Abstract
Textile technology is advancing by incorporating the benefits of nanotechnology into its diverse fields. Nanoparticle inclusion is one of the potential finishing processes in textile treatment to create multifunctional textiles. The goal of this research is to impart antibacterial, stain-release, and UV (ultra-violet) protection properties on cotton fabric by depositing ZnO nanoparticles on the fabric surface. Following the synthesis of the nanoparticles by the use of a sonochemical strategy, the nanoparticles are then fixed onto the fabric through the application of a mechanical thermo-fixation technique. The nanoparticles are applied to the fabric at three different concentrations: 0.5%, 1%, and 2%. The surface characterization shows that nanoparticles are of an average size of 40–100 nm and nearly spherical in shape. The colony count method is used to quantify the antibacterial activity of the nano-treated fabric. The results indicate that the treated fabric is effective against both gram-positive (S. aureus) and gram-negative (E. coli) bacteria. The color stain of the direct blue dye of treated fabric has degraded under UV irradiation, indicating the stain release property. The results demonstrate considerable stain release that rises with the amount of ZnO nanoparticles. Excellent UV protection is also found since 2%-ZnO nano-coated fabric shows around 96% blockage of UV rays.
Introduction
Cotton, one of the most significant natural resources, is frequently utilized to create textiles for use in daily life. 1 It is very strong, chemically stable, flexible, hygroscopic, dyeable, comfortable, and biodegradable. 2 Due to its improved value for textiles and contribution to a healthier and more pleasant life, cotton fabrics offer a wide range of potential applications.2–4 Thus, the product development and diversification of cotton goods are very important to achieving a growing international market. Moreover, modern cotton should be functional, durable, comfortable, hygienic, and smart-looking. 5 To meet cotton cloth advancement, nanotechnology is a very challenging issue.6,7 Nanotechnology creates unique materials at the nanoscale. Nanomaterials are small particles with a large specific surface area and unique optical, mechanical, catalytic, and biological capabilities. 8 The use of nanomaterials concerns creating functional properties without altering the comfort properties of the textile substrate. 9 The textile finishing section mainly focuses on using metallic nanoparticles to create value-added functional textiles.10,11 Several studies have been conducted on the application of nanoparticles to textile materials to develop finished fabrics with various properties. 12 For instance, nanoparticles can make textiles water-resistant, soil-resistant, wrinkle-resistant, antimicrobial, antistatic, UV-protective, flame retardant, and more.13–15
Further, the use of nanoparticles can overcome the inherent and existing problems of cotton fabric. Since cotton is a fibrous material and provides a large surface area, it has an excellent environment to promote bacterial growth16,17 which is responsible for the spread of microorganism-caused infection and causes serious health problems. People in modern society are very concerned about their health, so the demand for health-safety products is growing by the day. 18 The inclusion of nanoparticles on cotton fabric solves this serious problem and ensures environmental safety. Tania et al. 19 revealed that Ag nanoparticles synthesized via the in situ method on dopamine-modified cotton fabric have outstanding antibacterial activity. The treated fabric showed a 100% bacterial reduction for both S. aureus and E.coli bacteria after 24 h, demonstrating that it is completely bacterial resistant. According to Mehravani et al., 20 gold nanoparticles and textile materials have enormous potential for producing antimicrobial materials for health and hygiene goods, such as infection control and barrier materials, with improved properties. Based on the findings of the research21–23 metal nanoparticles are a more promising substance to use in the production of antimicrobial fabrics. However, the high cost of silver and gold nanoparticles restricts their application in industry as antibacterial agents, although these particles are extremely effective against microorganisms. 18 On the other hand, metal oxide nanoparticles (ZnO, CuO, and MgO) are now frequently used in the textile industry to manufacture functional textiles. The explosive growth of functional materials, which are created by loading various metal oxides onto fabrics, has attracted attention in recent years. Importantly, the metal oxide nanoparticle is a widely used antimicrobial agent12,24 since it is effective against a wide range of microorganisms. The inorganic nano metal oxides ZnO, TiO2, MgO, and CaO are particularly efficient as they are stable under harsh process conditions and safe for human beings and animals.25–28 Among the mentioned metal oxides, ZnO is gaining more interest in the textile research field due to its cost-effectiveness, accessible availability, and unique chemical and physical features. 18 Nano ZnO is utilized in photocatalysis, solar cells sensors, displays,29,30 electroacoustic transducers, photodiodes, anti-reflection coatings, 31 sunscreens, UV absorbers, 32 piezoelectric devices, and UV light-emitting devices 32 and many more. Besides, ZnO is a non-toxic, bio-safe, and biocompatible material and is used in biomedical applications without any external coating. 20 Therefore, ZnO nanoparticles are considered raw materials for cosmetics, polymers, food packaging, 33 medicine, 34 etc., to incorporate various functional properties. Textile fabric is regarded as vital material among others that are gradually advancing by utilizing the perspective of nano ZnO. Most importantly, nano ZnO can protect the textile fabric from the harmful effect of UV rays. Sunburn, skin cancer, and allergies are all caused by excessive exposure to UV radiation. Our body gets protection from skin-related diseases by wearing sun-protective clothes made of nano ZnO. Jin et al. 35 reported that the cotton fabric which protects UV rays is an important component of outdoor textiles. UV protection is discovered due to the nano ZnO bandgap of 3.37 eV and the ability to absorb light at 376 nm, 36 which is the wavelength range of ultraviolet rays. 37 Additionally, when exposed to ultraviolet light, the photo-catalytic characteristic of zinc oxide allows it to act as an antibacterial and self-cleaning agent. 27 Self-cleaning is one of the important functional properties of textiles 36 because it makes the fabric resistant to coffee and red wine stains while also repellant to water, dirt, odor, and so on. Stain release is one of the main phenomena of self-cleaning properties which refers to the degradation of various color stains. By examining the removal of color from a fabric that had been dyed and treated with nanoparticles, Montazer and Seifollahzadeh’s 38 research was capable of pinpointing the self-cleaning characteristic. This emphasizes the interrelation between the stain-release and self-cleaning properties. Kathirvelu et al. 39 studied the stain elimination property of nano-treated cotton-polyester blended fabric and found a good result. Other related work also reported the self-cleaning and stain release properties of textiles by using ZnO nanoparticles.36,40
Since ZnO has several functionalities, current research is being done to embed this nanoparticle into the cotton fabric to create a protective textile. Herein, nanoparticles are applied using a thermal-fixation approach 41 in conjunction with an acrylic binder. Binder can enhance the fastness of nanoparticles against washing. 28 As one of our previous related research 42 reveals that binder is an essential auxiliary chemical for the fixation of ZnO nanoparticles, where we found the excellent antimicrobial activity of ZnO nano-treated cotton. Along with antimicrobial activity, this study also reveals the stain release characteristics and UV protection properties of treated cotton fabric. Although conventional finishing agents can impart the functional properties mentioned on cotton fabric, most of these finishing agents are toxic and hazardous to the environment. Furthermore, a separate chemical is needed for separate functions. So we are inspired to work with nano ZnO to make a value-added and protective textile. Additionally, three different ZnO nanoparticle concentrations have been employed, which is novel because it was not identified in the previous researcher’s study, to highlight the impact of nanoparticle concentration on the mentioned functional attributes.
Materials and methods
Materials
For the experiment, grey cotton fabric of plain weave structure manufactured by Paramount Textile Ltd. (Gazipur, Bangladesh) is used. The specification of the fabric is 100% cotton with 73 ends per inch (EPI) and 68 picks per inch (PPI) and a weight of 151 GSM (grams per square meter). Pretreatment of the fabric is required prior to the application of nanoparticles.
The chemicals named Acrylic Binder: Helizarin SFT (Brand name), Detergent: Imerol PCLF, Wetting Agent: Leonil EHC, Peroxide Killer: Bactosol SAP, Desizing Agent: Bactosol HP2A, and Peroxide Stabilizer: Pretogen FCB are supplied by Archroma Bangladesh Ltd. The other chemicals that are needed to synthesize nanoparticles such as Zinc acetate dihydrate [Zn (CH3CO2)2·2H2O], NaOH and ethanol are purchased from Sigma Aldrich, Germany. Direct-blue dyestuff used for the color staining test is provided by Orient Chem., Bangladesh.
Methods
Preparation of ZnO nanoparticles
Zinc oxide nanoparticles are produced with some modification utilizing Zn(CH3COO)2·NaOH as described by Shahidi et al. 42 Ethanol is used as a solvent for this purpose. The experimental set-up for the preparation of ZnO nanoparticles is shown in Figure 1. First, prepare 0.5 M solutions of Zn (CH3COO)2·2H2O in ethanol at room temperature, followed by 100 ml of 0.1 M NaOH aqueous solution in a separate bucket. The NaOH solution is then added slowly, drop by drop, to the 25 ml Zn acetate solution and stirred continuously for 15 min. Then the mixture is sonicated for 1 h with a 60-watt ultrasound bath. All these processes are carried out at room temperature without any specific conditions. The sonochemical process is constantly in motion. After 1 h of ultrasonic treatment, the solution is heated at 75°C for another hour to evaporate the ethanol, and finally, the ZnO nanoparticle is obtained in powder form by oven drying.

Experimental set-up for the preparation of ZnO nanoparticles (NPs).
Characterization of nanoparticles
The nanoparticles are characterized using a scanning electron microscope (SEM) and X-ray diffraction analysis (XRD). An extremely concentrated scanning electron beam is utilized in SEM examination to bombard a sample’s surface, producing a huge number of secondary electrons whose intensities are varied by surface topography. Sampling is needed before the SEM analysis which is done by following the previous work. 43 The nanoparticles are coated with a gold layer by vaporization of gold in an argon environment, followed by drying using the JFC 600 fine auto coater, Malaysia. In a coating machine, nanoparticles are deposited on copper-covered carbon tap double faces and coated with a gold layer by vaporization of gold in an argon environment, followed by drying. The discrete particles are scanned using a field emission electron microscope (JSM-600, Tokyo, Japan). Figure 2 shows the image of nanoparticles at different magnifications, from 10,000 to 50,000. The morphology, structure, and size of prepared nanoparticles are clearly shown in the images in Figure 2(a)–(c). The approximate size of a nanoparticle is 40–150 nm.

SEM (scanning electron microscope) images of ZnO nanoparticles in different magnification: (a) ×10,000, (b) ×30,000, and (c) ×50,000.
XRD (X-ray diffraction) analysis is performed using an X-Ray Diffractometer from Phillips, Expert Pro, Holland. This approach is used to determine whether a substance is crystalline or amorphous at the atomic level. 44 The XRD peak of ZnO nanoparticles showing the crystalline size and shape are shown in Figure 3. The mean size of ZnO nanoparticles is determined by XRD patterns using the Debye–Equation Scherrer’s, 15 which is shown in equation (1):
where θ = 2θ/C, d = the full width at half the maximum intensity of the peak, and λ = the wavelength of the X-ray source (24056°A) (as shown in Figure 3). It can be found from this equation that the average size of a ZnO nanoparticle is 54.33 nm.

XRD pattern of ZnO nanoparticles.
Pretreatment of cotton fabric
The pretreatment processes called desizing, scouring, and bleaching are necessary to incorporate to prepare grey cotton fabric for proper nano inclusion. Those treatments remove the sizing material, dirt, dust, oil, wax, and impurities, destroying the natural grey color of the raw cotton fabric. The pre-treatment process has a great impact on impurities removal, enhancing the whiteness index, increasing absorbency, and imparting a permanent whiteness to the fabric. 45 The pretreatment strategy is based on previous work,46,47 with certain modifications. The combined pretreatment for cotton fabric follows the process curve indicated in Figure 4. The bath with the fabric is initially set at room temperature with the necessary amounts of wetting agent, detergent, glauber salt, acetic acid, and desizing agent. The H2O2 stabilizer and alkalis are added after 20 min and run for 30–60 min. The bath is then drained, and the fabric is given hot rinses to wash. Finally, the pre-treated fabric is collected after being dried at a 60°C temperature.

Combine desizing, scouring, and bleaching of cotton fabric.
Coating of cotton fabric with ZnO nanoparticles
A technique known as thermo-fixation 41 is used to apply ZnO nanoparticles to fabric with a padding machine, followed by drying and curing. The nanoparticles are dispersed in a 50% aqueous solution of ethanol. Three solutions of ZnO nanoparticles are formed with concentrations of 0.5%, 1%, and 2% on a weight basis of the fabric, along with a 1% acrylic binder. The prepared working solution is kept in a magnetic stirring machine at a 60°C temperature for 15 min before padding. Then it is padded on a laboratory padding machine by maintaining a 73% pick up. The fabrics are dried at 70°C in a preheated oven. Finally, it is cured in a Warner Mathis curing machine at a temperature of 150°C for 5 min to fix the nanoparticles inside the fabric. The coated samples are named Cot-1 for 0.5%, Cot-2 for 1%, and Cot-3 for 2% ZnO nanoparticle.
Functional property analysis
The current study evaluated the performance of ZnO nano-coated fabric by measuring antibacterial activity, UV protection, and stain release properties. The standard technique is used here to compare the antibacterial activity of nano-treated and untreated cotton fabric. 48 Antimicrobial activity is evaluated against gram-positive: S. aureus and gram-negative: E. coli by the colony counting method and results are expressed as bacterial reduction percentage (R%). For this test, the number of viable species in suspension is estimated, and the percentage reduction is measured based on recoveries from the untreated sample. This approach is intended for those surfaces with a 50–100% reduction capacity for the required contact time. 49 The following formula calculates the percentage of bacteria killed 50 within the designated time as shown in equation (2):
The bacterial reduction percentage
“B” is the number of colonies in control and “A” Number of colonies after the selected hour of introducing samples.
For analysis of UV protection properties, the UV radiation of solar light is divided into three different categories: (i) UV-A (320–400 nm), (ii) UV-B (280–320 nm), and (iii) UV-C (200–280 nm). The UV protection of our treated and untreated fabric is measured by a PerkinElmer UV visible machine from the USA, model- LAMBDA 1050+. It provides the transmittance curve of every sample. By analyzing the transmittance curve, the UV protection power is evaluated here.
For measuring the stain release property we were motivated by earlier research, where the stain release properties of nano-treated and untreated fabrics were examined as the reduction of color stains. 51 Herein, the samples with dye stained are exposed under a light source (CAC 60, Verivide, UK) for 24 h using a lightbox. Before that, direct blue dye (direct blue 5 SD, CI. 199) is used for the color-staining of both treated and untreated fabric at a concentration of 1%. For the preparation of the color stain sample, the cotton fabric is dyed with direct dyestuff in a laboratory dyeing machine using the procedure described by Chattopadhyay and Patel. 52 The dyeing conditions are maintained at a temperature of 100°C, time 30 min with a 1:30 material to liquid ratio. A spectrophotometer (SF 600, Data Color, USA) assesses color strength (K/S) in samples to determine whether the color has been photodegraded. The Kubelka and Munk equation (equation (3)) shows how the ratio of K to S affects the depth of color: 53
where R is the reflectance of the dyed fabric, K is the sorption coefficient, and S is the scattering coefficient. For a single color, the value of K/S is higher for a deep color and lower for a light color.
Results and discussion
SEM analysis of untreated and ZnO nano-treated fabric is done to observe the nano deposition and surface morphology of treated fabric. The antibacterial activity is assessed to determine the bacterial resistance power, and the stain release property is measured to express the self-cleaning activity of nano ZnO-coated fabric. Finally, the UV protection property is tested to see how well nano-coated fabric can stand up to the harmful UV rays of solar light.
Surface morphology analysis by SEM
A scanning electron microscope (SEM) with an accelerating voltage of 20 kV and a current of 10 mA is used to analyze the surface morphology of cotton fabric at a magnification of 1000. As shown in Figure 5, different percentages of ZnO nanoparticles are deposited. The untreated fabric shows (Figure 5(a)) a smooth and clear image of the cotton fibril structure. Three treated samples are shown in Figure 5(b)–(d) with different concentrations of ZnO nanoparticles. Those figures show a rough surface with the presence of nanoparticles in various shapes. The sample with 2% ZnO nanoparticles in Figure 5(d) shows more nano deposition and agglomeration. The highest concentration of ZnO nanoparticles causes more deposition of nanoparticles on fabric.

SEM image of (a) untreated fabric, (b) 0.5% nano ZnO coated fabric, (c) 1% nano ZnO coated fabric, and (d) 2% nano ZnO coated fabric.
Antibacterial activity
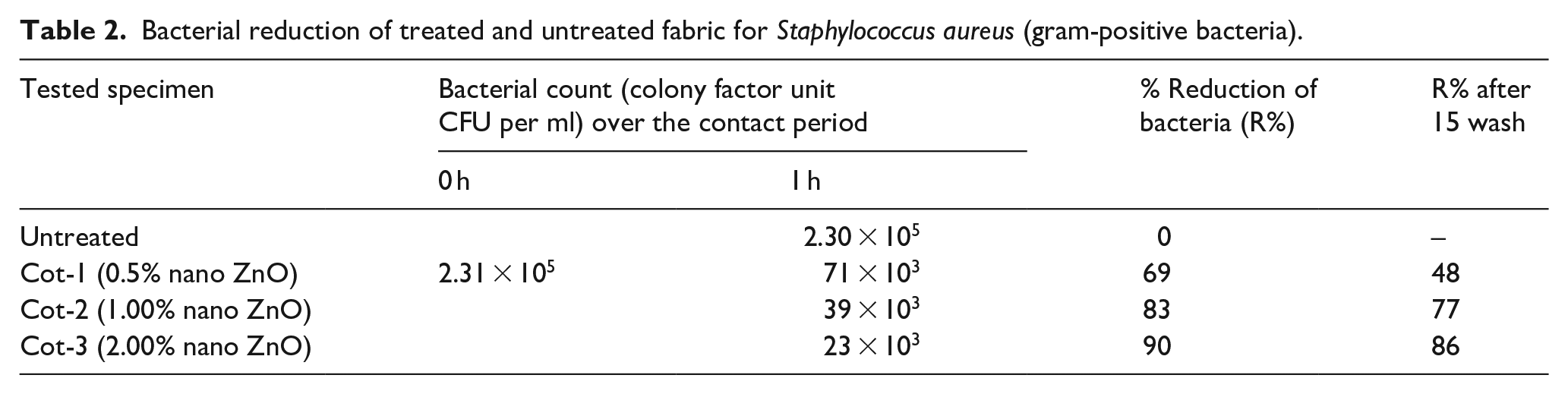
Antibacterial active textile substrates are capable of killing or inhibiting the growth of microorganisms. Nanoparticles of ZnO kill bacteria more selectively than bigger particles because they are small (less than 100 nm) and have a high ratio of surface to volume. 54 A quantitative antimicrobial test method developed to assess the resistance of non-leaching antimicrobial treated specimens to the development of germs under dynamic contact conditions is used to assess the antibacterial characteristics of nano ZnO treated fabrics. The results of bacterial reduction for gram-positive bacteria are represented in Table 1 and for gram-negative bacteria in Table 2. In addition, Figure 6 demonstrates the disk image of bacterial cells tested for treated and untreated fabrics. These are the images of the 2% nano ZnO coated sample called Cot-3 after 1 h of contact time. By comparing the existence of bacteria by pairs in Figure 6(a)–(d), it can be observed that the CFU (colony factor unit) notably reduces for both bacteria in the treated fabric. So, the treated fabric is capable of killing a sufficient amount of bacteria. The result of bacterial reduction from Table 1, and Table 2 shows that the bacterial reduction is highest for higher ZnO nano-coating and lower for a lower percentage of the nano-coated fabric. Gram-positive bacteria named S. aureus give the highest amount of bacterial reduction up to 90% after 1 h for sample Cot-3, which is very significant in antibacterial activity. For evaluating the durability of nano-ZnO treated cotton fabric toward repeated laundering, the treated sample is submerged in a washing solution containing non-ionic detergent (2 g/liter). The samples are stirred for 15 min at 50°C. Then, it is rinsed with tap water and air-dried. This procedure is repeated up to 15 times and the antimicrobial property of the washed sample is determined. The wash durability is checked only for the sample against S. aureus and the result is shown in Table 2. The obtained result indicates that a satisfactory reduction of microorganisms is found after 15 washes. Sample Cot-3 shows an 86% bacterial reduction after washing. Due to the use of a binder as a fixing agent, we found satisfactory wash durability. Our prior work 47 also supports the effect of binder on the durability of antibacterial action. Our result of antibacterial activity was found to be accurate when we compared it to other relevant publications. According to El-Nahhal et al., 55 90–93% of bacteria are killed on the surface of the ZnO nano-treated cotton fabric. But the wash’s durability was so bad when the washing cycle was extended. However, our wash stability is higher than that as a result of the use of a binder. According to published research, 56 for our samples, the acrylic binder fixes more nanoparticles, producing a strong antibacterial effect.
Bacterial reduction of treated and untreated fabric for Escherichia coli (gram-negative bacteria).

Bacterial reduction of treated and untreated fabric for Staphylococcus aureus (gram-positive bacteria).

The number of survival cells after 1-h contact: untreated fabric (a) S. aureus, (b) E. coli and treated fabric with 2% nano ZnO (Cot-3) (c) S. aureus, and (d) E. coli.
Stain release property of ZnO nano-coated fabric
Stain-release fabrics are those that clean themselves without any substantial physical assistance. It is one of the measures of self-cleanable fabric. It means the stains will leave the fabric more efficiently and faster without hampering the fabric quality. The degradation of dye stains is used to investigate the release of chemical stains. 51 For the assessment, the coated samples are subjected to an artificial UV light source for variable amounts of time at different intervals (up to 24 h) using a lightbox (CAC 60, Verivide, UK). Spectrophotometers measure the color strength (K/S) of the samples to determine whether or not there has been photodegradation of color (SF 600, Data Color, USA). Figure 7 shows how the color strength of an untreated fabric and three treated fabrics, Cot-1, Cot-2, and Cot-3, changes over time. It also shows what happens when the color strength changes.
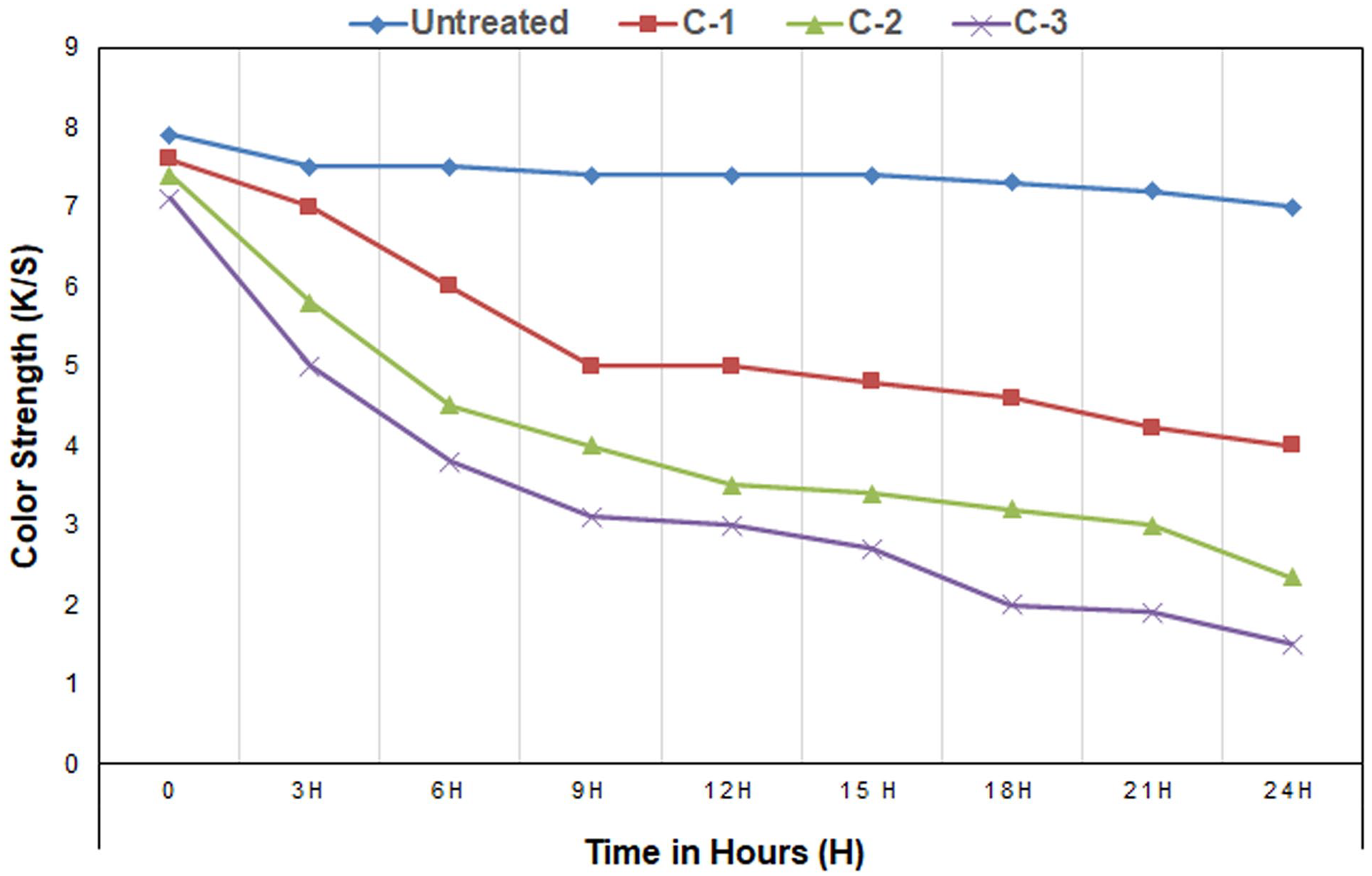
Color strength (K/S) values of untreated and ZnO nano-treated fabric at different intervals of time represent the dye stain degradation.
The decrease in K/S value is calculated by dividing (the initial value minus the instantaneous value) by the initial value. The result is given as a percentage (%). The curve of untreated fabric in Figure 7 demonstrates that after 24 h of UV light exposure, there is no substantial reduction in color staining. On the other hand, the treated fabrics Cot-1, Cot-2, and Cot-3 show a very notable reduction in color stain after 3–24 h of light exposure. The rate of reduction increases with time. For example, after 24 h of light exposure, a sample called Cot-3 with 2% ZnO nanoparticles shows nearly 78% color degradation. So, ZnO nanoparticle deposition causes color degradation easily, but the reduction rate is not linear with time. It is found that the color reduction abilities of textiles are correlated with the amount of ZnO nanoparticles present in the impregnation solution. The findings revealed the color reduction capabilities of the textiles are enhanced when the concentration of nano ZnO nanoparticles in the impregnation solution is increased. This is similar for other nanoparticles too. Montazer and Pakdel 55 applied TiO2 nanoparticles to wool fabric and found similar findings for color reduction, which supports our findings.
UV protection
The UV protection of treated and untreated fabric is shown in Figure 8, which is represented as the transmittance value of wavelength 200–700 nm. The treated fabrics, Cot-1, Cot-2, and Cot-3, show lower UV transmittance than the untreated fabric. The lower transmittance reflects the higher UV blockage as the UV rays do not pass through the fabric surface. The quantitate figure is shown in Table 3. The table indicates that Cot-1 gives UV transmittance of 40% in the UVA region. It means that 60% of UV light is blocked by the treated fabric having 0.5% nano ZnO. For the most concerning UVB region, Cot-1 gives 68% UV blockage. The second sample, Cot-2, shows 22% transmittance, which means 78% of UV blockage in the UVB region. The highest concentration of ZnO lies in the samples, Cot-3 shows the highest 98% UV blockage in the UVB region since it gives only 6% transmittance of UVB light (Table 3).
Average UV transmittance (%) of different wavelengths with UPF range.


UV transmittance curve of untreated fabric and nano ZnO coated fabric.
Table 3 also shows the typical UPF (ultraviolet protection factor) ranges, which are calculated using the literature 57 and the AS/NZS 4399 standard. The previous study 58 shows that 30 ± 6 UPF indicates very good UV protection with 6% UV transmission. Our coated sample Cot-3 provides around 31 ± 3 (average UPF range), representing that we have revealed outstanding UV protection by using a higher concentration of nano ZnO. When all of the data in Table 3 is compared, it is clear that the UV protection of cotton fabric increases as the amount of nano zinc-oxide increases.
Conclusions
The sonochemical approach produces almost spherical ZnO nanoparticles with a size range of 40–150 nm which are applied to the cotton fabric with the aid of a cross-linking agent known as an acrylic binder. The presence of nanoparticle deposition on fabric surfaces of various shapes and sizes is confirmed by SEM analysis. The surface morphology of treated cotton fabric obtained by SEM images also shows that the increasing amounts of nano ZnO, cause the agglomeration of nanoparticles on the fabric surface. The ZnO nano-coating imparts some functional properties to the cotton fabric, which are found by quality evaluation of treated fabric and compared with that of untreated fabric. The results demonstrate that the treated fabric shows excellent antibacterial activity as well as a high stain release rate. Every coated sample shows good antibacterial activity against both gram-positive, S. aureus and gram-negative, E. coli bacteria. The sample coated with 2% ZnO nanoparticle has the highest antibacterial activity, killing 90% of bacteria after 1 h. The wash durability of antimicrobial property is also found to be good as the sample Cot-3 shows 86% bacterial reduction after 15 washings. The washability is ensured due to the use of binder as a fixing agent during nano-coating. The self-cleaning property expressed by dye stain degradation is also very satisfactory for all treated fabrics. Approximately 78% of the color stains on the treated cotton fabric have been degraded within 24 h of UV irradiation. The UV protection of nanocoated fabric is found to be excellent. As the sample having the highest nano concentration, Cot-3 shows 96% UV blockage. Therefore, the overall results indicated that ZnO nanoparticles can be used to create a functional cotton textile with antimicrobial, self-cleaning, and UV protective properties. As a result, the treated fabric may be an excellent choice for use as a high-tech functional, protective, and value-added textile. The potential of ZnO nanoparticles can be used in future studies to improve the functionality of other natural fibers like jute, wool, and silk, thereby increasing the use of natural products in modern markets. According to all the information provided, nano ZnO may be utilized more frequently in textiles than other metallic nanoparticles like silver and gold due to its inexpensive cost and multifunctional properties. ZnO nanoparticles have the potential to be used in the textile industry much more than it is now, according to all the facts presented.
Footnotes
Acknowledgements
The authors would like to thank BUET (Bangladesh University of Engineering) and BUTEX (Bangladesh University of Textiles) for providing the testing facilities for this research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.



