Abstract
The tight junction (TJ) regulates epithelial cell polarity and paracellular permeability. In the present study, to investigate whether the second extracellular loop of occludin affects the localization of carcinoembryonic antigen (CEA) and CD26 expressed on apical membranes, and the fence function of the TJ, the human intestinal epithelial cell line T84 was treated with the monoclonal anti-occludin antibody (MAb) 1H8, corresponding to the second extracellular loop of occludin. In T84 cells treated with MAb 1H8, occludin disappeared, and CEA and CD26 were observed to diffuse from the apical membrane to the basolateral membrane. Furthermore, a decrease in the fence function of TJ was observed without changes in the TJ strands and barrier function. When T84 cells precultured in low calcium (Ca) medium were recultured in normal Ca medium in the presence of MAb 1H8, recruitment of occludin to the apical-most membranes and recovery in distribution of CEA and CD26 were markedly retarded compared with the control. These results suggested that MAb 1H8 against the second extracellular loop of occludin selectively affected formation of the apical/basolateral intramembrane diffusion barrier and that the second extracellular loop of occludin plays a crucial role in the maintenance of epithelial cell polarity by the TJ.
T
Occludin, the first integral TJ protein to be identified, is the most ubiquitously expressed at the apical-most membranes (Furuse et al. 1993; Matter and Balda 1999). It is a 65-kDa tetraspan membrane protein that has two extracellular loops, a long carboxy-terminal cytoplasmic domain, and a short N-terminal cytoplasmic domain (Furuse et al. 1993; Ando-Akatsuka et al. 1996). The two extracellular loops have unusual amino acid compositions consisting of a high content of tyro-sine and glycine residues in the first loop, whereas the second loop is rich in tyrosine residues (Furuse et al. 1993; Schneeberger and Lynch 2004). Further evidence for an adhesive function of occludin was observed in experiments in which the addition of a peptide, whose amino acid sequence corresponded to that of the second extracellular loop of occludin, was associated with disruption of the TJ barrier (Wong and Gumbiner 1997). The second extracellular domain is required for stable assembly of occludin in TJ areas (Medina et al. 2000). More recently, the second extracellular domain of occludin was shown to regulate TGF-β type I receptor localization for efficient TGF-β-dependent dissolution of the TJ during epithelial-mesenchymal transition (EMT) (Barrios-Rodiles et al. 2005).
By overexpression of occludin, the barrier function of the TJ, measured by transepithelial electric resistance (TER) and the transepithelial flux of mannitol, was increased in mammalian epithelial cells (Balda et al. 1996; McCarthy et al. 1996). The fence function of TJ was not impaired in suppressed occludin clones, as determined by BODIPY-sphingomyelin diffusion in the membrane (Yu et al. 2005). Occludin-deficient mice express a complex phenotype, but one without obvious structural or functional TJ abnormalities (Saitou et al. 2000). We have shown that occludin is relevant to apoptosis via MAPK and Akt signaling pathways in occludin-deficient mouse hepatocytes in vitro (Murata et al. 2005). However, the physiological function of occludin in the TJ is still undefined.
Carcinoembryonic antigen (CEA) and CD26/dipeptidyl peptidase IV (DPPIV) are 180-kDa and 110-kDa glycoproteins, respectively, with ubiquitous expression in the apical domain in well-differentiated cancers (Gold and Freedman 1965a, b; Ahnen et al. 1982; Baricault et al. 1995; Decaens et al. 1996). We previously found that the distribution of CEA may contribute to expression of occludin in human colorectal adenocarcinoma (Tobioka et al. 2002). In the present study, to investigate whether the second extracellular loop of occludin affected the localization of CEA and CD26, and the fence function of the TJ, human intestinal epithelial cell line T84 was treated with a monoclonal anti-occludin antibody, MAb 1H8, corresponding to the second extracellular loop of occludin. In T84 cells treated with MAb 1H8, the disappearance of occludin at cell borders, changes in the localization of CEA and CD26, and a decrease in the fence function of TJ were observed.
Materials and Methods
Production of MAbs
All animal procedures were performed in accordance with the Guide to the Care and Use of Experimental Animals (Japanese Council on Animal Care) and approved protocols from the Animal Care Committee of the Research Institute of the Sapporo Medical University, Sapporo, Japan.
Nucleotide sequences and the deduced amino acid sequences of rat occludin cDNA are available in the DDBJ/EMBL/GENEBANK database under accession number AB016425. cDNA encoding the second extracellular loop of rat occludin was obtained by PCR and introduced into pET-21a (+) vectors (Novagen; Madison, WI). Histidine-tagged protein was produced in E. coli BL21 (DE3) as described by the manufacturer and then purified using a column containing Ni-NTA agarose beads (Qiagen; Valencia, CA). Monoclonal antibodies were obtained essentially according to the procedure of Kohler and Milstein (1975). BALB/c mice (Shizuoka Laboratory Animal Center; Hamamatsu, Japan) were immunized with the purified protein, and hybridomas were prepared by fusion between mouse lymphocytes and NS-1 myeloma cells. The culture supernatant of each hybridoma was screened by ELISA using the immunogen. We selected the monoclonal anti-1H8 antibody (MAb 1H8) by Western blotting and immunocytochemistry.
Peptide Synthesis and SPOTs Analysis
Based on the assumed amino acid sequence of the extracellular loop of occludin, individual peptides were synthesized on a membrane using a SPOTs kit (Sigma; St Louis, MO), which contains a derivative cellulose membrane (SPOTs membrane). After washing with methanol, the SPOTs membrane was blocked overnight at 4C with blocking buffer (Sigma). The membrane was washed with Tris-buffered saline containing 0.05% Tween 20 for 10 min at room temperature and then incubated with MAb 1H8 for 1 hr at room temperature. The membrane was next incubated with horseradish peroxidase (HRP)-conjugated mouse IgG for 1 hr at room temperature. Detection was carried out using an enhanced chemiluminescence (ECL) Western blotting system (Amersham; Buckinghamshire, UK).
Cell Lines and Culture Conditions
Human intestinal epithelial cell line T84 obtained from the American Type Culture Collection (Rockville, MD) was cultured in DMEM/F12 (Gibco; Grand Island, NY) supplemented with 7% fetal bovine serum (Cansera International; Ontario, Canada), 100 U/ml penicillin, and 100 mg/ml streptomycin in a humidified atmosphere with 5% CO2 at 37C. The medium was replaced with fresh medium every other day. Confluent T84 cells were treated with MAb 1H8 for 2 hr at 37C.
Calcium (Ca) Switch Assay
For the Ca switch assay, confluent T84 cells were treated in low Ca medium (5 μM Ca2+) overnight, after which the low Ca medium was replaced by normal Ca medium (1.8 mM Ca2+).
Immunofluorescence Microscopy
Cells grown on glass coverslips were fixed with cold acetone and ethanol (1:1) mixture for 15 min at −20C or 4% para-formaldehyde in PBS for 20 min at room temperature. In the unfixed cells, we performed all processes at 4C. As primary antibodies, rabbit polyclonal anti-CEA, mouse monoclonal anti-CD26 (Dako; Carpinteria, CA), mouse monoclonal anti-occludin, rabbit polyclonal anti-claudin-1, mouse monoclonal anti-claudin-4, rabbit polyclonal anti-JAM-A, rabbit polyclonal anti-ZO-1 (Zymed; South San Francisco, CA), and mouse monoclonal anti-E-cadherin antibodies (BD Transduction Laboratories; San Jose, CA) were used. As secondary antibodies, Alexa 488 (green)-conjugated anti-rabbit or mouse IgG against polyclonal or monoclonal antibodies and Alexa 592 (red)-conjugated anti-rabbit or mouse IgG against polyclonal antibodies were used. Cells were also used for double staining of CEA and occludin. Cells were incubated with the primary and secondary antibodies for 1 hr at room temperature. Specimens were examined using a laser-scanning con-focal microscope (MRC 1024; Bio-Rad, Hercules, CA).
Western Blot Analysis
T84 cells in 60-mm dishes were washed with PBS, and 800 μl of buffer (1 mM NaHCO3 and 2 mM phenylmethylsulfonyl fluoride) was added. Cells were scraped and collected in microcentrifuge tubes and then sonicated for 10 sec. Protein concentration of each sample was determined using a BCA Protein Assay Reagent Kit (Pierce Chemical; Rockford, IL). Aliquots of 15 μg protein/lane for each sample were separated by electrophoresis in 4/20% SDS-polyacrylamide gels (Daiichi Pure Chemicals Co.; Tokyo, Japan). After electrophoretic transfer to a nitrocellulose membrane (Immobilon; Millipore, Billerica, MA), the membrane was saturated for 30 min at room temperature with blocking buffer (PBS, 0.1% Tween 20, and 4% skim milk) and incubated with anti-occludin, anti-claudin-1, anti-claudin-4, anti-JAM-A, anti-ZO-1, anti-E-cadherin, and anti-actin antibodies at room temperature for 1 hr. The membrane was incubated with HRP-conjugated anti-rabbit or mouse IgG at room temperature for 1 hr. Detection was carried out using an ECL Western blotting system (Amersham).
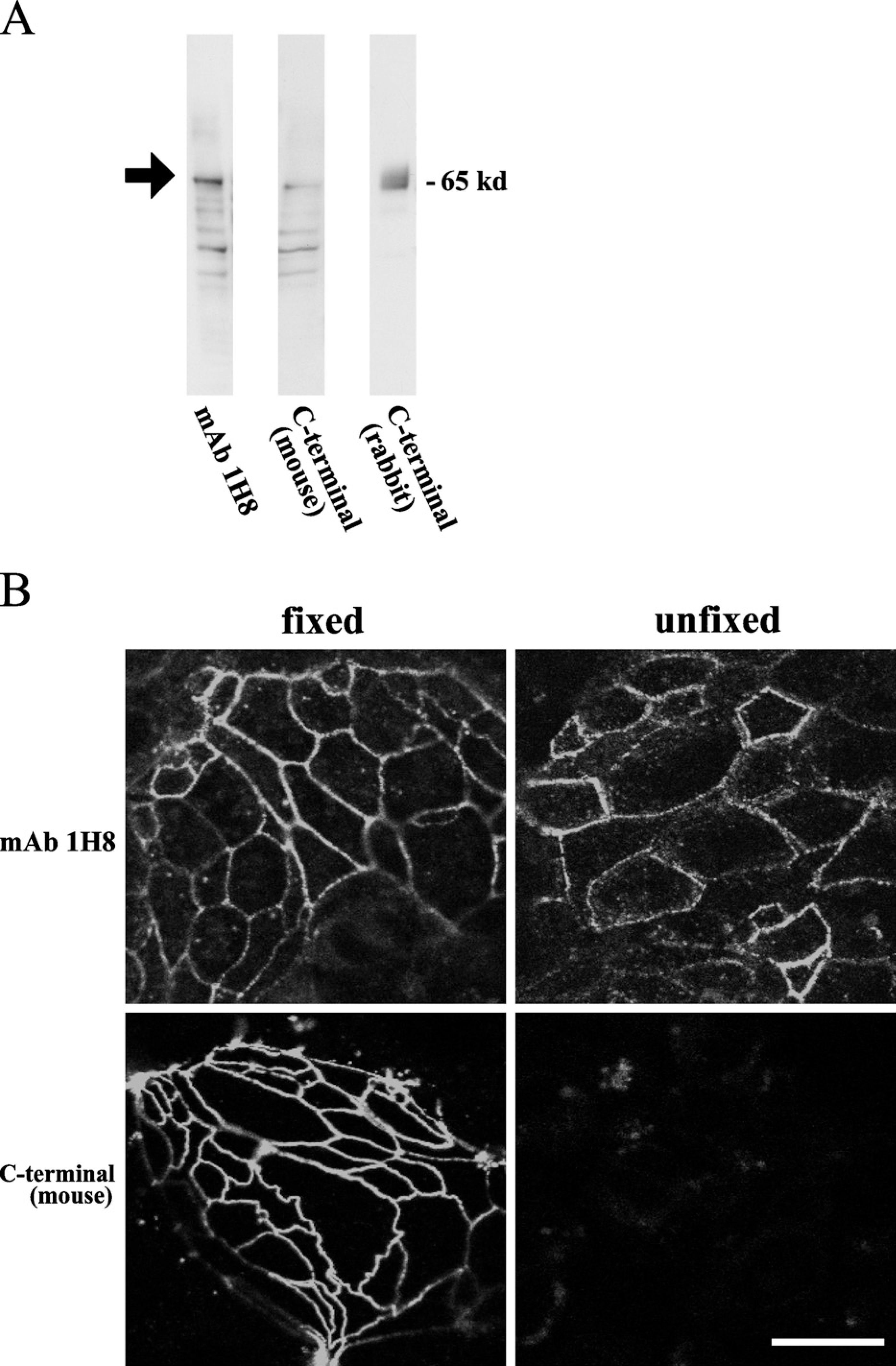
Monoclonal antibody (MAb) 1H8. (
Fence Function Assay (Diffusion of BODIPY-sphingomyelin)
This was performed according to Balda et al. (1996). Sphingomyelin/BSA complex (5 nM/ml) was prepared in P buffer (10 nM HEPES, pH 7.4, 1 mM sodium pyruvate, 10 mM glucose, 3 mM CaCl, and 145 mM NaCl) using BODIPY FL-sphingomyelin (Molecular Probes; Eugene, OR) and defatted BSA. Cells plated on glass-bottomed microwell plates (Mat Tek Corp.; Ashland, MA) were loaded with BODIPY-sphingomyelin/BSA complex for 1 min on ice, after which they were rinsed with cold DMEM/F-12 and mounted in DMEM/F-12 on a glass slide. Samples were analyzed by con-focal laser-scanning microscopy.
Measurement of TER
T84 cells were cultured to confluence on 6.5-mm Transwell (0.4-mm pore size) filters (Corning; Corning, NY) coated with rat tail collagen. TER was measured using an EVOM voltameter with an EVDOHM-12 (World Precision Instruments; Sarasota, FL) on a heating plate (Fine; Tokyo, Japan) adjusted to 37C. Values are expressed in standard units of ohms/cm2. For calculation, the resistance of blank filters was subtracted from that of filters covered with cells.
Freeze-fracture Analysis
For freeze-fracture experiments, T84 cells were fixed in 40% glycerin solution after fixation in 2.5% glutaraldehyde/0.1 M PBS (pH 7.3). The specimens were mounted on a copper stage, frozen in liquid nitrogen, fractured at −170 to −180C, replicated by platinum/carbon from an electron beam gun positioned at a 45° angle followed by carbon applied from overhead in a JFD-7000 freeze-fracture device (JEOL; Tokyo, Japan). After the replicas were thawed, they were floated on filtered 10% sodium hypochlorite solution for 10 min in a Teflon dish. Replicas were washed in distilled water for 30 min, mounted on copper grids, and examined at 80 kV on a JEOL 1200EX transmission electron microscope.
Data Analysis
Results are given as mean ± SEM. Differences between groups were tested by the two-tailed Student's t-test for unpaired data; p<0.05 was considered significant.
Results
Specificity of MAb 1H8
We produced a fusion protein with the second extracellular loop of rat occludin with the first loop in E. coli and raised a mouse monoclonal antibody, MAb 1H8. To examine the specificity of MAb 1H8, we carried out Western blotting and immunocytochemistry using cell line T84. MAb 1H8 was detected as a 65-kDa band, like mouse and rabbit anti-occludin antibodies against C-terminal domain (Figure 1A). Furthermore, MAb 1H8 was detected at cell borders of both fixed and unfixed T84 cells, whereas a mouse anti-occludin antibody against the C-terminal domain was detected at cell borders of only fixed T84 cells (Figure 1B). These results indicated that MAb 1H8 was an antibody against the extracellular loop region of occludin.
To confirm whether MAb 1H8 was such an antibody against the second extracellular loop of occludin, we performed the SPOTs analysis (Sigma) using synthetic-comprised 10–14 polypeptides against the first or second extracellular loop of human occludin (Figure 2A). In the immunoblotting, the spots of No. 3 and No. 4 in loop 2 were detected by MAb 1H8 (Figure 2B). This result indicated that MAb 1H8 could recognize the polypeptide YGSQIYALCNQFYTP of the amino acids 208–222 in the second loop of human occludin (Figure 2C).
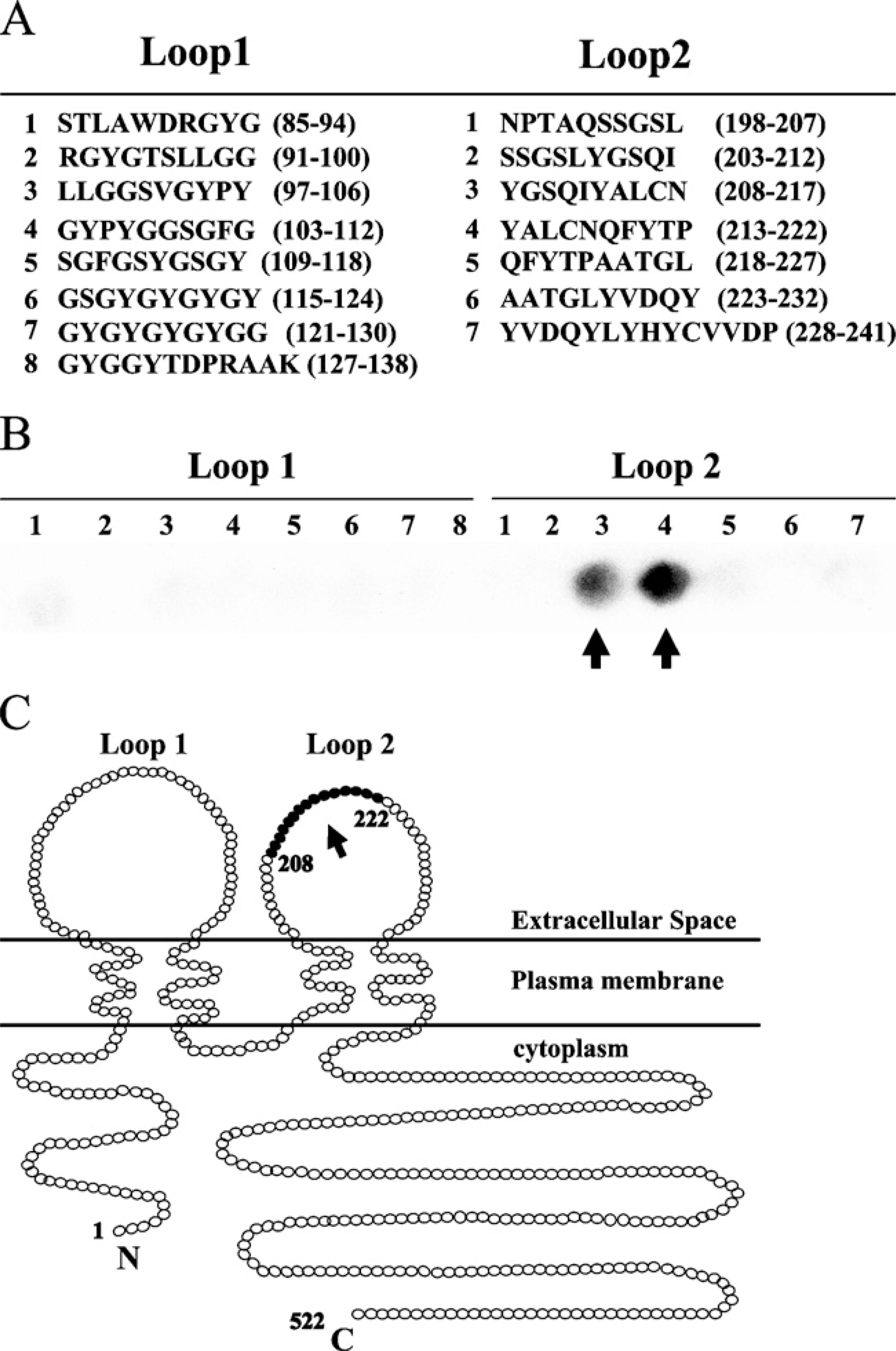
SPOTs analysis for specificity of MAb 1H8. (
(
Changes in Localization of CEA, CD26, and Occludin but Not Claudin-1, −4, JAM-A, ZO-1, and E-cadherin by Treatment With MAb 1H8 in T84 Cells
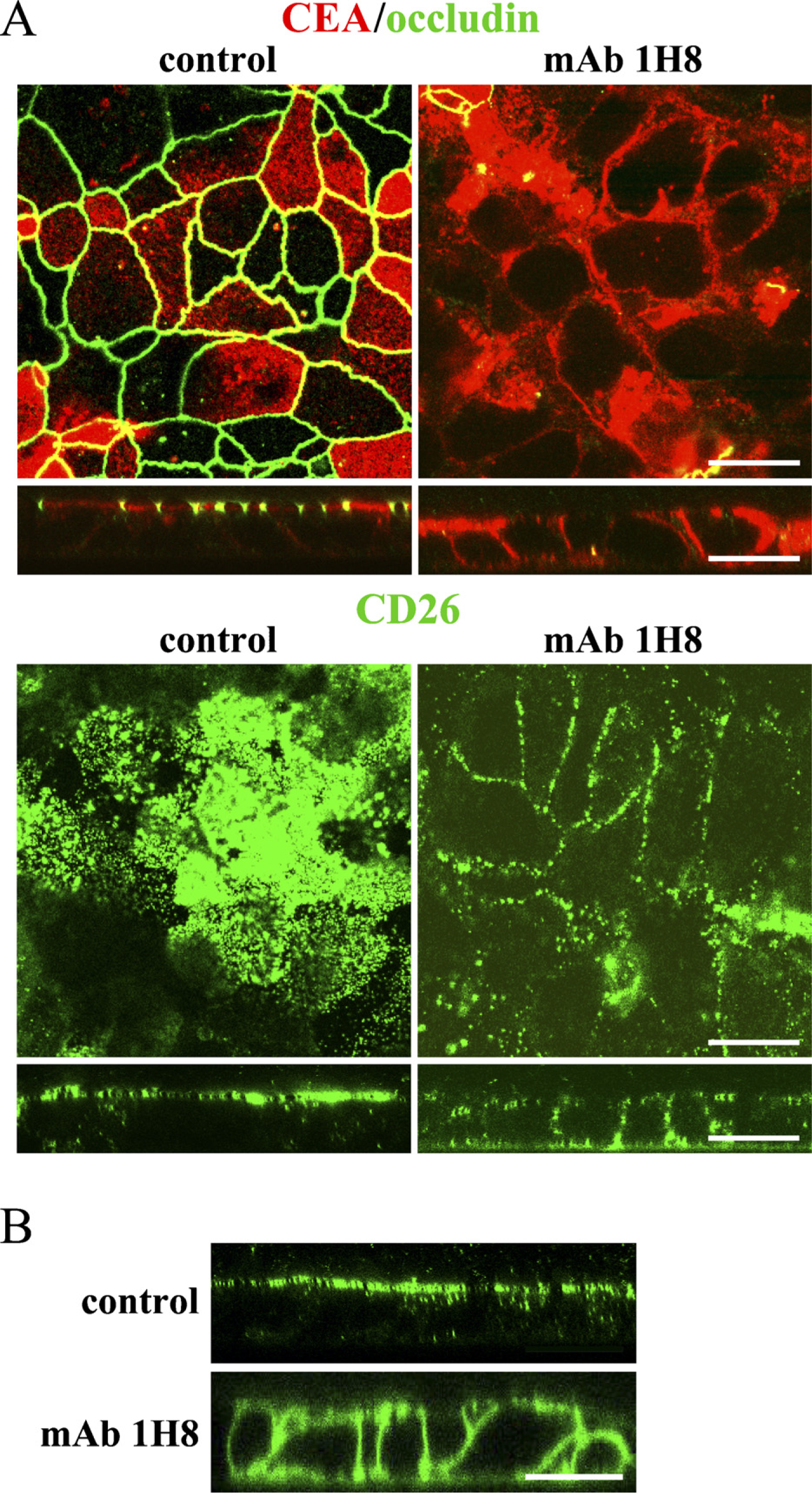
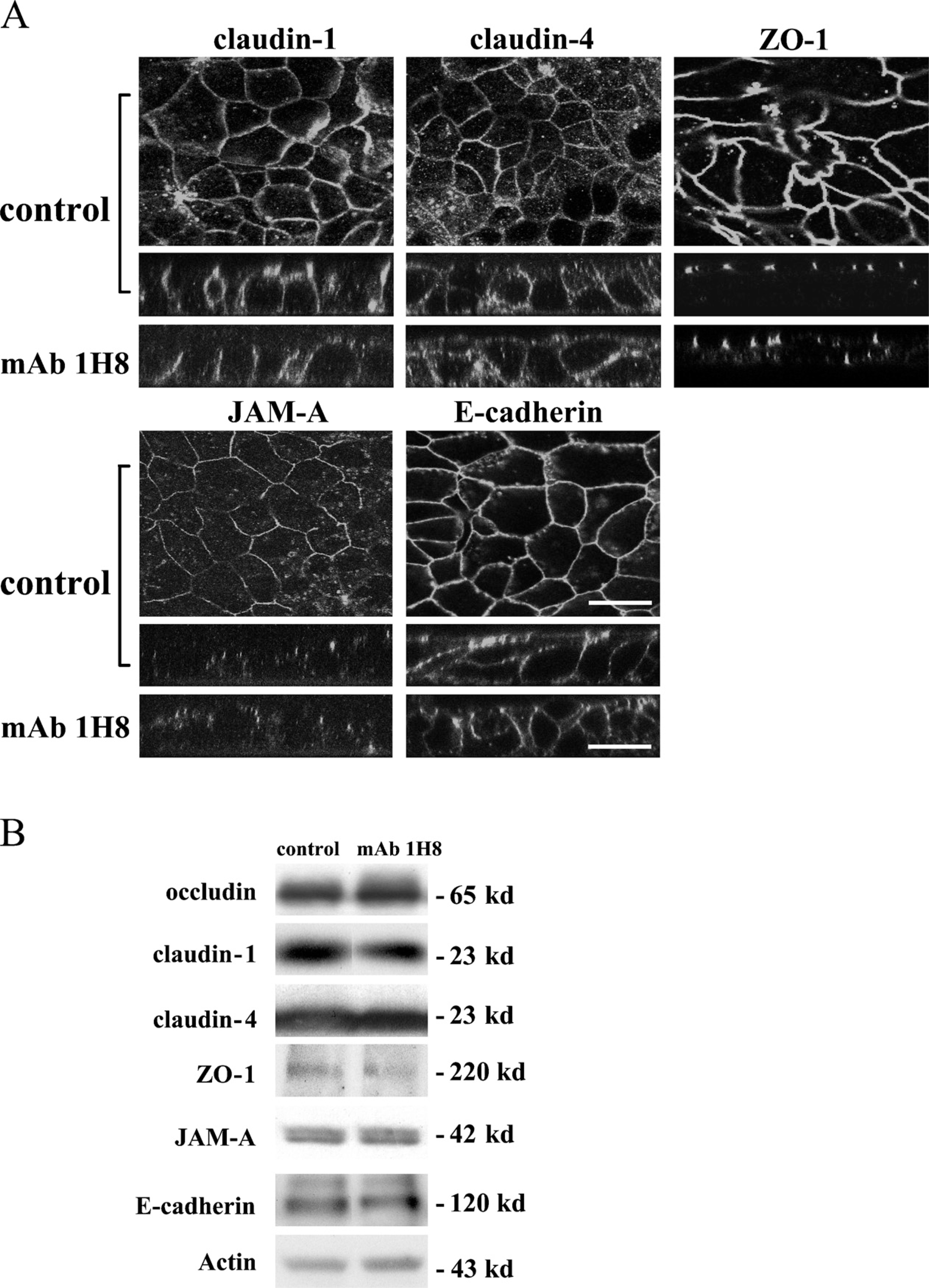
To examine whether MAb 1H8 affected the localization of CEA and CD26 expressed at the apical domain, T84 cells were treated with MAb 1H8 for 2 hr and then subjected to immunocytochemistry for occludin, CEA, and CD26. In control T84 cells, occludin was expressed at cell borders and at the apical-most region (Figure 3A). CEA and CD26 were expressed at the cell surface and at the apical membranes (Figure 3A). After treatment with MAb 1H8, occludin disappeared. CEA and CD26 were clearly seen between the cells, and the expression was observed from the apical membrane to the basolateral membrane (Figure 3A). In control T84 cells, claudin-1, −4, JAM-A, ZO-1, and E-cadherin were expressed at cell borders, and the expression was observed from the apical membrane to the basolateral membrane (Figure 4A). After treatment with MAb 1H8, no localization change was observed (Figure 4A).
No Changes in Expression of Occludin, Claudin-1, −4, JAM-A, ZO-1, and E-cadherin by Treatment With MAb 1H8 in T84 Cells
To examine changes in expression of occludin, claudin-1, −4, JAM-A, ZO-1, and E-cadherin in T84 cells treated with MAb 1H8 for 2 hr, we performed Western blotting. After treatment with MAb 1H8, no change of occludin, claudin-1, −4, JAM-A, ZO-1, or E-cadherin was observed compared with the control (Figure 4B).
Changes in Fence Function but Not Barrier Function of TJs Caused by Treatment With MAb 1H8 in T84 cells
To examine whether MAb 1H8 affected the fence and barrier functions of TJs in T84 cells, we performed diffusion assay with BODIPY-sphingomyelin for fence function assay and examined TER for barrier function assay. The BODIPY-sphingomyelin of control T84 cells was effectively retained in the apical domain (Figure 3B). After treatment with MAb 1H8 for 2 hr, the probe diffused through the TJ, strongly labeled the basolateral and basal surfaces, and appeared to penetrate the cells (Figure 3B). However, when we measured TER at 1, 2, 3, and 24 hr after MAb 1H8 treatment, no change of TER was observed (Figure 5A).
Changes of TJ Strands Caused by Treatment With MAb 1H8 in T84 Cells
To examine changes of TJ strands caused by treatment with MAb 1H8, we made freeze-fracture replicas. In freeze-fracture replicas of both the control and cells treated with MAb 1H8, developed networks of continuous TJ strands were observed (Figure 5B).
Changes in Localization of CEA, CD26, and Occludin Caused by Treatment With MAb 1H8 in T84 Cells During Ca Switch
We examined the effects of MAb 1H8 on the expression and localization of CEA, CD26, and occludin during development of newly formed TJs in a Ca-switch assay. When T84 cells were replaced with normal Ca medium after incubation with low Ca medium overnight, they were or were not treated with MAb 1H8. In T84 cells after incubation with low Ca medium overnight, occludin disappeared, and CEA and CD26 were observed from the apical membrane to the basolateral membrane (Figure 6). In the control, occludin appeared at the apical-most region, and CEA and CD26 were expressed at the cell surface and at the apical membranes from 4 hr after replacement with normal Ca medium (Figure 6). However, in treatment with MAb 1H8, occludin appeared at the apical-most region, and CEA and CD26 were expressed at the cell surface and at the apical membranes 12 hr after replacement with normal Ca medium, whereas occludin disappeared at 4 hr and 8 hr after treatment, and CEA and CD26 were observed from the apical membrane to the basolateral membrane (Figure 6).
Discussion
The TJ regulates epithelial cell polarity and paracellular permeability. Occludin was the first integral membrane protein of the TJ to be discovered and is involved in permeability barrier function (Furuse et al. 1993; Ando-Akatsuka et al. 1996; Balda et al. 1996; McCarthy et al. 1996; Matter and Balda 1999). The second extracellular loop is required for stable assembly of occludin in the TJ and organization of the paracellular barrier (Medina et al. 2000), and it regulates the localization of TGF-β type I receptors expressed on the basolateral membranes during EMT (Barrios-Rodiles et al. 2005). However, the role of the second extracellular loop of occludin in the localization of the proteins expressed on the apical membranes and the fence function, which may be closely related to epithelial cell polarity, is still unclear. In the present study, to investigate whether the second extracellular loop of occludin affected the localization of CEA and CD26 expressed on the apical membranes, and the fence function of TJ as determined by BODIPY-sphingomyelin diffusion, the human intestinal epithelial cell line T84 was treated with the monoclonal anti-occludin antibody 1H8, corresponding to the second extracellular loop of occludin. In treatment with MAb 1H8, occludin disappeared, and CEA and CD26 were observed from the apical membrane to the basolateral membrane, whereas in controls occludin was expressed at the apical-most region of the cell borders, and CEA and CD26 were expressed at the cell surface and at the apical membranes. Furthermore, a decrease in the fence function of TJ was observed without changes in the TJ strands or barrier function.
Immunocytochemistry (
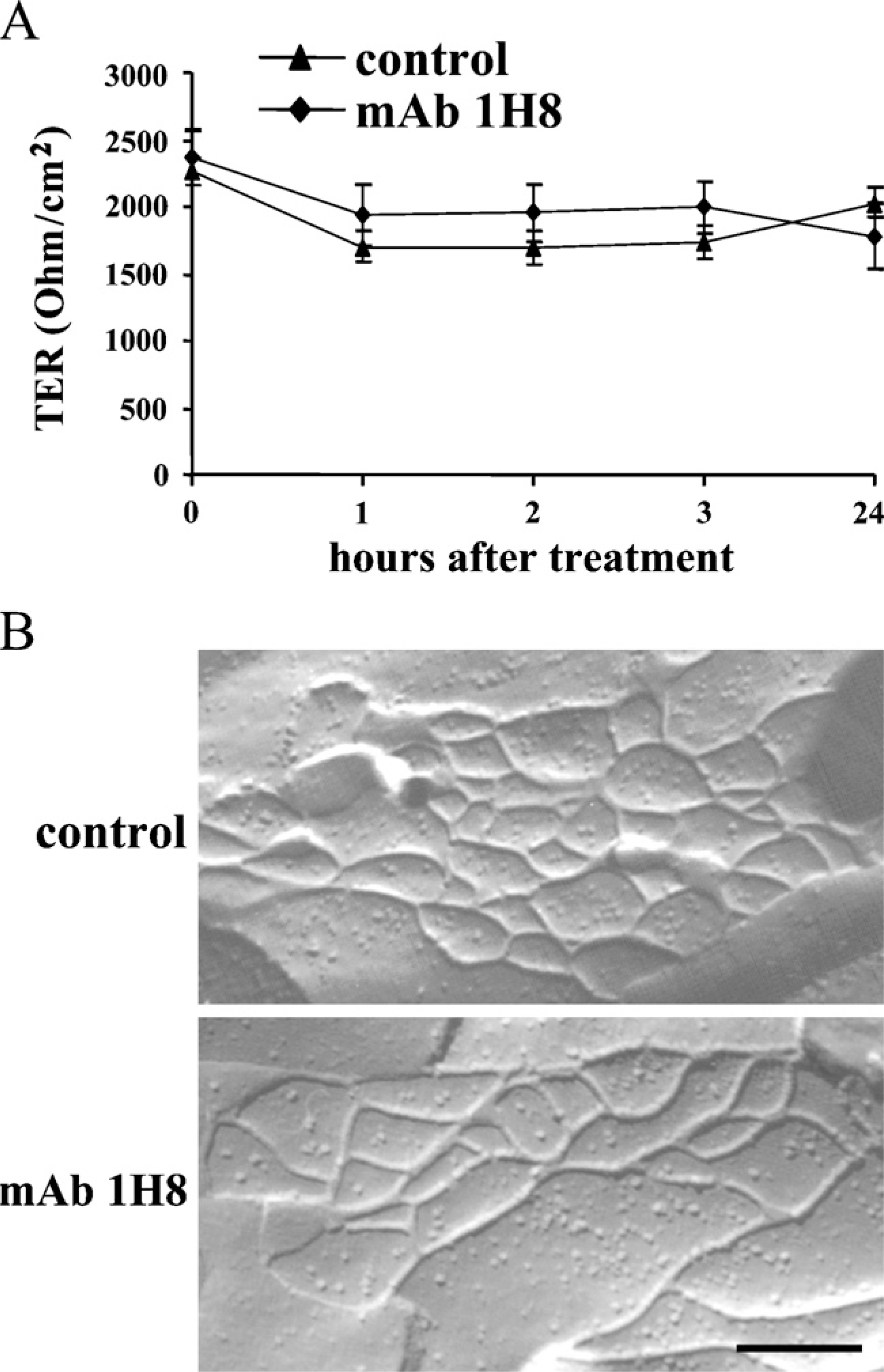
Barrier function (
When a synthetic peptide corresponding to the second extracellular loop of occludin was added to the culture medium, it drove the endogenous occludin from TJ areas of cultured epithelial cells without affecting the gross epithelial cell morphology (Wong and Gumbiner 1997). Peptides corresponding to the second extracellular loop of occludin are associated with disruption of the TJ barrier (Wong and Gumbiner 1997). Expression of COOH terminally truncated occludin leads to an increase in paracellular flux of small molecular tracers and a decrease in fence function as determined by BODIPY-sphingomyelin diffusion in the membrane (Balda et al. 1996). Knockdown of occludin by siRNA in MDCK II cells is associated with decreases in claudin-1 and −7 and increases in claudin-3 and −4, whereas the fence function of TJ is not impaired by the suppression of occludin (Yu et al. 2005). In the present study, when MAb 1H8 was added to the culture medium, endogenous occludin disappeared from TJ areas of T84 cells without the change of protein expression, whereas no changes in the expression and distribution of the TJ proteins claudin-1, −4, JAM-A, ZO-1, and the adherens junction protein E-cadherin were observed compared with the control. Although the mechanisms of the disappearance of occludin after treatment with MAb 1H8 were unclear, it specifically affected the expression and function of occludin.
During Ca depletion in MDCK cells, an initial quick drop of TER is accompanied by tyrosine dephosphorylation and the disappearance of occludin (Chen et al. 2002). In the present study, during Ca depletion of T84 cells by treatment with EDTA, the disappearance and tyrosine dephosphorylation of occludin were observed together with changes in localization of CEA and CD26 (data not shown). Furthermore, when T84 cells pre-cultured in low Ca medium were recultured in normal Ca medium in the presence of MAb 1H8, recruitment of occludin to the apical-most membranes and restoration of the distribution of CEA and CD26 were markedly retarded compared with the control. These results indicated that MAb 1H8 might affect the development of epithelial cell polarity by inhibition of the recruitment of occludin during TJ assembly.
It is thought that loss of functional TJs is associated with epithelial cell-derived cancers as loss of epithelial cell polarity (Li and Mrsny 2000). Downregulation or disappearance of occludin was observed during tumor progression of human colorectal carcinoma, human endometrial carcinoma, and human lung carcinoma (Tobioka et al. 2002, 2004a, b). Downregulation of occludin was also observed during oncogenic Raf-1-induced EMT, which is associated with loss of epithelial cell polarity (Li and Mrsny 2000; Lan et al. 2004). Furthermore, our recent study has revealed that the methylator phenotype of occludin provides enhanced tumorigenic, invasive, and metastatic properties of cancer cells (Osanai et al. 2006). These findings suggest that abnormality in expression of occludin may be related to tumor progression.
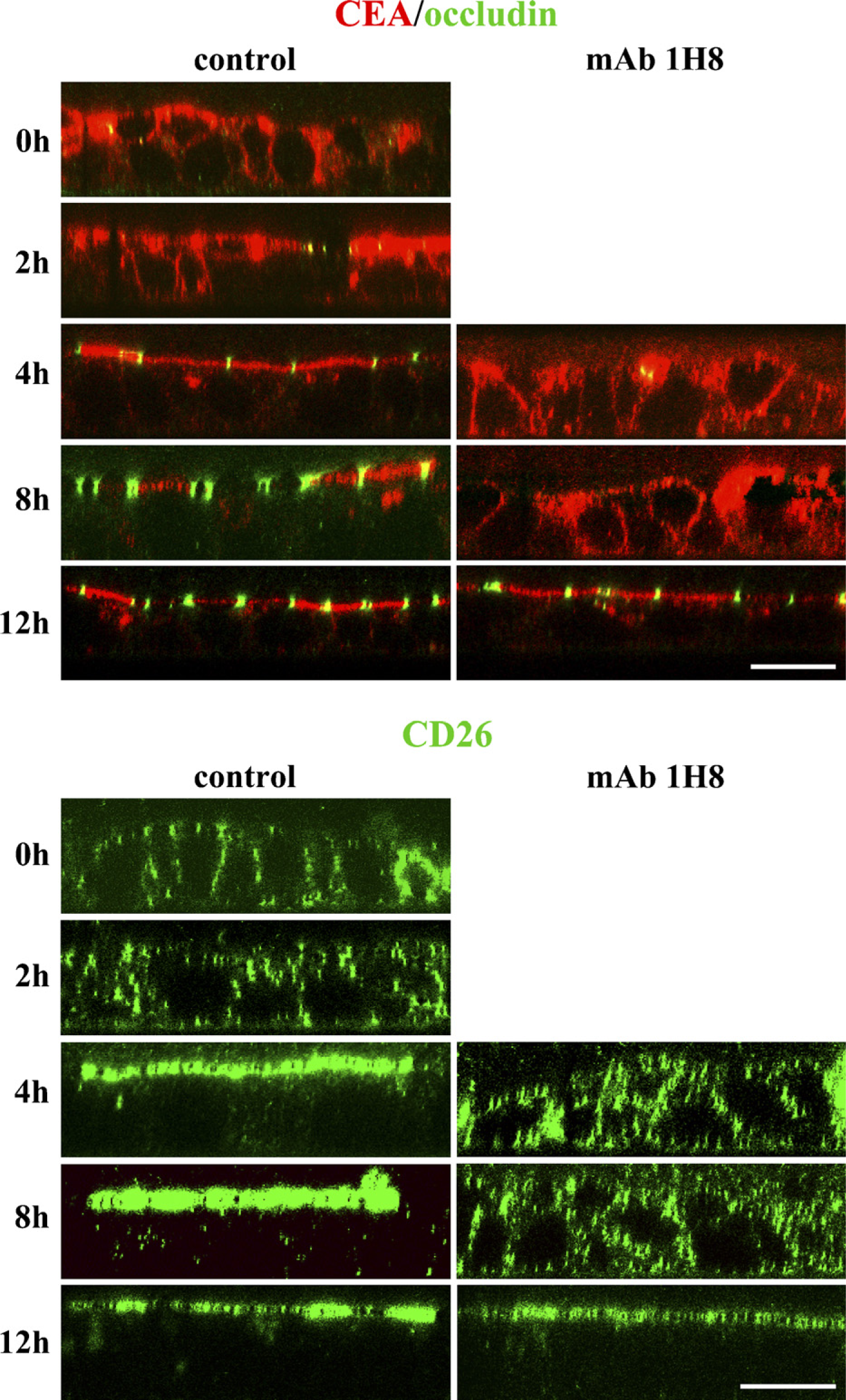
Immunocytochemistry for CEA, CD26, and occludin in T84 cells during calcium (Ca) switch. In the control, occludin appeared at the apical-most region, and CEA and CD26 were expressed at the cell surface and at the apical membranes from 4 hr after replacement with normal Ca medium (4 hr). After treatment with MAb 1H8, occludin appeared at the apical-most region, and CEA and CD26 were expressed at the cell surface and at the apical membranes at 12 hr after replacement with normal Ca medium (12 hr). Bar = 20 μm.
In conclusion, MAb 1H8 against the second extracellular loop of occludin selectively affects formation of the apical/basolateral intramembrane diffusion barrier, and the second extracellular loop of occludin plays a crucial role in maintenance of epithelial cell polarity by TJ. It is reported that the PAR-3-aPKC-PAR-6 complex is involved in the establishment of apical-basal polarity of epithelial cells (Ohno 2001). Furthermore, Crumbs-PALS1-PATJ complex regulates TJ formation and epithelial cell polarity (Shin et al. 2005). In the present study, although we never examined the changes in expression of these molecules, it is possible that MAb 1H8 may have affected them.
