Abstract
Dyeing of cellulosic materials with reactive and direct dyes is commercially very popular, but is not eco-friendly On the other hand, pigment colors cannot be applied by conventional exhaust methods. These problems can be solved or minimized by increasing the affinity of cellulosic materials. Cationization of cellulosic fibers improves affinity towards anionic dyes like reactive dyes, direct dyes, and pigment colors dispersed with anionic dispersants. This review paper discusses various studies relating to cationization of cotton, linen, and ramie fibers using various chemicals (e.g., quaternary ammonium compounds, chitosan, CHPTAC, other epoxy compounds, and triazine) as well as non-reactive pretreatments with cationic polymers.
Keywords
Cotton Cationization
At the present time, dyeing cotton fabrics with direct and reactive dyes is a very energy- and water-intensive, pollution-generating process. Most commercially available direct and reactive dyes have only a moderate affinity for cotton. Cotton fibers build up negative surface charges in water and these charges act to repel anionic dyes and retard exhaustion. This lack of affinity is currently overcome by the use of high concentrations of electrolytes in the dyebath (such as sodium chloride and sodium sulfate) resulting in serious environmental problems when disposing the dyebath effluent and by the use of extended dyeing times at elevated temperatures. The high concentration of electrolytes serves a dual role, overcoming the negative charges built up on the cotton fiber and reducing dye solubility.
Cotton pretreatment before dyeing can offer a simple and effective method of improving dye-fiber affinity, avoiding the need for salt in the dyebath. Cationizing cotton fiber increases the substantivity of anionic dyes due to presence of positive charges imparted to the fiber. Cationization is the chemical modification of cotton to produce cationic (positively charged) dyeing sites in place of existing hydroxyl (-OH) sites. 1 , 2 Because protein fibers like wool and silk were known to have good dyeability, efforts were made, as early as the mid-1800s, to animalize cotton by depositing protein material on it to make it more dyeable. Treatment of cellulose with cationic, nucleophilic polymers also enables reactive dyeing at neutral pH without electrolyte addition. Dyeing cationic cotton results in greater use of dye and higher color values. In addition, the strong dye-fiber interactions resulting from cationizing cotton allow dyeing with no added electrolytes and minimal rinsing and afterwashing. However, a fiber with great dye-attracting properties may continue to exhibit those attributes in later use and become scavengers during laundering.
Lei and Lewis
3
,
4
reviewed numerous chemicals that can be used to provide cationic sites in cotton fiber, namely glycidyl trimethylammonium chloride,
Chemical modification of cotton by etherification, esterification, grafting, and crosslinking reactions is generally performed by reaction with functional groups (hydroxy groups) present in the fiber. These chemical modifications of cotton are subjects of extensive studies. 5 Lewis and McIlroy 6 explain the effect of chemical modification on subsequent dyeing processes. Some developments in cotton cationization to improve dyeability towards anionic dyes are reviewed by others as well. 7 By introduction of negative groups such as carboxy groups through cellulose carboxymethylation, the uptake of direct dyes decreases in proportion to carboxymethyl-group content. 8 On the other hand, exhaustion of anionic dyes, including reactive, direct, and acid dyes, improves greatly by cotton cationization. The most common approach is via reaction of various types of fiber-reactive substituted amino compounds with cotton. By introducing amino groups, the cellulosic fiber is cationized, resulting in high substantivity for anionic dyes due to the Coulombic attraction between the positive charge on the fiber and the negative charge on the anionic dyes. Such cationized cotton would be dyeable with reactive dyes under neutral or mildly acidic conditions in the absence of electrolyte and alkali in the dyebath.
Cationized cotton has been available for many years and is being used commercially in a number of mills for dyeing yarns, knits, wovens, and garments. However, implementation remains at a relatively low level, for various reasons: 9
Low fixation of cationic agents by exhaust methods can increase chemical and processing costs.
Cationic cotton requires some modification of existing dyeing procedures.
Production of seconds may be high until a mill has substantial experience with the process.
Dye selection is critical, as some dyes work better with cationic cotton than others. Selection can be limited by requirements for color, depth of shade, and fastness (especially lightfastness).
For safety reasons, the cationic agent should be applied in a closed system.
Cationized cotton has a very high ecological impact—the amount of chemicals required to dye cotton textiles can potentially be reduced by as much as 50%. Further research and development continues to refine the application process and improve product quality.
One study deals with modifying the chemical structure of the cellulosic cotton fabrics by introducing amino groups as new active centers. 10 The properties of the resulting aminated cotton, dimethylaminoethyl (DMAE)-cotton, are evaluated and discussed by studying the dyeability, lightfastness characteristics, and fading kinetics. The modification succeeds in saving energy by decreasing both the dyeing time and temperature, and the dyeability and lightfastness characteristics are improved.
Cationization is superficial since X-ray diffraction shows that it has no effect on cotton crystalline structure. 11 Dyeing of cationized cotton without salt indicates that reactive group type does not affect the dye yield on cationized cotton and colorfastness is not changed. 12
Quaternary Ammonium Compounds Treatments
Many cationic treatments make use of quaternary amine residues. Pretreatment agents of this type include glycidyltrimethylammonium chloride (Glytac A) and its epichlorohydrin analogue. 13 Unfortunately, these compounds are characterized by low substantivity, an unpleasant odor, and poor thermal stability during application—to some extent this has restricted their use. 14 To avoid these disadvantages, polymeric derivatives of Glytac-type agents were developed for cotton cationization.
In another study, a new fiber-reactive quaternary compound containing an acrylamide residue, 1-acrylamido-2-hydroxy-3-trimethylammoniumpropane chloride (AAHTAPC), is synthesized by reacting an α-aminoalkyl-ω-quaternaryamine compound with acryloyl chloride. It is applied to cotton fabrics via pad-bake under alkaline conditions. The treated fiber is dyed with reactive dyes without any other dyebath additions such as salt or alkali. Reactive dyes are exhausted almost entirely onto the treated cellulosic fiber. The color yield is better than that on untreated cotton by the conventional method. The shades are bright, level, and gave good washfastness and lightfastness. 15
Five cationizing agents are synthesized, through the reaction of various trialkylamines with epichlorohydrin, and then used for cellulosic fiber cationization. Cationized cellulosic fibers are dyed with acid dyes and show excellent antimicrobial activity, in spite of the low degree of cationization. 16
Cotton can be pretreated with a commercial cationic agent Sintegal V7conc (Chemapol), which is a quaternized polyglykol ether of fatty amine with cationic character. 17 Dyeing of cationized fabrics using reactive dyes, in the absence of salts, increases color intensities progressively with the increase of cationic agent concentration. Dye exhaustion is increased due to the ionic attraction of Sintegal V7conc cationic groups and reactive dye anionic groups. Compared with the original fabric, cationized samples have higher color uniformity and standard fastness level, indicating that surfactant cationic groups have an importance for reactive dye absorption and that dye fixation is realized by covalent bonding with cotton hydroxyl groups, similar to standard dyeing.
Health Hazards
Quaternary ammonium compounds can display a range of health effects, among which are mild skin and respiratory irritation up to severe caustic burns on skin and gastrointestinal lining (depending on concentration), gastrointestinal symptoms (e.g., nausea and vomiting), coma, convulsions, hypotension, and death. 18
Quaternary ammonium groups are thought to be responsible for anaphylactic reactions that occur with use of neuromuscular blocking drugs during general anesthesia in surgery 19 Quaternium-15 is the single most often found cause of allergic contact dermatitis of the hands (16.5% in 959 cases). 20
Chitosan Derivatives
Chitosan is a linear polysaccharide composed of randomly distributed β-(1-4)-linked D- glucosamine (deacetylated unit) and
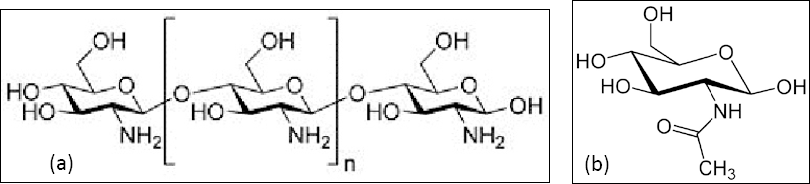
Chemical structure of chitosan (a) D-glucosamine (b)
(3-Chloro-2-hydroxypropyl)-trimethylammonium chloride (CHPTAC)
A typical cationization process involves the reaction of CHPTAC with alkali to form 2,3-epoxypropyltrimethylammonium chloride (EPTAC, C6H14NOCl, Fig. 2), which then reacts with cellulose to form cationic cotton. As EPTAC is a non-volatile organic salt handled in aqueous solutions, inhalation exposure to this chemical does not occur. Since EPTAC is a genotoxic carcinogen, this property alone is sufficient to lead to the strictest measures for risk management in work places. The environmental risk assessment concludes that there is concern for the aquatic ecosystem (including marine environment) from exposure arising from cationization of starch. There is also concern for humans exposed via the environment with regard to carcinogenicity and mutagenicity, however the risks are very low. For consumers there is no concern. 22
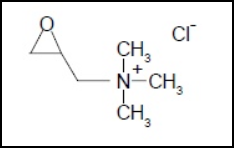
Chemical structure of EPTAC
Chemical modification of cotton fabric via cationization with CHPTAC is studied by Hashem, Smith, and Hauser. 23 The reaction is carried out in a strong alkaline medium (usually sodium hydroxide) either by the exhaustion method at high temperature or by the cold pad-batch method. The quaternary ammonium group ((CH3)3N+), has a persistent positive charge and can thereby lead to the formation of ionic bonds (salt linkages) with negatively charged anionic groups, such as those found in a wide array of anionic dye classes or carboxy-containing compounds. The former imparts no-salt dyeing properties for cotton fabric. Moreover, the presence of cationized groups in the cellulose also imparts anti-microbial properties to the cotton fabric.
Reaction of CHPTAC with cotton cellulose is the focus of a number of studies. 24 The reaction mechanisms are summarized in Eq. 1–3.
The chlorohydrin form of the reagent is converted to the epoxy intermediate EPTAC; Eq. 1);
The epoxy reacts with the cellulose itself (Eq. 2);
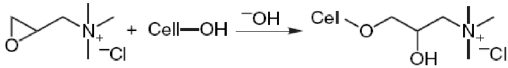
epoxy is converted via hydrolysis to the nonreactive 2,3-dihydroxy derivatives (Eq. 3).
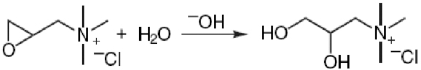
Considering these reaction schemes, the magnitude of cationization of cotton fabric relies on the sodium hydroxide concentration, reaction temperature, reaction time, solvent used, and liquor to goods ratio, as well as the method of application.
Montazer, Malek, and Rahimi 12 cationized cotton with CHPTAC by a pad-batch process. Cationized cotton samples are dyed with reactive dyes containing various reactive groups. Dyeability of the cationized cotton samples with reactive dyes without salt is significantly improved due to an increase in the ionic attraction between the dye and cationized cotton. Results show that the wash and dry crockfastness of cationized cotton dyed with reactive dyes are similar to those of the untreated cotton. However, the lightfastness of some cationized fabric samples is improved.
Hashem reports the possibility of one-stage preparation and cationization. 25 A one-stage process for cationization with CHPTAC and pretreatment of cotton involves enzymatic and oxidative desizing, scouring, bleaching with hydrogen peroxide, and combined desizing-scouring, as well as combined desizing-scouring-bleaching. Each of these pretreatment operations or their combination, with and without cationization, is carried out using three techniques, namely, cold pad-batch, pad-steam, and exhaustion. The cationization chemicals are compatible with the scouring ingredients and, to a lesser extent, with the formulation of enzymatic desizing. On the other hand, cationization chemicals are not compatible with oxidative desizing or peroxide bleaching ingredients. Efficiency of the one-step process for scouring and cationization is maximized through detailed investigation of the process parameters. The treated fabric can be dyed without electrolyte and the fabrics show a high levelness of color shade, residual wax content, and nitrogen content. 25 For the exhaustion method, the greatest efficiency of scouring and cationization could be attained when the desized cotton fabric is treated with an aqueous bath containing sodium hydroxide (5 g/L) and CHPTAC (10 g/L) using a liquor to goods ratio of 20:1 at 80 °C for 90 min. In the cold pad-batch technique, maximum scouring and cationization efficiency is observed at CHPTAC and sodium hydroxide concentrations of 100 g/L and 49.3 g/L, respectively. The cold pad-batch technique gives a higher fixation percentage (50%) compared with the exhaustion technique (25%), whereas the scouring efficiency is comparable in the two cases. Treated fabric can be dyed without electrolyte and the fabrics show levelness of dye shade, nitrogen content, and residual wax content. 26
Ramasamy and Kandasaamy 27 report the influence of process variables on the cationization efficiency of two cationic agents: CHPTAC and a polyamino chlorohydrin quaternary ammonium compound (Cibafix WFF). Cationization increases dye use by 30% with no change in colorfastness and reduces environmental pollution to a great extent. Cationized cotton dyeing consumes only 40% of the steam and 50% of the time required by normal dyeing. The effluent of cationized cotton contains less pollution load than that of conventional dyeing as no salt and soda is added during cationized cotton dyeing. The most beneficial aspect of the cationization technique is the reduction of total dissolved solid (TDS) in the effluent as TDS cannot be removed from the effluent easily. A similar study with Cibafix WFF was also made by Subramanian, et al. 28
Other Epoxy Compounds
Two epoxy reagents,
Triazine Derivatives
Cellulose is chemically modified with triazine derivatives containing cationic and anionic groups. Dyeing and diffusion properties of modified cellulose are discussed. The exhaustion and fixation of reactive dyes on modified cellulose are greater than those on unmodified cellulose.
Compared with unmodified cellulose, the dyed modified cellulose also possesses good colorfastness to washing. The diffusion coefficients of dyes at different temperatures are calculated. Compared with unmodified cellulose, the diffusion of dyes in the modified cellulose shows significant change. 30
Cationic Agent CA200
Cotton fabric is treated with reactive cationic agent CA200 at five different concentrations (1-5% w⁄w) using the exhaust method at 40 °C and at a liquor ratio (LR) of 50:1 without the addition of salt. After 1 h, sodium carbonate (20 g/L) is added in portions, while the temperature is increased to 80 °C over 30 min. The fabric is neutralized with 2% w⁄w aqueous acetic acid for 5 min at 40 °C, and then rinsed in tap water and dried at room temperature (RT). The cationized fabric is dyed with vinyl sulfone based reactive dyes (e.g., Remazol Blue R and Remazol Red RB, Dystar) without salt. Cotton pretreated with CA200 displays an improved rate of dyeing (even in the absence of salt when compared with conventional dyeing in the presence of salt). Dyeing on pretreated cotton exhibits good properties of wetfastness and lightfastness. Because of the improved dye substantivity caused by the introduction of cationic groups into cotton, cationization enhances the color strength. In terms of chemical oxygen demand (COD), biochemical oxygen demand (BOD), and TDS of the dyeing effluent, the degree of pollution is reduced after cationization. 31
Non-reactive Pretreatments
Some polymers with affinity for cellulose tend to be desorbed during dyeing and inhibit uptake of the dye or cause it to precipitate. 32 Recent work has established the value of polymeric quaternary ammonium compounds, amines, or amides, which may be attached to cotton by non-chemical mechanisms. 33 Despite the encouraging results obtained with nonreactive polymers, in the salt-free dyeing of cotton, problems remain in dye selection and in obtaining a level result. 34
Cationic Polymers PT1 and PT2
Two cationic polymers (Fig. 3) were studied by Blackburn and Burkinshaw: a copolymer of diallyldimethylammonium chloride and 3-aminoprop-1-ene (PT1) and a copolymer of 4-vinylpyridine quaternized with 1-amino-2-chloroethane (PT2). 33 Both PT1 and PT2 pre-treatment polymers are highly substantive to cellulosic fiber. The anionic carboxylic acid groups in the substrate, because of their relatively low pKa values, are ionized at the pH values of the application (pH 6-7). Ion-ion interactions occur between cationic groups in the agent and the anionic carboxylic acid groups in the substrate. The other forces of attraction involve hydrogen bonding and Van der Waals forces.
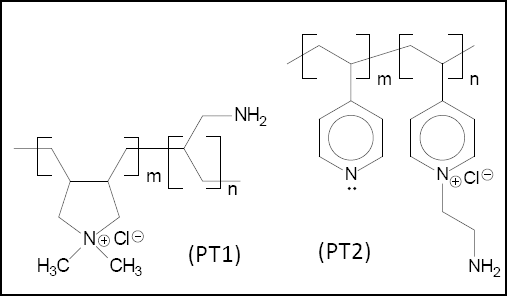
Chemical structures of cationic polymers, PT1 and PT2.
The ionic interaction between positively charged nitrogen of PT1 and hydroxyl group of cellulose is shown in Fig. 4. In case of PT2, the interactions may be:
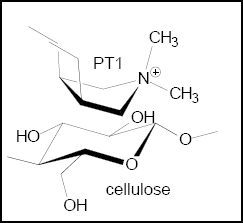
Interaction of PT1 and cellulose
Ion-dipole interactions between cellulose hydroxyl groups and pyridinium residues of PT2 or;
Yoshida H-bonding between cellulose hydroxyl groups and pyridine residues in PT2
Little or no salt is required during dyeing with reactive dyes. Nucleophiles in the polymer are more reactive than hydroxyl groups in the fiber. Consequently:
Application may be done at neutral pH
Hydrolysis of dyes is minimized
Color fixation is maximized
Less color occurs in effluent
Less wash-off is required
Significant reduction in operation time is observed
Significant reduction in water consumption is observed.
Poly(vinylamine chloride) Polymer
Ma, et al, investigated the pretreatment of cotton with poly(vinylamine chloride) (PVAC, Fig. 5) for the salt-free dyeing of cotton with reactive dyes. 35 The presence of a large number of cationic sites involving primary amino groups (H3N+) within PVAC molecule are of particular value for achieving salt-free dyeing of cotton with reactive dyes. As the pH increases, the proportion of H3N+ groups in the molecule decreases and that of H2N groups increases.
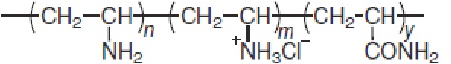
Chemical structure of poly(vinylamine chloride).
A pad-bake method is used for cotton pretreatment with PVAC. In most cases, 5 g/L PVAC is padded on to the cotton fabric to give a wet pickup of 80%. The pH of the pretreatment liquor is maintained at 7 using a buffer comprised of potassium dihydrogen phosphate (7 g/L) and sodium hydroxide (1.39 g/L). The fabric samples are pre-dried at RT and then baked at 100 °C for 10 min in a rapid baker. The PVAC-treated cotton fabric is dyed with cold brand, hot brand, and vinyl sulfone using respective standard dyeing procedures. When compared with reactive dyes conventionally dyed in the presence of salts, cotton pretreated with PVAC displays improved reactivity and fixation for most reactive dyes and are dyed without salt. The application of PVAC is straightforward and uniform dyeing is easily achieved using the prad/air dry/bake method for pretreatment. Typical Langmuir-type adsorption is obtained on cotton pretreated with PVAC, confirming its effectiveness at lower concentrations. The reactive dyes penetrate the fiber thoroughly. Dyeings on pretreated cotton exhibit excellent washfastness and good crockfastness. Residual dye in the effluent is markedly reduced, confirming the commercial potential of PVAC as a pretreatment chemical for cotton.
Pre-treatment with Cationic Starch
About 15% of reactive dyes worldwide are currently applied by continuous dyeing. In continuous dyeing, reactive dyes are directly padded onto cotton by means of a pad mangle. This avoids the high concentrations of salts (40-100 g/L) required to overcome dye–fiber repulsion in exhaust dyeing of reactive dyes. A certain amount of salt (10-50 g/L) is however still needed to reduce migration. It was previously reported that higher salt concentrations are more effective for this purpose. 36 A cationic starch is prepared using glycidyltrimethylammonium chloride (Glytac A) and its effect on the dyeing of cotton is investigated using a series of eight reactive dyes. Pretreatment of cotton with cationic starch improves the level of dye fixation of reactive dyes applied by continuous dyeing. Better results are obtained at higher degrees of substitution. Concentration of cationic starch is optimal between 0.5% and 1.0%. 37
Application of Direct Dyes
Direct dyes require high electrolyte concentration for dyeing and direct dyed fabrics show low wetfastness properties. Pre-treatment of cellulosic fibers with cationic agents is reported to diminish or eliminate the amount of electrolyte required, and improve the wetfastness of direct dyes.6,38Pre-treatment of cotton fabrics with mono- and bis-reactive cationic agents produces a fiber that may be dyed with direct dyes under neutral conditions in the absence of salt.
The cationic component of monochlorotriazine mono-reactive cationic agent is shown in Fig. 6. Fairly high degrees of exhaustion, fixation, and improved wetfastness are achieved for all cases of cationized cotton fabrics compared to untreated samples. The results also indicate that cotton pre-treated with the bisreactive cationic agent shows higher degrees of dye exhaustion and fixation relative to cotton pretreated with the mono-reactive agent. 39
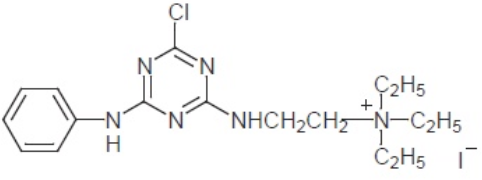
Monochlorotriazine mono-reactive cationic agent.
Pigment Dyeing
The use of pigments can provide a weathered or distressed look to garments without subsequent garment washing. In general, the procedure is to dye fabric in piece form, makeup garments, and then launder, washing down the color to produce the desired look. This process involves strict control over dye selection and application to ensure uniform and reproducible results. As an alternative, a garment-dyeing process has been developed to produce garments with the popular weathered or distressed look without subsequent garment washing. Since pigments are insoluble in water, they have no substantivity or affinity for cotton and a binder is required to fix the pigment particles on the fiber surface to give colorfastness. It is possible to dye cotton fabric with pigments by exhaust method, provided the fabric has been pretreated with a cationic auxiliary to provide the necessary affinity 40
Most companies offer a complete system of pigment or cationic agent evaluated for effectiveness and application conditions. It is important to choose a system that yields high pigment exhaustion and is reproducible. Other important considerations are color range, fastness, cost, and desired results. It is important to choose fabrics such as twills, single knit piques, or other types of fabric with a textured surface to achieve the best look with the pigment-dyeing process. 40
The recommended piece of equipment for this process is generally a rotary drum machine capable of being programmed with a variable drum speed during the dyeing cycle. However, other types of equipment, such as paddle machines, are also used in pigment garment dyeing.
The cationizing agent is exhausted onto the garment first. It acts as a link between fabric and pigment. After cationic treatment, the bath is dropped, the garment rinsed, and the pigment applied. LR is kept as high as 20:1 and the initial bath temperature is kept low (27 °C) to gain uniform exhaustion of the pigment onto the garment. A slow rate of temperature rise (1 °C/min) aids uniform exhaustion of the pigment.
After rinsing off any excess color, a low temperature or air-curable binder, usually an acrylic-based product, must be applied to the garments to improve crockfastness. It is preferable that a cationic rather than a nonionic binder is exhausted onto the fabric. Because reproducibility from one dye lot to the next may be difficult, it is important to carefully select garment components, dyes, and chemicals to be used and to monitor the dyeing process. Only with precise process control is it possible to reproduce colors between dye lots within currently accepted tolerances.
This exhaust method of pigment dyeing is widely used for the dyeing of T-shirts, sportswear, jackets, and trousers on fibers such as cotton, rayon, acrylic, polyester, nylon, and protein fibers. Conventional pigment dyeing has drawbacks in terms of wet crockfastness and coarse handle. The exhaust method of pigment dyeing can achieve moderate dry crockfastness at medium depths of color. 14 These problems can be alleviated by pretreating the fabric with cationic reagents and by increasing the dispersion of the pigment. By modifying the pigment surface, the average diameter of the pigment particles can be reduced to 100-200 nm, giving the pigment dispersion greater stability and color strength approaching that of dyes. 41
Pigments are insoluble in water. For dyeing, they are mixed with dispersing agent and cationic or anionic polymers. This is a very complex method and usually a trade secret for many companies. However, cationized pigment emulsions are not very marketable as the value added benefits are much less than their counterparts (i.e., anionized pigment emulsions). New kinds of pigments are formulated for use in textile dyeing. These are maintained in a stable dispersion in the medium of water by anionic surfactants. This type of pigment is known as a pigment resin color (PRC). 42
A solid anionic pigment preparation, consisting essentially of: (A) from 60% to 95% by weight of at least one pigment, (B) from 5% to 40% by weight of at least one water-soluble anionic surface-active additive (selected from the group consisting of homo- and copolymers of ethylenically-unsaturated monocarboxylic acids and/or ethylenically-unsaturated dicarboxylic acids with or without vinyl monomers comprising no acid function and alkoxylation products of these homo- and copolymers and their salts), and (C) from 0% to 20% by weight of at least one nonionic surface-active additive based on polyethers. 43
A conventional pigment, C.I. Pigment Red 22, is stirred with an aqueous solution of an anionic polymeric dispersant XG-1 (3% on weight of pigment) on an IKA high-speed mixer for 30 min at 10,000 rpm. The resulting conventional pigment dispersion has an average particle diameter of ∼1500 nm. This dispersion is converted to a nanoscale dispersion on an M-110EHI microfluidiser (Microfluidics International), processing at a pressure of 22,000 Pa for 2.5 h, which resulted in an average particle diameter of 128 nm with a final pigment dispersion of 40% solid content. 44 Cotton fabric is treated with a cationic agent at pH 11 and 80 °C for 30 min. After rinsing, the cationized fabric is exhaust dyed with the nano pigment dispersion for 10 min at RT and then heated to 70-80 °C over 10 min. After 20 min, binder is added and this temperature is maintained for an additional 10 min. The fabric is then washed with nonionic detergent and dried.
The study observed that pigment uptake is closely related to the concentration of cationic reagent, the pH, temperature of the pretreatment bath, and the pretreatment time. These factors also have an effect on the colorfastness of pigment-dyed fabrics. The nanoscale pigment dispersion gives a much higher color yield than that from dyeing with a conventional pigment dispersion. The nanoscale dispersion also produces a softer handle and more brilliant shade. Colorfastness properties are acceptable for wash-down effects. Exhaust dyeing with nanoscale pigments offers a number of advantages, including reduced pigment requirement, improved appearance and handle, and also environmental advantages. 45
Pigment Printing
Solfix E (CIBA, 40% solid content) is a polyaminochlorohydrin quaternary ammonium polymer with epoxide functionality that can react with cellulose via ether formation in the presence of alkali. 46 Cotton fabric is pre-treated with Solfix E at a concentration of 10-40% owf at 50 °C in a bath (LR = 30:1) for 1 h. Then, sodium carbonate solution (14% w/v) is added to the treatment bath in portions at the same temperature and the reaction is continued for a further 2 h. The bath is then drained, the fabric thoroughly rinsed with water, followed by soaping with 3 g/L of non-ionic detergent at the boil for 30 min, rinsed with water, and air dried. The cationized fabric is then printed with pigment, acid, and direct dyes in the usual way. The pigment and anionic dyes on cationized fabric show better fastness properties than untreated cotton fabric. The color loss after repeated washing is also less. The improvement in printability is a direct result of the formation of ionic bonds between the cationized cotton fabric and the anionic dyes or the print paste of the pigment used. 47
Cationized Linen
Unmodified linen fabric is cationized through the reaction of two different commercial cationizing agents: CHPTAC and polyaminochlorohydrin quaternary ammonium salt with epoxide functionality (PAQAC). Uncationized and cationized linen fabrics are dyed with four different types of fiber reactive dyes by using the cold pad batch dyeing method. The cold pad batch method for cationization with CHPTAC is a pH dependent reaction process that requires the addition of alkali. Fastness properties of the dyed cationized linen fabrics are evaluated. The results show that better
The lightfastness of the dyed fabrics treated with CHPTAC is better than that when PAQAC is used; indicating that the cationization reaction with CHPTAC takes place through the fiber, not just on the surface. The poor lightfastness of the fabric treated with PAQAC suggests that surface dyeing occurs in this case. 48
Two acid dyes and two direct dyes are used for dyeing linen fabrics with the assist of cationization using two different commercial cationizing agents, CHPTAC and polyhexamethlene biguanides (PHMB).
Cold Pad Batch Method
Linen fabric is padded with dye, 250 gm/L of CHPTAC, and 180 gm/L sodium hydroxide. The wrapped fabric is stored at RT for 12 h, then rinsed several times with water, and finally with diluted (1 g/L) acetic acid solution, and air dried. The final fabric pH was 7.4.
Pad Dry Cure Method
Linen fabric is padded with the above solution to 95% pickup. dried at 60 °C for 5 min, the dried fabric cured at 160 °C for 2 min, the fabric is rinsed several times with water, and finally with diluted (1 g/L) acetic acid solution, and air dried. The final fabric pH was 7.4.
Uncationized and cationized linen fabrics are dyed with the selected dyes using three different dyeing methods. The fastness properties of the dyed, uncationized, and cationized linen fabrics are evaluated. Cationic treatment of linen fabric with CHPTAC and PHMB produces deeper shades when exhaust dyed with acid and direct dyes compared to the uncationized linen fabric. Uncationized linen fabrics have lighter shades when dyed with the same dyes under the same dyeing conditions. 49
Cationized Ramie Fiber
A modified procedure for ramie fiber using CHPTAC as the cationic agent and caustic soda as the catalyst is available.
42
The morphological and structural transformations of the fiber induced by modification are determined by X-ray Diffraction (XRD), differential scanning calorimetry (DSC), and thermogravimetric analysis (TGA). XRD results show that the crystal structure of the modified fiber is still preserved although its crystallinity decreased. This was confirmed by the TGA results. The mechanisms for the modification and dyeing of ramie fiber are analyzed. Optimal modification conditions are 30 g/L of CHPTAC, 15 g/L of NaOH, reaction temperature of 50 °C, and reaction time of 60 min. Raw and modified fibers are dyed with C.I. Reactive Red 2. The
Conclusions
It is apparent that cationization of cellulosic fibers has generated tremendous enthusiasm among researchers— enough for them to address various problems of textile chemical processing related to eco-friendliness. Dyeing of cationized fiber material is not well standardized and is restricted to a limited number of small dyers. Cationization of cotton is not fully commercial yet. However, a great amount of research has been performed in this area. The time has come to coordinate and integrate various studies in this field.
