Abstract
This study identifies the actin-binding protein, coronin-1a, as a novel and effective immunohistochemical marker for microglia in both cell cultures and in formaldehyde-fixed, paraffin-embedded tissue. Antibodies to coronin-1a effectively immunostained microglia in human, monkey, horse, rat, and mouse tissues, even in tissues stored for long periods of time. The identity of coronin-1a-immunoreactive cells as microglia was confirmed using double immunolabeling with cell type-specific markers as well as by morphological features and the distribution of immunoreactive cells. These properties are shared by another actin-binding protein, IBA-1. Unlike IBA-1, coronin-1a immunoreactivity was also detected in lymphocytes and certain other hematopoietic cells. The results indicate that both coronin-1a and IBA-1 are robust markers for microglia that can be used in routinely processed tissue of humans and animals. Because both coronin-1a and IBA-1 are actin-binding proteins that play a role in rearrangement of the membrane cytoskeleton, it suggests that these proteins are critical to dynamic properties of microglia.
C
The mammalian genome contains seven distinct, but related, coronin family members (de Hostos et al. 1993; Rybakin and Clemen 2005), each of which is an abundant cytoplasmic protein with cell type-specific expression patterns. One mammalian coronin family member, referred to here as coronin-1a, is restricted in expression to hematopoietic cells. This protein has been identified in different ways by several groups and is consequently also known as coronin 1, p57, Clabp, clipinA, TACO, or simply coronin (reviewed by de Hostos 1999). In macrophages and lymphocytes, coronin-1a accumulates at sites of rearrangements of the actin cytoskeleton (Rybakin and Clemen 2005). Studies looking at the biological role of coronin-1a have implicated coronin-1a in phagocytosis (Ferrari et al. 1999), T-cell activation (Nal et al. 2004), and integration of extracellular signals to the actin cytoskeleton in leukocytes (Gatfield et al. 2005). Recently, gene knockout of coronin-1a in murine T cells leads to reduced ability to respond to chemotactic gradients and also increased susceptibility to apoptotic stimuli (Foger et al. 2006). Although the relationship between the actin cytoskeleton and coronin-1a is incompletely understood, these properties have lead researchers to believe that coronin-1a is involved in modulating rearrangement of the actin cytoskeleton during immune-specific functions (Oku et al. 2005).
There are no previous reports on coronin-1a immunoreactivity in the central nervous system (CNS). Because coronin-1a is found in cells of hematopoietic lineage, we wondered if it might be expressed in microglia. Labeling of microglia with coronin-1a antibodies might be particularly useful because coronins are abundant and evolutionarily conserved, so that robust recognition of microglia might be readily achieved in tissues from a wide variety of species. This is the first study to demonstrate expression of coronin-1a in microglia of multiple species, including humans.
Microglia are cells of the mononuclear phagocyte system and have been proposed to be of hematopoietic origin, but this remains controversial (Jordan and Thomas 1988; Cuadros and Navascues 1998). Micro-glia are thought to represent 5–20% of glia (Lawson et al. 1990, 1992), which may account for the relatively low levels of coronin-1a expression detected in Western blots and Northern blots of brain (Okumura et al. 1998; Ferrari et al. 1999). Microglia and macrophages rely heavily on dynamic changes in membrane cytoskeleton for their functions. When microglia become activated, they undergo dramatic changes in cell shape, proliferation, migration and phagocytosis (Nakajima and Kohsaka 1993; Kreutzberg 1996), which are all closely related to rearrangements of the actin cytoskeleton (Greenberg 1995; Mitchison and Cramer 1996). The biological properties of coronin-1a suggest that it is likely to play an important role in rearrangement actin in the membrane cytoskeleton of microglia.
Ionized calcium-binding adaptor molecule 1 (IBA-1), also know as allograft inflammatory factor-1 (AIF1), is another recently discovered protein with actin-binding properties that is expressed in hematopoietic cells (Sasaki et al. 2001). A number of immunohistochemical (IHC) studies, mostly in non-human animals, have utilized IBA-1 as a marker for microglia (Ito et al. 1998, 2001; Okere and Kaba 2000; Hirayama et al. 2001). Very few studies have reported IBA-1 as a microglial marker in human tissue or in formaldehyde-fixed, paraffin-embedded tissue (Ohyu et al. 1999). In this study we compare and contrast the staining properties of coronin-1a and IBA-1, highlighting these actin-binding proteins as effective microglial markers for routine IHC.
Materials and Methods
Generation and Characterization of Coronin-1a Antibodies
A cDNA encoding human coronin-1a was isolated by PCR from a human leukocyte large insert cDNA library (Clontech; Mountain View, CA) using primers designed to amplify the full-length coding sequence. A band of the expected size was isolated and ligated into PCR2.1 T overhang shuttle vector, and the sequence was verified by direct sequencing. The insert was found to be 100% identical to the human coronin-1a sequence (Genbank accession NM_007074). The full-length open-reading frame of the sequence was further subjected to PCR to add a 5′ EcoRI site immediately in front of the initiator methionine codon and a 3′ SalI site immediately after the stop codon. This PCR product was then ligated into pATH11 vector for expression of the full-length protein in Escherichia coli. A Trp-E-coronin-1a fusion protein was generated essentially as described previously (Harris et al. 1991). Briefly, fusion protein expression was induced by indoleacrylic acid, and the fusion protein was enriched by subjecting the bacteria to an inclusion-body preparation. This inclusion-body material was dissolved in 6 M urea and further purified by preparative SDS-PAGE. The purified Trp-E-coronin-1a construct was injected into rabbits to generate a polyclonal antibody.
For immunoblotting, HL60 cells were differentiated by a 7-day treatment with DMSO and then stimulated with 100 nM formyl-Met-Leu-Phe (fMLP; Sigma, St Louis, MO) for 2 min. HL60 cells were lysed in Laemmli's SDS buffer by boiling for 4 min. Lysates were separated with 7.5% SDS-PAGE and transferred to PVDF, blocked in 5% non-fat dry milk in Tris-buffered saline (TBS) for 1 hr, and incubated with 1:10,000 rabbit coronin-1a antisera overnight at 4C. The PVDF membrane was washed three times in 0.05% Tween-20 in TBS and then incubated with 1:10,000 horseradish peroxidase-conjugated anti-rabbit antibody (Amersham Biosciences; Pittsburgh, PA) for 1 hr at room temperature. After further washing in TBS plus Tween-20, the signal was detected with reagents in the Amersham chemiluminescence kit according to the manufacturer's instructions.
Cell Cultures
HL60 is a human cell line that differentiates to form neutrophil-like cells following treatment with DMSO. They were obtained from Orion Weiner (Harvard Medical School; Boston, MA). After 6 days of DMSO treatment, HL60 cells were plated onto glass coverslips, stimulated with 100 nM fMLP for 2 min, and fixed in 4% paraformaldehyde for 10 min. Cells were permeabilized with 0.05% Triton X-100 solution in PBS and incubated with rabbit coronin-1a antibody (1:100) at 4C overnight. Cells were washed three times in TBS, incubated with Alexa Fluor 488 goat anti-rabbit and red-fluorescent Alexa Fluor 594 phalloidin (both at 1:2000; Molecular Probes, Invitrogen, Carlsbad, CA) for 4 hr at room temperature. Cells were washed three times in TBS and mounted on a coverglass. Cells were imaged in a Bio-Rad MRC 1024 confocal microscope (Bio-Rad Laboratories; Hercules, CA).
Mixed cultures of neurons and glia were derived from embryonic day 18 (E18) rat embryos and grown as described (Lu et al. 1998). Cells were fixed in 3.7% formaldehyde in 0.1 M PBS for 1 min, followed by washing in 220C methanol for 1 min. Cells were incubated with rabbit coronin-1a antiserum at a dilution of 1:1000 for 1 hr at room temperature and then incubated with 1:2000 goat anti-rabbit Alexa Fluor 488 conjugate (Molecular Probes). For double-labeling experiments, the coronin-1a antibody was incubated as described, along with an appropriate chicken antibody. The neuronal intermediate filament protein α-internexin was detected (Figure 1E) with a chicken antibody IgY preparation used at a dilution of 1:2000 (EnCor Biotechnology Inc.; Gainesville, FL). We also used chicken IgY antibodies to the neuronal marker microtubule-associated protein 2 (MAP2); neurofilament subunits (low, middle, and high); and the glial markers, glial fibrillary acidic protein (GFAP), vimentin, and myelin basic protein (data not shown). These antibodies were obtained from EnCor Biotechnology Inc. and were used at dilutions between 1:1000 and 1:100,000, according to the manufacturer's data sheets. In double-labeling experiments, goat-anti rabbit Alexa Fluor 488 conjugate was incubated with 1:2000 Alexa Fluor 594 goat anti-chicken conjugate (Molecular Probes). Cultures were also incubated with coronin-1a antibody as described above and simultaneously incubated with fluorescein-labeled Griffonia simplicifolia isolectin-B4 (isoB4) at a concentration of 1:100 (Sigma). In this case, the coronin-1a antibody localization was visualized using 1:2000 goat ant-rabbit Alexa Fluor 594 (Molecular Probes). Cells were imaged with a Zeiss Axioskop 2 fitted with a 2 megapixel Spot RT-slider camera (Diagnostic Instruments; Sterling Heights, MI).
Human Tissues
A range of autopsy-confirmed neurological disorders including inflammatory, demyelinating, and degenerative conditions were studied for immunoreactivity with coronin-1a and IBA-1. Human material used is summarized in Table 1. Archival paraffin tissue that had been stored as glass-mounted sections or obtained from freshly cut paraffin blocks were studied to address the stability of the antigen with respect to tissue storage. Glass-mounted tissue sections ranged in storage from 5 months to 20 years. Samples from the nervous system included a variety of neuroanatomic regions including gray and white matter in cortical and subcortical regions, as well as the spinal cord and peripheral nerve roots. All archival human tissue had been fixed in formaldehyde and embedded in paraffin by routine histological practice.
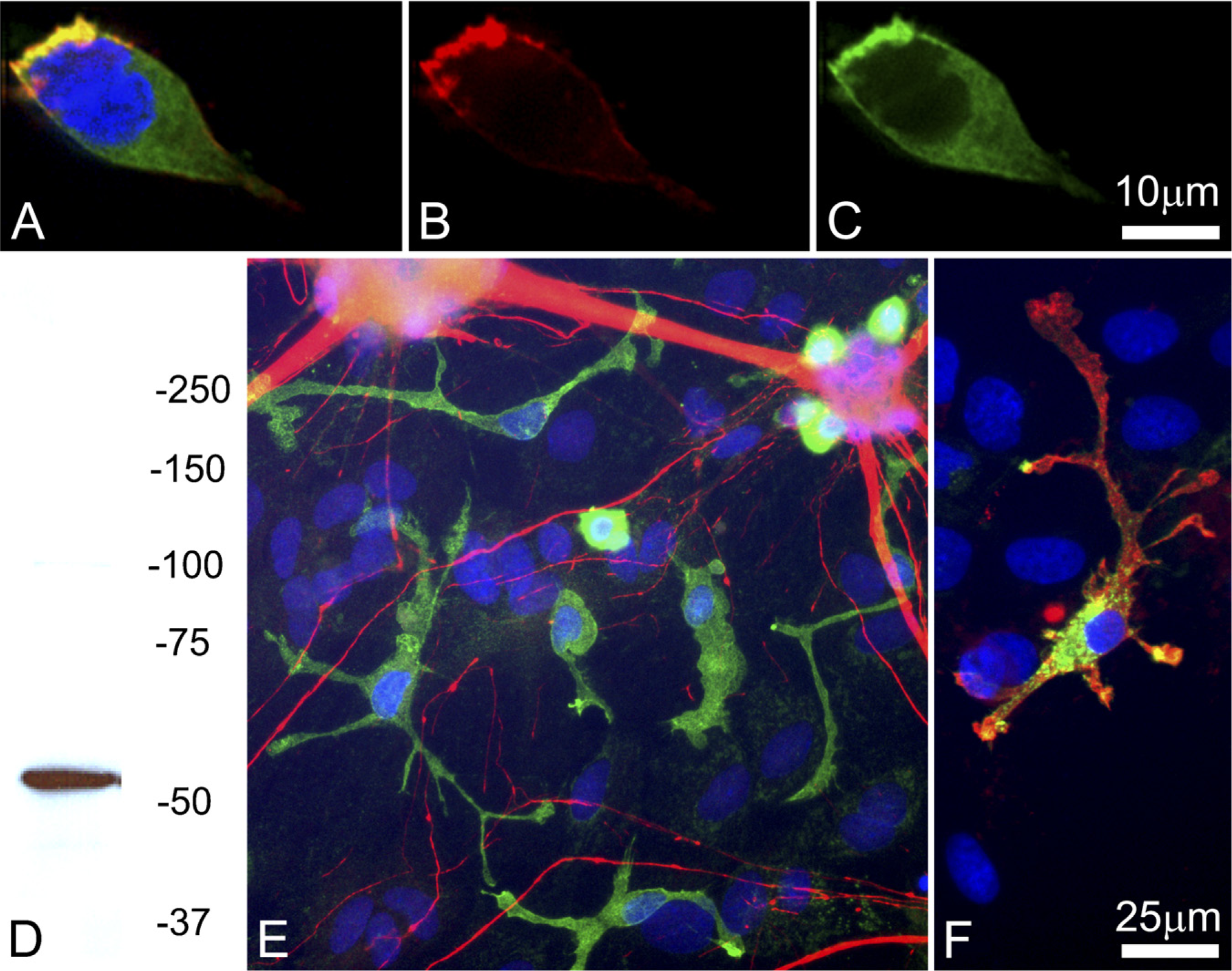
Images of differentiated and fMLP-stimulated HL60 cells stained with antibody to coronin-1a (red channel in A, B), fluorescent phalloidin (green channel in
Human tissue
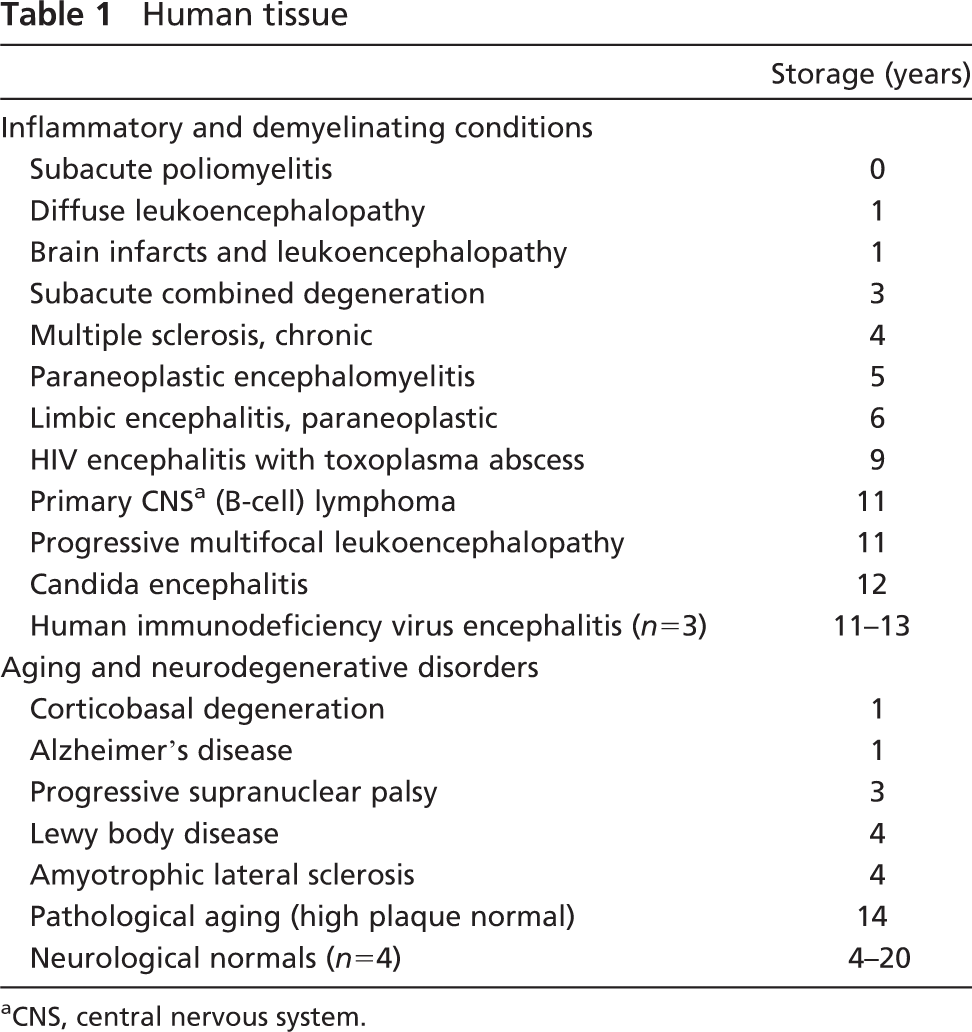
CNS, central nervous system.
To investigate the staining pattern in non-CNS tissues, several other organs from a case with limbic encephalitis were also sampled, including tissue from pancreas, bone marrow, thyroid, prostate, stomach, esophagus, liver, spleen, lung, and heart.
Animal Tissues
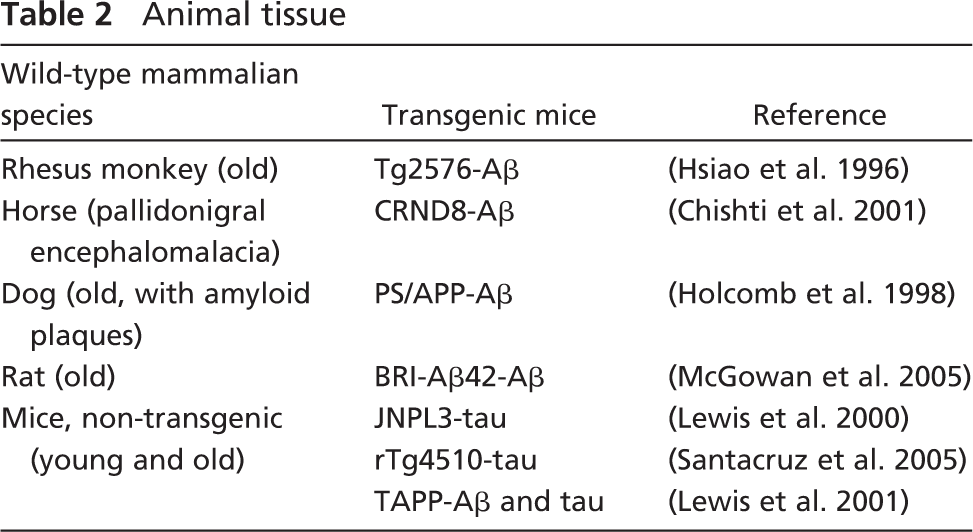
To analyze the species specificity of coronin-1a and IBA-1, brain tissue from several other mammalian species was studied (Table 2) including several different types of transgenic mice, as well as age- and background strain-matched non-transgenic controls. Most animal tissue was fixed and processed in the same manner as the human tissues. All rodent specimens were obtained from animals handled by procedures approved by the Mayo Clinic Institutional Animal Care and Use Committee. Other animal tissue was available from veterinarians or from referral sources with approved research programs. The aged Rhesus monkey sample was generously supplied by Alan Peters (Boston University; Boston, MA). Samples of brain tissue from equine nigropallidal encephalo-malacia were from the University of Minnesota veterinary service and provided by Dr. Demetrius Maraganore (Mayo Clinic College of Medicine). The aged dog brain was provided by a local veterinarian for diagnostic evaluation from a humanely sacrificed animal.
Animal tissue
Optimal Fixation for Coronin-1a IHC
Three commonly used fixatives, paraformaldehyde (4%), formaldehyde (10%), and ethanol (100%), were used to determine the optimal fixation for coronin-1a IHC. Non-transgenic mouse brain tissue was embedded in paraffin after fixation with these three fixatives. No qualitative difference was detected in the number or morphology of microglia; however, intensity of the microglial staining appeared slightly better with formaldehyde fixation compared with paraformaldehyde and ethanol fixation.
The length of time in fixative was also studied for formaldehyde and ethanol fixation. No qualitative difference was detected in microglial staining for tissue fixed for 24 hr or for 1 week.
IHC
Paraffin-embedded tissue was sectioned at a thickness of 5 μm and mounted on glass slides. After removal of paraffin in xylene and rehydration in a graded series of alcohols (100%, 100%, 95%), sections were steamed at high power for 30 min in distilled water for antigen retrieval. The primary antibodies were rabbit antisera raised against coronin-1a as described above and a commercial antibody to IBA-1 (1:1000; Wako Chemicals USA, Richmond, VA). To confirm specificity of the rabbit antiserum to coronin-1a, some sections were also stained with a commercially available coronin-1a rabbit antiserum raised against recombinant full-length human coronin-1a (1:250; Upstate USA, Chicago, IL). Immunostaining was performed on a DAKO Autostainer according to the manufacturer's instructions using DAKO Envision + System peroxidase kit, with 3,3′-diaminobenzidine as the chromogen (DAB; Dako, Carpinteria, CA). After immunostaining, sections were briefly counterstained with hematoxylin and coverslipped.
Several commonly used heat- and chemical antigen-retrieval techniques, along with a range of primary antisera dilutions, were tested to determine the optimal staining conditions for both coronin-1a and IBA-1.
Double Immunofluorescence
In a selection of human and mouse tissues, sections were stained with coronin-1a and either a glial- or lymphocyte-specific mouse monoclonal antibody to confirm the identity of coronin-1a-positive cells. Paraffin-embedded tissue sections were processed as above, except that they were incubated in normal goat serum (1:20; Sigma) for 20 min, followed by overnight incubation in a cocktail of coronin-1a and a primary monoclonal mouse antibody (Table 3). After washing thoroughly in PBS, sections were incubated in a cocktail of fluorochrome-conjugated goat anti-mouse (fluorescein isothiocyanate; 1:100, Pierce Biotechnology, Rockford, IL) and goat anti-rabbit (Alexa Fluor 568; 1:200, Molecular Probes) secondary antibodies for 1 hr at room temperature. Sections were washed in distilled water before being coverslipped using Vectorshield mounting media (Vector Laboratories; Burlingame, CA). Slides were examined using a confocal fluorescent microscope (Fluoview, Version 2.0; Olympus America Inc., Melville, NY). Images were collected using single excitation for each wavelength, separately, and channels were subsequently merged using an image-processing program (Olympus Fluoview).
Results
Coronin-1a in Cultured Cells-Western Blot and Immunocytochemistry
We expected that coronin-1a antibodies would stain the leading edge of migrating neutrophils and therefore grew cultures of HL60 cells, a human cell line that can be differentiated to a neutrophil morphology. Cells were stimulated to differentiate by treatment with DMSO, and addition of the peptide fMLP further activated a migratory and chemotactic morphology. The coronin-1a antibody stained the leading edge of these cells, which was associated with actin as revealed by fluorescent phalloidin (Figure 1). Coronin-1a immuno-reactivity was clearly concentrated at the leading edge of this migratory cell in a pattern overlapping actin. Western blotting of crude homogenates of HL60 cells with the coronin-1a antibody revealed a single strong band at ~57 kDa, the expected molecular mass for coronin-1a (Figure 1D). No band could be detected in similar homogenates of the non-hematopoietic cell line, Hek293, which also showed no coronin-1a immunoreactivity with immunocytochemistry (not shown). In mixed cultures derived from E18 rat cortex grown for 1 week in tissue culture, a class of small cells with short, often bulbous processes was immunoreactive for coronin-1a (Figure 1E). These cells did not stain with antibodies to MAP2, neurofilament subunits, GFAP, or myelin basic protein, although they were positive for vimentin (not shown). The coronin-1a staining appeared to be primarily associated with the plasma membrane; however, in contrast to the HL60 cells, coronin-1a staining appeared on all extensions of these cells. In both cell types, coronin-1a staining was excluded from the nucleus. The coronin-1a-positive cells had small, compact, and elongated nuclei, typical for microglial cells. When cultures were double labeled with isoB4, a lectin specific to microglia (Streit and Kreutzberg 1987), the coronin-1a-positive cells (shown in the red channel in Figure 1F) were clearly also positive for isoB4 (green channel in Figure 1F). In summary, all findings show that the coronin-1a-positive cells do not express markers specific for neurons, astrocytes, or oligodendrocytes but do express microglial markers (Figure 1F).
Coronin-1a Immunoreactivity in Human Tissue
Coronin-1a IHC revealed cells with the morphology and distribution consistent with microglia (Dickson and Lee 1996), including ramified and bipolar parenchymal microglia, as well as perivascular cells and histiocyte-like cells in the leptomeninges and choroid plexus. The cellular localization of coronin-1a was usually most intense at the plasma membrane but also present in cytoplasm. In non-CNS tissues, coronin-1a expression was restricted to hematopoietic cells, including lymphocytes and tissue histiocytes (Figure 2). Erythrocytes and normocytes in bone marrow were negative, whereas immunoreactivity was detected in megakaryocytes and myeloblasts. Interestingly, certain coronin-1a-positive cells in peripheral tissues such as tissue histiocytes of the heart, Kupffer cells of the liver, and Langerhans cells of the esophagus had a ramified or dendritic appearance similar to microglia in the CNS.
Primary antibodies and their specificity
IF, immunofluorescence; IHC immunohistochemistry; IC, immunocytochemistry; GFAP, glial fibrillary acidic protein.
In brain tissue from neurologically normal individuals, coronin-1a labeled cells with multiple long, thin, branching processes consistent with ramified microglia (Figure 3). Coronin-1a-positive cells were detected in both white matter and gray matter of cortical and subcortical regions and the spinal cord. In general, micro-glial staining was less prominent in cortical gray matter than subcortical gray matter and white matter.
Consistent with results of cell culture experiments, neurons did not show coronin-1a immunoreactivity. At high antibody concentrations, weak neuronal staining was sometimes detected, but this was not detected at higher dilutions when there was still robust microglial immunoreactivity.
In pathological tissues such as Alzheimer's disease and other neurodegenerative disorders, many of the coronin-1a-immunoreactive cells resembled so-called “activated microglia,” with fewer and shorter cell processes (Figure 3). This was most evident in the hippocampus of advanced Alzheimer's disease where coronin-1a IHC labeled clusters of activated microglia associated with β-amyloid plaques, one of the pathological hallmarks of Alzheimer's disease. Brain macrophages in infarcts, abscesses, and demyelinating disorders, which are mainly derived from blood mono-cytes (Fujita and Kitamura 1976), had a foamy appearance and sometimes were lipid laden (Figure 3). In most inflammatory disorders, it was difficult to differentiate lymphocytes from small microglia, but coronin-1a-positive lymphocytes were clearly shown to present in perivascular and parenchymal infiltrates in paraneoplastic, fungal, and viral encephalitides and primary CNS lymphoma (Figure 3). These results suggest that coronin-1a has an immune cell-specific pattern of expression that includes microglia and tissue macrophages, as well as lymphocytes.
Comparable coronin-1a staining was observed in paraffin-embedded tissue that had been sectioned and mounted on slides 20 years prior to staining when compared with recently sectioned tissue, indicating that the coronin-1a epitope is stable to long storage, even in tissue mounted on glass slides.
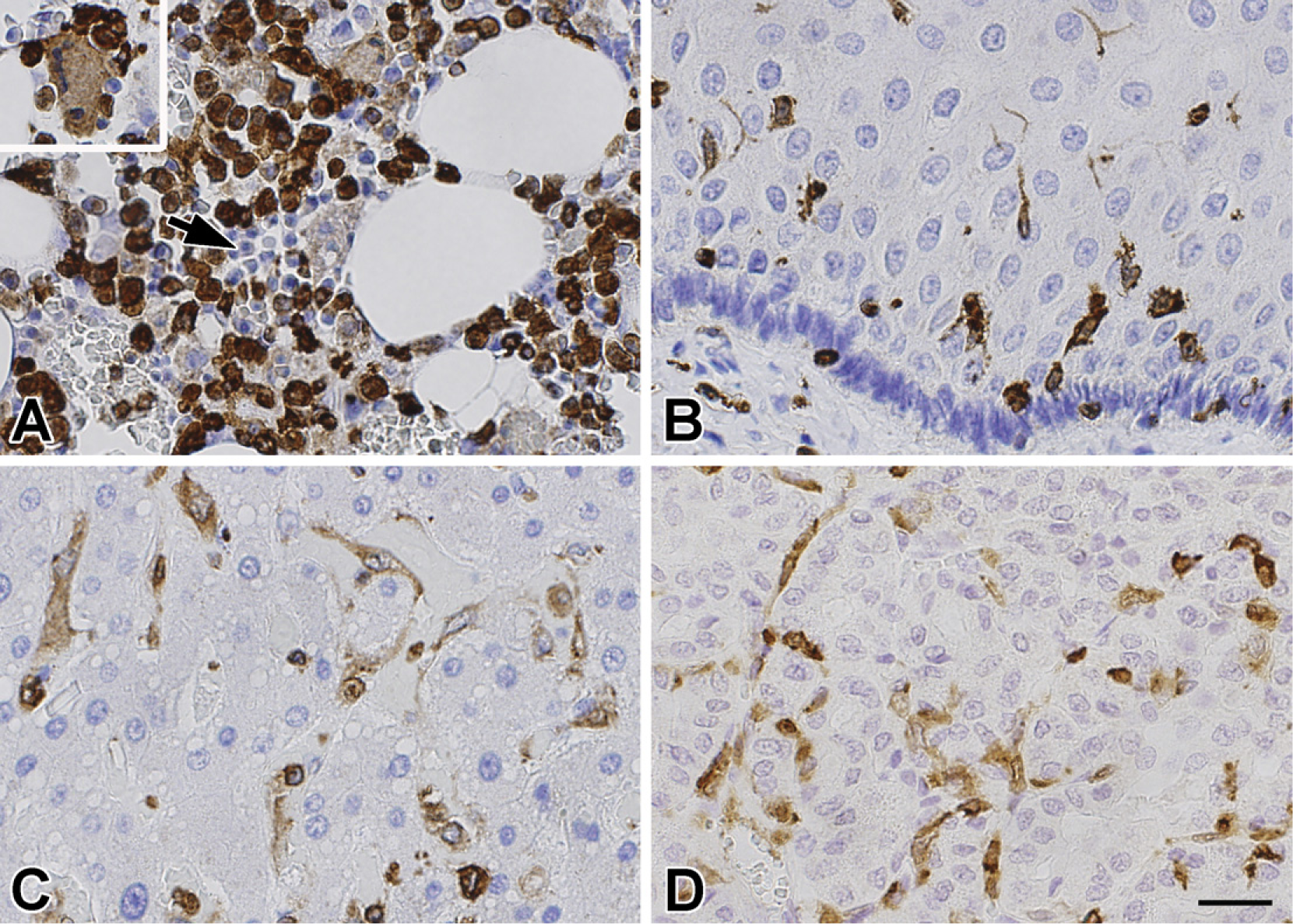
Immunohistochemical staining of non-neural tissues using coronin-1a. In bone marrow (
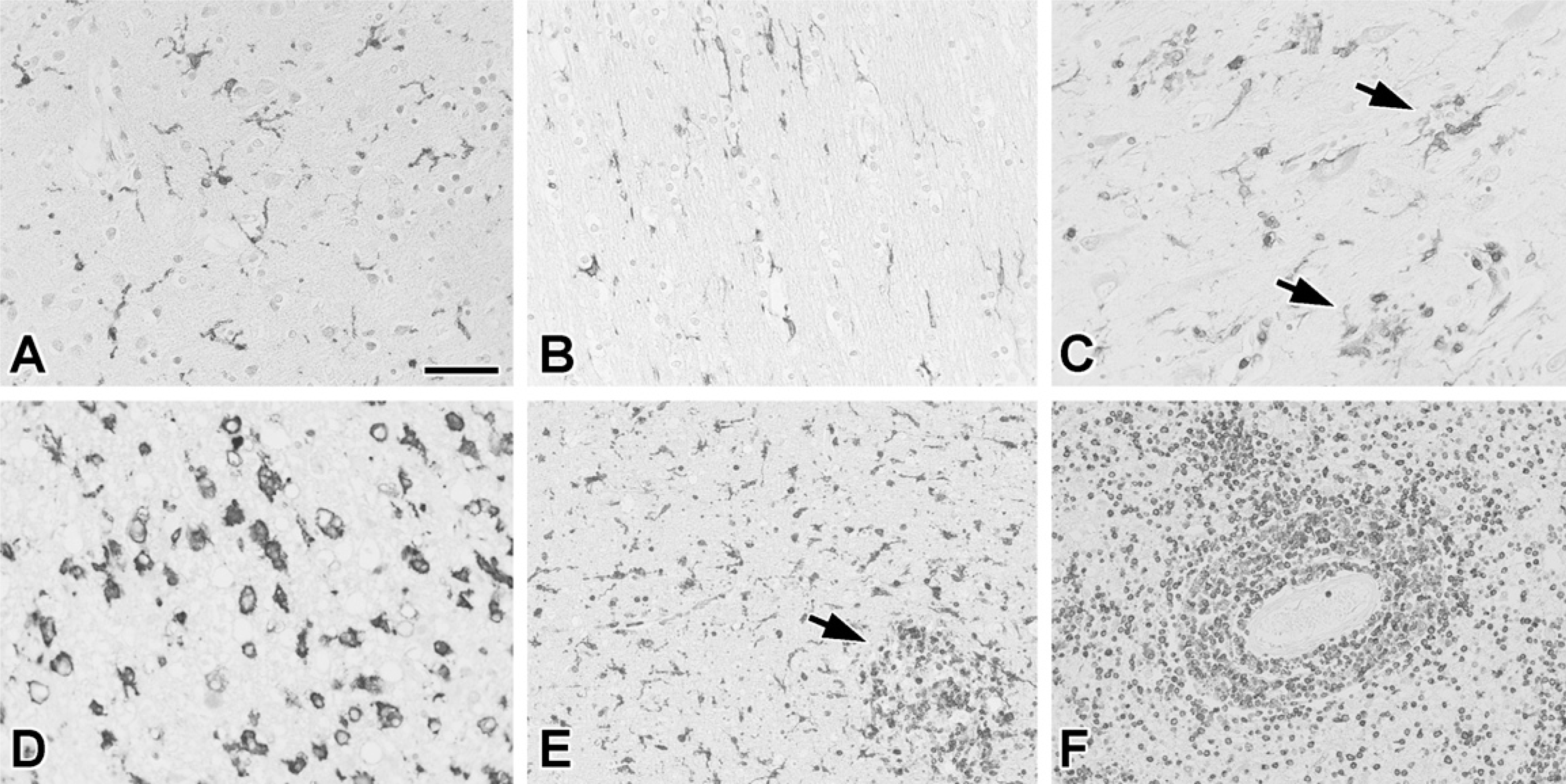
Normal and pathological human brain tissue immunostained with coronin-1a. Ramified small parenchymal cells with the morphology and distribution consistent with microglia were stained in the gray matter (
Coronin-1a Immunoreactivity in Animal Tissue
Coronin-1a is an evolutionary conserved protein (Okumura et al. 1998), suggesting that antibodies to coronin-1a might be useful in studies of a range of species. To investigate the possible species cross-reactivity, coronin-1a IHC was tested in a variety of different mammalian tissues. In line with the results on human tissues, coronin-1a immunoreactivity in CNS of all mammalian species examined was consistent with microglia (Figure 4).
Brain sections of an aged Rhesus monkey had predominantly ramified coronin-1a cells in the white and gray matter, in a staining pattern similar to that detected in neurologically normal elderly human brains. In the aged dog brain, which had numerous β-amyloid plaques, ramified microglia were prominent in the cortex where they formed clusters around plaques, similar to those found in similarly processed sections of Alzheimer's disease.
In the brain of a horse with equine nigropallidal encephalomalacia, there were subacute to chronic infarcts in subcortical regions characteristic of this disease (Farrell et al. 1971). Interestingly, coronin-1a immunoreactivity in these infarcts demonstrated morphological heterogeneity of microglia. Within and directly adjacent to the area of infarction were lipid-laden macrophages. These were surrounded by ameboid-type microglia with short, stubby processes, and these were surrounded by ramified microglia in the adjacent unaffected brain parenchyma. The appearance of morphologically distinct microglia/macrophages illustrates the functional diversity of microglia similar to that shown previously in demyelinating conditions (Dickson and Lee 1996). To reiterate, all morphological variants of microglia and macrophages were immunoreactive for coronin-1a.
Coronin-1a Immunoreactivity in Transgenic Mice
Detecting microglia in formaldehyde-fixed, paraffin-embedded mouse tissue has traditionally been difficult. To determine if coronin-1a IHC was effective under these conditions, we used a range of different transgenic (Tg) mice that exhibits pathology commonly seen in Alzheimer's and other neurodegenerative diseases. Tg mice were separated into two categories: those characterized by abnormal tau deposition (Tg-Tau) and those with β-amyloid plaques (Tg-Aβ). These were complemented with age- and strain-matched non-transgenic (NTg) controls.
Ramified microglia were consistently seen in the white matter, midbrain, and spinal cord of all the NTg controls that had no detectable pathology. In older NTg mice (20 months), coronin-1a-positive microglia were more numerous and less ramified compared with younger NTg mice (6 months). This age-related increase in microgliosis has been reported previously (Sturrock 1980). Ameboid microglia were found in areas vulnerable to tau- and β-amyloid-related pathology in Tg mice. For example, in Tg-Aβ mice, ameboid microglia were present in and around β-amyloid plaques in cortical and subcortical regions of the brain. In Tg-tau mice coronin-1a-immunoreactive microglia were present in regions that were vulnerable to tau-related neurodegeneration such as amygdala, brainstem, and spinal cord. In double (tau and Aβ) transgenic mice, so-called TAPP mice (Lewis et al. 2001), which have tau pathology in the spinal cord and brain as well as β-amyloid plaques in the cortex and hippocampus, a prominent microglial reaction was detected in older mice (18 months), with intense staining of ameboid-type microglia around plaques in the cortex and macrophage-like cells in the fiber tracts in the spinal cord that are known to undergo degeneration in this model (Figure 4).
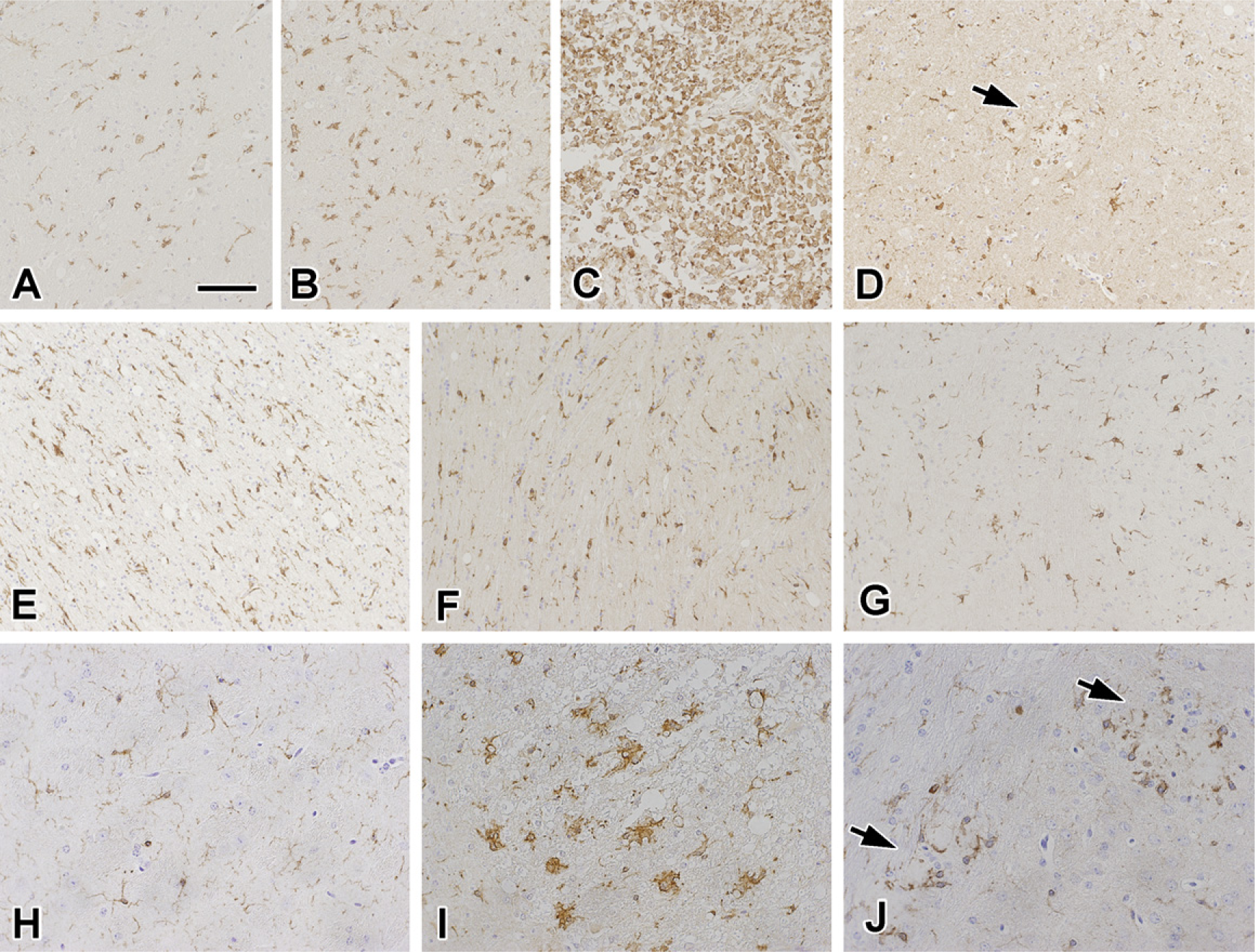
Coronin-1a immunostaining of brain tissue of several mammalian species. In equine nigropallidal encephalomalacia (
Cell Type-Specific Expression of Coronin-1a
In humans and mice, coronin-1a has previously been expressed in leukocytes. To confirm that coronin-1a specifically labeled microglia and lymphocytes, double-immunofluorescence staining was performed in human tissues using well-characterized microglial and lymphocyte markers. Not surprisingly, cells fitting the morphological description of microglia were also positive for the human microglia markers HLA-DR and CD68 (Figure 5). HLA-DR, which is a plasma membrane protein, colocalized with coronin-1a on the plasma membrane of microglia with little or no colocalization in the cytoplasm. Conversely, CD68, which is mainly expressed in the lysosomal compartment in the cytoplasm, gave a granular intracellular staining, with coronin-1a immunoreactivity on the cell surface.
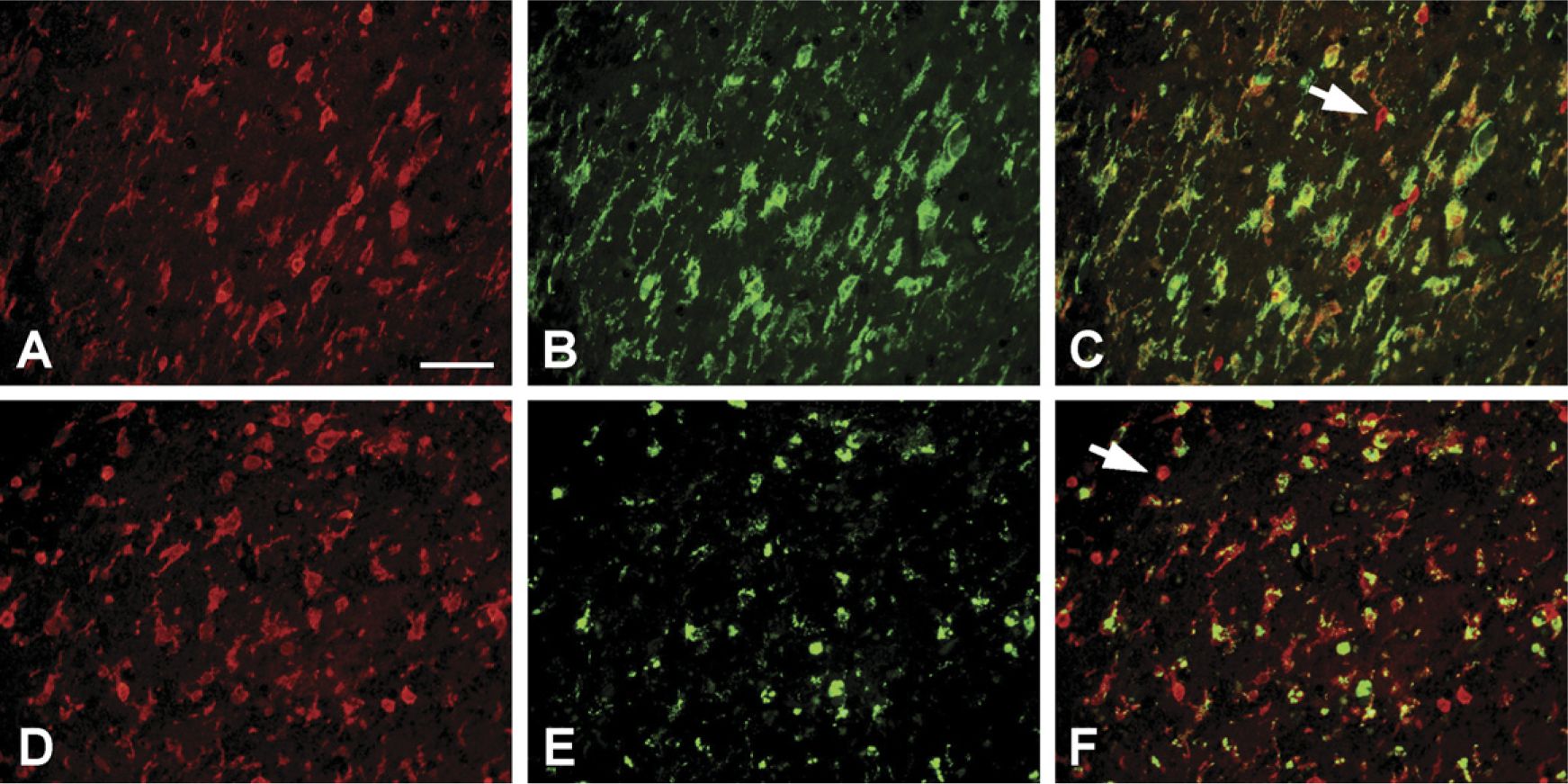
Double-immunofluorescence staining for coronin-1a (red) and for microglial markers (green) in a case of limbic encephalitis. Coronin-1a immunoreactivity (
Some cells had intense coronin-1a reactivity but were negative for both CD68 and HLA-DR. The widespread inflammation in these particular tissues and morphology of these cells suggested that these were lymphocytes. In sections of tonsil from humans (Figure 6), there was abundant coronin-1a immunoreactivity in cells of the corona and center of germinal follicles. At the cellular level, this staining was mainly at the plasma membrane and colocalized with CD20-positive B-lymphocytes in the center of germinal follicles and CD3-positive T-lymphocytes in the corona of germinal follicles. These findings were supported by IHC results from a case of primary CNS lymphoma, which also showed coronin-1a-positive perivascular and parenchymal neoplastic B-lymphocytes (Figure 3).
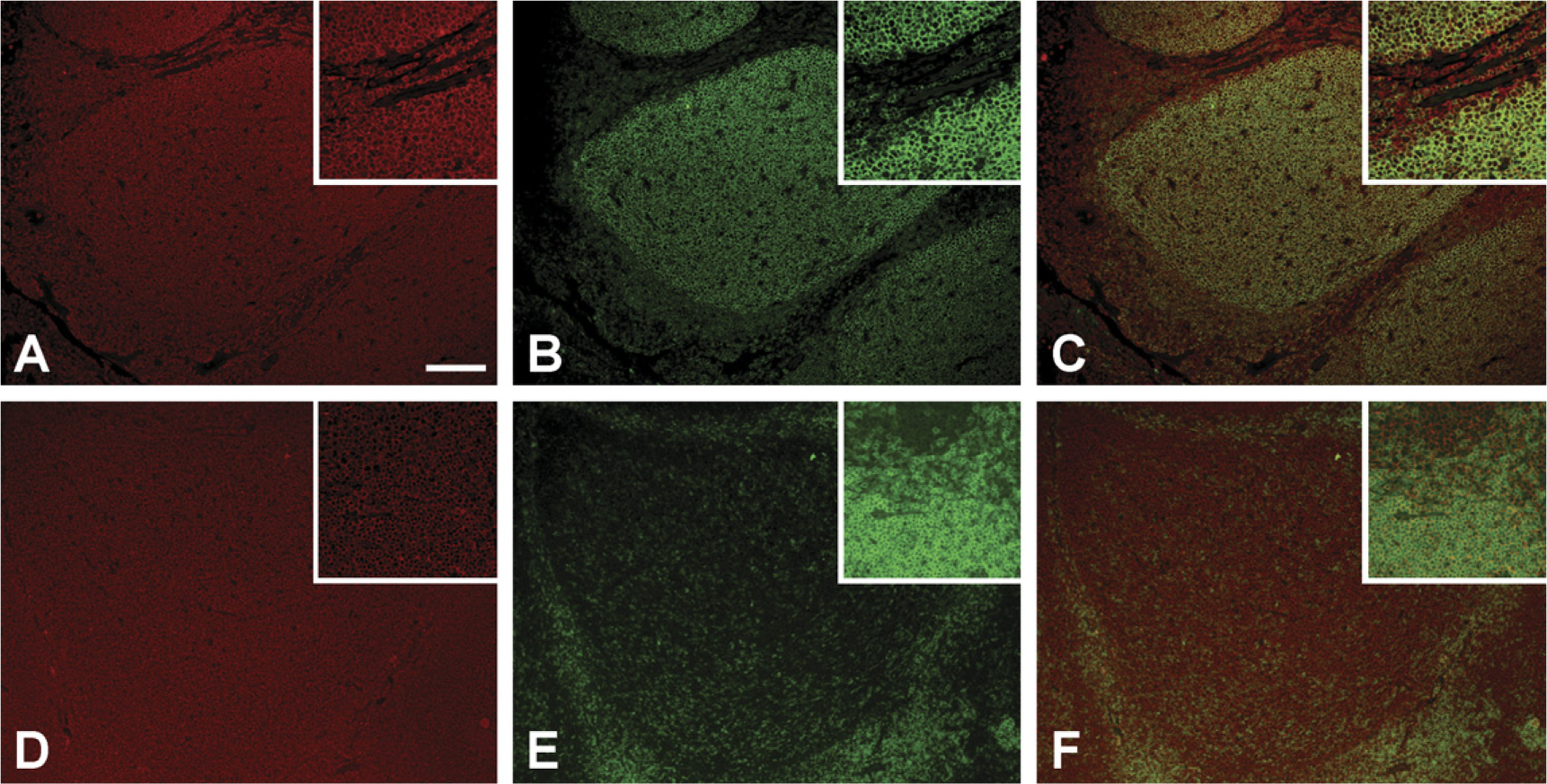
Double-immunofluorescence staining for coronin-1a (red in
The other major glial constituents of the brain are astrocytes and oligodendroglia, which are derived from the neural tube rather than from hematopoietic precursors. To rule out the possibility that coronin-1a is expressed in astrocytes, human and mouse tissues were double immunostained for coronin-1a and GFAP. In all sampled tissues, cells positive for coronin-1a were negative for GFAP and vice versa, indicating that coronin-1a does not label astrocytes and can be considered specific for microglia (and lymphocytes). In Alzheimer's disease and Tg-Aβ mice, both coronin-1a-positive microglia and GFAP-positive astrocytes were close to each other around amyloid plaques but could be clearly differentiated with double immunofluorescence (Figure 7).
Carbonic anhydrase II (CAII) has been used to label oligodendroglia in paraffin-embedded tissues (Ghandour et al. 1980). Using IHC for CAII and coronin-1a on adjacent sections of non-transgenic mouse tissue (both antibodies are generated in rabbits), the staining patterns for CAII and coronin-1a were distinctly different (not shown). These results complement results from cell culture experiments using myelin basic protein as an oligodendroglial marker (see above) and indicate that coronin-1a does not recognize oligodendrocytes.
Comparison of Coronin-1a and IBA-1
IBA-1 is a well-established IHC marker for microglia (Ito et al. 1998, 2001; Ohyu et al. 1999; Okere and Kaba 2000; Hirayama et al. 2001) that has been used to study microglial reactions in a number of different mammalian species, but less is known about its immunoreactivity in human tissues. In most reports of IBA-1 immunoreactivity of microglia in animal studies, it has not been used in formaldehyde-fixed, paraffin-embedded tissue (Ohyu et al. 1999). To confirm the identity of coronin-1a-positive cells as microglia, IBA-1 IHC was performed on adjacent sections of coronin-1a-stained tissues. Direct double labeling was problematic because both antibodies were generated in rabbits. Qualitative comparison showed that the staining pattern for IBA-1 was almost identical to that of coronin-1a, being expressed on both ramified and ameboid microglia. One major difference between coronin-1a and IBA-1 was the lack of lymphocyte immunoreactivity for IBA-1. This was evident in the human tonsil and spleen, which showed widespread immunoreactivity for coronin-1a, but IBA-1 immunoreactivity only in dendritic cells and macrophages (Figure 8). Apart from these differences, IBA-1 immunoreactivity in microglia was identical to that of coronin-1a. The antibody to IBA-1 also stained microglia in the human tissues archived for long periods of time, indicating that like coronin-1a, IBA-1 epitopes are also stable over time.
Discussion
This study identified antibodies to coronin-1a and IBA-1 as novel and effective IHC markers for microglia. In tissue culture as well as in formaldehyde-fixed, paraffin-embedded mammalian brain tissue, we show that anti-bodies to coronin-1a label microglia. Antibodies to IBA-1 were also useful IHC markers for microglia in similar tissue. In human, monkey, horse, rat, and mouse tissue, we demonstrated that antibodies to coronin-1a and IBA-1 specifically labeled cells with the morphology and distribution of microglia, even in sections archived for long periods of time. The identity of coronin-1a-and IBA-1-immunoreactive cells as microglia was confirmed using microglial-specific markers (e.g., HLA-DR and CD68), as well as the lectin, isoB4. Neither coronin-1a nor IBA-1 was detected in astrocytes or oligodendrocytes. The only significant difference between coronin-1a and IBA-1 was that coronin-1a immunoreactivity was also present in lymphocytes, whereas IBA-1 was not.
The fact that coronin-1a was effective as a microglial marker in a wide range of mammalian tissues is likely related to coronin-1a being a highly conserved protein. In fact, sequence analysis shows that human, rat, mouse, cow, and other mammalian coronin-1a sequences are exactly 461 amino acids long and can be perfectly aligned without deletions or insertions. Any sequence divergences are conservative or semiconservative, and each sequence is 94–98% identical to the others. These findings suggest that antibodies to coronin-1a are likely to be useful as a pan-microglial marker. This evolutionary conservation also suggests that coronin-1a has an important functional role in microglia.
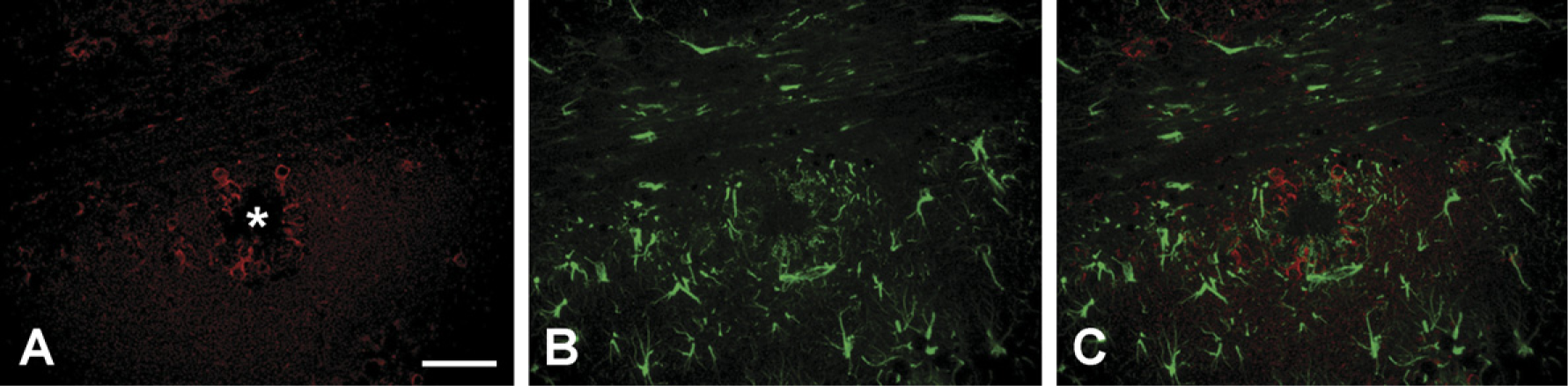
Double-immunofluorescence staining of transgenic mouse brain for coronin-1a (red in
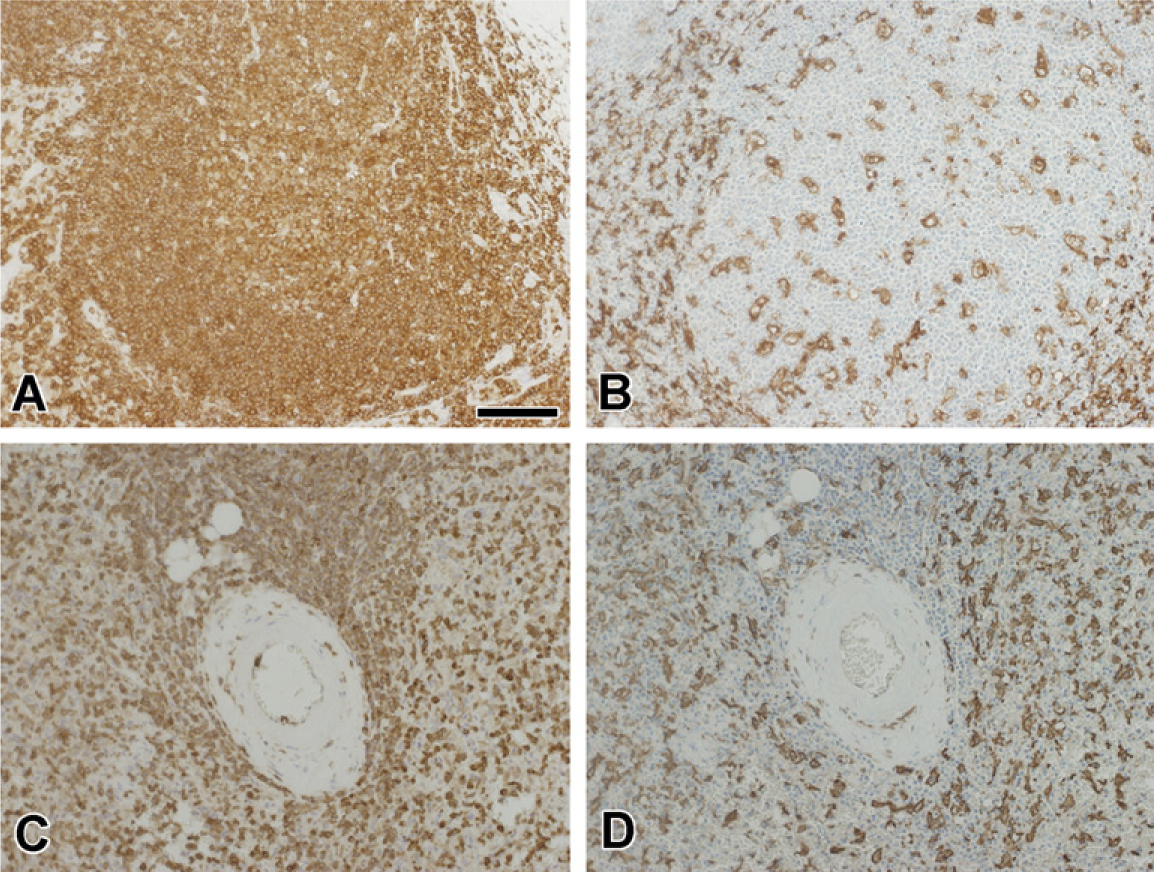
Comparison of coronin-1a (
Initial studies reported high expression of coronin-1a in immune-related tissues such as spleen, tonsil, and lymph nodes, but relatively low levels of expression in the brain (Suzuki et al. 1995; Okumura et al. 1998; Ferrari et al. 1999). At the cellular level, coronin-1a expression was largely restricted to immune system cells such as macrophages and lymphocytes. Microglia are immune cells of the brain. In mammalian tissue we demonstrated coronin-1a in both ramified and ameboid microglia. Microglia were predominantly ramified in unaffected regions of the brains, whereas ameboid microglia were common in areas undergoing some form of tissue damage. This specific pattern of microglial morphology is consistent with the pleomorphic functions of microglia (reviewed by Imai et al. 1996), with ramified microglia representing a quiescent state in the healthy brain and activation associated with ameboid morphology in response to CNS insults. Expression of coronin-1a in the different morphological subtypes of microglia, which are thought to represent different states of activation, suggest that coronin-1a is constitutively expressed in microglia. This notion fits with the biological function of coronin-1a, which has been shown to be constitutively associated with the actin cytoskeleton in macrophages and lymphocytes (Gatfield et al. 2005). The present results confirm the membrane and subplasma membrane cytoplasmic localization of coronin-1a in microglia similar to those reported previously for macrophages and lymphocytes, which is consistent with evidence that coronin-1a binds to cell membrane and membrane actin cytoskeleton through two distinct binding domains (Gatfield et al. 2005).
IBA-1 is an EF-hand protein expressed specifically by cells of the monocytic lineage, including microglia (Imai et al. 1996). The present study demonstrates that coronin-1a has a very similar immunoreactivity to IBA-1, being expressed in both ramified and ameboid microglia in formaldehyde-fixed, paraffin-embedded tissues. Interestingly, IBA-1 is also an evolutionary conserved protein (Deininger et al. 2002) that has actin-binding properties. IBA-1 has been shown to colocalize with F-actin and to be essential for membrane ruffling in response to macrophage colony-stimulating factor and phagocytosis in cultured cells (Ohsawa et al. 2000). Previous studies indicate that IBA-1 is involved in actin cross-linking and bundling (Sasaki et al. 2001), which intriguingly are properties common to proteins in the coronin family (Rybakin and Clemen 2005). More recent studies have also implicated IBA-1 in regulating the rearrangement of the actin cytoskeleton (Ohsawa et al. 2004), which is similar to the biological function attributed to coronin-1a (Oku et al. 2005).
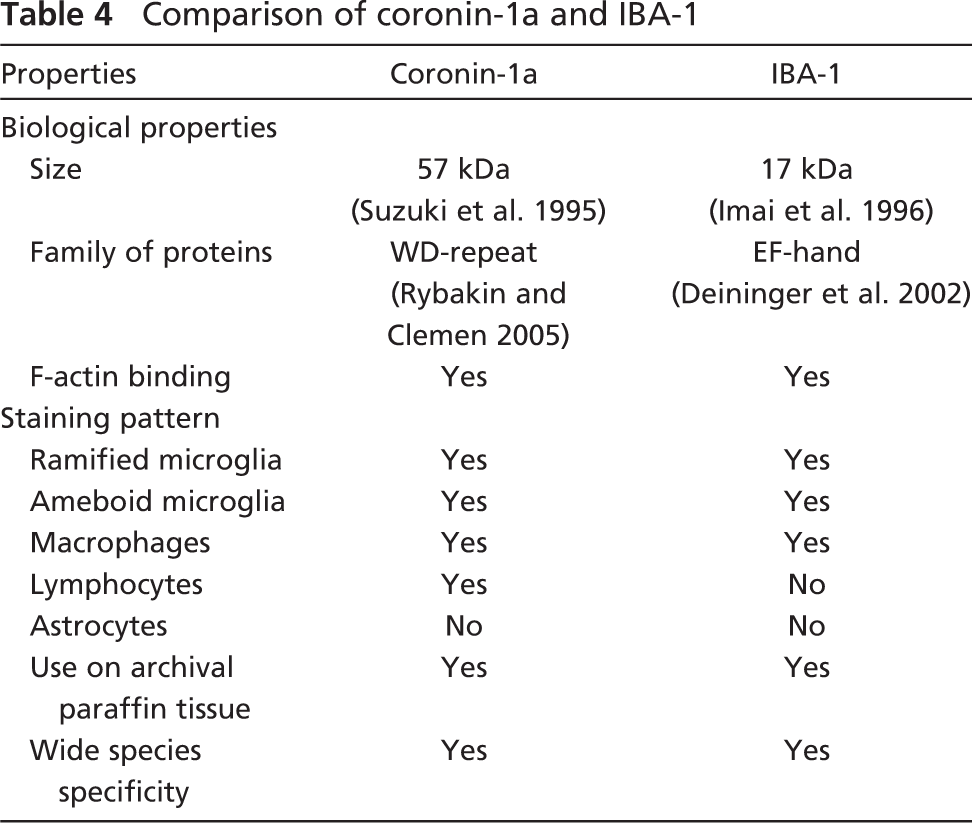
Table 4 summarizes the biological similarities and differences between coronin-1a and IBA-1. The fact that coronin-1a is expressed in microglia and macrophages as well as lymphocytes (Okumura et al. 1998; Ferrari et al. 1999), whereas IBA-1 expression is restricted to microglia and macrophages (Imai et al. 1996), may provide insight into possible differences in their biological functions as actin-binding proteins. Microglia rely on their ability to react to changes in their microenvironment by changing morphology, becoming motile, and gaining phagocytic capacity. These changes all require significant rearrangements of the actin cytoskeleton, highlighting the importance of coronin-1a and IBA-1 in microglia function.
Comparison of coronin-1a and IBA-1
IHC for actin has been reported to be a marker for microglia (Plantier et al. 1998); however, actin IHC is not specific to microglia. From a practical point of view, antibodies to both IBA-1 and coronin-1a provide more specific means of detecting microglia. It is important to acknowledge that coronin-1a should be considered specific for microglia only in disorders where the presence of lymphocytes in the parenchyma is minimal (Perry 1998). In the setting of neuroinflam-matory diseases, coronin-1a would be better described as a marker for generalized inflammation because it is expressed in lymphocytes and macrophages (Okumura et al. 1998; Ferrari et al. 1999). The same properties are characteristic of another microglial marker, leukocyte common antigen (CD45, see Dickson and Lee 1996).
Tissue sections stored for long periods of time can show masking or degradation of microglial antigens, especially for membrane-associated antigens, making IHC detection difficult. In contrast, the antigenicity of coronin-1a and IBA-1 were preserved in tissue sections stored for even long periods of time. Unlike more commonly used microglial markers (e.g., HLA-DR and CD45) that are often susceptible to postmortem degradation or epitope masking by tissue processing, coronin-1a and IBA-1 are associated with the membrane cytoskeleton, which may be the reason for their greater stability in archival tissue samples. For humans there are a number of well-characterized microglial markers suitable for IHC, although some of these are not effective markers in routinely processed tissue or in archival sections (Dickson and Lee 1996). Similarly, most of the widely used microglial markers for mouse studies are ineffective in formaldehyde-fixed, paraffin-embedded tissue and therefore require frozen or free-floating sections for IHC (for example, see Morgan et al. 2005). Previous reports of IHC using IBA-1 on non-human, paraffin-embedded tissues are few (Ohyu et al. 1999). The present results demonstrate that not only IBA-1, but also coronin-1a, are useful reagents for detecting microglia in paraffin-embedded tissues in a range of species. The abundance and stability of cell type-specific expression of the various members of the coronin family suggest that other coronin family members may be useful markers of other cell types in future studies.
The use of transgenic mouse models in research on neurodegenerative disorders has increased substantially, and there is interest in the role of microglia in these models. In Alzheimer's disease, extracellular deposits of β-amyloid protein and neurofibrillary tangles composed of intraneuronal aggregates of tau protein are both associated with reactive microglia (Dickson 2001). The results of the present study demonstrate that antibodies to actin-binding proteins coronin-1a and IBA-1 are effective means of demonstrating microglia associated with β-amyloid plaques and neurodegeneration associated with tau pathology in a range of transgenic models, independent of background strain. The discovery of coronin-1a as a microglial marker and confirmation that IBA-1 is also a useful microglial marker provide researchers with several tools to study the role of microglia in the pathogenesis of Alzheimer's disease and other neurodegenerative diseases, especially when using formaldehyde-fixed, paraffin-embedded tissues.
It is of interest that coronin-1a was one of the molecules, along with Aβ, tau, and GFAP, that unbiased proteomic studies have shown to be enriched in Alzheimer β-amyloid plaques compared with non-plaque-surrounding tissue (Liao et al. 2004). The results of the present study suggest that coronin-1a is enriched in β-amyloid plaques because of its association with microglia.
Footnotes
Acknowledgements
This study was supported by National Institutes of Health Grants R01-AG22595, R01-AG20216, P50-AG16574, P50-AG25711, P50-NS40256, P01-AG17216, and P01-AG03949 and by private funding from EnCor Biotechnology, Inc. G.S. holds equity in EnCor Biotechnology, Inc., a company commercializing certain antibodies used in this study, and may benefit by receiving royalties or equity growth.
Z.A. is a Doctoral Candidate for the MRC Centre for Neurodegeneration Research, King's College London, Institute of Psychiatry, Department of Neuroscience, De Crespigny Park, London, SE5 8AF, UK. The authors acknowledge the valuable histological support of Monica Casey-Castanedes, Virginia Phillips, and Linda Rousseau. The authors appreciate donation of HL60 cells from Dr. Orion Weiner, Harvard Medical School, Boston, MA.
