Abstract
The deterioration of organic film due to chemical and UV attack is an increasing concern in paint technology. Thus, the development of new material for UV blocking anticorrosive film draws significant attention in materials science research. This can be achieved by the incorporation of wide band gap nanoparticles like titania (TiO2NPs) and zirconia (ZrO2NPs) in electroactive polymer namely poly(pyridine-4-yl-methyl) methacrylate-co-butyl methacrylate (poly(PyMMA-co-BMA)) film (hybrid film) for the protection of mild steel (MS) from corrosion. The TiO2NPs and ZrO2NPs in combination with polymer absorb more UV light which prevents the deterioration of film. The hybrid material made of poly(PyMMA-co-BMA) and wide band gap nanoparticles was prepared by in situ solution polymerization. The resultant hybrid materials were characterized by various techniques namely X-ray diffraction studies (XRD) and transmission electron microscopy (TEM). The hybrid materials were deposited as film on the MS by spin coating method. The anticorrosive performance of hybrid films was analysed out using potentiodynamic polarization and electrochemical impedance spectroscopy (EIS) studies. The surface examination of films were characterized with scanning electron microscope to confirm the formation of poly(PyMMA-co-BMA) and its different nanocomposite films on MS. The UV blocking studies were also carried out using UV-visible spectroscopy. The electrochemical and optical studies reveals that the poly(PyMMA-co-BMA)/TiO2 film on MS in 3.5% (w/v) NaCl provides better protection against corrosion than ZrO2 based nanocomposite hybrid film.
Introduction
The protection of steel surface against corrosion in marine environment is typically done by using organic coatings. Nevertheless, degradation of organic coating takes place upon exposure to UV radiation on its coated surface. Hence, researchers are combining the inorganic nanoparticle with the organic polymer attributes to boost the anticorrosive properties and stability of organic coating materials.1-5 Ammar et al. reported that the incorporated nano ZnO particles in the hybrid polymeric matrix had improved the hydrophobicity, anticorrosive and thermal properties of coating. 2 The inclusion of titanium dioxide nanotubes significantly enhances the thermal stability, corrosion and heat resistant ability of epoxy resin. 3 It is apparent that the addition of nanoparticles in polymers could improve the anticorrosive property of resultant organic hybrid coatings. According to Ramezanzadeh et al. the progress in corrosion protection performance of the organic hybrid coatings (epoxy based) can be explained with the following mechanism: first, well-dispersed nanoparticles within the polymeric matrix lead to improve the quality of coating by reducing the porosity and tortuousness in the diffusion pathways. Secondly, the inclusion of the nanoparticles strengthens the adherence of the cured epoxy on the surface of the substrate. 6
Generally, conducting polymers and their nanocomposites are the best choices for anticorrosive application but possess poor solubility, hence they are coated by electrodeposition method.
In this context, the research is targeted to study the influence of other metal oxide nanoparticles having wide band gap like titania (TiO2NPs) and zirconia (ZrO2NPs), which are the UV light absorber, in the UV blocking anticorrosive performance of electroactive methacrylate polymer. It is expected that different metal oxide nanoparticles in electroactive polymer experience diverse passive layers formation and leads to unique mechanism of corrosion prevention. Thus, an attempt has been taken to study and compare the anticorrosive properties of poly(pyridine-4-yl-methyl) methacrylate-co-butyl methacrylate) (poly(PyMMA-co-BMA) and its metal oxide nanocomposites in 3.5% (w/v) NaCl medium.
Materials and methods
Materials
Titanium isopropoxide, zirconium isopropoxide and nitric acid were purchased from Sigma Aldrich, India. Pyridine-4-methanol, methacrylic acid and n-butyl methacrylate were obtained from Merck, India. Azobisisobutyronitrile, (AIBN) (Sigma Aldrich, India) was crystallized from ethanol at 50°C. Oleic acid was obtained from Sigma Aldrich, India. Tetra hydro furan (THF), n-propanol, chloroform and ethanol were acquired from Merck and dried using sodium metal. All the other chemicals were procured from Merck and purified by standard methods.
Preparation of TiO2 nanoparticles
0.1 ml of 8 M Nitric acid was added drop by drop into 34 ml ethanol under vigorous stirring. 9.75 ml of titanium isopropoxide were slowly added to the solution followed by stirring for 10 minutes. The obtained pale-yellow mixture was added into 150 ml of distilled water slowly under stirring in room temperature. The resultant solution was treated at 50°C in the oven for 12 hours to complete crystallization. After the gelation, the wet gels were washed with distilled water and dried at 80°C. Finally, the powder was grounded to obtain fine particles.11,12
Preparation of ZrO2 nanoparticles
Zirconium isopropoxide (17.7 g) was diluted with n-propanol (41.3 g) and hydrolyzed by drop wise addition of aqueous ammonia under continuous stirring until the pH 10−10.5. After the completion of the hydrolysis, the sol was continuously stirred for an hour in room temperature to get polymerized. The obtained gel was dried in an oven at a temperature of 110°C for 12 h and under vacuum at 70°C temperature in a Rotavapor. 13
Surface modification of metal oxide nanoparticles
Surface modification of metal oxide nanoparticles (NPs) (TiO2 and ZrO2) with the oleic acid were carried out. 50 ml of 2% oleic acid solution in ethanol was prepared and 0.5 g NPs was added under stirring at 50°C. After 4 hours, the contents were centrifuged (1 × 106 rpm) and the modified NPs were collected, washed with ethanol followed by acetone (5 × 30 ml) and dried under reduced pressure. 8
The surface of the metal oxide nanoparticle is modified to decrease or inhibit the agglomeration throughout the course of the in situ polymerization process. Introduction of the unsaturated double bonds on metal oxide nanoparticles facilitated by the reaction of carboxylic acid group of oleic acid with hydroxyl groups of metal oxide nanoparticles (Figure 1). 14 Surface modified metal oxides are mainly organophilic due to oleic acid. The coordination of oleic acid to the surface of nanoparticles exhibits good dispersion in the organic phase with a tendency towards the interface.

Surface functionalization of nanoparticles by using oleic acid. M = TiO2Nps and ZrO2NPs.
Preparation of poly(PyMMA-co-BMA) metal oxide nanocomposites
A random copolymer namely poly(PyMMA-co-BMA) and its nanocomposites (TiO2 and ZrO2) (2.0 wt%) were prepared by in situ solution polymerization as reported earlier.8,9 The nanocomposites were prepared by the addition of 10 mmol of (pyridine-4-yl)methyl methacrylate and n-butyl methacrylate with 2.0 wt% of NPs (TiO2 and ZrO2) (Figure 2) in THF solvent and ultrasonicated for about 30 minutes followed by polymerization using AIBN as radical initiator under N2 atmosphere at 60°C. After the predetermined time, the obtained mixture was allowed to cool and hexane was added, a white coloured amorphous powder (10−20 µm particle size) was obtained. The resultant material was further reprecipitated using chloroform to get the pure product.
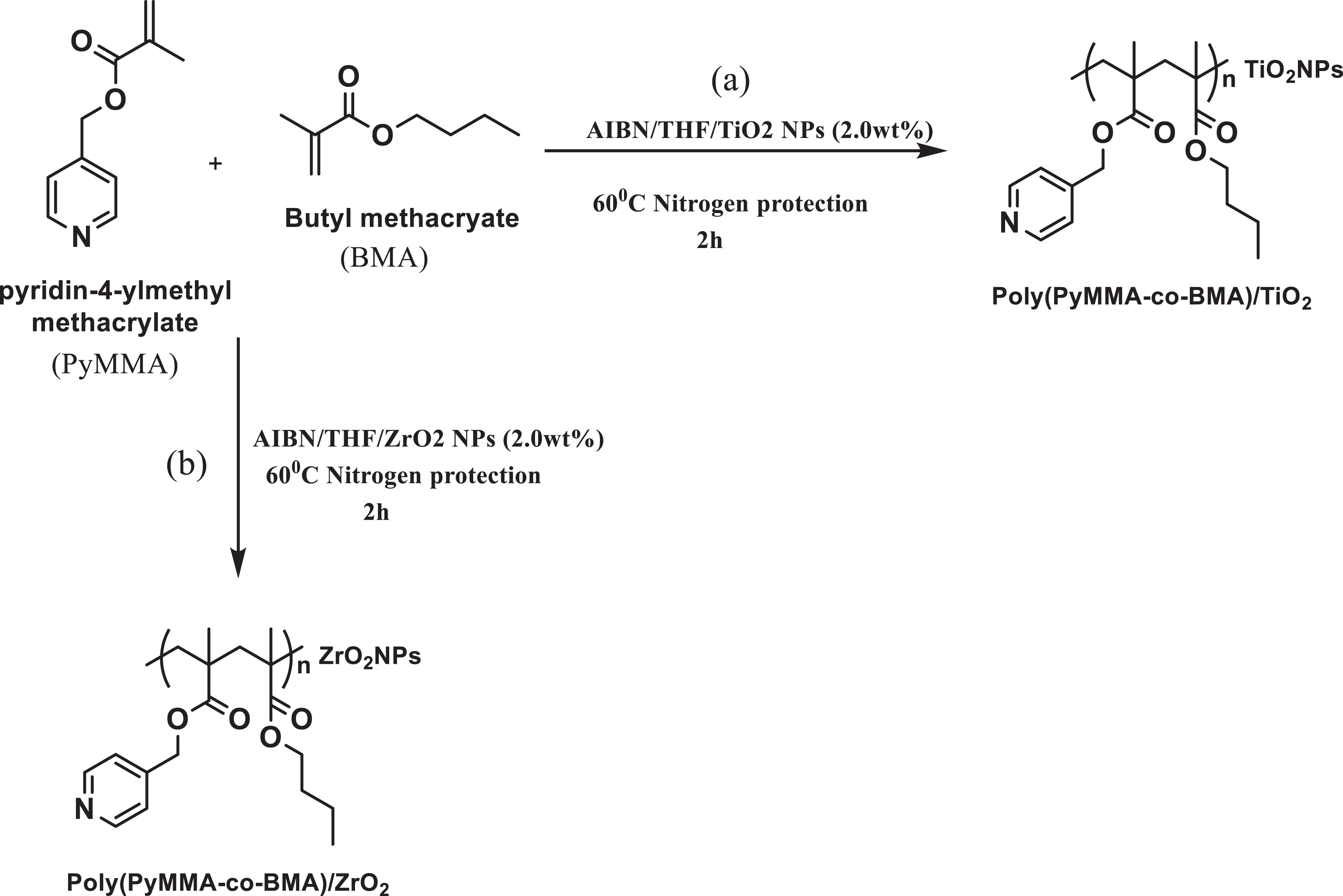
Schematic representation of synthesis of (a) poly(PyMMA-co-BMA)/TiO2 and (b) poly(PyMMMA-co-BMA)/ZrO2.
Characterization
The poly(PyMMA-co-BMA), poly(PyMMA-co-BMA)/TiO2 and poly(PyMMA-co-BMA)/ZrO2 were characterized by XRD (Bruker-D8 Advanced X-ray diffractometer with Cu-Kα (1.5418 Å). TEM (FEI-TECNAI G2-20 TWIN 200 kV) micrographs of poly(PyMMA-co-BMA) metal oxide nanocomposites were taken to study the dispersion of nanoparticles in polymer matrix. The Fourier transform infrared (FTIR) spectra were recorded in the range of 450 to 4000 cm−1 using a Shimadzu IR Affinity-1S Spectrometer. For all the measurements, the samples were grounded with KBr and made into a pellet. FESEM images were taken using (Carl Zeiss Supra 55 VP operating at 20 KeV) to examine the surface features of polymer and its metal oxide nanocomposites coating on metal surface. TEM images of PPyMMA/ZnO were taken using FEI-TECNAI G2-20 TWIN 200 kV instrument, using an acceleration voltage of 80 kV. Diluted suspensions (concentration 0.1 mg (mL)) of pristine and grafted nanoparticles in Ethanol or THF were applied onto ultrathin carbon-coated copper grids (Ted Pella, Inc.) and examined in, the microscope after drying. Diffuse reflectance UV/VIS spectra were used to evaluate the photophysical properties of coatings in the diffuse reflectance mode (R) and transformed into absorption spectra through the Kubelka–Munk function. The UV-visible absorption and transmittance spectra were recorded (DRS) on JASCO–V 670 spectrophotometer in the wavelength range 200−700 nm. The thickness of poly(PyMMA-co-BMA) and its nanocomposites (TiO2 and ZrO2) coatings were measured by digital coating thickness gauge Elcometer model 456. Averages of three consecutive readings were taken into consideration.
Corrosion studies
The electrochemical measurements were carried out using the Electrochemical Workstation (Model No: 604D CH Instruments) at a constant temperature of 28 ± 1°C with an electrolyte (3.5% (w/v) NaCl solution as corrosive media). A three-electrode cell assembly comprising of graphite as counter electrode, a saturated calomel electrode as reference electrode, a mild steel specimen as working electrode. The tip of the reference electrode is placed very close to the surface of the working electrode to minimize the ohmic potential drop. The remaining uncompensated resistance is also reduced by the electrochemical workstation. The Tafel plots were scanned at the scan rate of 0.01 mVsec−1. The electrochemical impedance studies were performed using the same setup as that of Potentiodynamic Polarization studies and the applied ac perturbation signal of about 5 mV within the frequency range of 100 kHz to 0.001 Hz was applied to the coated mild steel at open circuit potential. CHI 604D fits the selected electric circuit to the experimental impedance spectrum data and displays the value of the parameter of each electrical circuit element in the best-fitting condition. 15
Results and discussion
Prerequisite assessment of poly(PyMMA-co-BMA) metal oxide nanocomposite coatings
FTIR spectra of the TiO2NP as synthesized and surface treated samples were taken as shown in Figure 3. The peaks corresponding to C=O stretching (Figure 3(b)) of oleic acid moiety occurred at 1719.65 cm−1 which is absent in the spectrum (Figure 3(a)) of unmodified TiO2 microcrystal, indicating that the chemical reaction took place between oleic acid and TiO2 microcrystal. According to the FTIR analysis, the band corresponding to the Ti-O-Ti bond generally appeared at ∼800 cm−1–500 cm−1. Similarly, the observed FTIR peak in the region of 498–502 cm−1 for the synthesized ZrO2NP is attributed to the vibration modes of ZrO2 groups, which confirm the formation of ZrO2NP structure. Figure 4 shows the TEM images of pristine TiO2 and ZrO2 nanoparticles. It was found that synthesized nanoparticles are in granular shape with diameter in the range of 50−100 nm.

FTIR spectra of (a) TiO2, (b) functionalized TiO2 (OA-TiO2), (c) ZrO2 and (d) functionalized ZrO2 (OA-ZrO2).

TEM micrographs of synthesized (a) TiO2NPs and (b) ZrO2NPs.
The Figure 5 is the X-ray diffractogram of poly(PyMMA-co-BMA) metal oxide nanocomposites and their respective nanoparticles. The Figure 5(a) and (b) show the XRD pattern of poly(PyMMA-co-BMA)/TiO2 and TiO2NPs respectively. It was observed that the XRD pattern of poly(PyMMA-co-BMA)/TiO2 include characteristic peaks ((2θ = 25.28 (101), 37.94 (004), 47.82 (200), 54.39 (105) and 62.45 (204) and the diffraction information were in good agreement with JCPDS No # 21-1272) of crystalline anatase TiO2NPs structure. 11

XRD patterns of (a) Poly(PyMMA-co-BMA)/TiO2, (b) TiO2NPs, (c) poly(PyMMA-co-BMA)/ZrO2 and (d) ZrO2NPs.
The XRD patterns (Figure 5(c) and (d)) of poly(PyMMA-co-BMA)/ZrO2 (2.0 wt%) and ZrO2NPs show the coexistence of broad amorphous peak of the poly(PyMMA-co-BMA) and suppressed crystalline peaks of monoclinic ZrO2NPs. 16 The XRD studies confirm that the integration of nanoparticles in the polymer host does not influence any change in crystallinity, hence it is revealed that the distribution of nanoparticles in the polymer is homogenous.
It is well known that the effectiveness in anticorrosive properties of hybrid material relies on nanoparticle’s morphology, 9 the TEM micrographs of poly(PyMMA-co-BMA) metal oxide nanocomposites were taken (Figure 6). It can be seen in Figure 6(a) and (b) that the TiO2NPs and ZrO2NPs were uniformly dispersed in the poly(PyMMA-co-BMA) matrix and they have spherical granular structure with diameter of about 10−20 nm. The morphological studies revealed that the size of TiO2NPs and ZrO2NPs were comparatively equivalent to ZnONRs as reported earlier. 9

TEM micrographs of (a) poly(PyMMA-co-BMA)/TiO2 and (b) poly(PyMMA-co-BMA)/ZrO2.
The quality of coating reflects on the size of the nanoparticle and homogeneous distribution of nanoparticles in the polymer matrix, which lead to improve the anticorrosive behaviour by reducing the porosity and tortuousness in the diffusion pathways. 6 Thus, it is clear that the prepared hybrid materials may be used as an efficient anticorrosive materials.
Anticorrosive evaluation of poly(PyMMA-co-BMA) and its metal oxide nanocomposites coatings
Potentiodynamic polarization studies
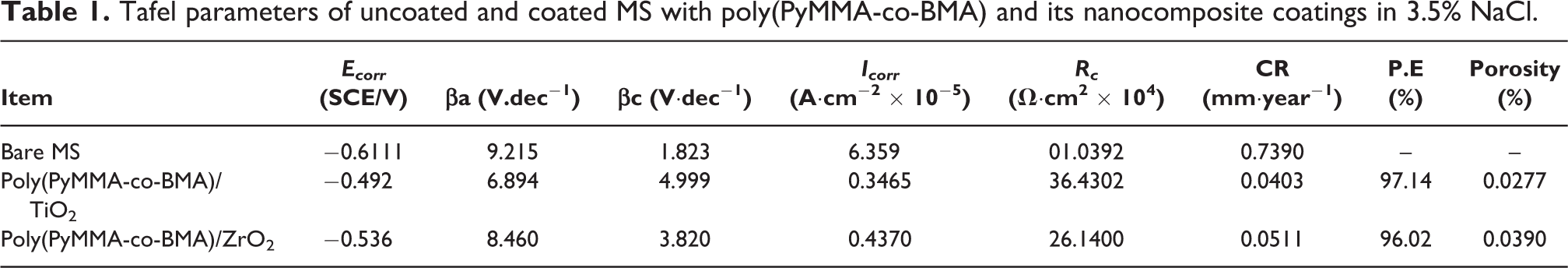
Figure 7 shows the Tafel plots of bare, poly(PyMMA-co-BMA) and its metal oxide nancomposites coated MS. Tafel parameters obtained from these plots have been listed in Table 1. The more positive Ecorr value indicate a slower corrosion rate. 17 It is known from the Table 1 that the Ecorr of nanocomposites coated specimens improved positively due to the integration of nanoparticles, which suppressed the anodic reactions and consequently inhibited the corrosion. 18 The well dispersed nanoparticles in poly(PyMMA-co-BMA) matrix reduces the small pores (Table 1) and provides a tortuousness, which block/delay the electrolyte access to the steel surface. 10 The TiO2 based nanocomposites offer the best blocking temperament due to its effective nuclear charge that promotes strong interlayer adhesion and a uniform deposition with the polymer matrix leads to network formation. 19

Tafel plots of uncoated and coated MS with poly(PyMMA-co-BMA) and its nanocomposite.
Tafel parameters of uncoated and coated MS with poly(PyMMA-co-BMA) and its nanocomposite coatings in 3.5% NaCl.
Electrochemical impedance spectroscopic analysis
Nyquist plots
Figure 8 shows the Nyquist plots of poly(PyMMA-co-BMA) and its metal nanocomposites coatings. Our previous report reveals that the chemical interaction between ZnONRs and functional groups on the polymer matrix effectively improved the barrier properties and ionic resistance of the hybrid coating. 10 But the slow deterioration of coatings occurs, which was confirmed by appearance of two capacitive loops in Nyquist plots20,21 and depressed semicircles in the impedance plot.22,23

Nyquist plots of uncoated and coated MS with poly(PyMMA-co-BMA) and its nanocomposite.
For the poly(PyMMA-co-BMA)/TiO2 coated MS, equivalent circuit (Figure 9(a)) includes the component of oxidation film and polymer nanocomposite film. The circuit comprises of charge transfer resistance (Rct), coating resistance (Rc), coating capacitance (Cc), solution resistance (Rs), and double layer capacitance (Cdl). The Q represents constant phase element (CPE) used to modify the double layer capacitance for obtaining more accurate fitting results and the Warburg impedance (W) is added to an equivalent circuit because of the appearance of a linear region at lower frequencies. The Nyquist plot of poly(PyMMA-co-BMA)/TiO2 (2.0 wt%) coating has greater Rct, W and lower Q values compared to other metal oxide nanocomposites.
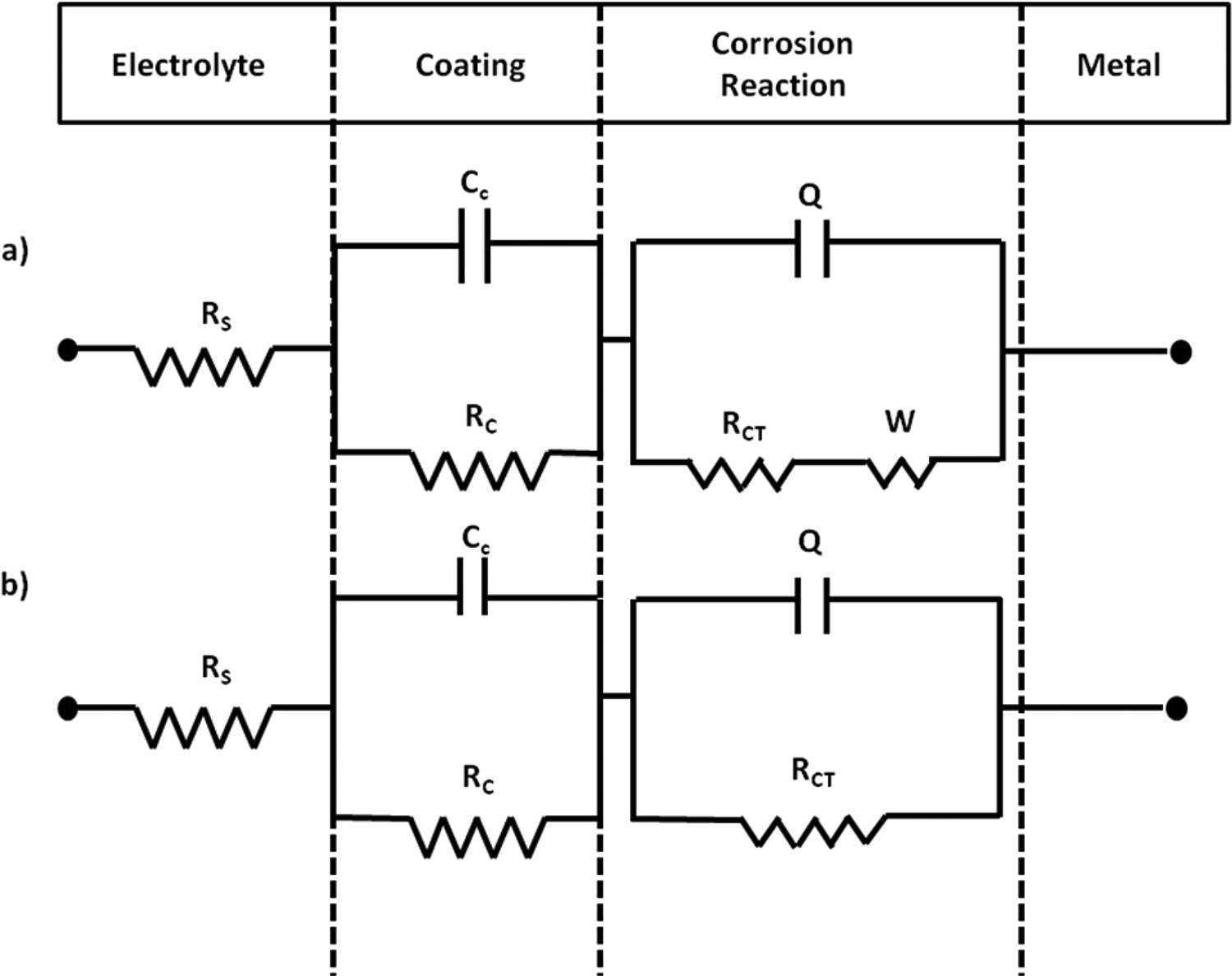
Equivalent circuit model used in modeling for a) poly(PyMMA-co-BMA)/TiO2 coated and b) poly(PyMMA-co-BMA) coated mild steel.
From the Table 2, it is found that the poly(PyMMA-co-BMA)/TiO2 coating possesses higher Rct compared to other nanocomposites coatings and hence it has slower corrosion rate. 24 The linear region in the plot is related to the diffusion resistance against corrosive species through the porous coating and oxide layer by passive layer formation. 25 It is known that the proportion of the Warburg region in the Nyquist plot is the restrictive factor for ion diffusion/transport from electrolyte to the electrode surface. 26 Therefore, the higher value of W restricts the access of corrosive ions to the MS surface and indicates greater difference in ion diffusion path lengths and increased barrier of ion movement to the MS. The Nyquist plot (Table 2) of poly(PyMMA-co-BMA)/ZrO2 (2.0 wt%) coating possess greater Rc compared to other metal oxide nanocomposites coating. The higher Rc reveals that the surface blocking tendency of poly(PyMMA-co-BMA)/ZrO2 (2.0 wt%) coating is higher compare to other metal oxide nanocomposites.
EIS parameters of uncoated and coated MS with poly(PyMMA-co-BMA) and its nanocomposite coatings in 3.5% NaCl.

Bode plots
Bode and Phase angle plots of bare MS, poly(PyMMA-co-BMA) and its nanocomposite coated MS were presented in Figure 10(a) and (b) respectively. The bode plots of coated and uncoated MS revealed that the coated specimens exhibited higher impedance values at low frequencies and higher impedance values were attained by incorporating metal oxide nanoparticles. Phase angle (theta) at higher frequencies has been considered as a helpful parameter to evaluate the protective performances of coatings. 27 If the coating shows high resistance, the current prefers to pass through dielectric pathways and higher phase angles (near 90°) between the current and the voltage result. In low resistant coating, current prefers to pass through conductive pathways and the result is lower phase angles (near 0°). 24
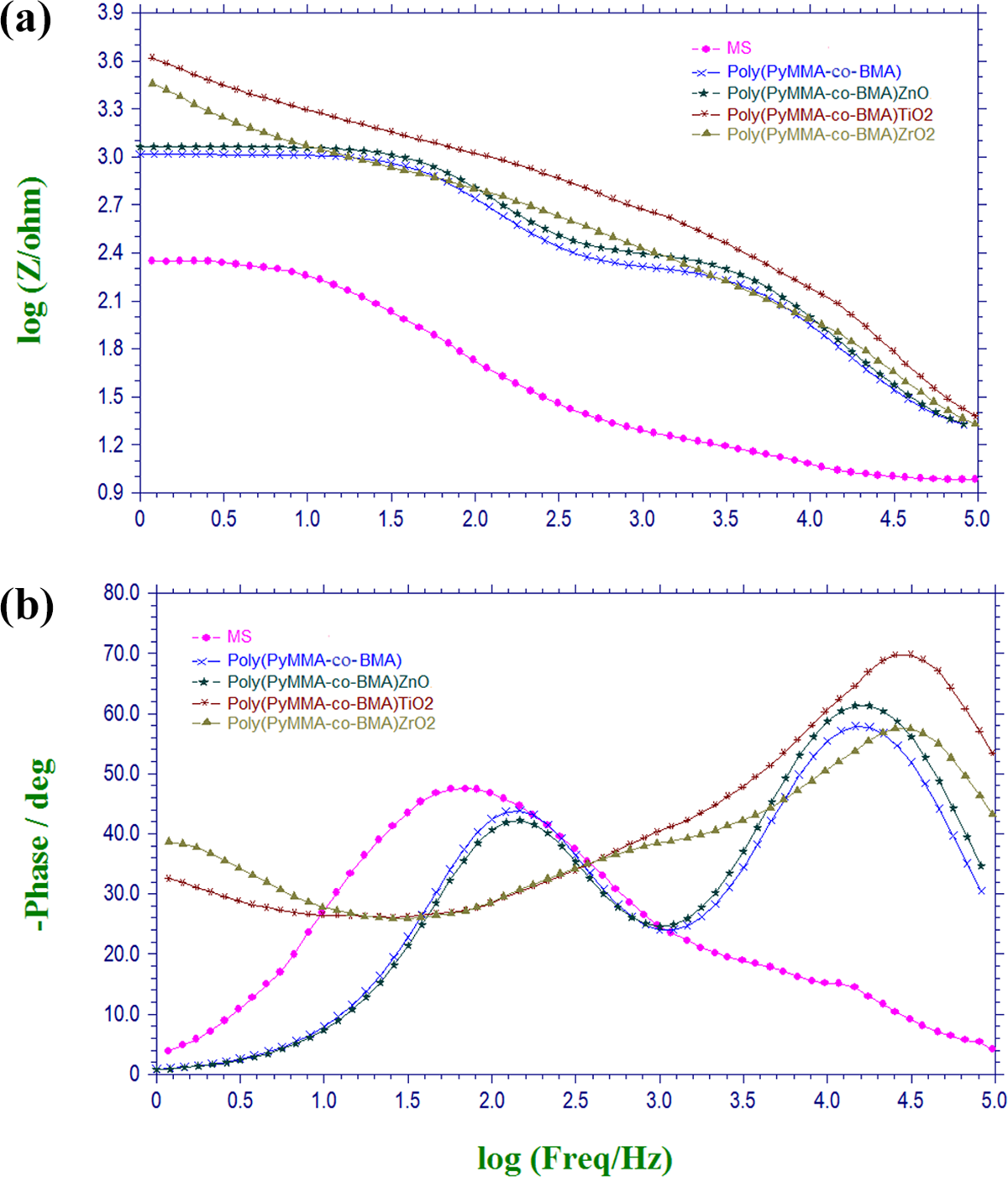
a) Bode plots of uncoated and coated MS with poly(PyMMA-co-BMA) and its nanocomposite. b) Phase angle Plots of uncoated and coated MS with poly(PyMMA-co-BMA) and its nanocomposite.
It is reported that, an increase in phase angle value from 57.8° to 60.9° at high frequency region (10 kHz) was achieved by incorporating 2.0 wt% of ZnO nanoparticle in the polymer. This concludes that the coatings demonstrated capacitive behaviour at high frequency but conductive behaviour at low frequency respectively. 10 The phase angle of poly(PyMMA-co-BMA)/TiO2 (2.0 wt%) coatings have a single band with large scale (Figure 10(b)) (10 KHz) at 69°, which is higher than that of reported. 10 This suggest that TiO2 nanocomposites coating do not show conductive behaviour and it is considered to form a barrier layer with high resistance and low capacitance. The enhanced corrosion resistant behaviour of TiO2 nanocomposite is due to the effective nuclear charge of TiO2 that promotes strong interlayer adhesion and a uniform deposition process. 20 Thus, network reinforcement might be a significant factor that contributes to the enhanced total resistance.
The poly(PyMMA-co-BMA)/ZrO2 (2.0 wt%) coating (Figure 10(b)) shows the phase angle at 58° (10 kHz), which is very close to that of polymer and ZnO nanocomposite but exhibit at higher frequency suggesting that the poly(PyMMA-co-BMA)/ZrO2 coatings show capacitive behaviour.
Surface examination of poly(PyMMA-co-BMA) and its metal oxide nanocomposites coatings
Thickness of coatings
The poly(PyMMA-co-BMA) and its nanocomposite coatings thickness were measured using elcometer. The mean of three successive readings were taken into consideration and the coating thicknesses were found to be around 0.8–1.3 μm.
Surface morphology of coatings
The surface morphology of poly(PyMMA-co-BMA) and its nanocomposites coated MS before and after performing the Tafel experiment is shown in Figure 11. As it was observed earlier 10 neither agglomeration nor two phase system in the TiO2 and ZrO2 nanocomposite coatings were found in Figure 11(a) and (c). No pinhole, pits or any heterogeneity on the surface of nanocomposite coatings were set up as well. This signifies that uniform and homogenous coatings without any curing defects were produced. Figure 11(b) and (d) showed the surface morphology of poly(PyMMA-co-BMA) and its nanocomposite (TiO2 and ZrO2) coatings after performing Tafel in 3.5% (w/v) NaCl medium, which reveals a slight crazing and deposition of NaCl crystals on the surface of coated specimens as found previously. 10

FESEM micrographs of coatings on MS. (a) poly(PyMMA-co-BMA)/TiO2 (before Tafel studies), (b) poly(PyMMA-co-BMA)/TiO2 (after Tafel studies), (c) poly(PyMMA-co-BMA)/ZrO2 (before Tafel studies) and (d) poly(PyMMA-co-BMA)/ZrO2 (after Tafel studies).
UV stability of coatings
The UV-visible spectroscopy provide information about the UV blocking characteristics of poly(PyMMA-co-BMA) and its nanocomposite (TiO2 and ZrO2) coatings. The UV-visible absorption spectra for poly(PyMMA-co-BMA) before and after coating on MS, demonstrates that there were no significant shift. But it is reported that there is an increment in the absorption maxima of poly(PyMMA-co-BMA) coated MS at 252 nm validate the complex formation between the MS and functional polymer.10,28,29
The similar behaviour was observed (Figure 12(a)) in the case of poly(PyMMA-co-BMA)/TiO2 coating, where there is a change in the position of absorption maximum of poly(PyMMA-co-BMA)/TiO2 coated MS from 328 nm to 314 nm indicates the formation of a complex between nanocomposite and MS. This blue shift provides a strong evidence for the enhanced interaction between poly(PyMMA-co-BMA)/TiO2 on MS surface. Moreover, the small surface area per unit mass and volume increases the effectiveness of the UV blocking radiation. 20

UV-visible absorption spectra of (a) poly(PyMMA-co-BMA)/TiO2 and (b) poly(PyMMA-co-BMA)/ZrO2 coatings on MS.
Generally, the monoclinic crystalline structures of pristine ZrO2NPs have a sharp and intense band at 212 nm with an absorption edge around 300 nm. But, it was overlapped with the Π→Π* absorption bands of poly(PyMMA-co-BMA) matrix. 30 Further the change in the (Figure 12(b)) absorption intensity and the position of absorption maximum of poly(PyMMA-co-BMA)/ZrO2 coated MS from 264 nm to 255 nm indicate the formation of a complex between nanocomposite and MS.
This study explains the facts that (i) the establishment of complexation between poly(PyMMA-co-BMA) and nanocomposite (TiO2 and ZrO2) coatings with MS and (ii) the improvement of UV absorption characteristic by poly(PyMMA-co-BMA) and its nanocomposite (TiO2 and ZrO2) coatings on MS.
The comparative studies of UV absorption spectra shows that the change in the position of absorption maxima of poly(PyMMA-co-BMA)/TiO2 coating is lesser than that of other nanocomposite coatings. This phenomenon can be explained by high complex forming ability of TiO2NPs with the polymeric ligands. 20 The equal contribution of polymeric ligands towards MS and TiO2NPs for complexation leads to the formation of close packed and adhesive nanocomposite coating. Thus, all the nanocomposite coating provides the good shielding property against UV radiation and inherently improves the durability of the system by absorbing UV radiation that can protect the polymer coating from photo degradation.
Interfacial mechanism
The pyridine moiety and carbonyl groups of the copolymer forms effective binding with metal. The barrier formation is possible mechanism behind the enhanced corrosion resistance behaviours of the polymer and its metal oxide (TiO2 and ZrO2) nanocomposite film coated MS. The coatings restrict the reach of corrosive ions like OH−, Cl− and H2O species at metal/coating interfaces. The EIS results also confirmed that the presence of nanopartcles in poly(PyMMA-co-BMA) coating played a major role at metal/coating interface, they get dispersed within the polymeric matrix lead to improve the quality of coating by lowering the porosity and zigzagging the diffusion pathways.31,32
However, in the case of TiO2 based nanocomposite, the strengthening of the coating tackiness leads. The equivalent circuit model for poly(PyMMA-co-BMA)/TiO2 coated MS is fitted with the circuit shown in Figure 9(a), which is due to uniform film and the diffusion of corrosive species is restricted due to the passive oxide layer formation. It can be attributed to the TiO2 of poly(PyMMA-co-BMA)/TiO2 coating, which attracts cathode-generated OH− of the electrolyte and reacts according to the reaction given below.

The as-formed TiO32−ions react with the iron ions at the adjacent anodes, thereby forming a passive film. The anodic inhibitions confirmed by the greater shift of the Ecorr towards the positive direction compared to composite coating of the other fillers (Figure 7). The passive film prevents the movement of the corrosive medium, such as Cl−, H2O, and O2, to the metal surface. Simultaneously, this film restrains the mass transfer between the anode and the cathode, thereby greatly weakening the transport of electrons from anode to cathode and controlled the electrochemical reaction. 32 The poly(PyMMA-co-BMA)/ZrO2 coated film is uniform and protective as it is explained in the equivalent circuit shown in Figure 9(b).
The surface examination of films were characterized with UV-visible spectroscopy and scanning electron microscope to confirm the formation of poly(PyMMA-co-BMA) and its different nanocomposites film on MS. The UV blocking studies were also carried out using UV-visible spectroscopy. The electrochemical and optical studies reveals that the poly(PyMMA-co-BMA)/TiO2 film on MS in 3.5% (w/v) NaCl provides better protection against corrosion than ZrO2 based nanocomposites hybrid film.
Conclusion
The prepared nanocomposite materials were characterized by XRD and TEM analysis and they are found to be homogenous, which is an important parameter for an organic film to perform its action effectively. The nanocomposites were coated as film on the MS by spin coating method. The anticorrosive performance of hybrid films was carried out using Potentiodynamic Polarization and Electrochemical Impedance Spectroscopy (EIS) studies. The electrochemical studies display that the incorporation of nanoparticles (TiO2 and ZrO2) in the poly(PyMMA-co-BMA) film exhibited a good anticorrosive effect. TiO2 nanocomposite coating has proved to be an effective way to fabricate a good anticorrosive coating in aggressive conditions by forming a passive layer at coating metal interface. The surface examination of coatings were characterized with scanning electron microscope to confirm the formation of poly(PyMMA-co-BMA) and its different nanocomposite films on MS, which showed that the coatings were found to be uniform and good adherent. The results obtained from UV-visible studies provided the information about the UV stability of the organically bound nanocomposites coatings on MS upon exposure to the UV radiation, that in turn protect the underlying mild steel from corrosion and UV degradation (i.e weathering). Thus, the comparative studies of anticorrosive behaviour of poly(PyMMA-co-BMA) TiO2 and ZrO2 nanocomposites films endow different mechanism of corrosion protection, which provides new prospects to material scientist and technologist to develop an UV blocking anticorrosive coating materials for diverse aggressive environments. Hence, stability of organic coating against the chemical and photochemical attack enhances the its durability of it and protects the underlying metal substrate from corrosion for a long time.
Footnotes
Acknowledgement
I Pugazhenthi would like to thank the Management, Annamacharya Institute of Technology and Sciences (Autonomous), Rajampet, Andhra Pradesh, India. The authors would like to thank the Defense Research and Development Organization (DRDO) INDIA for their financial support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Defense Research and Development Organization (DRDO) INDIA (Ref. No. ERIP/ER/1204672/M/01/1525).
