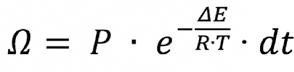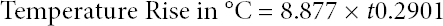Abstract
Current application of phase change materials (PCMs) in protective clothing is mostly limited to providing comfort through temperature regulation in extremely hot or cold weather. In this research work, PCMs were incorporated into a firefighters garment fabrics to enhance its thermal protective performance. Woven and knit Spentex (carbon-based flame resistant (FR) fiber) fabrics were permeated with selected hydrated salt PCMs. The rate of temperature rise on the wearer side was traced. Inclusion of PCMs increased the fabric thickness and its thermal conductivity which, in turn, resulted in a slight increase in thermal resistance. Thermal protective performance (TPP) testing showed a significant increase in protection time and performance with use of PCMs. Such extension in the tolerable rescue time can be important in preventing firefighters’ burns.
Introduction
At a certain constant temperature, a phase change material (PCM) goes from solid phase to liquid phase by absorbing thermal energy called latent heat of fusion. Below this temperature, this material changes from liquid to solid by giving back the energy absorbed in melting. The property of absorbing thermal energy and releasing it back at a constant temperature led to calling these materials “cold storages” or “heat storages” based on the plausible melting or freezing points. For example, ice can be referred to as a cold storage material. At a certain higher temperature, a material can go from liquid to vapor if given the latent heat of evaporation. It is well known that the latent heat of evaporation is much higher than the latent heat of fusion, but difficulties associated with the condensation process makes it hard to use the same material multiple times using liquid-gas transitions. In this study, investigation was limited to solid-liquid phase change materials for use in firefighter turnout gear.
The idea of using PCMs in clothing is fairly new 14 Microencapsulated paraffins are usually incorporated into fabrics to keep the body warm when moving from warm to cold environments. This effect will last as long as the time paraffin takes to change phase from liquid to solid (inside a capsule), releasing its latent heat of fusion, in less than 15 min. The opposite scenario of using PCMs as a cooling aid can be visualized.
In their work of modeling the effect of fire exposure on protective clothing embedded with phase change material, Mercer and Sidhu 5 in 2008 theoretically proved that PCM addition makes firefighter clothing more protective. Paraffins that normally would be used in garments cannot be used for this purpose because of their low melting point as well as their flammability Zelba, et al., 6 and Mehling, et al., 7 classified substances and eutectics with potential PCM use as organics and inorganics. Organics consist mostly of paraffins, which have melting points less than 70 °C Propionamide and naphthalene melt near 80 °C, while high density polyethylene (HDPE) and erythritol melt slightly over 100 °C Inorganic materials are mostly hydrated salts. Some hydrated salts have high potential to be used as PCM in firefighter gear.
Henriques 8 in 1947 introduced an integral model (Eq. 1) that is still effectively used to predict skin damage and the time to a second degree burn on human skin as a measure of thermal protective performance (TPP).
Ω is a quantitative measure of burn damage at the skin. When Ω is less than 0.5, there is no damage, but when Ω is between 0.5 and 1.0 there are first-degree burns. When Ω reaches a value of 1.0, there are second-degree burns; P is the frequency factor, S−1; ∆E is the activation energy for skin, J/mol; R is the universal gas constant, 8.315 J/(mol·K); T is the absolute temperature at the basal layer; and t is the total time for which T is above 44 °C.
The TPP value is determined as the intersection of the time-dependent increase of temperature as measured by the calorimeter, placed in place of the skin, to a time-dependent empirical performance curve known as “Stoll criteria” (Eq. 2).
This criterion was developed from the second-degree burn injury model generated from experimental burn energy data by Alice Stoll and Maria Chianta 8 based on the work of Henriques. 9 , 10 It is used in ASTM standard D4108-8711, NFPA 197112, and ISO 1749213. This criterion will be denoted as SDB in the article graphs.
The objective of the current work was to study the effect of selected PCMs embedded in firefighter fabrics on the protection performance of the permeated fabrics against heat and fire in terms of thermal conductivity and TPP.
Materials
Fabrics
The material used in firefighter turnout gear (ensembles) consists of three different layers: an outer shell, a moisture barrier, and a thermal liner. The thermal liner layer is where PCM was added in this study. Various Spentex (carbon-based flame resistant (FR) fiber) fabrics were tested in the thermal liner layer of firefighter ensembles. Fabrics were donated by Global Safety Labs Inc. (Tulsa, OK, USA).
For this study, two knitted carbon-based FR fiber fabrics (6.5 and 6.2 oz/yd 2 ) and a woven carbon-based FR fiber fabric (6.5 oz/yd 2 ) were used.
PCMs
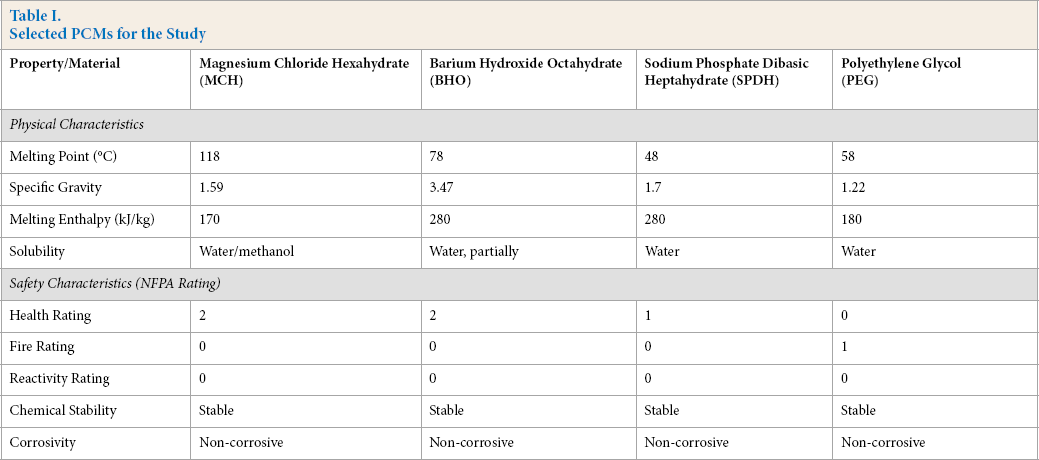
For phase change materials, inorganic materials were selected with the potential of being used in firefighter ensembles, considering their safety characteristics related to fire exposure. Selected PCM materials with different melting temperatures are given in Table I.
Selected PCMs for the Study
Experimental
Thermal Conductivity
Measurement of thermal conductivity of raw fabrics and PCM-permeated fabrics was carried out on an Alambeta instrument. 14 The fabric is placed between two plates of different temperatures where the heat flow between them is measured. The upper plate, equipped with a direct heat flow sensor, is heated and kept at 32 °C (human skin temperature), and the lower plate starts at room temperature (RT). Simultaneously, at a pressure similar to that of fabric thickness micrometers, the device measures the distance between the two plates as the fabric thickness (h) and measures the thermal conductivity coefficient (k) in W/(m·K). Thermal resistivity (r in K/W) is calculated as in Eq. 3.
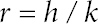
TPP
The TPP test was conducted according to ASTM D4108-87. 11 In this test (Fig. 1), the sample was placed in the sample holder having fabric outer side down facing the flame, while the sensor was placed on the top of the inner side of the fabric, making a gap of 0.25 in. if the tested fabric was single layered. If the tested material was multilayered, the sensor was placed directly on the back face of the nearest layer. The sensor measured temperature gradient when the sample was exposed to a flame with an energy level of 8.3 W/cm2(2.0 cal/cm2·s). The data was recorded as a temperature increase versus time and called heat flux curves. The intersections of heat flux curves with the SDB curve “Stoll criteria” gave the time to a second-degree burn in seconds. The result of multiplying this time (in seconds) by the heat flux (cal/cm2·s) is the TPP factor.
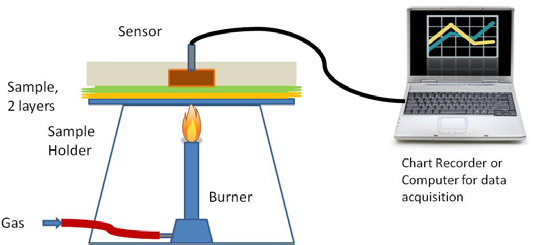
Schematic illustration of TPP test set-up.
Results and Discussion
In the first set of samples, knit and woven fabrics of the same material and mass per unit area were used. The starting thickness of the knit fabric was double that of the woven fabric. PCMs were dissolved in water until saturation and fabric specimens were then submerged in the solution until complete soaking was achieved. Specimens were removed fro m the PCM solution, put on padding paper, an d left to dry under the laboratory's standard conditions. The weight gain was considered as the amount of PCM added to the dry sample. Magnesium chloride hexahydrate (MCH) samples did not dry under the, standard lab conditions and had to be oven dried. The amount of PCM shown on the graphs in grams was evenly distributed over a fabric specimen area of 5 × 5 in.
Thermal Resistivity
Results of thickness, thermal conductivity, and thermal resistivity of the permeated fabrics are shown in Fig. 2. The relative increase in the woven fabric thickness with the increase in the amount of various PCMs was more profound than that for the knit fabric, which could be due to the special open structure of the latter. These results also show that thermal conductivity increased with the increased amount of PCMs in the permeated fabric. This is consistent with the fact that the PCMs used had higher thermal conductivities than the fabric itself. Thermal resistivity, as a result of the thickness divided by the thermal conductivity, considerably increased for woven fabric while slightly decreased for knit fabric in two of the three cases.
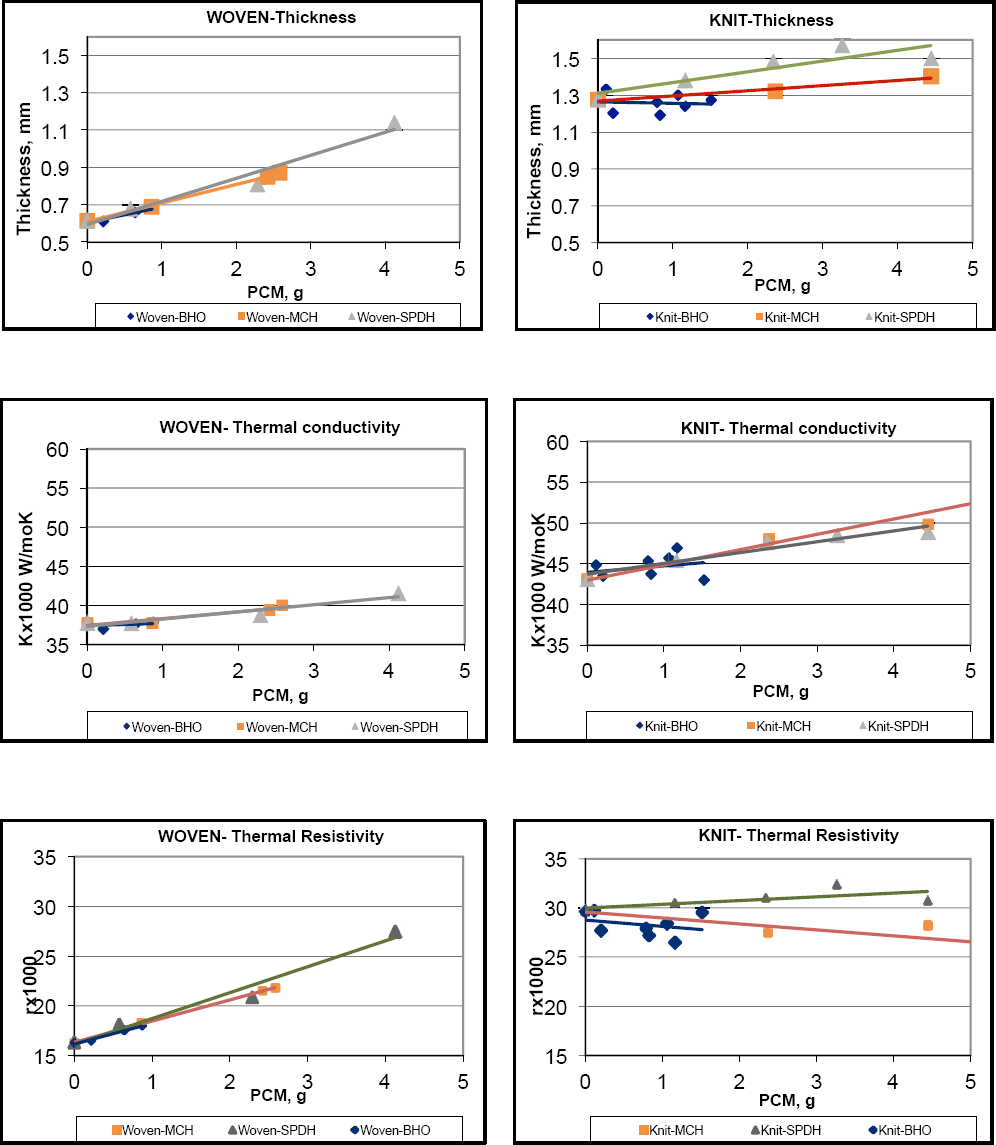
Effect of PCM on thickness (h), thermal conductivity (k), and thermal resistivity (r) when added to woven and knit carbon-based FR fiber fabrics.
TPP Results
TPP results are shown in Fig. 3. The heat flux was set at 2.0 cal/cm2·s (8.3 W/cm2) as directed by ASTM D4108-87. 11 Knit fabric samples showed greater increases in the protection time than the woven samples because they were able to carry more PCM than the woven samples in addition to the fact that the bulkiness and thickness were sources of more protection. Barium hydroxide octahydrate (BHO) showed more protection than sodium phosphate dibasic heptahydrate (SPDH). Magnesium chloride hexahydrate (MCH) samples absorbed moisture from the atmosphere, which affected the consistency of testing results, and consequently, were not included here.
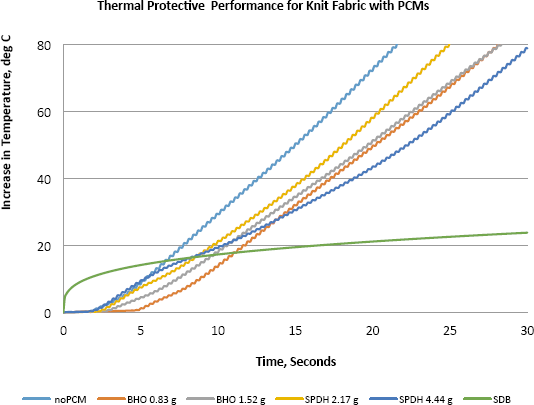
Effect of PCM on TPP.
In another set of samples, PCMs were distributed evenly between two fabric layers of knit carbon-based FR fiber fabric of 6.2 oz/yd 2 . In this case, the PCM amount as described by areal density was set at three levels: 0 g/m2(no-PCM), 236 g/m2, and 472 g/m2, where the higher density was twice the middle density. Table II shows the results of the time to second-degree burn. The resultant TPP factor was calculated at a heat flux of 2.0 cal/cm2·s (8.3 W/cm2).
TPP for Double Layer Carbon-Based FR Fiber Knit Permeated with Various PCMs at Various Concentrationsa
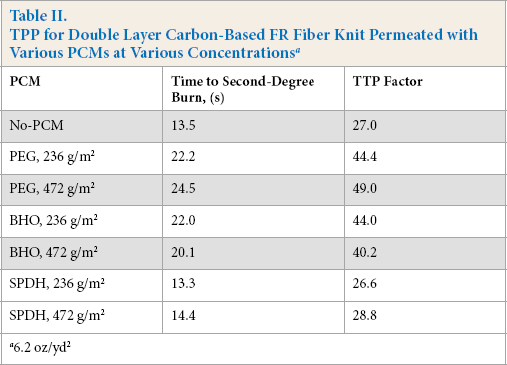
6.2 oz/yd 2
Despite the difference in melting temperature and melting enthalpy, polyethylene glycol (PEG)-treated samples showed a gain in protection comparable to BHO-treated samples containing the same amount of PCM. The SPDH-treated samples showed little gain in protection. As a generalized result, the TPP increased significantly when certain PCMs were added. Among these, melting temperature, melting enthalpy, and specific gravity were the determining factors for the PCM choice.
Figs. 4–6 show the change in temperature as well as the rate of this change with time for different densities of PCMs. It is evident that the increase in temperature with time for fabric with no PCM was greater than that of the fabric with PCMs in all cases. As shown here for the fabrics with no PCM, the dT/dt value was distinctly higher during the first few seconds and kept increasing to a certain point when it started to decrease as the temperature increased. The inclusion of PCMs obviously slowed down the increase in temperature on the side of the fabric close to the human body. After only 40s, the PCMs helped to keep the temperature lower by 30 to 40 degrees, which can be an important difference. Results show that the benefit from increasing the PCM areal density was not linear. This may be an indication of working at the high end of areal density levels.
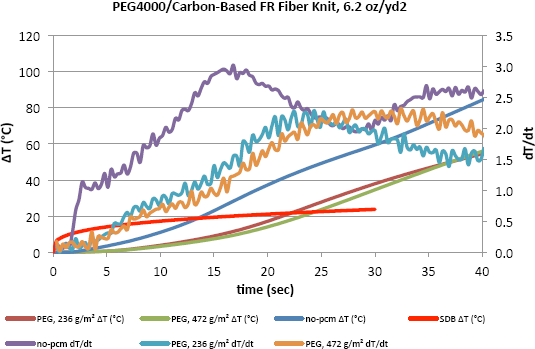
Temperature change and dT/dt vs. time of carbon-based FR fiber knit (6.2 oz/yd 2 ) fabric incorporated with PEG4000 and tested on the TPP test set-up.
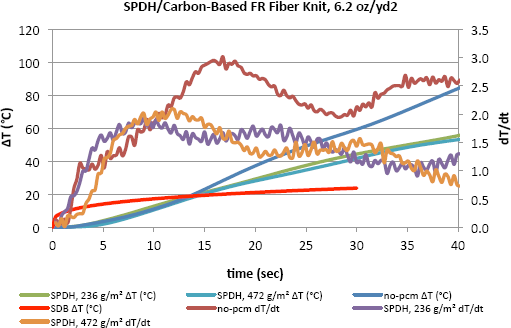
Temperature change and dT/dt vs. time of carbon-based FR fiber knit (6.2 oz/yd 2 ) fabric incorporated with SPDH and tested on the TPP test set-up.
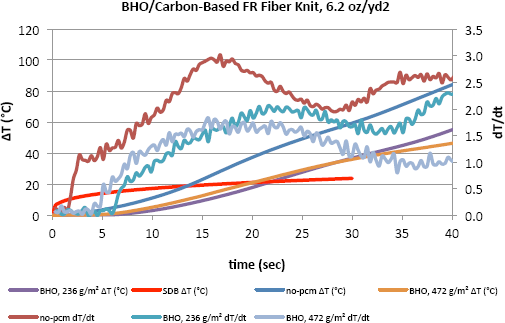
Temperature change and dT/dt vs. time of carbon-based FR fiber knit (6.2 oz/yd 2 ) fabric incorporated with BHO and tested on the TPP test set-up.
A thorough investigation, with a wider range and more levels of PCMs, is essential to clarify this relationship—an objective of our future work. Results also show that the benefit from increasing the PCM density becomes more profound with time. This is, of course, different from one fabric to another and from one PCM to another in addition to the amount of PCM used.
Conclusions and Future Work
In this work, PCMs were used with a flame resistant material for the purpose of increasing its TPP when used in firefighter gear. Results show that the inclusion of PCMs slowed down the increase in temperature on the side of the fabric close to the human body. After only 40 seconds, the PCMs helped to keep the temperature lower by 30 to 40 degrees. The results also show the need of optimizing the PCM areal density over the fabric for the sake of having lighter weight firefighter ensembles.
In our future work, we are planning to widen the base of the PCM selection. Silica encapsulation of chosen hydrated salts will be investigated with other ways for incorporating the PCMs in the flame resistant fabrics. In addition to the TPP test by open-flame only (ASTM D4108-87), an additional radiant heat source will be used at the same time (ISO 14792).