Abstract
There is a paucity of information of tight junction (TJ) proteins in gallbladder epithelium, and disturbances in the structure of these proteins may play a role in the pathogenesis of acute acalculous cholecystitis (AAC) and acute calculous cholecystitis (ACC). Using immunohistochemistry, we investigated the expression of TJ proteins claudin-1, −2, −3, and −4, occludin, zonula occludens (ZO-1), and E-cadherin in 9 normal gallbladders, 30 gallbladders with AAC, and 21 gallbladders with ACC. The number of positive epithelial and endothelial cells and the intensity of the immunoreaction were determined. Membrane-bound and cytoplasmic immunoreactivities were separately assessed. We found that TJ proteins were uniformly expressed in normal gallbladder epithelium, with the exception of claudin-2, which was present in less than half of the cells. In AAC, expression of cytoplasmic occludin and claudin-1 were decreased, as compared with normal gallbladder. In ACC, expression of claudin-2 was increased, and expression of claudin-1, −3, and −4, occludin, and ZO-1 were decreased, as compared with normal gallbladder or AAC. We conclude that there are significant differences in expression of TJ proteins in AAC and ACC, supporting the idea that AAC represents a manifestation of systemic inflammatory disease, whereas ACC is a local inflammatory and often infectious disease.
Keywords
Tight junctions (TJ) are important apical cell-cell contacts in epithelial and endothelial cell sheets and have at least two functions: the barrier function and the fence function (Tsukita et al. 2001). The barrier function regulates the passage of ions, water, various macromolecules, and inflammatory cells through the paracellular spaces and is thus relevant to permeability disorders, leading to, for instance, the development of tissue edema and diarrhea. The fence function maintains cell polarity and may be impaired in cancer cell invasion. The major integral membrane proteins forming continuous TJ strands are occludin, claudins, and junctional adhesion molecules (JAMs) (Furuse et al. 1993, 1998; Martin-Padura et al. 1998; Morita et al. 1999). The principal cytoplasmic structural proteins associated with TJs are ZO-1 (zonula occludens), ZO-2, and ZO-3, which all bind to the cytoplasmic tails of occludin and claudins (Balda and Matter 2000). These proteins together are thought to maintain the integrity of TJs, and their expression and localization are presumed to have an important role in the regulation of paracellular permeability. E-cadherin is a transmembrane protein present in adherens junctions where it functions as an adhesion molecule and also interacts with the actin cytoskeleton to stabilize TJs (Tunggal et al. 2005).
Expression of TJ proteins has mainly been studied in laboratory animals and in cell cultures (Furuse et al. 1993, 1998; Enck et al. 2001; Rahner et al. 2001; Kiuchi-Saishin et al. 2002; Han et al. 2004a, b, c). In patients, TJ proteins have been studied in inflammatory bowel disease (IBD) and in some neoplasms (Kucharzik et al. 2001; Prasad et al. 2005; Soini 2005). In critically ill patients, disturbances in the formation and function of TJs can be important factors leading to permeability disorders and end organ dysfunctions (Fink and Delude 2005). To our knowledge, there are no studies published concerning TJ proteins in critically ill patients. Expression of TJ proteins in normal or inflamed gallbladder has also not been studied.
We hypothesized that the expression of TJ proteins in gallbladder of critically ill patients with AAC is different from expression in normal gallbladder and from expression in ACC. Therefore, we analyzed by immunohistochemistry the expression of claudins 1–4, occludin, E-cadherin, and ZO-1 in gallbladders removed from critically ill patients with AAC and in gallbladders removed from outpatients with ACC, and we compared them with the expression in normal gallbladder.
Materials and Methods
Patients
This study was approved by the Ethics Committee of Oulu University Hospital. Our setting is a 20-bed medical-surgical intensive care unit (ICU) in a university hospital. In the years 2000 and 2001, 39/3984 ICU patients underwent cholecystectomy because of AAC during their ICU stay. A detailed report of the clinical features of these 39 patients has been previously published (Laurila et al. 2004). Typical histopathological features in the gallbladder wall in AAC are bile infiltration, lymphatic dilatation, and leukocyte margination of blood vessels, whereas typical features in ACC are epithelial degeneration and defects and widespread occurrence of inflammatory cells (Laurila et al. 2005). In the present study, the AAC group consisted of 30 randomly chosen gallbladders of the 39 ICU patients operated because of AAC during the years 2000–2001. The mean (SD) age of these patients was 60 (12.5) years, 19/30 were men, and the mean (SD) APACHE II (Acute Physiology And Chronic Health Evaluation) and SAPS II (Simplified Acute Physiology Score) scores on admission were 23.6 (6.1) and 47.2 (12.3). Sepsis (10/30), cardiovascular surgery (8/30), and pneumonia (5/30) were the most common admission diagnoses. The median (25th, 75th percentile) length of ICU stay before cholecystectomy was 7.5 (2.8, 15.3) days. Two of the 30 bile samples (6.7%) taken during cholecystectomy were positive for bacterial growth.
The ACC group in the present study consisted of 21 gallbladders of consecutive patients operated at our hospital within the study period. An operative finding of inflamed gallbladder with gallstones was used as the inclusion criterion. All these patients were admitted to the hospital because of ACC and were treated on the normal hospital ward. None needed intensive care. The median (25th, 75th percentile) time from start of symptoms to the operation was 3 days (2, 4.5) and median (25th, 75th percentile) time from admission to hospital to the operation was 2 (1, 2) days. The mean (SD) age of these patients was 57.9 (10.3) years, and 6/21 were men. Of 16 bile cultures, nine were positive for bacterial growth (56%).
Samples of normal gallbladders were removed during pancreatic tumor surgery. These patients had a local disease remote from the gallbladder and had no history of biliary obstruction. No signs of acute inflammation were seen in the basic histological examination of these gallbladder samples. The mean (SD) age of the patients in this group was 59.1 (17.8) years, and 3/9 were men.
Immunohistochemistry
Gallbladder samples were fixed in neutral phosphate-buffered 10% formalin and embedded in paraffin. Fixation time was ~24 hr in most cases (range: 24 — 66 hr). Sections were stained in hematoxylin and eosin for conventional histopathological diagnosis (Laurila et al. 2005). Epithelial expression of junctional proteins was investigated as follows: claudin-1, −2, −3, and −4, occludin, and ZO-1 as elements of TJs, and E-cadherin as an element of adherens junctions. Primary antibodies were purchased from Zymed Laboratories (South San Francisco, CA). Antibodies were used according to the manufacturer's recommendations and consisted of polyclonal rabbit anti-claudin-1, monoclonal mouse anti-claudin-2, polyclonal rabbit anti-claudin-3, monoclonal mouse anti-claudin-4, polyclonal rabbit anti-occludin, polyclonal rabbit anti-ZO-1, and monoclonal mouse anti-E-cadherin. Dilution used was 1:100 for all antibodies and the dilution solution was purchased from Dako (S2022; Dako, Carpinteria, CA). Sections were pretreated with Tris/EDTA, except in the case of occludin, for which the sections were pretreated with pronase. Sections for ZO-1 immunohistochemistry were heated in a microwave oven for 15 min before the application of the primary antibodies, after which Envision kit (K5007; Dako) was used. This kit detects both bound mouse and rabbit primary antibodies. The color was developed by diaminobenzidine. Negative control stainings were carried out by substituting non-immune rabbit or mouse serum and PBS for the primary antibodies.
Results are presented as proportions (percentage) of epithelial cells expressing the studied antigens (intensity >0). Membrane-bound and cytoplasmic immunoreactivities were separately assessed. Staining intensity was categorized into four classes for analysis (0 = absent, 1 = weak, 2 = moderate, 3 = strong). Immunoreactivity in the vascular endothelial cells in the gallbladder wall was analyzed as well. All assessments were made blindly by two investigators (JL and either TK or YS).
Statistical Analysis
Statistical analyses were performed using SPSS 12.0.1 for Windows (SPSS Inc.; Chicago, IL). Data are expressed as percentage, mean, and standard deviation (SD) in the case of normally distributed data and as median and the 25th and 75th percentiles in the case of non-normally distributed data. Normality of distribution was assessed with the Kolmogorov-Smirnov test. Categorical data were analyzed using Fisher's exact test. Kruskall-Wallis test was used to describe differences in the percentage of positively stained epithelial cells between the AAC, ACC, and control groups. Mann-Whitney U-test was applied to analyze differences between groups (AAC vs. control, ACC vs. control, and AAC vs. ACC). Differences were considered significant at
Results
We found both membrane-bound and cytoplasmic immunoreactivity for all studied TJ proteins in our gallbladder samples. The percentages of epithelial cells with positive staining in the different groups are presented in Figure 1 and Figure 2, and the distribution of staining intensity is presented in Table 1. In endothelial cells, marked immunoreactivity was detectable for ZO-1 and occludin, and there were no significant differences between groups (data not shown).
Claudins
Membrane-bound staining for claudin-1, −3, and −4 was present in practically all epithelial cells in AAC and controls, but in ACC it was not so prevalent (Figure 1, top). Cytoplasmic staining for claudin-1, −3, and −4 was clearly less prevalent than membrane-bound staining in all groups (Figure 1, bottom). Expression of claudin-2 differed from the other claudins; in the control and AAC groups, only about half of the cells showed membrane-bound or cytoplasmic staining, but a significantly greater part of cells stained positively in ACC (Figure 1 and Figure 3), and also the intensity of staining was stronger in ACC than in AAC or controls (Table 1).
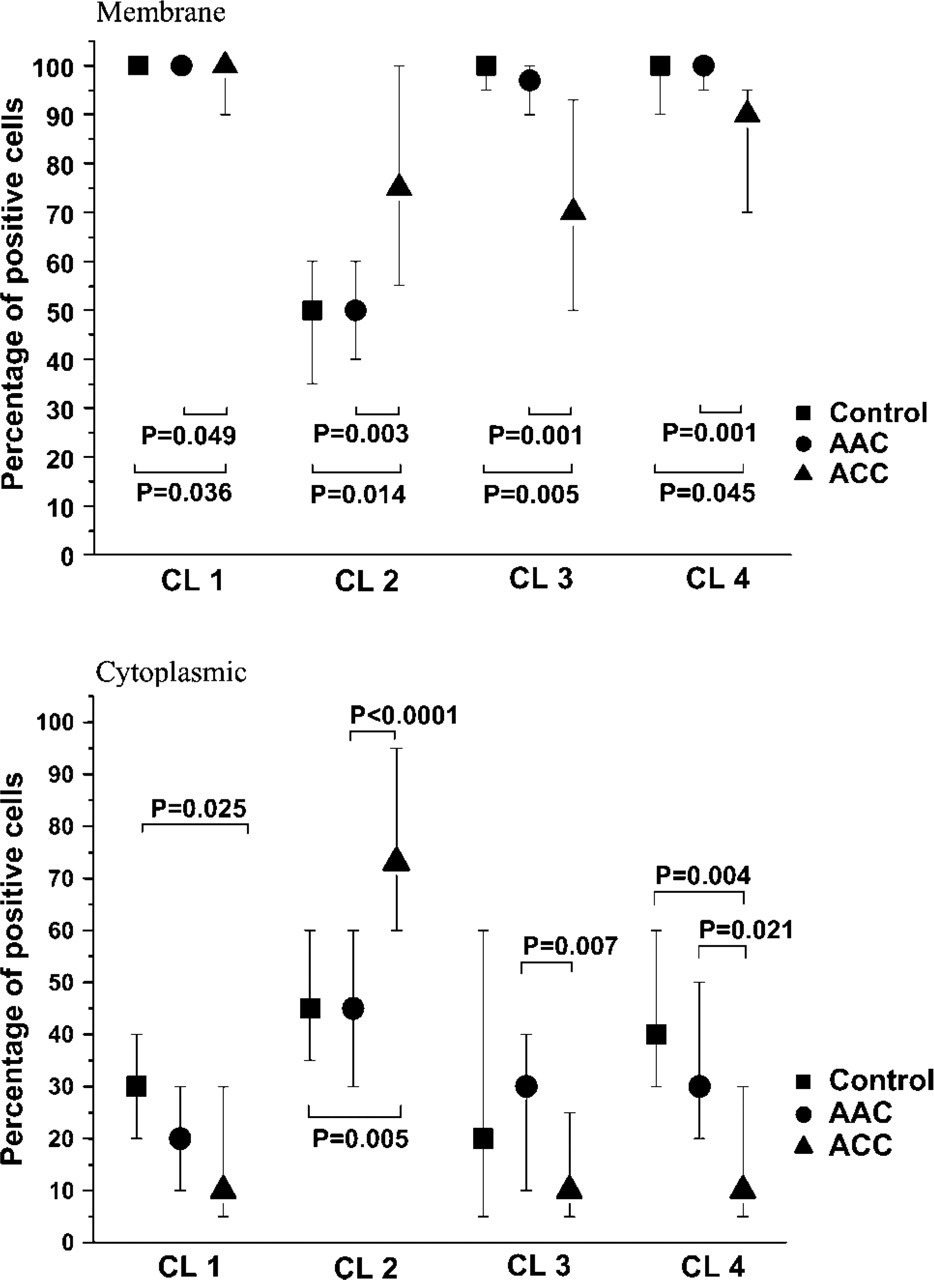
Median (25th, 75th percentiles) percentages of cells staining positively for claudins. CL 1, claudin-1; CL 2, claudin-2; CL 3, claudin-3; CL 4, claudin-4.
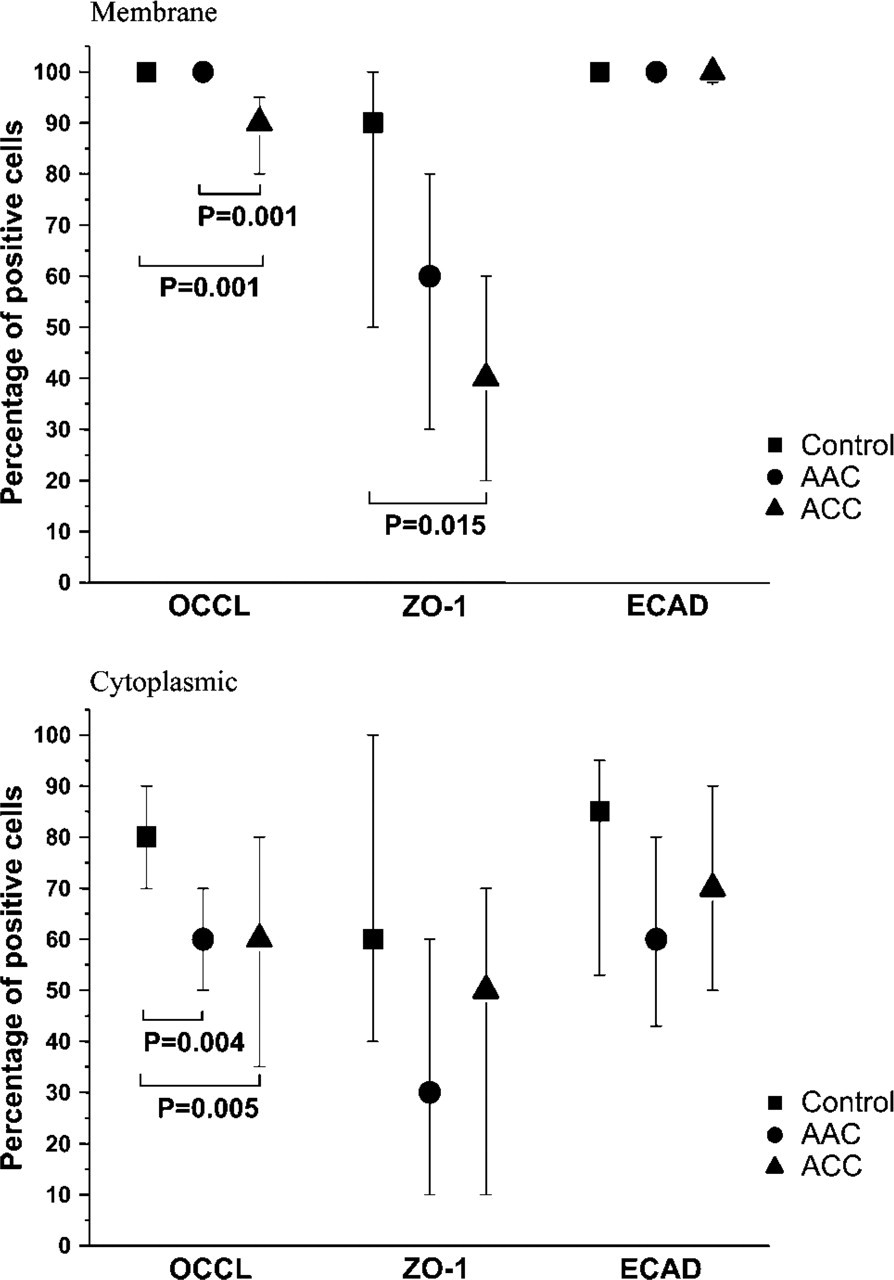
Median (25th, 75th percentiles) percentages of cells staining positively for occludin, ZO-1, and E-cadherin. OCCL, occludin; ECAD, E-cadherin.
Occludin
The extent of membrane-bound positivity for occludin was similar in the control and AAC groups but significantly diminished in the ACC group (Figure 2, top). Cytoplasmic positivity for occludin was significantly less prevalent both in AAC and ACC compared with the control group (Figure 2, bottom, and Figure 4).
ZO-1 and E-cadherin
ZO-1 expression was most evident in the apical regions of epithelial cells. The control group had an intensive membrane-bound immunostaining for ZO-1, whereas positively stained cells were less abundant in ACC (Figure 2, top, and Figure 4). Cytoplasmic ZO-1 positivity was present in half of the cells in all groups, and there were no significant differences between groups (Figure 2, bottom). Staining for E-cadherin was widespread both as membrane-bound and as cytoplasmic, and there were no remarkable differences between groups (Figure 2 and Figure 4).
Intensity of the staining of different TJ proteins a
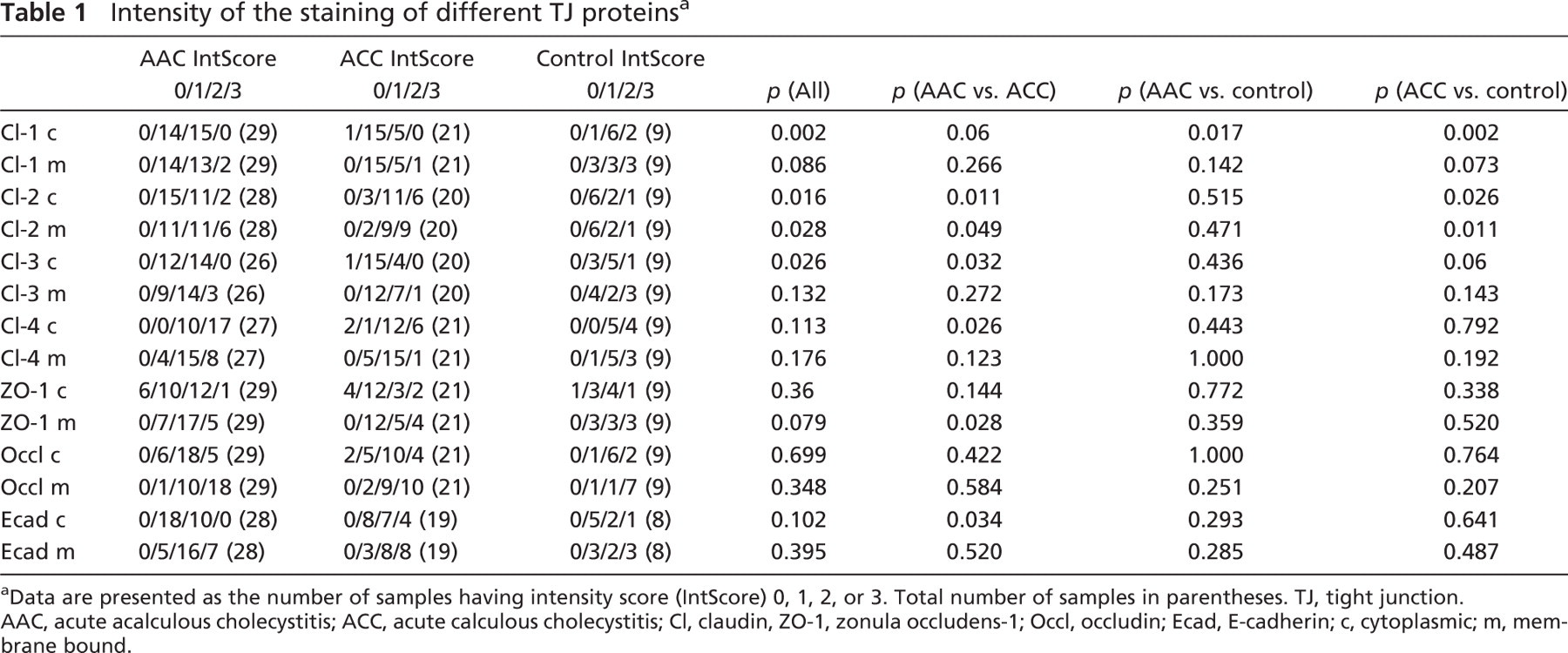
Data are presented as the number of samples having intensity score (IntScore) 0, 1, 2, or 3. Total number of samples in parentheses. TJ, tight junction.
AAC, acute acalculous cholecystitis; ACC, acute calculous cholecystitis; Cl, claudin, ZO-1, zonula occludens-1; Occl, occludin; Ecad, E-cadherin; c, cytoplasmic; m, membrane bound.
In summary, in the control group, membranous staining for all studied TJ proteins except claudin-2 was wide, whereas cytoplasmic staining was less common. In AAC, expression of membrane-bound claudins was not different from that in control gallbladder samples. However, expression of cytoplasmic occludin (percentage of cells staining positively) and intensity of cytoplasmic claudin-1 staining were decreased compared with control gallbladder samples. In ACC, there was increased expression of both the membrane- and cytoplasmic-bound claudin-2, and there was a decrease in the expression of both membrane- and cytoplasmic-bound claudin-1, claudin-4, and occludin.
Discussion
TJs are important cell-cell contacts regulating permeability across epithelial and endothelial cell sheets. Our study showed that TJ proteins are widely expressed in normal human gallbladder epithelium, with the exception of claudin-2, which was detectable in only about half of the cells. In ACC, both cytoplasmic and membranous expression of claudin-2 were significantly increased, and expression of claudin-1, −3, and −4, occludin, and ZO-1 were significantly decreased compared with normal gallbladders or AAC. In AAC of critically ill patients, expression of cytoplasmic occludin and intensity of cytoplasmic claudin-1 staining were lower than in the normal gallbladder. There were, however, no differences in the membranous expression of TJ proteins between AAC and normal gallbladders. There were no differences in the expression of E-cadherin among AAC, ACC, or normal gallbladders.
In previous studies, claudins were expressed in different epithelial types and also in vascular endothelial cells. However, there is variation in expression of different claudins, depending on the location and site of the epithelia (Enck et al. 2001; Rahner et al. 2001; Kiuchi-Saishin et al. 2002). Furthermore, the presence of different claudin types correlates with the permeability of cellular sheets, and it has been shown that different sets of claudins may associate with each other, and that the composition of these complexes determines the permeability of TJs (Furuse et al. 1999; Turksen and Troy 2004). In renal tubular epithelium, for instance, different claudins are present in specific tubular segments (Rahner et al. 2001; Kiuchi-Saishin et al. 2002). We found that normal gallbladder epithelium showed intensive expression of claudin-1, −3, and −4, whereas claudin-2 was expressed more weakly. The observed wide expression of ZO-1 and E-cadherin in normal gallbladder epithelium is in agreement with the previous observations on gallbladder epithelium (Torres et al. 1996; Choi et al. 2004).
In the present study, the decrease in cytoplasmic expression of occludin and claudin-1 was characteristic for AAC. This might suggest increased degradation or decreased synthesis of these proteins during systemic inflammation. One plausible mechanism for this is the effect of cytokines. Mankertz et al. (2000) found that pro-inflammatory cytokines, tumor necrosis factor α, and interferon γ downregulated the expression of occludin in human intestinal cell culture. Han et al. (2004a, b, c) have shown in endotoxemic mice that endotoxemia caused, along with permeability abnormalities, a marked decrease in the expression of occludin in intestine, liver, and lung. Samonte et al. (2004) studied the expression of TJ proteins in two-hit injury in rats and found that burn injury and
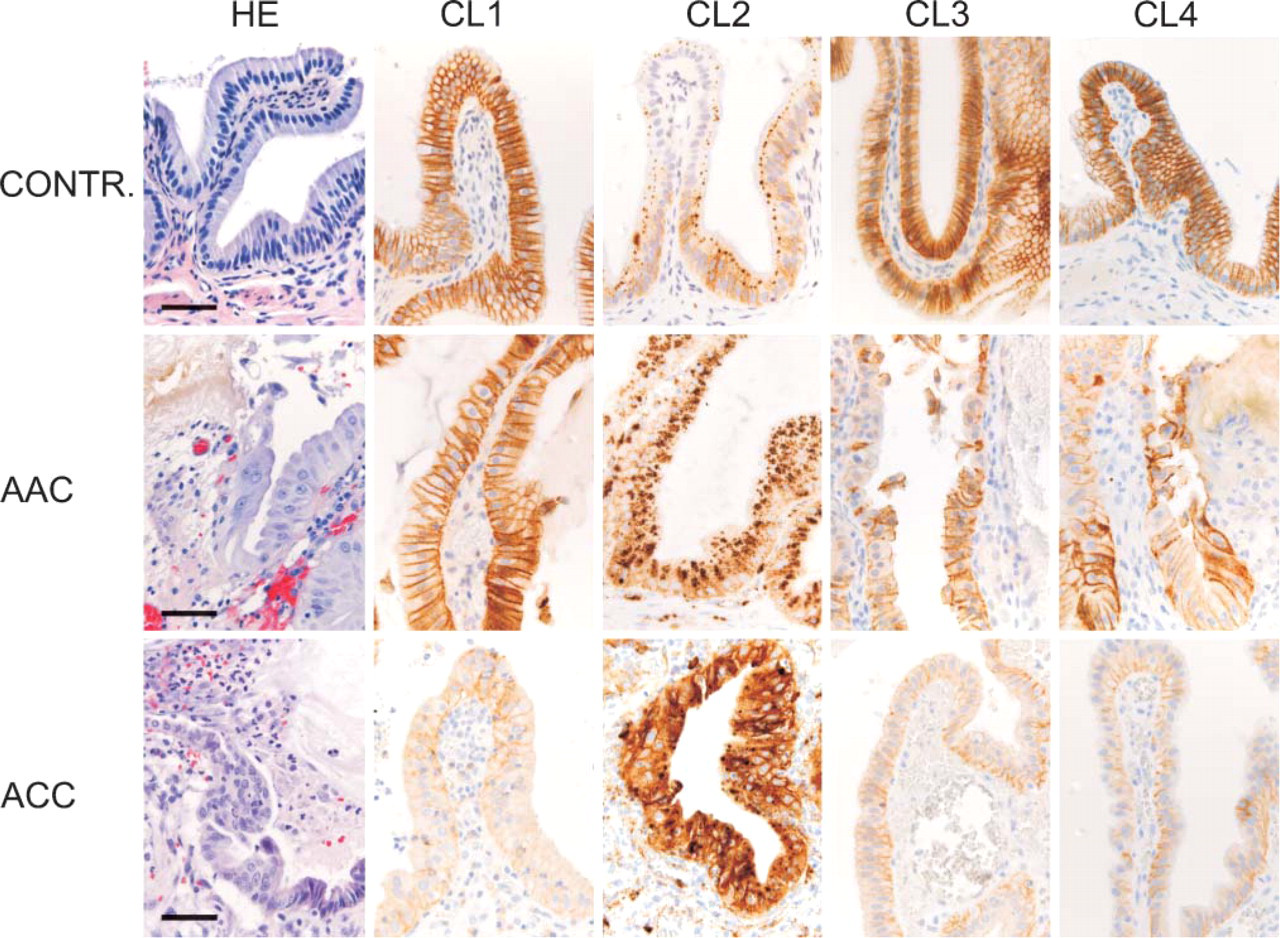
Micrographs of normal gallbladder (CONTR.), acute acalculous cholcystitis (AAC), and acute calculous cholecystitis (ACC). First panel (left) shows hematoxylin/eosin (HE)-stained sections to demonstrate normal histology (CONTR.), characteristic mucosal bile infiltrate in AAC, and neutrophilic inflammatory response in ACC (27). The next panels show immunohistochemical stainings for claudins 1–4 (CL 1, CL 2, CL 3, CL 4). Bar = 0.05 mm.
In ACC, the observed decrease in expression of several TJ proteins and increase in expression of claudin-2 may be related to intense local purulent inflammation and bacterial infection. More than half of the gallbladders in the ACC group had bacterial growth, in contrast to only 7% in the AAC group. Bacteria may be able to invade tissue compartments and degrade or damage TJ structures (Berkes et al. 2003). It has also been shown that claudin-3 and −4 may serve as ligands for some bacterial toxins such as Clostridium perfringens entero-toxin (Berkes et al. 2003). Upregulation of claudin-2 seems to be a typical response in inflammatory reactions involving neutrophilic leucocytes. Prasad et al. (2005) studied the expression of claudin-2, −3, and −4 in colonic epithelial cells and found that claudin-2 was absent or barely detectable in normal human colonic epithelial cells but was strongly expressed along the inflamed crypt epithelium in Crohn's disease and in ulcerative colitis. Claudin-3 and −4, on the other hand, were present throughout normal colonic epithelium but were reduced or redistributed in diseased epithelium. Furthermore, Kucharzik et al. (2001) showed diffuse global downregulation of occludin in IBD, but expression of claudin-1, ZO-1, JAM, and E-cadherin was reduced only in epithelial cells immediately adjacent to transmigrating neutrophils in the diseased part of the colon. We conclude that the observed changes of TJ proteins in ACC are largely similar to those in other acute inflammatory epithelial reactions and probably mediated by proinflammatory factors. In comparison with AAC, the more intensive and partly divergent reaction in ACC is likely to be caused by high concentrations of proinflammatory factors originating locally, which contrast with the presumably lower concentrations of proinflammatory mediators with vascular entry into gallbladder mucosa in AAC. Additionally, hypoxic conditions in AAC might further modify the gallbladder response.
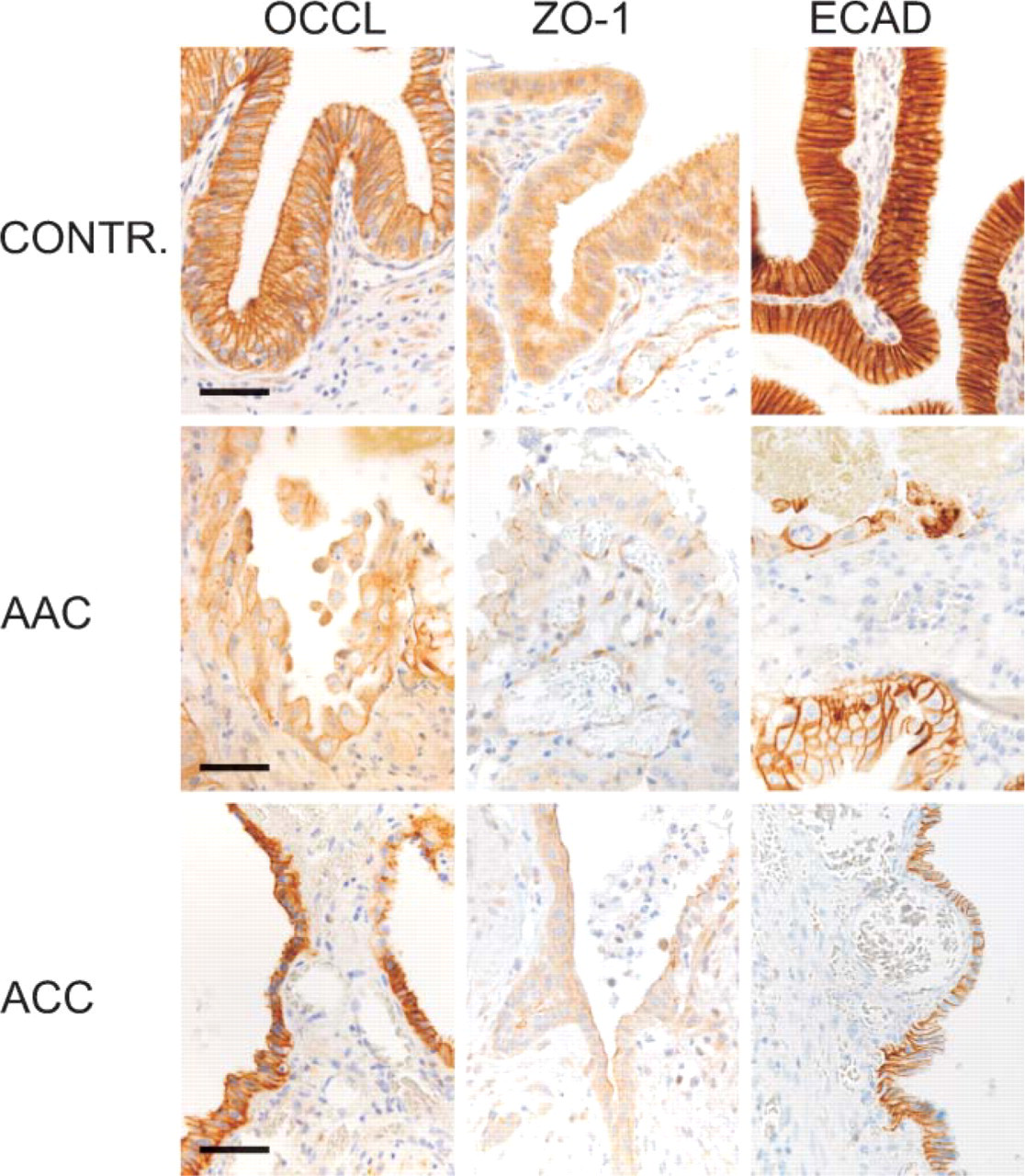
Micrographs of normal gallbladder (CONTR.), acute acalculous cholcystitis (AAC), and acute calculous cholecystitis (ACC). Panels show immunohistochemical stainings for occludin (OCCL), ZO-1, and E-cadherin (ECAD). Bar = 0.05 mm.
In conclusion, we demonstrated that TJ proteins are widely expressed in normal gallbladder epithelium. In both AAC and ACC, TJ protein expression is changed, and there are interesting qualitative and quantitative differences in the pattern of abnormal expression between these entities. In AAC, the observed slight decrease in the expression of occludin and claudin-1 could be a local manifestation of a systemic inflammatory response possibly associated with the effects of visceral hypoxia. In ACC, more extensive decrease in the expression of several TJ proteins and increase in the expression of claudin-2 are consistent with the intensive effects of local acute inflammation and infection and likely to be mediated by the effects of locally released proinflammatory mediators and inflammatory cells and possibly by infectious agents.
Footnotes
Acknowledgements
We thank Professor Juha Peltonen for constructive comments on this study and Ms. Riitta Vuento for skillful technical assistance in immunohistochemistry.
