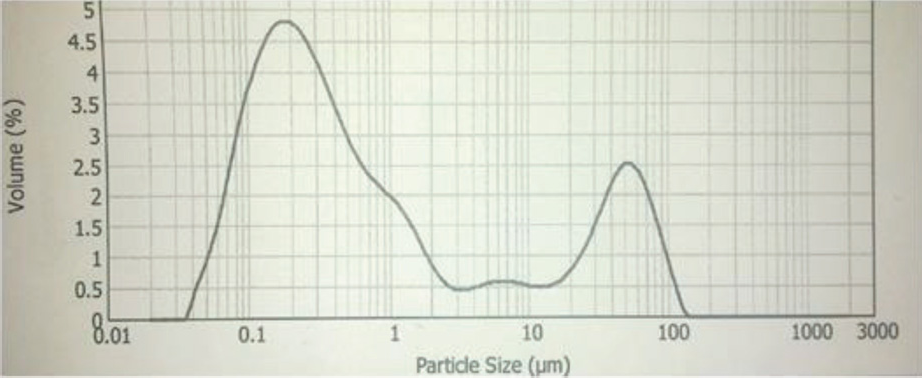
ZnPc-op-eugenol particle size distribution.
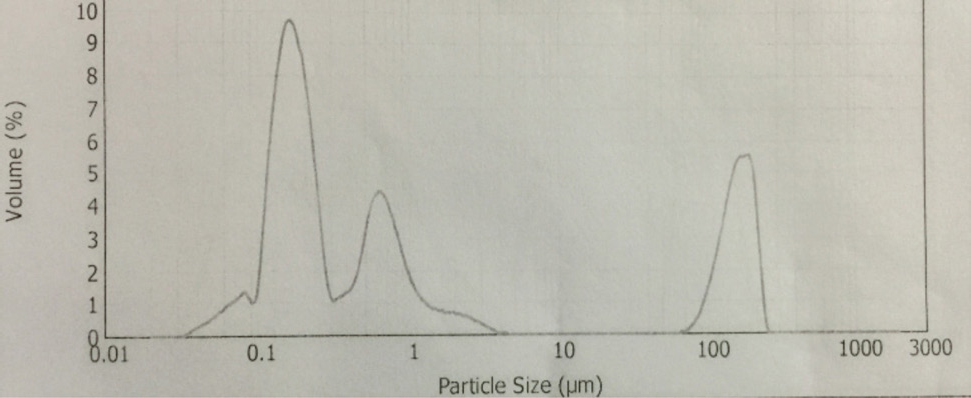
CoPc particle size distribution.
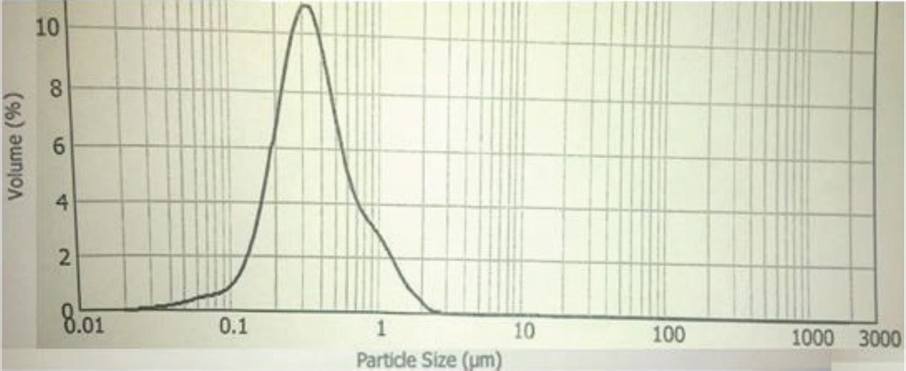
CoPc-t-eugenol particle size distribution.
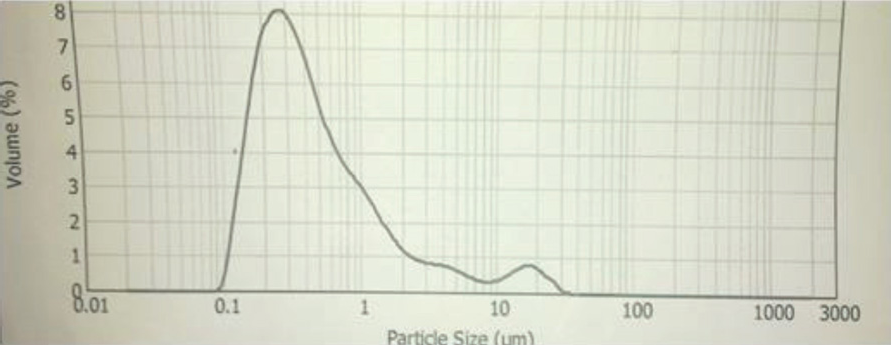
CoPc-op-eugenol particle size distribution.
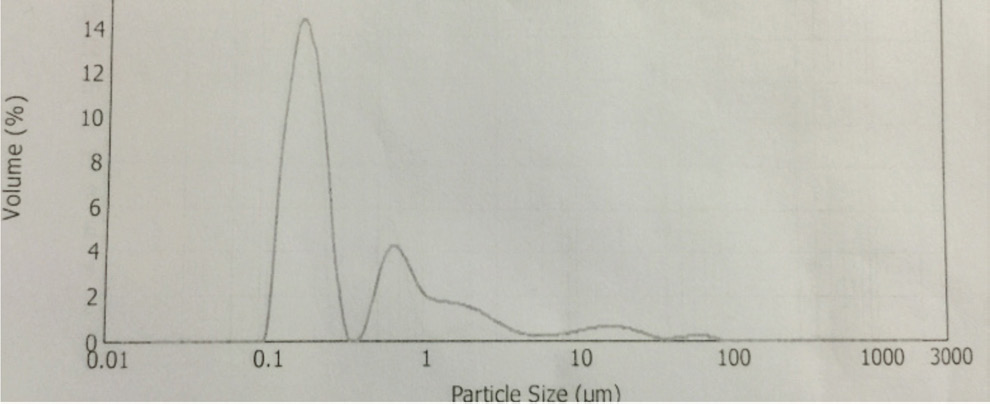
CuPc particle size distribution.
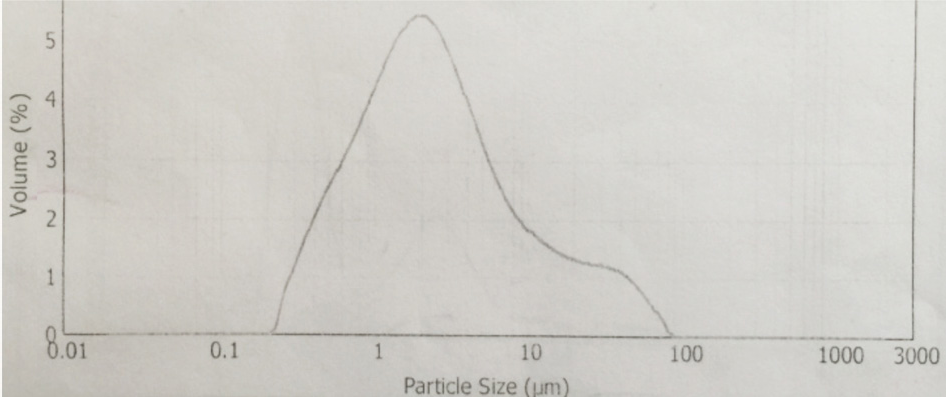
CuPc-t-eugenol particle size distribution.
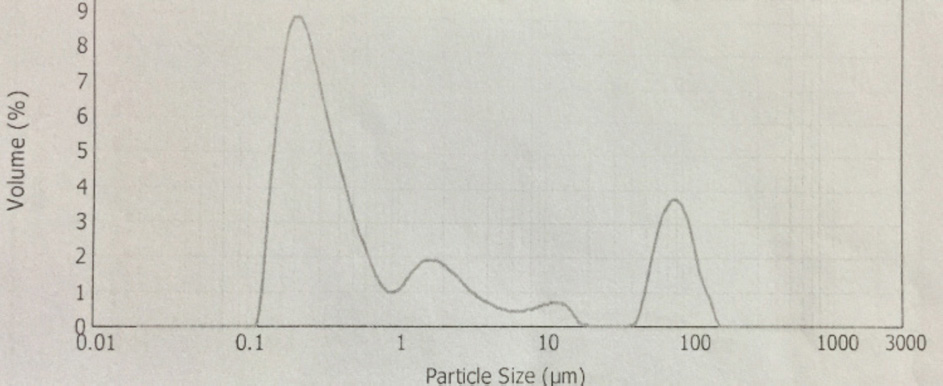
CuPc-op-eugenol particle size distribution.
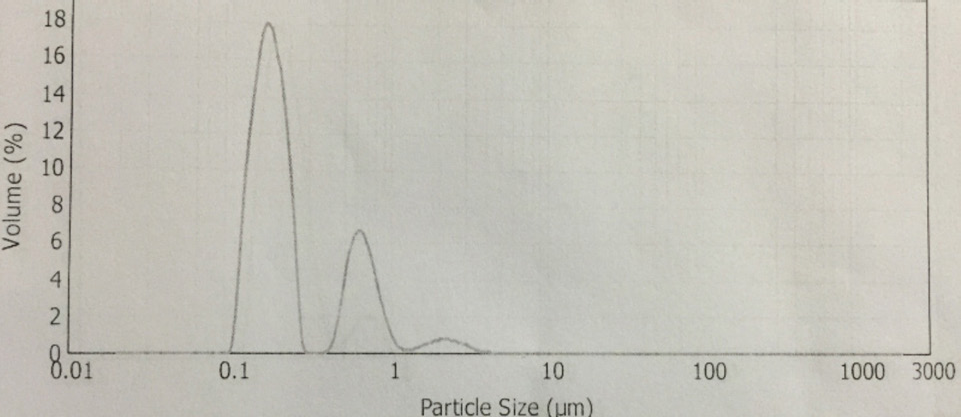
NiPc particle size distribution.
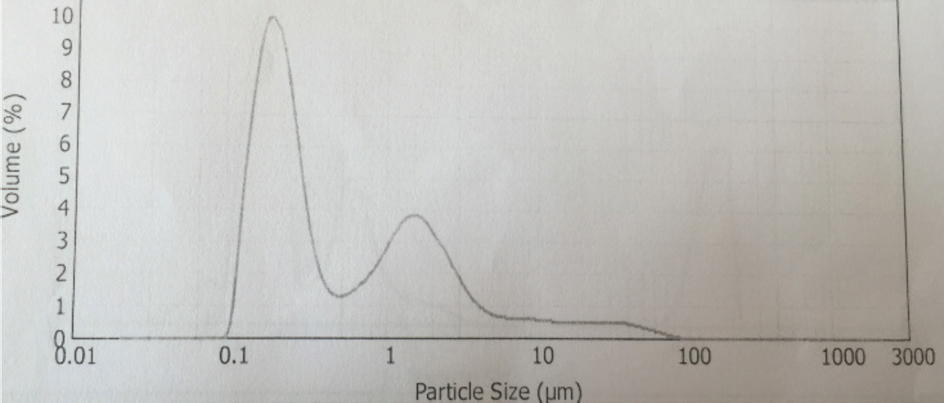
NiPc-t-eugenol particle size distribution.
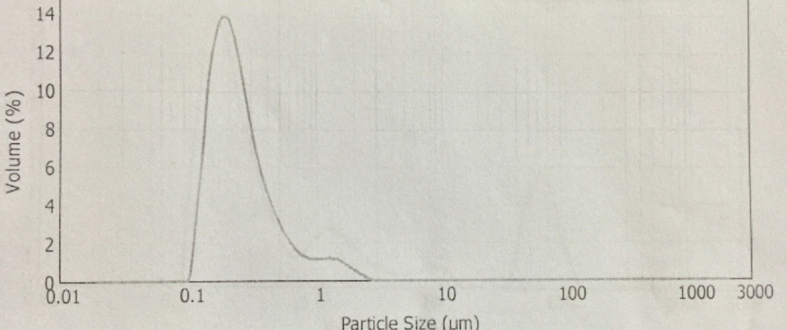
NiPc-op-eugenol particle size distribution.
Color Strength
The synthesized pigments were insoluble in water, so pigment printing, which is applicable to textile coloration without rinsing or washing, was used. The cotton fabrics had a uniformly-colored surface in different shades of green or blue. The color of the pigments on the fabrics varied with different central metal ions and with the number of eugenol groups in phthalocyanine structure due to changes in the wavelength of maximum UV-Vis absorption. The λmax values of the fabrics ranged from 615 to 690 nm. Penetration degree and color shade of the printed fabrics were given with K/S and λmax values in Table III. With an increasing number of eugenol groups in the same metal phthalocyanine structure (e.g., NiPc, NiPc-t-eugenol, and Ni-op-eugenol), the spectral bands shifted to longer UV-Vis wavelengths (bathochromic shift), changing the color from blue to green.
Penetration Degree and Color Shade of Printed Fabrics

The penetration degree decreased with an increasing number of eugenol groups in the same metal phthalocyanine structure; CoPc, CoPc-t-eugenol, and CoPc-op-eugenol values were 96.64%, 52.5%, and 9.28%, respectively.
Antibacterial Activity
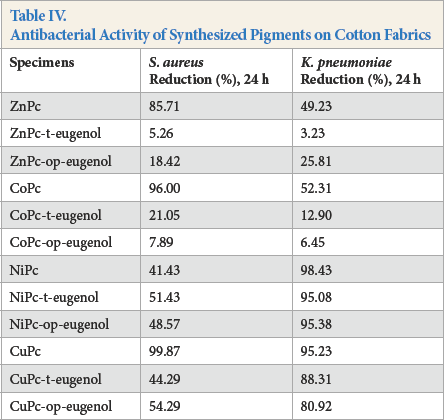
The antibacterial activity of the printed cotton fabrics (Table IV) were tested according to the shake flask test method against S. aureus and K. pneumoniae.
Antibacterial Activity of Synthesized Pigments on Cotton Fabrics
S. aureus bacteria can cause numerous infections, including impetigo, scalded skin syndrome, folliculitis, furuncle, carbuncle, osteomyelitis, septic arthritis, endocarditis, toxic shock syndrome, pneumonia, thrombophlebitis, and deep tissue abscess and infection. 33 K. pneumoniae bacteria can cause different types of infections, including pneumonia, bloodstream infections, wound or surgical site infections, and meningitis. 34
The antibacterial properties of the pigments varied significantly with the choice of central metal ion depending on the bacteria tested (Table IV). CuPc and CoPc pigments gave the most effective antibacterial activity against S. aureus on cotton fabric, 99.87% and 96.00%, respectively. NiPc, NiPc-op-eugenol, and CuPc pigments showed the most effective antibacterial action against K. pneumoniae on cotton fabric, 98.43%, 95.38%, and 95.23%, respectively. The eugenol substituted and non-substituted nickel and copper phthalocyanines were especially effective antibacterials against the more resistant K. pneumoniae.
Colorfastness
The lightfastness and crockfastness results are given in Table V. The dry crockfastness values of the printed cotton fabrics were in the range of 4–5 and 5 gray scale ratings, which were commercially-acceptable levels. Wet crockfastness values were slightly less (3–4 to 5) compared to dry crockfastness values.
Crockfastness and Lightfastness Values of Printed Fabrics
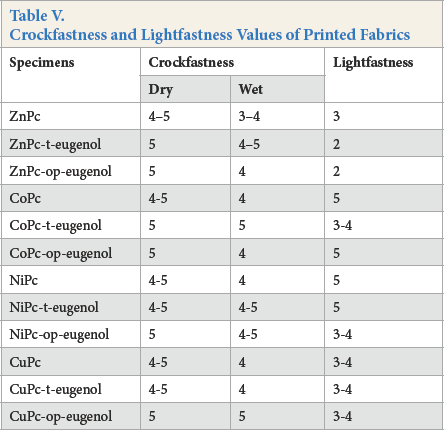
The printed cotton fabrics exhibited different lightfastness levels according to the wool scale rating based on central metal ion types. In all synthesized pigments, lightfastness values for cobalt and nickel metal ligands were medium (3–4 to 5), while the values for zinc metallophthalocyanines (2–3) were poor. The poor lightfastness of zinc metalloph-thalocyanines can largely be explained by a higher quantum yield production of singlet oxygen, which causes photo-oxidation of cellulose and/or the dye. A long triplet lifetime and a relatively high triplet quantum yield, which are useful qualities for a photosensitizer, characterize metallophthalo-cyanines containing diamagnetic metal ions, such as Zn+2. Complexes with paramagnetic transition metal ions, such as Co+2, Ni+2, and Cu+2, however, have much shorter triplet lifetimes and this difference is reflected in the quantum yields of singlet oxygen formation. 35
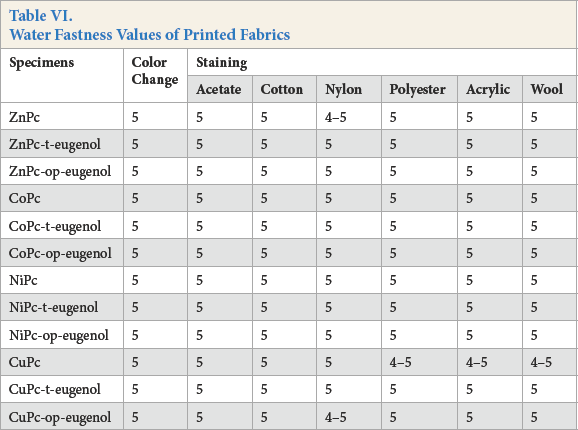
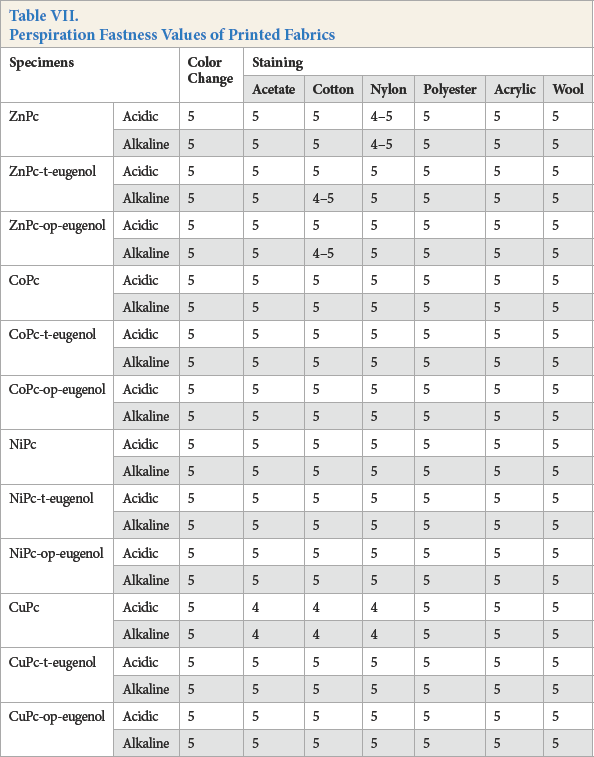
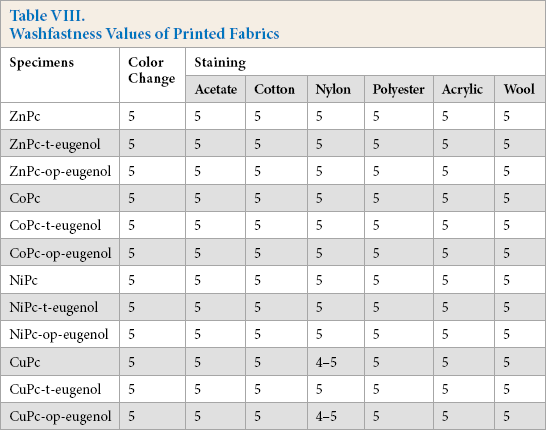
Water (Table VI), perspiration (Table VII), and washfastness gray scale ratings (Table VIII) were good (4 and 5).
Water Fastness Values of Printed Fabrics
Perspiration Fastness Values of Printed Fabrics
Washfastness Values of Printed Fabrics
Conclusions
The effect of eugenol group substitutions (tetra and octa) on fastness and antibacterial properties of metallophthalocyanines (M: Zn, Co, Ni, and Cu) printed on cotton fabric were examined. Different shades of blue- to green-colored antibacterial fabrics were obtained. Antibacterial protection was achieved only by coloration with these pigments without any additional antibacterial finishing. The synthesized pigments had various levels of antibacterial action against S. aureus and K. pneumoniae bacteria. Tis property of the pigments varied significantly with type of central metal ion and the bacteria tested. Fastness to perspiration and water, washfastness, and crockfastness values were at commercially-acceptable levels.
Potential use of these colored antibacterial fabrics might include military and healthcare applications (especially medical textiles), workwear, domestic products, and sports apparel.
Footnotes
Acknowledgements
The authors would like to acknowledge and thank the research and development department of Setaş Technology Center (Setaş Kimya Industry Co.) Tekirdağ in Turkey for their kind guidance and technical support. Thanks also for their valuable comments, which helped shape this study.
This work has been supported by Recep Tayyip Erdogan University in the context of Scientific Research Project 2014.102.02.04.