Abstract
A silver-tolerant (3.5 mM silver nitrate) marine bacterial isolate (Bacillus sp.) was used for biosynthesis of Ag-nanoparticles. UV-Vis spectra showed an absorption peak between 410 and 440 nm. Transmittance electron microscopic (TEM) analysis indicated the presence of 2 to 7 nm Ag-nanoparticles. O2-plasma pretreatment, followed by subsequent treatment with biosynthesized Ag-nanoparticles, improved loading and fixation onto the activated surfaces of the cellulosic fabrics (linen, cotton, and lyocell), and improved antibacterial activities against Gram-positive (Staphylococcus aureus) and Gram-negative (Escherichia coli) bacteria. Presence of Ag-nanoparticles on linen and lyocell surfaces was confirmed using scanning electron microscope (SEM) images. Antibacterial activity showed no severe reduction even after 15 washing cycles. Surface modification extent, Ag-nanoparticle loading, as well as antibacterial activity, was determined by the nature of cellulosic substrate.
Introduction
Textile materials based on cellulosic fibers are an excellent media for generation, growth, and spreading of harmful microorganisms (e.g., bacteria, fungi, and yeast) under appropriate nutrient, moisture, and temperature conditions.1–3 Harmful microorganism growth results in staining, unpleasant smell, discoloration, loss in mechanical properties, and/or health related problems. 4
Consumers’ attitudes toward the removal of harmful microbes from fabrics has stimulated intensive research and development efforts to develop functional finishes for high performance textile products that exhibit excellent antimicrobial activity.4–7
A wide range of antimicrobial agents based on natural products (e.g., chitosan, neem, aloe vera, sericin, natural dyes, and enzymes),8–12 or synthetic bioactive compounds (e.g., metal salts, quaternary ammonium compounds, 13 N-halamines, triclosan, antibiotics, noble metal nanoparticles, and metal oxides)5,14–20 are available.
Ag-nanoparticles (Ag-NPs) are a promising option for antimicrobial finishing of textile materials due to their proven antimicrobial activity against many pathogenic microorganisms, stability, and high-surface-to volume ratio.5,21Biosynthesis of Ag-NPs provides a cost-effective and energy-saving alternative to chemical and physical methods.22–27
In addition, numerous methods have been developed for loading and binding Ag-NPs onto textile substrates including dip-coating methods, sonochemical coating, sputter deposition, in situ Ag-NP synthesis, electrospinning, and plasma treatment.5,28,29 Among these methods, plasma treatment provides an eco-friendly, economically feasible, and dry process for superficial fiber modification, surface activation, and functionalization. This method can positively affect the extent of Ag-NP loading and fixation onto modified substrates, by adequate control of plasma parameters (i.e., gas type, exposure time, gas flow, and pressure).30–32
In the present work, the biosynthesis of Ag-NPs using marine Bacillus sp. has been investigated. Ag-NPs were characterized by UV-Visible spectroscopy, scanning electron microscopy (SEM), and TEM imaging. Loading of Ag-NPs onto cellulosic-substrates via O2-plasma pretreatment has been facilitated to impart high antimicrobial protection.
Experimental
Materials
100% mill-scoured and bleached plain weave linen (207 g/m2), cotton (120 g/m2) and lyocell (135 g/m2, Lenzing AG) fabrics were used. Silver nitrate (AgNO3, Aldrich) and other chemicals used during this study were analytical grade.
Staphylococcus aureus and Escherichia coli for antibacterial testing were provided by the Microbial Chemistry Department of the National Research Centre.
Methods
Bacterial Isolation, Culturing, and Screening
Bacteria used to produce silver nanoparticles were obtained from seawater, algae, and sediment samples collected from different locations (Ras Sidr, Ain Sokhna, and Hurgada) throughout the Red Sea area in Egypt. Sediment samples were collected at a depth of 10 m. The salinity was 3.8% and the pH 8.0. Sediment and water samples were collected in sterile 50 mL conical tubes. Samples were kept cool during the expedition and processed as soon as possible upon return to the laboratory. Approximately 50 mg of wet sediment (or 1 mL of seawater) and both soft coral and algal homogenate samples were used to inoculate agar plates supplemented with 2 g/L sodium caseinate, 0.1 g/L asparagine, 0.001 g/L ferrous sulfate, and 0.075 g/L ferric citrate dissolved in natural seawater. The final pH of the medium was adjusted to 8.0 before sterilization. The agar medium was prepared with 100% natural seawater sterilized by filtration. The inoculated plates were incubated at 28 °C for three weeks. After isolation, the colonies were purified by streak plate method and kept in 30% glycerol diluted in seawater medium at –70 °C and recultured as required.33,34
The isolated bacteria were grown on nutrient agar plates containing a final concentration of 3.5 mM AgNO3. AgNO3 (the exact weight) was dissolved in a small amount of distilled water and sterilized separately by autoclaving at 121 °C for 20 min, then left to cool. This was added to the nutrient agar media just before solidification and then poured into Petri dishes. The nutrient agar plates (Sigma-Aldrich) had the following ingredients (g/L): agar (15), meat extract (1), peptone (5), NaCl (5), and yeast extract (2). Each plate was inoculated with a loop full of each bacterial isolate. The plates were incubated under aerobic conditions at 30 °C for 7 days. One of bacterial isolates that could grow on these plates was selected for Ag-NP biosynthesis studies. 33
Ag-NP Biosynthesis
Scale up fermentation was performed using ten 1 L Erlenmeyer flasks each containing 200 mL of nutrient broth medium (silver nitrate was sterilized and added separately to give a final concentration of 3.5 mM) and each was inoculated using 10% of 24 h old inoculums prepared by inoculating a 250 mL Erlenmeyer flask containing 50 mL nutrient broth medium with a freshly prepared bacterial slant (24 hold).
The biomass formed was then harvested by centrifugation (2739g) at room temperature for 15 min. The culture supernatant was analyzed by UV/Vis spectroscopy as well as TEM. The Ag-NPs were harvested by centrifugation at 9447g, but for UV measurements, the culture supernatant was used as is.
For comparison, four Erlenmeyer flasks containing nutrient broth, nutrient broth with AgNO3, culture medium with isolate and nutrient broth, as well as culture medium containing a final concentration of 3.5 mM AgNO3, Fig. 1b (1-4), were incubated under similar experimental conditions. Upon visual observation, the culture medium (nutrient broth inoculated with bacterial cells) incubated in the presence of AgNO3 showed a color change from yellow to brown, whereas no color change was observed in culture medium without AgNO3 and in AgNO3 without the culture medium. These control experiments indicate that Ag+ ion reduction was not just a thermal process. The bacterial isolate was partially identified. 35

Bacteria isolate growth (a) and color change (b).
Plasma Pretreatment
Cellulosic substrates were placed between two electrodes of the atmospheric pressure dielectric barrier discharge reactor (APDBD, Fig. 2). The flow rate of the oxygen working gas was kept constant at 3 L/min. Samples were exposed to the plasma for 45 s. 36
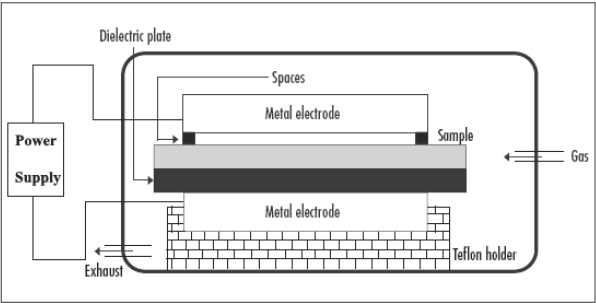
A schematic diagram of the dielectric barrier discharge plasma system.
Post-treatment with Ag-NPs
O2-plasma treated and untreated fabric samples were post-treated with an Ag-NP solution using a 20:1 liquor-to-fabric ratio, pH 9 at 65 °C for 30 min, followed by thoroughly washing with distilled water to remove excess and unfixed nanoparticles. 18
Testing
UV-Visible spectroscopy (Shimadzu UV-2450) was used to qualitatively confirm the presence of Ag-NPs in the reaction medium after 7 days. Ag-NPs have the ability to produce absorption peak in the wave length region 400–440 nm.
Durability to washing was conducted according to AATCC Test Method 61(2A)-1996, where each cycle was equivalent to five home laundries under ambient conditions.
TEM analysis was used to determine the size of the bio-synthesized Ag-NPs using a JEOL model 1200EX electron microscope operated at an accelerating voltage at 120 k V. Specimens for TEM measurements were prepared by dissolving a drop of colloid solution on a 400-mesh copper grid coated by an amorphous carbon film and evaporating the solvent in air at room temperature.
SEM images of plasma-treated, Ag-NP loaded linen, cotton, and lyocell were obtained with a JEOL JXL-840A electron probe microanalyser, equipped with energy disperse X-ray spectroscopy (EDX) for composition analysis. Samples were mounted on the stub with double stick adhesive tape and coated with gold in a S150A sputter coater unit (Edwards). The gold film thickness was 150 Å.
The Ag content in the post-treated fabric samples was determined by a fame atomic absorption spectrophotometer (GBC-Avanta) as follows: 0.5 g of dried fabric sample was dissolved in 10 mL of 72% sulfuric acid at 3 °C. A 0.5 mL aliquot of this solution was diluted up to 25 mL with a 0.06 M Na2HPO4 /0.02 M NaOH buffer solution before analysis.
The agar plate method was used to evaluate the antimicrobial activities of untreated and treated substrates. This disc diffusion test was done according to Collins and Lyne. 37 The antimicrobial activities of the textile specimens were tested against two strains of microorganism: S. aureus (G+ve bacteria) and E. Coli (G-ve bacteria). The bacterial test microbes were grown on nutrient agar (DSMZ1) medium (g/L): beef extract (3), peptone (10), and agar (20). The culture of each microorganism was diluted with sterile distilled water to 107–108 CFU/mL. Sample (film) discs (10-mm diameter) were located on the surface of the agar plates (10-cm diameter containing 25 mL of solidified media). The discs were placed on inoculated agar plates and incubated for 24 h at 37 °C. A corresponding plate without any discs was used as a negative control. An antimicrobial test on untreated textile was considered as a positive control. Sample antimicrobial activities were evaluated by the diameter clear zone that appeared around the film on the test microorganism. The results were averaged.
Results and Discussion
Bacterial Strain Selection
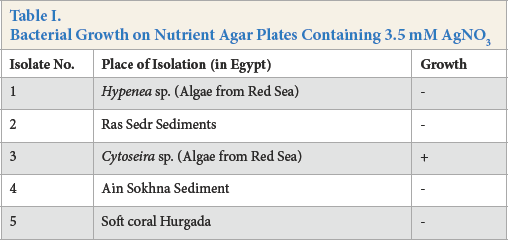
It is well known that silver is highly toxic to most microbial cells. However, several Ag-resistant bacterial strains are known. 33 In this work, a total of five marine bacterial isolates were screened for their ability to grow on nutrient agar plate medium containing a final concentration of 3.5 mM AgNO3 for 7 days under aerobic conditions at 30 °C.
Experimental results given in Table I signify that, among the five examined strains, only isolate No. 3 showed growth under the given conditions, and was selected for Ag-NP biosynthesis. Isolate No. 3 showed noticeable growth on the Ag-treated nutrient agar plates (Fig. 1a).
Bacterial Growth on Nutrient Agar Plates Containing 3.5 mM AgNO3
Moreover, the inset photograph (Fig. 1b) shows that there was a noticeable change in color from pale yellow to deep brown, especially in the case of sample No. 4, as a direct consequence of Ag-reduction and formation of Ag-NPs. 38
Isolated bacterial strain No. 3 was identified as a Bacillus sp. according to the method described by Holt, et al., 35 and was selected for Ag-NP biosynthesis.
Ag-NP Characterization
The UV-Visible spectrum of the biosynthesized Ag-NPs is shown in Fig. 3a. Absorption spectra of the formed Ag-NPs in the culture filtrate showed an absorption peak at about 410 nm due to surface plasmon excitation, which confirms the formation of Ag-NPs.
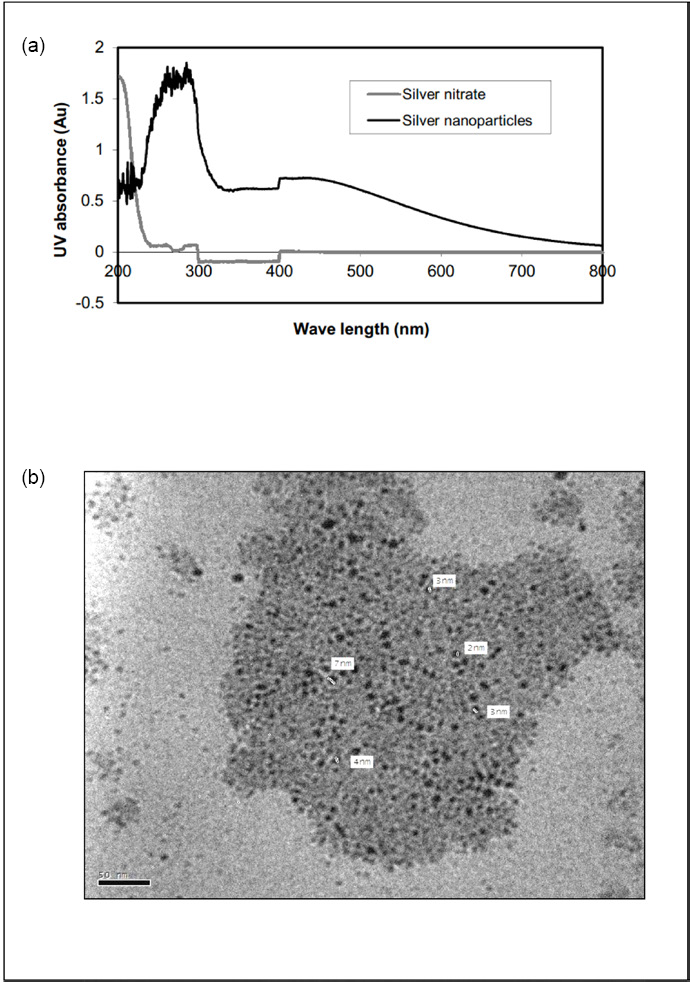
(a) UV-Vis spectra recorded as a function of 3.5 mM aqueous solution of AgNO3 with Bacillus sp. culture supernatant, (b) TEM image of Ag+ reduction to Ag0 by Bacillus sp.
The TEM image of Bacillus sp. reduced Ag-NPs is shown in Fig. 3b. This image showed spherical Ag-NPs in the range of 2 to 7 nm.
Based on the abovementioned results, the formation of Ag-NPs by the selected Ag-resistant bacteria (i.e., Bacillus sp.) reflect their ability to reduce water-soluble Ag+ions to form Ag-NPs, and to produce capping effects in Ag-N synthesis.24,33
Antibacterial Functionality and Durability
Antibacterial activity of linen, cotton, and lyocell cellulosic fabrics loaded with bio-synthesized Ag-NPs, without or with O2-plasma pretreatment, was tested against G+ve (S. aureus) and G-ve (E. coli) bacteria. For a given set of pre- and post-treatment conditions, results in Table II showed that O2-plasma pretreatment followed by Ag-NP treatment resulted in an increase in the Ag-content as well as a remarkable improvement in the antimicrobial activity of the treated substrates. Enhancement of the abovementioned properties reflects the positive impact O2-plasma pre-treatment had on surface modification and activation of the treated substrates.
Antibacterial Activity of Treated Cellulosic Fibers
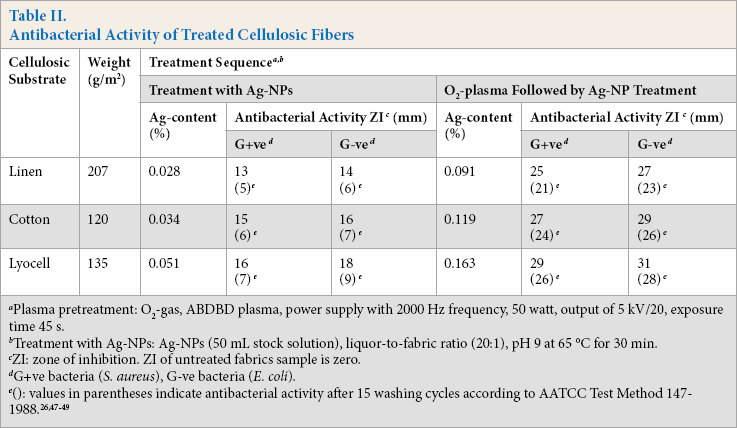
Plasma pretreatment: O2-gas, ABDBD plasma, power supply with 2000 Hz frequency, 50 watt, output of 5 kV/20, exposure time 45s.
Treatment with Ag-NPs: Ag-NPs (50 mL stock solution), liquor-to-fabric ratio (20:1), pH 9 at 65 °C for 30 min.
ZI: zone of inhibition. ZI of untreated fabrics sample is zero.
G+ve bacteria (S. aureus), G-ve bacteria (E. coli).
Generation of oxygen-functional groups by O2-plasma treatment, especially –COOH groups, 18 enhanced the extent of hydrophilicity, wettability, and Ag-NP loading onto the cellulosic structure.10,18,39 Greater antibacterial activity of plasma-pretreated samples was attributed to a larger amount of Ag-NP loaded on the treated substrate. The antibacterial activity of the treated cellulosic substrates followed the decreasing order lyocell > cotton > linen regardless of the treatment sequence. Differences among treated samples in Ag-content, as well as in imparted antibacterial activity, can be discussed in terms of surface morphology, cellulose content, amorphous/ crystalline regions, fabric weight, and extent of surface modification and functionalization, which in turn affected the extent of Ag-NP retention and antibacterial efficacy.18,40 The noticeable increase in the antibacterial efficacy of plasma-treated, Ag-NP loaded fabric samples, even after 15 washing cycles, is a direct consequence of increased bonding of Ag-NPs via the plasma-generated polar groups, i.e. –COOH groups. The remarkable decrease in the antibacterial activity of Ag-NP treated fabric samples that were not plasma pre-treated is attributed to the physically adhered Ag-NPs onto the fabric structure, resulting in a lower extent of fixation and easier removability compared with plasma-treated samples.
The significant antibacterial effect of Ag-NPs against both G+ve and G-ve bacteria results from damage to the bacterial membrane followed by an increase in cell permeability, and an uncontrolled transport through the cytoplasmic membrane that could result in cell lysis and ultimately cell death. Also, Ag-NPs (Ag0) release Ag+ ions, as shown in Eq. 1.41–45
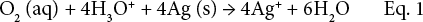
Ag+ ions can interact with the phosphorus moiety in DNA, leading to DNA replication inactivation, enzyme inhibition via reaction with cellular enzyme –SH groups,20,29 and/ or generation of oxygen radicals that can oxidize bacterial molecular structure.3,5 This means that antibacterial activity of the loaded Ag-NPs can be due to both the Ag-NPs as well as Ag+ ions.
Additionally, the results in Table II demonstrate that the G-ve bacterium has lower resistance against the imparted antibacterial property of the loaded Ag-NPs than the G+ve bacterium, which can be attributed to the thicker cell wall of G+ve (S. aureus) than G-ve (E. coli), keeping other parameters constant.28,46
SEM & EDX
Fig. 4 illustrates the SEM images and the corresponding EDX spectra for plasma-treated Ag-NP loaded linen, cotton, and lyocell fabric samples. The EDX spectra confirmed the existence of silver, along with carbon and oxygen, on the surface of treated samples.
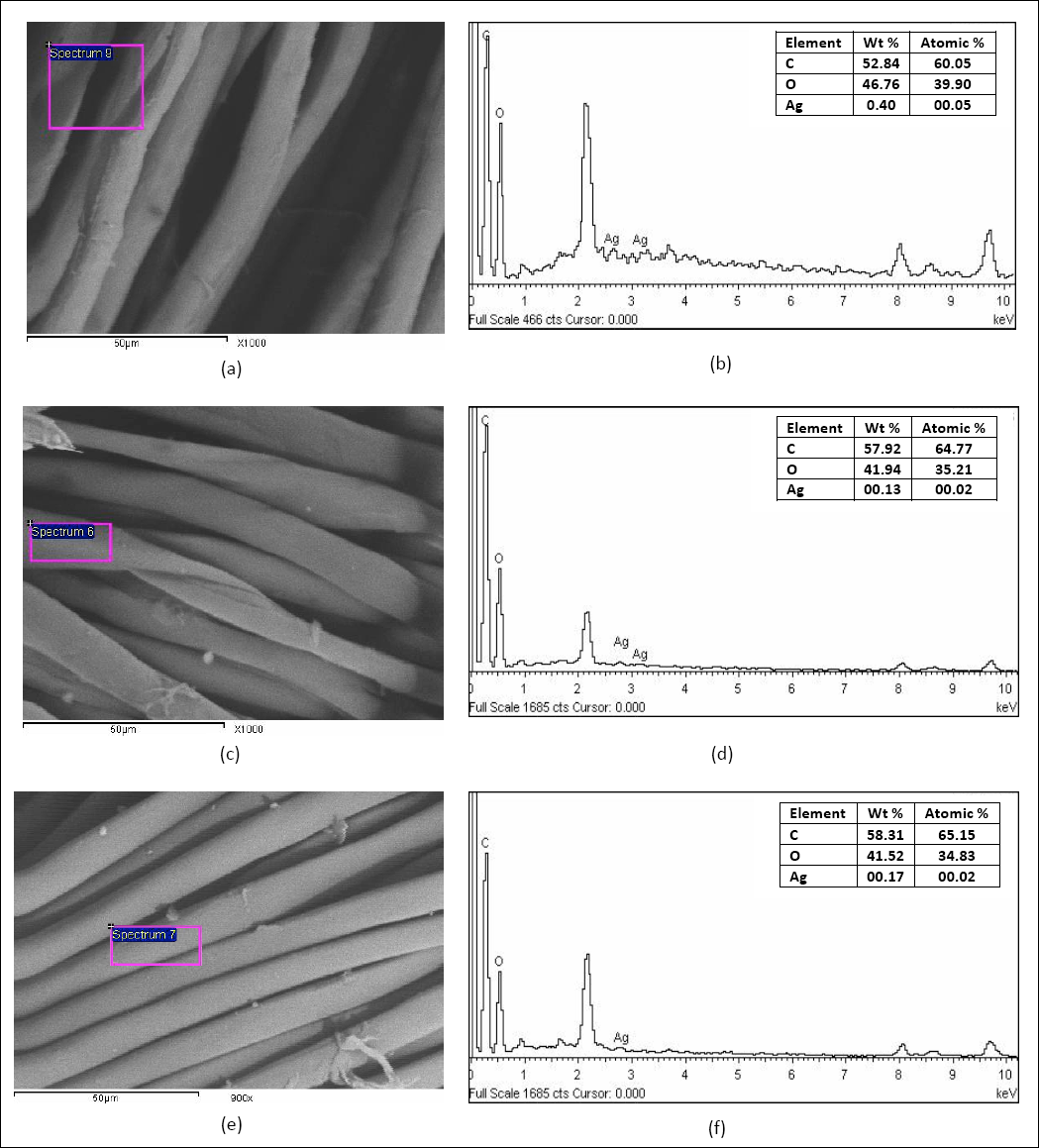
(a, b) SEM images and EDX spectrum of plasma-treated AgNP-loaded linen, (c, d) SEM images and EDX spectrum plasma-treated AgNPs-loaded cotton with Ag-NPs, and (e, f) SEM images and EDX spectrum plasma-treated AgNPs-loaded lyocell with Ag-NPs.
Conclusion
Ag-NPs of 2 to 7 nm in size were biosynthesized using Bacillus sp. and subsequently loaded on O2-plasma pre-activated linen, cotton, and lyocell fabrics. Ag-NP loaded cellulosic fabrics exhibited a remarkable antibacterial activity against S. aureus (G+ve) and E. coli (G-ve). The O2-plasma pretreatment positively affected the durability of the imparted antibacterial activity even after 15 washing cycles. The extent of surface activation, and Ag-NP deposition onto plasma-treated fabric surface, as well as the extent of the enhancement in the antibacterial functionality, was governed by the type of cellulosic substrate.
Future work will include analysis of antibacterial activity using AATCC Test Method 100, statistical analysis of antibacterial activity and Ag-content determination, and pilot scale studies to evaluate potential economic and ecological advantages.
Footnotes
Acknowledgment
The authors gratefully acknowledge financial support from National Research Centre, Cairo, Egypt, through grant number P90302.
