Abstract
Novel heterocyclic arylazo-disperse dyes containing pyrazolo[1,5-a]pyrimidine structures were prepared via conventional refluxing and microwave heating. Starting from cyanoacetic acid, 4-arylazo-2H-pyrazol-3-ylamine was obtained from a sequence of reactions. This pyrazole intermediate afforded the target arylazo-pyrazolo[1,5-a]pyrimidines and pyrazolo[1,5-a] pyrimidin-7-ones upon cyclization with 3-piperidinylacrylonitrile, ethyl acetoacetate, ethyl(arylhydrazono)acetoacetates, or (arylhydrazono)propanals. The structures of these compounds were confirmed by 1 H and 13 C nuclear magnetic resonance (NMR) spectroscopy, mass spectroscopy, infrared (IR) analysis, and elemental analysis. The disperse colorants produced were applied to printing on polyester fabrics to give satisfactory results. All the synthesized products were tested as antimicrobial agents. Most compounds exhibited relatively strong antibacterial activity against a panel of Gram-positive and Gram-negative bacteria.
Introduction
Many nitrogen-containing heterocyclic materials1,2 have biological activity and their role as pharmacophores are of considerable historical importance.3–5 Nitriles have been broadly used as intermediates in the synthesis of various heterocycles, such as aminopyrazoles, that can be easily prepared by the reaction of nitrile derivatives with hydra-zine.6,7 The reaction of aminopyrazole derivatives with electrophilic reagents affords a variety of fused heterocycles with a broad spectrum of antimicrobial activity.8–14 Furthermore, these fused heterocycles are also used as intermediates in the dye industry.15–17 Arylazopyrazolo[1,5-a]pyrimidines were prepared by condensing aminoarylazopyrazoles with β-bifunctional reagents using methodologies initially established in our laboratories.18–20
In the present work, we report the synthesis of arylazopyrazolo[1,5-a]pyrimidines and their potential application as disperse dyes for printing polyester fabrics. These fused systems displayed considerable antimicrobial activity and gave satisfactory printing results on polyester fabrics. A comparison of conventional and microwave heating use during syntheses was also made. Fastness properties and spectral data for these new dyes were also evaluated. The UV-Vis absorption spectra of these dyes with electron-withdrawing and electron-donating groups were also examined in detail.
Experimental
Materials and Apparatus
All melting points are uncorrected and are expressed in °C. They were determined on a Stuart melting point apparatus. Infrared (IR) spectra were recorded as KBr pellets on a Fourier transform infrared (FTIR) Nicolet 670 spectrometer and
1
H nuclear magnetic resonance (NMR) spectra were recorded on a Varian GEMINI 200 MHz NMR spectrometer, using tetramethylsilane (TMS) as the internal standard, and chemical shifts were given in δ (ppm).
13
C NMR spectra were obtained on a JEOL EX-400 MHz NMR spectrometer, the chemical shifts are expressed in δ (ppm) using TMS as the internal standard. Mass spectra were obtained from a Shimadzu GCMS-QP-1000 EX spectrometer (70 eV). The maximum absorption wavelength (
Unless otherwise noted, all chemicals were supplied by Sigma Aldrich. Dry solvents for syntheses and spectroscopic measurements (spectroscopic grade) were used without further purification. Scoured and bleached 100% polyester fabrics were supplied by El-Mahalla El-Kobra Co. The fabrics were scoured in and aqueous solution having a liquor ratio (LR) of 1:50 and containing 2 g/L of non-ionic detergent solution (Hostapal, Clariant) and 2 g/L of sodium carbonate at 50 °C for 30 min to remove waxes and impurities, followed by rinsing in cold water, and finally drying at room temperature (RT).
Printing
Dyes were ground up using a small mortar and pestle and then sieved using a USA standard sieve with 0.0029 in. (75 μm) holes. A paste composed of synthetic thickener stock paste (3.5 g Alcoprint DT-CS with 95 g water) and the ground dye (1.5 g) was prepared by stirring the mixture well. The printing paste was applied to the fabrics using the fat screen printing technique. White polyester fabric (100%) plain weave (149 g/m2) was supplied by Miser Co. Print fixation was done by thermofixation at 180–200 °C for 2–3 min in an automatic thermostatic oven (Werner Mathis Co.). Washing of involved cold water rinsing, followed by washing with soap at 40 °C using 1 g/L Ciba Eriopon OS. Reduction clear was performed at 50 °C using 1–2 g/L sodium hydrosulfite, 1–2 g/L caustic soda at 66 °Tw (36 °Bé), and 1 g/L Ciba Eriopon OS. The printed fabric was rinsed at 50 °C, then with cold water, and finally dried. 6
Colorfastness
Fastness to washing, perspiration, light, and sublimation was tested according to the reported methods.21–24
Color Strength
The color strength of the printed samples, expressed as
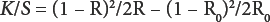
Crockfastness
Crockfastness was determined according to the ISO test meth-od. 22 In this test, the color is transferred from the surface of the colored fabric to another surface by rubbing. A colored test specimen fastened to the base of a crockmeter (SDL Atlas) was rubbed on a white crock test cloth under controlled conditions.
Dry Crocking Test
The test specimen was placed on the base of the crockmeter. A white testing cloth was mounted. The covered finger was lowered onto the test specimen and caused to slide back and forth 20 times by making ten complete turns at a rate of one turn per second. The white test sample was then removed for evaluation using the Gray Scale for Staining.
Wet Crocking Test
The white test sample was thoroughly wetted out in water to a 65% pickup. The procedure was run as before and the white test samples were then air dried before evaluation.
Washfastness
Washfastness was determined according to ISO 105-C02:1989 using a Launder-Ometer (SDL-Atlas). 21 The specimens (5 × 10 cm) were sewn between two similar pieces of bleached cotton fabric. The composite specimen was immersed into an aqueous solution containing 5 g/L soap and 2 g/L of sodium carbonate at an LR of 50:1. The bath was thermostatically adjusted to 45 °C. The test was run for 30 min at 42 rpm. The samples were then removed, rinsed twice with occasional stirring or hand squeezing, and then dried. Washfastness was assessed using the Gray Scale for Color Change.
Colorfastness to Perspiration
Alkaline Solution
L-Histidine monohydrochloride monohydrate (0.25 g/L), sodium chloride (10 g/L), and sodium dihydrogen phosphate (1 g/L) were dissolved in 1 L of distilled water. The pH was adjusted to 8 with sodium hydroxide solution (0.1N).
Acidic Solution
L-Histidine monohydrochloride monohydrate (0.25 g/L), sodium chloride (10 g/L), and sodium dihydrogen phosphate (1 g/L) were dissolved in 1 L of distilled water. The pH was adjusted to 4.3 with an acetic acid solution (10%). The colored specimen (5 × 4 cm) was sewn between two pieces of uncolored specimens to form a composite specimen. The composite specimen was then immersed for 15–30 min in each solution with occasional agitation and squeezing to insure complete wetting. The test specimen was placed between two glass plates under a force of ∼4–5 kg. The plates containing the composite specimen were then held vertical in the oven at 37 ± 2 °C for 4 h. The effect on the color of the test specimen was determined by reference to the Gray Scale for Color Change.
Lightfastness
Lightfastness was determined according to AATCC TM 16A-1989 (ISO 105-B02:1988). 24 The evaluation was established using the blue scale (SDL Atlas) as the reference of color change. For lightfastness testing, a Fade-Ometer 18-F (Aldrich) equipped with a UV lamp was used, with light exposures conducted for 35 h.
Antimicrobial Activity Evaluation of Synthesized Dyes
Some of the synthesized compounds were evaluated for antimicrobial activity against
Synthesis-Intermediates
Note that data relating to the characterization of the following intermediates are available from the authors. All proper safety precautions, including wearing safety glasses, use of fume hoods, and shielding, must be observed when working in the laboratory.
3-Piperidin-1-yl-acrylonitrile
A mixture of cyanoacetic acid (0.05 mol), triethyl orthofor-mate (0.05 mol), and piperidine (0.5 mL) was heated under reflux for 2 h, allowed to cool to RT, then poured into cold water. The reaction mixture was treated with 1 M sodium carbonate solution (50 mL), extracted with dichlorometh-ane (100 mL), and the extract was left overnight to dry. The solvent was evaporated in vacuo. Piperidin-1-yl-acrylonitrile (
Phenyl Substituted and Unsubtituted 2-(phenylhydrazono)-3-oxo-propionitriles
An aryldiazonium salt (0.01 mol) solution was prepared by adding sodium nitrite solution (0.7 g in 10 mL H2O) to a chilled solution of arylamine hydrochloride (0.01 mol of arylamine in 5 mL conc. HCl) with stirring. The resulting aryldiazonium solution was then added to a cold solution of
Ethyl 2-(4-nitrophenylhydrazono)acetoactate
4-Nitroaniline (1.38 g, 0.01 mol) was dissolved in conc. HCl (10 mL) and the solution was then cooled to 0–5 °C. Sodium nitrite (0.01 mol) in water (3 mL) was then added to this solution dropwise with vigorous stirring over 1 h, while cooling at 0–5 °C. The clear diazonium salt solution was then added dropwise to a well-cooled (0–5 °C) and stirred solution of ethyl acetoacetate (1.3 g, 0.01 mol) in sodium acetate (2 g, dissolved in 10 mL of 50% aqueous ethanol). The pH of the coupling mixture, in each case, was maintained at 5–6 throughout the coupling process by adding sodium acetate. Stirring was continued for 4 h at 0–5 °C and the precipitated products separated upon dilution with cold water (50 mL) were filtered of, washed with water several times, dried, and recrystallized from ethanol-H2O to give ethyl 2-(4-nitrophenylhydrazono)acetoactate (
4-(Arylazo)-2H-pyrazol-3-ylamine
A mixture of 2 (0.01 mol) and hydrazine hydrate (0.01 mol) in ethanol (25 mL) was refluxed for 2 hours then poured into H2O. The solid 4-(arylazo)-2H-pyrazol-3-ylamine (
Synthesis-Dyes
3-Phenylazo-pyrazolo[1,5-a]pyrmidin-7-ylamine
A mixture of 3-piperidinoacrylonitrile
5-Methyl-3-(4-nitro-phenylazo)-4H-pyrazolo[1,5-a] pyrimidin-7-one
A mixture of ethyl acetoacetate
Procedures for 3,6-Bis(arylazo)pyrazolo[1,5-a]pyrimidin-7-ylamine Syntheses
Method A
A mixture of
Method B
A mixture of
Method C
A solution of the aryldiazonium salt, which was prepared from the appropriate aniline derivative (0.01 mol), sodium nitrite (0.01 mol), and concentrated HCl (25 mL) in water (10 mL) was added slowly to a cold 5 °C solution of
6-(4-Nitrophenylazo)-3-phenylazo-pyrazolo[1,5-a] pyrimidin-7-ylamine
6-(4-Nitrophenylazo)-3-phenylazo-pyrazolo[1,5-a]pyrim-idin-7-ylamine (
3,6-Bis-(2-nitrophenylazo)-pyrazolo[1,5-a]pyrimidin-7-ylamine
3,6-Bis-(2-nitrophenylazo)-pyrazolo[1,5-a]pyrimidin-7-ylamine (
3,6-Bis-(phenylazo)-pyrazolo[1,5-a]pyrimidin-7-ylamine
3,6-Bis-(phenylazo)-pyrazolo[1,5-a]pyrimidin-7-ylamine (
3,6-Bis-(4-nitrophenylazo)-pyrazolo[1,5-a]pyrimidin-7-ylamine
3,6-Bis-(4-nitrophenylazo)-pyrazolo[1,5-a]pyrimidin-7-yl-amine (
3,6-Bis-(p -tolylazo)-pyrazolo [1,5-a]pyrimidin-7-ylamine
3,6-Bis-(
Procedure for 3,6-bis-(arylazo)pyrazolo[1,5-a]pyrimidin-7-one Syntheses
A mixture of
5-Methyl-6-[(4-nitrophenyl)-hydrazono]-3-phenyl-azo-6H-pyrazolo[1,5-a]pyrimidin-7-one
5-Methyl-6-[(4-nitrophenyl)-hydrazono]-3-phenylazo-6H-pyrazolo[1,5-a]pyrimidin-7-one (
5-Methyl-3-(4-nitrophenylazo)-6-[(4-nitrophenyl)-hydrazono]-6H-pyrazolo[1,5-a]pyrimidin-7-one
5-Methyl-3-(4-nitrophenylazo)-6-[(4-nitrophenyl)-hydrazono]-6H-pyrazolo[1,5-a]pyrimidin-7-one (
(11,12,14,15-tetraaza-cyclopenta[a]phenanthren-17-yl) -p-tolyl-diazene
Aqueous sodium nitrite (0.7 g in 5 mL H2O) was added to a cold (0 °C) stirred solution of
Results and Discussion
Synthesis
The structures of synthesized compounds were confirmed by a variety of spectroscopic techniques, including
1
H and
13
C NMR, mass spectroscopy, IR, and elemental analysis. Elnagdi et al.6,18–20 reported that the reaction of enaminoni-trile
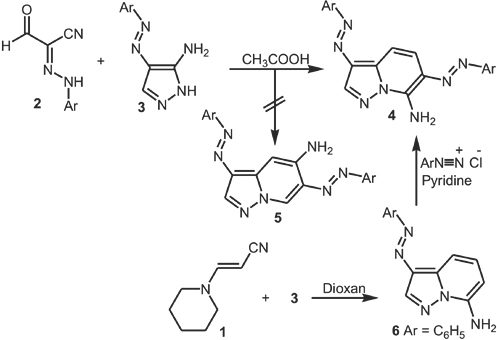
Synthesis of arylazo-pyrazolo[1,5-a]pyrimidin-7-ylamines starting from piperidinylacrylonitrile (
Product
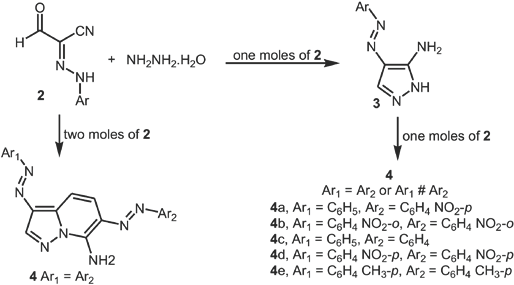
One-pot synthesis of arylazo-pyrazolo[1,5-a]pyrimidin-7-ylamines (
Aminopyrazoles
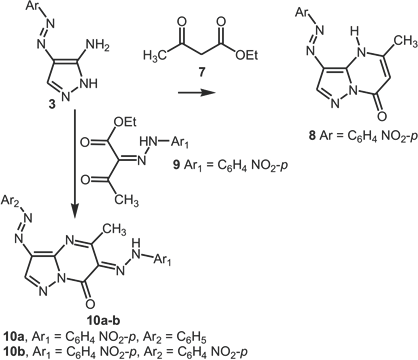
Synthesis of arylazo-pyrazolo[1,5-a]pyrimidin-7-ones (
Consistent with the previously reported reactivity of β-naphthol toward heterocyclic diazonium salts,
31
compound
Trials to isolate acyclic hydrazones failed. It is believed that direct formation of
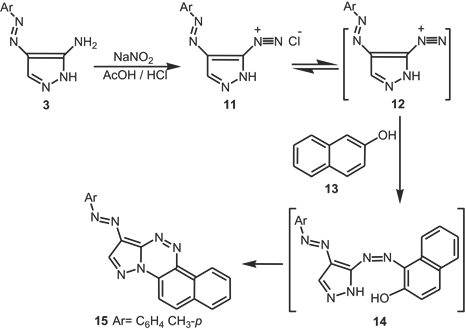
Synthesis of pyrazolo[5,1-c]-1,2,3-triazines (
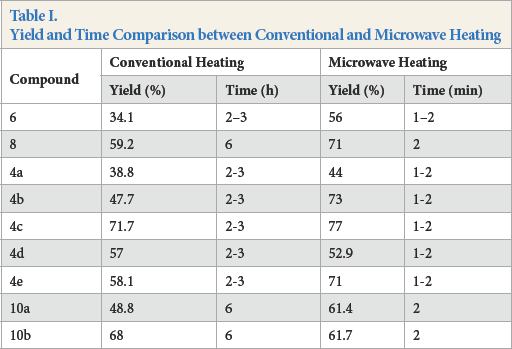
Use of microwave heating has environmental advantages over use of conventional energy resources, such as low energy consumption, milder reaction conditions, acceleration of reaction rates, increased reaction yield, and less or solvent-free reaction conditions.6,9,33 Products
Yield and Time Comparison between Conventional and Microwave Heating
Dyeing Polyester Fabrics
Color Properties
The arylazopyrazolopyrimidines synthesized in this work can be considered disperse dyes because they are water-insoluble, small molecules able to dye polyester fibers. Polyester has a high affinity for disperse dyes compared to other hydrophobic textile fibers. 34 Therefore, these arylazopyrazolo[1,5-a]pyrimidine disperse dyes were applied to polyester fabrics.
UV-Vis Absorption Spectra and Color Strength of the Prepared Dyes
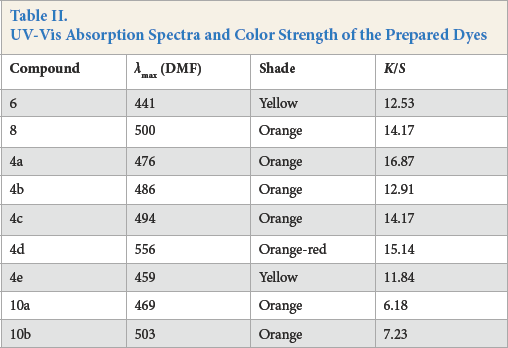
The hues of the printed polyester fabrics were consistent with the observed
Fastness Properties
The fastness of the printed polyester samples was generally satisfactory; the depths of shade and dye structure influenced the fastness results.
Washfastness
The rate of movement of dyes out of polyester fibers during the washing process depends on the molecular size of dye, the nature of mechanical link formed between the dye and the fiber, and the dye's charge, which is affected by presence of electron donating or electron withdrawing substituents. The washfastness of the printed polyester fabrics tested, including color change and stain on cotton, ranged from poor to very good (Table III). Better washfastness results were obtained for dyes with electron-donating substituents, indicating their higher substantivity to hydrophobic fibers. On the other hand, poorer washfastness results were obtained for dyes with electron-withdrawing substituents that consequently had poor substantivity to hydrophobic fibers.
Fastness Properties of the Synthesized Dyes on Polyester Fabrics a
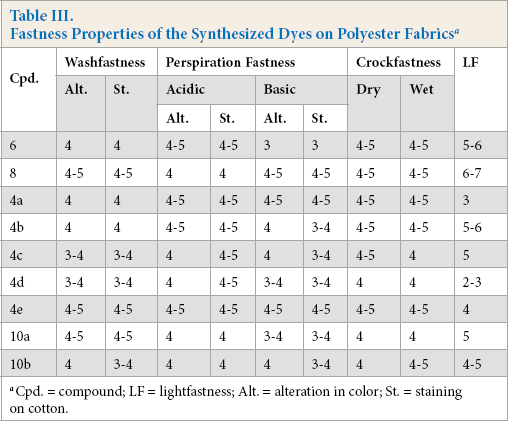
Cpd. = compound; LF = lightfastness; Alt. = alteration in color; St. = staining on cotton.
Perspiration Fastness
The extent of dye removal from polyester fabric due to perspiration is shown in Table III. The results show that dye removal was mostly dependent on the dye substituents. Dyes with electron-withdrawing groups were less stable to perspiration. The perspiration fastness values varied from poor to very good, with higher ratings for dyes with electron-donating groups.
Crockfastness
Crockfastness evaluates the removal of loosely adhered dye molecules from the surface of the polyester fabric. The higher molecular weight dyes had the higher crockfastness values. All of the dyed fabrics tested generally gave good crockfastness results as shown in Table III.
Lightfastness
Lightfastness values for the dyed fabrics tested varied from poor to very good depending on the dye‘s molecular structure and depth of shade (Table II). For the aminoazobenzene derivatives, an increase in the electron-withdrawing effect of the diazo component led to a decrease in the electron density on the amino nitrogen atom with photofading taking place due to singlet oxygen attack at this site.16,17 Generally, the same photofading effect was observed with the dyes containing the electron-withdrawing
Antibacterial Activity and Structure-Activity Relationship
Dyes
The percentage activity index was calculated by the formula shown in Table IV. Compared to the bisazo dyes
Dye Antibacterial Properties
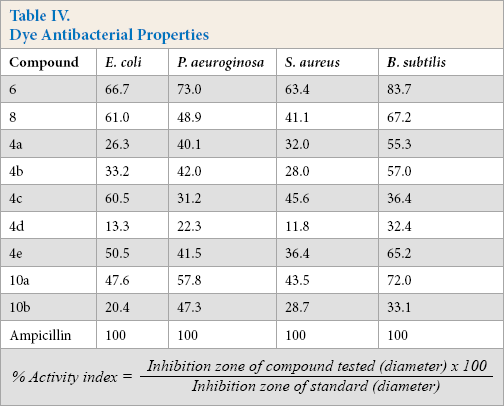
It is notable that these dyes exhibited antimicrobial activity. For this to be useful in practice, it would be necessary that, once applied, the dyes migrate out of the polyester. The generally good fastness results suggest that this migration would be limited. Future work is needed that tests the antimicrobial activity of the dyed material (as opposed to the dyes present on a paper substrate) to substantiate this conclusion. 35
Conclusion
In this work, a series of arylazo-pyrazolo[1,5-a]pyrimdin-7-ylamine and arylazo-pyrazolo[1,5-a]pyrimidin-7-one disperse dyes have been synthesized using both conventional refluxing and microwave heating. Use of microwave heating reduced the time required for synthesis from hours to minutes and reduced energy consumption. These yellow, orange, and orange-red dye printed polyester fabrics gave solid shades with satisfactory depth of shade, levelness, and fastness properties. The fastness properties tested (lightfast-ness, crockfastness, washfastness, and perspiration fastness) were, in most cases, rated acceptable to very good. Furthermore, most of these compounds showed relatively strong antibacterial activity.
Footnotes
Acknowledgement
We are grateful to National Research Centre, Cairo, Egypt; for their financial support.
