Abstract
We undertook this study to evaluate the expression of septin family members SEPT5, SEPT8, and SEPT11 in proliferative retinal membranes. Epiretinal membranes (ERM) were obtained from seven patients with proliferative vitreoretinopathy (PVR) and from four patients and four postmortem eyes with proliferative diabetic retinopathy (PDR). Subretinal membranes (SRM) were obtained from one patient and six postmortem eyes. Membranes were examined by immunohistochemical staining of paraffin sections using polyclonal antibodies against SEPT5, SEPT8, and SEPT11 and an ABC detection system. SEPT8 expression was detected in all ERM and SRM, with an exceptionally strong expression of 100% for ERM of PVR, 63% for PDR membranes, and 57% for SRM. SEPT11 was identified in 91% of all cases, with strong expression of 14%, 25%, and 14% in ERM of PVR, PDR, and SRM, respectively. SEPT5 was seen in 50% of all cases; strong immunostaining was found in only one case of PVR membranes. Our finding suggests a role for members of the septin family in the development of proliferative retinal membranes.
Keywords
P
In terms of pathogenesis and composition, retinal membranes can be divided into (a) vascularized membranes induced by retinal ischemia (as in PDR or retinal vein occlusion) and (b) avascular membranes induced by rhegmatogenous RD or after retinal surgery (Meyer et al. 2002). Contraction of proliferative retinal membranes leads to distortion or detachment of the retina. Surgery for retinal membranes is often dissatisfying both anatomically and functionally. It is therefore essential to advance the research into the pathological mechanisms involved in the development of theses membranes.
Septins are a novel family of GTP-binding proteins broadly distributed in many eukaryotes, except plants. It was recently demonstrated that they are involved in multiple fundamental cellular processes such as cytokinesis, membrane dynamics, vesicle trafficking, apoptosis, and cell polarity (Ito et al. 2005). Moreover, ample data implicate the septin family to be involved in the pathogenesis of diverse disease states such as leukemia (Megonigal et al. 1998) and tumors of the breast (Kalikin et al. 2000; Montagna et al. 2003), ovary (Burrows et al. 2003), and brain (Sakai et al. 2002). Septins are also associated with neurodegenerative conditions such as Alzheimer's disease (Kinoshita et al. 1998), Down's syndrome (Cheon et al. 2001; Engidawork et al. 2003), and Parkinson's disease (Ihara et al. 2003).
Septins (SEPT5, SEPT8, and SEPT11) are expressed in human brain (Hall et al. 2005). The presence of SEPT5 and SEPT8 has been described in heart, aorta, prostate, testis, and ovary (Blaser et al. 2003). Meanwhile, SEPT5 has been reported to be a parkin-binding protein. A mutation of the parkin leads to an increase of SEPT5 and causes autosomal recessive juvenile Parkinson (Zhang et al. 2000). Septins SEPT5 and SEPT8 are involved in granula secretion in platelets (Dent et al. 2002; Blaser et al. 2004).
Proliferative retinal membranes in clinical conditions

ERM, epiretinal membrane; PVR, proliferative vitreoretinopathy; PDR, proliferative diabetic retinopathy; SRM, subretinal membrane; RD, retinal detachment.
We recently demonstrated SEPT5 and SEPT8 expression in the human eye. Both SEPT8 and SEPT5 were expressed in corneal endothelial cells, iris tissue, lens epithelium, and non-pigmented ciliary epithelium. In addition, SEPT8 was expressed in conjunctival epithelial cells, corneal epithelial cells, trabecular meshwork, iris vessel endothelium, retina, and in the optic nerve (Pache et al. 2005).
The exact role of septins in the human eye both under physiological and pathophysiological conditions is, however, still poorly understood. We therefore examined expression of SEPT5, SEPT8, and the newly available SEPT11 in proliferative retinal membranes.
Materials and Methods
Human Specimens
ERM and SRM were obtained from 12 eyes of 12 patients who underwent vitrectomy and from 10 postmortem eye globes. In all cases, the cause of death was either heart failure or pneumonia. Sample collection was approved by the Ethics Committee criteria and followed the tenets of the Helsinki Declaration. Clinical data of the patients are shown in Table 1. All samples were fixed in 4% formalin dehydrated by graded alcohol and embedded in paraffin. Four-μm sections were cut for immunohistochemistry.
Immunohistochemistry
Consecutive sections of paraffin-embedded eyes were deparaffinized and rehydrated. Antigen recovery was enhanced by oven heating the sections. Samples were incubated with primary antibodies against SEPT5, SEPT8, and SEPT11 overnight at 4C. The concentration of primary antibody was previously determined to be 1:200. Immunohistochemical staining was performed with the Elite ABC Kit (Vector Laboratories; Burlingame, CA) according to the manufacturer's protocol. The antigen was finally visualized by peroxidase staining using the peroxidase substrate 3,3′-diaminobenzidine tetrahydrochloride. Sections were counterstained with hematoxylin, dehydrated, and mounted. Incubation without primary antibodies served as negative control. Uveal melanoma tissue served as positive control.
Expression of SEPT5, SEPT8, and SEPT11 in proliferative retinal membranes

a One sample not evaluated.
+++, strong expression; ++, moderate expression; +, weak expression; −, absent expression.
The polyclonal antibody was used to specifically detect SEPT5, SEPT8, and SEPT11. The IgG fraction of the anti-SEPT5 and anti-SEPT8 antibody, respectively, was prepared from rabbit serum as previously described (Blaser et al. 2003).
The polyclonal anti-SEPT5 antibody was directed against the recombinant fragment of SEPT5 containing 387 residues (complete reading frame). The recombinant fragment had been cloned in a pET30b vector (65 N-terminal residues) (Zieger et al. 2000). The antibody was generated in rabbit (Blaser et al. 2002). This antibody recognizes a 45-kDa protein. Anti-SEPT5 antibody recognizes both SEPT5 isoforms because the two isoforms differ only concerning 18 N-terminal residues. Anti-SEPT5 antibody did not cross-react with SEPT8 and SEPT11.
The polyclonal anti-SEPT8 antibody was directed against the recombinant fragment of SEPT8 containing 105 N-terminal residues. The recombinant fragment had been cloned in a pET30c vector. The antibody was raised in rabbit and recognized a 60-kDa protein (Blaser et al. 2002). Anti-SEPT8 antibody recognizes the isoforms SEPT8_v1 (58 kDa) and SEPT8_v2 (64 kDa). Anti-SEPT8 antibody showed no cross-reactivity against SEPT5 and SETP11.
The SEPT11 peptide LVSNGVQIYQFPT (accession #NP 018 713.1; aa 204–216) was generated to establish a polyclonal antibody against SEPT11. Polyclonal antibodies were raised in rabbits and affinity purified (Biogenes; Berlin, Germany). Antibodies against SEPT5, SEPT8, and SEPT11 do not cross-react with the other septin proteins.
All sections were assessed for localization and intensity of specific immunoreactivity on a semiquantitative scale of − to +++ by two blinded, experienced observers (XX, PM). In case of disagreement, consent was achieved by discussion.
Results
SEPT5
SEPT5 was weakly expressed in four of seven cases of PVR, in two of eight cases of PDR, and in one of seven cases of SRM; moderate expression was found in two ERM of PVR and two ERM of PDR; strong expression was found in one case of PVR (Table 2; Figure 1 and Figure 2).
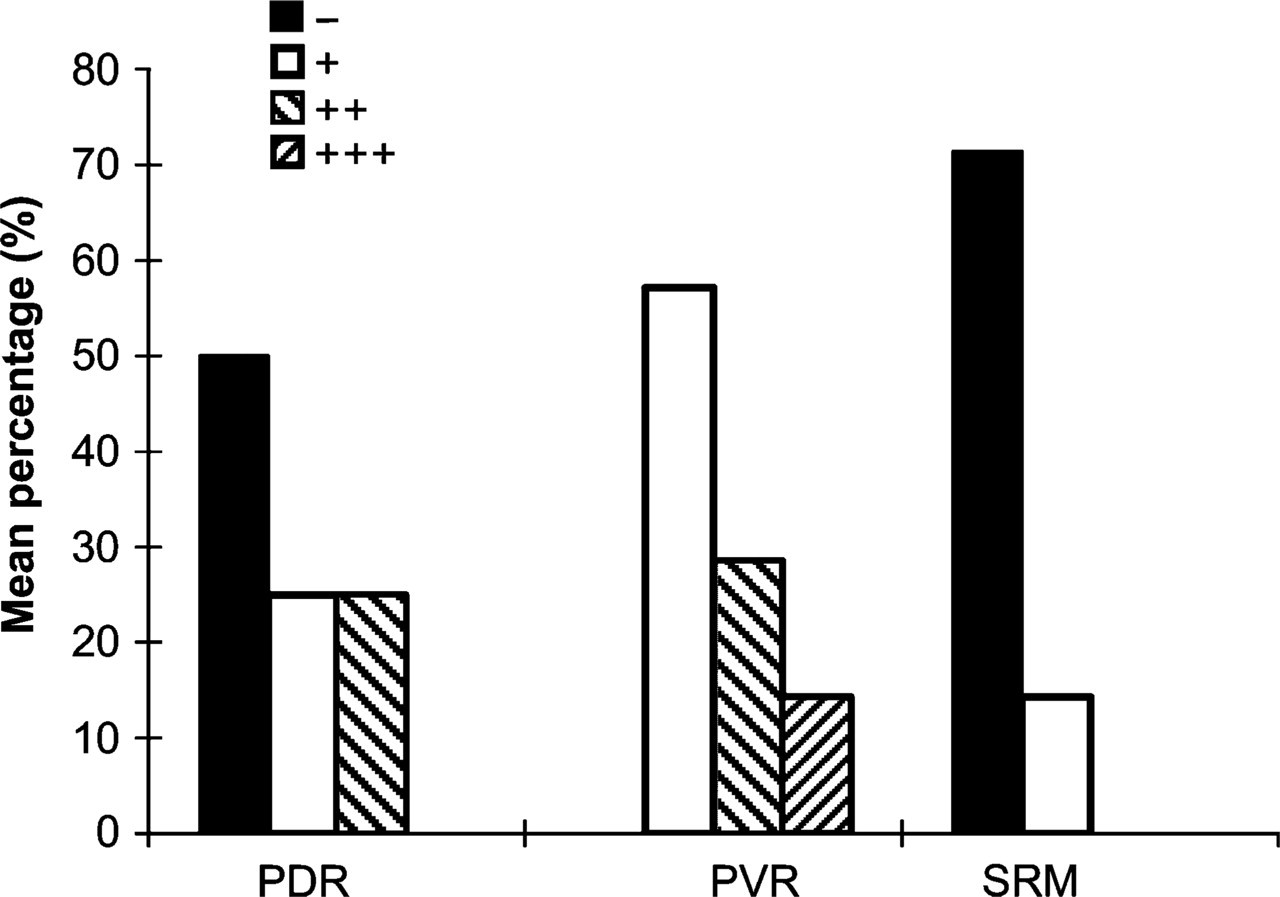
Expression of SEPT5 in proliferative retinal membranes. PDR, proliferative diabetic retinopathy; PVR, proliferative vitreoretinopathy; SRM, subretinal membrane. +++, strong expression; ++, moderate expression; +, weak expression; −, absent expression.
SEPT8
Weak positive expression of SEPT8 was observed in one case of PDR and one case of RD. Moderate positive expression of SEPT8 was detected in two cases of PDR and two cases of RD. Strong expression was seen in all seven PVR membranes, five PDR membranes, and four SRM of RD (Table 2; Figure 3 and Figure 4). Some cells showed nuclear staining for SEPT8, whereas in others it was cytoplasmic.
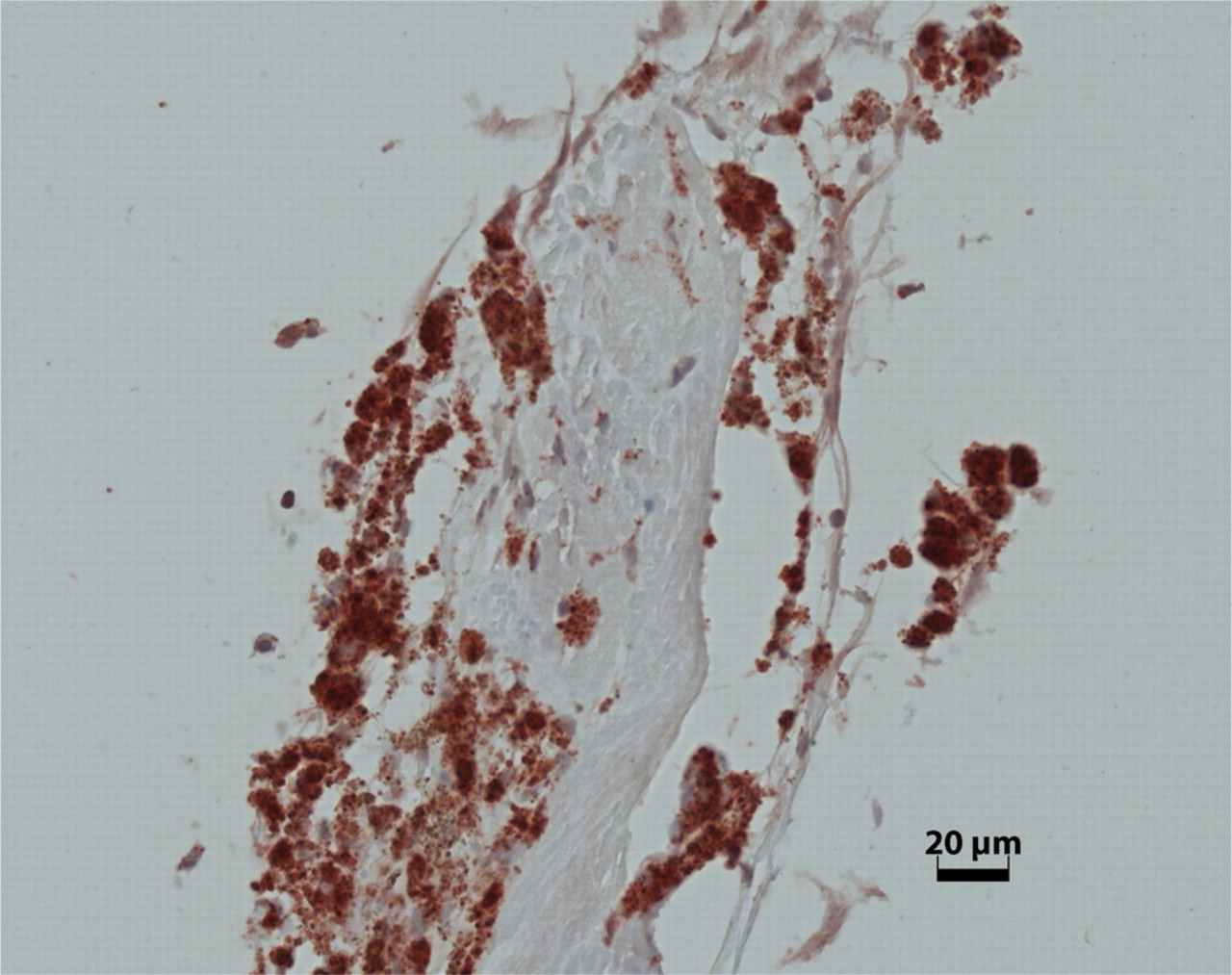
SEPT5 expression in an epiretinal membrane (ERM) of PVR.
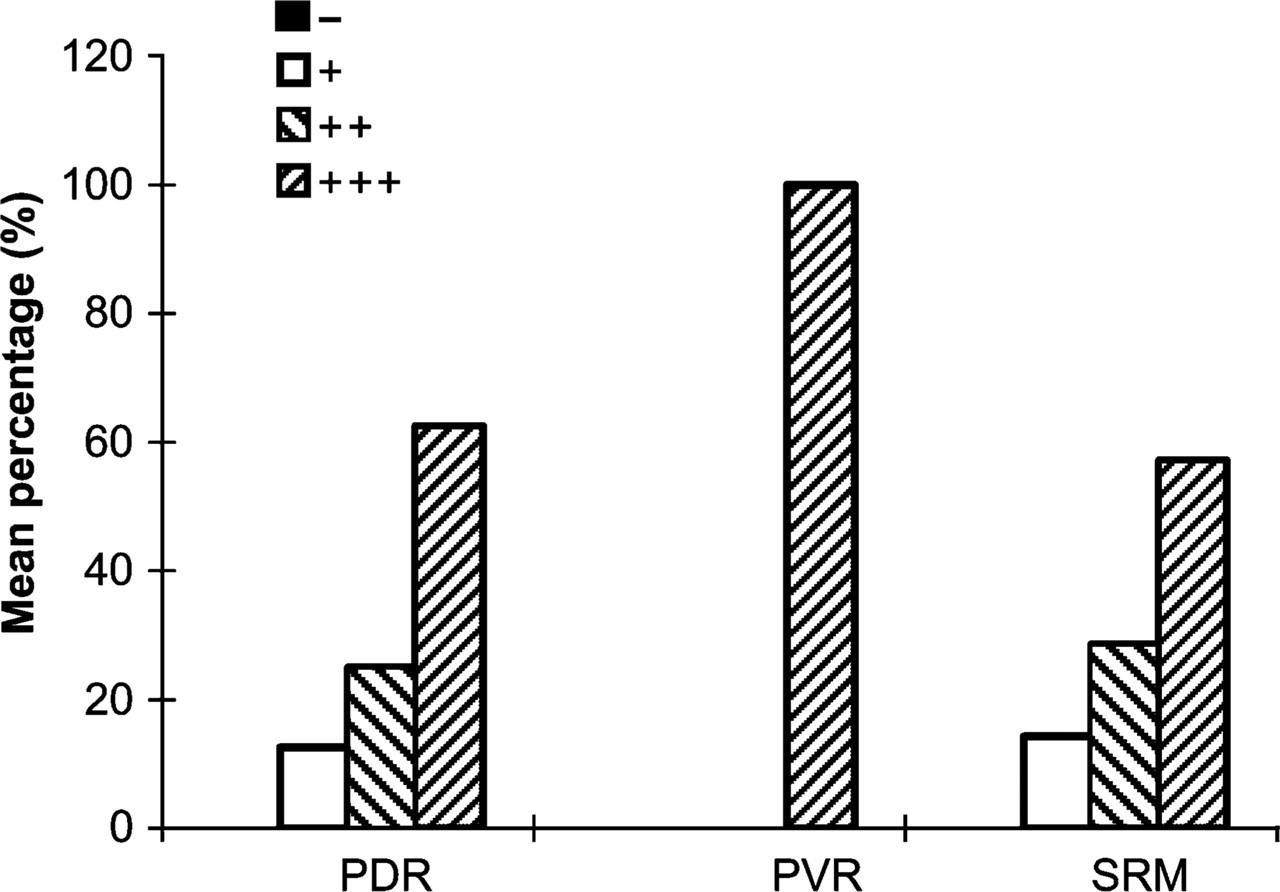
Expression of SEPT8 in proliferative retinal membranes. PDR, proliferative diabetic retinopathy; PVR, proliferative vitreoretinopathy; SRM, subretinal membrane. +++, strong expression; ++, moderate expression; +, weak expression; −, absent expression.
SEPT11
Of the 22 proliferative retinal membranes, SEPT11 weakly immunostained in one case of PVR, four cases of PDR, and five cases of SRM of RD; moderate staining of SEPT11 was seen in four ERM of PVR, one case of PDR, and one sample of RD; and strong expression was found in one case of ERM from PVR, two cases of PDR, and one case of RD (Table 2; Figure 5 and Figure 6).
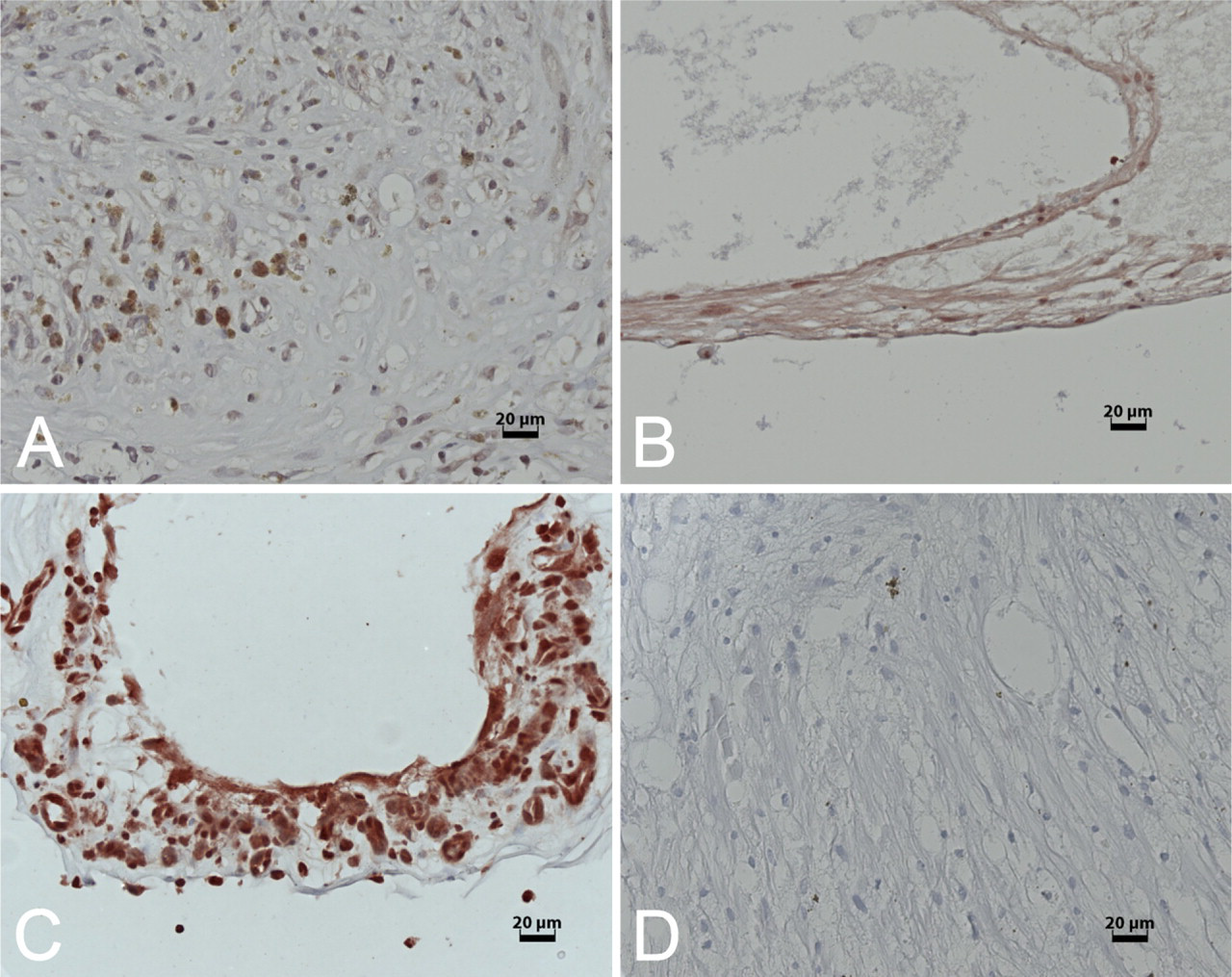
SEPT8: mild (+,
Discussion
We examined the expression of the three mammalian septins (SEPT5, SEPT8, and SEPT11) in ERM and SRM. All three septins were expressed in these membranes; however, at different percentages. SEPT5 was expressed in ∼54% of all cases, SEPT8 was strongly expressed in most cases, and SEPT11 expression was moderate.
SEPT8 expression was particularly strong in ERM and SRM, mainly in PVR (100%) and PDR (63%). The fact that SEPT8 expression is more pronounced in PVR than in ERM of PDR and in SRM of RD might imply far-ranging differences of the cellular components among these three membrane types. We recently demonstrated that in normal human eyes, SEPT8 is expressed in the optic nerve head axons as well as in the inner layer of the optic nerve head (Pache et al. 2005), which is composed of astrocytes. Astrocytes have also been demonstrated to be present in proliferative membranes (Schwartz et al. 1988; Gandorfer et al. 2002). They possibly originate from Elschnig's membrane at the optic nerve head. We therefore hypothesize that the positive staining of SEPT8 demonstrated in the present study suggests a role for SEPT8 in the pathogenesis of proliferation and migration of astrocytes. Interestingly, some cells showed nuclear staining for SEPT8, whereas in others it was cytoplasmic. This is in agreement with the observation of other researchers who showed that septins can be expressed in cytoplasma and in the nucleus, dependent on the cell's state of the cell cycle (Nagata et al. 2004).
SEPT11 is widely expressed in mammalian tissues and distributes along with cytoskeletons in a cell type-specific manner by colocalizing with stress fibers and microtubules in cells (Hanai et al. 2004). In our investigation, SEPT11 was observed in 20/22 cases, and its expression in these membranes ranged from weak (46%), to moderate (27%), and strong (18%). Strong expression of SEPT11 in ERM of PVR, PDR, and SRM was 14%, 25%, and 14%, respectively. According to sequence similarity, the septin family has been classified into four subgroups. Due to its highest homology to SEPT8, SEPT11 has been placed in the same subgroup as SEPT6 and SEPT8 (Kinoshita 2003); therefore, the role of SEPT11 in proliferative retinal membranes may also be similar to SEPT8.
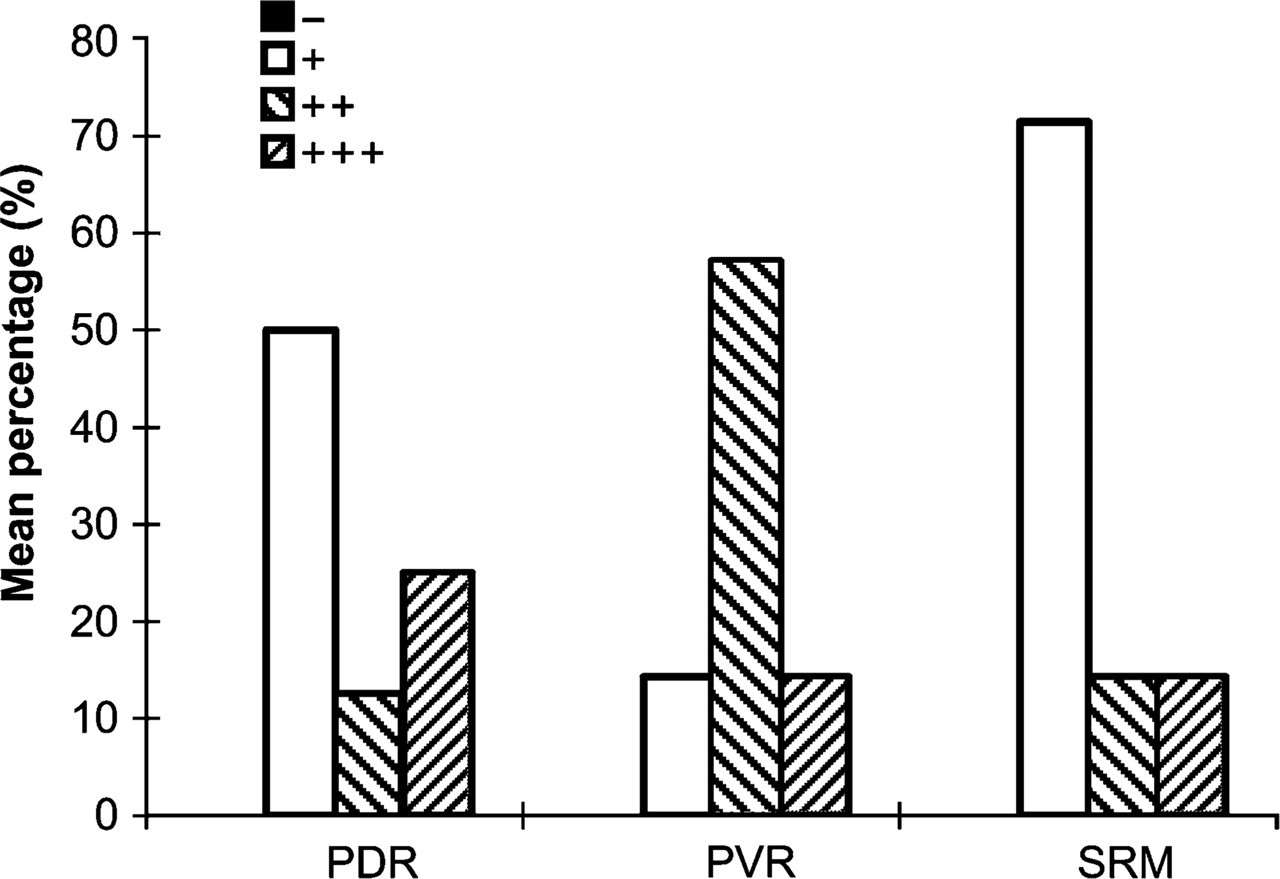
Expression of SEPT11 in proliferative retinal membranes. PDR, proliferative diabetic retinopathy; PVR, proliferative vitreoretinopathy; SRM, subretinal membrane. +++, strong expression; ++, moderate expression; +, weak expression; −, absent expression.
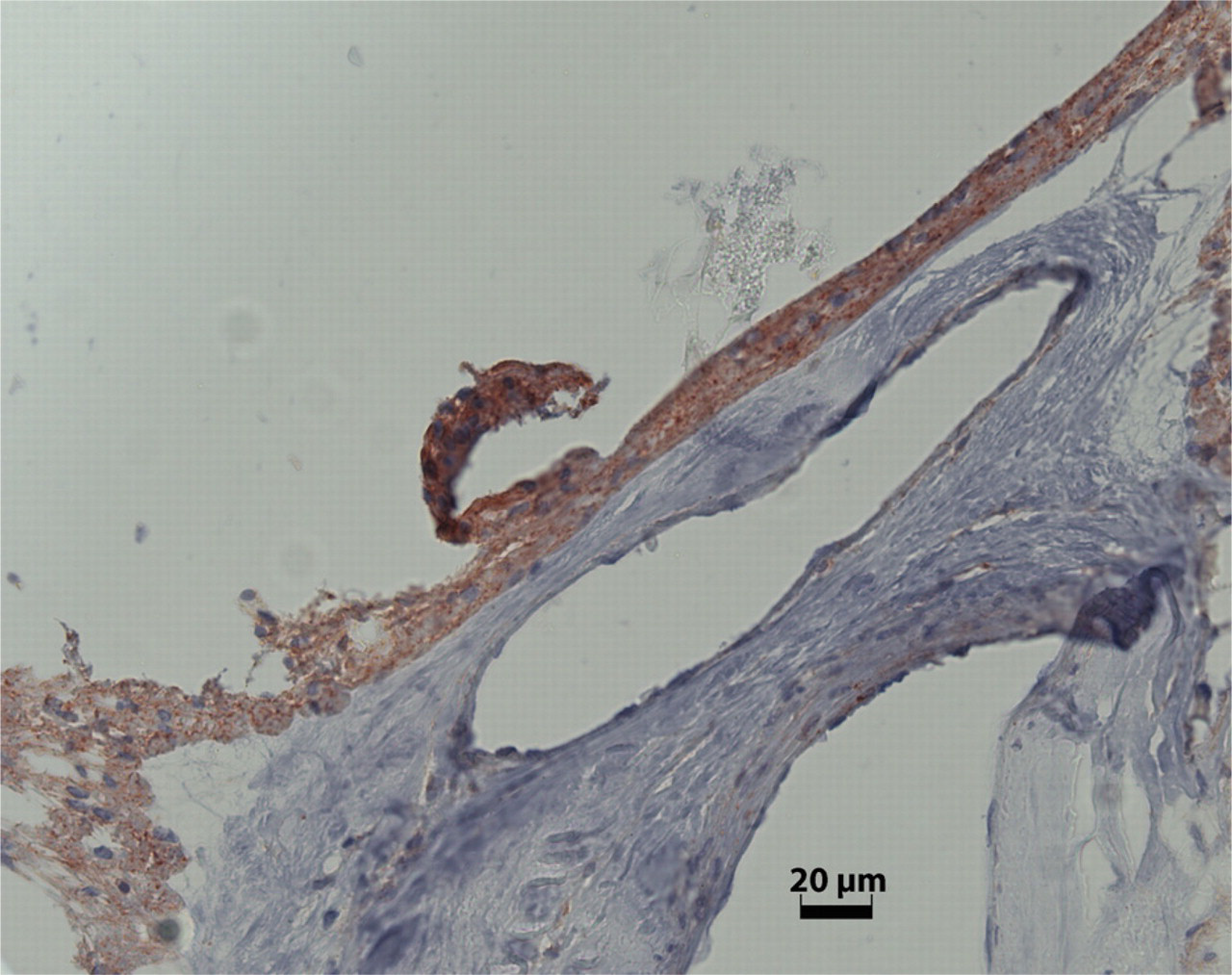
SEPT11 expression in an ERM of PDR.
Immunostaining of SEPT5 was observed in 100% of the PVR cases and in 50% of the cases of PDR. Only one case of SRM showed a weak positive staining for SEPT5. SEPT5 expression in proliferative membranes was weaker than that of SEPT8 and SEPT11. In a previous study on healthy eyes we were able to demonstrate that SEPT5 expression was restricted to the anterior segment (Pache et al. 2005). The fact that SEPT5 is expressed in the posterior part of the globe under pathological conditions is, therefore, highly interesting and deserves further investigation.
We speculate that the clue to the septins' contribution to membrane formation lies in their principal function in cytokinesis and apoptosis. The distinct characteristic of ERM and SRM is that the physiological homeostatic equilibrium is disturbed by cell proliferation. Septin filaments can assemble in bundles tightly associated with the actin-based structures such as contractile ring, cortical actin, and stress fibers (Trimble 1999; Kinoshita 2003). In addition, septin-dependent recruitment of a class of kinases to the bud neck region is a checkpoint for cell cycle progression. It has been demonstrated that septins can produce neuronal toxicity and motor behavioral dysfunction in dopaminergic neurons (Son et al. 2005). They may also contribute to neoplasia by enhancing membrane dynamics (Polo et al. 2004). Moreover, it has been reported that one isoform of SEPT4 can promote apoptosis. A loss of septin function may promote an increase in cell number (Larisch et al. 2000). Dysregulation in retinal membrane growth kinetics due to loss of cell death control has been implicated in the development and progression of human proliferative disorders. This proliferative process is associated with a rate of cell loss that occurs via apoptosis.
Recent investigations have shown that SEPT5 is an interaction partner with both SEPT8 (Blaser et al. 2003) and SEPT11 (Blaser et al. 2006). These studies suggest that SEPT5 and SEPT8, as well as SEPT11, are components of a cell-specific complex and function as regulative scaffolds for recruitment of other proteins. Expression of SEPT5, SEPT8, and SEPT11 in ERM and SRM implies that they may also act as a cell cycle-regulating complex.
In summary, we were able to demonstrate an involvement of certain members of the septin family in the development of proliferative retinal membranes of the human eye. The exact mode of action of these proteins, however, has not yet been disclosed and deserves further intense research. Hopefully, in the future a better understanding of the role of septins in proliferative retinal membranes may help to facilitate both therapy and prevention of this sight-threatening eye disease.
Footnotes
Acknowledgements
This study was supported by the Swiss Federal Commission for Scholarships for Foreign Students (FCS).
