Abstract
Polyhydroxyalkanoates (PHAs) are a family of linear biodegradable thermoplastic polyesters that are synthesized in nature by bacterial fermentation. Their formation is triggered by a deficiency of nutrients and an excess supply of carbon sources. Despite the high yield of PHAs via bacterial fermentation, it is not an economically viable route. To reduce the cost, photoautotrophic production is thought to be a desired route. Cyanobacteria are one of the only prokaryotic species that naturally produces PHAs (under stress conditions) by photosynthesis. The current study describes the mixotrophic biosynthesis of poly(3-hydroxybutyrate) (PHB) in cyanobacteria (blue green algae) Nostoc muscorum followed by polymer extraction. The extracted polymers (in the fiber/yarn form) may have uses in various textile-related medical applications such as tissue scaffolds, sutures threads, and surgical meshes.
Keywords
Introduction
The mid-1900s was known for many significant inventions and discoveries, but was popularly known as the Age of Plastics due to massive advancements in the field of synthetic polymers. 1 Soon, the whole world was captivated by the wonders of synthetic polymeric materials due to their versatility, flexibility, resistance to harsh solvents, and alterable features. In the following decades, pollution created by these indestructible synthetic polymers led researchers to explore other avenues of plastics production.
Polymers synthesized by macro and microorganisms can be easily broken down in the environment into water and carbon dioxide—such polymers are termed biopolymers. Poly-hydroxyalkanoates (PHA) are a class of linear polyester biopolymers that are commonly produced intracellularly in microorganisms (usually bacteria) under stress conditions. Among macroorganisms, several strains of fungi 2 and trans-genic plants 3 are known to biosynthesize PHAs. However, there are significant negative effects of PHA production reported 4 on growth and development of transgenic plants, which made microorganisms a more viable candidate for production of PHAs.
PHAs act as a food reserve that is collected as water-insoluble granules in the cytoplasm of the microorganism. Poly(3-hydroxybutyrate) (PHB) was one of the first and the most commonly found PHA polymers to be isolated (Fig. 1 and Table I) by Lemoigne from Bacillus megate-rium. 5 Synthesis of PHAs in microbes results from the limitation of essential growth nutrients such as phosphorus, nitrogen, and iron in the presence of surplus carbon sources. When the environment is deprived of carbon food sources, the microbes metabolize PHAs through intracel-lular depolymerases. Hence, these polyesters act as energy reserve molecules. 6 The cost of PHA production via bacterial fermentation is high due to the expensive carbon sources and rich oxygen supply required during the process. These economic constraints make low-cost photosynthetic alternatives of PHA production more desirable.
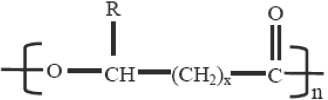
General chemical structure of the poly-hydroxyalkanoate family of polymers. 7
Constituents of the PHA family 7

Cyanobacteria and PHA
Cyanobacteria are one of the only prokaryotic species that naturally produce PHAs in nutrient deficient environments by photosynthesis and require fewer resources for growth and biomass production. Cyanobacterial cells can accumulate the homopolymer of PHB under photoautotrophic and chemoheterotrophic conditions. 7 PHB in the cyanobacterial species Chloroglea fritschii was first reported by N. G. Carr using UV spectroscopy. 8 Since then, several researchers have studied the intracellular occurrence of PHAs in multiple algal strains such as Gleocapsa sp. PCC 6501, 9 Oscillatoria limosa, 10 Microcystis aeruginosa, 11 Spirulina plantensis, 12 Syn-echococcus sp. PCC 6803, 13 and Synechococcus sp. MA 19. 14
Pure PHB polymers possess various attractive properties (e.g., thermoplastic processability, hydrophobicity, optical purity, complete biodegradability, and biocompat-ibility) making it a reasonable substitute for commercial textile fiber materials such as polypropylene. 15 However, it is noteworthy that, in spite of the increasing number of algal strains examined to grow PHB, most of the literature deals only with cyanobacterial physiology and related biochemical aspects of the microorganisms. Very limited research has been done on the biosynthesis and characterization of PHAs in Nostoc muscorum for use in textile fiber-related applications. 16 The Nostoc genus comprises colonies of filamentous cyanobacteria found in a variety of environmental habitats and also live within host organisms through a symbiotic relationship. 7 Versatility and robust growing patterns makes N. muscorum an attractive choice for production of PHAs.
The primary goal of this research was to investigate the biosynthesis and extraction PHB homopolymers from Nostoc cultures for future use in textiles. This paper also examines the physicochemical properties of the extracted polyesters and, based on these properties, recommendations for use in fiber-related applications are discussed.
Experimental
Materials
Test Organism and Growth Conditions
The N. muscorum culture was obtained from the algae culture collection in the Bioconversion Lab at the University of Georgia, Athens. The culture was axenically grown in BG11 medium17 in a controlled conditions growth chamber at 25 °C ± 1 °C and pH 7–8, with light:dark cycles of 12 h:12 h and under a light intensity of 80–100 μmol photon m−2 s−2 photosynthetically active radiation (PAR) for 10 days to allow full growth and proliferation. The culture was continuously bubbled with a filtered and sterilized atmospheric air mixture containing 20 parts of CO2 per 100 parts of mixture at a flowrate of 1.89 L/min.
PHB production was induced in N. muscorum by subjecting the culture to nitrogen (N) and phosphorus (P)-starvation by separation of the culture from the medium through centrifugation and re-suspension of the centrifuged culture pellets into a pH-adjusted N- and P-free BG11 medium.
Mixotrophic Biosynthesis of PHA
To enhance the production of PHB in the culture, the cells grown in BG-11 medium for 10 days were transferred to a carbon-rich (and N- and P-deficient) medium. Here, in addition to the bubbling of CO2, acetate (0.1% w/v) as an organic carbon source was supplemented into the deficient medium. The culture was then incubated in a growth chamber for 7 days under the same conditions as mentioned previously.
Methods
Culture Growth
To monitor growth of the Nostoc culture, sampling to check culture concentration was done every 48 h and the optical density (OD) of the culture was recorded at 750 nm using a UV-Vis spectrophotometer (Cary-50 Bio). All results were based on detection in triplicate of the culture samples. Additionally, the change in morphology of the growing Nostoc cells was visualized under a light microscope (Zeiss Primo Star).
PHB Extraction
Methanolysis was used to degrade the culture cells and extract PHB polymers from the intracellular inclusions in the culture. 5 A known amount of algal culture was centri-fuged (at 4700 rpm for 10 min) and, after discarding the supernatant, the cell pellet was suspended in methanol at 4 °C overnight to remove the pigments. The pellet thus obtained was dried at 60 °C and the polymer was extracted in hot chloroform followed by precipitation of PHB in chilled methanol. The methanol-chloroform mixture was decanted and centrifugation was used to separate the precipitated polymer. The polymer was further purified by dissolving in chloroform and evaporation of the solvent to obtain a polymer film.
Estimation of PHB Accumulation
An algal sample (5 mL) was centrifuged. The resulting pellet, after weighing, was re-suspended in 1 mL of deionized water and transferred into a 2-mL screw cap microtube of known weight. The sample was then centrifuged and, after removal of the supernatant, the pellet was dried overnight at 80 °C in an oven. The dried pellet was reweighed to determine the average biomass or dry cellular weight (dcw). Upon extraction of PHB from the dried pellet using the previously mentioned methanolysis method, the pellet containing pure polymer was weighed to determine the yield of PHB as percent dry weight of the biomass.
Spectrophotometric Assay of PHB
The presence of PHB can be further confirmed according to Law and Slepecky's spectrophotometric assay. 2 A dried pellet of culture sample was subjected to cellular destruction by mixing the pellet with 1 mL of 96% H2SO4 and heating the mixture up to 100 °C for 1 h. The culture sample underwent digestion and removal of pigments in the presence of heat and concentrated acid leading to the formation of crotonic acid. The concentration of crotonic acid was then determined at 235 nm on the UV-Vis spectrophotometer using standard crotonic acid as the control.
Gas Chromatography-Mass Spectroscopic (GC-MS) Analysis of PHB
The culture was separated from the stationary media phase by centrifugation (at 4700 rpm for 10 min) before subjecting it to a standard methanol-esterification reaction. 17 Approximately 30 mg of cells were methanolyzed in a 10-mL screw cap test tube solution consisting of 15% (v/v) sulfuric acid and 85% (v/v) methanol for 140 min at 100 °C in a water bath, with constant stirring. After esterification, the mixture was cooled, 1 mL of distilled water was added, and the test tube was vigorously shaken. The bottom organic layer consisting of hydroxyacyl methyl esters were then subjected to GC-MS analysis 6 on an HP series GC system with an HP INNOWAX capillary column (30 m × 0.25 mm × 0.25 μm) using nitrogen as the carrier gas with a constant flow rate of 1.5 cm/min.
Results and Discussion
The growth pattern of the N. muscorum algal cells in BG-11 medium is shown in Fig. 2. In the initial growing stages, the cells were seen to thrive at an exponential rate, with a large increase in concentration (mg of cells/L of medium) due to the abundant availability of essential nutrients. Upon transfer of the culture into an N- and P-deficient medium (and addition of acetate) on the tenth day, the growth profile was seen to plateau and eventually decline. There were no noticeable morphological changes seen in the size and shape of the cells under a light microscope, except that the deficient cells indulged in more clustering and coiling (due to N- and P-deficiency) rather than in the previously recorded long, filamentous state of the normal cells.
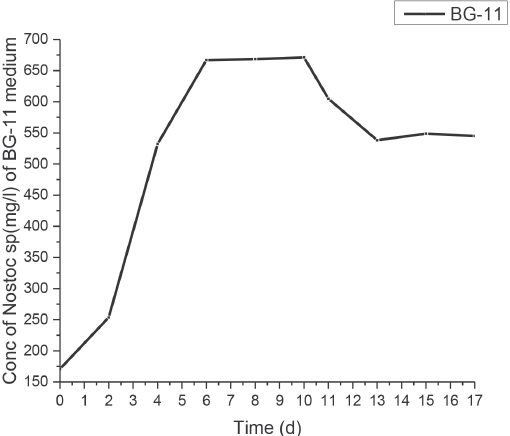
Growth profile of N. muscorum culture in standard BG-11 medium for 10 days and in N- and P-free medium supplemented with 0.1% (w/v) of acetate.
It was also noted that, under N- and P-free conditions (and excess of carbon), bleaching along with flocculation and sedimentation of the Nostoc cells occurred with a shift to a more alkaline pH (9–10). This bleaching effect was also visualized as a decrease in chlorophyll content (cells changed color from blue-green to yellow-green) as seen under the light microscope (Fig. 3).
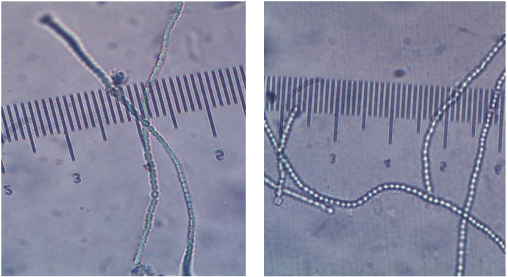
Morphology and change in chlorophyll content of N. muscorum cells in standard medium (left) and in N- and P-deficient medium (right).
N. muscorum is a non-diazotrophic microorganism, which under N-starved conditions, responds to the change by degrading its blue-green pigments (called phycobilip-roteins) through a process called chlorosis16,18 which is observed as bleaching. The degraded proteins are then used to synthesize new materials to maintain cell growth in N-deficient environments.
Fig. 4 illustrates the PHB content (%w/w of dcw) of the Nostoc biomass vs. the number of days it was incubated in N- and P-deficient medium in the presence of acetate. While the growth rate of the test organism after introduction to the deficient environment did not positively increase (Fig. 2), the intracellular accumulation of PHB steadily increased until day 6 to reach a maximum accumulation of 8.389% (w/w of dcw). The PHB content then declined slightly after day 6. This decline in PHB accumulation of the biomass was attributed to the role played by the polymer in the organism's metabolic pathway. 10 In cyanobacteria, PHB granules serve as a specific carbon reserve that can be used to produce acetyl-CoA, which is a precursor to various biosynthetic products. When the cyanobacterial cells accumulated an adequate amount of carbon compounds (until day 6), they eventually degraded the energy-rich polymers to generate metabolic intermediates for the synthesis of vital amino acids.
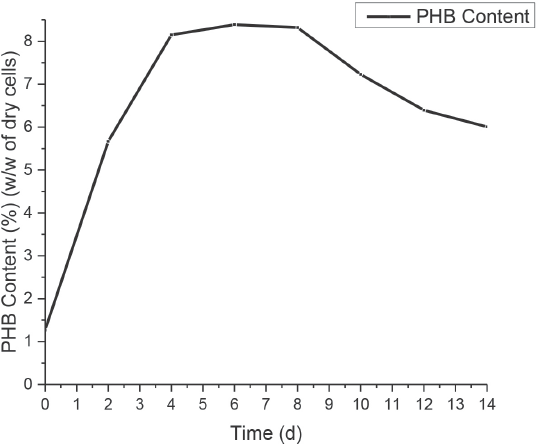
PHB accumulation profile of N. muscorum culture in N- and P-deficient medium (PHB content %w/w of dry cells with respect to days of incubation).
Acetate is an important carbon source for the cyanobacte-rial culture to synthesize PHB. In the presence of acetate, the algal cells achieved a maximum PHB accumulation of 8.389% (w/w of dcw) in 7 days as opposed to 7.31% (w/w of dcw) after 14 days in acetate-deficient conditions as reported in the literature. 16 The positive impact of acetate on PHB accumulation in the Nostoc cells could be credited to the stimulating effect of acetate—there is a direct link between acetate consumption and PHB synthesis in cyanobacteria. The bacterial cells synthesize the precursor of PHB, acetyl-CoA, at a faster and efficient rate when acetate is present in the culture medium, thus enhancing the production of the linear polyester material. 19
The spectrophotometric assay of PHB extracted from Nostoc cells was performed as described in the experimental section. PHB polymers, in the presence of sulfuric acid, degrade to form crotonic acid, which can be detected using a UV spectrophotometer at 235 nm (Fig. 5). Fig. 6 shows the absorption spectrum of the acid digested sample along with the standard—a high degree of similarity was observed between the two spectra. The high and narrow absorption peak of the sample after acid digestion and the absence of any other peaks indicate that the PHA formed in the Nostoc cells was only PHB and not a mixture of other hydroxyalkanoates. 20
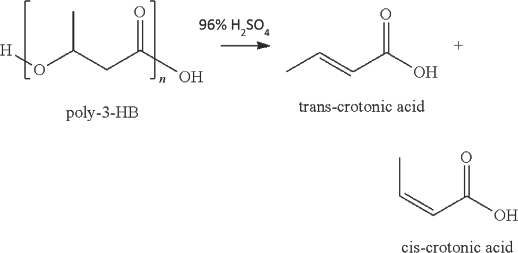
Chemical equation illustrating formation of crotonic acid from polyhydroxybutyrate.
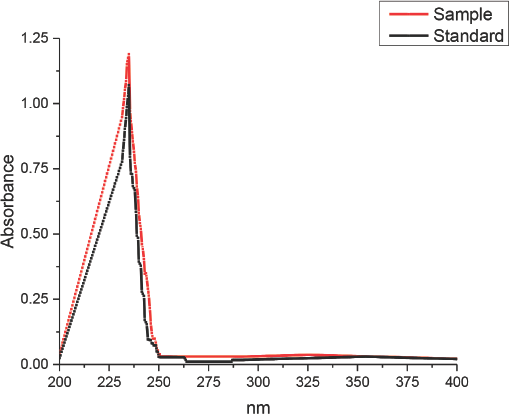
Comparison of the absorption spectrum of N. muscorum sample after acid digestion and standard acid.
The results of the GC-MS analysis of the extracted PHB polymers are given in Figs. 7A–C. Normal and N- and P-starved cells were subjected to methanolysis and analyzed by GC-MS. While no PHB accumulation was detected in the normally cultured cells, the characteristic peak of 3-hydroxybutanoic acid methyl ester (3-HB-Me) at 5.67 min in the gas chromatogram (Fig. 7A and B) of the N- and P-deficient cells confirmed the presence and accumulation of PHB polymers, which is also corroborated by reports in the literature.6,13 In the mass spectrum of the algal-PHB sample (Fig. 7C), the mass-to-charge ratio (m/z) value of 92 was predominantly detected. This indicated the presence of 3-hydroxyacids in the sample by comparison to a reference mass spectrum. Other novel peaks found in the mass spectra (e.g., 88 and 116) were due to derivatives of 3-hydroxyacids, which were most likely formed during the methanolysis process. 20
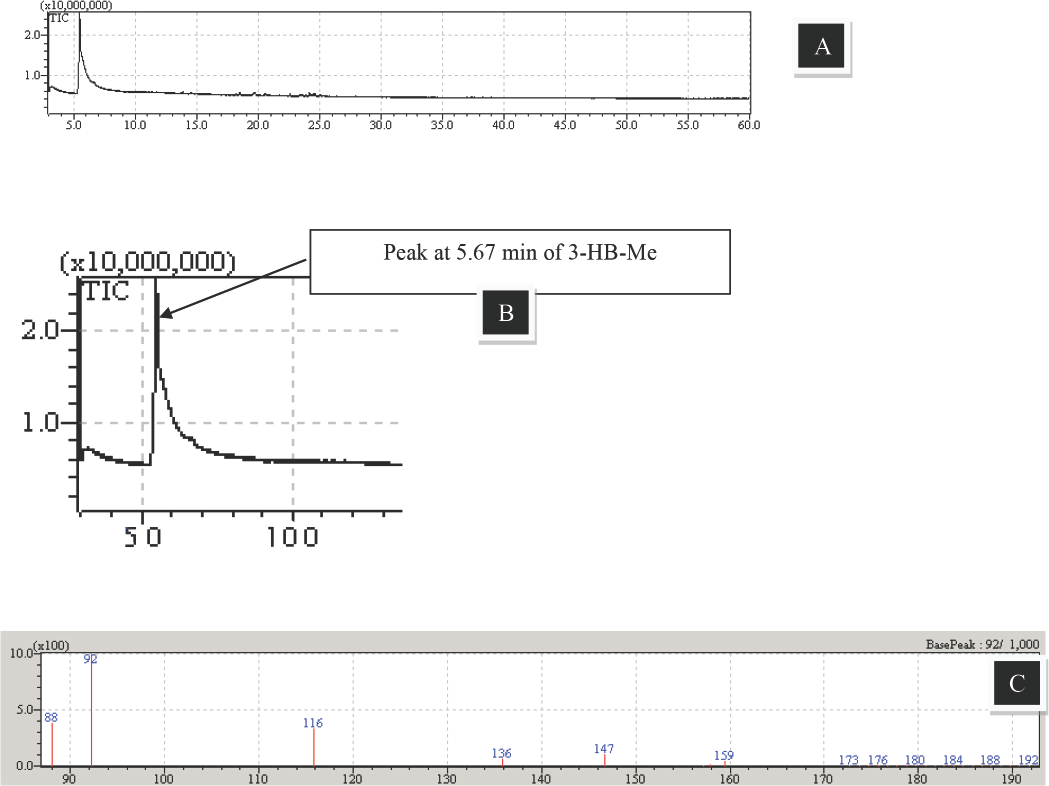
GC-MS analysis of PHB from N. muscorum A) total ion chromatogram of extracted PHB; B) peak at 5.67 min showing the characteristic ion fragment for 3-hydroxybutanoic acid methyl ester (3-HB- Me), C) Mass spectrum (m/z) of the sample.
On the basis of results pursued in this study (that should be supported by further characterization) and related literature,7,16,20,21 PHB can be recommended for use in medical textile applications. This includes their potential use as cardiovascular products (i.e., pericardial and arterial septal repair patches, heart valves, and woven fabric scaffolds for regeneration of arterial tissues), sutures, and wound dressings. 21 Future investigation of the extracted PHB samples will involve characterization of thermal properties (e.g., melting point, glass transition temperature, and thermal degradation temperature), molecular weight analysis, antimicrobial analysis, and tensile and other fiber-related testing to solidify the foundation of its use in the medical textile field.
Conclusions
Polyhydroxyalkanoates are a class of macromolecules that can now be produced photo-autotrophically and mixotro-phically in cyanobacterial species as demonstrated by this study. The results of the physicochemical characterization techniques show poly(3-hydroxybutyrate) (PHB) polymers are rapidly accumulated in N. muscorum, making it a viable cyanobacterium choice for production of the linear polyester material. The exponential intracellular accumulation of PHB in the Nostoc cells over a short time period suggests the economic feasibility of large-scale PHB production using cyanobacterial strains.
With potential uses in biomedical (e.g., tissue scaffolding, prosthetic hernia meshes, wound healing, and drug delivery), personal care (e.g., disposable pads), and food packaging (e.g., food-contact packages) applications, use of PHB materials having appropriate physical and thermal properties may usher in an era of environmentally-friendly and sustainable materials. Further characterization of the N. muscorum generated PHB prepared in this study, including physical and thermal properties, is underway.
Footnotes
Acknowledgements
This research was supported by the AATCC Foundation Student Research Support Grant 2014. We would also like to acknowledge the contribution of The University of Georgia's Bioconversion Lab for their continuing support through facilities and guidance.
