Abstract
The decomposition of flame resistant (FR) materials, either through elevated temperature or contact with flames, results in a range of chemical species, some of which can be quite toxic to humans. Small scale or bench scale tests for decomposition products have been done in the past, but there were always questions as to whether these were representative of full-scale flash fire test results. To determine whether the decomposition products would be of sufficient quantity to measure and whether different FR materials would produce a “signature” set of compounds, full scale testing was undertaken. Coveralls constructed from four common FR materials were evaluated. The methodology used allowed the determination of the thermal decomposition products of FR materials when exposed to flash fire.
Keywords
Introduction
Flame resistant (FR) garments provide some protection against a short-duration hazard. Typically, the performance of single-layer garments is evaluated using test methods such as ASTM F1930 or ISO 13506, which use an instrumented manikin to measure energy transfer, and, in turn, predict a thermal injury. This garment testing is carried out using propane as a fuel and the duration of the exposure is typically three or four seconds. Tests can be performed with longer or shorter durations but, for most single layer garments, these longer and shorter tests will provide results that are close to the maximum or minimum burn injury predictions.
FR garments are constructed using a range of textile materials and, increasingly, blends of different materials that are either inherently flame resistant or natural fibers treated to make them flame resistant. Work done by the author several years ago indicated that, when heated, some FR materials would produce large quantities of toxic decomposition products. 1 These products included hydrogen cyanide (HCN) and hydrogen chloride (HC1), as well as a range of organic compounds that resulted from the decomposition. The purpose of that work was to ensure that people evaluating these textiles using fire or flame were aware of the potential for off gassing and would take appropriate precautions against accidental exposure.
Subsequent to that study, Bradham et al. 2 developed a method of exposing very small samples of FR materials to an open flame and capturing the decomposition products for analysis. That study used the same sampling technique for acid gases (a gas sample was drawn through a midget impinger with an NaOH matrix). An unexpected result in this paper was the presence of cyanide in cellulosic materials that did not have nitrogen in the molecule. The authors attributed this to reaction with atmospheric nitrogen. Chloride was also sampled in the off gas products and was only found in significant amounts from one fabric, the only modacrylic blend tested.
Gas chromatograph/mass spectroscopy results were presented for the decomposition products for selected compounds, but only the match quality was shown (not the apparent concentration). 2 As the authors noted, only FR materials containing cotton or rayon showed fanfural or levoglu-cosenone (expected from cellulosic materials). Only the modacrylic containing FR material produced significant benzonitrile, not unexpected given the composition of the parent polymer.
To extend this work further and determine what compounds are formed, and at what concentrations, when FR materials are exposed to a short duration, large scale, flash fire, simulations were undertaken at the University of Alberta's Protective Clothing and Equipment Research Facility (PCERF).
Material Evaluated
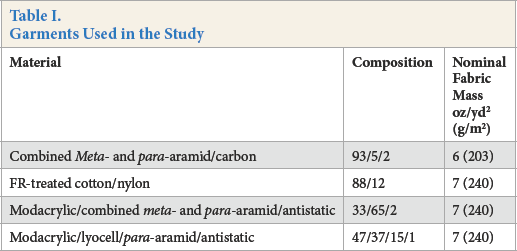
Garments were obtained commercially for use in the tests. The coveralls were tested as received from the reseller and were not laundered prior to testing. The tests were undertaken at the University of Alberta using an instrumented manikin and exposure system conforming to ASTM F1930 and ISO 13506. Each coverall was tested over 100% cotton underwear (T-shirt and brief) and gases were sampled from the room after exposure. Samples of the T-shirt (front and back) were sent to a commercial laboratory for the measurement of chloride (hydrochloric acid residue). The garments used in the study are shown in Table I.
Garments Used in the Study
Exposure Routes
Decomposition products of interest can be broadly divided into two categories; asphyxiants and irritants. 3 Asphyxiants inhibit the ability of cells to use oxygen, causing effects equivalent to oxygen deprivation. Carbon monoxide (CO) and hydrogen cyanide (HCN) are the primary asphyxiants considered in fire toxicity. Irritants, such as hydrochloric acid (HC1), in sufficient concentrations will cause pain and sensory interference along with the possibility of longer term pulmonary and respiratory effects.
Exposure to irritants or asphyxiants can also have an immediate effect on breathing, affecting both respiration rate and volume.4,5 An increase in respiration increases the exposure to harmful decomposition products, while decreasing respiration can exacerbate the effects of asphyxiants.
Dermal exposure or exposure to sensitive membranes like the eyes can result from the products of thermal decomposition contacting the skin or, if the materials are at concentrations higher than saturation at skin temperatures, via condensation on the skin surface. Acidic products, commonly hydrochloric acid, are produced in large quantities during the decomposition of materials containing modacrylic. Acids formed during decomposition are at high temperatures and are very reactive. In some modacrylic containing textiles, the acid deposition is sufficient to degrade 100% cotton underwear used under the FR materials during testing. This degradation results in the cotton losing significant strength to be torn as easily as tissue paper.
Methodology
Acid Gases
Acid gases, specifically HCN and HC1, were measured in the burn chamber following each exposure by drawing a sample of room air through a midget impinger containing 0.1 M sodium hydroxide. After the exposure, the room air was mixed using fans and 10 L of room air was drawn through the impinger over a 10 min time period at a flow rate of 1 L/min. Since the room was not perfectly sealed, there was some exchange with the outside, both during and after flame exposure. Some decomposition products were carried out of the room with the flow of the plume— the average concentration within the room exhibited some decline over the sampling period. Therefore, the measured concentrations were lower than would be obtained if the room had been perfectly sealed. Leakage tests done prior to sampling showed that the room had a relatively low exchange rate after the exposure, so the decline in concentration with time was not considered to be a significant factor. A hand-held analyzer (Sensit HCN analyzer) was also used to obtain concentrations of HCN following exposures. Comparisons of the concentrations measured with the hand-held unit and the impingers were made.
Sampling for CN− and Cl− specifically was undertaken due to their significance in fire toxicity for HCN and HC1, respectively. Both are also expected decomposition products among the garments evaluated in this study. 6
Volatile Organic Compounds
Sampling for volatile organic compounds (VOCs) was done by drawing a sample of air into a polyvinyl fluoride film sample bag using an evacuated sample system. Sampling was done from two locations: near the face of the manikin immediately following the exposure and from the room after a 5 min mixing period. Samples were sent to an analysis lab. The constituents were determined using a combined gas chromatograph/mass spectrometer (GC/ MS) system. The GC portion of the instrument was used to separate the compounds and the mass spectrometer was used to identify each compound in the sample by comparing the instrument response to a library of spectral responses. Samples taken near the face of the manikin were obtained over ∼3 s and were intended to sample the residual gases evolving from the still hot FR garments. It was expected that these samples would provide some indication of the maximum concentration a person wearing the garments might be exposed to. Samples from the room after mixing were taken to get some idea of potential yields of compound and the concentrations that might be expected given the volume of an enclosed space. By measuring the concentrations in the burn chamber (volume known) one could infer what concentrations would be obtained in other volume spaces.
Acidic Deposition on Undergarments
Samples of undergarments (100% cotton T-shirt and brief) were sent to an analysis lab to determine the chloride content. Samples, ∼6 ×10 in., were taken from the front and back of the T-shirt and combined as a single sample to ensure that an average concentration of chloride ion was measured. Fig. 1 shows where a typical underwear sample was taken.

Location of sampling of T-shirt after each exposure.
Experimental Results
Conditions Affecting Results
Note that all of the concentrations were approximate as there were no calibration standards available for these types of screening measurements. Table cells highlighted in green indicate compounds that were found across all materials at all exposure duration while the cells highlighted in blue indicate compounds that were detected across all exposure durations for a particular FR textile.
Note that samples taken near the manikin face immediately following exposure showed a larger number of compounds than those taken after mixing into the chamber volume. Mixing fans were used for 5 min to ensure uniform concentrations within the burn chamber prior to drawing an air sample Once the decomposition products were mixed into the 160 m3 chamber, the concentrations were significantly reduced. The analysis technique used (analysis of air samples with GC/MS) had detection limits for each type of chemical species. As a result of mixing chamber dilution, the number of chemical species in which the concentrations fell below the detectable limits increased and the number of compounds shown was reduced in the mixing chamber samples.
Propane was found in all samples as the exposure conditions were created using propane fuel. The fuel was released through the torches without any intentional mixing to create fuel rich diffusion flames. In diffusion flames, the reaction rates are limited by oxygen availability in the combustion zone, so the flames are orange, fuel rich, and sooty. This allows the creation of the 84 kW/m2 exposure condition, but because of the fuel rich condition, some of the fuel escapes the flames and is evident in the analysis.
Coverall Tests
Combined Meta- and Para-Aramid
Fig. 2 shows the 6 oz/yd 2 combined meta- and para-aramid coverall on the manikin after a 4 s exposure. Discoloration, shrinkage, and stiffening of this fabric was observed in all exposures, with the most significant effects resulting from the 4 s exposure. Underwear samples were from both the front and back of the T-shirt and combined as a single sample for the analysis of chloride residue.

Combined meta- and para-aramid coverall after 4 s exposure to propane diffusion flames.
Table II lists the compounds found in the samples taken near the manikin face immediately following the exposure. Table III lists the compounds found in the burn chamber air after a 5 min mixing period.
Measured Chemical Species from Combined Meta- and Para-Aramid Coverall (Manikin Face)
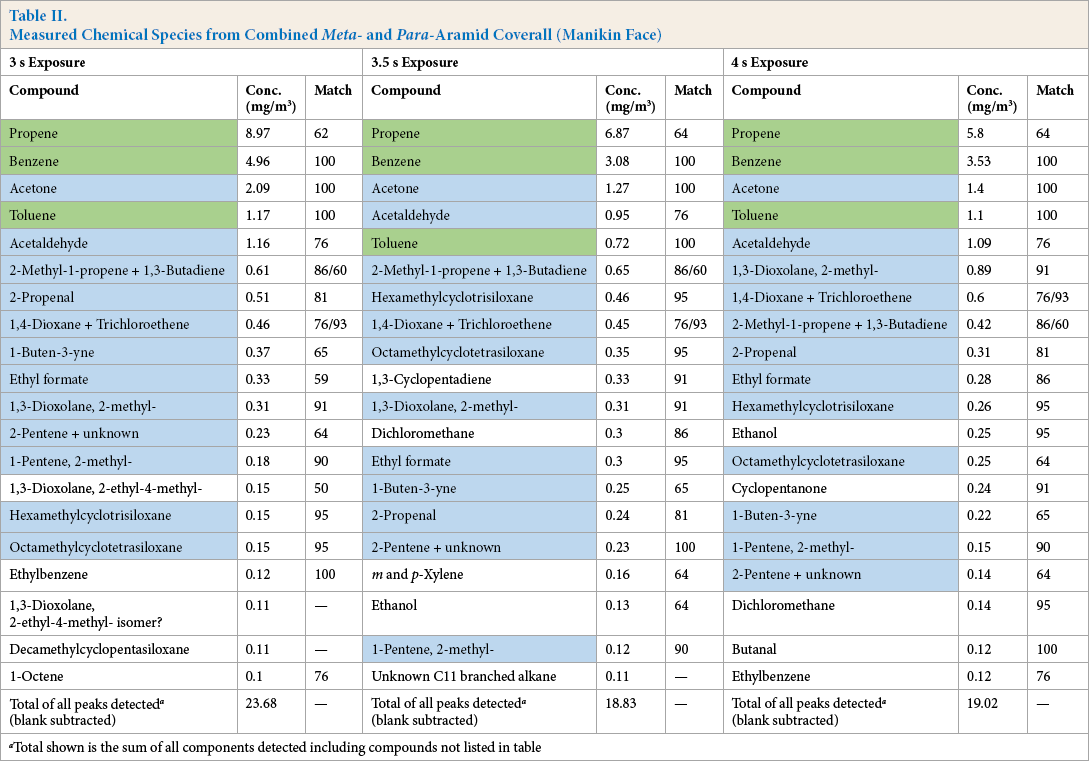
Total shown is the sum of all components detected including compounds not listed in table
Measured Chemical Species from Combined Meta- and Para-Aramid Coverall (Burn Chamber)
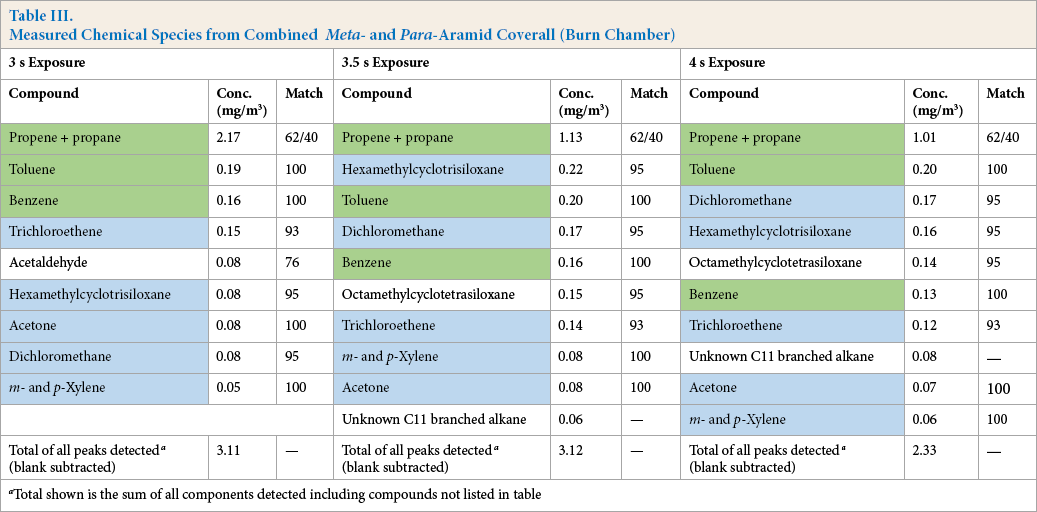
Total shown is the sum of all components detected including compounds not listed in table
FR-Treated Cotton/Nylon
The FR-treated cotton coverall showed increased damage and residue on the cotton underwear as the exposure was increased. Fig. 3 shows the result after a 4 s garment system exposure. Damage to the coverall was extensive (as would be expected from a lightweight FR cotton material) and the evidence of residues on the underwear increased with exposure level.

FR-treated cotton/nylon coverall after 4 s exposure.
Tables IV and V list the chemical species identified in samples from near the manikin face (Table IV) and in the chamber atmosphere (Table V) after mixing for 5 min. The range of compounds identified was quite different than that obtained from the combined meta- and para-aramid material and each of the materials tested produced a unique list of decomposition products.
Measured Chemical Species from FR-Treated Cotton/Nylon Coverall (Manikin Face)
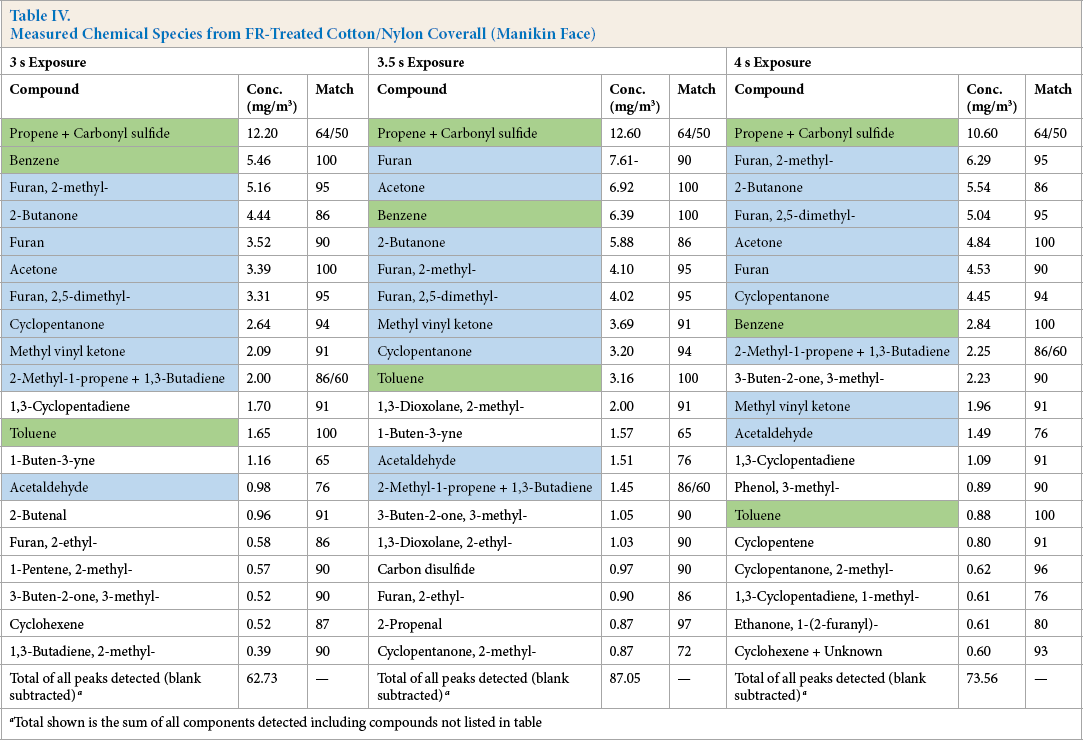
Total shown is the sum of all components detected including compounds not listed in table
Measured Chemical Species from FR-Treated Cotton/Nylon Coverall (Burn Chamber)
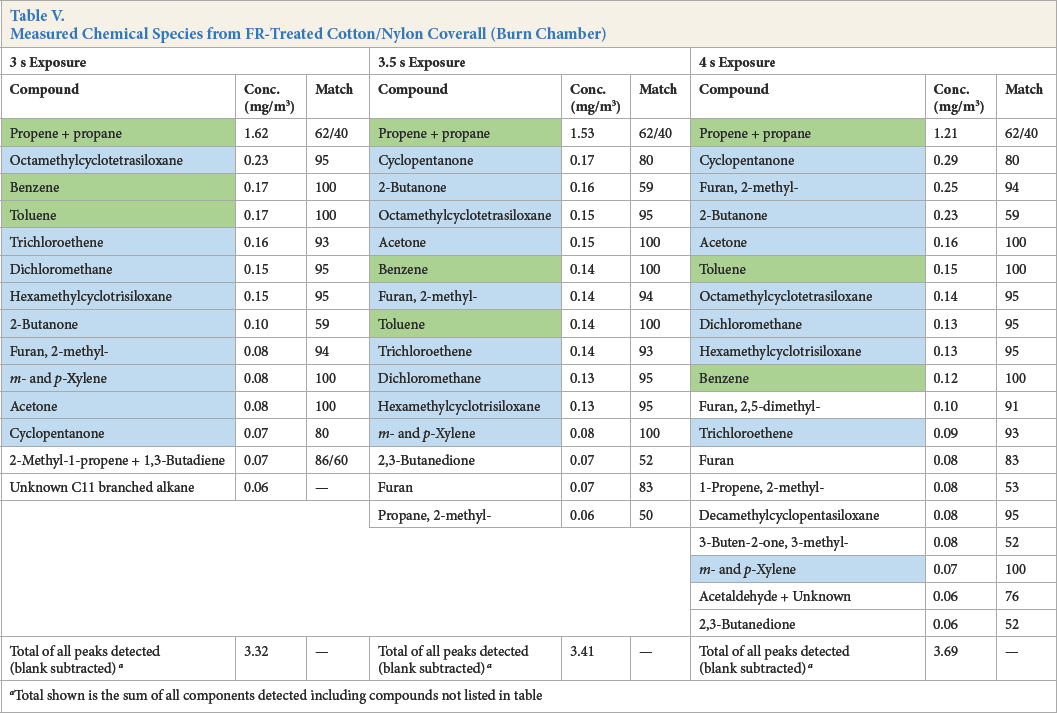
Total shown is the sum of all components detected including compounds not listed in table
Aramid/Modacrylic
Nominal 7 oz/yd 2 coveralls with 100% cotton T-shirt and brief were exposed using propane diffusion flames for durations of 3, 3.5, and 4 s. Fig. 4 shows the result after a 4 s garment system exposure. Damage to the garment, as expected, increased with exposure time and the apparent quantity of residue on the underwear, as indicated by color change, also increased.

Aramid/modacrylic coverall after 4 s exposure.
Exposure of the aramid/modacrylic blend coveralls produced the range of compounds shown in Tables VI and VII.
Measured Chemical Compounds from Aramid/Modacrylic Coverall (Manikin Face)
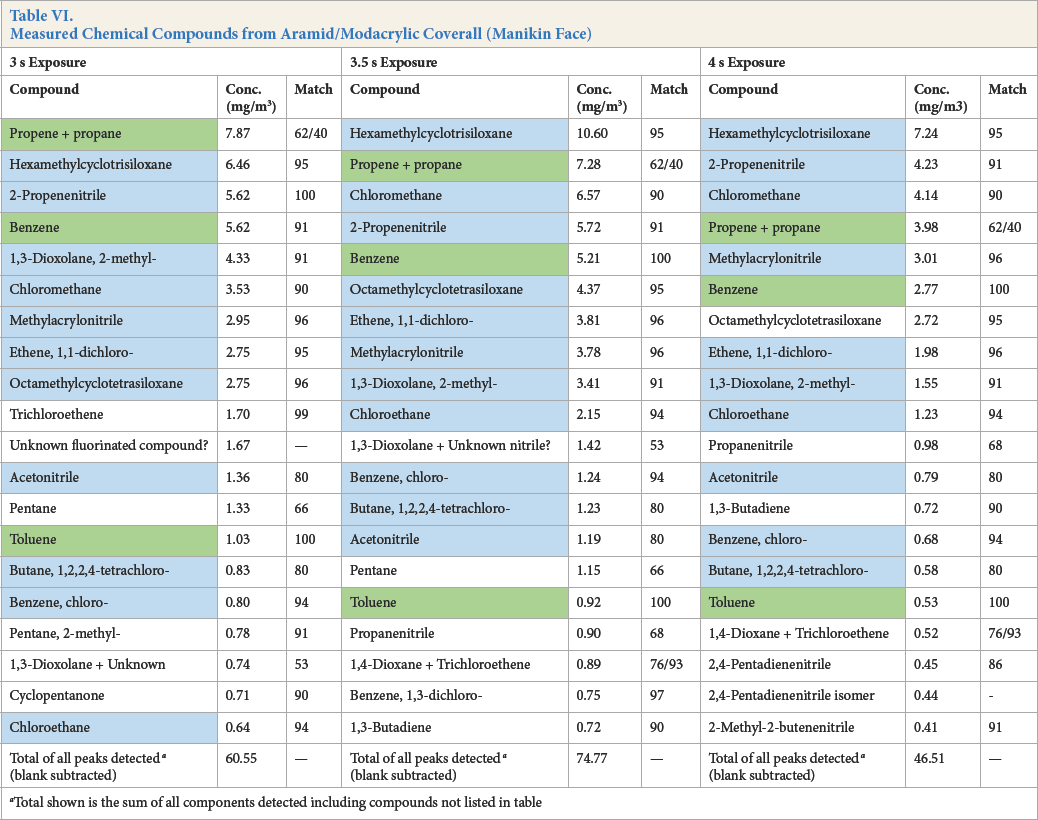
Total shown is the sum of all components detected including compounds not listed in table
Measured Chemical Compounds from Aramid/Modacrylic Coverall (Burn Chamber)
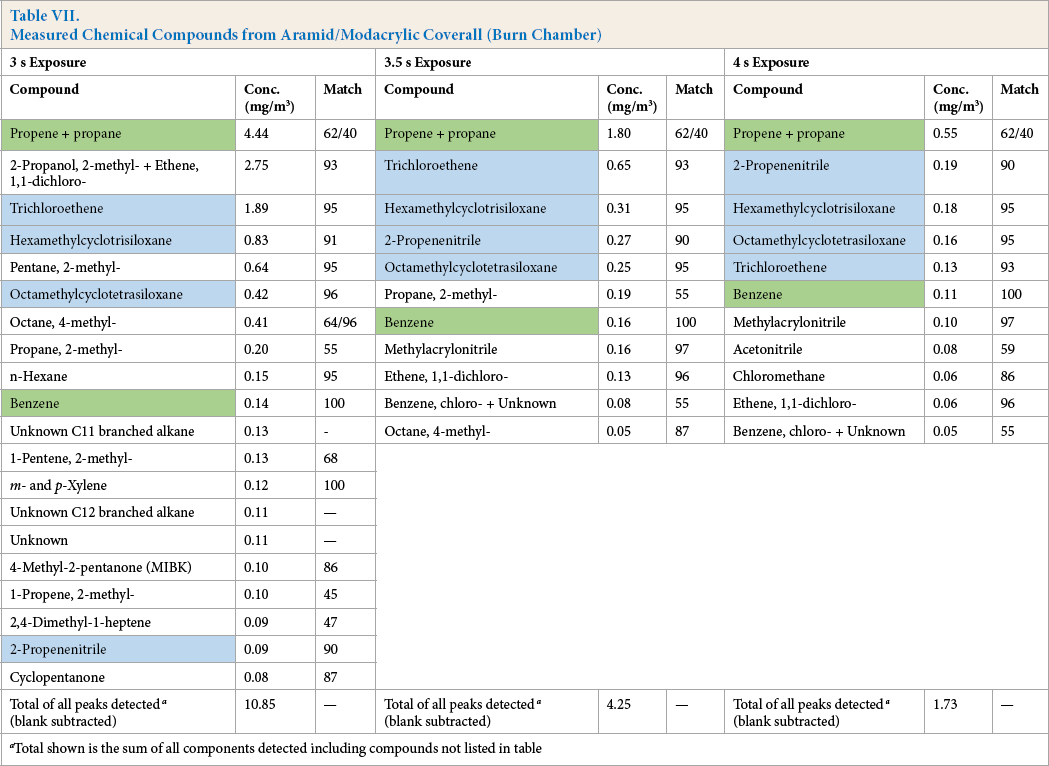
Total shown is the sum of all components detected including compounds not listed in table
Modacrylic/Lyocell/Para-Aramid/Antistatic
Three samples of nominal 7 oz/yd 2 modacrylic/lyocell/para-aramid coveralls were tested over 100% cotton underwear at exposure times of 3, 3.5, and 4 s. Fig. 5 shows the result after a 4 s garment system exposure.

Modacrylic/lyocell/para-aramid/antistatic coverall after 4 s exposure.
Exposure of modacrylic/lyocell blend FR coveralls resulted in the chemical species shown in Tables VIII and IX. It should be noted that no sample was obtained for the 4 s exposure in the room as the sample bag leaked before it could be analyzed.
Measured Chemical Species from Modacrylic/Lyocell/Para-Aramid/Antistatic Coverall (Manikin Face)
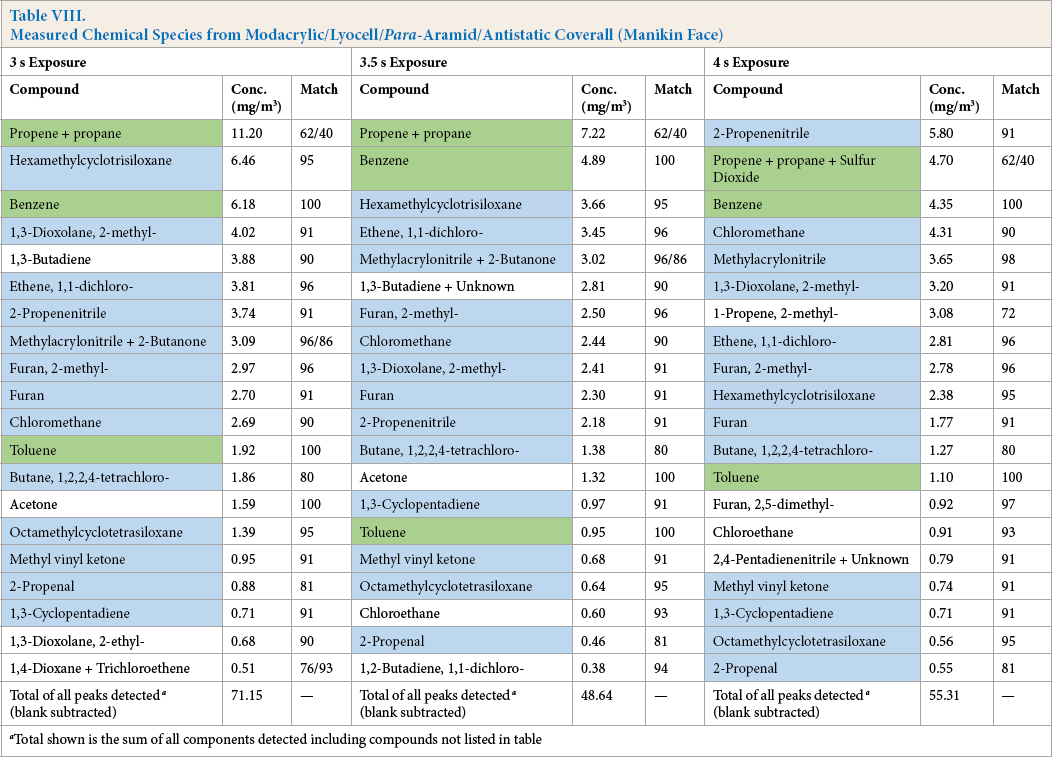
Total shown is the sum of all components detected including compounds not listed in table
Measured Chemical Species from Modacrylic/Lyocell/Para-Aramid/Antistatic Coverall (Burn Chamber)
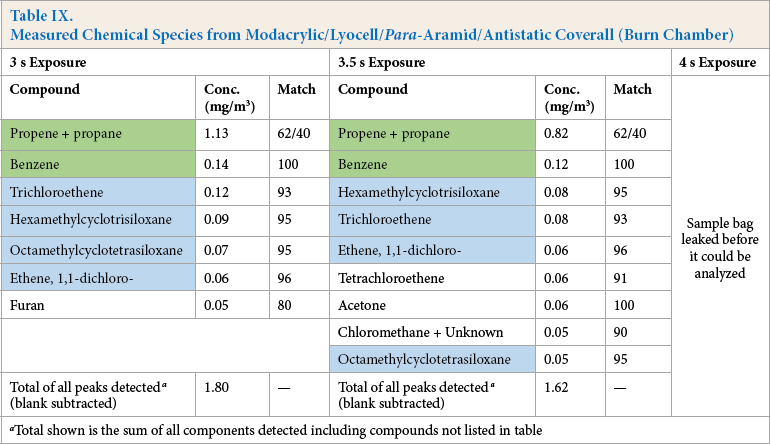
Total shown is the sum of all components detected including compounds not listed in table
Acid Gases Sampled from Burn Chamber
After each garment exposure, the atmosphere within the burn test chamber was mixed using fans for a period of 5 min. After mixing, gases were drawn through a midget impinger at a 1 L/min flow rate for a period of 10 min. The resulting sample was sent to a commercial laboratory for determination of chloride and cyanide.7,8
Table X lists the material tested, the impinger concentrations (mg/L), and the corresponding air concentration (ppm) based on impinger concentration. One should note that the burn chamber was not perfectly sealed and, as a result, the concentration decreased over time as a result of air exchange with outside air. Tracer gas (carbon dioxide) was used to evaluate the leakage prior to running these tests and the leakage rates were deemed acceptably low.
Sampling Results for Cyanide/Chloride from Burn Chamber after Test
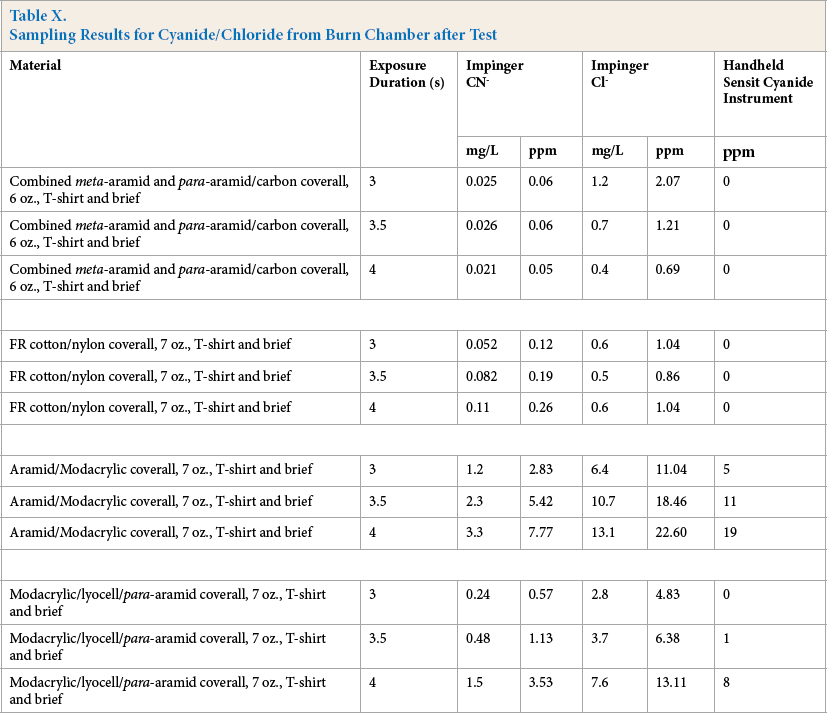
Results show that the concentrations of chloride and cyanide in the ambient air were relatively low (average cyanide < 8 ppm and average chloride < 23 ppm) and both cyanide and chloride were much higher where the material tested contained modacrylic (Table X). Average cyanide concentrations were greater than 150 times higher (at 4 s exposure) in the FR material that was a combination of modacrylic and aramid than the aramid (meta + para) alone. Hydrochloric acid measurements indicated a 35-fold increase in concentration for the same material comparison.
The burn chamber in which the tests were performed had a volume of ∼ 160 m3 (5650 ft 3 ), a large volume. As a result, decomposition into this volume produced low, but easily detectable, concentrations. Decomposition of the same materials into a smaller space would result in a corresponding increase in average concentration (halving the volume would give twice the concentration).
More importantly, there is reason to believe that the concentration of cyanide was significantly under reported using this method. The US EPA cautions that in a matrix containing both cyanide and chloride the chloride will oxidize the cyanide and the result will be a lower reported concentration than what was initially present. 9 This potential interference is outlined in Section 4 of EPA Method 335.4: “4.4 Oxidizing agents, such as chlorine, decompose most of the cyanides.” 9
This contention is supported by the concentrations measured during the exposure using a hand-held cyanide analyzer. The Sensit HCN analyzer is reported to have 2 ppm or 3% of reading (whichever is greater) accuracy and a range of 0-100 ppm. During the testing of these FR materials, the cyanide concentration indicated by the hand-held analyzer was on average 2.5 times higher than the impinger samples indicate. The relationship between cyanide concentration, as determined using impinger samples, to concentration determined using the hand-held analyzer is shown in Fig. 6.
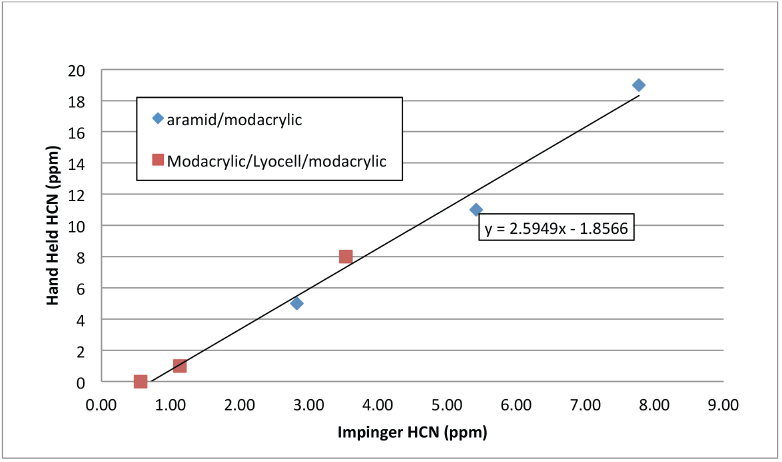
Relationship between cyanide concentration determined with a hand-held analyzer and midget impinger (25 mL).
Fig. 7 shows the relationship between acid gas production (as measured from the impinger samples) and exposure time for the same four materials (Table X). Acid gases were essentially undetectable from either meta/para aramid or FR-treated cotton materials. Acid gas production from the modacrylic-containing materials increased with exposure time. Exposures were not increased beyond 4 s because these single-layer coverall materials would show extensive burn injury predictions—they were not intended to provide protection beyond 3 or 4 s.
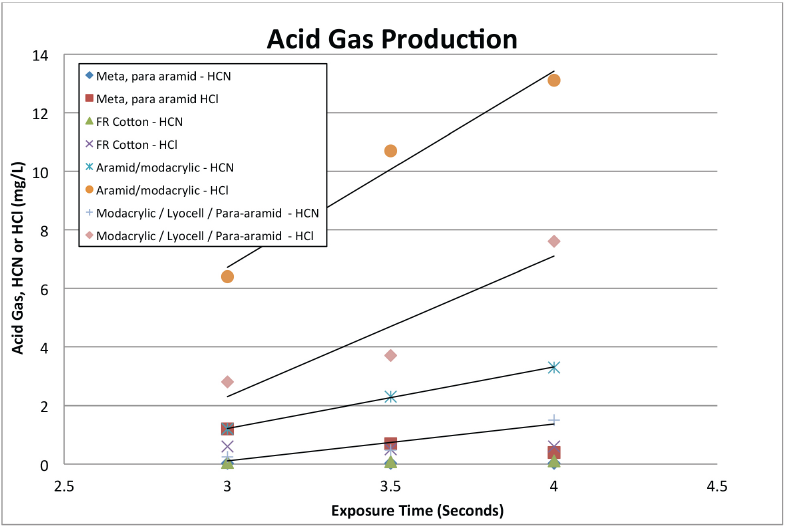
Changes in acid gas production (HC1, HCN) with exposure time, flash fire exposure.
Acidic Deposition on Cotton Underwear
Cotton underwear from each burn trial was sampled from both the front and back of the T-shirt and combined to obtain a representative sample. Samples were sent to an analysis laboratory for determination of chloride content post exposure. Chloride is a residual from hydrochloric acid produced primarily from the thermal decomposition of modacrylic containing fibers. The hydrogen ion in hydrochloric acid at elevated temperature hydrolyzes the cellulose molecules of cotton, fragmenting the polymer chain and resulting in rapid loss of strength. The chloride ion does not react with the cotton, but remains as a residual indicative of the quantity of acid initially present. 10
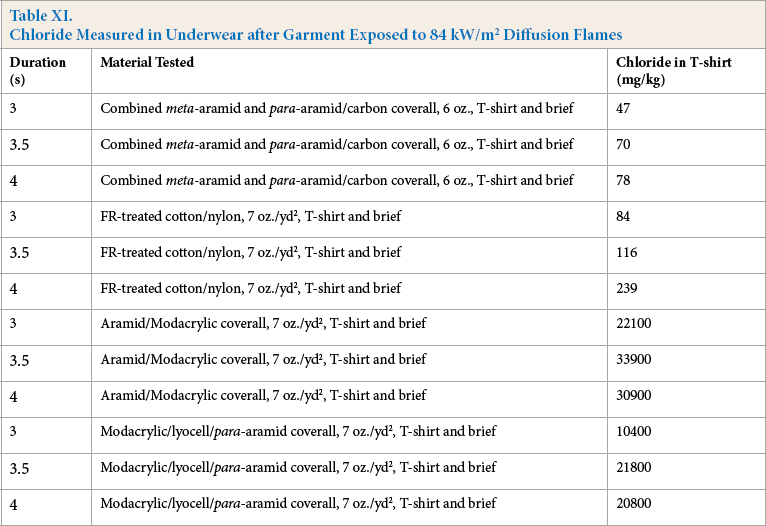
The quantity of acid deposited on the underwear strongly correlated with the amount of modacrylic fiber present in the textile (Table XI). With the combined meta- and para-aramid and the FR-treated cotton, there was essentially no acid deposition, whereas the aramid/modacrylic blend produced concentrations of chloride ion in the T-shirt that ranged from 22,000 mg/kg to more than 30,000 mg/ kg depending on the exposure duration and modacrylic content. With the modacrylic/lyocell blend the acid residues were lower, but still very high, ranging from 10,000 mg/ kg to more than 20,000 mg/kg. The hydrochloric acid was expected to react with the lyocell before reaching the underwear layer, resulting in the reduced chloride concentration measured in the underwear layer. These results show that the deposition of hydrochloric acid on the T-shirt material (or on the skin if no T-shirt was worn) was 200-400 times higher with the modacrylic-containing textiles than with the combined meta- and para-aramid blend and 100-200 times higher than with the FR-treated cotton. The acids deposited on the T-shirt (or skin) were at elevated temperature and hence were more reactive than at room temperature. The deposition of high temperature acid on the skin would certainly increase the severity of any burn injury received.
Chloride Measured in Underwear after Garment Exposed to 84 kW/m2 Diffusion Flames
Conclusion
The thermal decomposition of flame resistant (FR) textile materials, either in a fire or simply at elevated temperatures, produces a range of products, some of which are toxic to humans. Coveralls made from FR materials were exposed to flash fires ranging between 3 and 4 s and samples were obtained from near the manikin face immediately following the exposure and from the burn chamber after mixing. Samples of cotton underwear were analyzed for hydrochloric acid residue after exposure.
The methods used in this study showed that the materials produced could be measured and that each blend of FR material would produce a fairly unique set of compounds.
As expected, the measured concentrations of chemical species were much higher when sampled immediately following the exposure near the manikin face. Burn chamber samples were much lower after being mixed into the 160 m3chamber volume. Decomposition of these materials into a smaller space should result in corresponding increases of concentration (halving the room volume should double the concentrations measured).
Samples of cotton underwear analyzed for hydrochloric acid residue showed that materials containing modacrylic produced acid depositions that were 100-400 times higher than materials that did not contain modacrylic.
