Abstract
This paper is a follow up to previous work from these laboratories, which led to a method for simultaneous reactive dye fixation and DMDHEU finishing of cotton at significantly higher shade depths (
Introduction
A method for simultaneous dyeing and durable press (DP) finishing of cotton using reactive dyes and DMDHEU, in place of alkali, was reported in Part 1 of this series.
1
Using a pad-dry-steam-cure process, dyes such as C.I. Reactive Red 120 were applied to cotton at a high shade depth
In this article, the function of steaming in the one-step dyeing and DP finishing process is discussed. To help understand the basis for our enhanced dye fixation levels on cotton, the nature of the chemistry between dyes and DMDHEU on a glass surface, and the analysis of solvent extracts from dyed fabrics and their powdered samples were undertaken. Using the results of these experiments, the mechanism of dye fixation on cotton fabric via the one-step dyeing and DP finishing process was established.
Experimental
Materials, Processes, Formulations, and Tests
Mercerized and bleached woven cotton fabric (153 g/m2) were used in this study along with three reactive dyes: C.I. Reactive Red 177 (Avecia Inkjet Ltd.), C.I. Reactive Red 120 (Clariant International Ltd.), and CI. Reactive Blue 19 (DyStar Colours Deutschland GmbH) and were used as received. Dye selection was based on parent structure, reactive group, commercial popularity, and availability. Dyes, DP finish agent (DMDHEU, 44% aqueous solution), and DP catalyst (MgCl2·6H2O) were commercial products from NOVEON Inc. and Sigma Aldrich Chemical Co., respectively, and were used as received. The dyebaths contained 1-2% dyes based on weight of bath (owb) with various DMDHEU and catalyst levels. Details for the reactive dyeing, and one-step dyeing and DP finishing processes (direct cure and steam cure), as well as the test method for shade depth of dyed fabrics were reported in the previous paper. 1
Analysis of Dye and DMDHEU
Chemical Reaction
A dyebath aliquot (0.5 mL) containing 1% dye, 6% DMDHEU, and 1.8% MgCl2 was dropped on a glass slide. The slide was dried in an oven at 100 °C for 5 min. A pair of slides was prepared for each of the three dyes used. After drying, one of the pair slides was cured at 160 °C for 3 min. Each pair of slides was separately washed in 50 mL boiling water for 10 min. After washing, all components on the dried only slide dissolved in water, while an insoluble DMDHEU film remained on the cured slide. The dye concentration in the wash water was measured at the λmax of the corresponding dye using a BYK Gardner spectrophotometer. The level of dye unfixed on DMDHEU was calculated from the dye concentration in the wash bath from the cured slide versus the concentration of dye washed from the dried only slide.
Extraction of Dyed Fabrics and Fabric
Powder Samples
Dyed fabrics and the corresponding powdered samples (0.5 g each) were extracted with 50% dimethylformamide (DMF)/water solution (50 mL) at 100 °C for 10 min. The powdered samples were made by grinding dyed fabrics using a fabric mill. The dye concentration in the extracted baths was measured using the spectrophotometer.
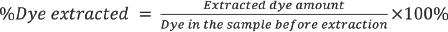
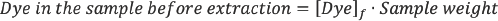
[Dye]
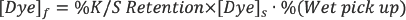
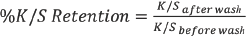
Results and Discussion
Various Processes and DMDHEU Level Effects on Dye Fixation
When a cotton fabric was dyed through the one-step dyeing and DP finishing process, its shade depth and
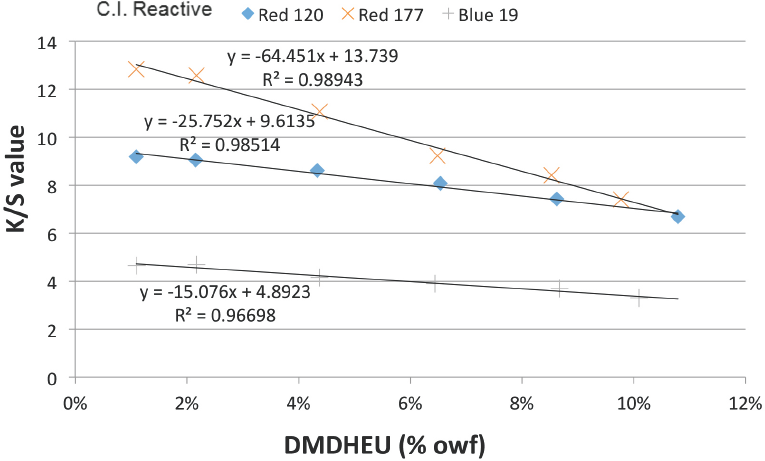
The absolute slope value (|Slope|) indicated the impact of DMDHEU concentration on the shade depth of dried fabrics. The highest |Slope| among the three dyes was obtained using Red 177, which indicated that the dye uptake during padding decreased the most as DMDHEU concentration increased. Variations in |Slope| may be caused by dye structure (size and shape). The molecular structures and models of the three dyes are shown in Fig. 2. Red 120 and Blue 19 had non-planar structures, while Red 177 was essentially planar, which is important for forming aggregates. At high DMDHEU concentrations, the level of free water molecules in the dyebath was reduced, increasing interactions among dye molecules. Hence, molecules of Red 177 may gradually form aggregates at increased DMDHEU concentrations. Since the larger the dye particles are in the dyebath, the slower the dye diffuses from dyebath to fiber, it was expected that the shade depths of Red 177 dyed fabrics would be significantly affected by increased DMDHEU concentrations in the dyebath.
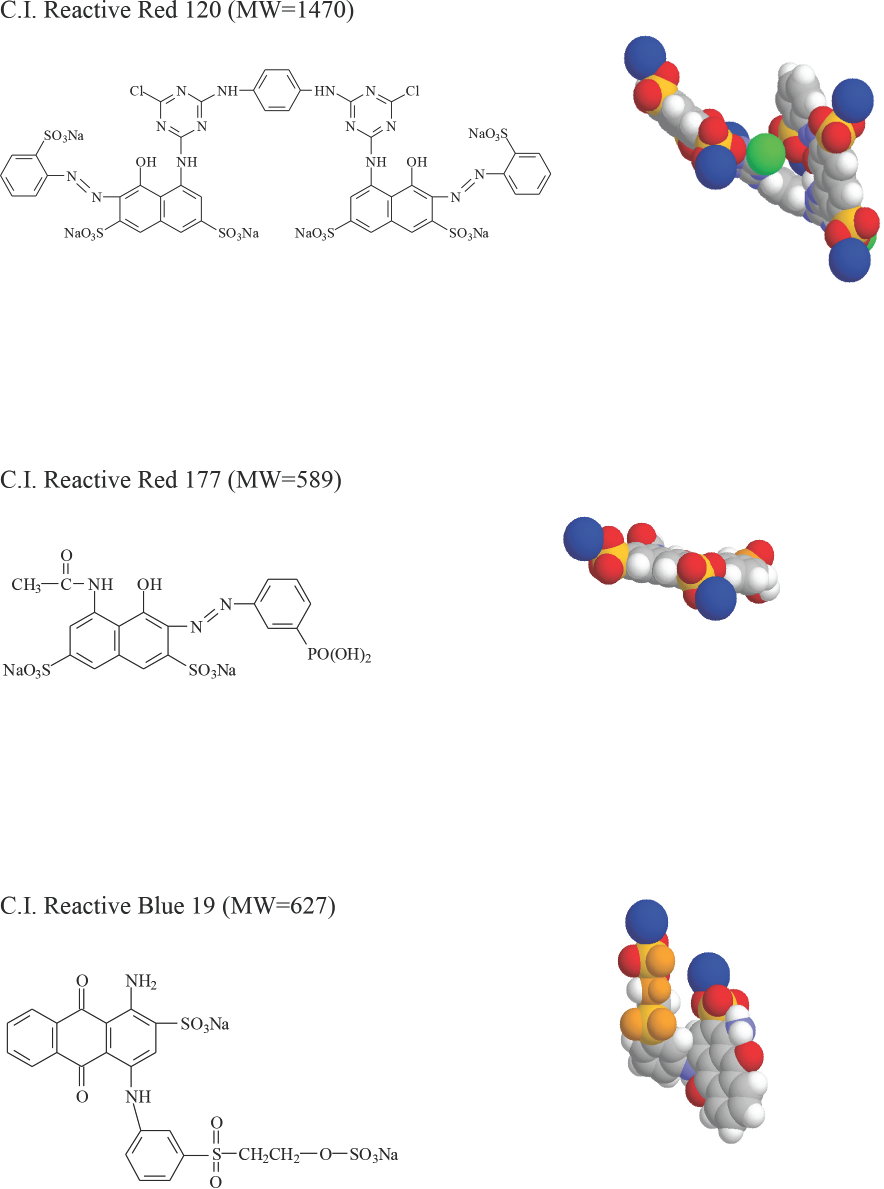
Molecular structures of Reactive Red 120, Red 177, and Blue 19 and their models at the lowest energy level, generated by ChemWindow.
Fig. 3 shows
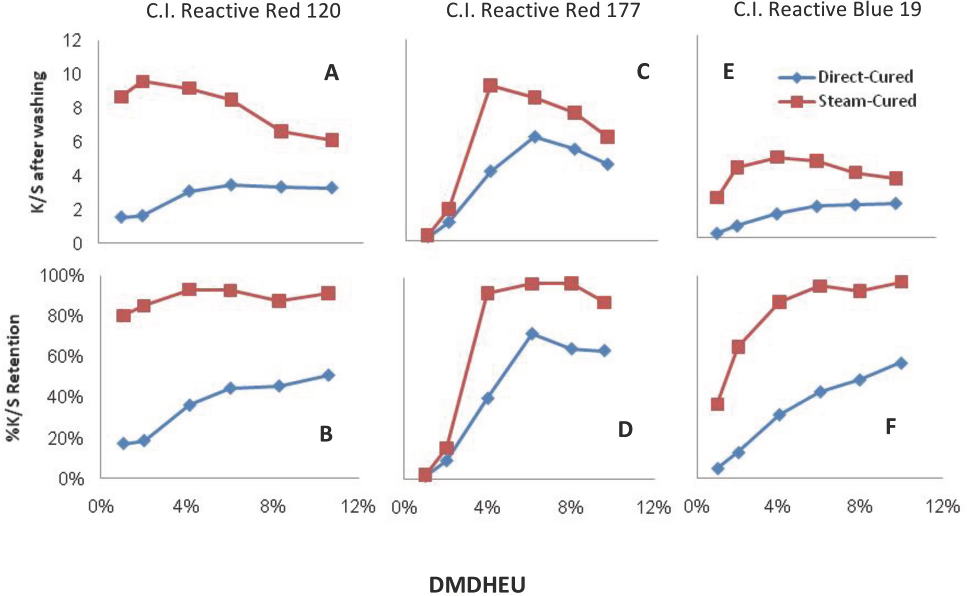
Similarly, the peak levels in the steam cure process were 4% DMDHEU and changed very slightly afterwards (Figs. 3C and D). For Blue 19 dyed and finished fabrics,
Based on all of the curves in Fig. 3,
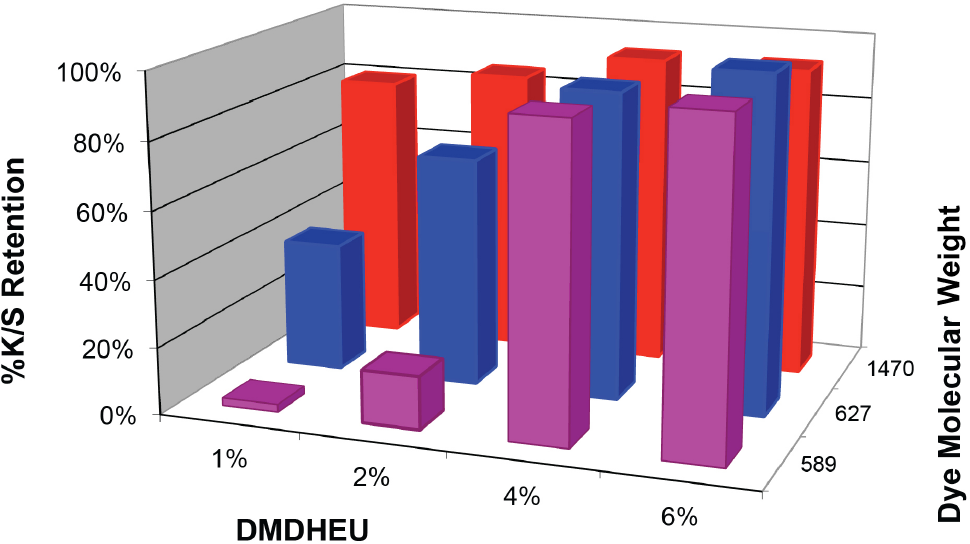
Effects of DMDHEU concentrations and dye molecular weight
Curing Dyes and DMDHEU in the Absence of Fabric
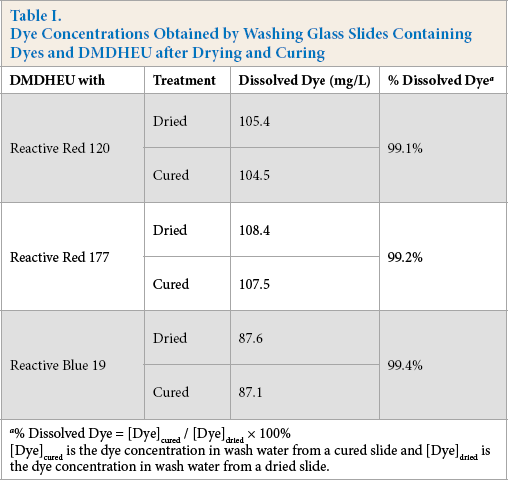
The relationship between dyes and DMDHEU was examined by curing dyebath contents on glass slides under the same fabric curing condition. The dyebath contained 6% DMDHEU and 1% dye. The molar ratios of DMDHEU to dye in the dyebath were 50:1, 20:1, and 21:1 for Red 120, Red 177, and Blue 19, respectively. It was anticipated that if dye bonded to the methylol groups of the DMDHEU film on a glass slide after curing, it would not dissolve in the wash water, while the dye in the residue that was dried on glass but not cured would fully dissolve in the wash water. The ratio of the dye concentrations in the two wash waters (cured / dried) would indicate the level of covalent bonding of dye to DMDHEU. Table I lists dye concentrations in the wash waters for the three dyes and the percent dissolved dye. The higher the percent dissolved dye, the higher the level of the dye washed off the glass surface and the lower the level of chemically reacted dye and DMDHEU.
Dye Concentrations Obtained by Washing Glass Slides Containing Dyes and DMDHEU after Drying and Curing
% Dissolved Dye = [Dye]cured / [Dye]dried × 100%
[Dye]cured is the dye concentration in wash water from a cured slide and [Dye]dried is the dye concentration in wash water from a dried slide.
Results in Table I indicate that all the % dissolved dye values were higher than 99%, which suggests the dyes did not covalently bond to DMDHEU during curing. Fig. 5 is an example of a half washed glass slide obtained from 1% Red 177 and 6% DMDHEU + 2% MgCl2 solution after drying and curing. It can be seen from the lower half of the slide that dye washed away leaving behind a film of crosslinked DMDHEU.
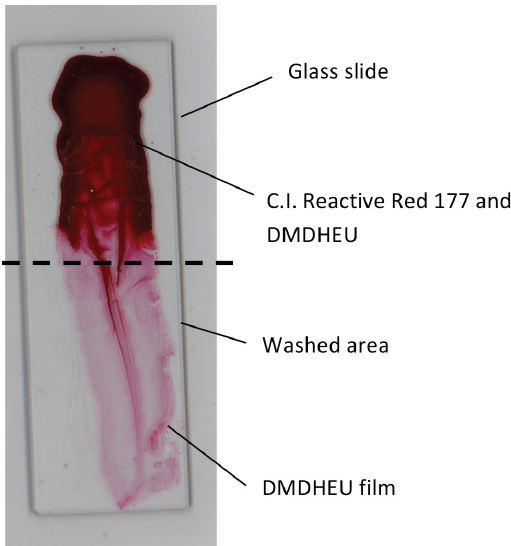
A partially washed glass slide containing Reactive Red 177 + DMDHEU after drying and curing.
DMF Extraction of Dyed Fabric and Fabric Powders
To help characterize the mechanism of dye fixation by DMDHEU, dyed fabrics and corresponding powdered samples were extracted with DMF/water (50/50) at 100 °C for 10 min. Fig. 6 shows the percent extraction values for Red 120 from dyed cotton fabrics after the AATCC Test Method (TM) 61 (2A) wash test. Fabrics were dyed with 2% Red 120 through either one-step steam cure process by DMDHEU fixation or by conventional pad dyeing using alkali fixation. As Red 120 contains monochlorotriazine groups that can chemically bond to cotton, the percent extracted dye from the alkali treated fabric and the corresponding powder were similar, 7.4% and 7.6%, respectively. However, 6% DMDHEU treated fabrics gave different levels of percent extracted dye from fabric and powder samples, 3.7% and 12.2%, respectively. Since the level of percent extracted dye from the powder sample was much higher than that from a fabric sample treated using the one-step process, fixation of Red 120 on the fabric by DMDHEU was not mainly due to chemical bonding.
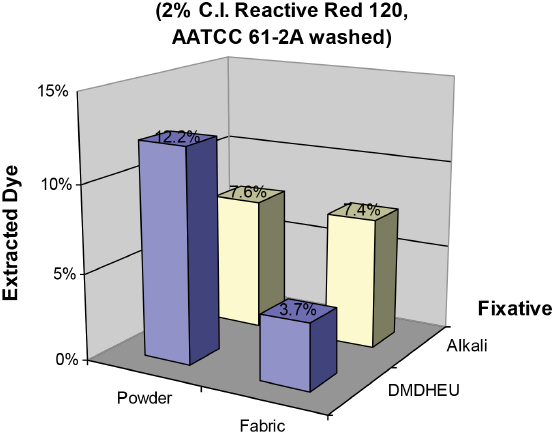
DMF/H2O extraction levels from 2% Reactive Red 120 dyed fabrics fixed by DMDHEU in the one-step steam cure process and by alkali in conventional pad dyeing.
Fig. 7 displays results from DMF/water (50/50) extracted Blue 19 dyed fabrics fixed by DMDHEU using a one-step steam cure process and by alkali using conventional pad dyeing. The percent extracted dye level on DMDHEU fixed fabric and on the corresponding powdered sample were 2.8% and 12.0%, respectively, while the percent extracted dye levels on alkali fixed fabric and on the corresponding powder were 2.8% and 4.5%, respectively. These results were consistent with those from Red 120 dyed samples, and indicated that the principal application of Blue 19 was not via chemical bonding to cotton or DMDHEU during one-step dyeing and DMDHEU finishing.
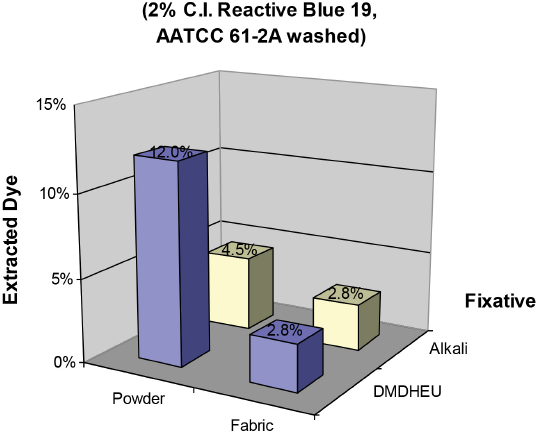
DMF/H2O extraction levels from 2% Reactive Blue 19 dyed fabrics fixed by DMDHEU in the one-step steam cure process and by alkali in conventional pad dyeing.
Figs. 6 and 7 suggest that dye was entrapped inside the fiber pores by DMDHEU crosslinking of cotton. When the fabric was powdered, various pores were opened and the dye could be extracted into DMF/water.
Table II shows the DMF/water extracted levels for Red 120 and Red 177 from fabric and from the corresponding powder produced by the direct cure and steam cure processes. The percent extracted dye values from the fabrics were found to be lower than those values obtained from the corresponding powdered samples.
Percent Extracted Dye from Fabrics and Powdered Samples
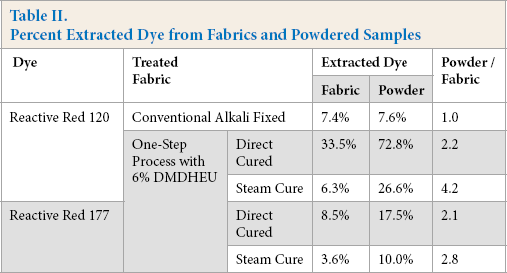
Following the one-step steam cure process, the amount of extracted Red 120 from the fabric sample was 6.3%, which indicates that most of the dye was entrapped by DMDHEU. After grinding the fabric to a powder, 26.6% of the dye in the fabric could be extracted away. For the one-step direct cure process, only a small amount of dye diffused into the fiber following the padding step, leaving most of the dye on or near the fiber surface. Subsequent DMDHEU crosslinking did not fix the dye inside fiber. Therefore, 33.5% of the dye on the fabric was extracted into DMF/water. When the fabric was ground to a powder, the extractable dye on the substrate reached 72.8%.
Similar results from DMF/water extraction of Red 177 dyed fabric and the corresponding powder can also be found in Table II. Following one-step direct cure and steam cure processes, the percent extracted levels of Red 177 from the direct-cure fabric and the corresponding powder were 8.5% and 17.5%, respectively, while the percent extracted levels of the dye from the steam cure fabric and its powder were 3.6% and 10.0%, respectively.
Although the percent extracted levels from Red 177 dyed fabrics were lower than those from Red 120 dyed fabrics, the ratios of percent extracted dye from fabrics and the corresponding powders were similar. As shown in Table II, the ratio of extracted Red 120 from powdered samples and dyed fabric after the one-step direct cure process was 2.2, while a similar ratio (2.1%) was obtained from Red 177 dyed samples. For the samples using the one-step steam cure process, the ratio of extracted Red 120 from dyed powder / fabric samples was 4.2, while the ratio from Red 177 dyed samples was 2.8.
For the conventional alkali fixed sample, reactive dye was chemically bonded to fiber so that a ratio of percent extracted dye levels on fabric and powdered forms was close to 1. For combined dyed and DMDHEU finished fabrics, the ratios were largely greater than 1, indicating that the dye did not chemically bond to fiber. Interestingly, comparing the data in Table II, dye fixation levels on fabrics using the one-step direct cure process were low, but those using the one-step steam-cure process were even greater than those using the conventional reactive dyeing process. This phenomena indicated that steaming was a key step to improve dye fixation in the one-step dyeing and DP finishing process. As steaming could efficiently promote dye diffusion from fiber surface into pores, dyes could be entrapped there by DMDHEU crosslinking inside the pores after curing.
Conclusion
Based on the results of this study, it is evident that the reactive dyes studied that contained monochlorotriazine, phosphonic acid, or sulfatoethyl sulfone groups did not bond to cotton or to the DP agent (DMDHEU) during one-step dyeing and DP finishing. Instead, dyes were fixed to the fiber by adsorption of dye and DP finish on fiber surface, followed by diffusion of dye and DP finish into the fiber pores, and finally, by DP-induced crosslinking of cellulosic chains during curing that locked the dye inside the fiber. It is also evident that dye fixation levels were greatly affected by dye size and DMDHEU concentration.
