Abstract
Vitamin D deficiency is strongly associated with the risk of developing colorectal cancer (CRC). Because of the propensity of bioactive 1,25-dihydroxyvitamin D3 to cause toxic hypercalcemia, considerable effort has been directed to identifying safer drugs while retaining the efficacy of the parent compound. However, vitamin D precursors do not present toxicity concerns and may be sufficient for CRC chemoprevention or chemotherapy, providing the appropriate enzymes are present in colonic epithelia. We previously showed that CYP27B1 is present at equally high levels in the colon and CRC irrespective of differentiation but was not present in metastases. In this study we used quantitative immunohistochemistry to show that CYP27A1, converting D3 to 25-hydroxycholecalciferol, is present in increasing concentrations in the nuclei of normal colonic epithelia, aberrant crypt foci (ACF), and adenomatous polyps. Whereas total cellular CYP27A1 remains high in CRC and lymph node metastases, the amount of enzyme present in the nuclei decreases with tumor cell dedifferentiation while rising in the cytoplasm. Similarly, increasing amounts of the deactivating enzyme CYP24 are present in the nuclei of normal colonic epithelia, ACFs, and adenomatous polyps. Although the amount of total CYP24 decreases slightly in CRC as a function of tumor cell dedifferentiation and metastasis, location of this enzyme shifts almost entirely from the nuclear compartment to the cytoplasmic compartment. These data indicate that non-toxic vitamin D precursors should be sufficient for CRC chemoprevention, but that neither vitamin D nor its precursors may be sufficient for CRC chemotherapy.
V
In classic vitamin D biochemistry, cholecalciferol (D3) is generated by ultraviolet light acting at the level of the skin or is consumed in the diet. Cholecalciferol is transported to the liver whereupon 25-hydroxylase (CYP27A1), a member of the cytochrome P450 enzyme family, converts it to 25-hydroxycholecalciferol [25(OH)D3]. This pro-hormone is the major circulating metabolite of vitamin D and is the form that best reflects overall vitamin D status (Heaney 1999, 2004). From the liver, 25(OH)D3 is transported to the kidneys where it is further metabolized by 25-hydroxyvitamin D3-1α-hydroxylase (CYP27B1) to its bioactive form, 1,25-dihydroxyvitamin D3, [1,25(OH)2D3]. Both 25 (OH)D3 and 1,25(OH)2D3 are degraded to inactive forms by 24-hydroxylase (CYP24).
We recently quantified vitamin D receptor (VDR) and CYP27B1 expression in non-malignant, premalignant, and malignant tissues from human colon (Matusiak et al. 2005). We showed that CYP27B1 is present at equally high levels in normal colonic epithelium as in aberrant crypt foci (ACF), the earliest histopathological lesion associated with malignant transformation, as well as in adenomatous polyps and CRC, irrespective of tumor cell differentiation. In contrast, VDR levels were low in normal colonic epithelial cells and were increased in ACFs, adenomatous polyps, and in better-differentiated tumor cells but then declined as a function of tumor cell dedifferentiation. Both 1α(OH)ase and VDR levels were negligible in tumor cells metastasizing to regional lymph nodes. Using our novel technique for quantitative immunohistochemistry (Q-IHC) (Matkowskyj et al. 2000, 2003a, 2004, 2006), we also showed that the location of VDR shifted from being primarily nuclear in location in non-malignant human colonic epithelial cells to being primarily cytoplasmic in location with malignant transformation (Matusiak et al. 2005).
Because 1,25(OH)2D3 causes toxic hypercalcemia, its clinical utility is limited. Consequently, considerable effort has been expended attempting to identify non-toxic vitamin D analogs possessing the efficacy of the parent compound. Because, as we have shown (Matusiak et al. 2005), CYP27B1 is present in colonic epithelia, D3 alone may be sufficient for CRC chemoprevention and/or chemotherapy, providing that CYP27A1 and CYP24 are present in appropriate quantities and cellular compartments. Yet, of the few studies that have been performed evaluating the expression of these enzymes in human CRC, most evaluated expression at the mRNA level (Anderson et al. 2006).
To determine whether cholecalciferol may be useful for CRC chemoprevention and/or chemotherapy, we report here on cellular CYP27A1 and CYP24 protein expression in human ACFs, polyps, and CRCs of defined differentiation along with associated lymph node metastases. Our observations are particularly germane, given the increasingly appreciated role cytochrome P450 enzymes play in attenuating the effects of anti-cancer drugs (Bruno and Njar 2007). In concert with our previous study characterizing CYP27B1 expression in these same tissues, our findings suggest that cholecalciferol has potential for use in CRC chemoprevention but may be less efficacious for CRC chemotherapy.
Materials and Methods
Materials
CYP27A1 and CYP24 antibodies, goat anti-sheep horseradish peroxidase (HRP)-labeled antibody, goat anti-mouse IgG-HRP, and rabbit anti-goat IgG-HRP were from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA). Tris-buffered saline Tween-20 (TBST) wash buffer, target retrieval solution, protein block serum, antibody diluent, EnVision+ HRP, (DAB)-Rabbit System, DAB chromogen, and automated hematoxylin were all from DAKO (Carpinteria, CA).
Tumor Specimens and Histological Grading
Colon cancers were randomly selected from the University of Illinois at Chicago (UIC) Gastrointestinal Tumor Bank. All cancers were isolated at the time of surgery, whereas all ACFs and polyps were isolated at the time of endoscopy. All were formalin-fixed and paraffin-embedded by the Department of Pathology staff at the Jessie Brown VA Medical Center. Differentiation was assessed as previously described (Carroll et al. 1999; Glover et al. 2003; Matkowskyj et al. 2003b): well-differentiated tumors possessed well-formed glands and small regular nuclei in the face of well-formed glands, whereas the complete absence of gland formation or the presence of bizarrely shaped glands identified poorly differentiated tumors. In contrast, cells in moderately differentiated tumors were less columnar or frankly cuboidal, were less polar, and contained more dysplastic nuclei than those observed in well-differentiated tumors.
Q-IHC
Tissues were sectioned to a thickness of 4 μm using a Sakura Accu-Cut SRM 200 Rotary Microtome (Torrance, CA). Tissues were processed for antigen retrieval by rehydrating in graded alcohols, placing in auto/iodine for 1 min, rinsing in TBST buffer followed by Redusol (Fisher Scientific; Pittsburg, PA) for 2 min, and once again in TBST buffer. Slides were then placed in target retrieval solution at 100C for 20 min, allowed to cool to room temperature, and rinsed once again in TBST.
IHC was performed by hand using a two-step indirect immunoperoxidase technique. To do this, tissues were incubated in a 3% H2O2 solution to quench endogenous peroxidase activity, rinsed with TBST, blocked with protein block serum for 30 min, and rinsed in TBST. CYP27A1 and CYP24 primary antibodies were applied (1:20) as determined by antibody titration, overnight at 4C before rinsing with TBST. Next, rabbit anti-sheep HRP-labeled antibody was added for 1 hr (1:2000), rinsed thoroughly with TBST followed by incubation with DAB-chromogen for 5 min, and then counterstained for 2 min with hematoxylin. All tissues were then dehydrated in graded alcohols and xylene and coverslipped using Permount (Fisher Scientific). For all specimens, control tissues were processed identically and at the same time, except that they were not exposed to primary antibody. Thus, all differences between the experimental tissue and the control tissue are ultimately due to DAB identification of the relevant protein.
Chromogen abundance was quantified by Q-IHC as previously described (Matkowskyj et al. 2000, 2003a, 2004, 2006). Images of the relevant cells were saved in TIFF format, and the amount of chromogen per pixel determined by subtracting the mathematical energy of the control slide (i.e., not exposed to primary antibody) from that in the homologous region of the experimental slide (i.e., exposed to primary antibody). In all instances the relevant region (i.e., cytoplasm vs. nuclei) was isolated from the rest of the image using Photoshop (Adobe Systems; Palo Alto, CA) and used for chromogen quantification as previously described (Matkowskyj et al. 2000, 2003a, 2004, 2006). Chromogen quantity is expressed as value less energy units per pixel.
Standards and Statistics
In all instances, IHC quality control was achieved as follows. Because all runs were performed using a computer-controlled Dako Autostainer, conditions were identical at all times. Additionally, a section from a single tumor was included in all runs to control for experimental variability. The amount of chromogen as determined by Q-IHC revealed run-to-run variability <5%. All data are presented as mean ± SE. Data were evaluated using Stata (College Station, TX) with p<0.05 considered significant.
Results
Of the few studies examining the expression of CYP27A1 and CYP24 in the colon, all focused on mRNA, and none has assessed protein expression as a function of histology or tumor cell differentiation. To evaluate the expression of CYP27A1 and CYP24, we utilized commercially available polyclonal antibodies. The CYP27A1 antibody is a goat polyclonal directed to a 17-amino acid region located within amino acids 1-50 of Q02318, whereas the CYP24 antibody is a goat polyclonal antibody directed to a 20-amino acid segment within amino acids 100-150 of Q07973 (with the exact epitope being a trade secret).
We used these antibodies to evaluate the expression of CYP27A1 and CYP24 in normal colonic epithelium and in CRC precursors such as ACF and adenomatous polyps, as well as in resected human colon cancers and in adjacent non-malignant tissues. To do this, we randomly selected five ACF, five adenomatous polyps, and 10 CRC from the UIC Gastrointestinal Tumor Bank. Because CRC are heterogeneously differentiated (Jass et al. 1986; Steinberg et al. 1986; Carroll et al. 1999), we also could assess expression of these proteins as a function of tumor cell differentiation. Overall, the 10 CRCs studied—approximately equal in number by stage—contained 35 separate and distinct regions of well-differentiated tumor, 19 that were moderately differentiated and 13 that were poorly differentiated (Table 1). Each region evaluated contained between 75 and 400 cells (Table 1). Because we used sections including the tumor margin for all CRCs studied, we also had access to the adjacent normal colonic epithelium. Finally, we also studied all lymph nodes containing metastatic deposits from the CRCs described above (Table 1). Overall, ∼5425 cells in total were evaluated.
CYP27A1 expression was detected immunohistochemically in all colonic tissues evaluated (Figure 1). As determined by Q-IHC, CYP27A1 expression was exclusively noted in the nucleus of normal colonic epithelial cells (Figure 1A). Increasing CYP27A1 expression was observed in ACF (Figure 1B), the earliest histopathological lesion associated with malignant transformation (Takayama et al. 1998; Bird and Good 2000) and in adenomatous polyps (Figure 1C); however, the expression profile was the same as observed in normal colonocytes (Figure 1A) insofar as all protein was present in the nucleus. In contrast, although total cellular CYP27A1 expression was increased in well-differentiated (Figure 1D), moderately differentiated (Figure 1E), and poorly differentiated (Figure 1F) CRC cells as compared with what was detected in premalignant lesions, decreasing amounts of this enzyme were detected in the nucleus, whereas increasing amounts were detected in the cytoplasm. We also evaluated expression in tumors metastatic to regional lymph nodes. In all lymph nodes containing metastases, CYP27A1 was apparent in both the nuclei and cytoplasm (Figure 2A).
Characterization of human CRC studied
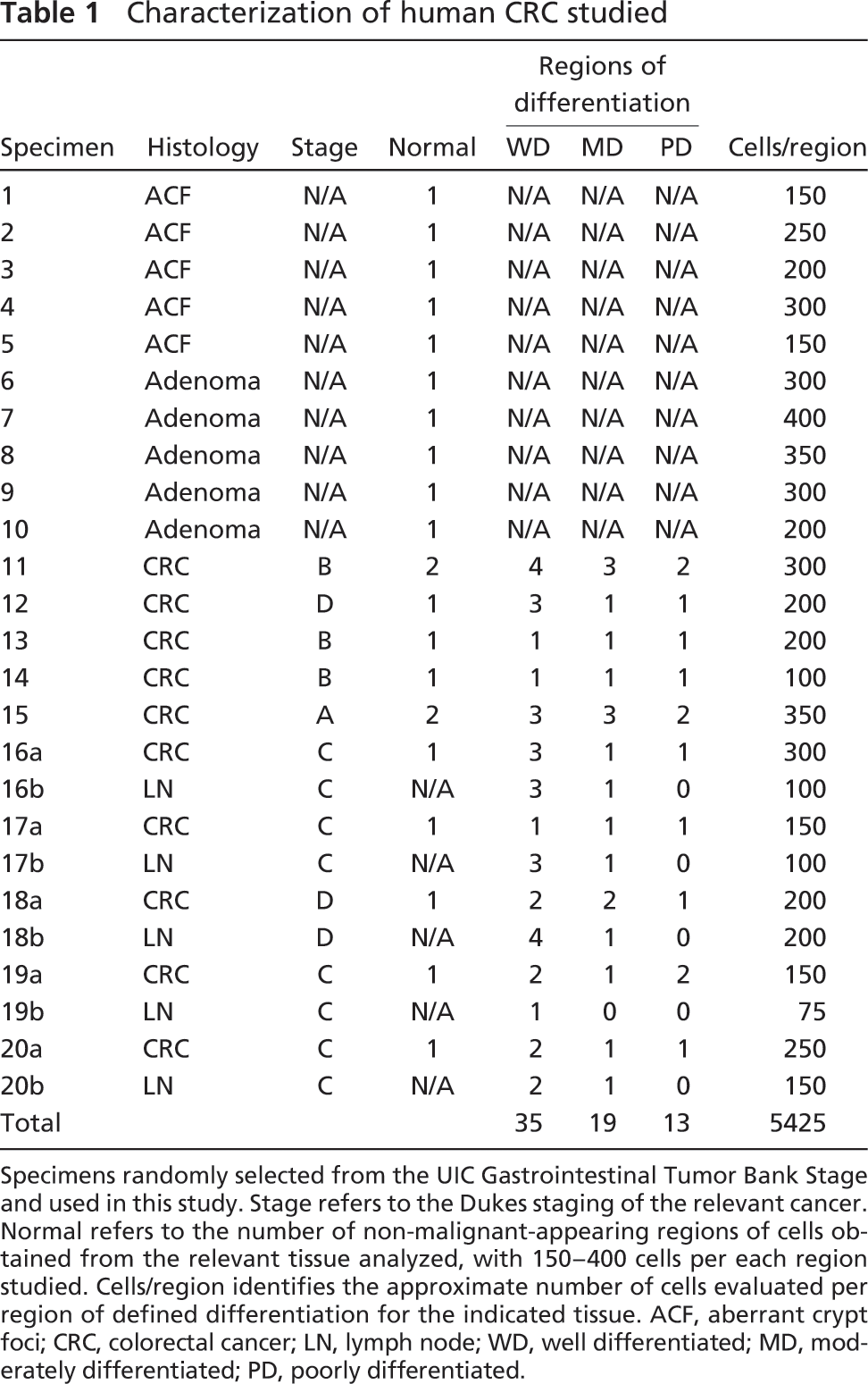
Specimens randomly selected from the UIC Gastrointestinal Tumor Bank Stage and used in this study. Stage refers to the Dukes staging of the relevant cancer. Normal refers to the number of non-malignant-appearing regions of cells obtained from the relevant tissue analyzed, with 150-400 cells per each region studied. Cells/region identifies the approximate number of cells evaluated per region of defined differentiation for the indicated tissue. ACF, aberrant crypt foci; CRC, colorectal cancer; LN, lymph node; WD, well differentiated; MD, moderately differentiated; PD, poorly differentiated.
We next evaluated CYP24 expression in these same tissues (Figure 3). Overall, the expression profile was similar to what we observed for CYP27A1 insofar as chromogen appeared to be restricted to the nuclei (Figure 3). Increasing nuclear CYP24 expression was observed in ACFs (Figure 3B) and in adenomatous polyps (Figure 3C). Following malignant transformation, CYP24 was also detected in the cytoplasm of primary CRC cells irrespective of differentiation (Figures 3D-3F) and in tumor cells metastatic to lymph nodes (Figure 2B).
We next quantified CYP27A1 and CYP24 expression in these tissues by Q-IHC (Matkowskyj et al. 2000, 2003a, 2004, 2006). We previously demonstrated that this approach quantifies immunohistochemically derived chromogen in a manner that accurately reflects the actual amount of protein present (Matkowskyj et al. 2004). Consistent with our empirical observations, CYP27A1 (Figure 4) and CYP24 (Figure 5) chromogen was increased in the nuclei of ACFs and adenomatous polyps (ANOVA, p<0.01) as compared with what was detected in normal colonic epithelial cells. Although nuclear levels of CYP27A1 and CYP24 in well-differentiated and moderately differentiated CRC cells was similar to what was observed in normal colonic epithelial cells, significantly decreased amounts were noted in poorly differentiated CRC cells (p<0.05). Whereas nuclear CYP27A1 remained detectable in tumor cells metastatic to lymph nodes (Figure 4), chromogen for CYP24 was not (Figure 5). In contrast to what we observed for nuclear expression of CYP27A1 and CYP24, neither could be detected in the cytoplasm of normal or premalignant neoplasms, whereas significant amounts of this protein were present in CRC cells irrespective of differentiation or metastasis (Figure 4 and Figure 5).
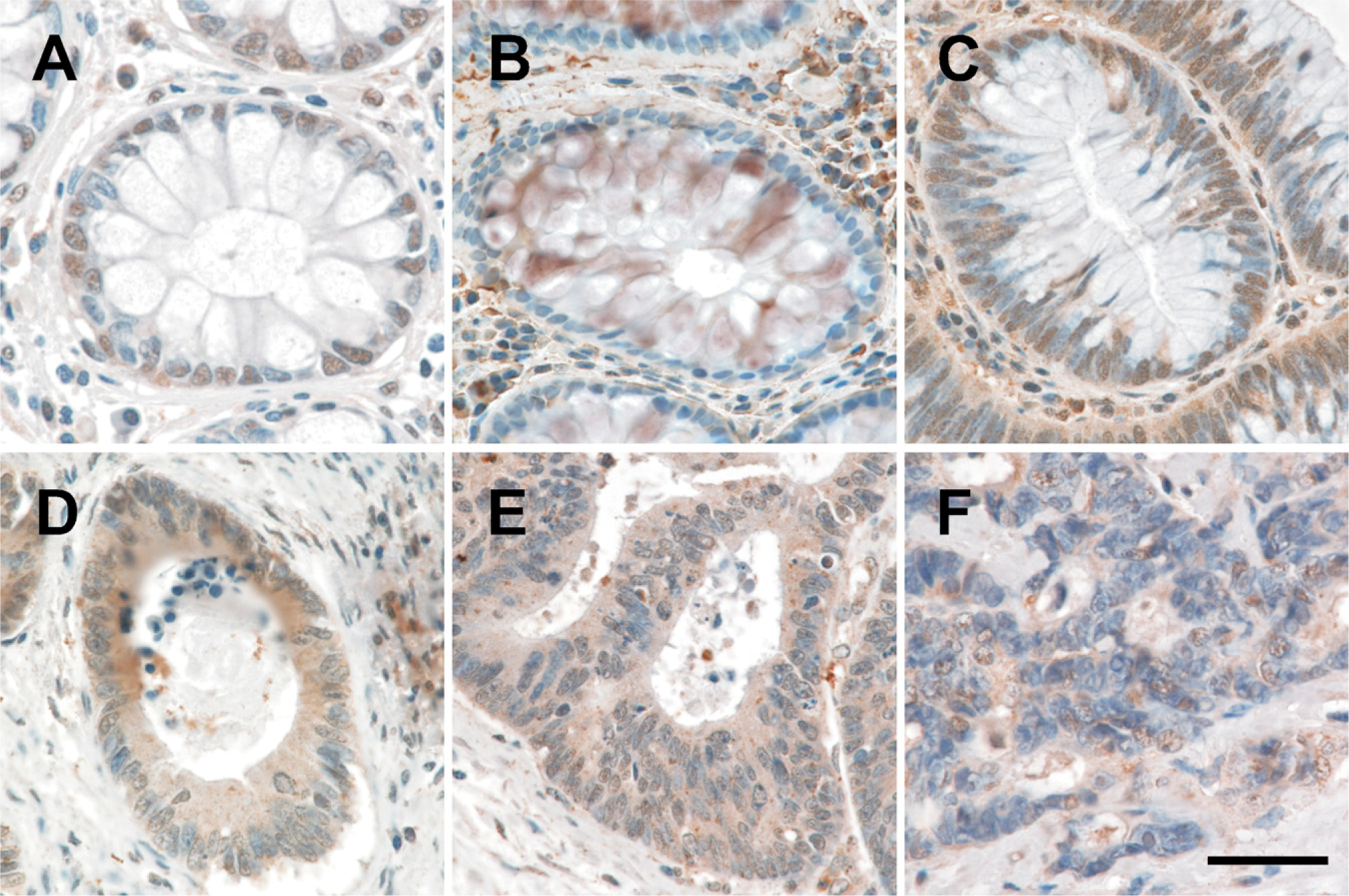
Expression of CYP27A1 in human colon cancer and adjacent non-malignant tissue. (
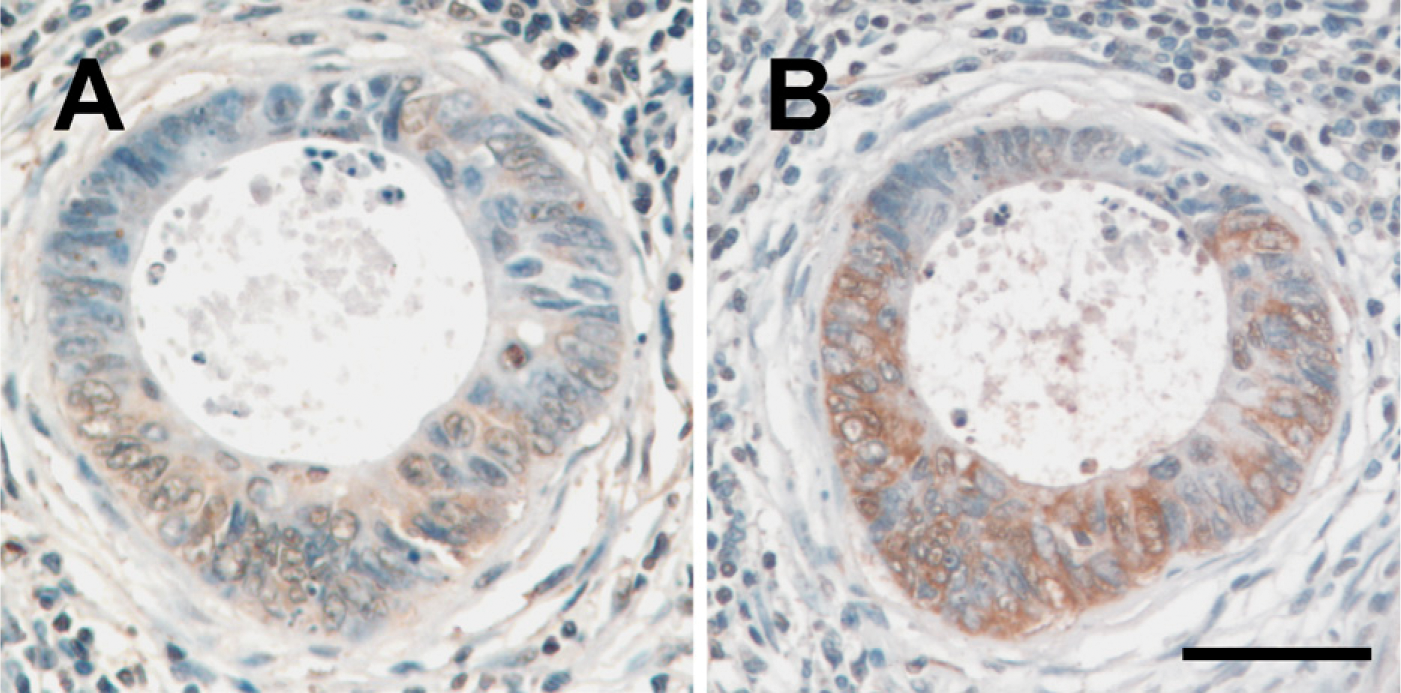
Expression of CYP27A1 (
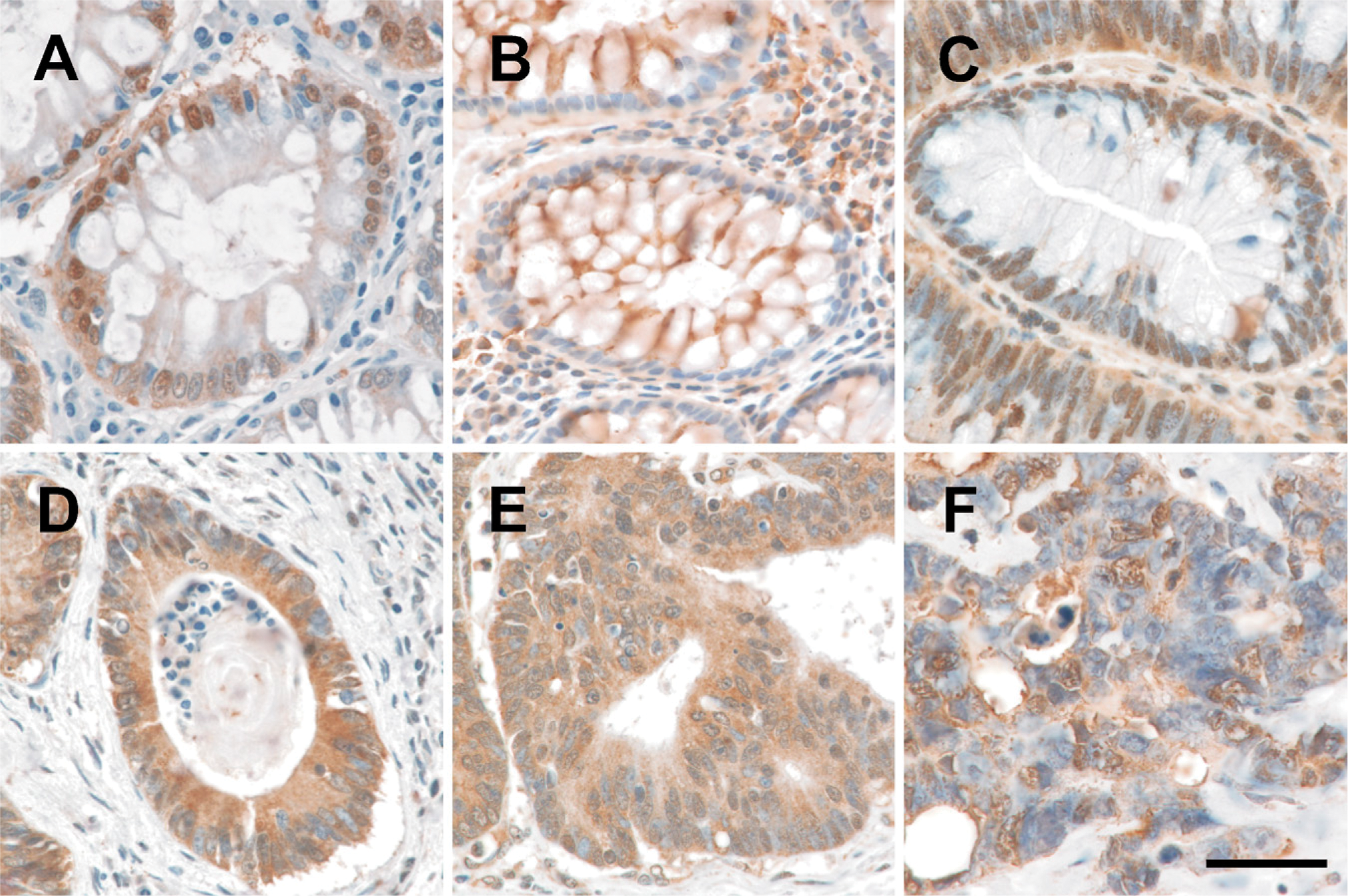
Expression of CYP24 in human colon cancer and adjacent non-malignant tissue. (
Translocation of CYP27A1 and CYP24 expression from the nucleus to the cytoplasm as a function of malignant progression can be more readily appreciated by expressing the chromogen values obtained by Q-IHC as a ratio (Figure 6). Whereas CYP27A1 and CYP24 in normal tissues and premalignant neoplasms were expressed entirely in the nucleus—yielding a ratio of ∞-the ratio of nuclear-to-cytoplasmic expression dropped to 1 or less postmalignant transformation.
Finally, we quantified nuclear and cytoplasmic CYP27A1 expression in lymph nodes and compared that to what was detected in the original tumor that gave rise to the metastasis. In all instances, whatever expression of CYP27A1 could be detected in the nuclei of the primary tumor decreased significantly in the nuclei of metastatic tumor cells (Figure 7, left panel). Similarly, whatever CYP27A1 was detected in the cytoplasm of cells within the primary tumor decreased significantly in that of the metastases (Figure 7, left panel). In contrast, whereas CYP24 nuclear expression likewise decreased in tumor cells contained within lymph nodes as compared with the primary tumor when they arrived, cytoplasmic expression increased (Figure 7, right panel).
Discussion
Vitamin D has long been suspected of preventing CRC, with hypovitaminosis D being a major risk factor for developing this type of malignancy (reviewed in Harris and Go 2004). Upon binding to its cognate receptor, bioactive 1,25(OH)2D3 decreases colon cancer cell proliferation (Lointier et al. 1987; Shabahang et al. 1994). Additionally, bioactive 1,25(OH)2D3 induces apoptosis (Vandewalle et al. 1995; Diaz et al. 2000) as well as having pro-differentiating effects (Vandewalle et al. 1995) on various CRC cells. Thus, vitamin D is suspected of having both CRC chemotherapeutic and chemopreventive properties. This is particularly important because vitamin D deficiency is increasing in the general population (Holick 1998; Tangpricha et al. 2002; Stokstad 2003).
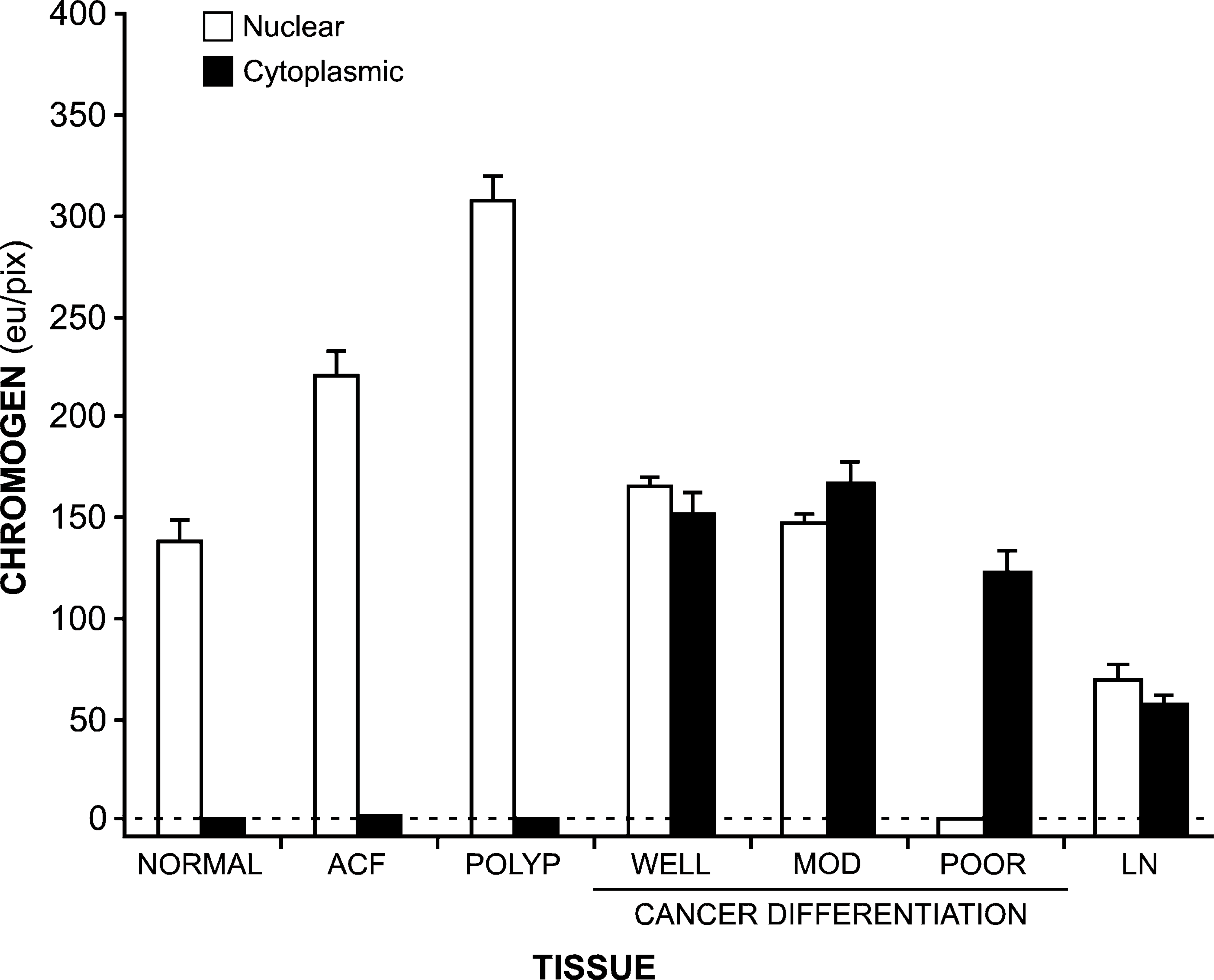
Amount of CYP27A1 present in the nuclei (open bars) and cytoplasm (closed bars) present in normal colonic epithelium (normal), ACF, adenomatous polyps (polyp), colon cancers of defined differentiation (WELL, well-differentiated; MOD, moderately differentiated; POOR, poorly differentiated), and tumors metastasizing to regional lymph nodes (LN). In all instances, immunohistochemistry was performed and chromogen amount quantified as described in Materials and Methods, with data shown as mean ± SE. The amount of nuclear CYP27A1 is significantly elevated in ACF and polyps and is significantly decreased in poorly differentiated cancer and lymph nodes, as compared with normal tissues (ANOVA, p<0.01). Likewise, the amount of cytoplasmic CYP27A1 is significantly elevated in all cancers and lymph nodes as compared with normal tissues (ANOVA, p<0.01).
Yet, bioactive 1,25(OH)2D3 is unlikely to be widely used because of its narrow therapeutic range and side effect profile. Indeed, even the pharmacological precursor to 1,25(OH)2D3, calcifediol [i.e., 25(OH)D3], can be calcemic and thus cause a number of side effects, potentially limiting its widespread use. As a result, >2000 1,25(OH)2D3 analogs have been synthesized in the hopes of generating an effective anti-neoplastic drug without the toxicity of the parent compound (Mehta and Mehta 2002).
However, it may not be necessary to identify such a compound, at least for CRC, if the appropriate enzyme complex is present within the colonic epithelium. Thus, the purpose of this study was to systematically assess CYP27A1 and CPY24 cellular expression in normal and neoplastic colonic epithelium. We demonstrate here that CYP27A1 is restricted to the nucleus of normal colonic epithelia, with increasing amounts detected in ACFs and adenomas (Figure 4). ACFs were first defined in the colons of rodents exposed to carcinogens and identified soon thereafter in human colons (Takayama et al. 1998). ACFs are associated with adenomatous polyp (Yokota et al. 1997) and CRC formation in humans (Takayama et al. 1998) and are an accepted CRC biomarker (Hawk et al. 2004). The finding of increased CYP27A1 expression in ACFs, as well as adenomatous polyps, in concert with our prior observation that CYP27B1 is also increased in precancerous colonic neoplasms (Matusiak et al. 2005), strongly suggests that the simplest and most widely available form of vitamin D, cholecalciferol, may be sufficient for CRC chemoprevention.
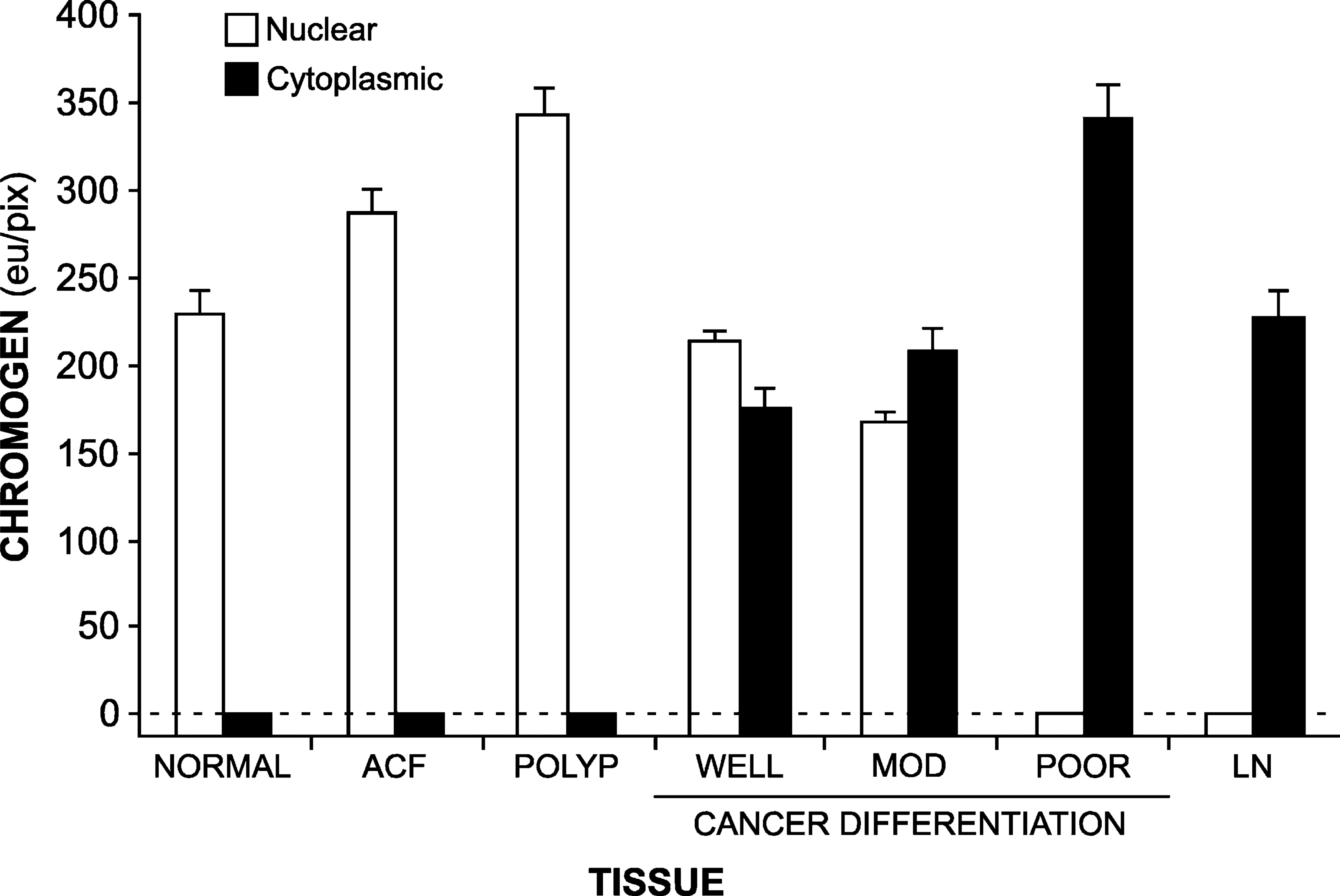
Amount of CYP24 present in the nuclei (open bars) and cytoplasm (closed bars) present in normal colonic epithelium (normal), ACF, adenomatous polyps (polyp), colon cancers of defined differentiation (WELL, well-differentiated; MOD, moderately differentiated; POOR, poorly differentiated), and tumors metastasizing to regional lymph nodes (LN). In all instances, immunohistochemistry was performed and chromogen amount quantified as described in Materials and Methods, with data shown as mean ± SE. The amount of nuclear CYP24 is significantly elevated in ACF and polyps and is significantly decreased in poorly differentiated cancer and lymph nodes, as compared with normal tissues (ANOVA, p<0.01). Likewise, the amount of cytoplasmic CYP24 is significantly elevated in all cancers and lymph nodes as compared with normal tissues (ANOVA, p<0.01).
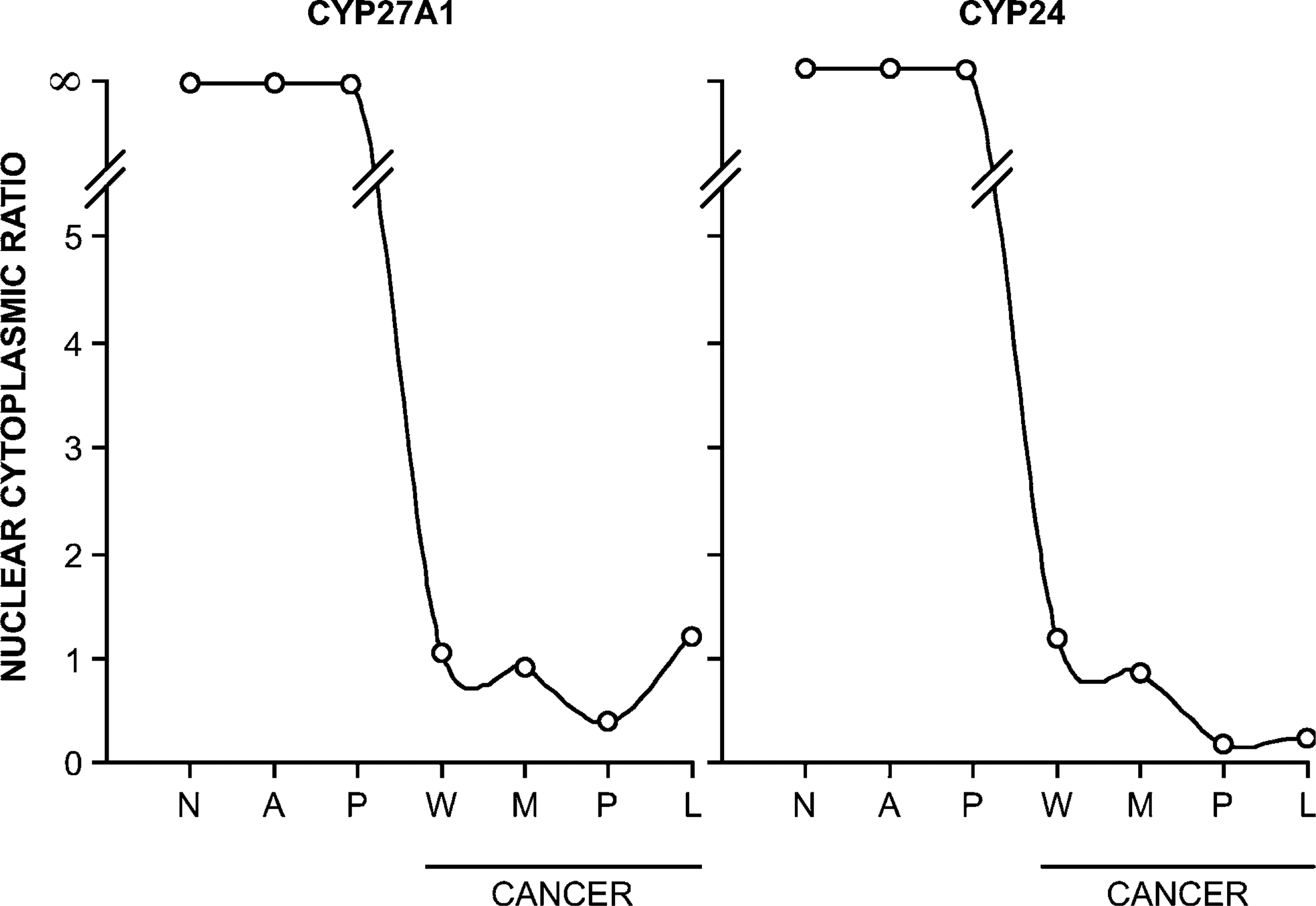
Ratio of nuclear to cytoplasmic CYP27A1 expression (left panel) and CYP24 expression (right panel). In all instances, the amount of the relevant protein was determined by quantitative immunohistochemistry as described in Materials and Methods. Data are expressed as mean ± SEM, whereas error bars cannot be seen due to their small size. N, normal tissue; A, ACF; P, adenomatous polyp; W, well-differentiated CRC; M, moderately differentiated CRC; P, poorly differentiated CRC; L, metastatic cancer to lymph node.
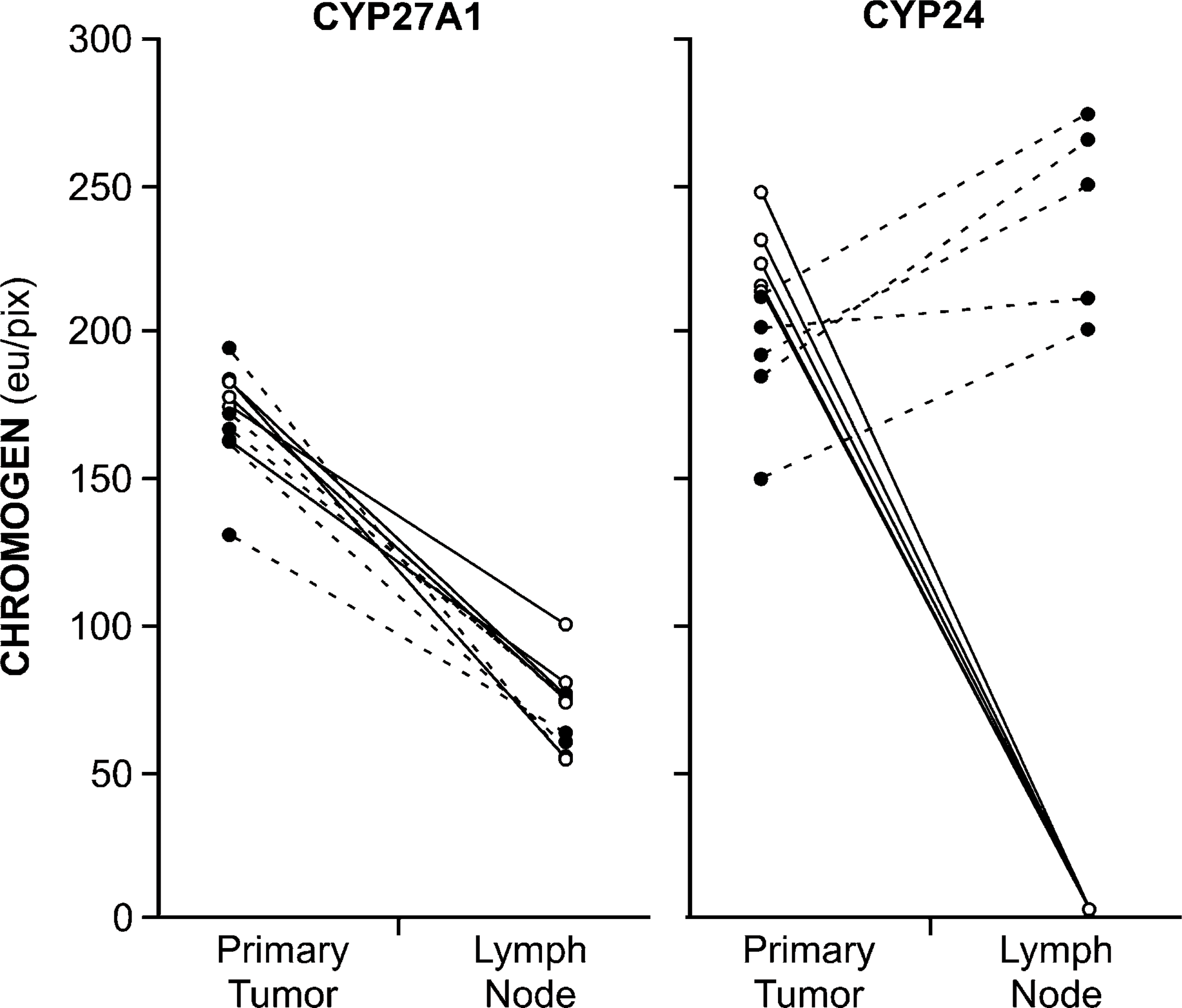
Amount of CYP27A1 chromogen (left panel) and CYP24 chromogen (right panel) present in the nuclei (open symbols and solid lines) and cytoplasm (closed symbols and dashed lines) in matched primary tumors and lymph nodes from the same patient. In all instances, analysis was restricted to tumors and lymph nodes containing well-differentiated tumor to facilitate data processing. Chromogen quantity shown as the mean ± SEM for well-differentiated regions within the primary tumors and for the well-differentiated tumor nest within the relevant lymph node. eu/pix, value-less energy units per pixel.
Yet, our data also suggest the possibility that neither cholecalciferol nor 1,25(OH)2D3 may be suitable for CRC chemotherapy. In this study we made use of our novel technique for Q-IHC (Matkowskyj et al. 2000, 2003a, 2004, 2006) to show, for the first time, the degree to which both CYP27A1 and CYP24 are expressed at the cellular level in CRC. Although a few other studies have addressed CYP27A1 and CYP24 expression in CRC (Cross et al. 2005; Anderson et al. 2006), ours is the first study to assess the presence of these proteins in the nucleus and cytoplasm at the cellular level and as a function of tumor cell differentiation. This is important because one study suggested that CYP24 expression was increased in CRC as compared with adjacent non-malignant tissue (Anderson et al. 2006); however, this study was performed using RT-PCR and could only detect relative amounts of mRNA present without information provided as a function of differentiation.
Tumor differentiation is important clinically because it predicts the development of metastases for most solid tumors (Birchmeier et al. 1996; Kowalski and Medina 1998) including CRC (Wood et al. 1981). Whereas CYP27A1 and CYP24 are expressed only in the nucleus of precancerous colonic tissues, in postmalignant transformation there is a decreased amount of nuclear protein present (Figure 6). With the increasing presence of the vitamin D-degrading enzyme CYP24 in the cytoplasm—especially apparent in tumor cells that have metastasized to regional lymph nodes (Figure 7)—it is unlikely that bioactive 1,25(OH)2D3 derived from orally consumed precursors will remain as such in its migration to its site of action in the nucleus.
This study, however, must be considered in the context of a number of limitations. First and foremost, we determined the amount of relevant protein by Q-IHC. As such, we have no information as to the enzymatic activity of CYP27A1 and CYP24 present. Additionally, protein was detected using commercially available antibodies, the characterization of which was not possible. The manufacturer, Santa Cruz Biotechnology, Inc., declined to identify the epitope to which these polyclonal antibodies were directed and would not provide samples of the synthetic peptide used for their generation. Although the manufacturer claims specificity for these reagents, it remains possible that such specificity does not, in fact, exist. Despite these limitations, however, the present data, in combination with data we have previously published (Matusiak et al. 2005), are consistent with one another and suggest that although pro-vitamin D analogs may be useful in CRC chemoprevention, they may be less useful for CRC chemotherapy.
Footnotes
Acknowledgments
This work was supported by NIH Grant CA-094346 and a VA Merit Review (to RVB).
