Abstract
In peripheral nerves, groups of Schwann cell–axon units are isolated from the adjacent tissues by the perineurium, which creates a diffusion barrier responsible for the maintenance of endoneurial homeostasis. The perineurium is formed by concentric layers of overlapping, polygonal perineurial cells that form tight junctions at their interdigitating cell borders. In this study, employing indirect immunofluorescence and immunoelectron microscopy, we demonstrate that claudin-1 and -3, ZO-1, and occludin, but not claudin-2, -4, and -5, are expressed in the perineurium of adult human peripheral nerve. We also describe the expression of occludin, ZO-1, claudin-1, -3, and -5 in the developing human perineurium, showing that the expressions of claudin-1 and -3, ZO-1, and occludin follow similar spatial developmental expression patterns but follow different timetables in achieving their respective adult distributions. Specifically, claudin-1 is already largely restricted to perineurium-derived structures at 11 fetal weeks, whereas claudin-3 and occludin are weakly expressed in the perineurial structures at this age and acquire a well-defined perineurial distribution only between 22 and 35 fetal weeks. ZO-1 appears to acquire its mature profile even later during the third trimester. The results of the present and previous studies show that the perineurial diffusion barrier matures relatively late during human peripheral nerve development.
I
Diffusion through the perineurium is affected by the exceptionally thick basement membranes present in the structure (Gamble and Eames 1964), whose negative charge functions as a molecular sieve, selectively preventing the diffusion of certain compounds. The perineurium also constitutes a metabolically active barrier, facilitating the transport of molecules such as glucose through the barrier, as indicated by the presence of glucose transporter 1 in the perineurium (Muona et al. 1993; Tserentsoodol et al. 1999). Furthermore, macromolecules can traverse the perineurial cell layer by transcytosis (Oldfors 1981).
The membranes of adjacent perineurial cells overlap within each perineurial cell layer, and transmission and scanning electron microscopy have revealed tight junctions at the interdigitating cell borders (Thomas 1963; Reale et al. 1975; Beamish et al. 1991). Functional studies using different tracers and electrophysiological studies examining the ionic permeability have shown that tight junctions selectively prevent the diffusion of molecules through the perineurium (Kristensson and Olsson 1971; Ghabriel et al. 1989; Todd et al. 2000a,b).
Recent studies of epithelial tight junctions have provided novel insight with regards to the structure and function of tight junctions in general. In epithelia, tight junctions form continuous seals around cells. However, far from being simple barriers, they display ion and size selectivity, thus serving as a regulated permeability barrier that prevents water, solutes, and immune cells from passing freely through the paracellular pathway (Anderson 2001; Tsukita and Furuse 2002). The best characterized tight junction components are occludin, ZO-1, and the claudin family. Localized exclusively in tight junctions, occludin is a transmembrane protein with four hydrophobic trans-membrane helixes, whose amino (N) and carboxy (C) terminals are located on the cytoplasmic side of the plasma membrane (Furuse et al. 1993; Tsukita et al. 2001). ZO-1, located in the cytoplasmic plaques of tight junctions, is believed to contribute to the correct organization of proteins within the tight junction plaque (Stevenson et al. 1986; Stevenson and Keon 1998; Gonzalez-Mariscal et al. 2003). Claudins constitute a family of at least 24 distinct members (Morita et al. 1999; Tsukita and Furuse 2002). They are small tetraspan membrane proteins with two short extracellular loops and short cytoplasmic N- and C termini (Furuse et al. 1998; Tsukita et al. 2001). Claudins can adhere to each other homotypically or heterotypically and play a crucial role in the formation of the tight junction barrier (Furuse et al. 1999; Tsukita and Furuse 2002).
Molecular cloning and the detailed characterization of various tight junction components have recently provided the means for a more specific analysis of tight junctions in the perineurium. Indirect immunofluorescence and immunoelectron microscopy have localized ZO-1, occludin, and claudin-1 to the perineurium of adult rat peripheral nerves (Nagaoka et al. 1999; Tserentsoodol et al. 1999; Rina-Susilowati et al. 2000; Hirakawa et al. 2003) and claudin-1 in human perineurium (Folpe et al. 2002).
Human fetal peripheral nerve undergoes significant morphological maturation until the third trimester. However, the differentiation of human perineurial cells has not been widely investigated. Previous morphological studies did not reveal cell–cell contacts between overlapping perineurial cells at 12 fetal weeks, whereas at 14 weeks intercellular contacts were observed in regions of cellular overlap (Gamble and Breathnach 1965; Gamble 1966; Jaakkola et al. 1993). Previous studies concentrating on the development of basement membranes (Jaakkola et al. 1993) and the expression of the glucose transporter 1 (Muona et al. 1993) have shown that the structure of the perineurium matures relatively late during human embryonic development. However, to our knowledge the expression of tight junction proteins has not been investigated in developing human peripheral nerves. In this study, we describe the expression of several tight junction components, including occludin, ZO-1, and claudins 1–5, in developing human peripheral nerve and compare the expression of these proteins to their expression in adult nerve.
Materials and Methods
Tissue Samples
Sciatic nerve samples from four healthy men (aged 16–40 years) who died in accidents were obtained from the Department of Pathology, Turku University Hospital, Turku, Finland, and were used for indirect immunofluorescence labeling (IIF). Great auricular nerve samples were obtained for immunoelectron microscopy (IEM) from eight tumor patients (aged 21–83 years) whose great auricular nerve had to be sacrificed for surgical reasons at parotidectomy in the Department of Otorhinolaryngology-Head and Neck Surgery, Turku University Hospital. Sciatic nerve samples from six normal fetuses of 11, 12, 21, 22, 34, and 35 fetal weeks were obtained from the Department of Obstetrics and Gynecology and the Department of Pathology, Turku University Hospital, and were used for IIF and toluidine blue staining. All tissue samples were obtained with the appropriate informed consent and approval of the Joint Ethical Committee of the Turku University Hospital and the University of Turku, Finland.
Histology
To demonstrate the light microscopic structure of developing nerves, 1-μm Epon sections of sciatic nerves were stained with 1% toluidine blue.
Antibodies
The following primary antibodies were used: affinity-purified rabbit polyclonal antibodies to human claudin-1 (51–9000; concentration 0.25 mg/ml; dilution 1:100 in IIF, 1:30 in IEM), claudin-2 (51–6100; 0.25 mg/ml; 1:100 in IIF), claudin-3 (34–1700; 0.25 mg/ml; 1:100 in IIF, 1:30 in IEM), claudin-5 (34–1600; 0.25 mg/ml; 1:100 in IIF), and occludin (71–1500; 0.25 mg/ml; 1:125 in IIF, 1:30 in IEM); mouse monoclonal antibodies to human claudin-4 (32–9400; 0.5 mg/ml; 1:100 in IIF), claudin-5 (35–2500; 0.5 mg/ml; 1:100 in IIF), and ZO-1 (33–9100; 0.5 mg/ml; 1:100 in IIF, 1:50 in IEM). All antibodies were from Zymed Laboratories (South San Francisco, CA). Mouse monoclonal antibody to human type IV collagen (M785; 1:100 in IIF) was purchased from DAKO (Glostrup, Denmark). Secondary antibodies for IIF were tetramethyl-rhodamine isothiocyanate (TRITC)-conjugated swine anti-rabbit (R0156; 0.2 mg/ml; 1:100) and rabbit anti-mouse IgG (R0270; 0.4 mg/ml; 1:100) from DAKO and Alexa Fluor 488-conjugated goat anti-mouse IgG (A-11029; 2 mg/ml; 1:100) from Molecular Probes (Eugene, OR). In double labelings, TRITC-conjugated swine anti-rabbit IgG was mixed with Alexa Fluor 488-conjugated goat anti-mouse IgG. Secondary antibodies for immunoelectron microscopy were goat anti-mouse IgG+IgM coupled to 18-nm gold particles (115–215–068; 1:30), and goat anti-rabbit IgG coupled to 12-nm gold particles (111–205–144; 1:30) from Jackson ImmunoResearch Laboratories (West Grove, PA).
Indirect Immunofluorescence Labeling
For IIF labeling, 7-μm transverse and oblique-longitudinal cryosections of adult and fetal sciatic nerves were mounted on silanated glass slides and fixed in 100% methyl alcohol at –20C for 10 min. To prevent nonspecific binding, the samples were preincubated in PBS supplemented with 1% BSA for 15 min. Primary antibodies were diluted in 1% BSA-PBS and incubated on the samples at 4C for 20 hr. After six 5-min washes in PBS, the samples were incubated with secondary antibodies at 20C for 1 hr and washed six times in PBS. In control immunoreactions, primary antibodies were replaced with 1% BSA-PBS.
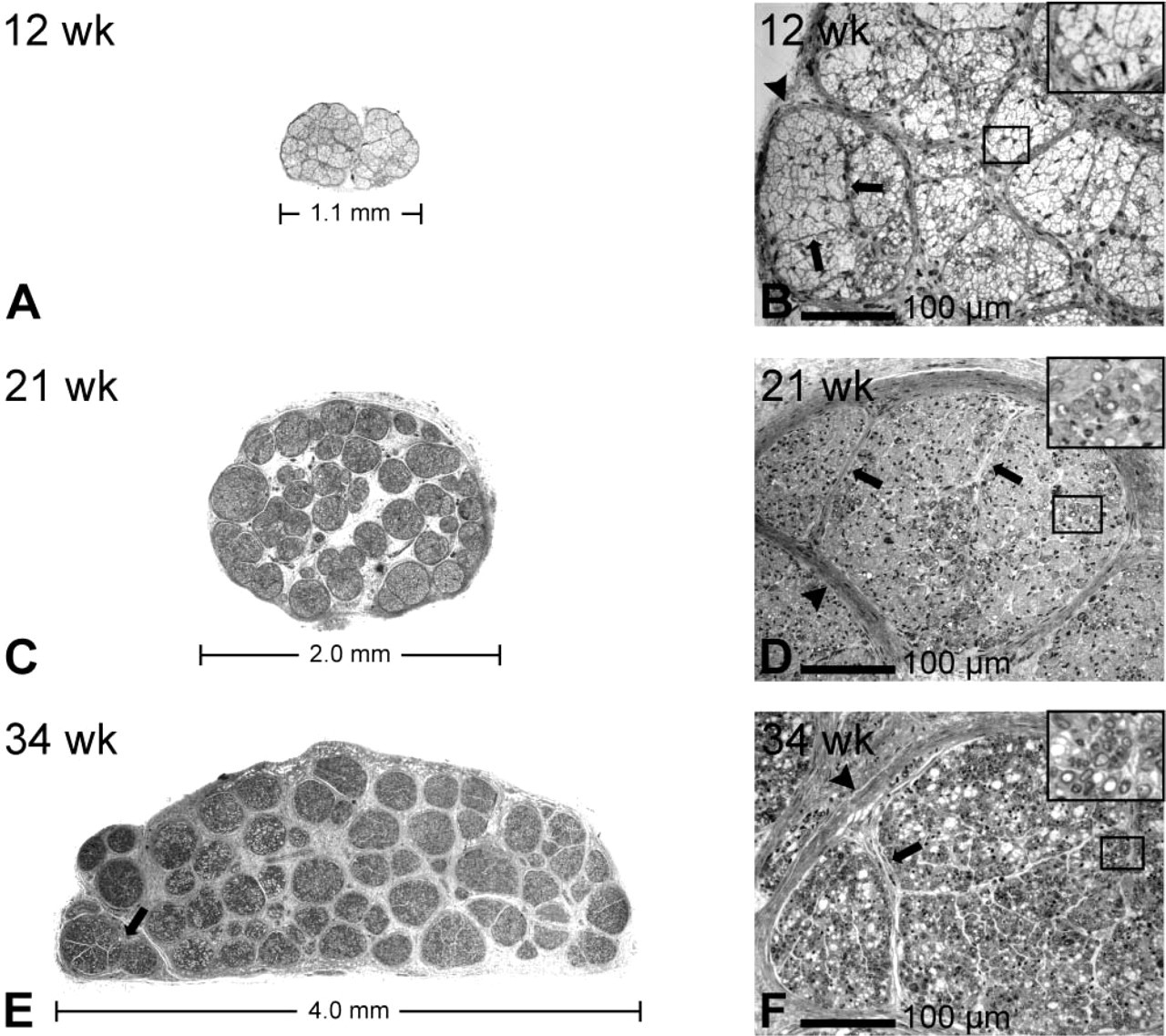
Toluidine blue staining of human fetal sciatic nerves at 12 (
Microscopy
IIF labeling and toluidine blue-stained slides were examined and photographed with a Leica DMRB fluorescence microscope equipped with a Leica DC 300F camera and Leica IM50 (version 1.20) software (Leica Microsystems; Heerbrugg, Switzerland). Resolutions of images were 1950 × 1545 pixels and images were saved in TIFF format.
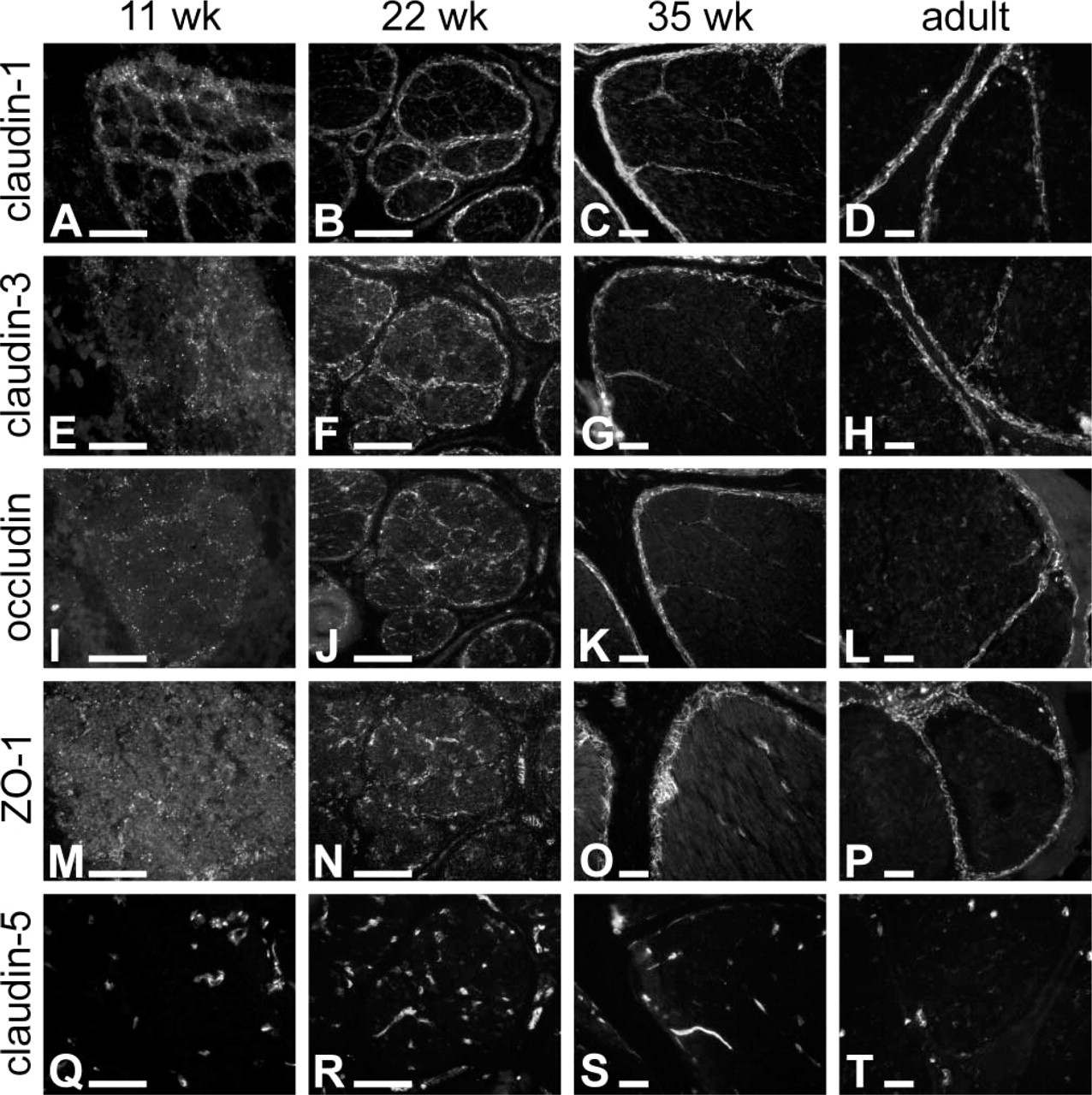
Immunofluorescence labeling of tight junction proteins in embryonic and adult human sciatic nerve. Eleven, 22, and 35 fetal weeks’ and adult nerves were immunolabeled for claudin-1, -3, and -5, ZO-1, and occludin. At 11 weeks, claudin-1, claudin-3, occludin, and ZO-1 display a discontinuous, punctate labeling pattern in the perineurium (
Confocal laser scanning microscopy was carried out with a Zeiss LSM 510 META confocal microscope equipped with argon ion and helium–neon lasers (Zeiss; Jena, Germany) and LSM 3.0 software. The objective magnification used was ×20 (numeric aperture 0.5), ×40 (oil immersion, numeric aperture 1.3), or ×63 (oil immersion, numeric aperture 1.4). For excitation of Alexa Fluor 488 the 488-nm line and for TRITC the 543-nm line was used. The beampath for Alexa Fluor 488 contained a 488-nm main dichroic mirror and a 500–530IR-nm bandpass filter and the beampath for TRITC contained a 543-nm main dichroic mirror and a 560-nm longpass filter for detection of the emitted fluorescences. The resolutions of original images were 1024 × 1024 pixels and images were saved in TIFF format.
All images were converted to grayscale format using Corel Photo-Paint (version 9) and imported to CorelDraw (version 9) to create composite photographs.
Immunoelectron Microscopy
Pieces of great auricular nerve 1 × 2 mm were fixed for 4 hr in freshly prepared 4% paraformaldehyde-2.5% sucrose-0.1 M phosphate buffer, pH 7.4, and then washed for 12 hr in phosphate buffer at 4C. Tissue samples were then dehydrated with increasing (30–100%) series of ethanol and infiltrated with LR White resin (London Resin; Berkshire, UK). Polymerization was performed in a 50C oven for 24 hr. Ultrathin sections were cut on coated nickel grids. To block nonspecific binding the sections were incubated with goat serum, diluted in 1% BSA-Tris-buffered saline (TBS)-0.3% Tween-20 for 30 min. Sections were then incubated with the primary antibodies diluted in 1% BSA-TBS-0.3% Tween-20 for 20 hr at 4C, followed by several washes in 1% BSA-TBS-0.3% Tween-20. Appropriate secondary antibodies were diluted in 1% BSA-TBS-0.3% Tween-20 and incubated for 1 hr at 20C. After several washes in TBS-0.3% Tween-20, the samples were postfixed in 2% glutaraldehyde in TBS-0.3% Tween-20 and counterstained with uranyl acetate and lead citrate. The sections were examined and photographed with a JEOL 1200EX electron microscope.
Results
Figure 1 shows the development of the fascicular morphology of human sciatic nerve. Between 11 and 35 fetal weeks, the diameter of the nerve increases due to growth of the fascicles and the synthesis of epineurial extracellular matrix (Figures 1A, 1C, and 1E). The increase in the diameter of the fascicles can be attributed to myelination of the axons and an increased amount of endoneurial extracellular matrix (Figures 1B, 1D, and 1F). During the first trimester, groups of axon–Schwann cell units are surrounded by perineurial sheaths comprising one or two cell layers (Figure 1B). Thin septae, extending from the perineurial sheath into the endoneurial space, subdivide the fascicles into smaller groups of nerve fibers. The nuclei observed in both the perineurium and the septae are spindle-shaped, resembling those of adult perineurial cells (Figures 1B, 1D, and 1F). The number of cell layers in the perineurium and in intrafascicular septae increases during development (Figures 1D and 1F). By 35 fetal weeks, perineurial sheaths surrounding adjacent fascicles tend to merge, leaving only small amounts of epineurial collagen between them (Figure 1E).
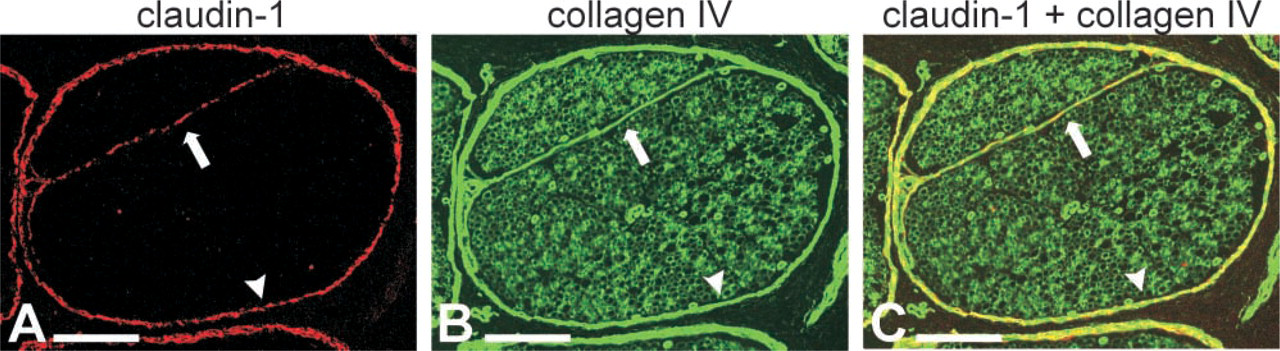
Confocal image of double immunolabeling of claudin-1 and type IV collagen in adult human peripheral nerve. Claudin-1 (
The blood–nerve barrier is constituted by the perineurium and the endothelium of endoneurial microvessels (Allt and Lawrenson 2000). An important role in the function of the blood–nerve barrier can be attributed to tight junctions that can regulate diffusion of various agents between the neighboring cells (Thomas et al. 1993). To investigate the molecular composition of the perineurial diffusion barrier, expression patterns of tight junction proteins, including claudins 1–5, ZO-1, and occludin, were examined by immunofluorescence microscopy in adult human sciatic nerve. Adult perineurium was negative for claudin-2 and -4 (not shown) and -5 (Figure 2T). Claudin-1 and -3, ZO-1 and occludin, on the other hand, were expressed in adult perineurium and in the intrafascicular septae (Figures 2D, 2H, 2L, 2P and 4C, 4F, 4I, and 4L). Double labeling for claudin-1 and basement membrane collagen IV revealed partial co-localization of type IV collagen and claudin-1, indicating that the cells forming the intrafascicular septae express a typical basement membrane component and represent perineurial cells (Figure 3).
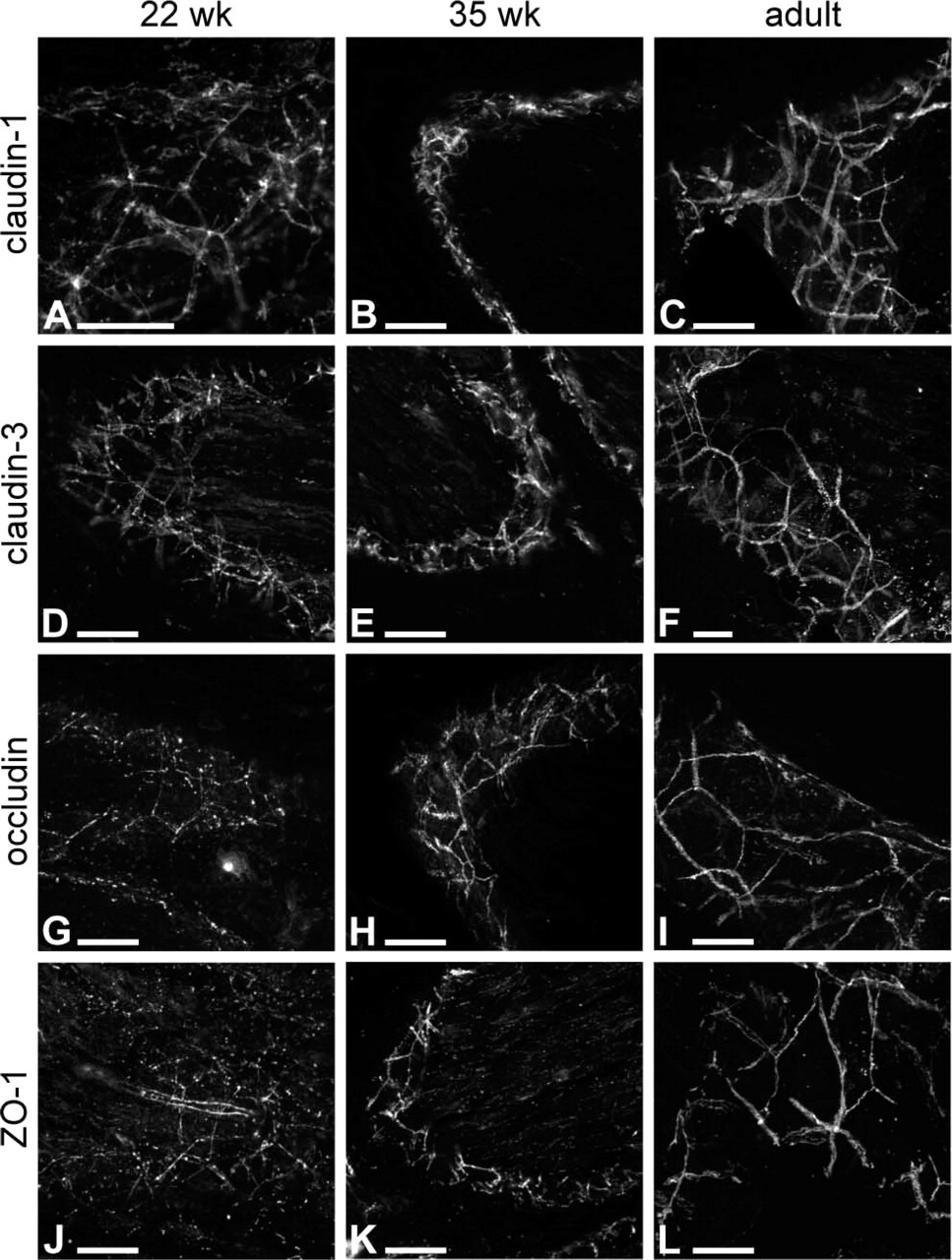
Immunofluorescence micrographs of tight junction proteins in longitudinal-oblique sections of embryonic and adult human sciatic nerve perineurium. Claudin-1 and -3, occludin, and ZO-1 are localized at sites of intercellular contact between perineurial cells at 22 and 35 weeks and in adult (
Previous ultrastructural studies have revealed that intercellular junctions appear between 12 and 14 fetal weeks in developing human perineurium (Gamble and Breathnach 1965; Gamble 1966; Jaakkola et al. 1993). To evaluate the expression of tight junction components in developing perineurium and to correlate these findings with the earlier ultrastructural studies, fetal nerves representing all trimesters were immunolabeled for tight junction components. In embryonic sciatic nerves, claudin-1 and -3, ZO-1, and occludin localized to the perineurium and the intrafascicular septae. The spatial developmental expression patterns of all four tight junction proteins were apparently similar (Figures 2 and 4), but they followed different timetables in achieving their adult distributions in the perineurium and in the intrafascicular cellular septae (Figures 2 and 4). The first to attain its adult distribution was claudin-1 which, as early as 11 fetal weeks, was localized to the perineurium in an intense punctate pattern and, to a somewhat lesser extent, to the intrafascicular septae (Figure 2A). Claudin-3 and occludin exhibited a more scattered punctate labeling of the perineurial sheath, while ZO-1 was hardly detectable at 11 weeks (Figures 2E, 2I, and 2M). At 22 weeks, ZO-1 displayed a poorly defined perineurial and intrafascicular septal labeling (Figure 2N) and occludin was discontinuous in the perineurium (Figure 2J), while claudin-1 and claudin-3 clearly delineated the individual fascicles (Figures 2B and 2F). Nevertheless, by 35 fetal weeks ZO-1 expression has been considerably upregulated in the perineurium and intrafascicular septae, although it persists in the endoneurial spaces (Figure 2O). These expression patterns are confirmed by oblique–longitudinal sections of the nerves (Figure 4), which also illustrate the characteristic continuous linear expression of all four tight junction proteins on the perineurial cell membranes by the age of 35 weeks (Figures 4B, 4C, 4E, 4F, 4H, 4I, 4K, and 4L). Here, again, expression of ZO-1 and occludin by the perineurial cells appears to lag behind that of claudin-1 and -3: ZO-1 and occludin show largely punctate pattern at 22 fetal weeks (Figures 4A, 4D, 4G, and 4J). High magnification of adult perineurium revealed a high density of tight junction proteins, especially for claudin-1 and -3, in the zones of convergence between several perineurial cells (Figures 4C, 4F, and 5). The endoneurial labeling for claudin-1 and -3 and occludin was faint compared with their labeling in the perineurium. As expected, the expression of claudin-5, characteristic of endothelial cells, was not associated with perineurial cells (Figures 2Q–2T), serving as a good negative control for the specificity of the perineurial cell protein expression patterns.
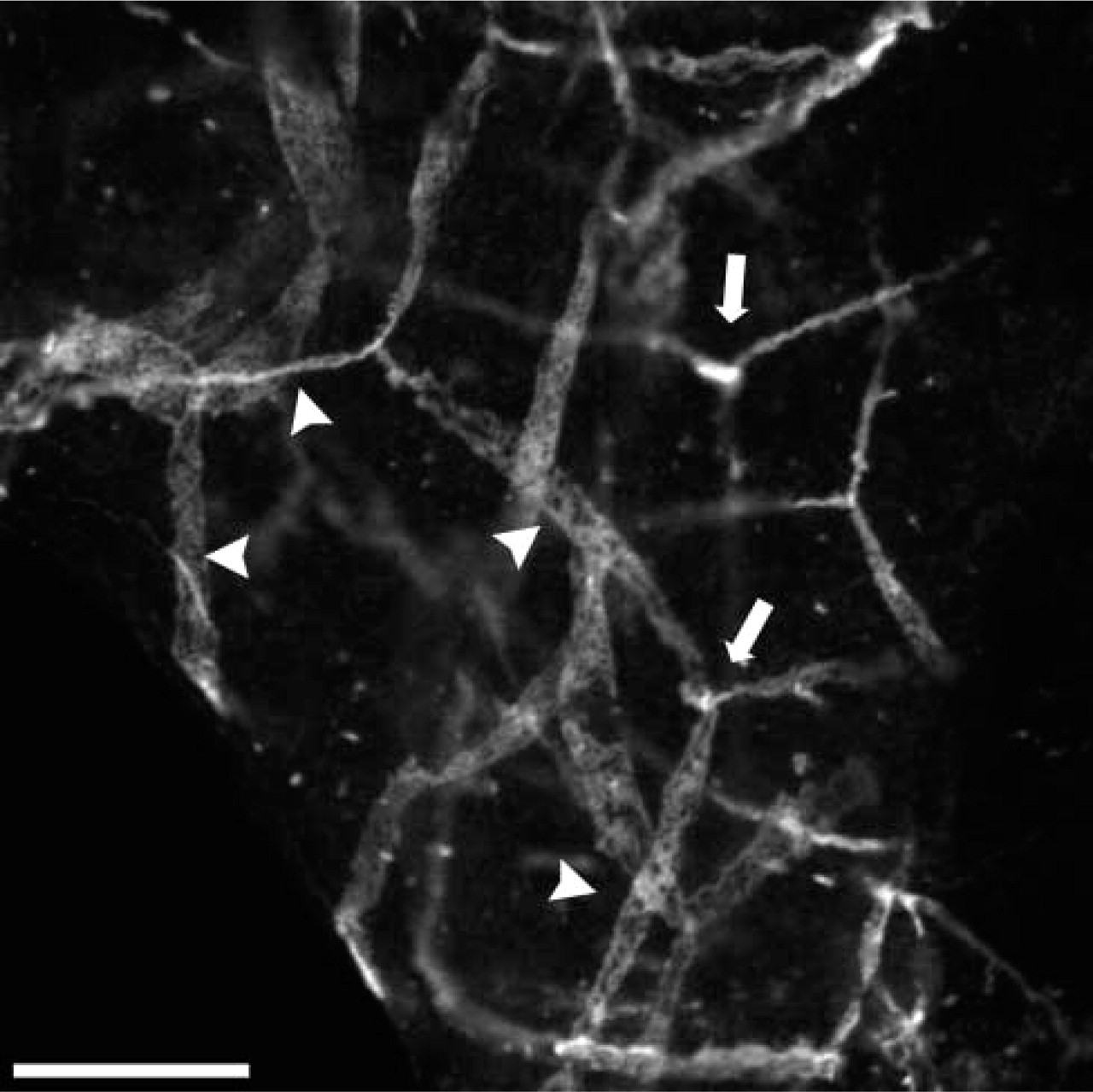
Partial magnification of Figure 4C shows high density of tight junction proteins for claudin-1 in the zones of convergence between several perineurial cells (arrows). Arrowheads point to the cell borders. Bar = 20 μm.
To correlate the findings of immunofluorescence studies with ultrastructure of the perineurium, post-embedding immunolabeling for ZO-1, occludin, and claudin-1 and -3 was performed. As expected, gold particles were detected at sites of close apposition of adjacent perineurial cells. The localization of the four perineurial tight junction proteins is shown in Figure 6.
Discussion
Together with endoneurial blood vessels, the perineurium forms a blood-nerve barrier that guarantees the constant endoneurial milieu (for review see Allt and Lawrenson 2000). Tight junctions constitute an essential part of this barrier in both perineurial and endothelial cells (Kristensson and Olsson 1971; Ghabriel et al. 1989; Todd et al. 2000a,b). The present work is a comprehensive mapping of several tight junction components in human nerve, including the developmental aspect (Table 1). This study demonstrates that claudin-1 and -3, ZO-1, and occludin are expressed in the perineurium of adult human peripheral nerve, whereas claudin-2, -4, and -5 are not detectable in mature perineurial tissue. Therefore, perineurial tight junctions can contain ZO-1, occludin, and three different claudin dimers, claudin-1/-1, claudin-1/-3, and claudin-3/-3. Claudin dimers are considered to be essential for the barrier function of tight junctions. The combination and mixing ratios of claudins within individual paired strands is considered to determine their tightness and selectivity (for review see Tsukita and Furuse 2002).
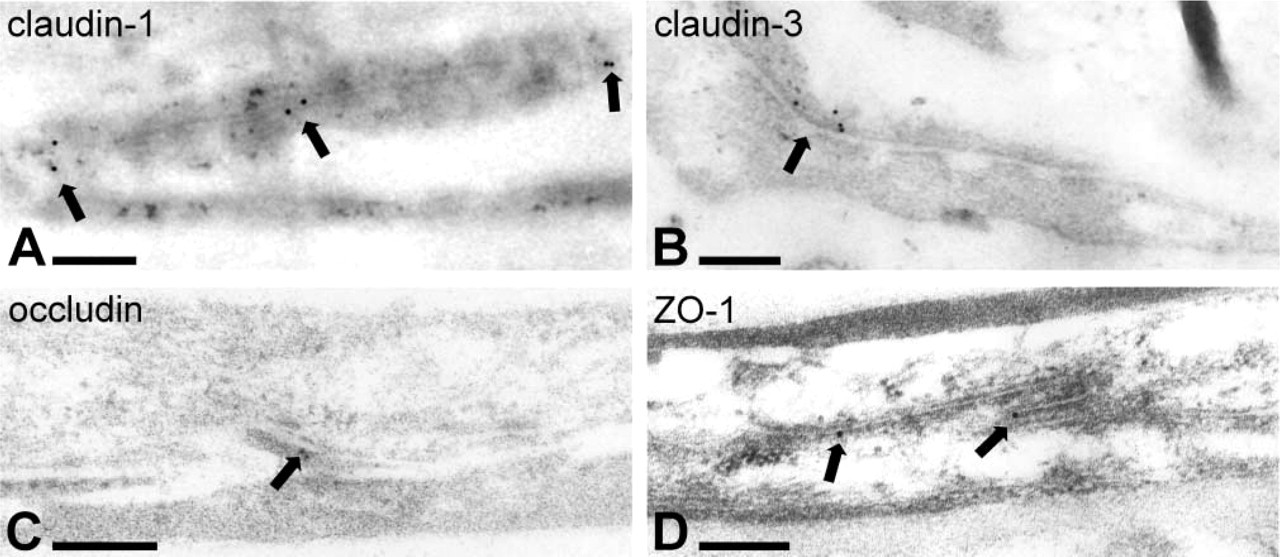
Immunoelectron microscopy of human adult great auricular nerve. Immunogold labeling for claudin-1 (
To our knowledge, only three articles concerning intercellular junctions of human fetal perineurium are available (Gamble and Breathnach 1965; Gamble 1966; Jaakkola et al. 1993). According to those morphological studies, intercellular contacts between overlapping perineurial cells begin to develop between 12 and 14 fetal weeks. Functional studies of rodent perineurial diffusion barrier have revealed species differences in the timetable of the perineurial development. Specifically, the perineurial diffusion barrier is not mature in newborn mice, whereas in newborn guinea pigs the barrier is fully functional (Kristensson and Olsson 1971). The present study evaluates the maturation of human perineurial tight junctions by analyzing the expression of several tight junction components at different time points. The results show that the four perineurial tight junction proteins (claudin-1 and -3, ZO-1, and occludin) follow slightly different expression timetables during human embryonic development. Specifically, antibodies to claudin-1 label the perineurium and intrafascicular septae as early as 11 fetal weeks, whereas ZO-1 acquires its mature profile markedly later. The expression pattern of ZO-1 in perineurium is still fragmented at the end of the second trimester and resembles the adult perineurial distribution only during the third trimester. Therefore, the full complement of proteins necessary to form mature, continuous tight junctions is reached relatively late during development and the barrier formed by tight junctions may not be completely functional until the third trimester. This is in line with previous observations on the expression of other proteins that participate in the formation of the perineurial diffusion barrier. Specifically, components of basement membranes, such as type IV collagen and laminin, appear concomitantly with the expression of tight junction proteins, whereas the structure of the basement membranes does not become continuous until the end of the third trimester (Jaakkola et al. 1993). Glucose transporter 1 gene expression is also turned on in the perineurium between 15 and 17 fetal weeks, and immunolabeling for glucose transporter 1 protein becomes intense and continuous by 24 fetal weeks (Muona et al. 1993).
The spatial expression pattern of all tight junction proteins studied here changed from punctate to linear during the maturation of the perineurium. From week 22 onwards, the zones of convergence between several perineurial cells are particularly intensely labeled for tight junction proteins; this is especially remarkable for claudin-1 and -3. On the basis of these results, it is reasonable to speculate that these zones of convergence between several cells are particularly well sealed by tight junctions.
Structural development of perineurium, with special reference to the expression of tight junction proteins

In conclusion, the results of this study show that the structural diffusion barrier in human perineurium matures quite late during fetal life. Immature perineurium may allow infectious agents or therapeutic agents to have easier access to the nervous system compared with what is normal in mature babies. This should be taken into account especially for fetuses during pregnancy and prematurely born babies.
Footnotes
Acknowledgements
Supported by the Cancer Societies of Finland, the Finnish Society of Dermatology, the Paijat-Hame Fund of the Finnish Cultural Foundation, the Turku University Foundation and Turku University Central Hospital (Turku, Finland), Oulu University Hospital, and the Department of Dermatology, University of Oulu (Oulu, Finland).
