Abstract
The bleaching of greige soybean knitted fabric using ultraviolet (UV) irradiation on hydrogen peroxide impregnated samples was studied. The effect of many variables on the samples’ whiteness, lightness, wettability, hydrophilic properties, cystine content, surface examination, and Fourier transform infrared (FTIR) properties was investigated. The UV/H2O2 bleaching process was optimized using a Box-Behnken experimental design and three optimal recipes were achieved. A novel bleaching process for greige soybean knitted fabric, which consists of sample impregnation in various peroxide baths, followed by UV irradiation, was developed. The improvement in whiteness of greige soybean samples, not easily bleached by conventional bleaching processes, was obtained within minutes, improving processing time and providing energy savings.
Introduction
Although petroleum-based fibers have been widely used in the textile industry, their long term production and sustain-ability is being reassessed due to increased oil costs and their relatively slow end-of-life biodegradability. Accordingly, in the light of these environmental and commercial factors and recent advances in the genetic modification of plant proteins, there has been renewed interest into regenerated proteins fibers. 1
Soybean fiber is a regenerated protein-based fiber, which has recently become commercially significant due to its luster, smooth surface, breathability, and comfort. These have been identified as desirable aesthetic properties in apparel. Together with its cost competitiveness and the potential for genetic engineering of the soybean protein to improve the material's performance, these offer commercial opportunities. 2
Soybean protein-based fiber is thought likely to replace certain sectors of the conventional natural fiber market, for example those involving cashmere and silk. Although natural protein fibers such as wool and silk have good physical properties and have been used extensively in the textile industry, they are relatively expensive to use and process. In silk, a large quantity of mulberry leaves is required for the production of a very small quantity of silk, resulting in an increased production cost. In addition, apart from the economic aspects, animal fibers are physically limited in several aspects. First, both wool and silk fiber vary in diameter and their performance profile is limited. Second, morphologically, the presence of scales on wool's surface results in felting shrinkage and difficulties in dyeing. In contrast, regenerated protein fibers, such as soybean fiber, do not have a theoretical limit in fineness to which fibers may be drawn.
In addition, soybean is a competitive production material for fibers in the textile industry since it is abundant and cost-effective. Another advantage of soybean protein is its higher protein content (40%), compared to peanuts (25%) and corn (10%), and the environmentally friendly production claimed by manufacturers. 3 Additionally, the possibility that a plant protein can be modified by molecular genetics provides the opportunity to improve fiber properties in specific applications.3,4 Soybean protein fiber is a potentially useful material for upscale and high-end clothing production. 5
Soybean fibers are beige, with a color similar to that of tus-sah silk. The reason soybean fibers are colored has not been completely ascertained. A very white product is difficult to obtain by conventional beaching methods.3,6 Although the dyeing and flammability of soybean fibers has been studied, little has been reported on the effect of bleaching with regard to reducing yellowness and maintaining fiber strength.1,4,7Other soy protein products are frequently treated with oxi-dative agents such as hydrogen peroxide, sodium peroxide, barium peroxide, or oxidizing salts, prior to industrial use.1,8Conventional bleaching of soybean fiber is thus a costly, non-environmentally friendly, and lengthy operation, since it is multi-stage in nature.
The development of advanced oxidation processes (AOPs) based on ultraviolet (UV) photolysis, capable of mineralizing a wide range of recalcitrant or hazardous organic compounds, had a strong impact on the progress of the technology associated with photochemistry and photochemical processes.9–11Because of its simplicity, oxidation is the most commonly used chemical decoloration process. In most cases the oxidizing agent is hydrogen peroxide, which, due to its relative stability, needs to be activated. Peroxide is activated by UV light.12–14 H2O2/UV treatment generates hydroxyl radicals—highly powerful oxidizing species with an oxidation potential of 1.78 V.15,16
The aim of this study is to develop an H2O2/UV treatment process for bleaching greige soybean fiber knitted fabric. This process was studied under a variety of conditions. The effectiveness of bleaching was evaluated using the whiteness index, lightness index, wettability, hydrophilic properties, cystine content, surface examination, and Fourier transform infrared (FTIR) properties of the soybean fabrics.
Experimental
Materials
Raw, 100% protein-based, soybean interlock-knitted fabric, mass per unit area 140 g/m2, constructed from spun yarns (linear density Ne 40/1), was obtained from a local supplier. Fabric samples (0.3 g) were used during the experiments. Non-ionic wetting agent, complexing agent, non-ionic washing agent, 50% hydrogen peroxide, sodium carbonate, and sodium peroxydisulfate were of laboratory reagent grade and obtained from a local provider. The UV lamp produced UV-B light, with a power consumption of 2000 W.
Procedures
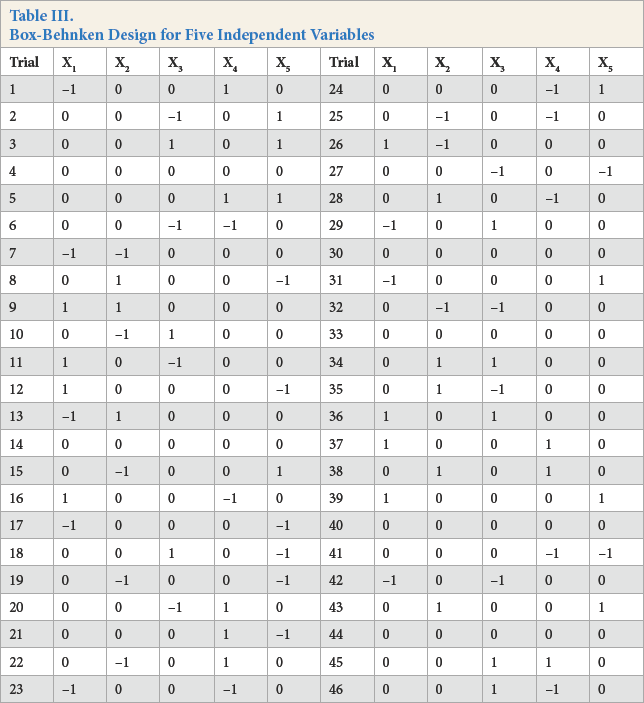
In accordance with the Box-Behnken design, 17 three different irradiation times (1, 2, and 3 min), hydrogen peroxide (40, 60, and 80 g/L), sodium carbonate (10, 15, and 20 g/L), and sodium peroxydisulfate (0, 6, and 12 g/L) concentrations, and distance between the UV lamp and fabric (10, 17, and 24 cm) were investigated. Hydrogen peroxide impregnating baths were prepared according to Tables I–III. First, the samples were immersed in one of the hydrogen peroxide bleaching baths at room temperature (RT) for 60 s. Then they were squeezed with a padding mangle until the wet pickup was 80–100%. Finally, impregnated samples were subjected to UV light in accordance with the Box-Behnken experimental design. The temperature of the impregnating baths was 20-25 °C. After the irradiation of both sides of the samples, they were rinsed with water at 90 °C for 10 min, washed with 0.06 g/L catalase enzyme at 40 °C for 20 min, washed with 1 g/L non-ionic washing agent at 95 °C for 10 min, rinsed with water at 95 °C for 10 min, and finally, cold rinsed with water for 10 min. The samples were then air dried.
Impregnation Bath Recipes

Variable Levels Chosen for Trials

Box-Behnken Design for Five Independent Variables
Experimental Design
Irradiation time, hydrogen peroxide concentration, sodium carbonate concentration, sodium peroxydisulfate concentration, and distance between UV lamp and fabric were chosen as the critical variables and designated as X1–X5, respectively. The low, middle, and high levels of each variable were designated as –1, 0, and +1 respectively (Table II). The actual design of experiments is given in Table III.
Analytical Methods
Whiteness and Lightness Determination
To evaluate the experimental results, the whiteness and lightness degree of samples was examined according to ASTM E31318 (D65, 10°) and the CIELab formula, respectively, with a Hunterlab Ultra Scan PRO spectrophotometer. Each sample was read in three different areas and the average value was calculated.
Wettability Determination
Wettability was examined by contact angle measurements using distilled water. Equipment consisting of a camera (PULNIX TM-765), a computer, and a monitor were used. The contact angles of water were measured by a sessile drop method. 19 The images of each drop were captured by the camera connected with a computer-based image capture system. The captured images were viewed on the monitor.
Hydrophilicity Determination
To measure the hydrophilicity of soybean fabrics, the sinking test method 20 was applied. Samples (1 cm2) were used. A flask, with a 15-cm diameter, was filled to a height of 10 cm with distilled water, and the sample put in the flask. When the sample contacted the distilled water and base of the flask, the chronometer was started and stopped, respectively. Finally, the immersion times of the samples were calculated.
FTIR Analysis
FTIR spectroscopy was used to determine the treatments that induced detectable modification to the fiber's internal physical microstructure. The measurements were carried out using a Perkin Elmer Spectrum 100 instrument. The specimens were analyzed at 4 cm−1resolution, and the recorded spectrum for each was the average of 4000 scans.
Cystine Content Determination
This method was modified from the cystine content determination of wool. 21 Determination of cystine content is particularly suitable for determining the extent of damage by alkali. For determination of cystine, the sample was hydrolyzed by heating with dilute sulfuric acid. The disulfide groups were then reductively cleaved by adding sodium sulfite. The cysteine thus formed, together with the cysteine originally present, gave a blue coloration on addition of phosphotungstic acid (Folin's reagent). The color was then assessed colorimetrically. 21
Microscopic Surface Examination
Surface examination of samples was carried out to assess whether the various treatments had caused any visible degradation to the fiber surfaces. A TM-1000 microscope (Hitachi) was used for this purpose.
Also, the results of all assays were compared using analysis of variance (ANOVA), followed by a post hoc test (Duncan's test).
Results and Discussion
By applying multiple regression analysis methods, the pre-dicted response for whiteness (WI) and lightness (L*) can be obtained and given as
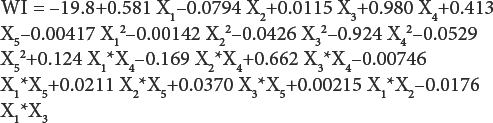
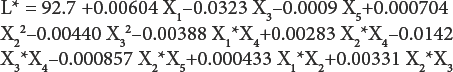
The statistical significance of Eqs. 1 and 2 was checked by F-test, and the ANOVA for response surface quadratic model is summarized in Table IV. The ANOVA of regression model demonstrates that the model was highly significant. The value of R 2 (99.8% for WI and 95% for L*) showed good correlation between the experimental and predicted values of the response (Table IV). The lack-of-fit measured the failure of the model to represent data in the experimental domain at points that were not included in the regression. The non-significant value of lack-of-fit (> 0.05) for WI and L* revealed that the quadratic model was statistically significant for the response and therefore, can be used for further analysis.
Regression Analysis for WI and L* a
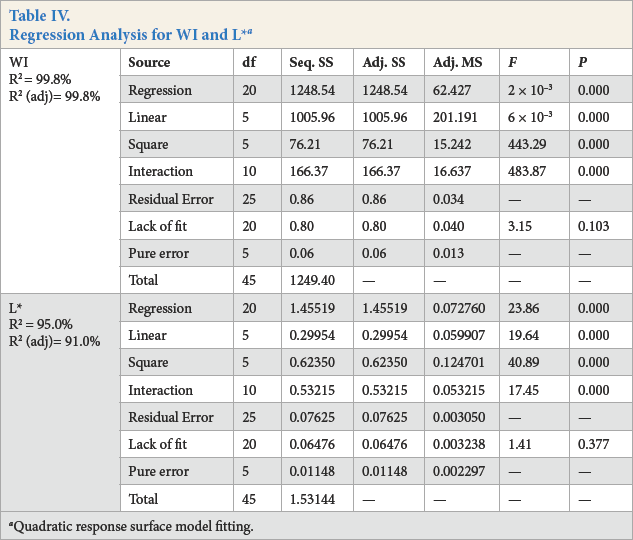
Quadratic response surface model fitting.
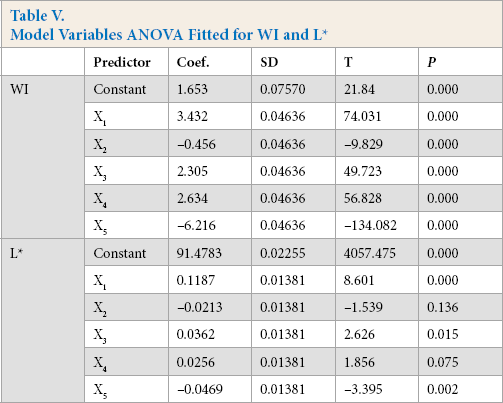
After ANOVA for the quadratic model, all model terms having P-values greater than 0.05 were eliminated, since they were not statistically significant at the 95% confidence level. Further ANOVA for the variables in the model used indicates that sodium carbonate concentration, distance between UV lamp and fabric (P = 0.000) for WI, hydrogen peroxide concentration (P = 0.000), sodium peroxydisulfate concentration (P = 0.015), and distance between UV lamp and fabric (P = 0.002) for L* had the greatest effect on the model (Table V).
Model Variables ANOVA Fitted for WI and L*
UV/H2O2 Treatment Results
WI and L*
The whiteness and lightness results of untreated and irradi-ated samples are given in Fig. 1.

Photographs of untreated and treated soybean fabrics. Sample number is final number in parentheses under each image. The experiment description is given above each pair of images, except for sample 1.
It is well known that UV irradiation alone cannot act as a bleaching agent. A photoinitiator, such as hydrogen per-oxide, must be used. 19 An increase in H2O2 concentration produces more hydroxyl radicals that can increase the reaction rate (Eqs. 3–6).
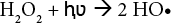
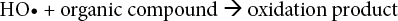
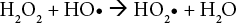
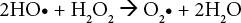
Consequently, higher WI values (greater than 5) were achieved working with 80 g/L hydrogen peroxide concentration. On the other hand, sodium peroxydisulfate concentration was important when the hydrogen peroxide concentration was less than 75 g/L. For example; if the aim is to achieve WI values greater than 0, an increase in sodium peroxydisulfate concentration from 0 to 6 and 12 g/L supported the decrease in hydrogen peroxide concentration from 65 to 50 and 40 g/L.
The increase in sodium carbonate concentration restricted sample whiteness improvement. For example, when the hydrogen peroxide concentration used was 60 g/L, an increase in the amount of sodium carbonate from 20 g/L to 50 g/L caused a decrease in WI values from 7.65 to 2.18. Use of a minimal amount of sodium carbonate provides chemical and cost savings along with soybean samples having high WI values.
It was sufficient to place the soybean fabric 10–14 cm from the UV lamp to obtain maximal WI values. Also, the increase in WI values of soybean samples was achieved with an increase in irradiation time from 1 min to 3 min.
The photographs of the untreated and treated samples demonstrated the achieved results (Fig. 1). Soybean sample 3 impregnated with a peroxide bath containing 80 g/L hydrogen peroxide, 6 g/L sodium peroxydisulfate, and 35 g/L sodium carbonate, and then irradiated for 3 min had the highest WI value. A similar result was found for sample 6. Although some of the treated samples (samples 2, 4, and 8) had a yellowish appearance, they were less yellow than the greige samples. There is no known method of improving the whiteness of greige soybean fabric in 3 min. The increase in irradiation time and placing the fabric 10 cm from the UV lamp increased the whiteness of soybean samples.
Fig. 1 also shows that an increase in hydrogen peroxide concentration, as well as decreasing the distance between the soybean fabric and the UV lamp, increased L* values. Maximum L* values can be achieved with irradiation (with fabric sample 14 cm from the UV lamp) of soybean fabric impregnated in the 60 g/L hydrogen peroxide bath.
Hydrophilicity and Wettability
To investigate the effect of H2O2/UV bleaching on the wet-tability properties of greige soybean fabrics, contact angle measurements were performed. The wettability properties of soybean fabrics improved after H2O2/UV treatment (Fig. 2). Better wettability properties and lower drop penetration times were obtained at increased irradiation times and at decreased distance between the fabric and UV lamp. The increase in sodium carbonate and sodium peroxydisulfate concentration resulted in the decrease in drop penetration time. For example; when the sodium carbonate concentration was increased from 20 g/L to 50 g/L, the drop penetration time decreased from 0.240 s to 0.096 s. When the contact angle measurements were evaluated based on hydrogen peroxide concentration, the photographs of the drops on the samples treated with 40 and 80 g/L hydrogen peroxide appeared different, and the drop penetration time of the sample, impregnated at the largest peroxide concentration, was slightly less.

Effect of treatment variables on wettabliliy properties of greige soybean samples. Measured as contact angle and drop penetration time. Sample number is final number in parentheses under each image. The experiment description is given above each pair of images, except for sample 1.
The sinking test method was used to observe the changes in the hydrophilic properties of treated soybean fabrics. Changes in irradiation time and distance between the fab-ric and the UV lamp caused no significant change in the sinking time and, therefore, to the hydrophilic properties of the soybean samples. The most important parameter in terms of hydrophilicity was the hydrogen peroxide concentration used in the impregnation bath. When the hydrogen peroxide concentration was increased from 40 g/L to 80 g/L, the sinking time of samples decreased from 15.5 s to 9 s (Fig. 3).
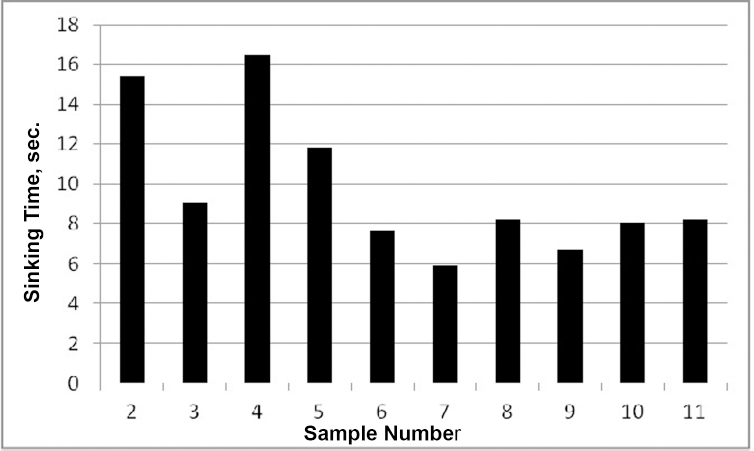
Effect of UV irradiation on hydrophilicity properties of greige soybean fabric.
FTIR Analysis
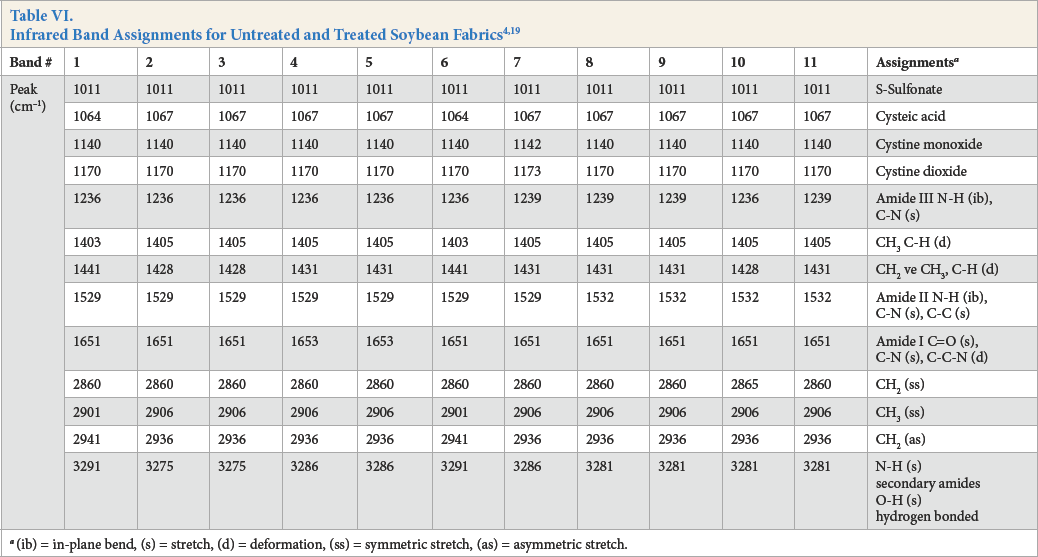
The infrared band assignments for greige and variously bleached soybean fabrics are shown in Table VI and Fig. 4. The strong broad band at around 3291 cm−1 in the spectrum of greige soybean fabric was due to hydrogen-bonded OH stretching and NH stretching of secondary amides. After various irradiation processes, the absorbance of OH and NH stretching decreases and the band moves from 3291 cm−1to 3275–3286 cm−1. The bands at 1064, 1140, and 1170 cm−1in the spectrum of the greige sample is attributed to cysteic acid, cystine monoxide, and cystine dioxide, respectively.19 Theincreased transmittance of these peaks indicates that the soybean sample was modified by UV irradiation. This result was demonstrated with the cystine content results of samples. The cystine content of greige soybean fabric decreased from 8.4% to 5.2% after UV irradiation. The 1651 cm−1 band indicates the C=O and C-N stretching, and C-C-N deformation, at amide I. After UV irradiation, the transmittance of this band increased.
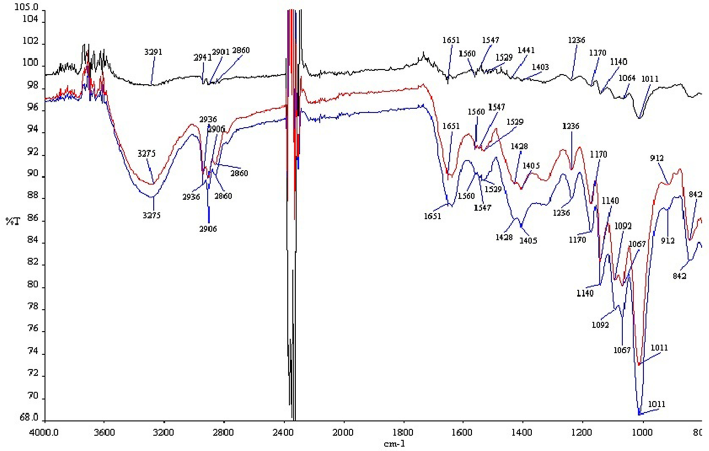
FTIR spectra for untreated and treated soybean fabrics. Black line is untreated soybean fabric (sample 1), red line is treated soybean fabric (sample 2), and blue line is treated soybean fabric (sample 3).
(ib) = in-plane bend, (s) = stretch, (d) = deformation, (ss) = symmetric stretch, (as) = asymmetric stretch.
Surface Examination
To characterize fiber damage, the surface morphology of H2O2/UV treated samples was also examined by tabletop microscope (Fig. 5). The micrographs of greige soybean fabric shows a smooth surface with mild longitudinal striations. This surface appearance is presumably inherent to the base fibers. Some signs of pitting, cracking, or similar features that would normally be associated with degradative damage was seen on the various irradiated samples.

Microscopic examination of untreated and treated soybean fabrics. See Fig. 1 for descriptions of all numbered samples
Conclusions
As a result of this study, a new H2O2/UV bleaching method for soybean fabric was developed. Improvement in the whiteness of greige soybean fabric was obtained within minutes, although soybean samples were degraded by UV irradiation and the cystine content decreased from 8.4% to 5.2% after irradiation. The content of the hydrogen peroxide impregnation bath affected the whiteness of irradiated soybean samples. To optimize the process, a Box-Behnken experimental design was applied and three optimal recipes were obtained.
The maximum WI (> 2 by ASTM E 313) and L* (> 92 by CIELab) values can be obtained using an impregnating bath consisting of 60 g/L hydrogen peroxide, 20 g/L sodium carbonate, 7.5 g/L sodium peroxydisulfate, with samples placed 14 cm from the UV lamp and irradiated for 2 min.
If the goal is to achieve a modest increase in WI (–2 to 0 by ASTM E 313) and L* (91.5 by CIELab) values, the greige soybean fabric should be impregnated in a bath consisting of 50 g/L hydrogen peroxide, 20 g/L sodium carbonate, 5 g/L sodium peroxydisulfate, and then placed 14 cm from the UV lamp and irradiated for 1 min.
To achieve a small improvement in WI (< –2 by ASTM E 313) and L* (< 91 by CIELab) values, the greige soybean fabric should be impregnated in a bath containing 40 g/L hydrogen peroxide, 20 g/L sodium carbonate, 5 g/L sodium peroxydisulfate, and then placed 14 cm from the UV lamp and irradiated for 1 min.
With this process, there is no requirement to heat the liquor, the process is fast (1–3 min), and energy is saved. This process has a promising future for fibers that cannot be bleached easily by conventional methods. Further research using this process should be performed.
Footnotes
Acknowledgements
This study has been prepared from the doctoral dissertation “An Investigation the Use of Eco-Friendly New Methods and Technologies at Pretreatment Process of Cellulosic Fibers and New Generated Fibers” by Seher Perincek.