Abstract
The functional recovery of skeletal muscles after peripheral nerve transection and microsurgical repair is generally incomplete. Several reinnervation abnormalities have been described even after nerve reconstruction surgery. Less is known, however, about the regenerative capacity of reinnervated muscles. Previously, we detected remarkable morphological and motor endplate alterations after inducing muscle necrosis and subsequent regeneration in the reinnervated rat soleus muscle. In the present study, we comparatively analyzed the morphometric properties of different fiber populations, as well as the expression pattern of myosin heavy chain isoforms at both immunohistochemical and mRNA levels in reinnervated versus reinnervated-regenerated muscles. A dramatic slow-to-fast fiber type transition was found in reinnervated soleus, and a further change toward the fast phenotype was observed in reinnervated-regenerated muscles. These findings suggest that the (fast) pattern of reinnervation plays a dominant role in the specification of fiber phenotype during regeneration, which can contribute to the long-lasting functional impairment of the reinnervated muscle. Moreover, because the fast II fibers (and selectively, a certain population of the fast IIB fibers) showed better recovery than did the slow type I fibers, the faster phenotype of the reinnervated-regenerated muscle seems to be actively maintained by selective yet undefined cues.
T
Reinnervation requires a fine interplay between nerve and muscle involving the regeneration of the injured nerve, the proper guidance of the regenerated axons to their target muscles, and muscle recovery from the denervation phenotype. Regarding nerve regeneration and guidance, several abnormalities have been described, such as axon loss after reconstructive surgery (Mackinnon et al. 1991; Lloyd et al. 2007), partial cross-reinnervation caused by foreign axons (Bodine-Fowler et al. 1997; Gramsbergen et al. 2000), or a high proportion of polyneurally innervated motor endplates (Ijkema-Paassen et al. 2002). In muscle recovery, microarray analysis has revealed that reinnervation may directly influence the molecular repertoire of myofibers by altering the expression of several factors involved in either neural regulation or fiber type determination (Zhou et al. 2006). It is known that the expression of slow myosin heavy chain (MHC) type I is absolutely dependent on slow-type nerve activity (Esser et al. 1993; Schiaffino and Reggiani 1996). In contrast, the fast-type innervation pattern maintains different fast isoforms (MHCIIA, IIX, and IIB). However, denervation or muscle inactivity has a similar effect on fast-isoform expression, inducing slow-to-fast shift with a concomitant increase in hybrid fibers in slow-type muscles (Ohira et al. 2006; Patterson et al. 2006). Cross-reinnervation studies have also proved the plasticity of muscles by adapting the fiber phenotype to the properties of the newly innervating motoneurons (Vrbová et al. 1995).
Reinnervation has been shown to change the MHC composition of the target muscles at both mRNA and protein levels, although the extent and quality of alterations were highly dependent on the experimental procedure and the type of muscles examined (Bodine and Pierotti 1996; Huey and Bodine 1996; Sterne et al. 1997; Ijkema-Paassen et al. 2001, 2005; Wang et al. 2002; Zhou et al. 2006). In contrast to mixed- or fast-type rat hindlimb muscles, which are characterized by an effective muscle recovery after reinnervation, we and others described long-lasting motor endplate and morphological abnormalities in the slow-type soleus after sciatic nerve transection and immediate repair by autologous graft (Ijkema-Paassen et al. 2001, 2005; Pintér et al. 2003). Moreover, a slow-to-fast fiber type transition was also observed, which seemed to be stable even 60 weeks after nerve repair (Ijkema-Paassen et al. 2001, 2005). Because satellite cells, the main source of postnatal muscle regeneration and growth (Bischoff 1994), have been described to be activated yet not fully differentiated in reinnervated rat muscles (Zhou et al. 2006), we hypothesized that the impairment of the muscle regenerative machinery may also be involved in the incomplete recovery of the soleus muscle.
To prove this hypothesis, we induced muscle necrosis and subsequent regeneration in the reinnervated soleus muscle 3 months after nerve repair by using notexin, the purified toxin of the Australian tiger snake, and followed the morphological changes during the course of muscle regeneration until day 28. Notexin destroys myofibers and motor endplate structures, leaving satellite cells and axons essentially intact (Harris et al. 1975, 2000; Davis et al. 1991; Grubb et al. 1991). Motor endplates are reestablished on the surface of the regenerating muscle fibers in a few days, again building contacts to nerve terminals in intact innervation (Harris et al. 2000). This leads to the normalization of the nerve–muscle interaction and to an almost complete muscle recovery, regaining or even strengthening the innervation-dependent slow character of soleus by day 28 (Whalen et al. 1990; Sesodia and Cullen 1991, Lefaucheur and Sebille 1995, Zádor et al. 1996).
As opposed to normal regeneration, we found remarkable morphological alterations and motor end-plate abnormalities throughout the regeneration process in reinnervated soleus (Pintér et al. 2003), which corroborated our hypothesis about the abnormalities of the regenerative machinery. To gain more insight into the underlying mechanisms, we performed in the present study morphometric, immunohistochemical, and mRNA analyses to investigate whether reinnervated soleus is able to reproduce its original fiber type pattern and size distribution after necrosis and regeneration. Here we show that the reinnervated fast-type soleus, unable to recover its original (slow) character, became even faster after muscle regeneration. This finding indicates that the (fast) pattern of reinnervation plays a dominant role in the specification of muscle phenotype during regeneration, which can also contribute to the long-lasting functional impairment of the reinnervated muscle.
Materials and Methods
Animals and Experimental Design
Adult male Wistar rats (270–340 g, n=21), obtained from the University of Szeged, Domaszék, Hungary, were used for the experiments. In the first group of animals (defined as reinnervated group, n=7), the left sciatic nerve was exposed at the proximal part of the thigh by splitting the gluteal muscle under intraperitoneal Nembutal anesthesia (0.5% Nembutal, 1 ml/100 g body weight). For denervation and immediate repair, a 12-mm nerve segment was resected and used as an autologous nerve graft in a reversed position to mimic a clinically relevant nerve reconstruction method (applying different peripheral nerves, such as sural nerve, to repair nerve injuries) and to avoid growth of axons through small side branches from the nerve graft (Mackinnon et al. 1991; Gramsbergen et al. 2000, Ijkema-Paassen et al. 2002; Pintér et al. 2003). The affected muscles first became denervated and atrophied, but after several weeks, reinnervation took place (Ijkema-Paassen et al. 2002; Pintér et al. 2003). Three months after the operation, the reinnervated soleus muscles and the contralateral untreated ones were removed. In the second group of rats (defined as reinnervated-regenerated group, n=8), sciatic nerve transection and immediate repair were carried out as described for the reinnervated group. However, 3 months after nerve repair, muscle necrosis was induced in the reinnervated soleus by intramuscular injection of 20 μg notexin as described previously (Zádor et al. 1996). On the 28th day of regeneration (4 months after nerve repair), the reinnervated-regenerated soleus muscles (and their contralateral counterparts) were removed. A third group of unoperated, age-matched rats (defined as normal group, 540–580 g, n=6) served as controls. All muscles were weighed, frozen in isopentane cooled by liquid nitrogen, and kept at −80C until further processing for morphological, immunohistochemical, and mRNA analysis. Animals were sacrificed with an overdose of Na-pentobarbital. All animal experiments were approved by the Institutional Animal Care and Use Committee at the University of Szeged in accordance with the U.S. National Institutes of Health guidelines for animal care.
Morphological and Morphometric Analysis of Muscles
For morphological analysis, cryostat sections of 15-μm thickness taken from the midbelly region of soleus muscles were stained with hematoxylin-eosin. In addition, motor endplates were visualized according to Tago's standard method (Tago et al. 1986) based on their acetylcholinesterase (AChE) activity. Total fiber number was counted in normal, reinnervated, and reinnervated-regenerated muscles analyzing the whole cross section of each muscle (three muscles for each group) with the help of Olympus DP Soft software, Version 3.2. (Soft Imaging System GmbH; Münster, Germany). With the same software, mean fiber cross-sectional area (CSA) was calculated by measuring fiber CSA in at least three to four microscopic fields for each muscle (at least 300 fibers per muscle and three muscles for each group). Due to the high variability of fiber CSA in the different areas of muscle sections in both reinnervated and reinnervated-regenerated muscles, the microscopic fields were carefully taken for CSA analysis to represent the characteristic fiber populations and their proper ratio.
MHC Immunohistochemistry
For fiber type analysis, serial cryosections of 15-μm thickness of normal, reinnervated, or reinnervated-regenerated soleus muscles were subjected to MHC immunohistochemistry (Mendler et al. 1998). Sections were blocked in 5% non-fat milk powder/PBS, then incubated with mouse monoclonal antibodies BA-D5 (purified, 1:50), sc-71 (1:25), BF-F3 (1:10), and sc-75 (1:15) specific for MHCI (slow oxidative), MHCIIA (fast oxidative), MHCIIB (fast glycolytic), and MHCII (all types of fast fibers), respectively (DSMZ [National Resource Centre for Biological Material], Germany; Schiaffino et al. 1989). After incubation with the peroxidase-conjugated secondary antibody (rabbit anti-mouse; Dako, Denmark), immunocomplexes were visualized with diaminobenzidine staining with (sc-75) or without (BA-D5, sc-71, and BF-F3) nickel enhancement (in the presence of 0.15% nickel ammonium sulfate). Control sections were immunostained without primary antibodies. The number and proportion of different fiber types were calculated by counting total fiber number and fibers expressing different MHC isoforms with the help of Olympus DP Soft software, Version 3.2. (Soft Imaging System GmbH). Because of the lack of antibodies specific to MHCIIX, the number of pure (non-hybrid) IIX fibers was estimated according to the relative proportion of II, IIA, and IIB fibers (IIX = II − IIA − IIB); however, the calculation of IIX fiber subtypes (pure and hybrid) could not be established. The proportion of I/II hybrid fibers was estimated as follows: I/II (%) = type I (%) + type II (%) − 100 (%). The size (CSA) of slow MHCI– and fast MHCII–expressing fibers was measured on muscle sections immunostained with the MHCII-specific sc-75 antibody, as described above. In addition, the CSA of IIB fibers was analyzed on muscle sections stained with the MHCIIB-specific BF-F3 antibody.
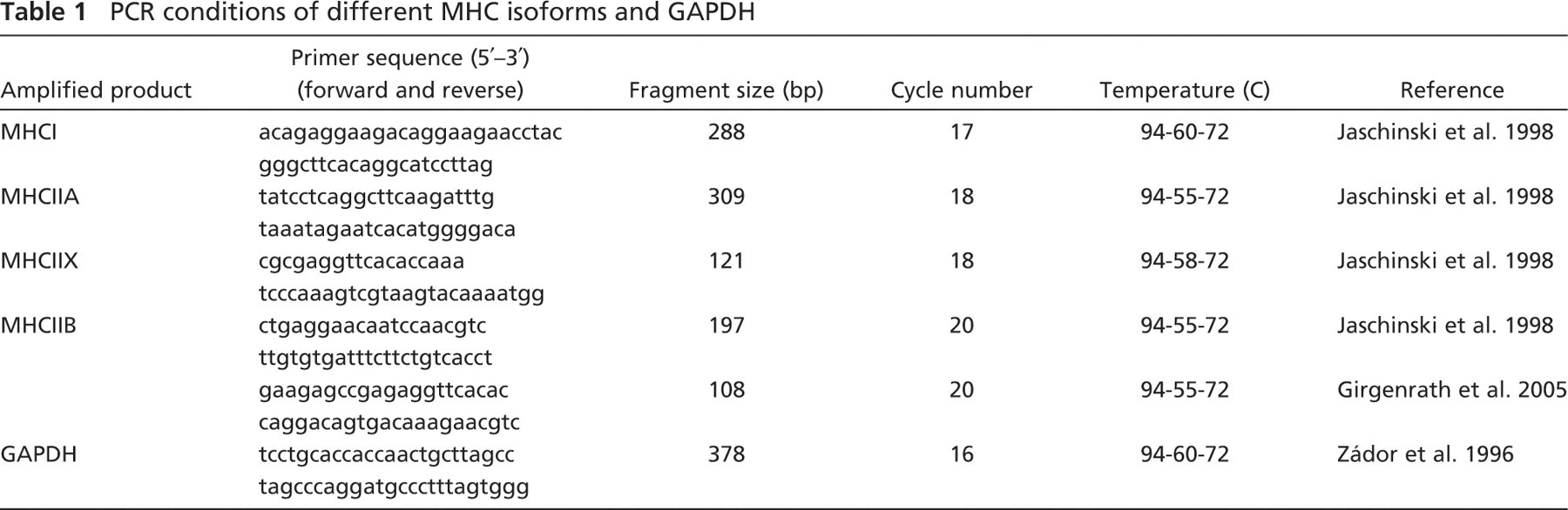
PCR conditions of different MHC isoforms and GAPDH
RT-PCR Analysis
Total RNA was isolated according to the acid-guanidinium-thiocyanate-phenol-chloroform method (Chomczynski and Sacchi 1987), and reverse transcription was performed from normal (n=3), reinnervated (n=4), and reinnervated-regenerated (n=5) muscles as previously described (Mendler et al. 2000). cDNAs for MHCI, MHCIIA, MHCIIX, and MHCIIB were amplified at 94C for 1 min, at the appropriate annealing temperature for 1 min, and at 72C for 1 min using 2 U/tube Taq polymerase (Table 1) (Zádor et al. 1996; Jaschinski et al. 1998; Girgenrath et al. 2005). We applied two different sets of primers for MHCIIB amplification, both of which produced essentially the same amplification pattern, outlined in the Results section. For quantification, we used the 197 bp–long PCR fragment of MHCIIB (Jaschinski et al. 1998). After polyacrylamide gel electrophoresis, specific PCR products stained with ethidium bromide were quantified with the help of Quantity One software (Bio-Rad Laboratories; Hercules, CA). For the normalization of MHC mRNA levels, the transcript level of the glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was proposed, which however, showed significant alterations in the different groups. Thus, total RNA gel electrophoresis was carried out, followed by the quantification of 28S rRNA, which served as an appropriate internal control because of its relative stability in the examined muscles.
Statistical Analysis
Each group of rats, for either protein or mRNA analysis, consisted of three to five animals. Data are reported as mean ± SEM. Statistical differences between groups were analyzed using either unpaired t-tests or one-way ANOVA, followed by a Newman-Keuls posttest (GraphPad Software, Inc.; San Diego, CA). For CSA measurements, 300–1200-fiber CSAs were summarized from three muscles per animal group. If fiber size distribution data violated the assumptions of normality and variance, they were also compared using a Kruskal-Wallis one-way ANOVA test, and differences between individual groups were analyzed by Dunn's posttest.
Results
Muscle Weight, Fiber Number, and Mean Fiber CSA
Because of nerve transection and immediate repair by graft, the sciatic nerve first degenerated, accompanied by the simultaneous transient denervation and inactivity of hindlimb muscles. However, the repaired nerve started to regenerate, reinnervating the hindlimb muscles ∼7 weeks after the operation (Ijkema-Paassen et al. 2002). In accordance with other morphological, motor endplate, and functional studies of sciatic nerve recovery (Hare et al. 1992; Sterne et al. 1997; Ijkema-Paassen et al. 2001, 2002), we found that by 3–4 months after microsurgery, the weight and morphology of reinnervated soleus muscles reached a near-optimal status and improved only slightly afterwards (Pintér et al. 2003). At this stage (3 months), the weight of the affected soleus was ∼72% that of its contralateral counterpart (Figure 1A, reinnervated group), and both total fiber number and mean CSA of reinnervated soleus were significantly lower than those of normal soleus (Figures 1B and 1C, respectively), showing a partial recovery of reinnervated soleus from the denervation phenotype. Upon inducing muscle necrosis and subsequent regeneration, the muscle weight of reinnervated-regenerated muscles (on day 28 of regeneration) tended to further decrease (from 72% to 65%) (Figure 1A, reinnervated-regenerated group), and even after 35 days of regeneration, it did not increase considerably (data not shown, Pintér et al. 2003). Similarly, a further significant drop was observed in the total fiber number of reinnervated-regenerated muscle (Figure 1B). Yet, the mean fiber CSA increased when compared with reinnervated muscles (Figure 1C). According to our previous findings, normally innervated soleus muscles undergo a near-complete regeneration, reaching the total fiber number and 70–80% of the mean fiber CSA by day 28 (Zádor et al. 1996 and unpublished data). Thus, our morphometric data indicate that there is a partial impairment of the regeneration process (involving mainly muscle weight and fiber number), because certain quantitative properties (i.e., fiber CSA) did not change the same way in reinnervated-regenerated soleus muscles when compared with reinnervated ones. However, the mean fiber CSA of all fiber types does not give information about the potentially altered recovery of different fiber populations (i.e., MHCI- versus MHCII- versus MHCIIB-expressing fibers) in reinnervated-regenerated muscle.
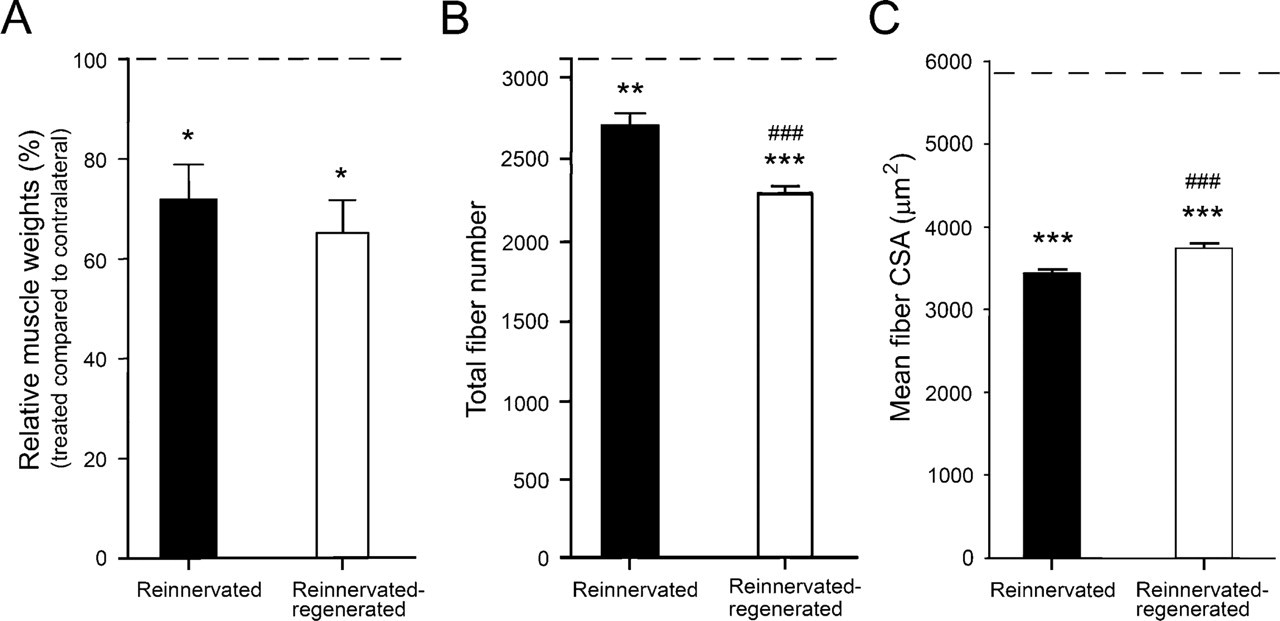
Relative muscle weights (
Muscle and Motor Endplate Morphology
As we revealed earlier in accordance with the literature (Ijkema-Paassen et al. 2001; Pintér et al. 2003), reinnervated soleus muscle became generally atrophied, but the extent of atrophy was variable in different fibers and fiber groups (compare Figures 2A and 2B). In addition, the accumulation of connective tissue was a characteristic feature of these muscles (Figure 2B, asterisk). This finding could also explain why the muscle weight of reinnervated soleus was not as much decreased (compared with contralateral) as expected on the basis of the remarkable decrease in total fiber number and mean fiber CSA in our experiments. After inducing muscle necrosis and regeneration, we found an even higher variability in fiber size compared with reinnervated muscles (Figure 2C). In addition, more than 80% of central-localized nuclei were counted, a parameter significantly different from that in normal regeneration (Figure 2C, arrows). The amount of connective tissue was also increased (Figure 2C, asterisk), and its accumulation pattern around single fibers was a novel finding distinct from that observed during reinnervation (compare Figures 2B and 2C) and normal regeneration (Whalen et al. 1990). In the morphological sense, these data are indicative of an altered, less-synchronous, and less-effective regeneration process in reinnervated soleus. To gain information about the innervation state of the affected muscles, we performed AChE staining of motor endplates as well (Figures 2D–2F; Pintér et al. 2003). In reinnervated muscles, we confirmed the findings of others (Ijkema-Paassen et al. 2002) who found highly variable motor endplates, several of which were characterized by abnormal (fragmented or granular) morphology (Figure 2E, arrowheads; see also inset). In reinnervated-regenerated muscles (i.e., on day 28 of regeneration), we observed motor endplates with similar morphological abnormalities coupled several times to smaller fibers (Figure 2F, arrowheads; Pintér et al. 2003). To summarize, our morphometric and morphological data revealed both quantitative and qualitative alterations in the regeneration process of reinnervated-regenerated muscles when compared with reinnervated and normal ones.
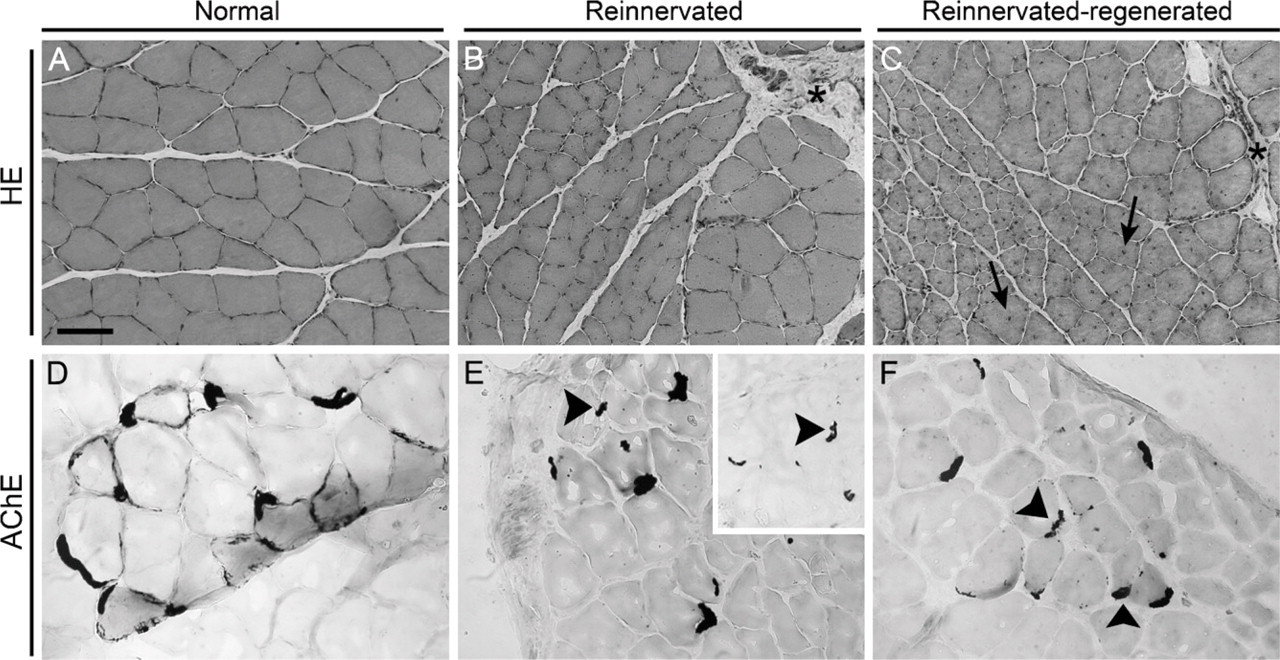
Morphology of the reinnervated and reinnervated-regenerated soleus muscles. Depicted are sections stained with either hematoxylin-eosin (HE) (
MHC Composition Based on Immunohistochemical Analysis
To analyze the possible effects of reinnervation and subsequent regeneration on MHC composition, serial crysections were immunostained with a set of monoclonal antibodies to distinguish and count (pure and hybrid) fibers expressing different MHC isoforms.
Reinnervated Muscles. In contrast to normal soleus, which contains 80–90% of slow type I fibers (not shown; Delp and Duan 1996), their number dramatically decreased to ∼33% after reinnervation (Figures 3A and 4A), and the amount of fast fibers significantly increased from ∼10–20% to 85% (Figures 3B and 4A). Although similar data have already been published in the literature (Bodine and Pierotti 1996; Huey and Bodine 1996; Ijkema-Paassen et al. 2002), the differential immunohistochemical analysis of the MHCII isoforms has not been carried out to date. We found that most of the fast II fibers were IIA fibers (44%) (Figures 3C and 4A), and no fibers expressed the fastest MHCIIB isoform (Figures 3D and 4A). The number of pure (not hybrid) IIX fibers was estimated according to the difference between II and IIA fibers, which proved to be ∼40% (Figures 3B–3D and 4A). Another notable change was the increased number (∼20%) of I/II hybrid fibers expressing both slow MHCI and fast MHCII (Figures 3A–3C and 4B) compared with those in normal soleus (less than 8%; Staron et al. 1999). Most of the I/II hybrids present in reinnervated soleus seemed to express MHCIIA as the fast isoform (Figures 3A and 3C). In addition, we observed variable staining intensity of the individual I/IIA hybrid fibers (Figures 3A and 3C, and insets), which may indicate a dynamic transition of type I to type IIA fibers, similar to the stepwise transformation of slow fibers toward fast ones in inactivity and cross-reinnervation studies (Pette and Vrbová 1992). Moreover, spatial grouping of the same MHC-expressing fibers was clearly detectable in all of the examined sections (Figures 3A–3C), which is a phenomenon seen after reinnervation (Ijkema-Paassen et al. 2001; Wang et al. 2002).
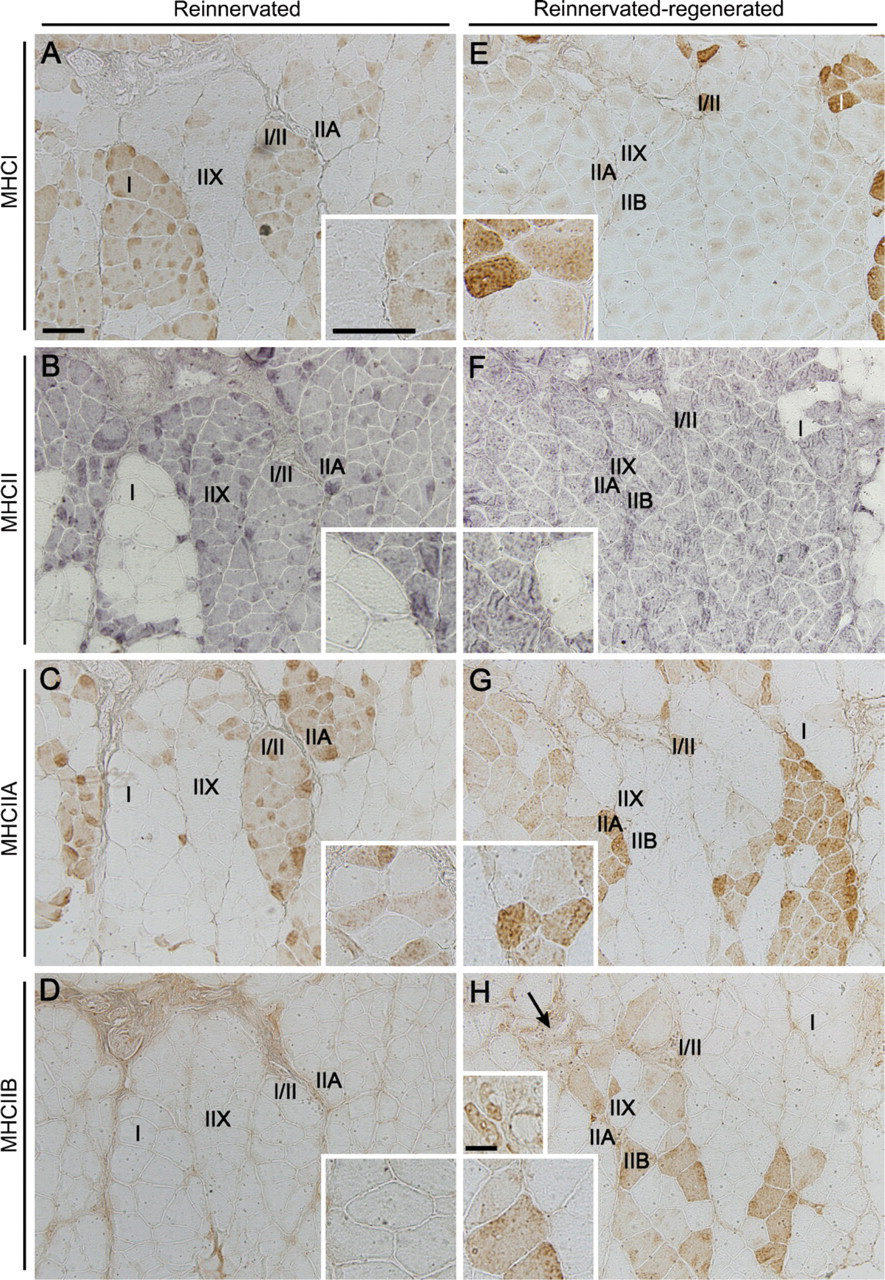
Immunohistochemical analysis of myosin heavy chain (MHC) isoforms in reinnervated versus reinnervated-regenerated soleus muscles.
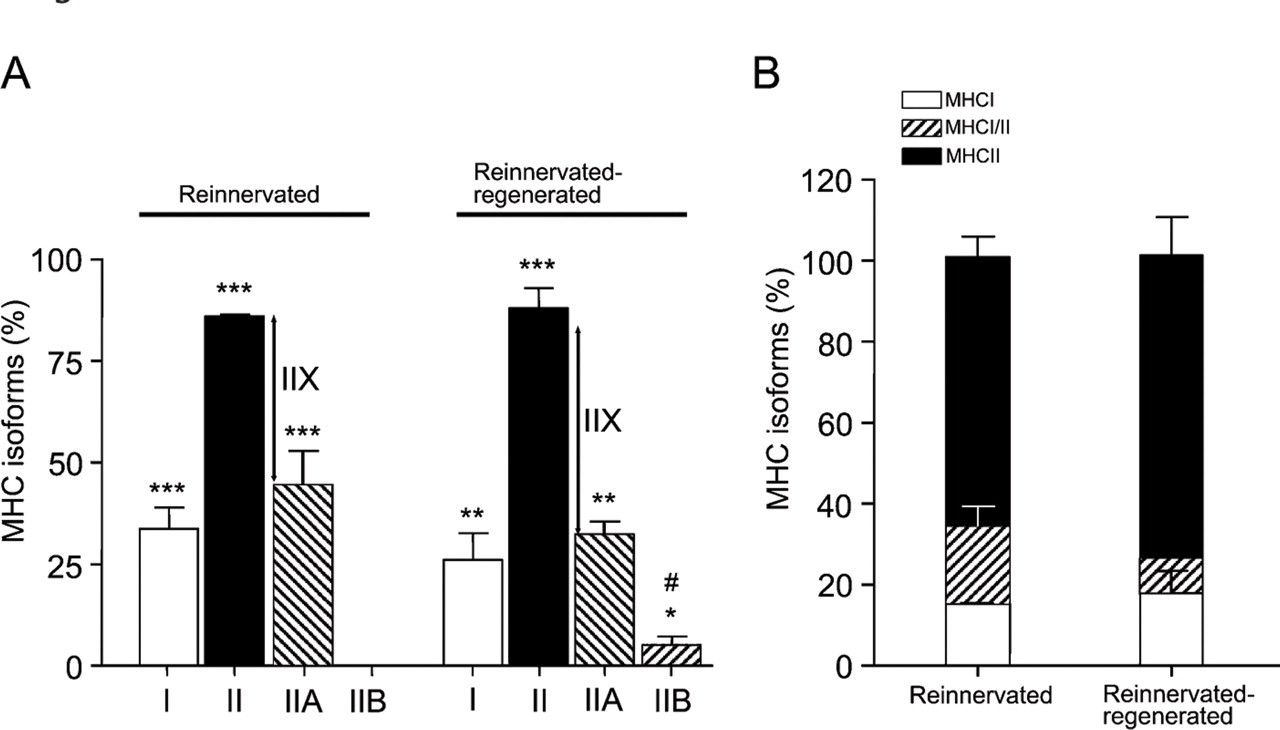
Fiber type distribution after reinnervation and subsequent regeneration of soleus muscle. (
Reinnervated–Regenerated Muscles. As a remarkable novel finding, we observed a further slow-to-fast (i.e., oxidative-to-glycolytic) shift in the phenotype of reinnervated soleus after regeneration took place. Although the ratio of slow type I to fast type II fibers (26/88%; Figures 3E and 3F and Figure 4A) did not differ significantly from that of the reinnervated muscles, it showed a clear tendency toward a faster phenotype. Among fast fibers, the number of fast oxidative IIA fibers tended to decrease from 44% to ∼32% (Figures 3G and 4A), and even the fastest glycolytic IIB isoform (undetectable in the reinnervated muscle) was expressed in a significant number of fibers after regeneration (∼5%; Figures 3H and 4A). The size and staining intensity of IIB fibers was notably variable. Very small, unmatured regenerated fibers with central nuclei were found mainly in areas where muscle regeneration was less effective, surrounded by increased amounts of connective tissue (Figure 3H, arrow; compare also small and large insets). On the other hand, larger fibers expressed MHCIIB in varying intensity (Figure 3H and large inset), which indicates that IIB fibers of fainter staining might be hybrid fibers coexpressing other fast isoforms (MHCIIX) as well. The estimated number of pure IIX fibers tended to increase from 40% to ∼51% (Figures 3F–3H and 4A). As further proof of a shift toward the faster phenotype, we found a relative increase in the ratio of pure fast II fibers at the expense of the number of I/II hybrid fibers, which decreased from 20% to ∼8% (Figure 4B). Although several type II fibers were probably hybrids coexpressing different fast isoforms (MHCIIA/IIX, MHCIIX/IIB, or MHCIIA/IIX/ IIB), we could not distinguish them, owing to the lack of a specific IIX antibody. To summarize our new findings, the regeneration process of the reinnervated soleus (reinnervation-regeneration model), in contrast to the uniformly slow-fiber composition of soleus muscle after normal regeneration, resulted in an even faster muscle phenotype with a notable presence of different populations of IIB fibers.
Mean Fiber CSA and Frequency Distribution of MHCI, MHCII, and MHCIIB Fibers
Because fiber size could be indicative of the innervation (atrophy) level of fibers, we aimed at analyzing whether there are different fiber populations in reinnervated-regenerated muscles compared with reinnervated ones. In normal slow-type soleus, CSAs of type I fibers were somewhat larger than those of type II fibers (5909 ± 55 μm2 and 5295 ± 79 μm2, respectively; Table 2; Delp and Duan 1996). In reinnervated muscles compared with normal muscles, type I fibers became smaller (4945 ± 110 μm2; Table 2). However, the frequency distribution revealed two main populations, one with a close-to-normal CSA and the other with a remarkably smaller CSA (Figure 5A, black bars, two peaks at ∼5500 μm2 and 3000–3500 μm2, respectively, n=318). These findings suggest that the larger fibers expressing MHCI were perfectly reinnervated by slow axons maintaining their original size, whereas smaller MHCI fibers may represent different populations, which have been either denervated but not transformed yet, or reinnervated by foreign (slow or fast) axons. Type II fibers seemed to be uniformly smaller-sized (2998 ± 31 μm2; Table 2; Figure 5B, black bars, n=1079) with less than 9% of fibers smaller than 1750 μm2 (Table 2; Figure 5B), indicating that most of them might be reinnervated.
Mean fiber CSA of MHCI, MHCII and MHCIIB fibers in normal, reinnervated, and reinnervated-regenerated soleus muscles
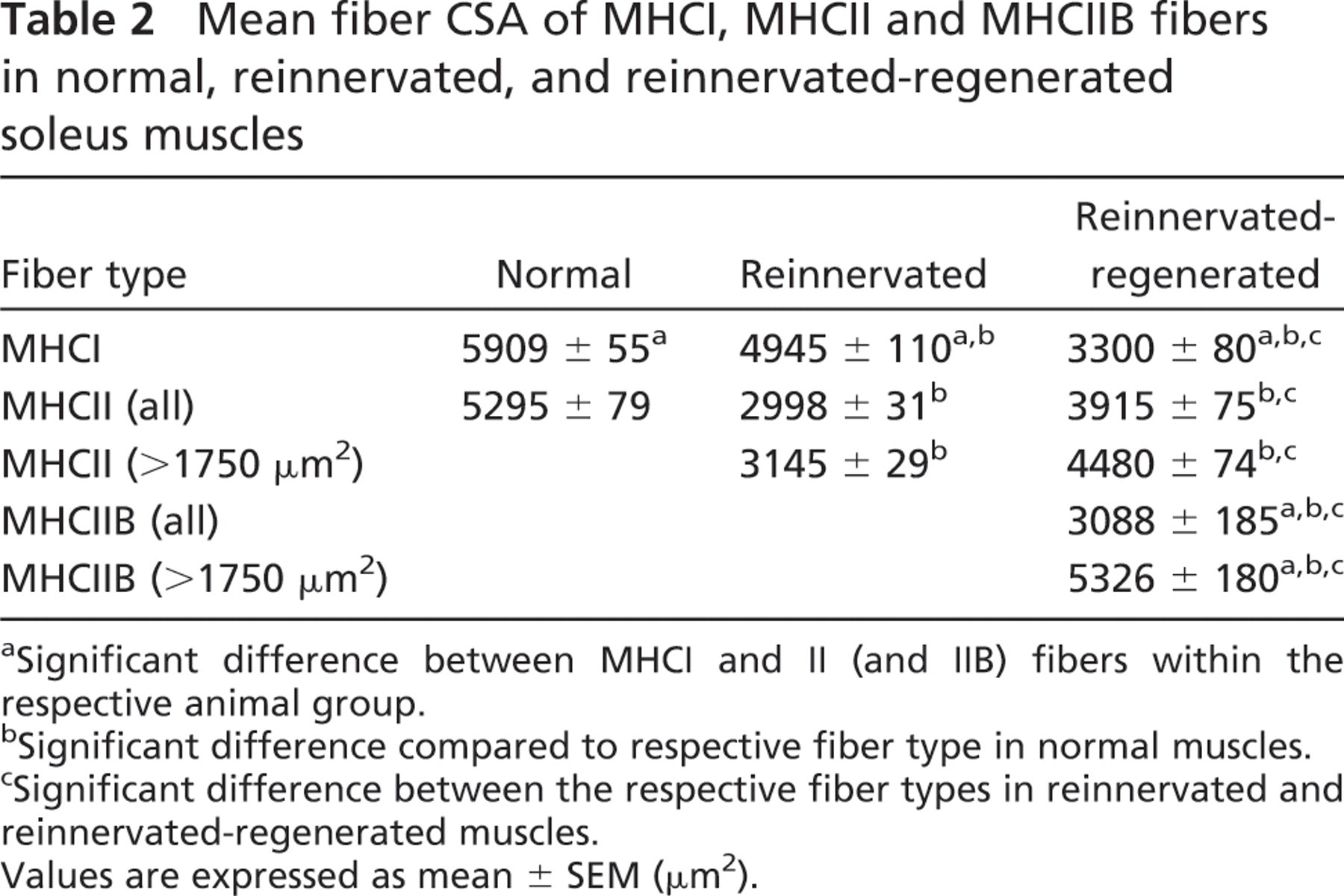
a Significant difference between MHCI and II (and IIB) fibers within the respective animal group.
b Significant difference compared to respective fiber type in normal muscles.
c Significant difference between the respective fiber types in reinnervated and reinnervated-regenerated muscles.
Values are expressed as mean ± SEM (μm2).
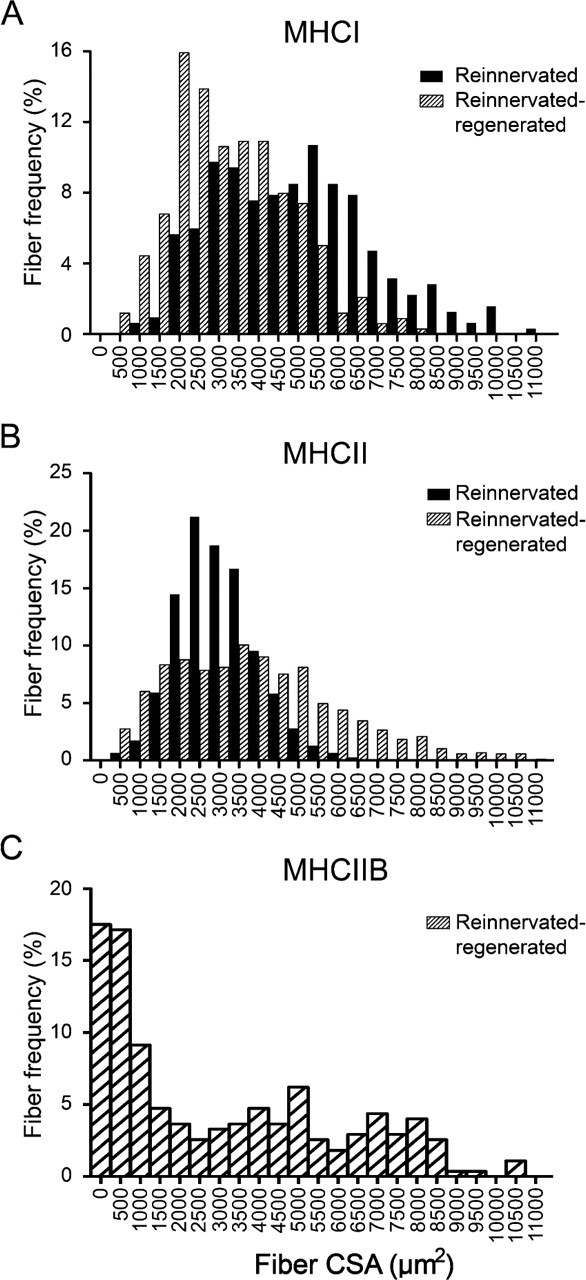
Frequency distribution of MHCI (
In reinnervated-regenerated muscles, the CSA of MHCI-expressing fibers became generally smaller, characterized by only one peak of a narrow-type distribution (3300 ± 80 μm2; Table 2; Figure 5A, hatched bars, n=339), showing that the recovery of slow fibers was impaired or delayed in the regeneration process. By contrast, MHCII fibers in reinnervated-regenerated soleus became larger than those in reinnervated soleus (3915 ± 75 μm2; Table 2; Figure 5B, hatched bars, n=865), and also larger than the regenerated MHCI fibers (Table 2; compare Figures 5A and 5B, hatched bars). The histogram of type II fibers showed a wide distribution profile starting at very small fibers on one tail and ending up in larger fibers on the other tail. The small fibers with characteristic central nuclei (CSA <1750 μm2) make up ∼17% of type II fibers and probably represent, at least a part of them, denervated unmatured fibers not able to fully differentiate in the absence of innervation. Indeed, as revealed by the size distribution of IIB fibers in reinnervated-regenerated muscles (3088 ± 185 μm2; Table 2; Figure 5C), about half of the fiber population was relatively small (CSA <1750 μm2), suggesting that a notable part of the smaller type II fibers expressed MHCIIB, a default isoform in the denervated regenerated fibers (see also Figure 3H, small inset; Jerkovic et al. 1997). However, in the other half of the IIB fibers (CSA >1750 μm2), mean fiber CSA was remarkably increased when compared with the similar population of MHCII fibers (5326 ± 180 μm2 and 4480 ± 74 μm2, respectively; Table 2). This interesting finding clearly indicates at least two populations of MHCIIB fibers (see also Figure 3H, small and large insets): a small one with denervation phenotype and a large one, which seems to be relatively hypertrophized compared with other type II fibers. In summary, we found in reinnervated-regenerated muscles that with the exception of the IIB-expressing small denervated fibers, fast type II fibers showed better recovery than did slow type I fibers at day 28 of regeneration, and a selective hypertrophy was seen in a certain population of IIB fibers.
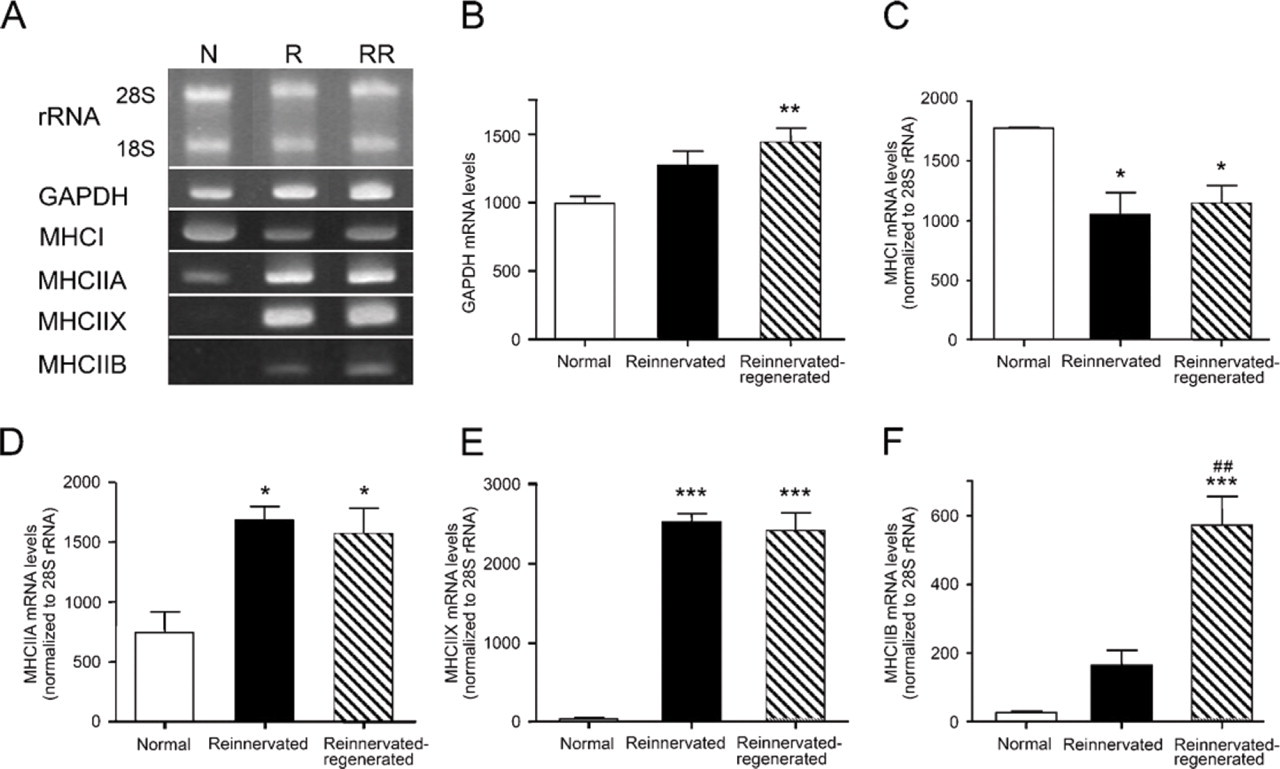
mRNA levels of MHC isoforms and GAPDH in normal, reinnervated, and reinnervated-regenerated soleus muscles.
mRNA Levels of MHC Isoforms Detected by RT-PCR
To determine whether the alterations detected at the level of fiber types are reflected in the mRNA level, we performed RT-PCR analysis for the different MHC isoforms in normal, reinnervated, and reinnervated-regenerated soleus. After total RNA gel electrophoresis, we used 28S rRNA as an internal control, the level of which proved to be stable in all of the examined animal groups (Figure 6A). By contrast, the mRNA levels of the glycolytic enzyme GAPDH, another internal control candidate, increased significantly after regeneration, indicating a glycolytic shift in the metabolism of the treated muscles (Figures 6A and 6B). In normal soleus, MHCI is known to be the predominant form; however, a significant decrease took place after reinnervation and subsequent regeneration (Figures 6A and 6C). In contrast, MHCIIA mRNA levels increased significantly with both treatments, compared with normal rats (Figures 6A and 6D), and a similar significant increase in MHCIIX expression was also observed in each treated group (Figures 6A and 6E). In contrast with the immunohistochemical alterations, no significant difference in MHCIIA or MHCIIX mRNA levels was found in the reinnervated versus reinnervated-regenerated groups, which is reasonable if one takes into account that total RNA was isolated from a whole muscle instead of performing single-fiber analysis. However, in accordance with immunohistochemical results, MHCIIB transcript levels were virtually not detectable in normal soleus, but a clear elevation (p=0.05) was seen in reinnervated muscles and a further highly significant increase took place in reinnervated-regenerated soleus (Figures 6A and 6F). These findings suggest that IIB fibers could be detectable with immunohistochemistry (in reinnervated-regenerated soleus) only when MHCIIB transcript levels were significantly elevated, whereas MHCIIB protein expression could not be visualized in individual fibers when the transcript levels were lower (in normal and reinnervated soleus). Our results are in agreement with the predominant transcriptional regulation of MHC isoforms described in slow-type skeletal muscle under several conditions (Schiaffino and Reggiani 1996; Baldwin and Haddad 2001; Pandorf et al. 2006).
Discussion
The functional impairment of skeletal muscles after nerve injury, despite optimized surgery, is an intriguing medical problem (Mackinnon and Dellon 1988). We hypothesized that in addition to several reinnervation disturbances, the regenerative capacity of the reinnervated muscle is also altered, as indicated previously by morphological and motor endplate abnormalities (Pintér et al. 2003). Indeed, here we provide evidence that fiber type composition of the reinnervated-regenerated rat soleus muscle is faster than that we observed in reinnervated soleus. This finding suggests that the (fast) pattern of reinnervation plays a dominant role in the specification of fiber phenotype during regeneration, which can also contribute to the long-lasting functional impairment of the reinnervated muscle. Moreover, because the fast II fibers (and selectively, a certain population of the fast IIB fibers) showed better recovery (larger CSA) than did slow type I fibers, the faster phenotype of the reinnervated-regenerated muscle seems to be actively maintained by selective yet undefined cues.
We applied a rat model of sciatic nerve transection and immediate repair by graft to analyze the regenerative machinery of reinnervated muscles (Pintér et al. 2003). In reinnervated soleus, our previous morphological findings, as well as the present results on the slow-to-fast fiber type transition, were consistent with those of another group (Ijkema-Paassen et al. 2001), who used a very similar rat model and described time-dependent abnormalities without a significant improvement in muscle recovery, even after 60 weeks of reinnervation (Ijkema-Paassen et al. 2005). In the present study, however, we performed a more detailed analysis of the slow, hybrid, and fast fibers at both immunohistochemical and mRNA levels, examining also the size (atrophy state) of the different fiber populations, because these data are necessary for judging the reinnervation level of fibers before the induction of regeneration. Our new results, namely the significantly high number of I/II hybrids, as well as the increase in the number of both MHCIIA- or MHCIIX-expressing fibers, accompanied by respective changes in their mRNA levels, suggest a gradual slow-to-fast fiber switch in reinnervated soleus, which is regulated predominantly at the mRNA level (Schiaffino and Reggiani 1996; Baldwin and Haddad 2001; Pandorf et al. 2006).
The explanation for fiber type transition in reinnervated soleus might be either the partial denervation of certain fiber populations because of axon loss (Mackinnon et al. 1991; Lloyd et al. 2007) and/or the random reinnervation of muscles by axons of different motoneuron pools (Bodine-Fowler et al. 1997; Gramsbergen et al. 2000; Ijkema-Paassen et al. 2001). In accordance with the literature (Ijkema-Paassen et al. 2001), we found that the total fiber number significantly decreased in reinnervated soleus when compared with normal soleus, which suggests that at least one population of denervated fibers, delayed in reinnervation, has been eliminated. However, the question remains: do all denervated fibers undergo complete elimination (by apoptosis or other mechanisms); are they replaced by new myofibers originating from proliferating satellite cells to some extent; or alternatively, do they exist for a longer time in the denervated stage (Tews et al. 1997; Borisov and Carlson 2000; Borisov et al. 2001, 2005; Tews 2002). In fact, Patterson et al. (2006) did not find any decrease in fiber number after 50-day denervation of rat soleus muscle, or any sign of the presence of regenerating fibers. On the basis of their findings, denervated fibers may exist for a longer time, expressing mainly fast MHC isoforms and forming several I/II hybrid fibers. However, we suggest that only a small proportion of fibers was still denervated 3 months after repair, as revealed by the difference in muscle weights and fiber diameter of denervated versus reinnervated soleus (Pintér et al. 2003), by the relatively small number of very small fibers in reinnervated muscles, and by the notably high proportion of muscle fibers recovering from notexin-induced necrosis. Thus, random reinnervation and the respective muscle activity pattern is a more likely explanation for fiber type and size transitions detected in reinnervated soleus (Bodine-Fowler et al. 1997; Gramsbergen et al. 2000; Ijkema-Paassen et al. 2002). It has been reported that only 14% of the original (slow) soleus motoneurons find their correct target following nerve repair, indicating that most of the reinnervating axons come from different motoneuron pools (Bodine-Fowler et al. 1997). In fact, slow fibers of normal soleus are remarkably larger than those of other hindlimb muscles and are also larger than most fast fibers, except IIB fibers (Delp and Duan 1996). We described here two populations of slow fibers, the larger one probably reinnervated by the original slow axons, and the smaller one, in which fibers may represent either denervated fibers on the way to transformation, or fibers reinnervated by foreign slow or (mismatched) fast axons. These mechanisms may even explain our results on the increased number of I/II hybrid fibers, a fiber pool that may match the one found to be polyneurally innervated in a similar animal model (Ijkema-Paassen et al. 2002). Similarly, the smaller yet remarkably uniform population of fast II fibers seems to be the consequence of a gradual fast transformation maintained by foreign axons reinnervating the soleus muscle. However, we could not detect the fastest IIB fibers in the reinnervated soleus. The reason might be that 3 months of reinnervation was not enough for a complete MHCI–MHCIIB fiber conversion and even longer reinnervation time would be required for this process (Ijkema-Paassen et al. 2001, 2005). On the other hand, even if not all the fast fibers were reinnervated, it seems that denervation does not favor the expression of those fast isoforms that are not present originally in the affected muscle, like the IIB fibers in soleus (Patterson et al. 2006).
Intriguingly, reinnervated fast-type muscles, in contrast with slow-type soleus, almost regained their normal phenotype after a transient change toward the slow character (Ijkema-Paassen et al. 2001; Wang et al. 2002; Zhou et al. 2006). The different reactions of slow- and fast-type muscles upon reinnervation may be explained by their different functions, because soleus is more dependent on the amount of mechanical activity (force generation), compared with fast-type muscles (Ohira et al. 2006). On the other hand, a few data support the hypothesis that the intrinsic (innervation-independent) properties of a particular muscle, e.g., different satellite cell populations and their functions, play a major role in the specification of fiber type, even after the removal of neural input (Patterson et al. 2006). If this is the case, alterations of the muscle regenerative capacity, influenced by both intrinsic and innervation-dependent mechanisms, can also contribute to the impaired phenotype of the reinnervated soleus.
The main question of our study was whether the regenerative machinery of the reinnervated soleus can reproduce the original muscle phenotype in both qualitative and quantitative senses. In the present study, we clarified that the total fiber number was significantly lower after 28 days of regeneration in reinnervated-regenerated muscles, compared with reinnervated ones. The “missing” fibers may represent a fiber population in reinnervated muscles that has not been properly reinnervated and that therefore could not maintain the subsequent growth of regenerated fibers. Otherwise, the mean fiber CSA of the existing fibers was even larger in the reinnervated-regenerated muscles than that in the reinnervated soleus (but smaller than that in normal soleus), indicating that the recovery of certain fiber populations was preferred after regeneration. Indeed, we detected here that even though slow-type regenerating fibers did not reach the size characteristic of those in reinnervated soleus, fast fibers were generally larger in reinnervated-regenerated soleus, compared with reinnervated muscles. Moreover, a clear transition toward a faster phenotype was observed, and even the fastest IIB fibers were present after regeneration. Because no reverse change occurred toward a slower phenotype even by day 35 (data not shown), it is unlikely that all these abnormalities are simple consequences of a delayed regeneration process resulting from additional innervation problems. Although notexin destroys motor endplates, several endplates were re-established at their original sites in both normally innervated and reinnervated soleus by day 5, and the axons seemed to recover by the end of regeneration (Harris et al. 2000; Pintér et al. 2003). Accordingly, the axon population reinnervating the soleus muscle after nerve repair should be essentially the same even after necrosis and subsequent regeneration.
Therefore, other explanations should exist for the phenotype shift accompanied by the appearance of IIB fibers during the regeneration process. In fact, it is the MHCIIB and IIX mRNAs and proteins that are expressed in the first few days of normal regeneration, at the time when destroyed motor endplates have not yet been re-established. After the recovery of motor endplates and slow-type innervation, both MHCIIX and IIB mRNAs and proteins decrease gradually in parallel with the increase in slow-type MHCI expression. In accordance with these data, denervated slow-type soleus expressed exclusively the fast MHCIIX and IIB mRNAs in the regeneration process (Esser et al. 1993; Jerkovic et al. 1997). Other reports also suggest that IIB fibers develop in the absence of neural stimulation (Butler-Browne et al. 1982; Schiaffino and Reggiani 1996; Fink et al. 2003). Thus, the relatively small MHCIIB-expressing fibers, often located in the connective tissue–rich areas in our experiments, certainly represent denervated fibers that could not accomplish MHC transitions and growth. However, the larger part of the MHCII-expressing fibers, and especially about half of the IIB fibers, reached a significantly larger size after regeneration, suggesting an active (fast-type) innervation of them. Thus, our results regarding fast-fiber transition can be interpreted as the impairment of the physiological fast-to-slow conversion during the regeneration process either due to denervation (small II and IIB fibers) or mainly, due to active maintenance of the (larger) fast fibers at the expense of the (smaller) slow ones. The latter finding is intriguing, particularly because it is the slow-fiber population that becomes dominant in normal soleus muscle regenerating from notexin-induced necrosis (Whalen et al. 1990). It cannot be ruled out that intrinsic muscle characteristics, such as a subpopulation of satellite cells determined to produce mainly slow-type or oxidative fibers (Kalhovde et al. 2005), are selectively impaired in reinnervated soleus, which in turn hampers the proper reaction to slow-type innervation, causing a delayed regeneration. This possibility might be tested in additional experiments at later stages of regeneration (i.e., several weeks after notexin treatment) to see whether the difference in the size of slow versus fast fibers would be still apparent. Another explanation for size differences might be that because of different cues, fast-type axons or their collaterals become more effective in reinnervated-regenerated muscles than do slow-type ones. Some recent reports have shown a definite role of active (fast-type) innervation in the expression of the MHCIIB isoform (Di Maso et al. 2000; Germinario et al. 2002). Moreover, from the view of several neurotrophic factors influencing peripheral nerve regeneration, neurotrophin-3 and neurotrophin-4 have been described to favor selectively the axon regeneration of fast (IIB) and slow motoneuron pools, respectively, playing an important role in determining the correct muscle phenotype (Sterne et al. 1997; Kingham and Terenghi 2006). Further investigations are needed to explore the significance of neurotrophins, especially of neurotrophin-3 in supporting the recovery of fast IIB (and II) fibers in the altered regeneration of the reinnervated soleus.
In conclusion, the morphological, fiber type, and size alterations detected in reinnervated-regenerated soleus could be the consequence of a complex process involving both innervation-dependent and/or -independent molecular mechanisms, the exploration of which may contribute to a more effective therapy for the functionally impaired reinnervated muscle.
Footnotes
Acknowledgements
This work was supported by grants from Medical Scientific Board, Hungary (ETT) 421/2003, ETT 008/2006, and Regional Science Center of the University of Szeged, Hungary (RET) 08/2004 National Technological Development Board, Hungary (OMFB)-0066/2005. We thank Balásházi Istvánné for the technical assistance in animal experiments.
