Abstract
UV protection is a key quality attribute for many consumer products such as sunscreens, paints, coatings, and textiles. One common UV protection mechanism, especially for sunscreens, uses metal oxides. Researchers have explored the use of metal oxides, in particular, TiO2 and ZnO, for their UV protection, antimicrobial efficacy, and self-cleaning properties on cotton specimens. This work examines whether three TiO2 extender pigments commonly used in the paints and coatings industry, calcium carbonate, kaolin clay, and barium sulfate, can impart UV-Vis opacity to a reactive dyed cotton/spandex knit fabric. Barium sulfate coatings were found to greatly improve dyed cotton/spandex fabric opacity in both the UV and visible light of the electromagnetic spectrum. Barium sulfate coatings yielded exceptional UV protection on dyed cotton/spandex fabric.
Keywords
Introduction
UV protection is an important aspect of many common consumer products. For example, sunscreens, paints, coatings, and textiles all require highly-efective UV protection. In the case of sunscreens, UV protection is critical in preventing damage to the human body caused by harmful UV radiation. For products such as paints, coatings, and textiles, UV protection is required to maintain polymer integrity and dye stability. Benzophenones and metal oxides are commonly used to impart UV protection. The metal oxides TiO2 and ZnO are extensively used as UV blocking agents in sun-screens.1–3 Sunscreens increasingly use inorganic pigments over organic absorbers due to the lower tendency of irritant reactions. 1 Paint and coating products routinely use large amounts of TiO2 and other inorganic pigments to control gloss and opacity attributes, albeit protective coatings typically use sterically-hindered phenols to prevent polymer breakdown from UV damage.
In the case of textiles, UV protection can be accomplished by fabric construction, dye selection, and by auxiliaries such as UV absorbers.4,5 A brief review of the literature shows that researchers have specifically explored the UV protection of zinc and titanium oxides, as well as other beneficial properties such as antimicrobial efficacy and self-cleaning potential arising from their use on cotton fabrics.6–12
A recent and growing trend in apparel is the athleisure market, a merging of traditional athletic and leisure wear. One area of concern for cotton garments in this market—thin form fitting athleisure wear (e.g., yoga pants) have a tendency to become transparent and may allow more penetration of incident UV light. Therefore, a need exists to easily reduce the transparency of these types of cotton garments.
The objective of this research is to determine if TiO2 extender pigments (i.e., kaolin clay, calcium carbonate, and barium sulfate), commonly used in the paints and coatings markets, show any reduction in transmission of visible and UV light and/or improvement in the ultraviolet protection factor (UPF) when applied to a reactive dyed cotton/spandex knit fabric.
Experimental
Materials
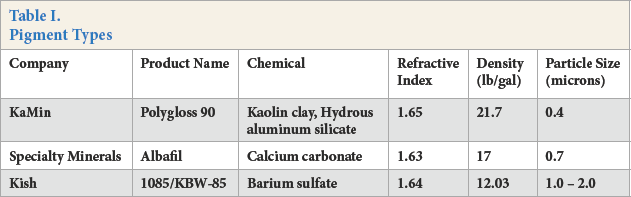
The pigments evaluated are shown in Table I with selected properties and manufacturer information. Pigments assessed included calcium carbonate, kaolin clay (hydrous aluminum silicate), and barium sulfate. In general, the refractive index and particle size were nearly identical for the three pigments, with variations in the bulk density. Other reagents used included defoamer (BYK-028, Altana), dispersing agent (Tamol 731A, Dow Chemical), and a -40 °C Tg vinyl acrylic latex binder (Saitack 3550, STI Polymer).
Pigment Types
The fabric used for testing (Cotton Incorporated) was a brown reactive dyed, plated single jersey, single knit with fiber content: 91% cotton/9% spandex; 60/1 Ne 100% cotton (combed and ring spun), and 1/20 den spandex, with a fin-ished fabric weight of 4.1 oz/yd2 (139 g/m2). This fabric was specifically identified as being highly transparent. The brown color yields a contrast for the white pigment.
Mixing Procedure
To prepare a pigmented stock binder formulation, latex was added to each pigment grind to help bind the pigments to the fabric. Tree different pigment formulations were prepared, one for each pigment type. The target pigment volume concentration (PVC) for each pigment formulation was 54.6%.
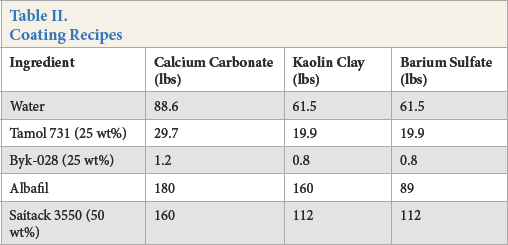
As a background, a typical paint or coating formulation consists of a pigment grind that disperses the pigment, and a letdown that incorporates the latex with the pigment grind. Typically, the batch size formulation is based on 100 gallons. Knowing the densities and accounting for the volume and weight of each ingredient, the volume and weight of solids are easily figured and allow a targeted pigment volume concentration of the dried coating to be ascertained. In the lab, a coating formulation can be made in small batches by weighing in grams the pounds called for in the coating formulation. Table II shows the formula for the full bath size coating of each pigment. In these experiments, one-quarter bath size mixes were made for each formula (i.e., one fourth of the pounds required in the recipe was added in grams).
Coating Recipes
Water, defoamer, and dispersing agent, were mixed for 1 min. The pigment was then sifed into this mixture under shear generated by a Cowls mixer. After grinding for 3 min, the latex was added and mixed for an additional 5 min. The mixture was removed from the mixer and stored in a capped container at room temperature.
Treatment Procedure
From the concentrated binder formulation, 10 g/L and 100 g/L of diluted solutions were prepared. To simulate padding, a 10 × 9.5 in. piece of cotton fabric was weighed and carefully pulled through a rectangular tray containing the appropriate diluted binder formulation. After wetting the fabric by sliding it through the tray, the sample was removed and laid on a plastic board for removal of the excess binder formulation by using a plastic rolling pin. The wet sample was then reweighed to determine wet pickup. The samples were hang dried in a constant temperature/constant humidity room for 24 h. The samples were then reweighed to determine dry pickup. A control fabric was generated using the same protocol with water only. Duplicate samples were made for each treatment and control.
Evaluation Procedures
The visible range (360 to 750 nm) optical transmission properties of the fabric samples were tested using an X-Rite Color iControl I7 bench-top reflectance spectrophotometer. A simple and quantifiable method of evaluating a textile for UV protection is use AATCC Test Method (TM) 183, Trans-mittance or Blocking of Erythemally Weighted Ultraviolet Radiation through Specimens. A numerical UV protection factor (UPF) value was assigned to the fabric. Treated specimens were evaluated for UV resistance using a Labsphere Transmittance Analyzer and assigned a UPF value. Per AATCC TM 183, three scans per specimen were completed to determine the UPF value of a treated fabric. The UPF ratings of two specimens were averaged for a final UPF value for each pigment at each treatment concentration.
Results and Discussion
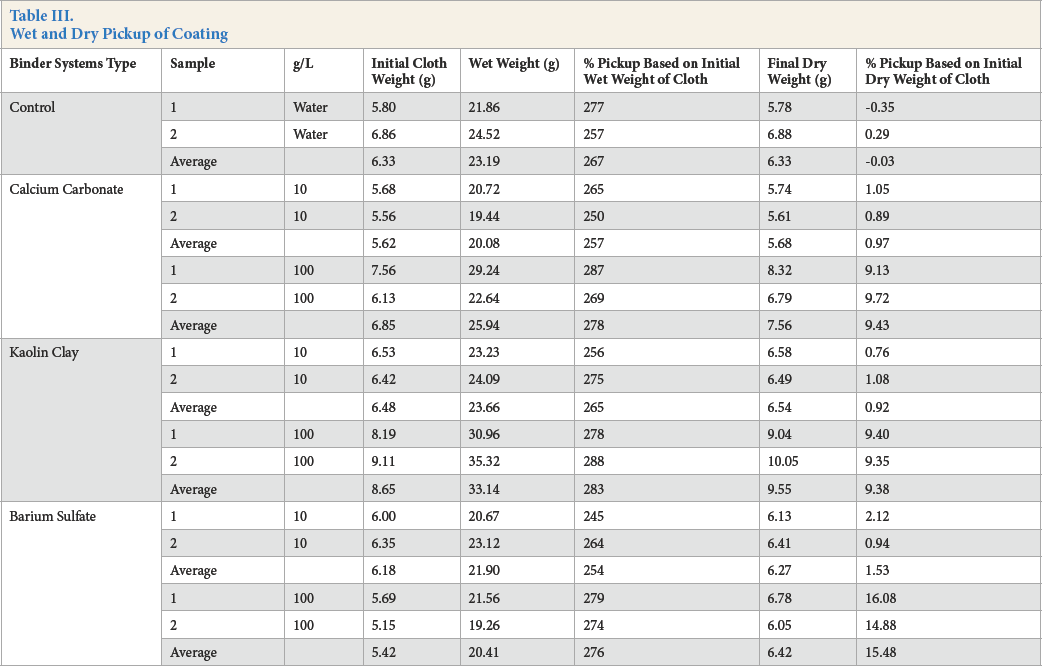
The pigment pickup results, including the average for duplicate treatments, are shown in Table III. Barium sulfate treatments yielded higher final add on than calcium carbonate and kaolin clay.
Wet and Dry Pickup of Coating
Figs. 1–3 show images of the treated and control specimens taken through a variable magnification light microscope.

Calcium carbonate treatments (L-R: control, 10 g/L, and 100 g/L).

Kaolin clay treatments (L-R: control, 10 g/L, and 100 g/L).

Barium sulfate treatments (L-R: control, 10 g/L, and 100 g/L).
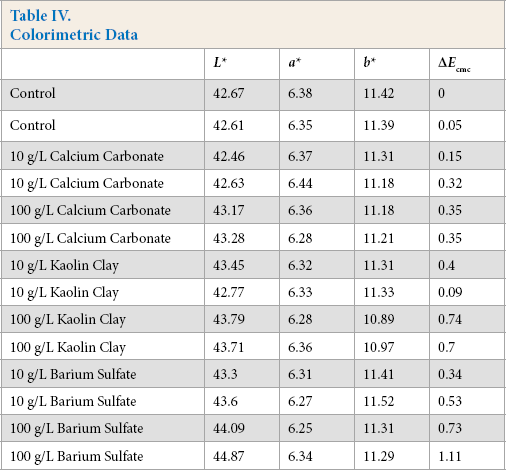
Each image represents ∼0.5-cm diameter. In general, except on the edges of the 100 g/L treatments, the pigments were evenly coated over the fibers without visual clumping or pigment coagulation. Further, as the colorimetric data in Table IV shows, all specimens, except one of the 100 g/L barium sulfate treatment specimens, were within one ΔEcmc unit of the control fabric. Only the L* value changed significantly with each pigment. Increasing the amount of pigment applied increased the L* value of the treated fabric, indicating a higher reflectance/refraction of incident light with increased pigment concentration.
Colorimetric Data
Visible Light Transmission
Fig. 4 shows the transmission of the specimens in the visible range from 360 to 750 nm. The graph indicates that all of the samples showed less transmittance than the control. Lower levels (10 g/L) of calcium carbonate and kaolin clay were only slightly better than the control. However, the barium sulfate showed much less transmission at this concentration. Using the 10 g/L barium sulfate binder mixture, incident transmission of light was less than the treatments at 100 g/L for calcium carbonate and kaolin clay. At 100 g/L level applications, the pigments were ranked for transmission values as barium sulfate < kaolin clay < calcium carbonate.
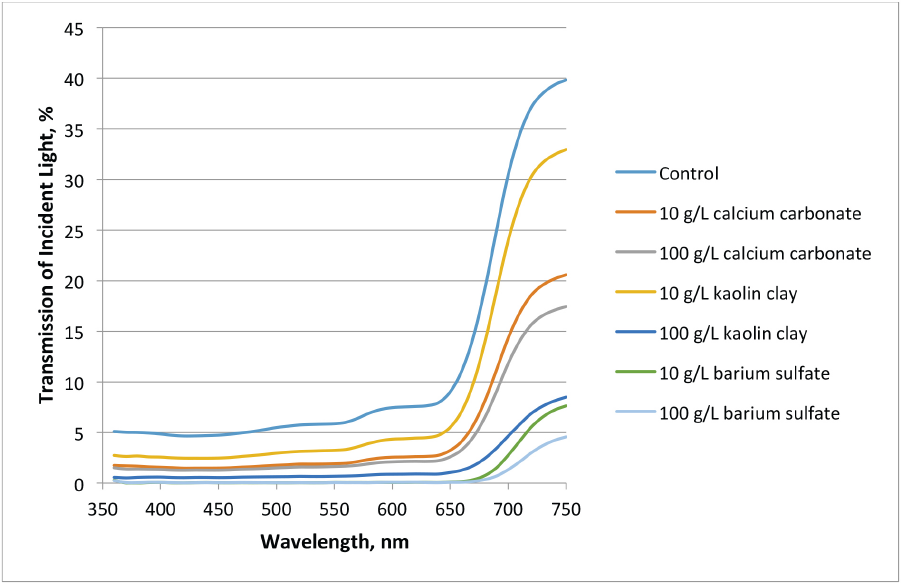
Visible light transmission of treated specimens.
Interestingly, barium sulfate allowed similar visible light transmission for both 10 g/L and 100 g/L applications. Roughly equal transmission was seen with 10 g/L of barium sulfate and 100 g/L of kaolin clay.
UV Light Transmission
Table V shows the individual and averaged UPF ratings obtained for these specimens. Very good results were obtained with barium sulfate, while marginal improvements were seen with calcium carbonate and kaolin clay. The untreated fabric itself provided fairly good UV protection. Although Table V shows that the lower pigment concentrations provided more UV protection, visual observation and some large variations in UPF ratings for duplicate samples suggested that migration and levelness of the higher concentration treatments may be suspect. It is possible that a more realistic pad/dry/cure treatment process may yield different results (i.e., greater UV protection with increased pigment concentration). Conversely, it is also possible that higher amounts of pigment are detrimental to UV protection once a critical concentration was reached.
UPF Ratings
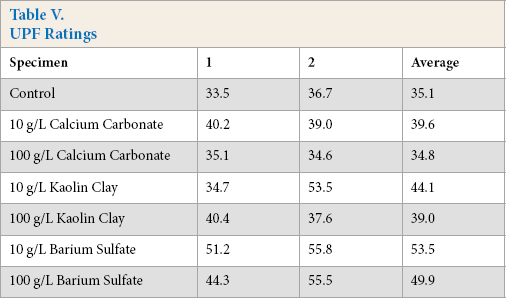
Fig. 5 shows the averaged UV scans of the data shown in Table V and reflects the pigment type with respect to the concentration of pigment and latex mixture used in the treatment. Clearly, barium sulfate allowed much less transmission of UV light than the control, calcium carbonate, and kaolin clay treatments.
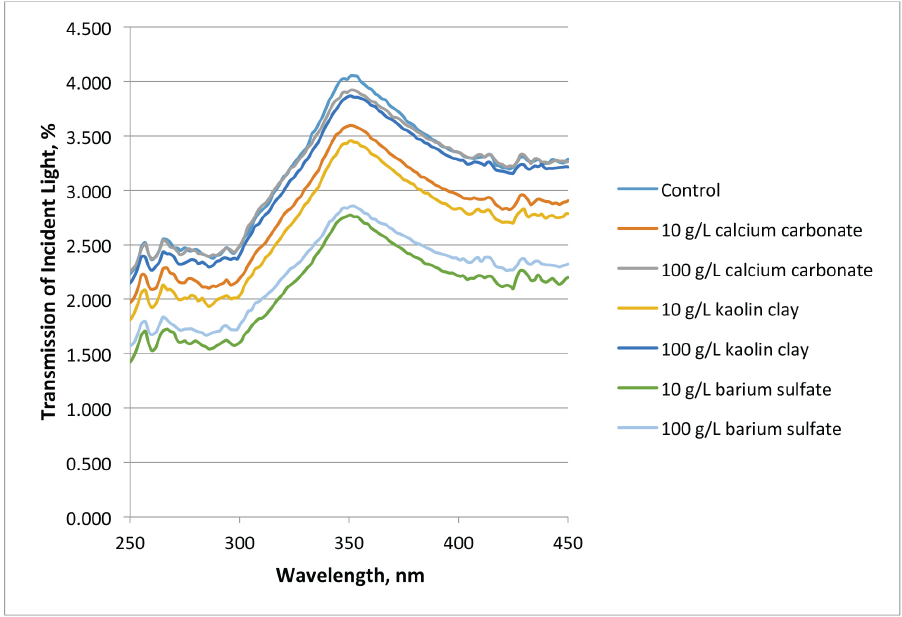
Ultraviolet light transmission of treated specimens.
Conclusions
It was shown that coatings consisting of traditional TiO2 pigment extenders and low Tg (-40 °C) vinyl acrylic latex binder can drastically lower the UV and visible light transmission properties of a dyed cotton/spandex fabric. This was especially true for barium sulfate pigment. Using 10 and 100 g/L of a barium sulfate coating, the UPF value was increased from 35 to 50+. Also, because of the use of a low Tg latex, hand was maintained and was not adversely affected. Further, color change was not highly affected by white pigment addition as only the L* value was increased, indicating a higher reflectance/refraction of incident light. However, at this time, durability of the coatings to laundering is not known. This treatment method may have potential for application in the growing athleisure market to provide modesty and protection to transparent cotton fabrics. Finally, TiO2 extender pigments are significantly cheaper than using TiO2 itself.
