Abstract
The study evaluates the simultaneous use of aerogel and phase change material (PCM) on the face cloth of thermal liner in firefighter’s protective garment. Aerogel is commonly used to resist incoming heat flux in the field of high heat protection and to prevent the loss of body heat in the cold environment clothing. In high heat protection clothing, aerogel not only resists the incoming heat fluxes but also blocks the outbound body heat. As a result the wearer suffers from internal increase of body temperature. Previous studies identified the potential use of aerogel in firefighter’s protective clothing. However there was no clear approach to resolve the problem associated with body heat release. Current study focuses on the problem by applying PCM along with aerogel on fabric. The ambient-side of a thermal liner face cloth was coated with silica aerogel particles; meanwhile, the next to skin side was coated with PCM/aerogel composite powder. The new thermal liner revealed superior thermal protection and comfort. It extended the time to reach pain threshold and increased the pain alarm time. The Fourier transform infrared analysis of the aerogel/PCM composite powder showed the presence of PCM in nanoporous aerogel particles while the differential scanning calorimeter quantified the heat absorbing capacity of the new composite powder. Scanning electron microscope, air permeability tester, and jPOR macro of ImageJ software were used for the surface characteristics and porosity analysis of coated liner. The thermal stability of the composite powder was investigated through an infrared thermal camera. No dripping or form deterioration was observed when the composite powder was heated over a temperature three times above the melting temperature of the pure PCM.
Keywords
Introduction
Designing protective clothing for firefighters is a challenging task as it requires making a compromise between two crucial but conflicting factors, i.e. maximizing thermal protection and minimizing heat strain [1,2]. Thermal protection is undoubtedly the primary concern; however, its effects on metabolic heat stress is also a very important consideration [3]. Hence the firefighter’s protective clothing (FFPC) needs to be built with a balance of these two factors. The current study aims to enhance the thermal protection of FFPC along with a balance of comfort by using aerogel and phase change material (PCM) simultaneously.
In case of thermal protection, the use of aerogel is a growing interest and several studies reported the use of aerogel in FFPC. IUPAC defined aerogel as a gel which is composed of a porous solid where the dispersed phase is a gas [4]. It is basically an extremely light weight nanoporous material, derived from a gel by replacing its liquid component with a gas [5]. Among different types of aerogel, silica aerogel has several fascinating properties. Silica is nonflammable and thermal conductivity of silica aerogel is significantly lower than air under same condition [6]. It has high specific surface area (∼1000 m2/g), high porosity (>90%), low thermal conductivity (∼0.015 W/mK), low bulk density (∼0.1 g/cm3), and hydrophobicity [7–13]. Hence, silica aerogel has bright prospect to be used in fire protective clothing. Qi et al. [14] investigated the thermal protective performance of aerogel in FFPC by placing aerogel beside thermal liner. Shaid et al. [15] investigated the thermophysiological properties of aerogel particle coated fabric. Jin et al. [16,17] padded nonwoven fabrics with aerogel particles and used it in thermal liner. They found that aerogel can improve the thermal protection but it reduces the water vapour transmission of the thermal barrier which in turns decreases the comfort. There is no doubt that aerogel resists heat flow, which is a desirable property for FFPC from the perspective of incoming heat flux. However, from the view of internal heat release from the body, it could be a detrimental property for FFPC. The firefighters burn considerable amount of calorie during their rigorous activities, causing excessive release of body heat. The protective gear they wear is usually thick and heavy multilayered garment to provide them necessary protection in their work environment. The combination of these two facts makes them experience discomfort and extra stress. Hence, the benefits of extraordinary flame protection and heat insulation properties of aerogel can only be effectively utilized when sufficient release of body heat can be assured. There are several reports available [14–17] where aerogel was used in FFPC to enhance the thermal protection. However, to the best of our knowledge, no literature discussed on what will be the means to minimize the body heat when aerogel blocks the heat release.
A newly focused idea is to use phase change material in the FFPC. PCMs are either organic, inorganic, or eutectics [18]. However the first two types are in main interest, especially the organic PCMs. Inorganic PCMs are generally various salt hydrates, nitrates and metallic compound. These types are not suitable in current application due to their supercooling and incongruent melting behavior [19]. Organic PCMs with phase transition around 18–65℃ are suitable for thermal comfort applications in textiles [20] as they show consistency in repeated use. In this study, eicosane (C20H42) was used as phase change material. It is an organic phase change material having phase transition temperature around 37℃, which is close to human body temperature. The use of PCM for fire protection garments has been gaining interest in recent years. McCarthy et al. [21] incorporated PCM in FFPC by sewing different types of commercially available form stable PCM between the batting and face cloth of the thermal liner whereas Zhu et al. [22] quilted the same between various layers. Isabel Cardoso et al. [23] and few other researchers used microencapsulated PCM (mPCM) in FFPC to enhance the thermal protection. Rossi and Bolli [24] integrated mPCM into foam and then coated the foam on a liner. To achieve higher vapour permeability they applied the foam as a form of blister or small bubble around 3 mm diameter and in a distance of 1 mm among them. Buhler et al. [25] also used the same application process and further studied the application for different types of PCM. There are few other studies where mathematical models showed the good prospect of PCM incorporation in FFPC [26–30]. In all these studies, PCMs were used as a shield for the protection from heat which is a passive way of protection from heat by absorbing external incoming heat flux. This paper shows the application of PCM as an absorber of outbound body heat. Nanoporous aerogel particle was used as thermal protection aid and an aerogel/PCM composite powder was used to absorb metabolic heat to enhance thermal comfort. Thus the research presents a practical approach to use PCM with aerogel as to achieve added thermal protection, without sacrificing comfort.
The temperature between the thermal liner and the firefighter’s under garment can reach from 48℃ to 62℃ before receiving burn [31]. In different studies [32,33], it has been identified that the pain threshold of human skin is around 44℃. When the skin temperature exceeds this threshold, the absorbed energy determines if and how severe burns will be received [34]. Then the skin receives second degree burns when skin temperature approaches to 55℃ [21]. Thus there is a time gap between start to feel pain and to receive second degree burn. The time between the two points, i.e. the time when the skin starts to feel pain and when it receives irreversible burns, is called the pain alarm time [24]. Hence in designing protective clothing, the temperature behind the thermal liner is decisive. The lower temperature behind the thermal liner, the cooler skin temperature will be. In the current study, skin temperature of any live human subject was not investigated, rather the temperature behind the thermal liner face cloth was considered as skin temperature. The time required to raise the temperature behind thermal liner was recorded and the performance of proposed thermal liner was analyzed in regards to time delay to pain threshold and pain alarm time.
Experimental
Materials
Laboratory grade PCM (n-eicosane), heptane and ammonium sulphate were obtained from Sigma-Aldrich. Enova aerogel particles of particle size 7 to 11 µm (average pore diameter of 20 nm) was sourced from Cabot Corporation and used as received. Coating paste was purchased from Permaset, Australia [35] and used without modification. The face cloth of the thermal liner was 135 g/m2 100% nomex plain woven fabric from Bruck, Australia; moisture barrier was “Steadair3000 moisture barrier” from Stedfast; and outer layer was 220 g/m2 100% Nomex plain woven fabric, also sourced from Bruck, Australia.
Methods
Eicosane was heated to 80℃, about two times its melting point, to achieve adequate fluidity of molten PCM as to ensure better penetration into nanoporous aerogel structure. Then aerogel particles were added slowly into the hot molten PCM with continuous stirring. The mixture was stirred with a high speed stirrer at 80℃ to prevent the aggregation of aerogel particles. After two hours it was filtered with a glass-filter paper with conjunction of a suction filtering mechanism. Then the residue was washed in heptane and dried under a fume hood at room temperature until the solvent evaporates. Finally it was dried at 120℃ in a vacuum oven to evaporate the excessive eicosane on the aerogel particle surface and residue solvent. A powdery eicosane/aerogel composite was obtained.
The face cloth of thermal liner was coated with prepared eicosane/aerogel composite powder on the next to skin side and with nanoporous aerogel particles on the other side by using SV-MATIS laboratory coating machine. The coated fabric was dried at 60℃ and cured at 130℃. For comparison, another piece of fabric was coated on both sides with the binder paste that contain neither aerogel nor composite powder, dried and finally cured. The thermal liner containing aerogel particle on ambient-side and PCM composite on skin-side, was coded as “S1” and the latter one was coded as S2 (Figure 1(b)).
Test sample combination: (a) actual view of different layers and a schematic diagram of multilayer fabric structure (b) liner coatings.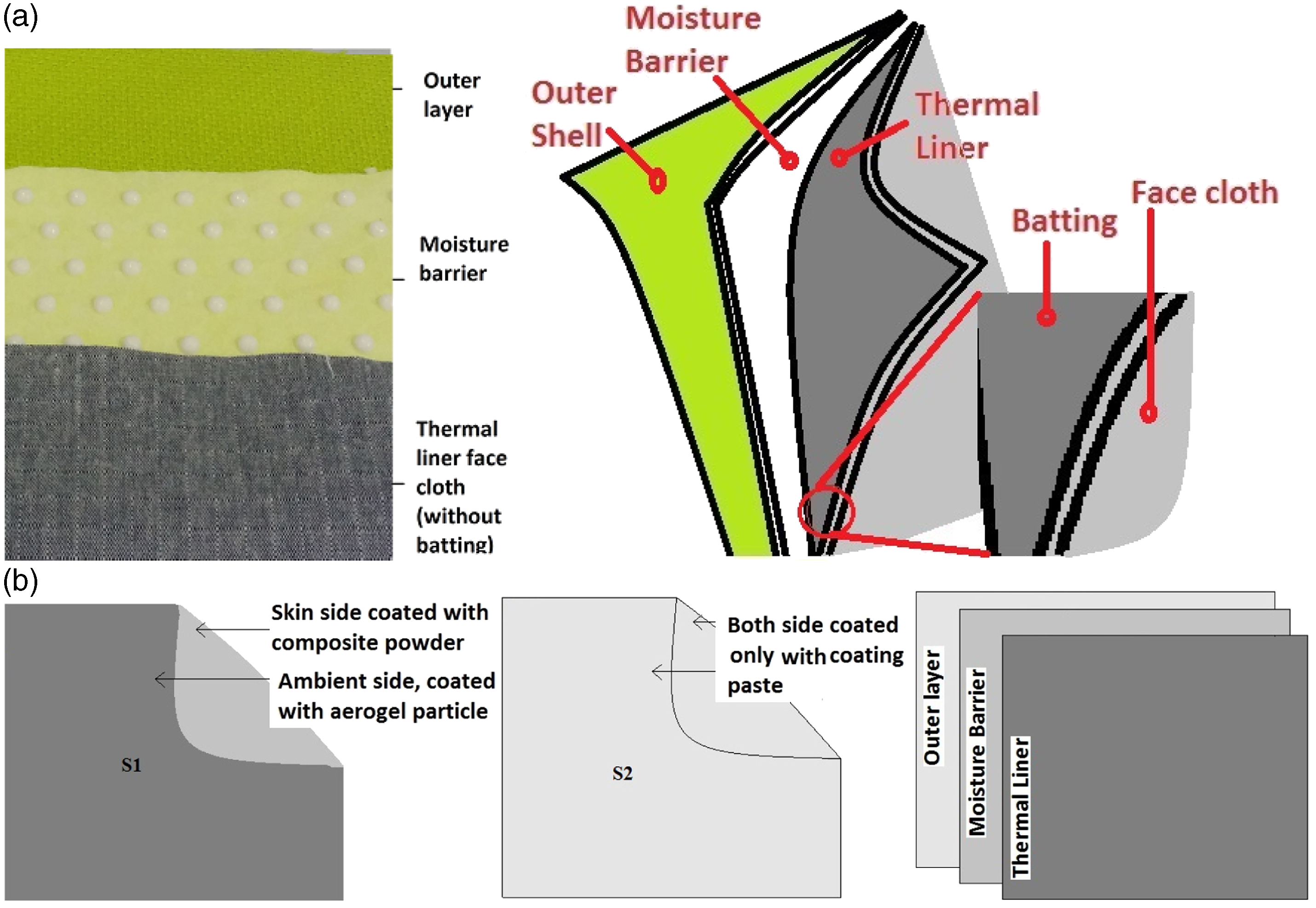
Characterization and thermal analysis
A Fourier transform infrared spectroscope (Perkin Elmer precisely Spectrum 400 FT-IR) was used for the chemical characterization of composite powder within the range of 650 cm−1 and 4000 cm−1. The total heat absorption capacity was determined with a differential scanning calorimeter (DSC). Infrared thermal imaging technique (by using a FLIR T400 IR camera) was used to observe the thermal stability of the composite powder in respect to pure PCM. A FEI Quanta200 scanning electron microscope was used to analyze the surface morphology of the coated liner. The porosity of the coated thermal liner fabric was analyzed by jPOR macro in
Finally, thermal performance of the coated fabric was investigated by a data-logger. The data logger was fitted with NI4350 high precision temperature and voltage meter (manufactured by National Instruments, Ireland) which records time–temperature curve through “VirtualBench-Logger” software. The test sample (150 mm× 100 mm) was same as firefighter’s clothing assembly which composed of an outer shell, moisture barrier and thermal liner as shown in Figure 1 where the thermal liner was prepared by joining S1 and S2 side-by-side (Figure 2). Normally a thermal liner is composed of one face cloth and one or more layers of batting material. In current study, the batting material from the thermal liner was separated to open the surface of the face cloth for coating. Hence, in the clothing assembly, only the coated face cloth was placed as thermal liner. The sample was exposed at a distance of 10 cm to a hotplate which was set at 250℃. The experimental layers were set as “Fabric-Exposed-No Space” configuration as described in NIST literature [37]. More or less similar bench-scale experimental configurations were also used previously by other researchers [21,38,39]. The heat source for the previous studies was produced by an air/natural gas fuelled radiant panel whereas in this case it was produced by a hot plate heated to 250℃. The data-logger recorded the temperature changes with the time. Sensors were placed behind each layer of the fabric assembly as shown in Figure 2.
Schematic diagram of the (a) thermocouple sensor’s position on various layers, (b) their cross-sectional view and experimental set up.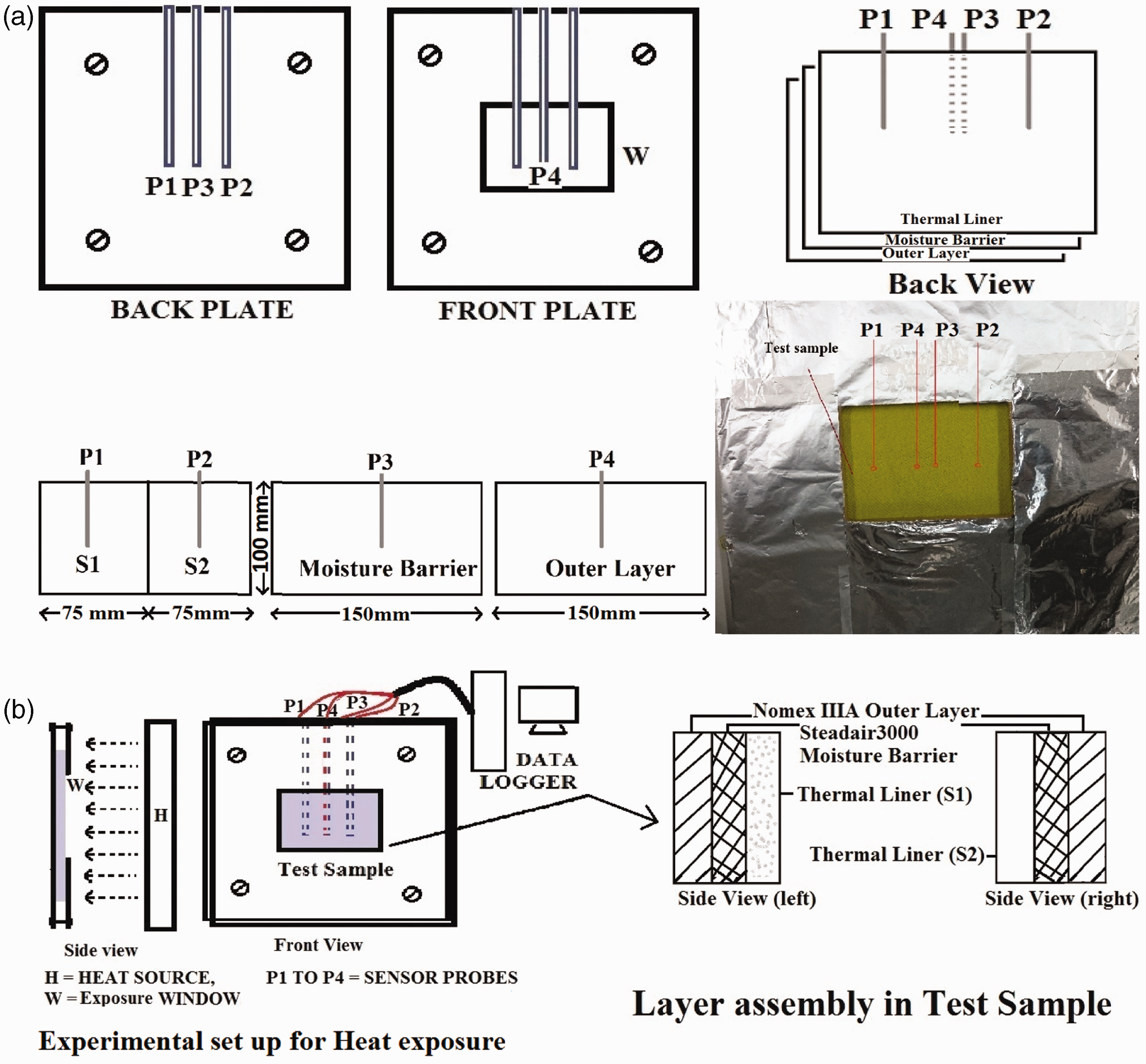
Results and discussion
The eicosane-aerogel composite powder had a calculated PCM loading of 550% on the weight of aerogel. The add-on of composite powder was around 12% on the weight of the fabric. The FTIR spectra, DSC thermo graph and form-stability of composite powder are discussed in the following sections. The thermal performance of the coated fabric is also analyzed at the end.
Characterization of composite powder
The presence of eicosane and silica structure in the composite powder was verified by comparing the FTIR spectrum (Figure 3) of pure eicosane and silica aerogel with that of the composite material.
FTIR spectra of pure eicosane, aerogel particle and eicosane-aerogel composite powder.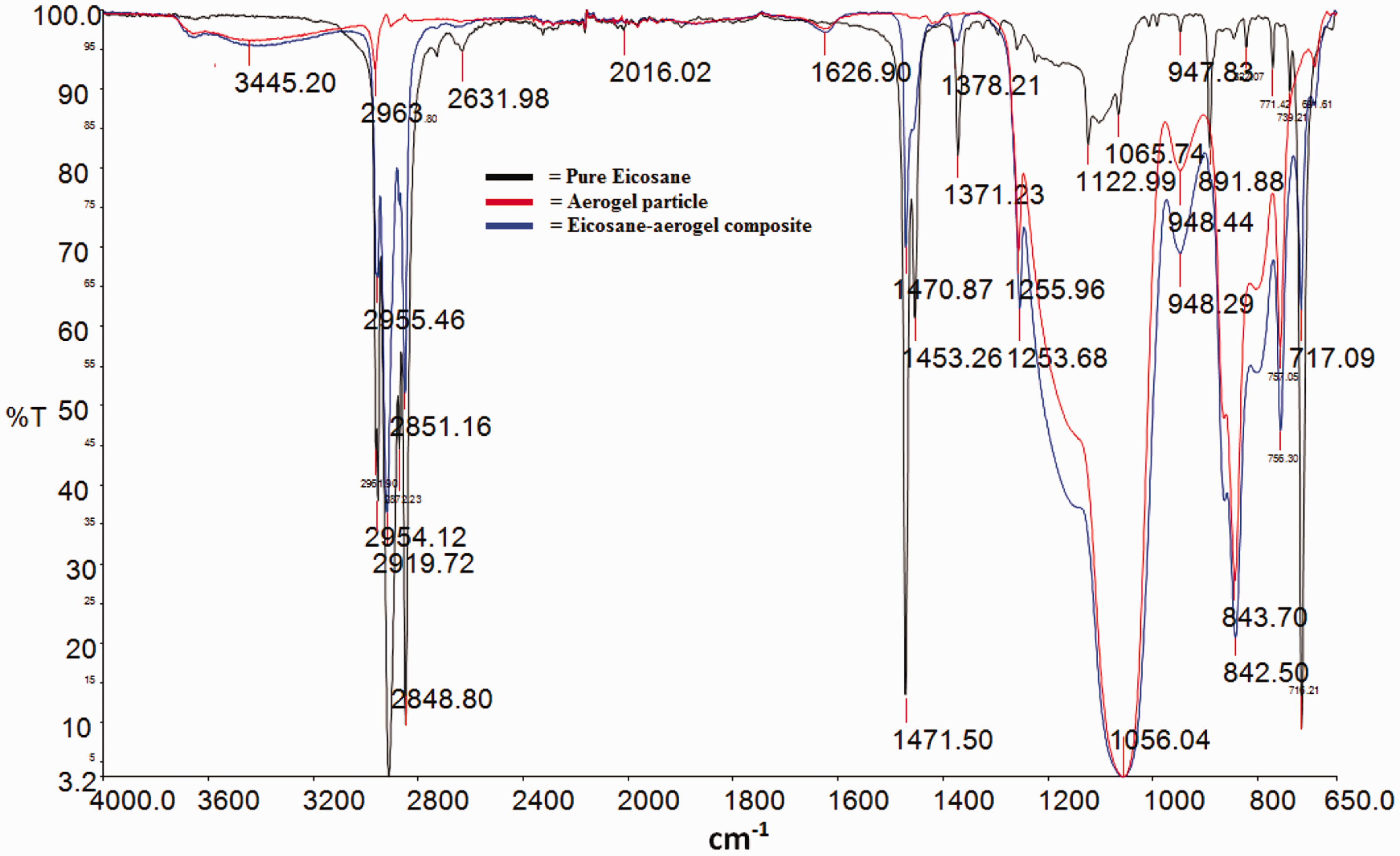
In FTIR spectra of eicosane, the peaks around 2848, 2912, 2954, and 2961 cm−1 are the stretching vibration of C–H bond [40] that presents in eicosane. The peak at 1471 cm−1 belongs to the bending/rocking vibration of –CH3. Peaks at 1371 cm−1 arose due to the characteristic bending absorption of methylene group. In addition, the peaks at 717 cm−1 is the long chain band rocking vibration of CH2 groups which is common in all alkanes [40]. The FTIR spectra of aerogel shows an intense peak around 1089 cm−1 which is the typical bending vibration of Si–O [41]. The peak at 843 cm−1 is due to the bending vibration of Si–O [41,42] and the peak at 948 cm−1 is for the stretching vibration of Si–OH [43].
For the composite, the peaks around the wave number of 2919, 2851, and 2955 cm−1 are the common peaks of eicosane with equivalent vibration. The composite also showed peaks at wave number 1056 and 842 cm−1 having corresponding vibration of pure silica aerogel. No significant new peaks were observed in composite powder. Thus it can be concluded that the composite powder is simply a physical macro-confinement and co-existence of silica aerogel and eicosane.
Analyzing thermal stability of eicosane/aerogel composite powder
The thermal image analysis of the composite powder showed (Figure 4) that the entrapped eicosane is stable even at a temperature more than three times above the melting point of pure eicosane. Aerogel does not melt at high temperature. Hence, the main concern of the current study was about the melting of eicosane which may result in the dripping out of PCM from aerogel particles. To compare the dripping behavior, the composites powder and pure eicosane were placed on a hotplate at a temperature around 120℃. Then the temperature change was recorded and thermal images were taken by a FLIR T400 Infrared thermal camera. Two observation spots were taken as “spot 1” and “spot 2” on composite powder and pure eicosane, respectively. It was found that, eicosane sample melt completely within 5 minutes while there was no visible change in composite powder. At this time, the temperature of the molten eicosane reached above 72℃ and the temperature of the composite powder was below 55℃. This temperature difference indicates the impressive thermal resistance of nanoporous aerogel particles and the evidence of the superior thermal resistance of composite powder.
Observation of the form stability of composite powder by heating at 120℃ and comparing the melting behavior with pure PCM.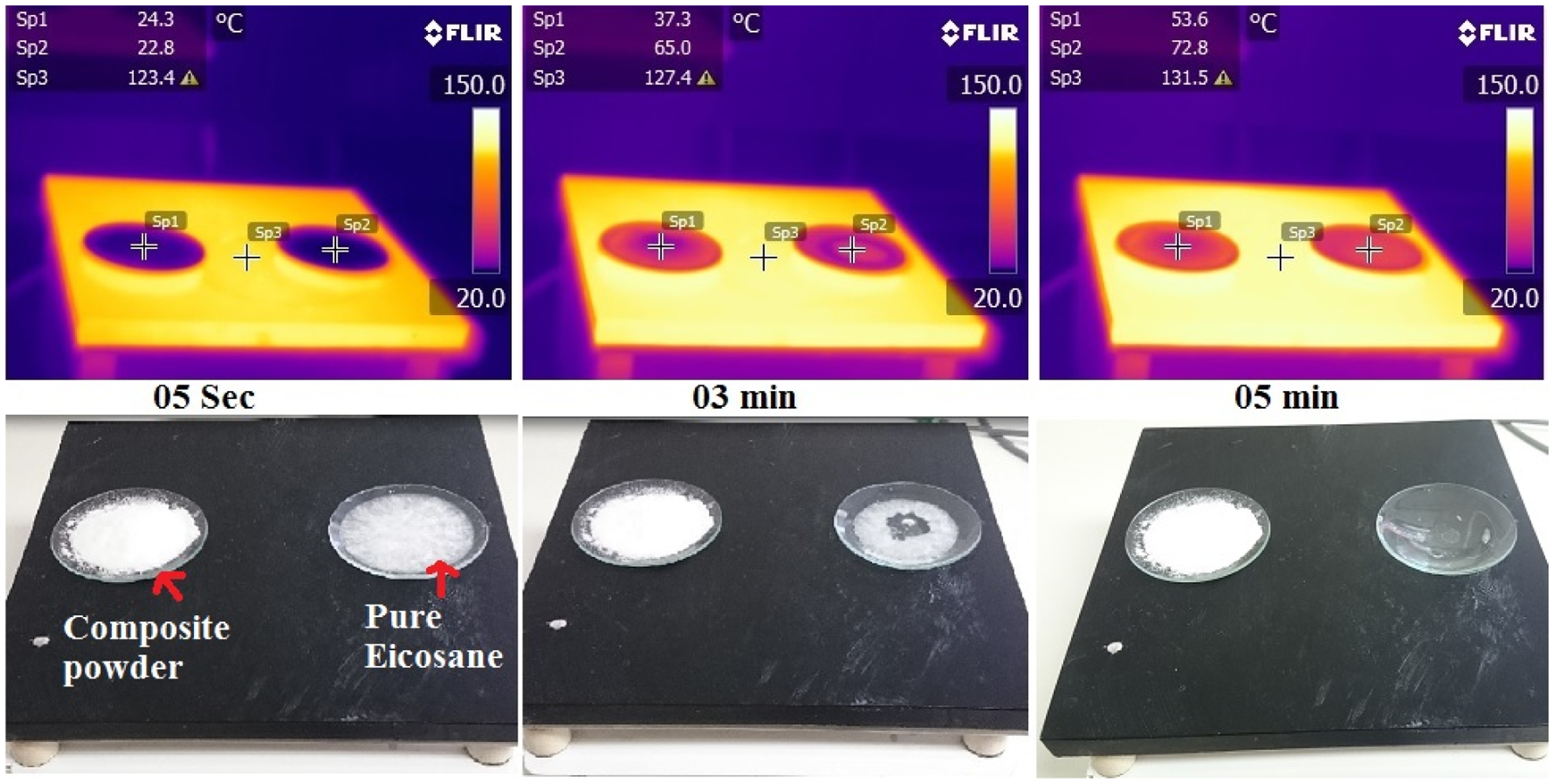
There were no sign of physical change or melt dripping in composite powder during heating experiment. Hence, it can be said that the PCM will not drip out from the applied thermal liner when it is exposed to high temperature.
One main concern of the proposed embodiment may be the completion of phase transition by the incoming external heat fluxes before the body temperature started to rise in phase transition zone. If it is the case, then it proves that some portion of external heat fluxes has to be absorbed by the PCM, which in turn will also slow down the temperature rise in skin-clothing microclimate. This slowing down of temperature in skin-clothing microclimate will again eventually help to cope with the heat stress. Hence, the simultaneous use of aerogel and PCM in firefighter protective clothing is undoubtedly capable of improving protection and comfort.
Analyzing thermal protection and comfort
The thermal protection and comfort of the thermal liner face cloth has been determined through analyzing the porosity, temperature profiles and DSC thermograph. In a complete FFPC, the face cloth of thermal liner stayed behind the moisture barrier which is wind and water proof. The face cloth S1 is expected to be impermeable to air as it was coated on both sides with aerogel and PCM that created binder films on both surfaces. The porosity measured by using jPOR macro in SEM images of coated and uncoated fabric surface showing the presence of binder film which covers the pores in fabric.
To test the thermal resistance, temperature changes behind each layer of FFPC assembly were analyzed. The time–temperature graph in Figure 6 shows that the face cloth S1 had superior thermal protection, in terms of temperature raise behind thermal liner, against incoming heat fluxes from an external heat source. The firefighter clothing assembly was exposed to a 250℃ heat source at 10 cm distance. The temperature behind the outer shell fabric (sensor P4) immediately raised above 70℃ within the first few minutes and then gradually became stable around 80℃. Temperature between moisture barrier (sensor P3) and thermal liners (sensor P1 and P2) rose comparatively at a slower rate. The temperature behind the face cloth of thermal liner increased very slowly and after 21 min 42 s it reached around 55℃ for S1 and around 65℃ for S2. Second degree burn takes place generally at 55℃. Hence, the test was stopped when the temperature behind the inner most layer reached to 55℃. Now two vital points, the time to reach pain threshold and time to receive burn, can be identified from Figure 6. Face cloth S2 took 4 min 42 s before reaching a point when the wearer would start to feel pain and took 9 min 45 s before the wearer would theoretically receive burn. This time frame was 10 min 30 s and 21 min 42 s respectively for the face cloth S1. Hence, it can be said that the face cloth S1 delayed the time to “pain threshold” for 5 min and would be able to provide extra 11 min to the wearer before receiving burn in the current experimental situation. In general, it may be argued that if the developed face cloth, i.e. S1 could actually provide extra 5 min to the firefighter before feeling thermal pain. The standard deviation of time to pain threshold was found only 0.3 min. Hence, it is certain that the face cloth S1 is capable of extending time to pain threshold and pain alarm time for firefighters. As a result, the temperature between skin-clothing microclimate will remain in comfort zone for longer duration and also the possibility of second degree burn injury will be lowered. This extended time frame or time delay to discomfort is the evidence of enhancing comfort and protection by the aerogel/PCM embedded face cloth.
Time–temperature profiles.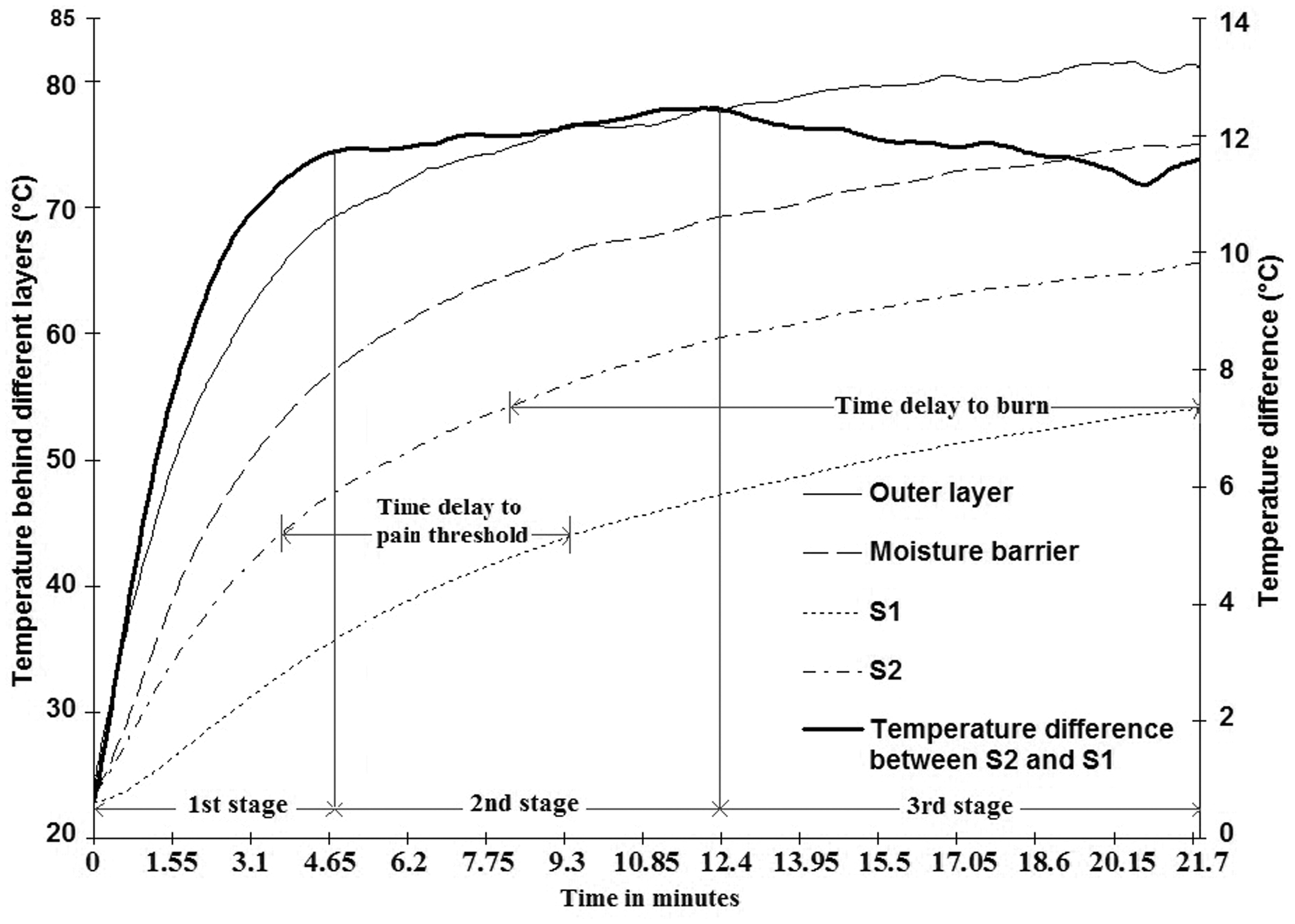
Furthermore, the phase change behavior of embedded PCM and the resultant extra time allowance in the comfort zone of wearer can be explained through the rate of temperature change. The temperature difference curve between S2 and S1 shows the rate of temperature change. The changes can be noted in three stages. In the first stage, higher rate of temperature change can be seen from a sharp upward trend in the temperature difference graph. The sharp upward trend indicates the slowest temperature increase of S1 in the melting zone of eicosane. Hence it can be assumed that the embedded PCM went through a phase change process at this stage, slowing down the temperature increase of S1 in comparison to S2. In the second stage, a relatively stable temperature difference (i.e., nearly zero rate of temperature change) between S2 and S1 was observed which is most likely due to the “near to completion” status of phase change process of the embedded PCM. Finally at the third stage when the phase changes completed, the temperature difference between S2 and S1 was minimizing, showed a down trend in the curve and rate of temperature change initiated with a reverse trend. As the average human body temperature fall in the range of 36.1–37.2℃ [44,45] and the maximum phase transition of the embedded PCM on skin-side occurs at 37–39℃, the body temperature will start to rise beyond this phase transition. However, the body temperature will not increase as long as the outbound heat fluxes will find any way to be mitigated. The embedded phase change material will act as a mitigation media by absorbing the outbound body heat. The PCM embodiment in the skin-side of face cloth offers the best opportunity to absorb outbound metabolic heat than its embodiment in any other layer. Hence, it is more likely that the embedded PCM will go for phase transition by metabolic heat fluxes and will slow down the body temperature raise.
A firefighter can produce around 300–500 W during their 15 min work [31] and Pause estimated that roughly around 40 to 60 kJ energy is required to be stored by PCM in order to provide cooling effect and to avoid heat stress over a desired period [46]. The DSC graphs in Figure 7 shows that each gram of composite powder is capable of taking 177 J of heat energy, which indicates that roughly 338 g of composite powder coating uptake will be required to provide effective cooling effect for the amount of time as described above.
DSC thermograph of the composite powder.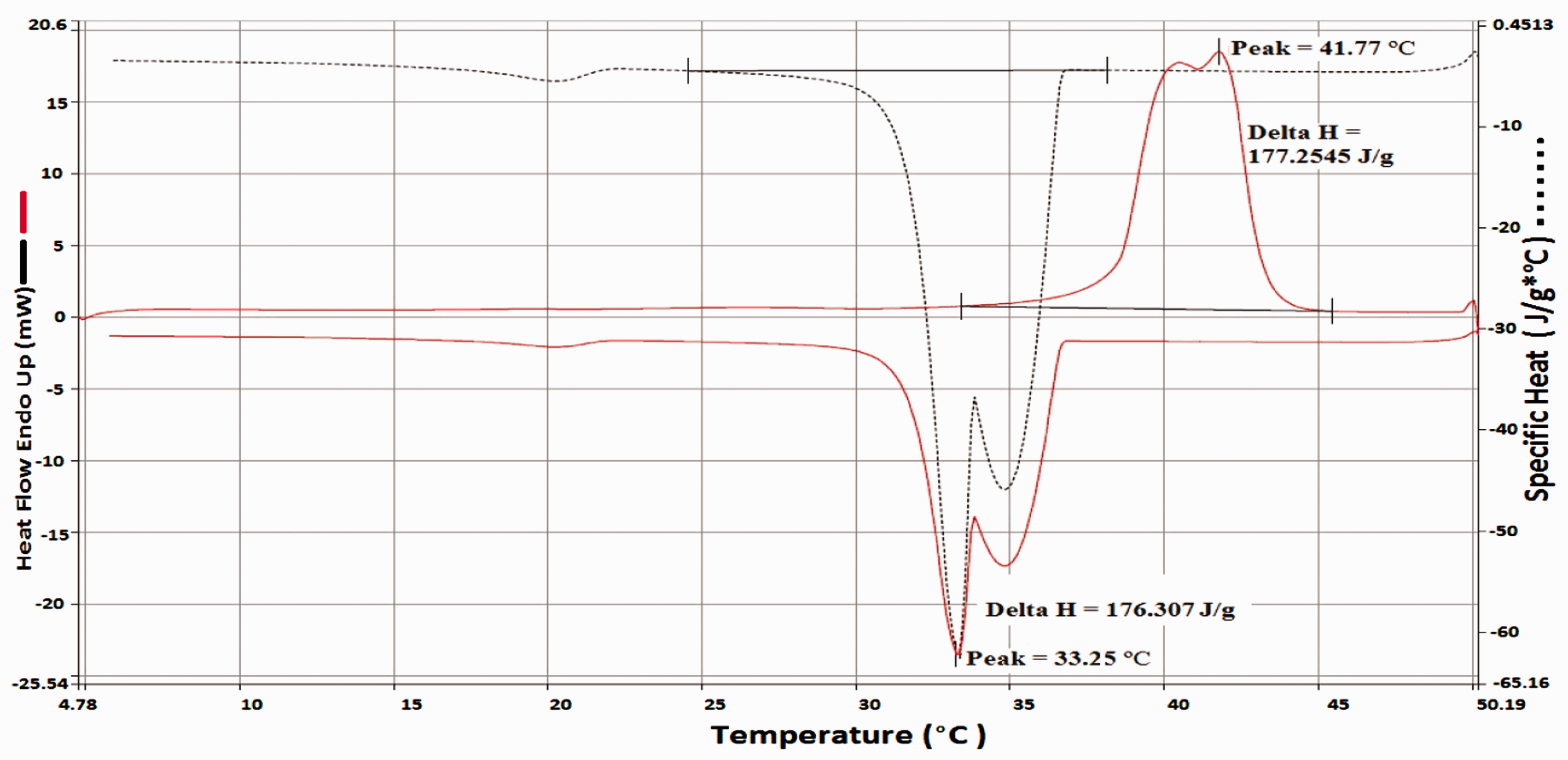
The amount of metabolic heat absorbed by the embedded PCM will mainly depend on the actual amount of PCM present to go through phase transition and on its melting enthalpy (Δ
Conclusion
The thermal protection and comfort of proposed thermal liner’s face cloth is investigated by applying nanoporous aerogel particles on the ambient side of the face cloth and composite of phase change material on the next to skin side. The aim of this study was to enhance protection against incoming external heat flux by the addition of aerogel and to overcome the body heat blockage problem of aerogel embedment through the incorporation of PCM. The study identified that the simultaneous use of PCM and aerogel can offer superior thermal protection and comfort in terms of pain threshold and pain alarm time. The PCM/aerogel composite powder was also found to be stable at elevated temperature which indicates its practicality and safe use in skin side of the face cloth. It is certain that the final performance of the proposed combination will depend on the amount of PCM for phase transition and its melting enthalpy. The enhancement of protection and comfort due to proposed combination is inevitable and proven by the current study.
Footnotes
Acknowledgement
The authors are grateful to Australian government for enabling this study through endeavour scholarship. They also like to thank Mr Martin Gregory for his outstanding support with laboratory instruments. Thanks to fellow doctoral researchers who extended their supporting hand to conduct tests. Special thanks to Mr Mac Furgusson and Mrs Siti Hana Nasir for their cooperation in coating and thermal imaging.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
