Abstract
Microglia, which are the resident macrophages and the first line of defense in the brain, can be activated within hours and migrate toward the injury sites after acute and chronic ischemic brain injury. However, a few studies have reported the interaction between microglia activation and necroptosis signaling following ischemic damage to the brain. In this study, chronic ischemic brain injury was induced by bilateral carotid artery stenosis (BCAS) and mice were sacrificed at 30 days after surgery. Ionized calcium-binding adaptor molecule 1 (IBA1) and glial fibrillary acidic protein (GFAP) immunostaining were performed to determine glial cell activation and inflammatory response. Tumor necrosis factor-α (TNF-α), interferon-γ (INF-γ), and interleukin-1β (IL-1β) proteins from the brains were examined to confirm inflammatory cytokines after BCAS. RIP1 and RIP3 proteins were detected to determine necroptosis signaling by Western blot. The data suggested that inflammatory responses, microglia activation, and necroptosis signaling are features of brain tissue pathology following BCAS-induced chronic ischemic brain injury.
Introduction
Microglia account for 5%–20% of the total glial population 1 and are quickly activated in the event of central nervous system (CNS) injuries and disorders, including inflammation, ischemic stroke, and so on, as the first line of immune response.1–5 In the past century, accumulated evidence demonstrated that this cell subpopulation has an important role in CNS homeostasis and are involved widely in CNS pathologies. However, it is still a subject of controversy whether activation of this microglial population following CNS insult is helpful or destructive. On one hand, there are some supporting evidences for the beneficial role of this population. The study demonstrated that ischemic brain injuries were exacerbated by the selective depletion of microglial cells, 6 whereas brain injuries were ameliorated by transplanting microglia into the ischemic brain.7–11 Microglial activation is considered as a benefit factor because they could remove cell debris and restore tissue integrity in the injured brain.12–14 On the other hand, there are also increasing evidence supporting the opposite view that excessive or inappropriate activation of microglial cells may contribute to secondary expansion of ischemic brain injury and exacerbation of neurological and pathological outcomes. 15 Therefore, microglial activation has dual role in nature, which depends on different cellular microenvironments and stages of pathological changes following brain injury.2,16
Necrotic cell death is a common phenomenon in a wide variety of pathological conditions,17–21 including ischemic stroke.19–21 It is possible that environment resulting from ischemic injury is suitable for necroptosis. 21 Necroptosis, an inflammatory pathological process,22,23 may exert a cellular “backup” mechanism to ensure the clearance of damaged cells under conditions of stress and inhibition of apoptosis, which is consistent with the character of slow kinetics. 21 Microglia, which function as scavenger cells in the brain,1,5,8,12–14 could be activated and produce pro-inflammatory cytokines (interleukin-1β (IL-1β), interleukin-6 (IL-6), interferon-γ (INF-γ), and tumor necrosis factor-α (TNF-α)) after ischemic brain injury, which lead to inflammatory response. The changes in neuropathology, including microglia reaction, which result from the arrest of blood flow circulation in the brain have been extensively studied. However, most studies on microglial activation and neuropathological changes in ischemic stroke focused on the acute brain injury, such as temporary or permanent ligation of the middle cerebral artery and middle cerebral artery occlusion (MCAO) surgery. There are only limited studies focusing on the chronic ischemic stroke between microglia activation and neuropathological alterations.
Herein, in order to determine the occurrence of both microglial reaction and necroptosis signaling involved in chronic ischemic stroke, we used the bilateral carotid artery stenosis (BCAS) model in mice. The occurrence of microglial reaction and necroptosis was studied 30 days after ischemic surgery.
Materials and methods
Animals and experimental design
Male C57BL/6 mice were purchased from the Shanghai Laboratory Animal Center (SLAC), Chinese Academy of Sciences. Animals used in the study were allowed freely to acquire food and water. Furthermore, all animals were kept in accredited facility of 12 h reverse light cycle. Mice 12 weeks old were used in the study. Mice were randomly divided into three groups: non-operated group, sham-operated group, and BCAS group (n = 15).
Surgical operation on the mice
The surgery for mouse model BCAS was performed using microcoils with the features of inner diameter of 0.18 mm, wire diameter of 0.08 mm, pitch of 0.50 mm, and total length of 2.5 mm (Sawane Spring Co., Wuxi city, China), according to previous publications.1,24–26 Briefly, all the mice were anesthetized with isoflurane (1.5%). After that, the mouse was fixed in the supine position. In order to fix microcoils, we exposed bilateral common carotid arteries following a midline incision in the cervical part. The body temperature was kept at 37.0 ± 0.5°C by a heating pad during the whole operation.
Determining the values of cerebral blood flow
Cerebral blood flow (CBF) was determined through the skull using a Laser Doppler Flowmetry (Omega Wave, Tokyo, Japan) when mouse was anesthetized with isoflurane (1.5%) and placed in the prone position before and after the surgery. The CBF values were presented as a percentage of the baseline value.
Fixation and processing of samples
All the mice were sacrificed under isoflurane (5%) anesthesia. The brains were removed and fixed in 4% buffered paraformaldehyde (PFA) at 4°C overnight, and then brains were dehydrated in 30% sucrose solution and were cut into 25-µm-thick frozen sections. These sections were collected and kept at −20°C.
Immunofluorescence histochemistry
Brain sections were rinsed with phosphate buffered saline (PBS) and blocked with 5% milk in PBS for 1 h and probed with primary antibodies in 3% bovine serum albumin (BSA) solution overnight at 4°C. The following day, secondary antibodies with fluorescence (1:500; Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA) were added and maintained for 1 h at room temperature. Finally, slides were mounted with ProLong Gold antifade reagent (Life Technologies, California, Carlsbad, America) and images were taken using a fluorescence microscope (Nikon ECLIPSE Ti-S; Nikon, Tokyo, Japan). The primary antibodies used were as follows: goat polyclonal anti–glial fibrillary acidic protein (GFAP) and ionized calcium-binding adaptor molecule 1 (IBA1) (1:300; Santa Cruz Biotechnology, Inc.).
Western blot analysis
Total protein extracts and Western blotting analysis were performed as previously described 27 following an abscission of hippocampus. Tissues were lysed in a homogenizer containing radioimmunoprecipitation assay (RIPA) buffer kept at 0°C–4°C (Beyotime Biotechnology, Shanghai City, China). Total 40 µg protein sample was added to the sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS/PAGE) gel well and separated by 10%–15% SDS/PAGE and transferred onto polyvinylidene difluoride (PVDF) membranes (Bio-Rad Laboratories, Hercules, CA, USA). And then, membranes were blocked with 3% BSA and incubated with primary and secondary antibodies, respectively. Protein levels of RIP1, RIP3, mixed lineage kinase domain-like (MLKL), and β-actin (Santa Cruz Biotechnology, Inc.) were assayed. To quantify the band intensity, we used ImageJ software and normalized with respect to the levels of β-actin as an internal control.
Enzyme-linked immunosorbent assay analysis
Concentrations of TNF-α, INF-γ, and IL-1β from the hippocampal homogenates were quantified using enzyme-linked immunosorbent assay analysis (ELISA) kits (Mouse TNF-α ELISA Kit, Mouse INF-γ ELISA Kit, Mouse IL-1β ELISA Kit, and Mouse IL-33 ELISA Kit; RayBiotech, Norcross, GA, USA) according to the manufacturer’s protocol. Result was expressed in picogram per milligram protein. Data from each sample were normalized to the protein concentration according to the standard curve.
Statistical analysis
Results were expressed as mean ± standard deviation (SD). A value of P < 0.05 was considered statistically significant. All statistical comparisons were conducted using the Statistical Package for the Social Sciences (SPSS 20.0; SPSS Inc., Chicago, IL, USA). All graphs were made in GraphPad Prism (version 5; GraphPad Software, San Diego, CA, USA). For comparison between the two groups or multiple groups, we determined the statistical significance using a Student’s t-test or one-way analysis of variance (ANOVA) followed by a Student–Newman–Keuls test.
Results
Experimental protocol
All the animals were randomly divided into three groups (BCAS group, sham-operation group, and non-operation group; n = 15). Every surgical operation for BCAS was completed within 15 min. Following the surgery, all the animals recovered within several hours except for some mental state, such as listlessness and loss of appetite. All of them showed no apparent motor weakness. We observed the physiological index for 30 days, for example, body weight and temperature, but there were no significant differences (data were not shown).
Alterations of CBF in the experimental mice
In Figure 1, data represented the mean values of CBF in mice after surgical operation. Both in the sham and non-operation groups, the mean values of CBF have no significant changes following operation between any points of time. In contrast, the CBF values reduced markedly before and after the surgery in the BCAS group, and we observed a sharp reduction in CBF values to 65 ± 7% within 1 h after surgery, and then, gradual recovery of CBF was observed. At 30 days, the CBF values in the BCAS group were still lower than in the other two groups. Intergroup differences in the CBF values were also detected, as shown in Figure 1; there were different alterations at the different points of time among the three groups.

The values of CBF in the experimental mice. There was a sharp reduction in CBF in the BCAS group after 1 h of surgery and then recovered gradually. Although there was a lower CBF in the BCAS operation group than in the other two groups, there was no statistical difference.
Microglia and astrocyte reaction in BCAS mice (IBA1/GFAP)
Strong microglial activation was observed after 30 days of BCAS surgery, as shown in Figure 2. Using antibodies incubated against IBA1 (Figure 2(a)–(c)) and GFAP (Figure 2(d)–(f)) antigens, strong immunoreactivity of microglia and astrocytes was seen at 30 days after BCAS surgery in the brain in contrast with the two control groups (Figure 2(g) and (h)).
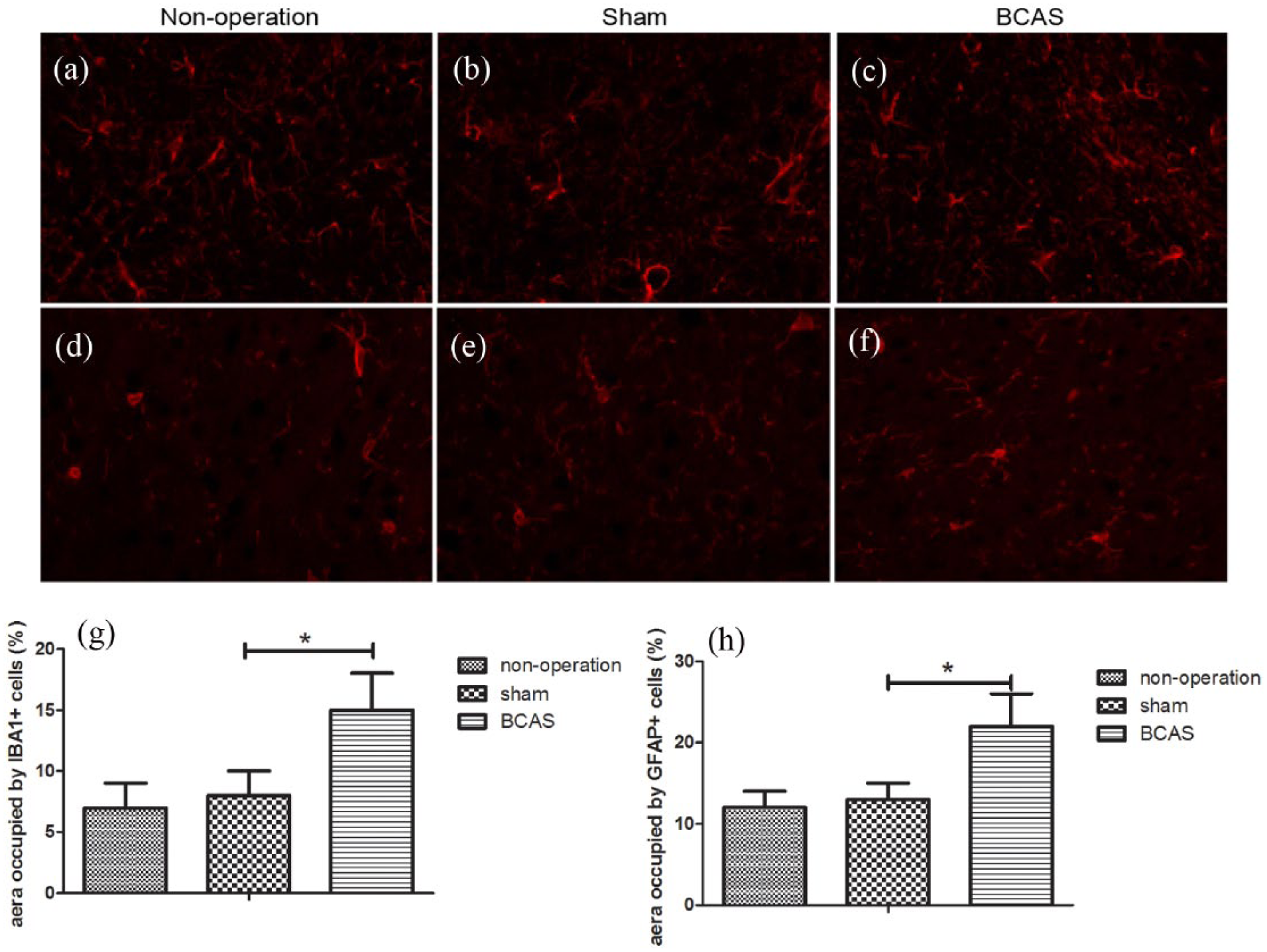
Microglial cells and astrocytes in the hippocampus of the mice. (a–c) Different degrees of astrocyte reaction were seen in three groups (400×). (d–f) Different degrees of microglia reaction were seen in three groups (400×). (g–h) Quantitative statistical data of astrocytes and microglial cells, respectively (*P < 0.05).
The alterations of RIP1, RIP3, and MLKL proteins from hippocampus
To understand whether necroptosis signaling was involved in the chronic ischemic stroke induced by BCAS surgery, we examined the expression of RIP1, RIP3, and MLKL proteins using Western blot. The kinase activities of RIP1 and RIP3 have been proposed to be required for necroptosis; 23 our results indicated that the levels of RIP1 and RIP3 proteins in the BCAS mice were higher than in the controls, as shown in Figure 3. As the substrate of RIP3, MLKL is also involved in necroptosis;23,28 a higher level of MLKL protein was seen in the BCAS mice, as shown in Figure 3.
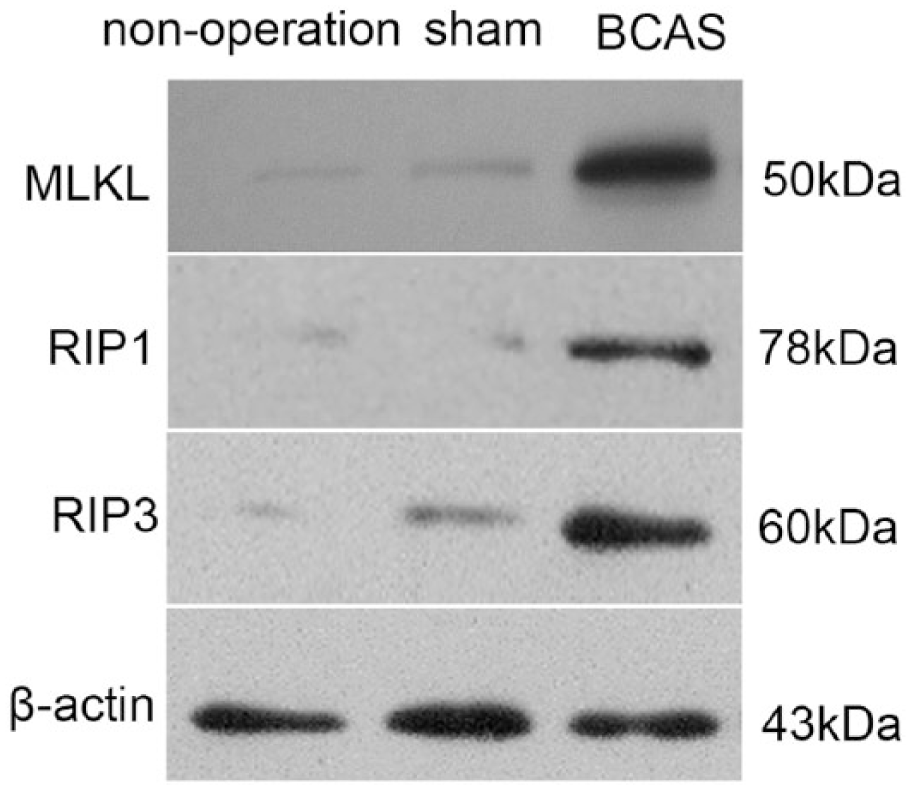
The changes of RIP1, RIP3, and MLKL proteins from hippocampus at 30 days after surgery. The left and right parts of the figure represent Western blot of the proteins and statistical data, respectively (*P < 0.05).
Inflammatory cytokines were increased after BCAS surgery
We used ELISA to evaluate alterations in the levels of some inflammatory cytokine expression. There was a stronger upregulation of TNF-α, INF-γ, and IL-1β protein expression at 30 days following BCAS surgery (Figure 4(a)–(c)).

Inflammatory-related cytokines were increased in BCAS mice. The results showed the protein concentrations of (a) TNF-α, (b) INF-γ, and (c) IL-1β in brain tissues at 30 days after BCAS surgery measured by ELISA (*P < 0.05).
Discussion
Herein, we investigated the pathological changes after chronic ischemic injury induced by BCAS surgery in mice. BCAS mice showed strong inflammatory response accompanied by activation of glial cells and involvement of necroptosis signaling following chronic ischemic injury. We observed that the levels of inflammatory cytokines expression, such as TNF-α, INF-γ, and IL-1β, were markedly increased in BCAS mice.
Microglia and astrocytes are considered as the primary immune effector cells with a function in immunological surveillance and contribute to the production of various cytokines in the CNS29–31 and are important for the homeostasis in brain, through balancing the inflammatory cytokines30,32,33 and exerting the interceptors in brain modulating the change of pH, CO2, or sodium ion, 34 respectively. The increased intensity of microglia reaction was also seen in this study. Microglial cells were specialized phagocytes in the brain and could devour neurons within several hours. 24 IBA1 is a marker of both microglia and macrophages; 25 according to the morphological difference and treatment of brain tissue, we can believe that the BIA1-positive cells are microglial cells. Furthermore, microglial cells are known for secreting cytokines in the CNS and protecting them against numerous pathological conditions, such as infectious diseases, neuroinflammation, and ischemia. 1 However, over-expressed or sustained stimulation of microglial cells can lead to a large number of cytokines, such as IL-1β and TNF-α,26,30 which may cause the deterioration of diseases, including severe neuroinflammation and subsequent cognitive dysfunction.
The type and duration of stimulus and its preceding, concomitant, and subsequent stimuli determine the immune response of microglia activation depending on producing pro-inflammatory and anti-inflammatory cytokines that control shift between the switching off and on of the immune response. In our study, we established chronic ischemic stroke induced by BCAS surgery in mice and sustained stimulation for 30 days, leading to a marked decrease in CBF. Furthermore, higher levels of inflammatory cytokines, such as TNF-α, INF-γ, and IL-1β, were observed in BCAS mice, which indicated the involvement of strong inflammatory response in BCAS mice.
Necrotic cell death is a common phenomenon in a wide variety of pathological conditions,17–21 including ischemic stroke.19–21 Necroptosis, a programmed necrotic cell death, can be induced by TNF35–39 and INF-γ, 19 and it is reported that RIP1, RIP3, and MLKL proteins are essential for the activation of necroptosis.40,41 So our data suggest that necroptosis is involved in the process of chronic ischemic injury. Tissue injury has a close connection with inflammation, and these two factors are reciprocal causation. 42 However, the cases of tissue injury previously reported were almost induced by strong and acute external factors, such as infectious, toxic, and traumatic injuries, which caused inflammation. Our results show that the changes in inflammatory cytokines are consistent with the alteration of necroptosis signaling and that of microglial cells following BCAS surgery, which suggests the tight link between the activation of microglial cells and necroptosis signaling in chronic ischemic brain injury.
Although our study demonstrated an important relationship between microglia activation and necroptosis signaling in chronic ischemic brain injury, there are some questions to be further researched, for example, elucidating how and when the activation of microglia affects necroptosis will aid our understanding of chronic ischemic brain injury, which should be important for clinical implications and social value.
This study demonstrated that microglia and astroglia cells were activated following BCAS mouse model and that the changes in necroptosis signaling were consistent with the activation of microglia.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the Natural Science Foundation of China (NSFC, nos. 81472150), the Key Projects of Shanghai Science and Technology on Biomedicine (nos. 13411951000), and the Key Construction Projects of Shanghai Health and Family Planning on Weak Discipline (nos. 2015ZB0401).
