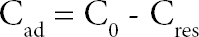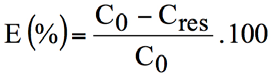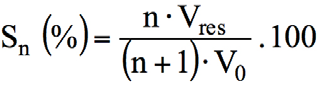Abstract
The feasibility of reconstitution and reuse of an acid dye dyebath for wool dyeing with different dye concentrations was studied. The residual dyebath was analyzed and reconstituted to the required concentration of dye, auxiliaries, and water for repeated reuse without deterioration of colorimetric properties. Analysis of CIELAB coordinates of dyed samples showed that the qualities of samples dyed in reused dyebaths were the same as those obtained from initial fabric dyeing, despite the very high number of reuse cycles. The influence of leveling agent on dyeing behavior remained acceptable. Analysis revealed that such reuse could reduce the amount of water, dye, and leveling agent to reduce effluent treatment costs. A reduction of 90.48% in water consumption was achieved after twenty successive reuses.
Introduction
The effluents that discharge from textile dyeing industries are complex and usually contain a wide variety of dyes and other auxiliaries such as dispersing agents, acids, bases, and salts. Dyes are major pollutants, causing environmental and health problems to human beings and aquatic animals. Thus, the removal of these materials from textile effluents is currently of great interest.
A wide range of methods has been developed for the removal of synthetic dyes from waste water including reverse osmosis, 1 ion exchange, 23 coagulation, 4 photodegradation, 5 adsorption, 6 decolorization by marine bacteria, 7 and oxidative 8 methods. Some of these mentioned processes are effective but relatively expensive. Therefore, the search for competitive technologies continues. Direct dyebath reuse is an effective way to reduce waste associated with textile dyeing.9-15 The advantages of this method are numerous and include reduction in the quantities of effluent discharged, the volume of water required, and the amounts of dyes, chemical auxiliaries, and energy consumed.
Khandaker, et al., 16 have used textile wash water for textile pretreatment instead of using fresh water to reduce the daily fresh water consumption. The pre-dyeing rinse wash water can be directly used for scouring and bleaching during the cotton yarn dyeing process. Raheel and Edgcomb 17 have applied the dyebath reuse method to dye cotton with class B direct and vinyl sulfone reactive dyes during four and eight reuse cycles, respectively. They examined the influence of impurity build up on color reproducibility by measuring color differences, color strength, and fastness, as well as the limits of dyebath reconstitution and reuse.
Jha 18 reused hydrolyzed reactive dyebaths for dyeing cotton fabric and wool fiber, at neutral and acidic pH values, respectively. Three types of reactive colors were used at different shade percentages. Dyed samples, analyzed in terms of K/S values, color difference, and washfastness, showed good color depth and tolerable fastness.
Koh, Kim, et al., 19 used this dyebath reuse technique to dye nylon microfiber with 1:2 metal complex dyes, reducing generated effluents. The spent dyebath was reused eight times, with fortification after each use, and obtained relatively acceptable color differences (ΔE values range from 0.11 to 1.6) between the samples dyed with the initial bath and those dyed with the reused baths.
Koh, Shim, et al., 20 tested dyebath reuse in the dyeing of polyamide with Acid Blue 113 dye. Reduction of dye and auxiliaries use was attempted for improving exhaustion by reusing 10 cycles of residual dyebaths.
Teli, et al., 21 tested this direct dyebath reuse technique for wool dyeing, with the aqueous extract of tea and madder as natural dyes, using ferrous sulfate and tannic acid as mordants. The number of dyebath reuses was four and two times for tea and madder, respectively. The resulting dyed samples presented some conformity problems, but had good to excellent washfastness and lightfastness.
Shams-Nateri 22 reused an exhausted dyebath for wool dyeing with natural madder dye. Madder quantities required to reconstitute the initial dyebath were determined by estimating the amount of reusable dye in the wastewater.
The reusable dye was evaluated using reflectance spectra of samples dyed in exhausted dyebaths and applying the Kubelka-Munk equation. Samples dyed in the reconstituted dyebath, repeated in three stages, showed approximately the same color and fastness as samples dyed in initial dyebath and offered an almost 20% cost saving.
In a previous study 23 the authors applied the dyebath reuse method in the dyeing of wool with acid and reactive dyes, at a dye concentration of 1% omf, to compare color properties of dyed samples. For both dyes studied, good results were obtained with an advantage for acid dyes in terms of color conformity and levelness.
The present study extended the previous work 23 to examine a wider range of concentrations of one of the dyes used. It confirmed the efficiency of acid dyebath reconstitution and reuse for wool dyeing with different dye concentrations. Dyebath exhaustions, color reproducibility, color conformity, levelness, and fastness of the dyed samples were analyzed. Reconstitution of the dyebath to the desired amounts of water, dyes, and auxiliaries was assessed after each dyeing cycle. This was performed in an attempt to obtain the same quality as the original dyed sample. The influence of leveling agent on dyeing behavior and its contribution to the obtained results were also studied. Savings in water, dye, and auxiliaries were evaluated after each dyebath reuse to confirm the utility of this method.
Materials and Methods
Materials
Bleached woven woolen samples were used for dyeing. Their principal physical characteristics were plain weave, 110.82 g/m2, fabric thickness of 0.66 mm, and warp and weft densities of 15 threads/cm and 14 threads/cm, respectively. Acetic acid (80%, Sigma-Aldrich) and Albegal A (alkyl amine polyglycol ether sulfate, amphoteric, Huntsman Textile Effects) were used as the fixing and the leveling agents, respectively. The acid dye tested in this work was Blue Marine Irganol 2RLS (λmax = 586 nm) supplied by CIBA (now Huntsman Textile Effects).
Methods
Dyeing
All dyeings were performed in an Ahiba Nuance Top Speed II-B laboratory dyeing machine (Datacolor) with a liquor ratio (LR) of 40:1. 22 , 23 A woven woolen sample (5 g) was immersed in the dyebath (200 mL) containing Albegal A (0.5% for 1% omf dyeing and 0.8% for 3% and 5% omf dyeings) and acetic acid (used to adjust the pH value between 5.5 and 6.5). The mixture was agitated for 10 min at 50 °C. Afterwards, the desired quantity of the acid dye (1%, 3%, and 5% omf), the temperature gradually increased from 50 °C to 100 °C in 45 min and maintained at 100 °C for 60 min. After dyeing, each sample was hot rinsed, soaped with 2 g/L soap solution at 40 °C, and finally washed with cold water.
Dyebath Reuse
Dyebath reuse was carried out as in the previous study 23 After dyeing, the residual dyebath was cooled to ambient temperature, filtered of pigment and waste material deposits, and analyzed with a Libra S6 spectrophotometer (Biochrom Ltd.) at the λmax (586 nm) of the test dye to measure the remaining concentration of dye. This allowed determination of the dye amount necessary to reconstitute the dyebath. The amount of dye available for dyeing in the reconstruction exhausted dyebath was calculated using Eq. 1.
C0 and Cres are the initial and the exhausted dye (dye remaining in the residual bath) solution concentrations (mg/L) respectively.
The exhausted dyebath was replenished with about 10 mL of fresh water to maintain a constant liquor ratio of the original volume. This allowed compensation of water lost due to removal of the fiber from the bath and evaporation during the dyeing cycle.
The residual dyebath pH was measured and adjusted to a value of 5.5-6.5 by the addition of acetic acid.
Albegal A was added based on the additional volume of fresh water added to reconstitute the original bath for 3% and 5% omf dye concentrations (for 1% omf, further addition of leveling agent was unnecessary throughout multiple dyebath reuses—no problems in evenness properties were found).
The procedures used as well as the parameters studied are represented in Fig. 1.
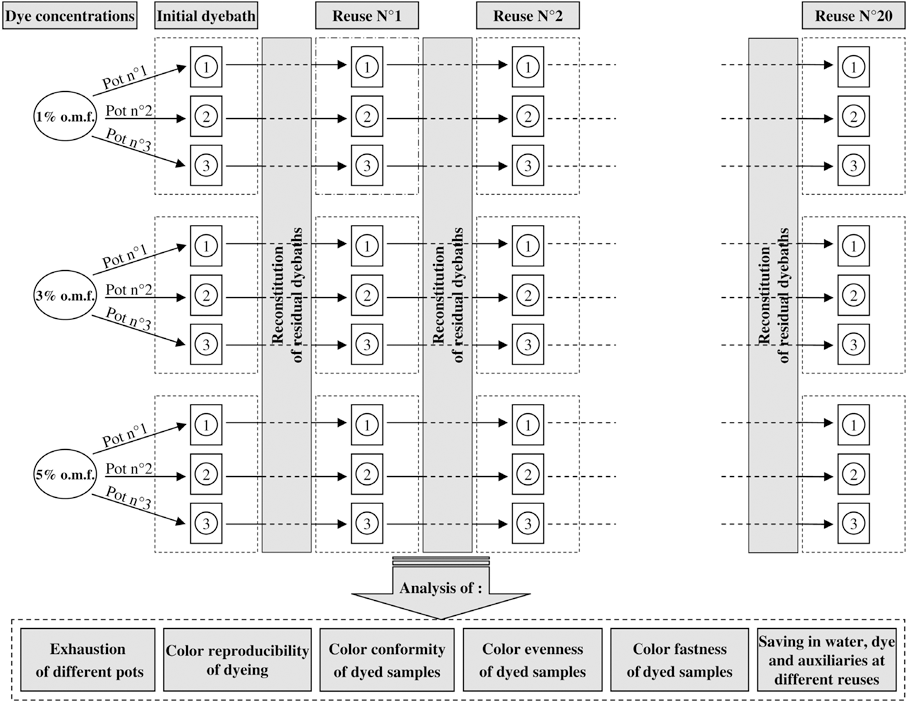
Dyebath reuse technique and studied parameters.
Dyebath Exhaustion
The extent of dye exhaustion (E, %) achieved for an appropriate dye concentration of the test dye was determined by measuring, using the Libra S6 spectrophotometer, the absorbance of the residual dyebath at λmax = 586 nm and deducing the remaining concentration of dye. E was calculated from Eq. 2.
Color Measurement
Color measurement of the dyed fabric was assessed using a Spectraflash 600 Plus (Datacolor) spectrophotometer coupled to a PC using a D65 light source and 10° standard observer condition. Each woven sample was folded twice to present a total of 4 layers of fabric for sufficient opacity. A series of color measurements was then performed in multiple locations. Three measurements were made per sample. This minimized the errors in reflectance measurements for this type of woven fabrics (i.e., starting from three measurements per sample, the obtained errors were quasi-constant.) 24 , 25
For color evaluation, CMC(2:1) 26 , 27 and CIEDE2000 28 , 29 color difference formulas were used (ISO and CIE recommendations, respectively). Color differences between dyed samples were calculated and analyzed to evaluate their color reproducibility, conformity, and levelness.
Colorfastness
Colorfastness was determined according to the respective French standard procedures: washfastness (NF G07-200), crockfastness (NF G07-016), and lightfastness (NF G07-067), which are equivalent to ISO standards: ISO 105-C06, ISO 105-X12, and ISO 105-B02, respectively. The changes in shade for washfastness and crockfastness tests were evaluated by gray scales. For lightfastness, a xenon test instrument (Suntest CPS Plus, Atlas Material Testing Technology) was used. The dyed samples and the blue scale were exposed to a xenon lamp light with a light intensity of 765 W/m2 for 72 h.
Savings Estimate
When reusing exhausted dyebaths, dyeing costs are usually reduced. In this section, estimates for savings in water, dye, and leveling agent use during various dyebath reuses are proposed. The percent saved water volume after n successive reuses of residual dyebath is given by Eq. 3.
V0 is the volume of the original dyebath (mL), Vres is the volume of the residual dyebath (mL), and n is the number of bath reuses. Indeed, when dyeing with the initial dyebath, the saving is equal to zero; both techniques consumed the same volume of water, V0. For dyebath reuse number 1, only a water volume of (V0- Vres) was added to maintain the original volume constant; so a save of Vres compared to the standard dyeing. After n successive reuses, the water savings can be given by Eq. 3. For dye and Albegal A, the corresponding savings depends on quantities added in each dyebath reuse.
Results and Discussion
Reuse Effect on Dyebath Exhaustion
Fig. 2 shows the exhaustion of the test dye with the number of dyebath-reuse cycles for the various dye concentrations (1%, 3%, and 5% omf) tested. For standard dyeing (dyeing using the initial dyebath), a high level of dyebath exhaustion (E, %) was obtained, with mean values of 91.39%, 95.39%, and 92.41% for 1%, 3%, and 5% omf respectively. For 1% omf dye concentration dyeings, exhaustion values were high and stable irrespective of the dyebath reuse time (mean value of 91.04% and a coefficient of variation of 1.47%).
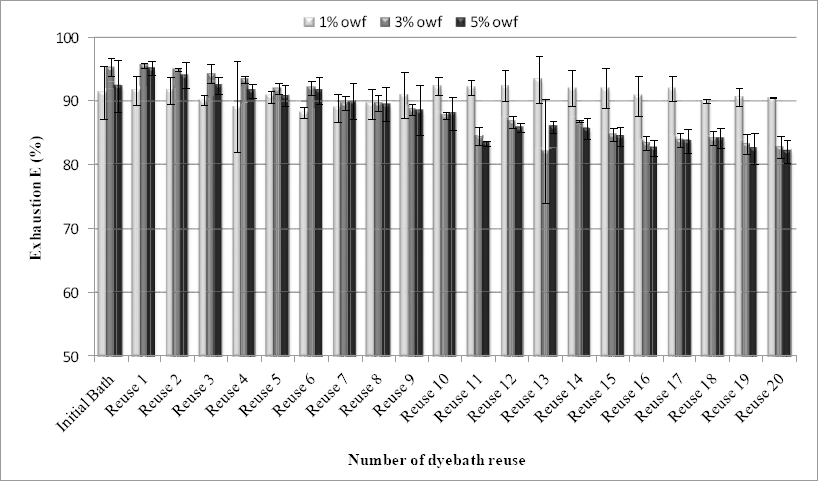
Exhaustion E (%) of test dye at different dyebath reuses (mean values with standard deviations).
However, when the samples were dyed at 3% and 5% omf, dyebath exhaustion decreased slightly as reuse proceeded. Although these exhaustions were lower than 95% (the usual level in wool dyed with acid dyes), 21 they can be considered good compared to other studies, especially those using reactive dyes in which exhaustion rates are very low and the integrity of the dyes is poor.17,18,23 Around 90% dyebath exhaustion means that 10% of the dye remained in the residual dyebath. This emphasizes the value of bath reuse to exploit the remaining quantity of dye and reduce the quantities of effluent discharged.
Color Evaluation
Color Reproducibility
Color reproducibility is one of the most important concerns in dyeing. Batch-to-batch reproducibility was examined in our study. For this, each dyeing was carried out three times in three different pots (Fig. 1). The color differences (ΔE) between the three obtained samples, based on two-by-two comparisons, were then calculated. The mean values of these color differences, expressed in AE CMC(2:1) and ΔE00 (CIEDE2000 color difference), are summarized in Table I.
Color Reproducibility for Various Dyebath Reuses a
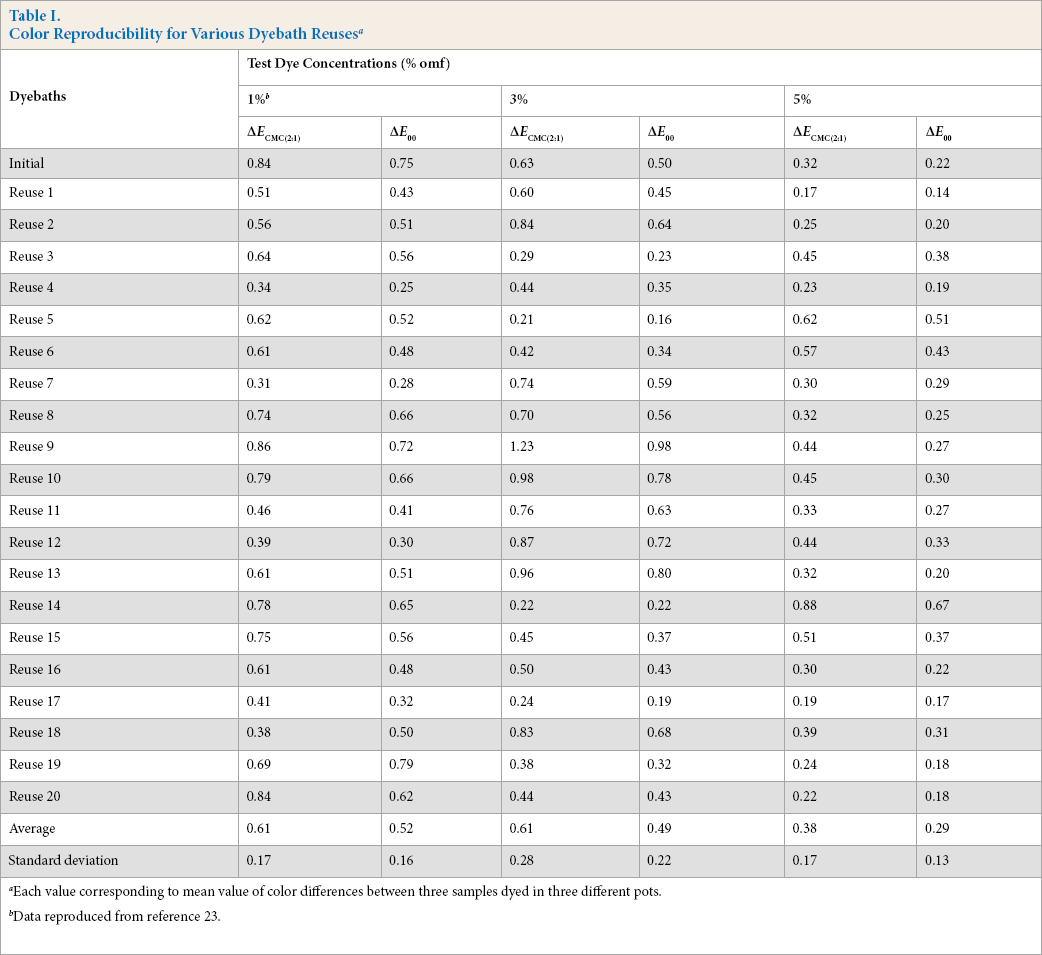
Each value corresponding to mean value of color differences between three samples dyed in three different pots.
Data reproduced from reference 23.
In the majority of cases, ΔE values were less than one. Average ΔECMC(2:1) and ΔE00 values ranged between 0.38 to 0.61 and 0.29 to 0.52, respectively. These values could be acceptable in the textile industry. In reality, a color tolerance for ΔE CMC(2:1) values as low as 0.5 was used, although 0.7 was more applicable in the textile industry. 27 Based on these color tolerances, dyeings in this paper were reproducible.
It is hard to distinguish these small ΔE values by eyesight. ΔE values for 1% and 3% omf dyeings were almost the same, while ΔE values for 5% omf dyeings were less (Table I). For the ΔECMC(2:1) and ΔE00 formulas, the dimensions of the tolerance ellipsoids are a function of the target position in the color space.26,28This is because color differences can be better discerned in some regions than in others. For saturated colors, these tolerances are less significant, which explains why ΔE values of 5% omf dyeings were lower than those of 1% and 3% omf dyeings.
Color Conformity
Table II shows, for the three studied dye concentrations, the colorimetric coordinates L*, a*, and b* of samples dyed in the initial dyebaths (considered as standard samples). It also presents ΔL*, Δa*, and Δb* values between samples dyed in each reuse dyebath and standard samples. For each dyeing, corresponding colorimetric coordinates were the mean values for three different samples (dyed separately).
Colorimetric Coordinates of Standard Samples a and Color Differences Between Reuse Dyed Samples and Standard Samples
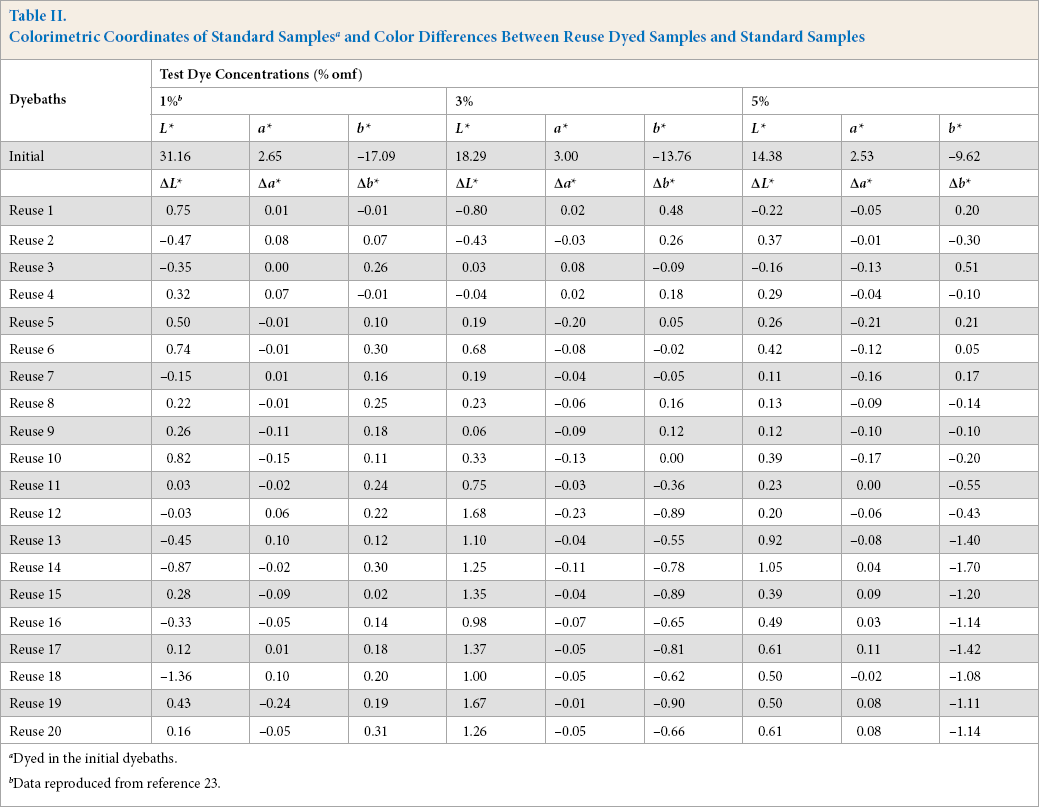
Dyed in the initial dyebaths.
Data reproduced from reference 23.
The differences between initial samples and samples dyed with reconstituted dyebaths were relatively small. The results showed small values of Δa* compared to ΔL* and Δb*. For blue colors, the a* component (red-green component) was insignificant. ΔL* and Δb* values appeared to be relatively high compared to others starting from the 12th and 13th reuses for 3% and 5% omf concentrations, respectively. These differences correspond to increased lightness (with relatively high values of ΔL*) and increased absolute values of the b* component (Δb* < 0). This could cause nonconformity problems.
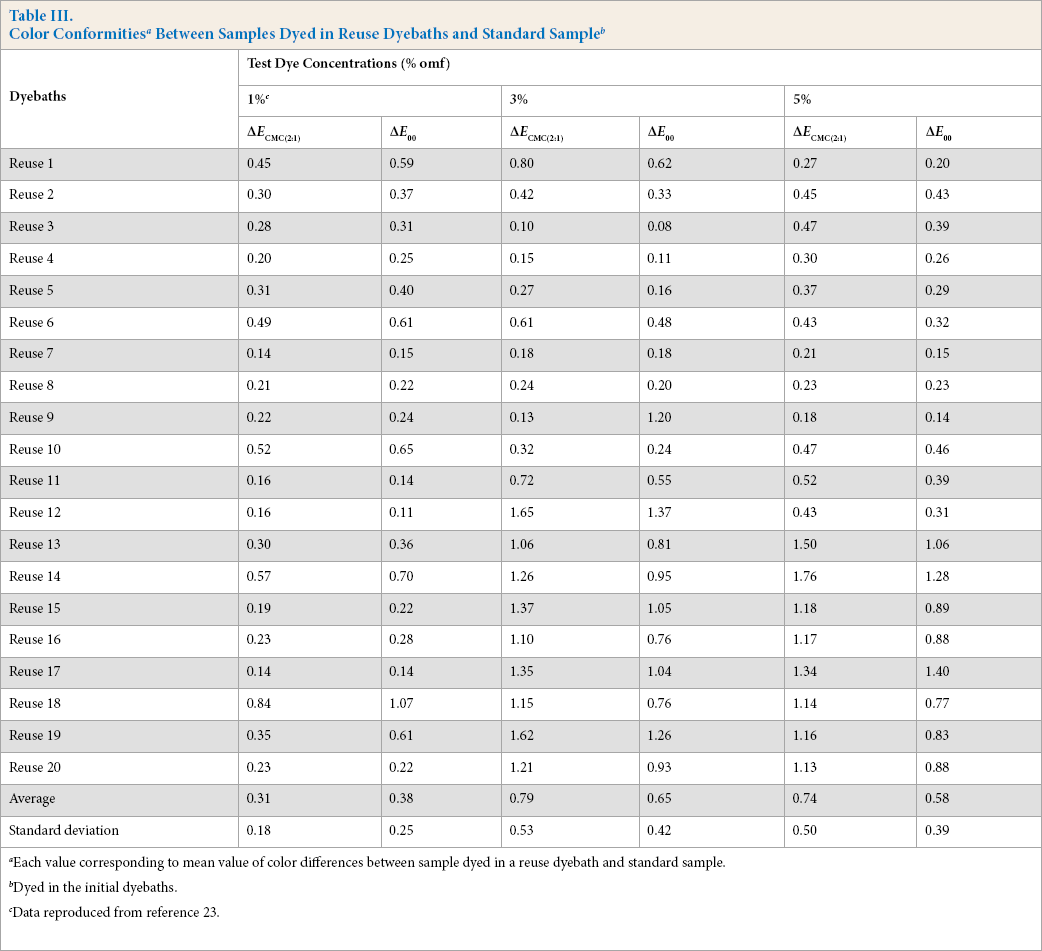
For each dyebath reuse, the total color differences between the samples dyed with reconstitued dyebaths and standard samples were calculated. Results, expressed as ΔECMC(2:1) and ΔE00 values, are summarized in Table III. The conformity of the dyed samples from the dyebath reuses compared to standard samples were evaluated.
Each value corresponding to mean value of color differences between sample dyed in a reuse dyebath and standard sample.
Dyed in the initial dyebaths.
Data reproduced from reference 23.
For 1% omf dyeings, ΔE CMC(2:1) and ΔE00 values were less than 0.84 and 1.07 with average values of 0.31 and 0.38, respectively. These differences were acceptable. This result confirmed the practicality of wool dyeing with reused acid dyebaths, with dyebath reuses in the literature ranging between 2 and 10.17–22 This number depends on the type of dyes used. In the present study, the number of reuses reached a value of 20 cycles. For 3% and 5%, omf dyeings, ΔE CMc(2:1) values were less than 0.80 and 0.52 until the 11th and 12th reuses, respectively. Later reuses showed some differences in the dyed fabrics when compared to fabric dyed in the original dyebath. However, these differences were still acceptable compared to results in other studies where color differences reached 1.6. 19 In addition, corresponding ΔE00 values were less significant and did not exceed 1.4 (5% omf, reuse no. 17). These results could be attributed to an excess of leveling agent (Albegal A) used that may cause a decrease in dyebath exhaustion and thus an increase in the clarity of the dyed sample. To confirm this hypothesis, the influence of the leveling agent on dyeing behavior was studied. Tests were carried out under 3% omf dyeing conditions. Results are shown in Figs. 3 and 4.
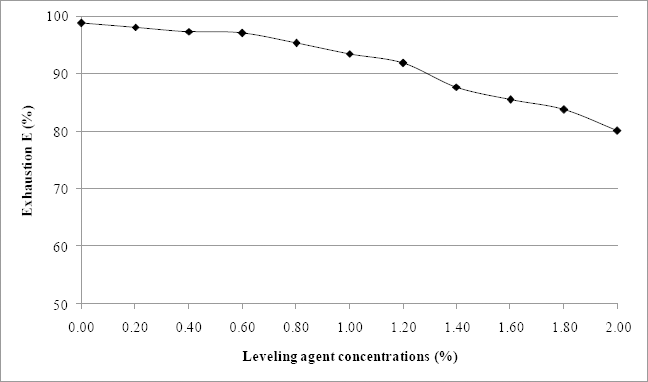
Exhaustion E (%) of test dye at various leveling agent concentrations using 3% omf dye.
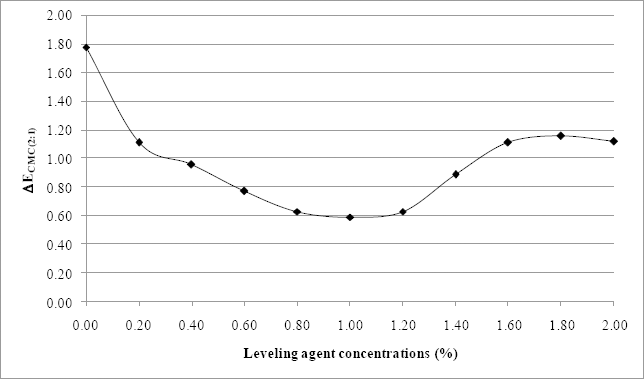
Color differences (ΔECMC(2:1)) between standard sample and samples dyed at various leveling agent concentrations using 3% omf test dye.
Fig. 3 reveals a gradual decrease of the exhaustion percentage with increasing concentration of leveling agent. This decrease can reach a dyebath exhaustion of 80%. However, it remains higher than exhaustions obtained for reactive dyes. 23 ΔECMC(2:1) values, determined between the sample dyed with a bath containing 0.8% of leveling agent (considered a standard sample) and samples dyed with other amounts of leveling agent are given in Fig. 4. When the amount of Albegal A ranged between 0.4% and 1.4%, the ΔECMC(2:1) values were below 1, indicating that the quantities of leveling agent did not cause differences between the dyed and standard samples. However, outside this range, ΔECMC(2:1) values were greater than 1. Indeed, when the Albegal A quantity was less than 0.4%, dyed samples were darker than the standard sample. The concentration of Albegal A used was likely insufficient to retard dye exhaustion on the material leading to higher values of ΔECMC(2:1). When the leveling agent concentration exceeded 1.4%, dyed samples became brighter than the standard one (with systematically larger values of ΔL*). This was the case with samples dyed using reconstituted dyebaths starting from the 12th and 13th reuses for 3% and 5% omf concentrations, respectively (Table II). Higher quantities of this agent may block the reactive sites present in the wool fabric, preventing fixation of dye molecules. This relationship between the amount of leveling agent and the color obtained is an interesting result and could be useful for researchers in this field.
Evenness
Evenness is the uniformity of dye distribution on the fabric. To assess the evenness of dyed samples, the following procedure was used.
Measure the color in three different areas within the surface of the sample;
Calculate ΔECMC(2:1) and ΔE00 values between these three areas taken two-by-two;
Estimate the evenness of each sample by the average value of these three color differences.
Evenness values of samples dyed in different dye baths are shown in Table IV. Results proved that evenness achieved over successive reuses was good to excellent, irrespective of the dye concentration tested. Indeed, for the three dye concentrations and with the exception of some samples, the majority of color differences were less than 0.5. This excellent result was maintained through all 20 reuses.
Evenness Achieved over Successive Reuses in the Dyeing of Wool Woven Samples a
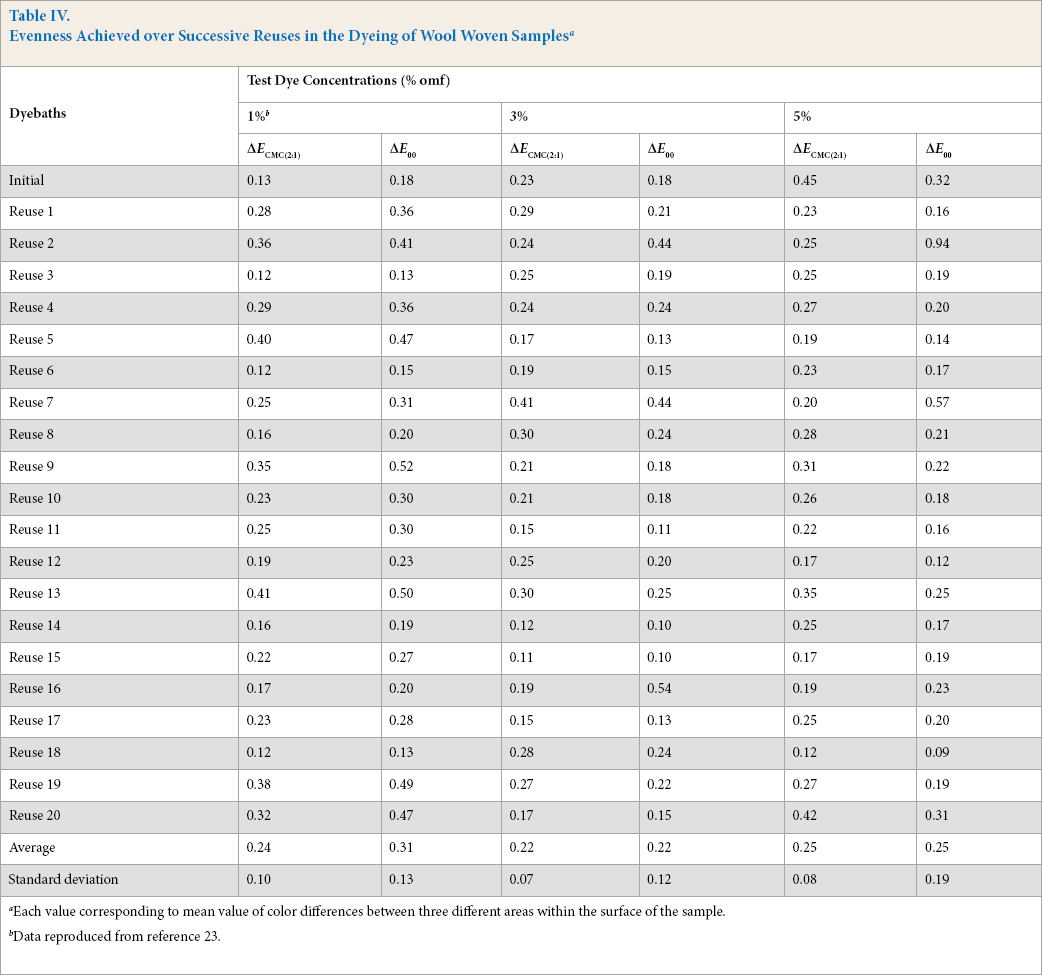
Each value corresponding to mean value of color differences between three different areas within the surface of the sample.
Data reproduced from reference 23.
Colorfastness
The washfastness, crockfastness, and lightfastness of the different dyed samples are shown in Table V. The colorfastness of different colored samples was good. In addition, the quality of samples dyed in reconstituted dyebaths was the same as samples dyed in the initial dyebath. In fact, all values were constant, except for 3% omf dye concentration use, where washfastness declined consistently from 5 to 4. No significant deterioration in colorfastness occurred during all cycles of dyebath reuse. These results confirm those found in other studies.17,22 However, in the present study, the number of dyebath reuses was even higher than in previous studies.
Colorfastness of Test Dyed Samples
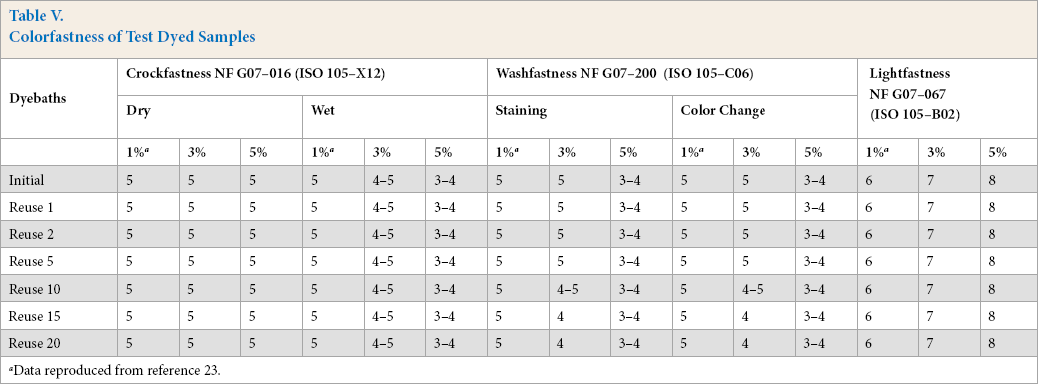
Data reproduced from reference 23.
Water, Dye and Auxiliaries Savings
Savings in water, dye, and leveling agent during the different dyebath reuses were evaluated. Table VI shows a comparison between the standard dyeing and the dye bath reuse technique in terms of water consumption. Results demonstrate that the savings in water (calculated according to Eq. 3) significantly increased with the rise in the number of reuse cycles and reached a value of 90.48% after twenty successive reuses on the laboratory scale.
Volumes (mL) of Used and Saved Water after the Different Dyebath Reuses
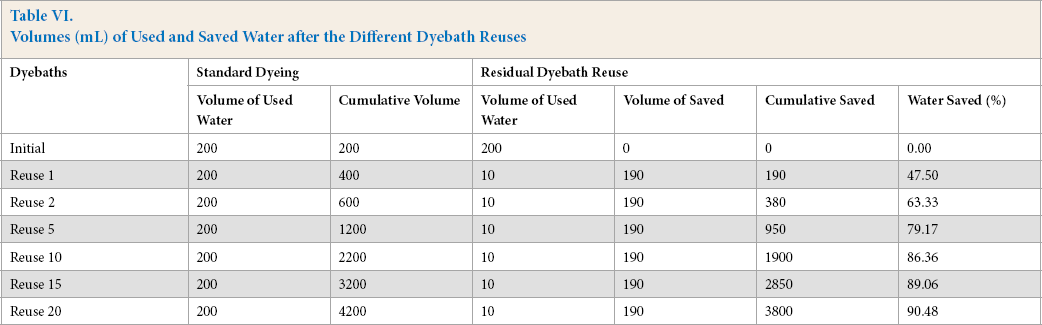
These results may be very useful in applications on an industrial scale. Indeed, with the reduction of water consumption, the dyebath reuse technique avoided treatment of many residual dyebaths and optimized the use of dyes and auxiliaries. Fig. 5 represents savings (in water, dye, and leveling agent) after each new reuse of the residual dyebath, for 3% omf dye concentration dyeings. Dyebath reuse offers, in this instance, a 10.21% dye savings compared to that of the conventional process. The total saving in leveling agent (Albegal A) was the same as that realized in water consumption. For 1% omf dye concentration dyeings, Albegal A was not added at all.
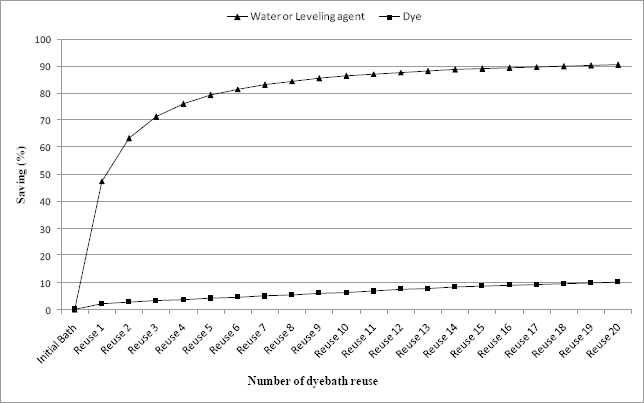
Total savings (%) in water, dye and leveling agent after each new reuse of the residual test dye bath (3% omf).
Conclusion
In the present study, wool fabrics were dyed with the test acid dye by applying the dyebath reuse method. Assessment of color parameters and colorfastness properties of obtained samples revealed that the quality of samples dyed in this study with recycled dyebaths was almost the same as samples dyed in the original bath. Indeed, after 20 reuse cycles, dyeing levelness and color consistency were very satisfactory. Differences expressed by ΔECMC(2:1) and ΔE00 values were hard to discern with the human eye. In addition, the colorfastness of all the samples were good and similar, irrespective of dye concentration and number of dyebath reuses. Analysis revealed that reuse of exhausted dyebaths can reduce of the amount of water, dye, and leveling agent and help reduce effluent treatment costs without sacrificing the quality of the finished goods.
Additional studies will be needed. Investigation of dyebath reuses with binary or trichromatic mixtures would help evaluate the performance of the applied method. Dealing with mixtures and different exhaust rates can be a challenge with regards to accurately reconstituting the bath. Also, application of this reuse technique in the semi-industrial and industrial scales are of particular interest. At these scales, other difficulties may appear, including storage of the residual dyebaths.