Abstract
This research studies the solubility properties of certain regenerated cellulose fibers in commonly-used acidic reagents, including formic acid/zinc chloride. A dissolution method for qualitative identification of Tencel A100 (lyocell) fiber in the presence of various other regenerated cellulose fibers was established. This qualitative method was simple, rapid, and had high sensitivity.
Keywords
Introduction
In recent years, the development and use of regenerated cellulose fibers has gained greater attention. One essential way of identifying textiles is fiber composition, which is mandatory for meeting many international import/export technical regulations. Due to large differences in perceived comfort for fabrics made from various regenerated cellulose fibers, and significant variance in the price of raw materials, there is an urgent need to develop qualitative and quantitative analysis techniques for the detection of blended products containing regenerated cellulose fibers.
There are no qualitative or quantitative dissolution analysis test methods and standards for regenerated cellulose fibers, not only in the GB/T 2910,1,2 GB/T 2911, 3 SN/T 3315,4,5 and FZ/T 010576,7 methods, but also in AATCC Test Method (TM) 20A 8 and ASTM D629. 9 There is a considerable amount of literature on qualitative and quantitative analysis of regenerated cellulose fibers and cotton10–18 or other fibers,19–22 such as rayon made from bamboo and cotton, 12 lyocell and cotton,11,14–17 viscose or modal and cotton,10,11,17,18lyocell and fax, 19 rayon made from bamboo and Shengma fiber (regenerated cellulose fiber made from jute and ambary), 20 and regenerated cellulose fiber and spandex.21,22There were also some studies among the regenerated cellulose fibers,23–25 such as lyocell, Modal,23,24 Tencel A100, and Tencel G100 fibers. 25 The methods used in these studies were mainly dissolution,10–19,22 diameter, 24 spectrophotometric, 25 and infrared spectroscopy21,26–28 methods. There were also studies reviewing various properties of regenerated cellulose fibers, including the effects of different treatments.29,30
The chemical characteristics of regenerated cellulose fibers are very similar, which is due to the structure of celluloses, which are polymers of beta-D-glucose. The degree of polymerization (n) of several common cellulose fibers are different. For instance, cotton fiber n values are generally 2000, lyocell and modal are ∼900, and viscose fiber is 250 to 500. 1 In acidic solvents, dissolution of cellulose fibers occurs when the large molecular chains of cellulose are shortened by breaking 1,4-glycosidic bonds, followed by degradation. During this process, the resulting n values become smaller until dissolution occurs. 12 Use of formic acid/zinc chloride to distinguish between regenerated cellulose fibers and cotton fiber relies on the large difference in n values between them based on acidic solvent solubility. Differences in the degree of polymerization between A100 fiber and other regenerated cellulose fibers may be exploited in practical work.
Therefore, it is important that qualitative and quantitative analytical methods with high efficiency, simple operation, and high sensitivity be established for these regenerated cellulose fibers. In this study, our lab developed a qualitative identification method for A100 fiber and certain other regenerated cellulose fibers by choosing a suitable acidic solvent and operating under appropriate experimental conditions. Use of this method will also help to improve the identification of these fibers in textile products and prevent commercial fraud. In a related study, a quantitative method has also been developed based on the same technology. 31
Experimental
Materials
Tencel A100, Tencel LF, Tencel G100, and Modal fibers were produced by Lenzing Co. Rayon made from bamboo (Hebei Jigao Chemical Fiber Co. Ltd.) and viscose fiber (standard material from the Shanghai Textile Institute of Technical Supervision, and separated into fibers before use) were also tested.
All proper safety precautions, including wearing safety glasses and protective clothing, must be taken when working with these reagents. Hydrochloric acid (36–38%, Beijing Chemical Works), formic acid/zinc chloride reagent (20 g of anhydrous zinc chloride (mass fraction > 98%, Tianjin Zhiyuan Chemical Reagent Co. Ltd.) and 68 g of anhydrous formic acid (Tianjin Zhiyuan Chemical Reagent Co. Ltd.), with water added to give a final weight of 100 g). Sulfu-ric acid (60%, Beijing Chemical Works) was prepared by slow addition of 343 mL of concentrated sulfuric acid (p = 1.84 g/mL) into ∼368 mL water, to reach a density of p = 1.4988 g/mL (20 °C) or p = 1.4948 g/mL (25 °C). 1/2 hydrochloric acid (1:1 (v/v) hydrochloric acid and water), and 1/4 hydrochloric acid (1:3 (v/v) hydrochloric acid and water) were prepared.
A thermostatic oscillator (SHA-BA, Jintan City Union Instrument Research Institute), a 100-mesh stainless steel screen, and an electronic analytical balance (AE160, Mettler) were used.
Procedures
The original regenerated cellulosic fibers given above and their mixtures were used in this work to simplify analysis. Fiber quantities were determined as the quantity after moisture regain, and the fiber samples in all evaluations (before and after testing) were weighed the same day to ensure consistency of moisture regain.
Qualitative Analysis
The fiber sample (0.5 g) was quickly added to the formic acid/ zinc chloride solution (50 mL) in an Erlenmeyer flask with a grinding plug, and placed in a water bath with the temperature preheated to and kept at 70 °C. The treatment time was 20 min, the samples oscillated at a frequency of 100 rpm, and a bath ratio of 1:100 was used. The reactant was poured onto the 100-mesh stainless steel screen, and the remaining insoluble matter was identified as A100 fiber by microscopic analysis.
Dissolution Characteristics and Differences of Recycled Cellulose Fibers
The insoluble material was washed with water, dried by heating in an oven, and then weighed after moisture regain.
Results and Discussion
Commonly-Used Acidic Reagents
The goal of a qualitative method is to be as simple, quick, and reliable as possible. Acids are commonly used reagents for regenerated cellulose fiber analysis. In this study, the various test regenerated cellulose fibers did not dissolve in formic acid, therefore, the reagents in Table I were used.
Solubility of Regenerated Cellulose Fibers in Commonly-Used Acid Reagents
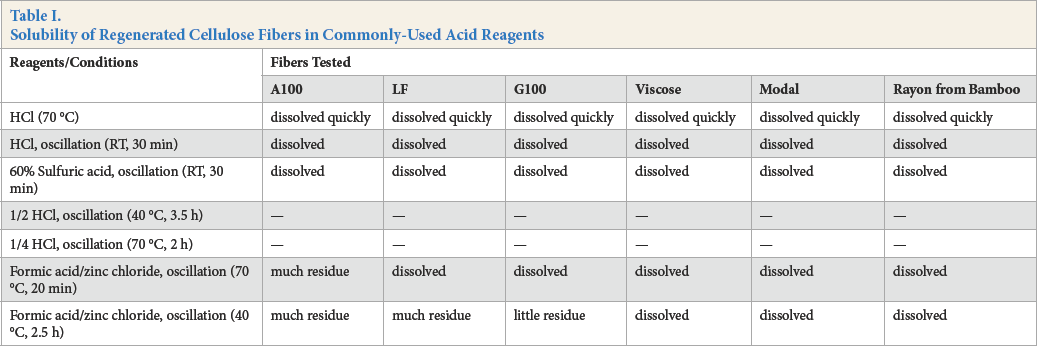
Hydrochloric acid (70 °C) caused all fibers to quickly (and violently; see precautions in experimental section) dissolve. Hydrochloric acid (room temperature (RT), 30 min) and 60% sulfuric acid (RT, 30 min) gave similar results. Formic acid/zinc chloride solution is commonly used in regenerated cellulose fiber composition analysis. The solubility of the regenerated cellulose fibers tested in formic acid/zinc chloride solution under different experimental conditions were different, thus this solution was chosen for further study.
Formic Acid/Zinc Chloride System
Experiments were carried out to study the solubility and dissolution process of the regenerated cellulose fibers being tested in formic acid/zinc chloride at different temperatures. The results are given in Table II. These data were obtained from 40 °C to 70 °C over 20–150 min.
Solubility of Regenerated Cellulose Fibers in Formic Acid/Zinc Chloride System
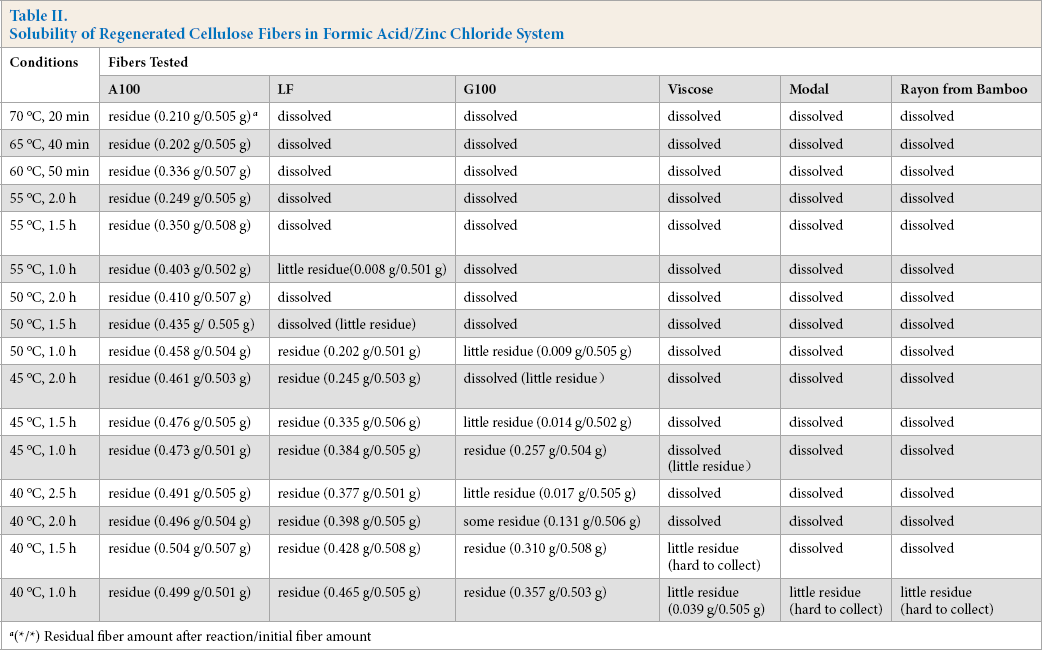
(*/*) Residual fiber amount after reaction/initial fiber amount
The solubility of the various regenerated cellulose fibers tested, when reacted with formic acid/zinc chloride, (from least soluble to most) was as follows: A100 < LF < G100 < viscose < Modal and rayon made from bamboo.
Temperature was an important factor in the dissolution of the regenerated cellulose fibers tested in formic acid/ zinc chloride. Besides the A100 fiber, the other regenerated cellulose fibers tested only needed 20 min to completely dissolve at 70 °C, and 2 h at 50 °C. Loss of residual A100 fiber increased with increased temperature.
The data in Table II show the dissolution of the regenerated cellulose fibers tested under various experimental conditions. Tests over time (1.0, 1.5, 2.0, and 2.5 h) at 40 °C showed the G100 fiber was more soluble after 1.5 h, and was almost completely dissolved at 2.5 h. The G100 fiber was more soluble after 1 h at 45 °C, and was almost completely dissolved at 1.5 h.
Method Establishment
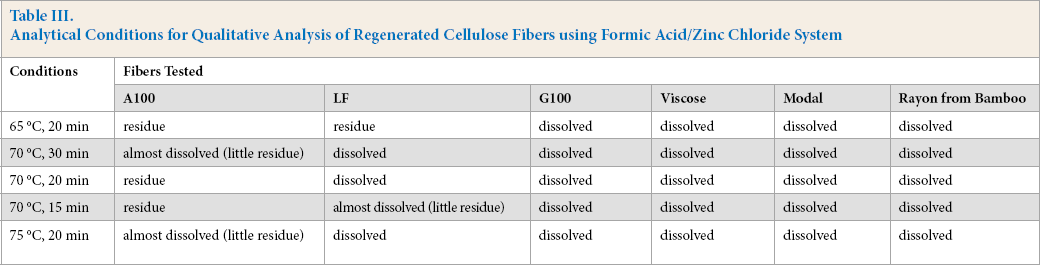
To decrease analysis time, qualitative analysis conditions for regenerated cellulose fiber analysis were refined around a temperature of 70 °C (Table III).
Analytical Conditions for Qualitative Analysis of Regenerated Cellulose Fibers using Formic Acid/Zinc Chloride System
Method Specificity
This experiment, using pure and mixed fiber samples under the previously determined test conditions, is given in Table IV. Tests 1 and 2 demonstrate that the insoluble matter was only A100 fiber. Tests 3–7 show the results for the five other fibers in the presence of 50% A100 fiber—residue was observed in all five cases. These results indicate that the method could distinguish A100 fiber from the other regenerated cellulose fibers tested.
Presence of Residue after Dissolution Testing of Various Regenerated Cellulose Fiber Mixtures
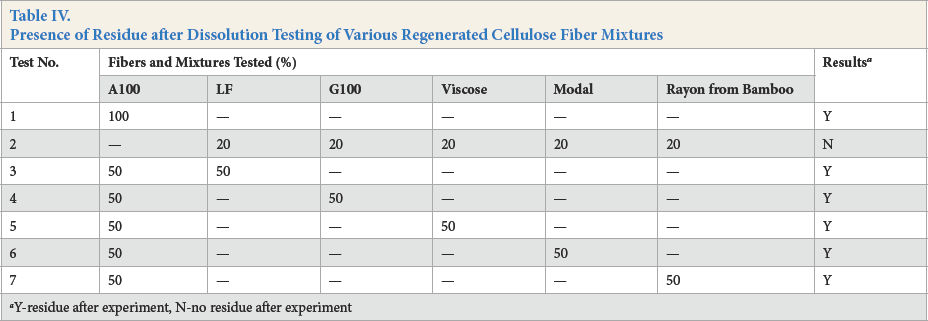
Y-residue after experiment, N-no residue after experiment
Experiments were carried out at 65 °C (20 min), 70 °C (15, 20, and 30 min), and 75 °C (20 min) as shown in Table III. The A100 fiber almost completely dissolved at 70 °C (30 min) and 75 °C (20 min). LF fiber could not be completely dissolved at 65 °C (20 min) or 70 °C (15 min). Thus, 70 °C (20 min) in formic acid/zinc chloride was suitable for qualitative analysis of A100 fiber versus the other regenerated cellulose fibers tested. While increasing the reaction temperature could shorten the reaction time, loss of residual A100 fiber would increase with increasing temperature, which would affect the detection limit.
Method Robustness
An orthogonal experiment was designed for investigating method robustness, including three factors (solution temperature, dissolution time, and oscillation frequency) (Table V). The results show that the conditions, including solution temperature of 70 °C, dissolution time of 20 min, and oscillation frequency of 100 rpm, were reproducible. The detection limit for A100 fiber content was greater than or equal to 1%, the detection range is the content of A100 fiber from 1% to 100%. In addition, different batches of reagents and different experimental personnel were also investigated, with the same results achieved.
Robustness of Qualitative Analysis Method for Regenerated Cellulose Fibers
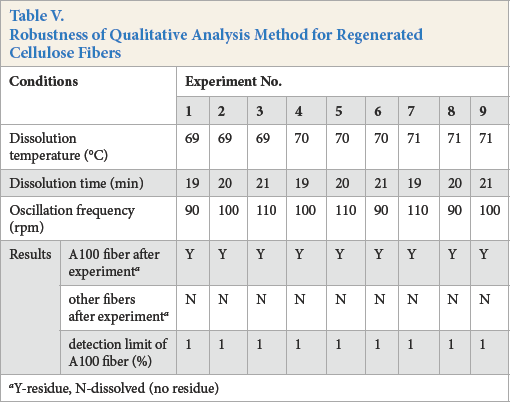
Y-residue, N-dissolved (noresidue)
Method Verification
Internal and external laboratory verification of the method was carried out. Eight laboratories, all of which are CNAS (China National Accreditation Service for Conformity Assessment) accredited fiber composition analysis item laboratories, were invited for the method verification and validation. One independent lab gave results of method robustness and sample verification and validation, and seven other coordinated laboratories gave sample verification and validation results. These results were consistent with results from our laboratory. The results indicated good reliability and general applicability of this method from different laboratories.
Conclusion
In this work, we studied the solubility of various regenerated cellulose fibers in commonly-used acidic reagents using the dissolution method, and established a qualitative method for detecting the presence of Tencel A100 fiber alone and in the presence of the other regenerated cellulose fibers tested. This method was simple, rapid, and highly sensitive. The dissolution characteristics of the regenerated cellulose fibers tested in formic acid/zinc chloride solution, including similarities and differences, were summarized. This study provided a solid foundation for the next step in establishing qualitative and quantitative analysis dissolution methods among other regenerated cellulose fibers.
