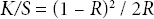Abstract
A Ag-nanoparticle (Ag-NP) dispersion was successfully synthesized using a glucose/Ag-nitrate/polyvinyl pyrrolidone (PVP)/sodium hydroxide reduction formulation. The resultant Ag-NP dispersion was successfully used alone and in combination with Disperse Orange 31 or Basic Blue 9 dyes to impart antibacterial properties against Gram-positive (Staphylococcus aureus) and Gram-negative (Escherichia coli) bacteria to polyester and polyacrylonitrile fabrics respectively using microwave treatment. An increase in Ag-NP dispersion concentration by 50% and prolonged microwave irradiation by two minutes significantly improved the antibacterial performance. The EDX spectrum of the Ag-NP loaded substrates confirmed the presence of elemental Ag (Ag0) on the fabrics’ surface. Addition of selected disperse or basic dye to the Ag-NP treatment bath produced disperse or basic dyeings with darker depth of shades and remarkable antibacterial functionality.
Introduction
Man-made synthetic polymer fibers include fibers such as polyester, polyacrylonitrile, and polyamide fibers. Polyester, the leading man-made fiber, is commercially known as polyethylene terephthalate (PET). PET is extensively used, due to its versatility alone or as a blend, in woven and knitted apparels, home furnishings, and industrial applications. Regular acrylic fibers are produced from copolymers containing more than 85% acrylonitrile monomer, along with other co-monomers used in polymerization (e.g., methacrylic acid, methyl methacrylate, and vinyl acetate) that are incorporated to improve dyeability and mechanical properties. Acrylic fibers have found a wide spectrum of uses including sweaters, skirts, dresses, suits, and knitted wear where soft and wool-like characteristics are desired. 13
Recently, attention was given to high added-value textile products that are able to combine traditional textile characteristics with modern multi-functionality Highly functional textiles are being used not only for clothing, but also in numerous industrial applications. 4 Nanostructured inorganic materials, metals, and metal oxides are of interest because of potential applications in functional textile finishing including self-cleaning, antimicrobial, and UV-protective textiles. 5 -9
Silver (Ag) is by far the metal most widely used as an antibacterial agent. 10 , 11 Several methods have been used to prepare Ag-nanoparticles (Ag-NPs) such as chemical, electrochemical, γ-irradiation, photochemical, laser ablation, and so forth. 12 The most common method of producing Ag-NPs is chemical reduction of Ag salts in water with reducing agents such as sodium borohydride, sodium citrate, polysaccharides, hydrazine, etc. 13 Incorporation of polyvinyl pyrrolidone (PVP) along with glucose resulted in accelerating an Ag-ion/glucose reaction, as well as protecting the formed Ag-NPs from further growth and agglomeration. 14
Recently, microwave irradiation has been widely used as an alternative to conventional heating and as a powerful technique of non-contact heating. Microwave heating was shown to be a more rapid, uniform, and efficient tool for reducing heat transfer problems. 15 , 16 Therefore, the present study is aimed at functionalizing polyester and polyacrylonitrile fabrics with Ag-NPs by microwave irradiation.
Experimental
Materials
Prewashed PET (177 g/m2) and polyacrylonitrile (PAN, 200 g/m2) woven fabrics were used for this study. All chemicals used such as AgNO3 (Sigma), PVP (40,000 Da, Merck), glucose (Merck), and NaOH (Merck) were analytical reagent grade. C.I. Basic Blue 9 (Merck) and C.I. Disperse Orange 31 (Clariant) were used for this research (Fig. 1). Doubly distilled water was used for preparation of Ag-NP dispersions.
Chemical structure of the dyes used.
Methods
Preparation of Ag-NP Dispersions
An aqueous solution (15 mL) containing glucose (10 mmol), PVP (11.5 mmol), and NaOH (7.5 mmol) was heated at 70 °C for 20 min under vigorous stirring. To this solution, 5 mL of AgNO3 (1.0 mol/L) was added, the solution refluxed for 40 min, and the colloid solution was cooled to room temperature (RT) in 30 min. 17 The prepared Ag-NP dispersion was brownish yellow, indicating the formation of Ag-NPs.18,19
Antibacterial Finishing
For microwave-assisted functionalization of these man-made substrates, fabric samples were immersed in a 100 mL Pyrex beaker containing different amounts of the prepared Ag-NP dispersion in aqueous solution using a liquor ratio (LR) of 30/1 at RT for 20 min, then placed in a commercial microwave at output power of 1725 W for various periods. After irradiation, treated fabric samples were thoroughly washed with hot distilled water to remove any unfixed Ag-NPs, thoroughly rinsed with distilled water, and finally dried at RT.
Concurrent Dyeing and Antibacterial Finishing
The feasibility of combined Ag-NP functional finishing and disperse or basic dyeing of PET or PAN substrates in one step was studied. Disperse dye or basic dye was included in the antibacterial-finishing formulations, followed by subsequent treatments given in the previous section.
Measurements
Ultraviolet-visible (UV-Vis) spectroscopy of the prepared Ag-NP dispersions was performed using a Perkin-Elmer Lambda spectrophotometer at a wavelength of 380 cm−1.
Transmission electron microscopy (TEM) was performed using a JEM 2100 F electron microscope (JEOL) at 200 kV. Specimens for TEM measurements were prepared by dissolving a drop of colloid solution on a 400-mesh copper grid coated by an amorphous carbon film and evaporating the solvent in air at RT.
Scanning electron microscope (SEM) images of untreated and treated fabric samples were obtained with a JXL-840A electron probe microanalyser (JEOL), equipped with an energy disperse X-ray spectroscopy (EDX) unit for the composition analysis. Samples were mounted on a stub with double stick adhesive tape and coated with gold in a S150A sputter coater unit (Edwards). The gold film thickness was 150 Å.
The antibacterial activities against Gram-positive (G+ve) S. aureus and Gram-negative (G-ve) E. coli bacteria were evaluated qualitatively according to AATCC Test Method (TM) 147-1988 and expressed as zone of growth inhibition (ZI, in mm units).
Color strength (K/S values) were determined from reflectance measurements using the Kubelka-Munk equation. 20
K/S is the ratio of absorption and scattering coefficient, and R is the reflectance at the wavelength of maximum absorbance of the dyes used.
The durability to wash (after 15 laundering cycles) was determined according to AATCC TM 61(2A)-1996.
Results and Discussion
Characterization of Prepared Ag-NP Dispersions
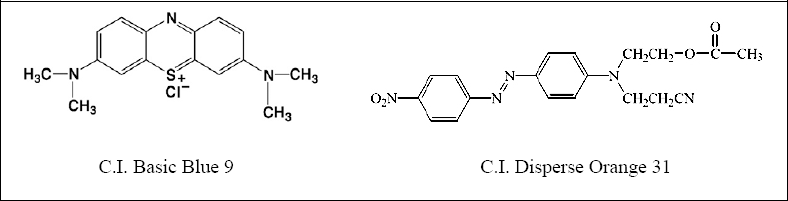
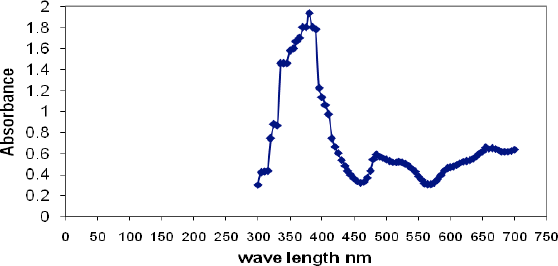
The Ag-NP dispersions were characterized using UV-Vis spectroscopy (Fig. 2) and TEM (Fig. 3). Fig. 2 shows a well defined plasmon band at 380 nm indicating Ag-NP generation and a decrease in Ag ion concentration, most probably due to the accelerating effect of PVP on the reaction between Ag ions and the reducing agent (i.e., glucose) used. 14 TEM analysis (Fig. 3) showed that the size of the Ag-NPs in the dispersion ranged from 12 to 42 nm.21,22
UV-Vis absorption spectrum of the prepared Ag-NP dispersion.
TEM image of the Ag-NPs prepared by glucose/PVP reduction of AgNO3 in alkaline medium.
Tentative Mechanism
Scheme 1 demonstrates the possible reactions among the AgNO3, glucose, and PVP in aqueous alkaline medium to give the final Ag-NP dispersion.
Before Addition of PVP:

Tentative mechanism.
The result was to decrease the presence of Ag ions and accelerate/enhance the extent of Ag-NP formation. 14 ,23-25
Functional Finishing
The Ag-NP dispersions were used to enhance the antibacterial properties of the chosen substrates by application of microwave irradiation. For a given set of treatment conditions, the data in Table I show that increasing the Ag-NP dispersion concentration up to 50% owf in the immersion bath resulted in significant enhancement in the antibacterial activity of the treated substrates (expressed as zone of inhibition—ZI). Treated (at 50% owf Ag-NP and 120s irradiation time) PET and PAN substrates gave maximal ZI values against both G+ve (17.5 mm and 21.0 mm) and G-ve bacteria (from 16.00 mm and 19.00 mm), respectively. Prolonged microwave irradiation time from 30s to 120s led to a remarkable improvement in the imparted antibacterial functionality, against G+ve (from 6.6 mm up to 17.5 mm and from 7.4 mm up to 21 mm for treated PET and PAN, respectively) and G-ve (from 5.5. mm up to 16 mm and 6.1 mm up to 19 mm for treated PET and PAN, respectively).
The extent of improvement in the imparted antibacterial activity was determined by type of substrate, fiber chemistry, surface morphology, active sites, extent of modification during irradiation, and subsequent attraction and fixation of Ag-NPs on and into the fabric structure (Eq. 8). This reaction follows the decreasing order of reactivity: PAN > PET26-29
The remarkable antibacterial activity by loading Ag-NP-dispersions onto the fabrics surfaces involved uptake of free Ag ions followed by protein denature and cell death. Ag-NPs interact with the bacterial membrane by penetrating inside the cell, thereby increasing permeability and disturbing respiration. Generation of reactive oxygen species such as hydrogen peroxide, in the presence of dissolved oxygen along with Ag-NP and/or Ag ion as catalysts, can also occur, oxidizing bacterial molecules.7,30
Table I reveals that the inactivation of G+ve (S. aureus) was more efficient than G-ve (E. coli) bacteria, keeping other parameters constant. This could be attributed to differences in cell wall structure, outer membrane, amenability to inhibition of enzymes functions, and/or inactivation of DNA replication between these organisms.30–32
The results obtained (Table I) disclose that the imparted antibacterial functionality was durable enough even after 15 washing cycles, regardless of the treated substrate tested. This can be explained as a strong interaction between the Ag+, Ag [PVP], and/or Ag0 or Ag0/PVP with the treated substrate active sites, especially -COOH groups via formation of ionic and/or coordination bonds. The enhancement of Ag ions or nanoparticle diffusion into fibers could also occur as a direct consequence of free volume generation within the polymer structure via segmental mobility during microwave irradiation in an aqueous medium. Partial hydrolysis of the PET and PAN fibers and the formation of additional functional groups on the fiber boundary, thereby increasing the extent of loading and binding of Ag onto/within the fiber structure, cannot be ruled out. 33
Effect of Ag-NP Dispersion Concentration and Irradiation Time on Antibacterial Activity of Treated Substrates a
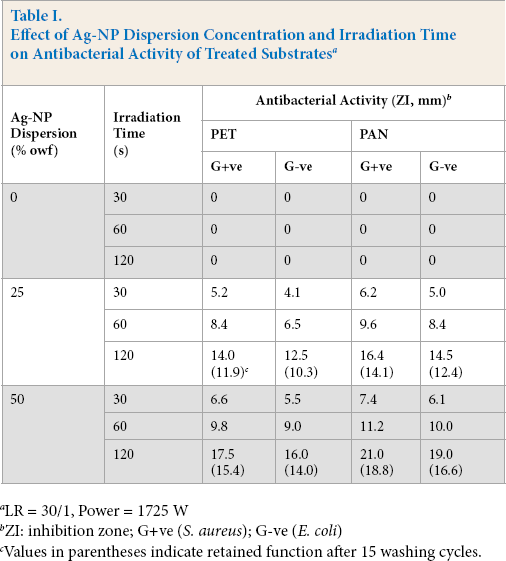
LR = 30/1, Power = 1725 W
ZI: inhibition zone; G+ve (S. aureus); G-ve (E. coli)
Values in parentheses indicate retained function after 15 washing cycles.
The fiber surface of untreated and Ag-NP finished fabrics were observed by SEM micrographs. The images (Figs. 4b and 5b) showed the deposition of well-dispersed nano-scale Ag-particles on PET and PAN surfaces compared to the untreated fabrics (Figs. 4a and 5a, respectively). The corresponding EDX spectra (Figs. 4c and 5c, respectively) showed the presence of Ag0 on the fiber surfaces. This provides evidence for Ag-NP loading on the selected substrates. The extent of Ag-NP loading on the treated substrates was governed by the surface morphology and the fiber chemistry in addition to the changes in fabric structure during microwave irradiation treatment.
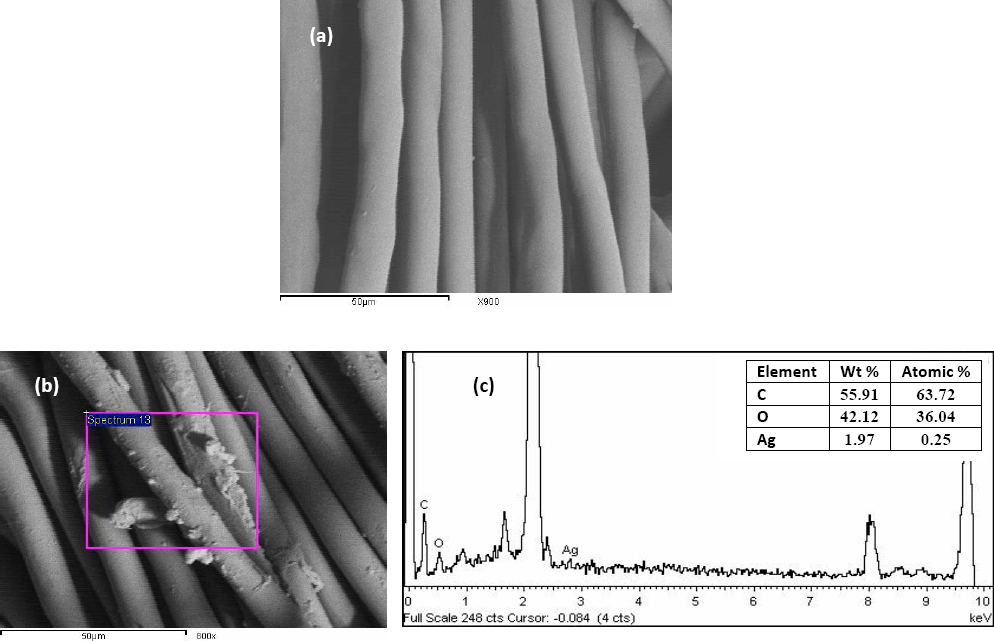
(a) SEM of untreated PET, (b) SEM of treated PET with Ag-NP dispersion, and (c) EDX image and Ag content of loaded PET. Ag-NP dispersion (50% owf); LR (20/1); irradiation time (120 s); power (1725 W).
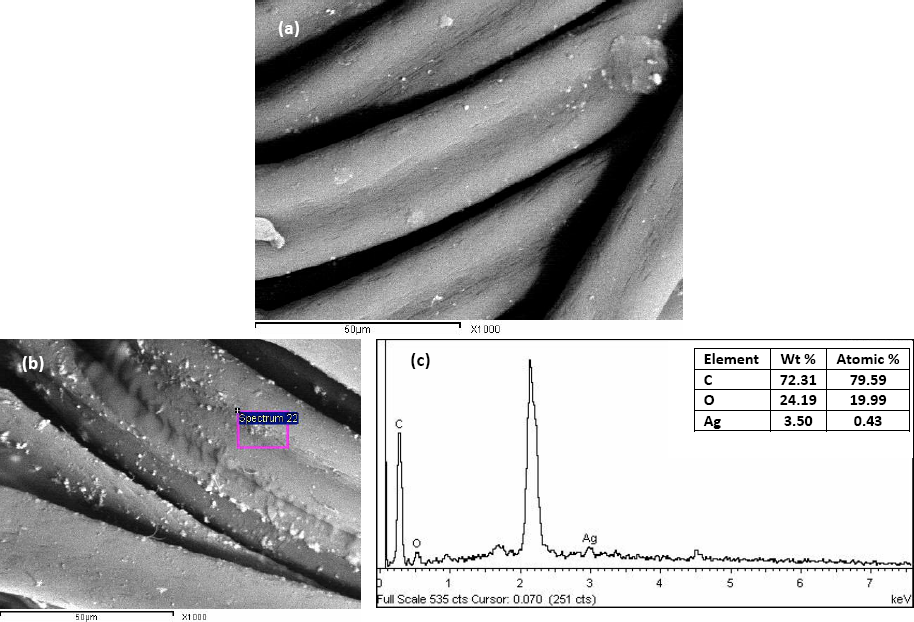
(a) SEM of untreated PAN, (b) SEM of treated PAN with Ag-NP dispersion, and (c) EDX image and Ag content of loaded PAN. Ag-NP dispersion (50% owf); LR (20/1); irradiation time (120 s); power (1725 W).
Concurrent Functional Finishing and Dyeing
The feasibility of carrying out antibacterial finishing and disperse or basic dyeing in a single stage using microwave irradiation was tested. K/S values and the antibacterial activities of the treated substrates are given in Table II.
Feasibility of Dyeing and Antibacterial Finishing in One Stagea
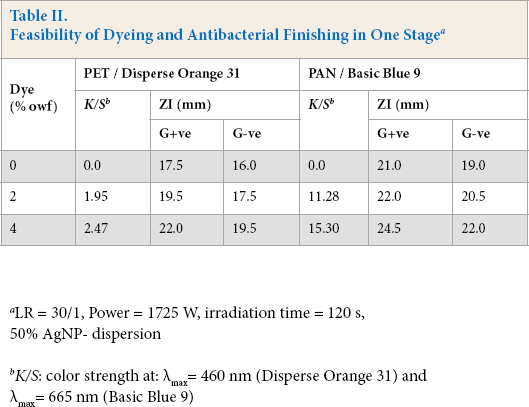
LR = 30/1, Power = 1725 W, irradiation time = 120 s, 50% AgNP- dispersion
K/S: color strength at: λmax= 460 nm (Disperse Orange 31) and λmax= 665 nm (Basic Blue 9)
The results show that incorporation of the disperse dye or basic dye in the finishing bath along with the Ag-NP dispersion led to a significant improvement in the K/S value along with an increase in the antibacterial activities of the obtained dyeings. The extent of improvement in the aforementioned properties was governed by both the type of substrate as well as the dye class. The higher the dye concentration used, the better the dyeing and functional properties. The imparted antibacterial properties against the G+ve bacteria were higher than that for the G-ve bacteria.
The extent of dyeing, expressed as a K/S value, was determined by the dye's chemical structure, functionality, and mode of interaction (physically as in case of disperse dyeing of PET, or by an ion exchange mechanism as in case of cat-ionic dyeing of PAN). The Ag-NP dispersion had a positive impact on enhancing the extent of dye uptake and fixation onto/within the irradiated synthetic fiber to make it more dye receptive. 34 Additionally, the rating of washfastness for the simultaneously dyed and finished fabric samples were quite good, irrespective of the dye type used and its concentration, according to AATCC TM 61-1996. 4
The enhancement of antibacterial activity that occurred by including the selected dyes along with the Ag-NP dispersion could be due to the synergetic effect of Ag-NP dispersion components and the active chemical groups in the basic (quaternary amine) and disperse (such as tertiary amine and -CN groups) dyes used. 8
Conclusion
Ag-NP dispersions of 15-42 nm size were prepared using a AgNO3/glucose/PVP/NaOH combination. They were used for imparting antibacterial functionality to PET and PAN fabrics using microwave treatment. The results demonstrate excellent durable antibacterial activity against G+ve (S. aureus) and G-ve (E. coli) bacteria (more than 85% retention of their original antibacterial activity even after 15 washing cycles). The PET and PAN fabrics treated with Ag-NPs along with disperse Orange 31 and Basic Blue 9 dyes respectively also demonstrate an enhancement in both the antibacterial and coloration properties.
Future work will include studies using the dispersion without Ag-NPs and AgNO3, at the same level used for Ag-NP preparation, and the addition of AgNO3 without dispersion.