Abstract
A bi-functional cationic agent was synthesized and was applied for cationization of cotton fabric. Pad-batch and exhaustion application processes were compared and dyeing levelness, adsorption isotherm, and effect of Na2CO3 on fastness were discussed. Results showed that the bi-functional cationic agent treated fabrics showed similar dye-uptake and slightly higher fastness than fabrics treated with a commercial mono-reactive cationic agent when applied by a pad-batch process, but displayed a higher color yield when the two agents were applied in an exhaust application.
Coloration of cotton fiber with reactive dyes continues to pose environmental concerns as large quantities of water, salts, and energy are consumed. Pollution-generating colored effluent is also produced. Cationic modification of cotton to improve its dyeability with anionic dyes has received considerable attention for some time. 16 Pretreatment of cotton fibers with cationic agents was reported to enhance the uptake of anionic dyes, resulting in no salt dyeings, reduced or no alkali use, reduced washing procedure, and high fixation levels. 7 , 8
Various chemicals have been used for cationic modification of cotton fibers.9-15 The most important and widely used commercial cationic agent is the quaternary ammonium salt 3-chloro-2-hydroxypropyltrimethylammonium chloride (CHPTAC). However, this cationic agent has some difficulties when applied; it is prone to hydrolysis under alkaline conditions, resulting in waste and environmental problems and lack of affinity to the cotton fabric. CHPTAC's use is more suited to cold pad-batch application than exhaust processes.
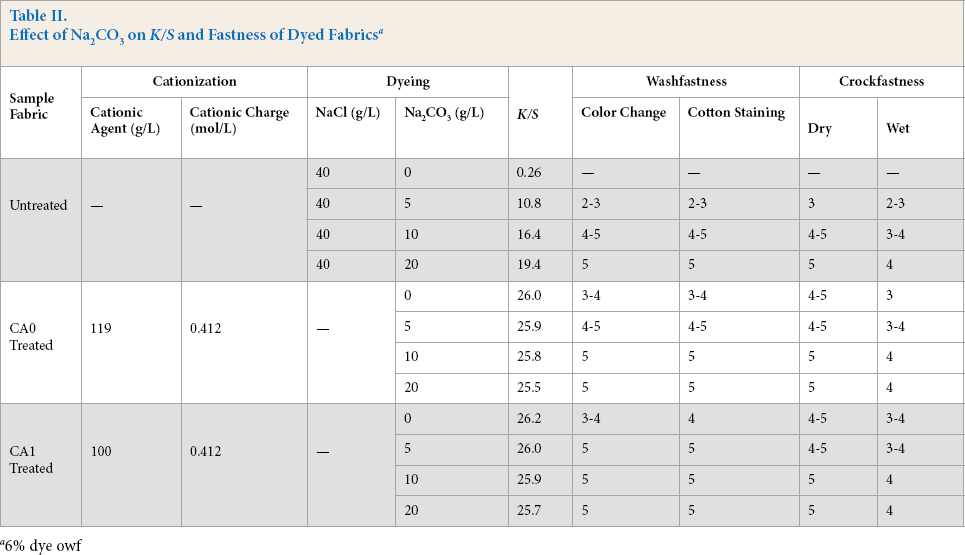
In this research, a bi-functional cationic reagent was synthesized (
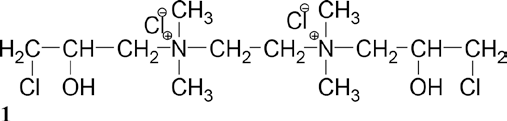
When applied under alkaline conditions, epoxide rings are formed to give
Compared with CHPTAC, this product has several advantages: 1) there are two epoxide groups, which improve the reaction efficiency of cotton fiber binding, 2) the two quaternary ammonium cationic dye sites not only can enhance dye uptake but may also improve colorfastness properties, and 3) the larger molecular structure may allow for more efficient exhaustion.
Experimental
Materials
A scoured and bleached plain woven, 100% cotton fabric with a weight of 124 g/m2 was used. The commercial cationic agent, CR2000 (3-chloro-2-hydroxypropyltrimeth-ylammonium chloride, CA0), available as a 65% solution in water, was supplied by Dow Chemical and used as received.
Synthesis of Bi-functional Cationic Agent
Under an argon blanket and ice-water bath cooling, TEMED was neutralized by conc. HCl, maintaining the temperature below 20 °C. After the addition, the reaction mixture was stirred continually until the reaction system had a neutral pH value. Epichlorohydrin was then added slowly with continuous stirring; the temperature was maintained at 40 ± 5 °C and the reaction mixture was stirred for 10-12 h.
The mole ratio of TEMED: HCl: epichlorohydrin was 1:2:2. The bi-functional cationic agent (CA1) was a yellowish, viscous liquid with 77.2% solid content. 16 , 17
Cationization
Pad-batch Application
The pad-batch application process was carried out on a Mathis HVF Lab Padder.
CA0 and CA1 were applied using respective stoichiometric sodium hydroxide concentrations. 18 To minimize reactant hydrolysis, alkali was added to the bath just prior to application. The fabrics were padded through the cationization baths at 100% wet pick-up, then wrapped in plastic, stored, and rotated at room temperature (RT) for 24 h. After removal from the plastic, the batched fabrics were rinsed several times with warm water at 40 °C, cold rinsed until the final fabric had a neutral pH, and then dried.
Exhaustion Application
The exhaustion process was carried out on a Mathis Labor Jumbo Jet Machine. Cotton fabric was treated with the CA0 or CA1 using a 10:1 liquor ratio (LR) at RT for 10 min, NaOH (50%) was added, the bath temperature was then gradually increased at 4 °C/min to 60 °C and held at 60 °C for 30 min. The bath was then cooled and the fabric was rinsed with 40 °C fresh water until the final fabric had a neutral pH, and then dried.
Dyeing Procedures
The dyeing process was carried on the Ahiba Texomat Labor Dyeing Machine at a 50:1 LR.
Dyeing of Untreated Cotton
Dyebaths were prepared and untreated fabrics were added at 28 °C. 40 g/L NaCl was added after 15 min and 10 g/L Na2CO3 was added separately in two equal amounts. The first half was added after 30 min. The bath temperature was gradually increased at 4 °C/min to 60 °C and a second amount of Na2CO3 was added. The bath was held at 60 °C for 30 min, then cooled and discarded. The fabrics were rinsed three times with tap water and twice with 50 °C water, then scoured in a bath containing 2 g/L scouring agent and Na2CO3 at 90 °C for 10 min, rinsed with water until a clear rinse was obtained, and allowed to air dry.
Dyeing of Cationic Cotton
Dyebaths were prepared and cationic fabrics were added at 28 °C. After dyeing at 28 °C for 30 min, the bath was gradually increased at 4 °C/min to 60 °C. The Na2CO3 was added (or not) and dyeing continued at 60 °C for 30 min (or for a certain period), then cooled and discarded. The fabrics were rinsed three times with tap water, once with 80 °C water for 10 min, and twice with tap water before drying.
Color Assessment
The whiteness of the cationized fabrics and
Colorfastness properties of the dyed fabrics were determined using AATCC Test Methods 61-2010 (2A) for washfastness and 8-2007 for crockfastness.
Results and Discussion
Effect of Pad-batch Cationization of Cotton Fabrics
Fabrics were treated with CA0 and CA1 using a pad-batch process and dyed as indicated in Table I. For comparison purposes, the corresponding concentrations of CA0 and CA1 were converted to the same molar concentration of cationic charge.
The whiteness of cationized fabrics was observed to decrease with the increase in CA concentration because the amino-functional group discolors with heat and aging. However, this degree of yellowing from cationic agent use can be accepted in practice.
The effect of cationic agent concentration on color yield was compared to the traditional dyeing process. The
Cationization Conditions and Whiteness of Samples
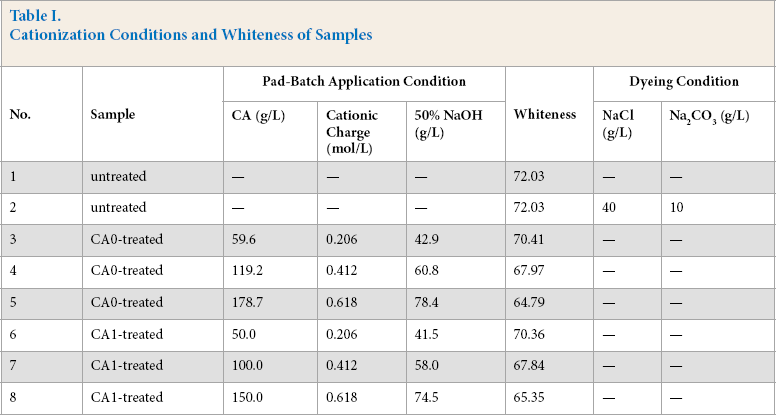
It can be seen from Fig. 1 that the Black B dye had almost no affinity to the cotton fabric without salt and alkali. A large increase occurred in
Dyeing Levelness of Cationized Cotton Fabrics
There were no levelness problems with excess dye application because all dye sites were occupied. However, excess cotton fabric cationization may result in a higher risk of dyeing unlevelness. Unlike dyeing nylon fabric with acid dyes, where the dye sites available can be controlled with pH adjustment or slow temperature gradients, the cationic sites of cationized cotton are permanent and cannot be controlled through pH or temperature manipulation. As a result, such immediate availability of cationic sites makes leveling cationized cotton very difficult. So it is necessary to use a retarder—such as carboxymethylcellulose (CMC)—that slows down the strike of the dye when excessive dye is not applied. 19 In practice, the dye depth should be matched with the degree of cationization.
Dyeing Kinetics and Thermodynamics Studies
Dye-fabric reaction rate can be expressed as the change in
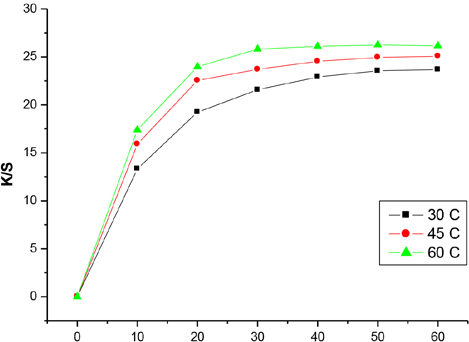
Effect of dyeing time and temperature on
As Fig. 2 shows, color strength
Sorption isotherms of the reactive dye on the cationic-pretreated cotton fabrics can be described by the Langmuir isotherm model. 12 Reactive anionic dye is quickly attracted to the positive sites on the cationized cotton before monolayer coverage is attained at equilibrium. Higher temperature promotes dye diffusion and reaction rate, but longer dyeing time at higher temperature may result in some desorption.
Effect of Na2CO3 on K/S Values and Fastness
For dyeing untreated cotton fabrics, addition of 10-20 g/L Na2CO3 is necessary to obtain reasonable fastness. In cationized cotton fiber, the positive charges act as dye sites, which can be dyed readily with anionic dyes by virtue of ion-ion interactions. Nevertheless, in the case of reactive dyeing, colorfastness may be affected to some degree, regardless of whether alkali is added or not. 19
It can be seen from Table II that increased Na2CO3 concentration greatly increased the
Effect of Cationization by Exhaustion
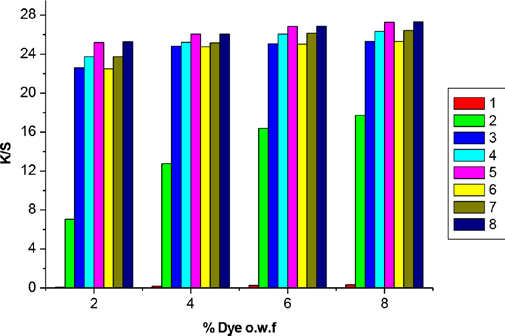
In this research, the exhaustion cationization process was used with CA0 and CA1, and dyeing properties of the cat-ionized fabrics were compared as shown in Fig. 3.
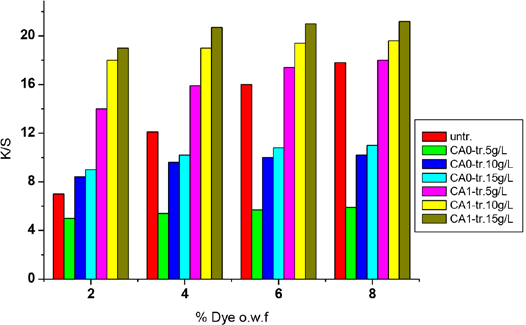
Both cationic treated fabrics showed an increase of
CA1 had more affinity to cotton, so it was exhausted more easily. On the other hand, CHPTAC (CA0) had only one epoxy reactive group that can react with -OH on cellulose macromolecules to form ether linkages. The desired reaction was accompanied by the side reaction of cationic agent with water, especially at higher temperature and alkali conditions, and lowered the efficiency of the treatment process. The bi-functional cationic agent (CA1) had two reactive groups: if one of the reactive groups was hydrolyzed during application, the other group would still react. Therefore, CA1 has a greater ability to resist hydrolysis and improved utilization efficiency in exhaustion processes when compared to CA0's properties.
Conclusion
CA1-treated fabrics showed Langmuir isotherm sorption when dyed with Remazol Black B Dye and had similar dye-uptake, and slightly higher fastness, than that of commercial cationic agent when applied by the pad-batch process, but gave much higher dye color yield values when used in exhaust application.
The dye depth and degree of cationization should be matched (or an appropriate amount of a retardant should be used) for level dyeing. Use of a low concentration of Na2CO3 can significantly improve the washfastness of dyed cationized fabrics.