Abstract
One-step dyeing and durable press (DP) finishing offers potential advantages such as energy savings, reduced production time, and quick response to the market. In the present research, six reactive dyes were used in combination with DMDHEU, a DP finishing agent, in one-step and conventional two-step dyeing and finishing processes. Colorfastness properties and DP ratings of fabrics treated by the one-step dyeing and finishing process were comparable to those fabrics dyed and finished in separate steps.
Introduction
Reactive dyes have many good properties, including bright colors, ease of application, excellent washfastness, and moderate cost. These features make reactive dye application a commercially viable option for cotton coloration. However, reactive dyes usually require a large amount of salt in exhaust dyeing processes, their fixation levels are often low, and it can be hard to achieve very deep shades. 1 Low fixation levels not only reduce dye efficiency, but also generate highly-colored wastewater, which can adversely impact the environment.
Durable press (DP) finishing is a conventional process for producing wrinkle-resistant cotton fabric, and uses padding, drying, and curing steps. During the curing step, the DP agent, the most common one being dimethyloldihydroxy-ethylene urea (DMDHEU), 2 crosslinks adjacent cellulose chains to enable cotton fabric to maintain a smooth appearance. Fig. 1 displays the structures for some crosslinking agents that have been used, which include various formaldehyde based and non-formaldehyde based products.
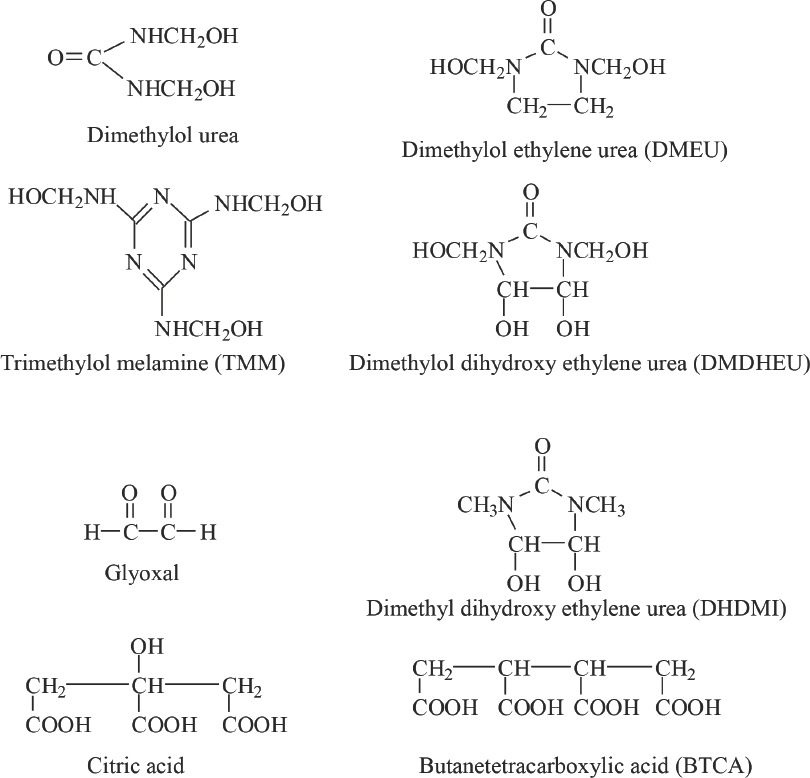
Crosslinking agents for dyeing and DP finishing.
In continuous dyeing with reactive dyes, the process involves padding, fixing, washing, and drying. The equipment used in a typical continuous dyeing process is similar to that used for DP finishing. Therefore, conducting reactive dye application and DP finishing in one step is plausible for commercial production. One-step dyeing and finishing has been of interest since the 1950s, and various crosslinking agents, reaction conditions, and application processes have been considered. Although the expected DP properties were achieved, the color strength of cotton fabric dyed through previous processes (e.g., 70%-80% wet pickup (WPU), dry for 7 min at 60 °C, cure for 3 min at 160 °C, using carbamoyl finishes and acid dye;
3
or at < 180 °C using reactive dyes,
4
or using reactive or direct dyes
5
) was typically low
A key barrier to simultaneous processing of cotton with reactive dyes and DP agents is that commonly used reactive dye systems react with cellulose only under alkaline conditions, while DMDHEU requires acidic conditions to crosslink cotton. Since a DP agent can also fix dyes, alkaline conditions may not be necessary for reactive dye application. Therefore, a one-step dyeing and DP finishing process merits revisiting with the goal of increasing dye fixation levels and shade depths on cotton.
The aim of the present study is to produce good DP performance and high shade depth on cotton fabrics through a commercially applicable, combined dyeing and DP finishing process. The benefits of this process would be energy savings, shorter production times, reduced machine occupancy, increased productivity, quick response, and reduced salt and color levels in dyebath effluents.
Experimental
Materials
Mercerized and bleached woven cotton fabric (153 g/m2) was used in this study. The six reactive dyes used in this study were C.I. Reactive Orange 11, C.I. Reactive Red 55, C.I. Reactive Red 120, CI. Reactive Blue 19, CI. Reactive Blue 21, and C.I. Reactive Black 5. Dye selection was based on parent structure, reactive group, commercial popularity, and availability. All dyes used, durable press finish agent (DMDHEU, 44% aqueous solution), DP catalyst (MgCl2·6H20), and cationic agent (choline chloride, HOCH2CH2N(CH3)3+C1−) were commercial products.
Processes
Dye and finish application were conducted using the following three general methods.
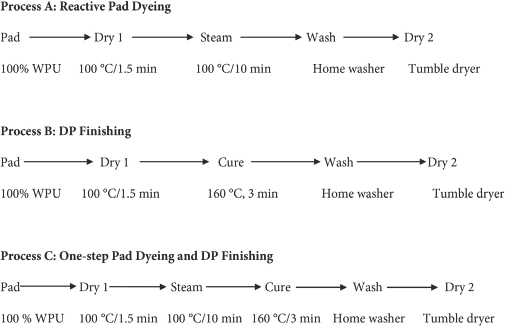
The padder and ovens used in the first drying and curing steps were obtained from Werner Mathis AG and the steamer was obtained from XORELLA AG. The steamed or cured fabrics were washed in a home washing machine (Whirlpool Cabrio) and dried in a home tumble dryer. The washing and drying conditions were those specified in AATCC Test Method (TM) 124 (permanent press conditions) to remove unfixed dyes and DP finish.
Bath Formulations
Bath 1 for Process A
Except for Black 5, the shade depth was 2% on weight of fabric (owf). The shade depth for the black dye was 3.4% owf The pad baths were prepared using 20 g/L dye (34 g/L Black 5), 30 g/L urea, and 20 g/L alkali (NaHCO3 for Blue 19, Blue 21, and Black 5, and Na2CO3 for Orange 11, Red 55, and Red 120).
Bath 2 for Process B
DMDHEU finish (6% solid, owf) and 30% MgCl2 catalyst (based on weight of DMDHEU) were used. Accordingly, the DP finishing bath contained 60 g/L DMDHEU and 18 g/L MgCl2.
Bath 3 for Process C
For simultaneous dyeing and DP finishing, dyes were added to Bath 2 to form Bath 3, which contained 20 g/L dye (34 g/L Black 5). To assess the effects of salt levels, 10, 20, and 50 g/L of Na2SO4 were also used in Bath 3. The fabrics were dyed according to the one-step Process C.
To study the effects of a cationic fixative on the dyeing and finishing system, choline chloride (CC), a quaternary ammonium compound, was added to Bath 3. This formed a bath containing 2% owf dye, 6% owf DMDHEU, and 2% owf CC. The selection of CC as a cationic fixative was based on its low toxicity and the presence of a hydroxyl group that could potentially react with DMDHEU 3 to enhance washfastness.
Test Methods
Washfastness and crockfastness were evaluated using AATCC TM 61(2A) and TM 8, respectively. Fabric DP ratings were measured using AATCC TM 124, with which fabric smoothness was graded. Fabric wrinkle recovery was examined using AATCC TM 128. The tenacity and tear strength of dyed and finished fabrics were measured using ASTM methods D5034 and D1424, respectively. Fabric shade depths were indicated by
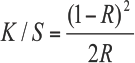
Results and Discussion
Shade Depth and Dye Fixation
A cotton fabric was dyed using the one-step dyeing and DP finishing process (Process C) and compared with the fabrics obtained using the conventional pad dyeing process (Process A). Table I contains
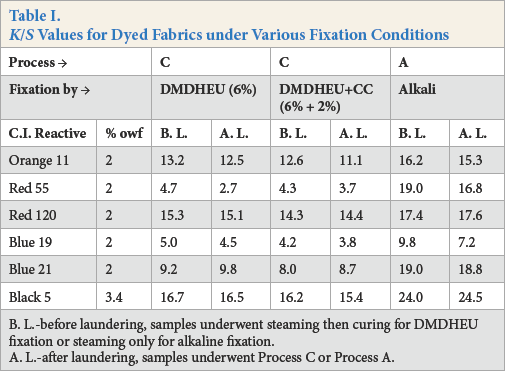
B. L.-before laundering, samples underwent steaming then curing for DMDHEU fixation or steaming only for alkaline fixation.
A. L.-after laundering, samples underwent Process C or Process A.
Dye fixation levels under different conditions were examined by comparing color strengths before and after laundering. Fig. 2 shows color strength retention values, where %
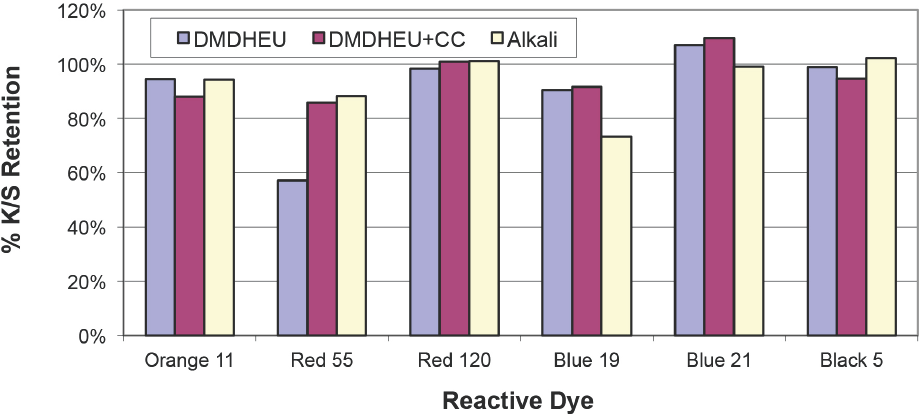
Color strength retention after laundering.
Bearing in mind that steaming is an important step in conventional pad dyeing, this step was used in the one-step dyeing and DP finishing process. Fig. 3 shows the effects of steaming on the shade depths of cotton fabrics dyed with Orange 11, using either the one-step process or conventional alkali fixation. For Condition Y fabrics (Fig. 3), steaming was omitted and the fabrics were cured after drying. A large difference in shade depths between the fabrics with and without steaming was observed, which indicates that without steaming, most of dye on the fiber surface was not fixed using either alkali or DMDHEU, and was washed off during the laundering step. Steaming provides moisture and thermal energy for fiber swelling and dye penetration, 17 which is the key for dye fixation by entrapping dyes inside fiber pores via DMDHEU crosslinking. Details of the mechanism of dye fixation in one-step dyeing and DP finishing processes will be presented in a separate paper.
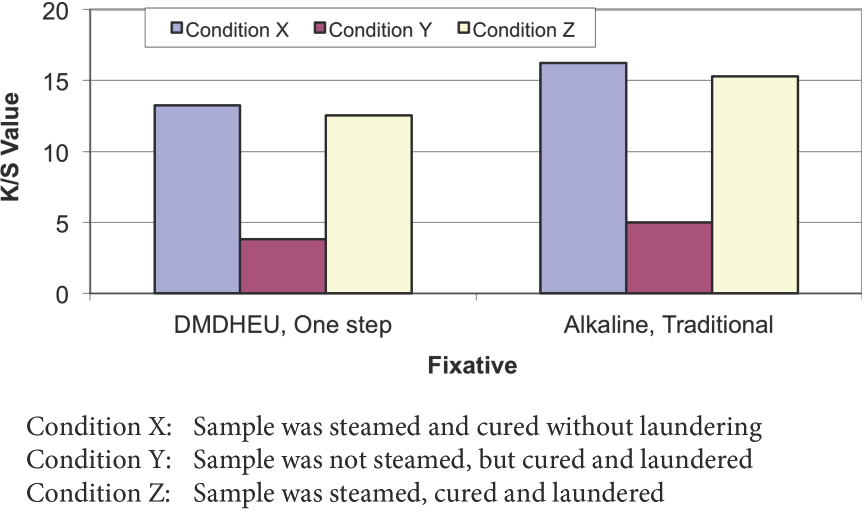
Effect of steaming on shade depth of Reactive Orange 11 dyed cotton.
Salt Concentration Effects on Shade Depth
A high concentration of salt is often used in conventional batch dyeing with reactive dyes because many reactive dyes have high water solubility and relative low substantivity for cotton. To illustrate our results in this area, Table II shows the effects of salt on Red 120 and Blue 19 dyeing of cotton through the one-step dyeing and DP finishing process. The effects of salt on the application of Red 120 were the same as in the conventional batch dyeing of cotton. The higher the salt concentration in these dyebaths, the higher the
Color Strength of Reactive Red 120 and Blue 19 Dyed Cotton at Various Salt Levels
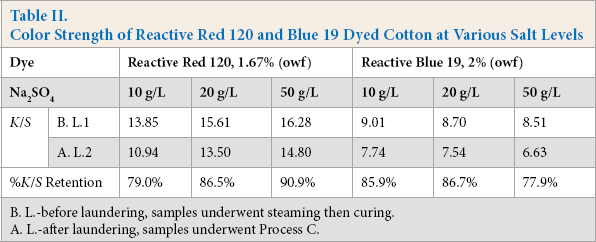
B. L.-before laundering, samples underwent steaming then curing.
A. L.-after laundering, samples underwent Process C.
Washfastness
Table III contains washfastness data for cotton fabrics dyed under different conditions. Most fabrics dyed in the presence of DMDHEU with or without CC had washfastness levels equal to or higher than those obtained using alkali fixation. Using DMDHEU fixation, fabrics dyed with Red 120 and Black 5 had washfastness ratings of 4.5 to 5 for both color change and staining. Orange 11 dyed fabrics gave the same staining ratings using the three fixation methods, while their color change ratings varied. The conventional alkali fixed fabric had the lowest color change rating (2.5), while the DMDHEU fixed fabric had the highest rating (4.5), making the one-step process more suitable for Orange 11 than the conventional method. Blue 19 dyed fabrics fixed under the three sets of conditions had the same color change rating, but different staining ratings. Conventional alkali fixed fabrics had high wool staining ratings but low nylon staining ratings. The one-step DMDHEU + CC fixed fabrics had the lowest stain fastness on wool (rating 2.5), while the DMDHEU only fixed fabric had high stain fastness on nylon (rating 4.0). Only Red 55 and Blue 21 applied using DMDHEU displayed lower washfastness than when applied under conventional conditions.
Washfastness Results
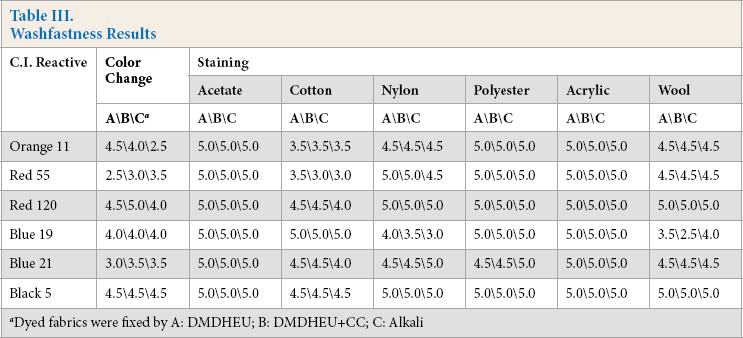
Dyed fabrics were fixed by A: DMDHEU; B: DMDHEU+CC; C: Alkali
Crockfastness
Table IV shows crockfastness data obtained from the application of six reactive dyes to cotton under three fixation conditions. The dry crockfastness of all dyed fabrics was excellent, with ratings of 4.5 or higher. However, wet crockfastness varied. Fabrics dyed with Red 120 and Black 5 in the presence of DMDHEU had wet crockfastness ratings of 3.5 or higher, which were slightly higher than the ratings from conventional alkali fixation (3.0). Comparing the fabrics dyed with Blue 19 and Blue 21 fixed under three different conditions indicated that fabrics treated with DMDHEU + CC had the highest wet crockfastness, while fabrics treated with DMDHEU alone had ratings equal to or lower than alkali fixed fabric. For the fabrics dyed with Orange 11 and Red 55, the use of DMDHEU with and without CC gave lower wet crockfastness than in the case involving alkali fixation. The results for average rating in Table IV indicate that dye fixation using DMDHEU gave wet crockfastness levels similar to those obtained using alkali fixation, while adding CC to DMDHEU usually improved wet crockfastness in the one-step process.
Crockfastness Data
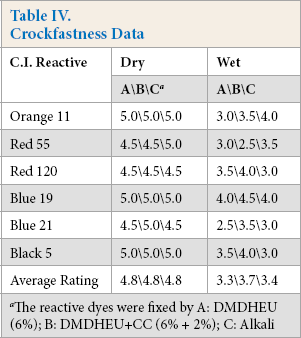
The reactive dyes were fixed by A: DMDHEU (6%); B: DMDHEU+CC (6% + 2%); C: Alkali
Lightfastness
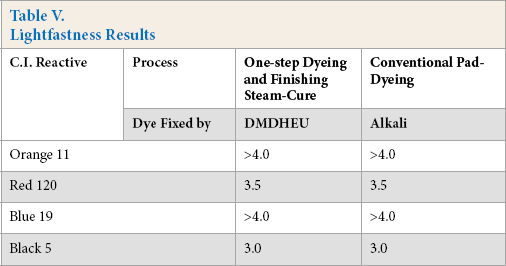
Table V displays the lightfastness data for the four reactive dyes (Orange 11, Red 120, Blue 19, and Black 5) dyed cotton fabrics through either the one-step dyeing and finishing procedure or the conventional pad dyeing processes. The data indicate that each dye of the four test dyes had the same lightfastness through the two processes. The lightfastness of dyed cotton was based on the reactive dye used, rather than the dye fixation methods.
Lightfastness Results
Dye Structure Effects on Dyeing Properties
Table VI shows shade depth and laundering fastness data for the six reactive dyes following DMDHEU fixation, along with dye structure information. Based on the dyeing results discussed above, Orange 11, Red 120, and Black 5 were found suitable for a one-step dyeing and DP finishing process. With DMDHEU fixation, fabrics dyed by these three dyes had good
Reactive Dye Structure Information, Shade Depth, and Washfastness
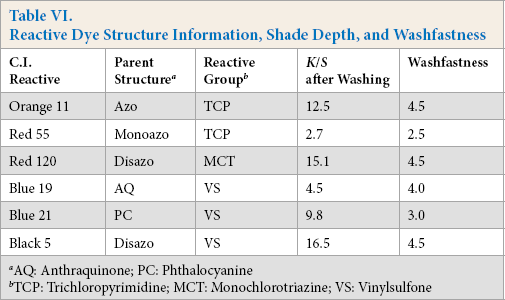
AQ: Anthraquinone; PC: Phthalocyanine
TCP: Trichloropyrimidine; MCT: Monochlorotriazine; VS: Vinylsulfone
Physical Properties
Table VII shows physical properties of Red 120 dyed fabrics through one-step application using DMDHEU or DMDHEU + CC, and conventional pad dyeing using alkali fixation. DMDHEU (6% owf) finished fabric and a control fabric were also evaluated for comparison. Fabrics dyed through the one-step process had a smooth appearance and good wrinkle recovery properties. Their DP ratings, wrinkle recovery angles, and fabric smoothness ratings were comparable to those of the conventional DMDHEU finished fabric. Tenacity and tear strength properties for the one-step dyed and finished fabrics were better than those from the traditional DMDHEU finishing process.
Physical Properties of Fabrics from One-Step Dyeing and Finishing, Conventional Pad Dyeing and Finishing Processes
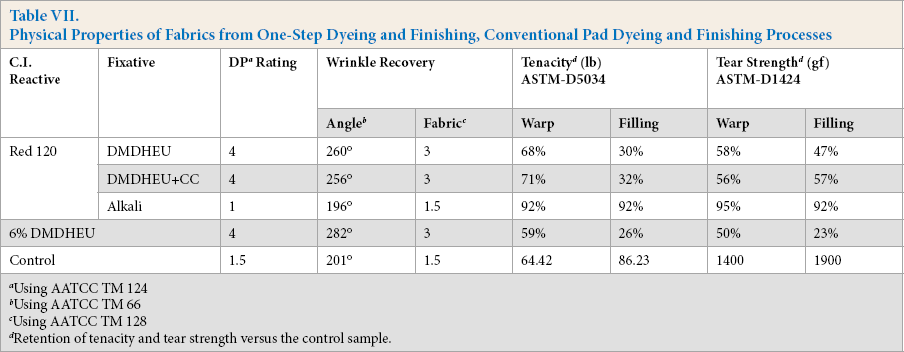
Using AATCC TM 124
Using AATCC TM 66
Using AATCC TM 128
Retention of tenacity and tear strength versus the control sample.
Conclusions
It was demonstrated that reactive dyes can be applied to cotton in a one-step process using DMDHEU in place of alkali. This method provides a one-step dyeing and finishing process that offers the potential for using any high temperature stable, water soluble dye to color cellulosic fibers. This means that acid dyes or direct dyes are potential alternatives to reactive dyes to reduce dye application costs. Adding choline chloride to the one-step dyeing system increased wet crockfastness of dyed fabric; however, dye uptake was slightly reduced. While salt was not necessary in the one-step process, dyes such as C.I. Reactive Red 120 benefited from adding salt to the dyebath, while high salt levels in the dyebath reduced dye absorption as well as dye fixation in other cases. Therefore, salt usage should be based on the dye selected for the dyeing process.
The dyed fabrics produced using the one-step dyeing system had comparable fastness properties and shade depths to fabrics dyed using conventional alkali fixation, and similar mechanical properties to fabrics that underwent conventional DP finishing.
Footnotes
Acknowledgements
The authors thank the Institute of Textile Technology (ITT) for full support of this research. A special appreciation goes to Judy Elson and Jeff Krauss for their invaluable help with lab scale studies.
