Abstract
Helicobacter pylori infects over half the population worldwide and is a leading cause of chronic gastritis and gastric cancer. However, the mechanism by which this organism induces inflammation and carcinogenesis is not fully understood. In the present study we used insulin-gastrin (INS-GAS) transgenic mice that fully develop gastric adenocarcinoma after infection of H. pylori-related Helicobacter felis. Histological examination revealed that more than half of those mice developed invasive adenocarcinoma after 8 months of infection. These carcinomas were stained by NCC-ST-439 and HECA-452 that recognize 6-sulfated and non-sulfated sialyl Lewis X. Lymphocytic infiltration predominantly to submucosa was observed in most H. felis-infected mice, and this was associated with the formation of peripheral lymph node addressin (PNAd) on high endothelial venule (HEV)-like vessels detected by MECA-79. Time-course analysis of gene expression by using gene microarray revealed upregulation of several inflammation-associated genes including chemokines, adhesion molecules, surfactant protein D (SP-D), and CD74 in the infected stomach. Immunohistochemical analysis demonstrated that SP-D is expressed in hyperplasia and adenocarcinoma whereas CD74 is expressed in adenocarcinoma in situ and invasive carcinoma. These results as a whole indicate that H. felis induces HEV-like vessels and inflammation-associated chemokines and chemokine receptors, followed by adenocarcinoma formation.
Keywords
H
H. pylori-associated gastritis is initiated by the host's strong neutrophilic response, and this response is largely replaced with lymphoplasmacytic infiltration into the lamina propria. This inflammatory response is followed by intestinal metaplasia and expression of intestine-specific genes, dysplasia, and eventually gastric adenocarcinoma (Peek and Blaser 2002; Yuasa 2003), suggesting an inflammation-carcinoma sequence in the infected stomach. Although a large number of individuals are infected with H. pylori, ∼3% and 1% of infected individuals develop peptic ulcer and adenocarcinoma, respectively (Peek and Blaser 2002). Recent studies showed that mucin proteins expressing αd,4-linked N-acetylglucosamine prevent or delay the progression of gastric diseases initiated by H. pylori infection (Kawakubo et al. 2004). On the other hand, it has been shown that H. pylori adheres to Lewis b blood group antigen and sialyl dimeric Lewis × carbohydrate on the gastric surface mucous cells, thus facilitating H. pylori infection (Ilver et al. 1998; Mahdavi et al. 2002). In addition, 6-sulfo sialyl Lewis X-capping structures on O-glycans function as a peripheral lymph node addressin (PNAd), which may recruit lymphocytes during inflammation (Renkonen et al. 2002; von Andrian and Mempel 2003; Rosen 2004).
Indeed, we have shown that H. pylori induces PNAd in inflamed human gastric mucosa (Kobayashi et al. 2004). The number of PNAd-expressing high endothelial venule (HEV)-like vessels increases with the progression of inflammation caused by H. pylori infection. Moreover, eradication of H. pylori by antibiotics results in disappearance of PNAd (Kobayashi et al. 2004). These results strongly suggest that PNAd induced by H. pylori infection may facilitate an inflammatory response by the host. However, molecular events taking place during the transition from inflammatory response to malignant transformation are not fully understood.
Several molecules have been reported to be associated with H. pylori-induced gastritis and gastric cancer (Rieder et al. 2001; Murray et al. 2002; Mueller et al. 2003). In humans, analysis of gastric mucosa from H. pylori-infected patients showed upregulation of IL-8 and ENA-78, which activates neutrophils (Rieder et al. 2001). Similarly, innate immune molecule surfactant protein D (SP-D) was upregulated in H. pylori-infected human gastric mucosa (Murray et al. 2002). Furthermore, in a BALB/c mouse model, gene microarray analysis showed that Helicobacter heilmannii-induced MALT lymphoma is characterized by upregulation of numerous genes (Mueller et al. 2003). Among them, the transition to destructive lymphoepithelial lesions and malignant lymphoma is marked by an increase in transcription of a single gene encoding calgranulin A/Mrp-8. However, no studies have been reported on how genes related to inflammatory responses are regulated during the course of the disease process initiated by Helicobacter infection. An early maker for gastric carcinoma has also not been reported.
In the present study we focused on how expressions of genes related to inflammation and lymphocyte recruitment are regulated during inflammation-carcinoma sequence initiated by Helicobacter infection. For this study we used insulin-gastrin (INS-GAS) transgenic mice of which transgene expresses human gastrin under influence of the rat insulin promoter (Wang et al. 1993). In INS-GAS mice, progression of inflammation and carcinoma formation is accelerated more than in wild-type mice, making this animal model suited for studying inflammation-carcinoma sequence (Wang et al. 2000). Using gene microarrays available from the Consortium for Functional Glycomics, we found that distinct expression profiles of genes related to inflammation are associated with H. felis-initiated inflammation and gastric carcinoma. The same analysis and histological examination showed SP-D (Persson et al. 1990) and CD74 (Cresswell 1996) increase in hyperplasia and adenocarcinoma and in dysplasia/adenocarcinoma in situ and invasive carcinoma, respectively. These results suggest that H. felis-induced disease progression requires expression of a distinct set of molecules in a temporally precise manner.
Materials and Methods
Animals
Forty six (24 male, 22 female) 8-week-old INS-GAS transgenic mice on FVB background were used in this study. For time course study, seven to eight mice were subjected to H. felis infection, and one or two mice served as control at each time point (0, 2, 4, 6, and 8 months after the start of the experiment). Mice were housed in autoclaved polycarbonate cages under conventional conditions, fed an autoclaved commercially prepared pelleted diet, and given autoclaved water ad libitum. Animals were treated in accordance with guidelines of the National Institutes of Health (Bethesda, MD), and experiments were approved by the Animal Research Committee of Burnham Institute for Medical Research.
Bacteria and Experimental Infection
H. felis (ATCC 49179; American Type Culture Collection, Manassas, VA) was cultured in Brucella broth (Becton Dickinson; Franklin Lakes, NJ) containing 5% horse serum (Irvine Scientific; Santa Ana, CA) at 37C in 15% CO2 with flat rotation at 100 rpm for 3 days. Bacteria [0.5 ml of Brucella broth containing 5 × 108 of H. felis per 1 ml (a density of 1 OD at 600 nm)] were introduced three times into the stomach by gavage every other day after 16 hr fasting. Mice were euthanized with CO2 at various intervals after inoculation (0, 2, 4, 6, and 8 months) and analyzed. Only those mice that exhibited Helicobacter colonization revealed by immunohistochemistry as described below were used.
Histopathological Evaluation
One half of the stomach, incised along the greater curvature, was fixed in 4% paraformaldehyde in PBS at 4C for 24 hr. These materials were embedded in paraffin, cut at 3 μm, and stained with hematoxylin and eosin (H-E). Due to the difficulty to distinguish adenocarcinoma in situ from severe dysplasia, we combined both lesions into one category, i.e., dysplasia/adenocarcinoma in situ. Histological hallmarks indicative for the dysplasia/adenocarcinoma in situ included (1) intraglandular folding, papillary projections, coalescent or cribriform glandular structure, (2) moderate to marked cellular pleomorphism with increased nuclear/cytoplasmic (N/C) ratio, (3) euchromatic nuclei with conspicuous nucleoli, and (4) more than two mitotic figures per high power field with or without occasional bizarre mitotic figures. On the other hand, invasive carcinoma was defined as invasion of glands with loss of the normal mucosal architecture and cellular atypia into submucosa or muscularis propria, mimicking non-carcinomatous lesions such as diverticulosis and gastritis cystica profunda (Shimizu et al. 1999; Wang et al. 2000). Incidence of dysplasia/carcinoma in situ and invasive carcinoma at each time point was compared with that of 0 time point using Fisher's exact test, and p values <0.05 were considered significant.
Immunohistochemistry
The following antibodies were used in this study: MECA-79 (rat IgM; BD Pharmingen, San Diego, CA) recognizing 6-sulfo N-acetyllactosamine on extended core 1 O-glycans that constitute PNAd (Yeh et al. 2001), HECA-452 (rat IgM, BD Pharmingen) recognizing sialyl Lewis × and sialyl Lewis a with or without 6-sulfate groups in O- and N-glycans (Berg et al. 1991; Kobayashi et al. 2004), NCC-ST-439 (mouse IgM, Nippon Kayaku, Tokyo, Japan) recognizing sialyl Lewis × on core 2 branched O-glycans (Kumamoto et al. 1998; Kobayashi et al. 2004), In-1 (rat IgG2b, κ, BD Pharmingen) recognizing mouse CD74 (Ii chain), anti-mouse SP-D (rabbit polyclonal; Chemicon, Temecula, CA), anti-H. pylori (rabbit polyclonal; Dako, Carpinteria, CA), which cross-reacts readily with H. felis, MECA-367 (rat IgG2a, κ, BD Pharmingen) recognizing mouse MAdCAM-1 (Nakache et al. 1989), and RA3-6B2 (rat IgG2a, κ, BD Pharmingen) recognizing mouse CD45R/B220 expressed on B-cell lineage (Coffman 1982).
Immunostaining was performed by the labeled streptavidin-biotin (LSAB) method on paraffin-embedded tissue sections as described previously (Kobayashi et al. 2004). Briefly, before staining, antigens were retrieved by boiling sections in a microwave oven in 10 mM citrate buffer (pH 6.0) for MAdCAM-1 or by 5-min treatment with 20 μg/ml proteinase K (Roche; Basel, Switzerland) for H. felis. After that, tissue sections were blocked with 3% fetal bovine serum (FBS) (Invitrogen; Carlsbad, CA) in PBS followed by blocking endogenous biotin using an Avidin/Biotin Blocking Kit (Vector Laboratories; Burlingame, CA) according to the manufacturer's instruction. After incubation with a primary antibody overnight at 4C, sections were incubated with host- and class-matched biotinylated secondary antibodies (Pierce; Rockford, IL) for 60 min. After washing with PBS, slides were incubated with horseradish peroxidase (HRP)-conjugated streptavidin (Dako). Color reaction was developed with 3,3′-DAB containing 0.02% H2O2. Sections were briefly counterstained with hematoxylin. Negative control experiments were carried out by using isotype-matched immunoglobulins (Beckman Coulter; Fullerton, CA), and no specific stainings were observed.
Selectin·IgM Chimera Binding Assay
L- and E-selectin·IgM chimeras were prepared as described (Kobayashi et al. 2004). After blocking endogenous peroxidase activity as described above, tissue sections were incubated with selectin·IgM chimera and washed with DMEM (Irvine Scientific). The sections were then incubated with HRP-conjugated goat anti-human IgM antibody (Pierce) and rinsed in DMEM. The color reaction was developed with DAB as described above. Sections were briefly counterstained with hematoxylin.
RNA Isolation and Transcription Microarray Analysis
Analysis of gene expression was conducted using a custom gene microarray (GLYCOv2 chip) produced by Affymetrix (Clara, CA) for the Consortium for Functional Glycomics (
RNA was extracted from the remaining half of each stomach using Trizol reagent (Invitrogen; Carlsbad, CA). After treatment with DNase I amplification grade (Invitrogen), total RNA was purified with RNeasy columns using the clean-up protocol (Qiagen; Valencia, CA) and its quality checked with an Agilent Bioanalyzer (Agilent Technologies; Palo Alto, CA). For expression analysis, 5 μg of total RNA was amplified and biotin labeled using the Bioarray High Yield RNA Transcript Labeling Kit (Enzo Life Sciences; Farmingdale, NY) and then hybridized to the GLYCOv2 chip. One chip per biological sample was run. Hybridization and scanning of the chip were performed according to recommended protocols of Affymetrix. Gene microarray data from this study has been deposited in the Consortium of Functional Glycomics database (
The microarray dataset was normalized using Robust Multichip Average (Bolstad et al. 2003) and then imported into GeneSpring (Silicon Genetics; Redwood City, CA) for ANOVA analysis. For each of the five time points (0, 2, 4, 6, and 8 months) the experiment consisted of three to five infected mice, chosen randomly from the mice displaying H. felis infection. For each time point (2, 4, 6, and 8 months), one non-infected mouse was subjected to analysis. The signals for each time point were normalized to their age-matched control signals that were represented by a mouse uninfected with H. felis and then normalized to the zero month expression values. ANOVA analysis was performed to test for genes that were differentially expressed at any time point compared with time zero at a significance level of 0.05. Hierarchical clustering was performed using Pearson correlation as a measure of similarity, after average linkage and median centering of values. To determine that the gene expression of the infected mouse was indeed increased relative to the uninfected mouse, profiles of the significant upregulated genes were plotted against their time-matched controls. These plots were used to determine which genes were consistent with a time-dependent pattern of expression such that the expression values of the uninfected mouse were outside the 95% confidence interval of the expression of the infected mice.
Semiquantitative RT-PCR of Glycosyltransferases and Sulfotransferase
The same RNA samples used for the gene microarray analysis mentioned above were treated with RNase-free DNase I (Roche), and first-strand cDNA was synthesized using Super-Script II RNase H− reverse transcriptase (Invitrogen). Negative controls were obtained by omitting reverse transcriptase. PCR was carried out using JumpStart REDTaq DNA polymerase (Sigma-Aldrich; St Louis, MO) and the following PCR primers: core 1 extension enzyme (Core1-β3GlcNAcT); 5′-ATGTGTTCGCACACACGGACAACA-3′ and 5′-GAACACGTCGTCGATTGGGAACAT-3′, Core2GlcNAcT-1; 5′-AGGGTGACCCAGAAGAAATCCAGA-3′ and 5′-CTCAGGAGCCTGTCAAGCATTTCA-3′, Core2GlcNAcT-2; 5′-ATTGCGTACTCCATGGTGGTGCAT-3′ and 5′-ACCAGGAAGCATAGACCACTGACA-3′, Core2GlcNAcT-3; 5′-TGGTTTCAAGGGAGGAAGAGGACT-3′ and 5′-GAGGAGGTCTGATAAGCAGTTCCA-3′, FucT-VII; 5′-GCTGGAGGAGCAACATTCATGGTA-3′ and 5′-ATCTGAATCACGCCGATAGCTCAG-3′,GlcNAc6ST-2 (LSST); 5′-AGCCCTTTGATATGGTGGAGAAGG-3′ and 5′-TGGTCTTCCTCCTTGATCGTTTCC-3′,MIP-3α 5′-AGAAGCAGCAAGCAACTACGACTG-3′and 5′-ACTCTTAGGCTGAGGAGGTTCACA-3′. Parallel RT-PCRs were performed with primers for the mouse glyceraldehyde-3-phosphate dehydrogenase (GAPDH) gene (5′-AAGCTTGTCATCAACGGGAAGCC-3′ and 5′-CTCCACCCTTCAAGTGGGCCC-3′) to serve as a reference. PCR amplification was performed with annealing at 60C for 30 sec, extension at 72C for 1 min, and denaturation at 94C for 30 sec. Cycle numbers varied depending on transcripts analyzed. The final extension step was 72C for 5 min. Negative and positive control amplifications were performed in every experiment. Each PCR product was run on a 2% (w/v) agarose gel containing 0.1 μg/ml of ethidium bromide and visualized under UV light. Digital images were obtained and densitometry was performed using NIH image software (h7tp://rsb.info.nih.gov/nih-image/). Semiquantitative measurement of each gene transcript was achieved by comparing the expression level of each gene with that of GAPDH and expressing the result as a simple ratio. For this analysis we first made a calibration curve for each gene using serially diluted plasmids harboring its cDNA as a template. Once a linear correlation between the fluorescence intensity of PCR product and the log of initial amount of plasmid was obtained, RT-PCR was undertaken, such that all products fell into the linear portion of that calibrated curve.
Results
Progression of Inflammation in Gastric Mucosa of INS-GAS Mice
The stomach of INS-GAS mice gradually developed mucosal hyperplasia with cystic dilated glands, and the mucosal thickness substantially increased as mice aged (Figure 1A, upper panel). Upon infection with H. felis (Figure 1B), mucosal hyperplasia became more prominent compared with uninfected INS-GAS mice (Figure 1A, lower panel). Whereas the ratio of surface mucous cells to fundic gland cells was ∼1:3 in uninfected INS-GAS mice (Figure 1C), it was almost 7-8:1 in INS-GAS mice 6 months after infection (Figure 1D). These results indicate that surface mucous cells proliferate preferentially after infection with H. felis.
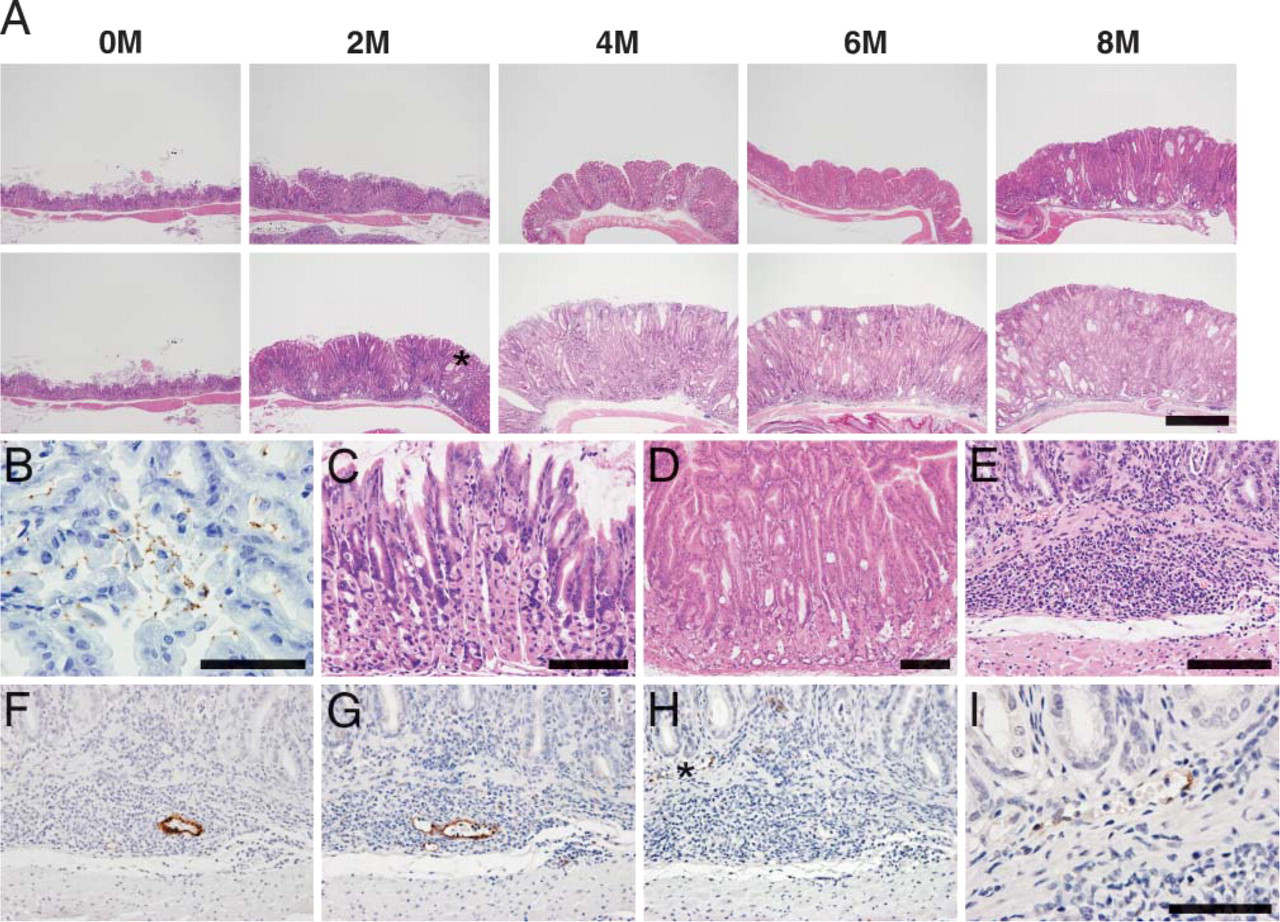
Progression of inflammation in the gastric mucosa of INS-GAS mice after infection with Helicobacter felis. (
In infected gastric mucosa, extensive lymphocyte recruitment was observed in the submucosa (Figure 1E) and associated with MECA-79-positive HEV-like vessels shown in a serial section (Figure 1F). These vessels were also bound by E-selectin·IgM chimera, indicating expression of selectin ligands on the vessels (Figure 1G). On the other hand, lymphocyte recruitment in the lamina propria was associated with MAdCAM-1-positive HEV-like vessels (Figures 1H and 1I) where MECA-79-positive HEV-like vessels were rarely found. In contrast to infected mice, no HEV-like vessels were observed in uninfected INS-GAS mice, even 8 months after the start of the experiment. These results indicate that H. felis infection induces pronounced inflammatory response in the gastric mucosa, and this response may be facilitated by lymphocyte recruitment through PNAd-expressing HEV-like vessels.
Gastric Adenocarcinoma After H. felis Infection
To determine the progression of gastric carcinoma after H. felis infection, the stomachs were histologically examined. Dysplasia/carcinoma in situ, largely characterized by proliferation of tubular or fused atypical glands, was observed in 25% of eight mice 2 months after infection (Figures 2A and 2B). Occasionally, small complex glands showing a cribriform appearance were observed (Figure 2C). These abnormal glands were not observed in control uninfected INS-GAS mice. Typical features of epithelial cells composing atypical glands found in different stages of disease progression are shown in Figures 2G-2I. It is apparent that N/C ratio of carcinoma cells (Figure 2I) is the largest, whereas that of normal epithelial cells (Figure 2G) is the smallest. N/C ratio of hyperplastic epithelia (Figure 2H) is in between. In addition, the nucleus of carcinoma cells (Figure 2I) had coarse chromatin and prominent nucleolus, whereas that of normal (Figure 2G) and hyperplastic epithelial cells (Figure 2H) did not. After 4 months of infection, adenocarcinoma invading the lamina propria (Figure 2D) and submucosa and muscularis propria (Figure 2E) was observed. In addition, vessel invasion was observed, and these carcinomas were surrounded by extensively recruited lymphocytes (Figure 2F).
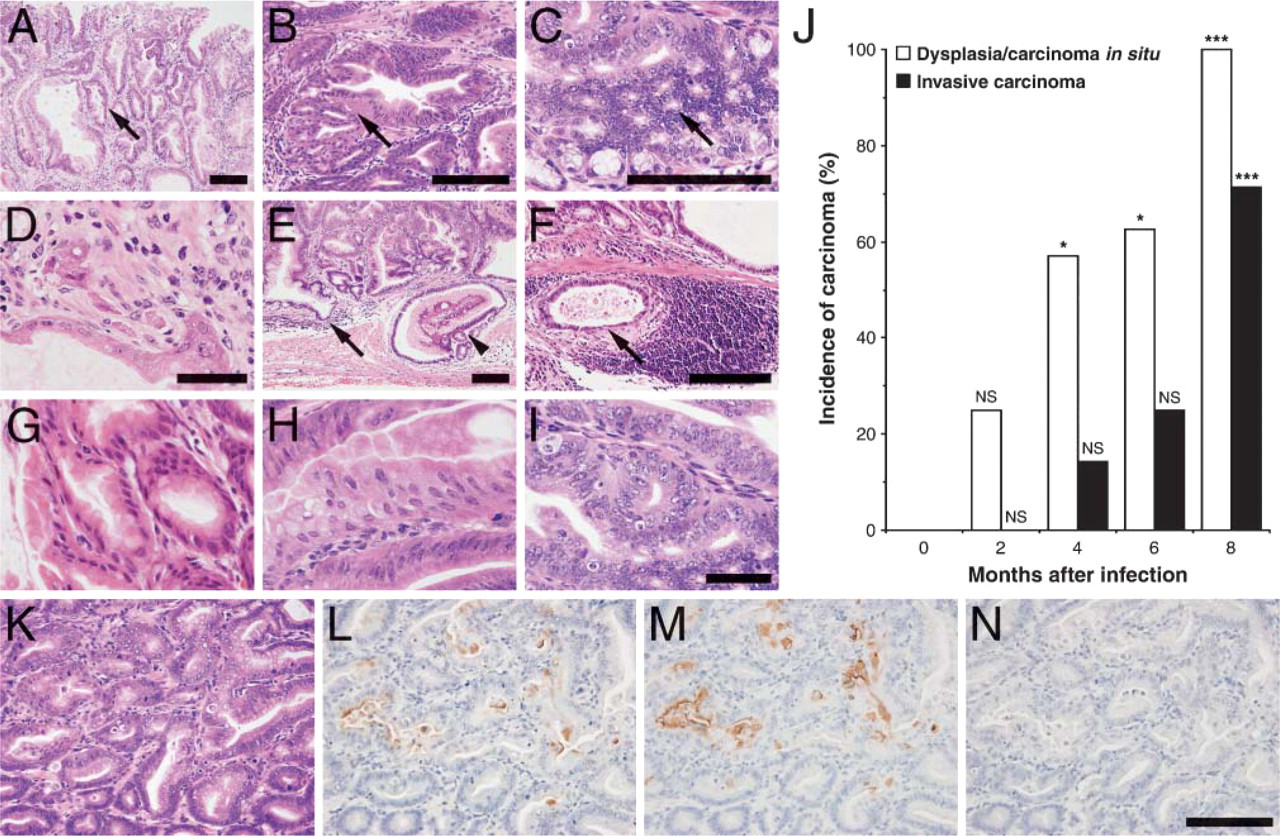
Progression of adenocarcinoma in H. felis-infected mouse stomach. (
When the incidence of dysplasia/carcinoma in situ and invasive carcinoma in INS-GAS mice was tabulated, it was concluded that such incidence was significantly increased as the observation period grew longer (Figure 2J). It was also apparent that the incidence of invasive carcinoma increased substantially later than the incidence of dysplasia/carcinoma in situ (Figure 2J). Indeed, 8 months after infection, gastric mucosa displayed invasive carcinoma in five of seven mice examined. These results combined indicate that H. felis infection significantly accelerates formation of gastric carcinoma and facilitates the establishment of invasive carcinoma in INS-GAS mice.
Expression of 6-Sulfated and Non-sulfated Sialyl Lewis × on Gastric Carcinoma
We previously showed that HEV-like vessels are formed in human gastric mucosa infected with H. pylori (Kobayashi et al. 2004). We also showed that those vessels characteristically express 6-sulfo sialyl Lewis × as detected by NCC-ST-439, MECA-79, and HECA-452 antibodies. Indeed, HEV-like vessels were detected by MECA-79 antibody in mouse gastric mucosa infected by H. felis (Figure 1F). However, these HEV-like vessels showed very little staining with HECA-452 or NCC-ST-439 antibodies. Instead, we found that gastric carcinomas were robustly stained by NCC-ST-439 and HECA-452 antibodies but not by MECA-79 antibody (Figures 2K-2N). It has been shown that NCC-ST-439 binds 6-sulfo sialyl Lewis × and non-sulfated sialyl Lewis × on core 2 branched O-glycans (Kumamoto et al. 1998; Kobayashi et al. 2004), whereas MECA-79 recognizes 6-sulfo N-acetyllactosamine attached to extended core 1 O-glycans (Yeh et al. 2001). HECA-452, on the other hand, recognizes sialyl Lewis × and sialyl Lewis a with or without 6-sulfate groups in O- and N-glycans (Berg et al. 1991; Kobayashi et al. 2004). Considering the carbohydrate epitopes recognized by these anti-carbohydrate antibodies, these results indicate that gastric carcinomas express 6-sulfated or non-sulfated sialyl Lewis × on core 2 branched O-glycans because they are positive for both NCC-ST-439 and HECA-452. 6-Sulfo sialyl Lewis X on extended core 1 O-glycans may be negligible because MECA-79 did not stain these gastric carcinomas.
A Distinctive Set of Genes Is Upregulated in Infected Stomach as Determined by Gene Microarrays
To determine if a characteristic expression profile of genes was observed in H. felis-induced inflammation and gastric adenocarcinoma formation, we isolated RNA from the stomach of INS-GAS mice at different time points following infection with H. felis. The expression profile of various genes in the infected stomach during the course of infection was analyzed using gene microarray. Consequently, ANOVA analysis identified 359/925 mouse genes on the chip that were significantly changed at a 0.05 p value level in expression at any time point (Figure 3). Gene expressions were grouped by hierarchical clustering algorithms to identify a gene expression profile characterizing disease progression as described previously (Mueller et al. 2003). In particular, we were interested in identifying genes upregulated in H. felis-infected mice (Figure 3, right panel). Profiles of all 170 significant genes that were upregulated were plotted against their matched controls, and 16 genes were identified from a total of 925 mouse genes that increased in a time-dependent profile (Table 1). Significantly, most upregulated genes were involved in inflammation. Among them, chemokine ENA-78 (CXCL5) was shown to attract neutrophils (Walz et al. 1991), and its expression level increases but does not change during the progression of H. pylori-infected patients (Rieder et al. 2001). CXCL13, also upregulated following infection, attracts B cells (Forster et al. 1996; Ansel et al. 2002) and plays a role in formation of Peyer's patches (Finke et al. 2002) (Table 1). The chemokine MIP-3α, which is expressed in the intestine under physiological conditions and attracts memory T cells, was also upregulated (Tanaka et al. 1999; Kunkel et al. 2003). At the same time, the chemokine MCP-2 and the chemokine receptor CXCR4 (Roth et al. 1995; Taub et al. 1995) were upregulated in the same analysis. CXCR4 is expressed in B cells and plays a role in B-cell adhesion to HEVs in the secondary lymphoid organs including Peyer's patches (Herbein et al. 1998; Okada et al. 2002). Upregulation of the chemokine receptor CXCR4 is noteworthy because this receptor facilitates HIV entry into CD4+ T cells (Herbein et al. 1998). Although there is no report on an increase of HIV-infected patients among H. pylori-infected individuals, further studies are worth pursuing to determine if such a correlation exists.
Upregulation of α2,6-sialyltransferase (Weinstein et al. 1987), which is required for formation of the ligand for CD22 (Sgroi et al. 1993), was detected. This increase is likely due to an increased B-cell recruitment because CD22 is mainly present on B cells. Upregulation of BDCA2/CLECSF11 indicates an increase in dendritic cells in lymphoid and non-lymphoid tissues (Dzionek et al. 2001). Sergycin, which is present in mast cell granules (Grujic et al. 2005), was upregulated during a 4- and 6-month period after infection, suggesting involvement of mast cells in H. felis-induced inflammation. In agreement, Nakajima et al. (1997) showed that mast cells might play an important role in the pathogenesis of H. pylori-associated gastritis and peptic ulcer in the human stomach. VCAM-1 on endothelial cells likely facilitates recruitment of leukocytes to inflammatory sites and enhances ongoing inflammatory processes (Nakache et al. 1989). N-acetylglucosaminyltransferase-IVa (GlcNAcT-IVa) forms tri-antennary N-glycans. It was recently discovered that gene inactivation or downregulation of GlcNAcT-IVa leads to diabetes mellitus (Ohtsubo et al. 2005).
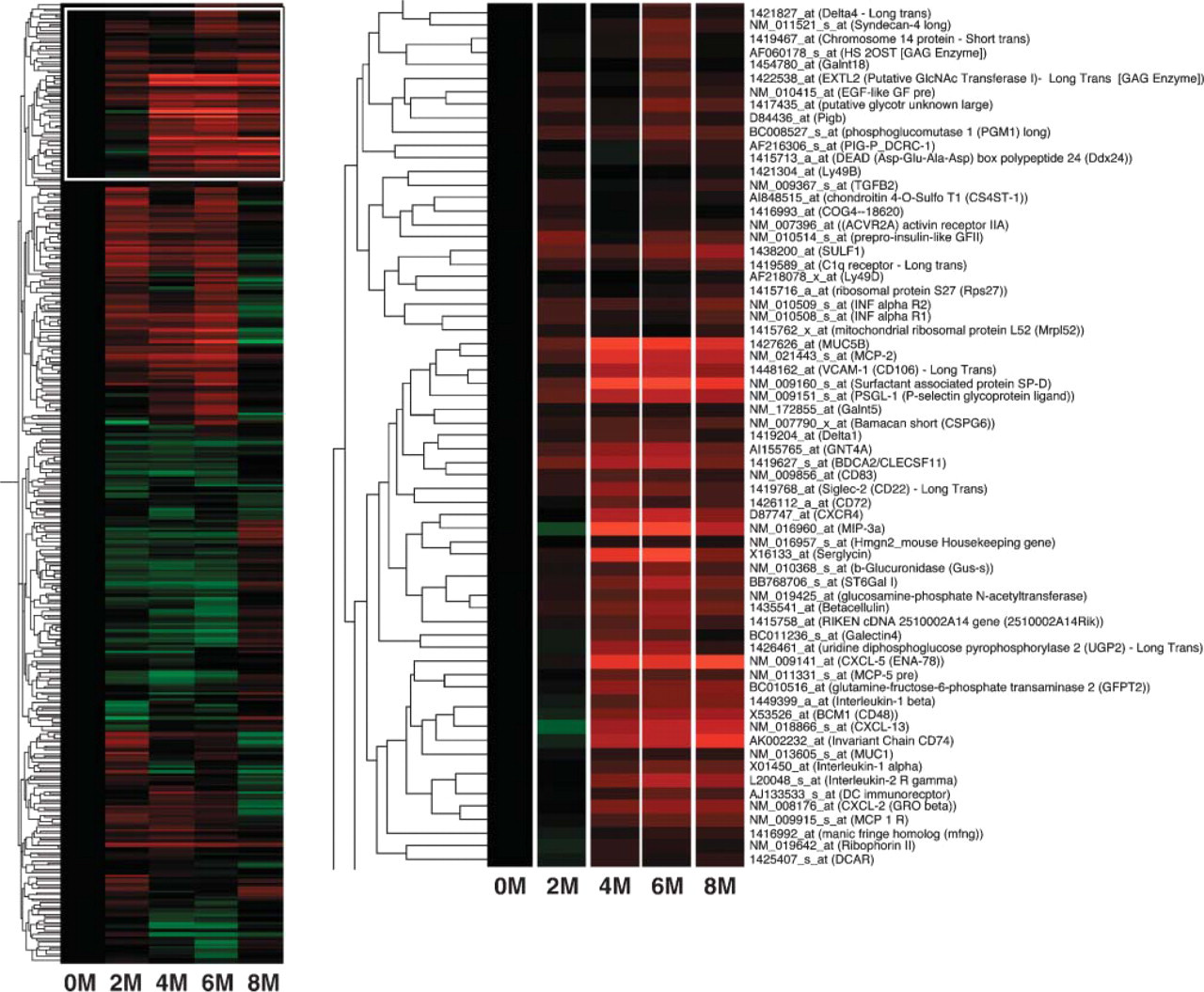
Hierarchical clustered expression profile of upregulated genes in H. felis-infected mouse stomachs. Heat map represents the average of three to five infected mice at each time point. Left panel contains an overview of 359 genes significant for differential regulation in infected vs uninfected animals. Red and green represent high and low ratios for the infected sample/uninfected control samples, respectively. Right panel is the enlarged figure white-boxed at left. These represent 65 upregulated genes compared with uninfected controls. The strongest red to weakest red corresponds to a scaled 5- to 2-fold increase normalized to the control-uninfected mouse, 2 to 8 months after infection.
Significantly, many upregulated genes have been reported to be relevant to H. pylori infection including SP-D, CD74, and MUC5B (Namavar et al. 1998) (Table 1). Upregulated expression of PSGL-1 (Moore et al. 1992) and IL-2R (Basso et al. 1996) may also be indicative of recruitment of immune cells to inflammatory-carcinoma sites.
Minimum Changes in Glycosyltransferase Gene Transcripts as Assessed by RT-PCR
The above results show that inflammation and carcinoma in infected gastric mucosa are correlated with formation of HEV-like vessels and expression of unique carcinoma-associated carbohydrate antigens, respectively. Formation of carbohydrate structures recognized by MECA-79, NCC-ST-439, and HECA-452 antibodies requires the sulfotransferase GlcNAc6ST-2 (LSST), FucT-VII, and Core2GlcNAcTs or Core1-β3GlcNAcT (Yeh et al. 2001; Kobayashi et al. 2004). Gene microarray analysis, however, revealed minimal increases in expression of these enzymes over control mice.
Upregulated genes revealed by gene microarray analysis a
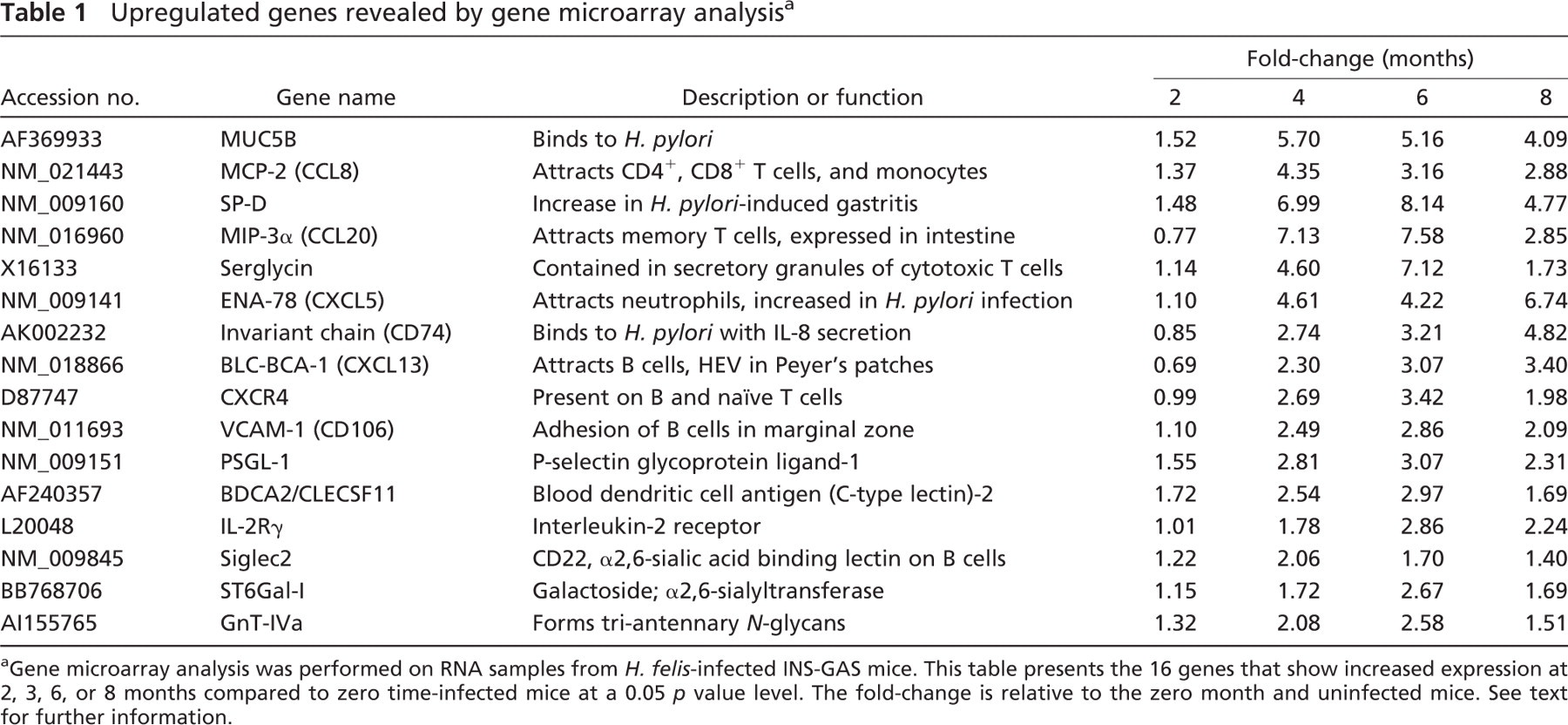
Gene microarray analysis was performed on RNA samples from H. felis-infected INS-GAS mice. This table presents the 16 genes that show increased expression at 2, 3, 6, or 8 months compared to zero time-infected mice at a 0.05 p value level. The fold-change is relative to the zero month and uninfected mice. See text for further information.
To determine the fidelity of gene microarray analysis and to obtain additional information on these carbohydrate-synthesizing enzymes, we carried out semiquantitative RT-PCR on Core1-β3GlcNAcT, Core2GlcNAcT-1, Core2GlcNAcT-2, GlcNAc6ST-2, and FucT-VII using the same total RNA samples used for gene microarray analysis as templates (Figure 4A). Among the transcripts analyzed, we found that Core2GlcNAcT-2 (mucin-type) (Yeh et al. 1999) is most abundant, followed by Core1-β3GlcNAcT, GlcNAc6ST-2, Core2GlcNAcT-1, and FucT-VII. Core2GlcNAcT-3 (Schwientek et al. 2000) was not detected. None of these genes showed substantial upregulation during disease progression (Figure 4B).
As a positive control, MIP-3α transcripts were significantly increased over control uninfected mice, confirming the results obtained by gene microarray analysis. Together these results indicate that global expression of the glycosyltransferases and sulfotransferase analyzed does not change as inflammation progresses, whereas the expression of the chemokine MIP-3α substantially increases during H. felis-induced inflammation.
Detection of CD74 and SP-D in Inflammatory Gastric Mucosa and Gastric Adenocarcinoma
Among upregulated genes, SP-D and CD74 increased substantially in gene microarray analysis. We thus explored whether expression of CD74 and SP-D reflects the progression of gastric disease. CD74 was immunohistochemically detected in regions colonized by H. felis (Figures 5B and 5C). SP-D expression was much higher in the surface mucous gel layer and lower part of the gastric mucosa than in the upper part of the gastric mucosa (Figure 5D). CD74 and SP-D showed very little staining in uninfected gastric mucosa (data not shown), indicating that CD74 and SP-D are upregulated in the region infected by H. felis.
Expression of CD74 and SP-D was also observed in gastric carcinoma (Figures 5E and 5F). CD74 was also seen in B cells (Figures 5G and 5H) as expected, consistent with a report that H. pylori infection recruits more B cells than T cells (Bayerdorffer et al. 1995). These results combined indicate that CD74 is upregulated where H. felis colonizes and at inflammatory sites and in adenocarcinoma throughout the disease. On the other hand, SP-D is upregulated at H. felis colonization sites but only weakly in gastric adenocarcinoma. These results as a whole indicate that a distinctive set of genes is upregulated during inflammation-carcinoma sequence of INS-GAS mice after infection with H. felis.
Discussion
This study demonstrates that H. felis sequentially induces inflammation and adenocarcinoma formation in the stomach of INS-GAS mice, and that progression of these diseases is closely associated with upregulation of a distinctive set of genes. Although our analysis was limited to 925 genes, results indicate that a distinctive set of genes is upregulated in a temporally specific manner (Table 1). INS-GAS mice infected with H. felis developed PNAd-expressing HEV-like vessels. Moreover, invasive adenocarcinomas to the submucosa and muscularis propria, as well as vessel invasion, were seen in the advanced stage of disease. In contrast, uninfected INS-GAS mice displayed negligible numbers of HEV-like vessels, and only a few non-invasive carcinomas were formed, even 8 months after the start of the experiment. These results clearly indicate that H. felis infection increases the inflammatory response from the host animal and leads to a highly advanced invasive or metastatic carcinoma, thus establishing sequential gene expression during the inflammation-carcinoma sequence in H. felis-induced carcinogenesis.
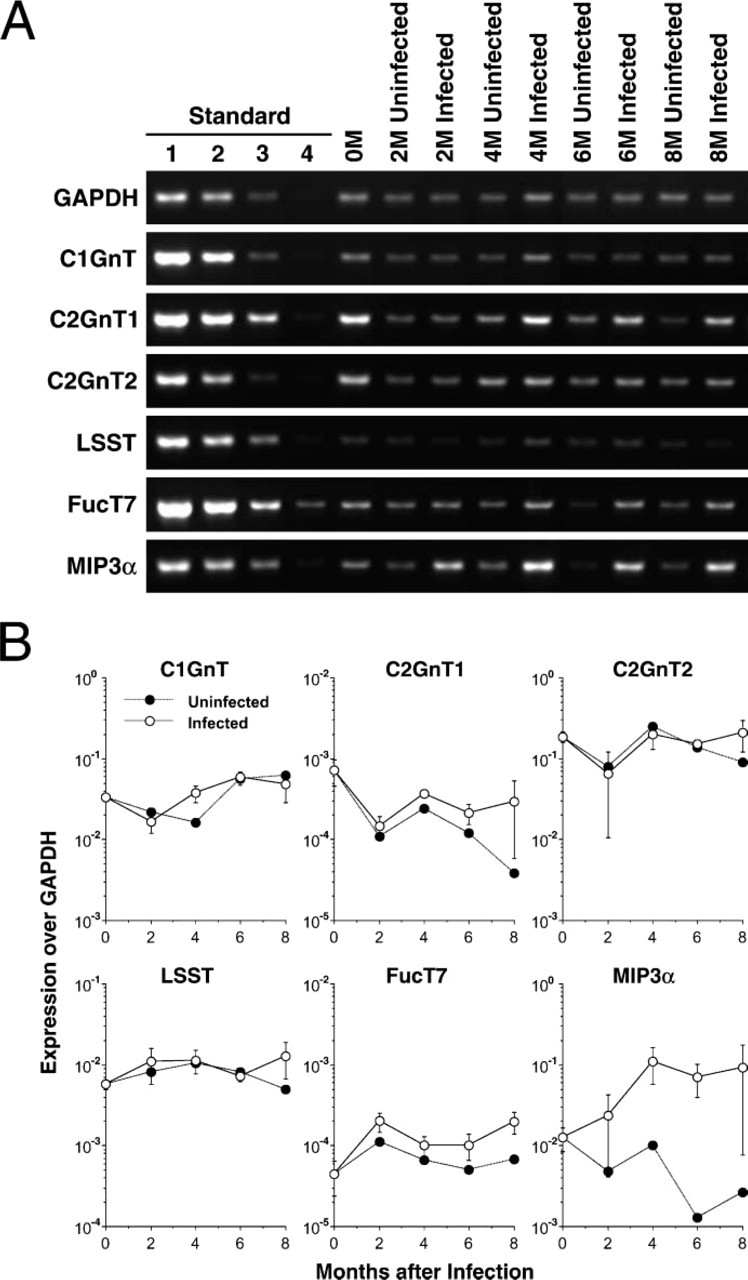
Estimation of the amount of different gene transcripts by semiquantitative RT-PCR. (
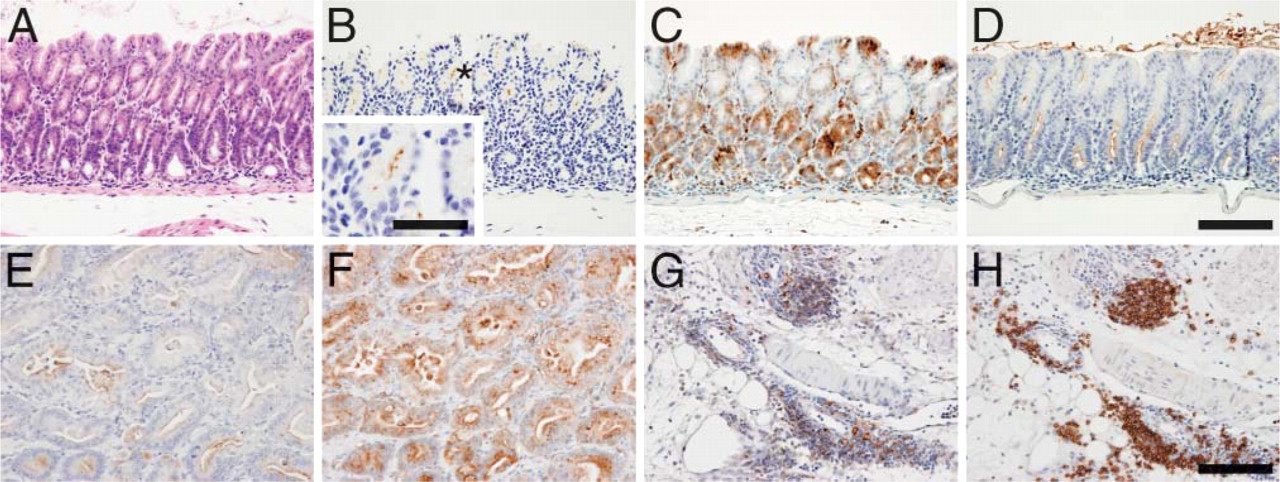
Expression of CD74, SP-D, and unique carbohydrate antigens associated with H. felis infection and resultant carcinoma. (
Gene microarray analysis revealed several important features about H. felis-induced inflammation and carcinoma. Among genes upregulated in response to the infection, CD74 and ENA-78 expression were apparently elevated throughout the disease process, whereas the majority of other upregulated genes are decreased at the 8-month point compared with 4- and 6-month points. CD74, also called invariant chain (Ii) protein, is a chaperone molecule that regulates antigen presentation of class II MHC molecules by inhibiting binding to endogenously derived antigenic peptides (Cresswell 1996). Overexpression of CD74 may then lead to decreased recognition of cancer cells by immune cells, thus increasing survival of cancer cells. In fact, the MHC class II-associated Ii protein was upregulated in chemically induced rat gastric cancer (Furihata et al. 1997). Our results are relevant to a report that H. pylori binds to CD74 expressed on gastric epithelial cells (Beswick et al. 2005). It was shown that H. pylori inhabits the mucous layer close to the epithelium but does not bind directly to the epithelium (Schreiber et al. 1999). On the other hand, it was reported that upregulation and secretion of ENA-78 (CXCL5) require attachment of H. pylori to epithelial cells (Rieder et al. 2001), suggesting that CD74 may function in both H. felis and H. pylori adhesion to gastric epithelial cells and inhibition of immune cell attack on gastric carcinoma cells in cancer progression. It is also noteworthy that CD74 was shown to play a role in pro-inflammatory signals and as a receptor for MIF, which is reported to have a pro-carcinogenic effect on gastric epithelial cells (Leng et al. 2003). These results as a whole indicate that increased expression of CD74 and ENA-78 after H. pylori infection likely plays a critical role in the progression of inflammation, and then in carcinogenesis.
SP-D is highly upregulated during a 4- and 6-month period. SP-D contains a C-type lectin domain and belongs to a family of proteins implicated in innate immunity, termed collectins (Persson et al. 1990). SP-D is thought to bind directly to lipopolysaccharide on Gram-negative bacteria such as H. felis, via the carbohydrate recognition domain (Murray et al. 2002). This observation is consistent with our findings that SP-D is found on the surface mucous gel layer as well as in a deeper part of the gastric mucosa colonized by H. felis. SP-D may inhibit progression of this disease process because binding by SP-D may result in H. felis aggregation, leading to enhanced killing and phagocytosis by neutrophils and macrophages.
It is noteworthy that MAdCAM-1-expressing HEV-like vessels were found in the lamina propria, whereas MECA-79-positive HEV-like vessels were more prevalent in the submucosa. Because the inflammatory response is thought to progress from the surface to a deeper part of the mucosa, MAdCAM-1-expressing and MECA-79-positive HEV-like vessels may represent HEVs in earlier and later inflammatory responses, respectively. This hypothesis is consistent with previous findings that HEVs expressing MAdCAM-1 are formed first, and those HEVs are gradually replaced by MECA-79-positive HEVs during ontogenesis (Mebius et al. 1996).
Although the inflammatory response induced by H. felis infection was associated with extensive lymphocyte recruitment, relatively fewer HEV-like vessels were detected in the animal model than in humans infected with H. pylori (Kobayashi et al. 2004). This may reflect a different time course for evolving diseases because humans usually develop disease after childhood, whereas inflammation takes place in mice <1 year after inoculation of H. felis. Because relatively few HEV-like vessels are formed in the mouse model, the expression profile of key glycosyltransferases that form selectin ligands did not show obvious upregulation. Our attempts to isolate HEV-like vessels by laser capture microdissection failed because the isolation of intact RNA was not possible after histological staining, which is necessary to identify HEV-like vessels. Additionally, gene transcripts of key enzymes that function in sialyl Lewis X biosynthesis did not show apparent upregulation despite the fact that gastric carcinomas were stained by NCC-ST-439 and HECA-452. It is possible that basal expression level of these enzymes might be enough to synthesize epitopes of those antibodies.
Recently, it has been reported that MECA-79 antibody has a significant therapeutic effect in a sheep model of asthma (Rosen et al. 2005). We also reported that inactivation of both GlcNAc6ST-1 and GlcNAc6ST-2 (LSST) results in loss of MECA-79 antigen in the lymph nodes and impaired contact hypersensitivity (Kawashima et al. 2005). Further studies are important to determine whether inhibition of L-selectin ligand synthesis in the Helicobacter-infected stomach leads to inhibition of H. felis- as well as H. pylori-induced chronic inflammation, thus preventing gastric carcinoma formation. The mouse model used here is likely applicable to those studies as well.
In conclusion, we have shown in this study that a set of genes is upregulated in H. felis-infected INS-GAS mice, and the majority of these upregulated genes is associated with inflammatory response. We also found that CD74 and possibly ENA-78 are indicative of gastric carcinoma formation, whereas SP-D is indicative of hyperplasia and early carcinoma. Because we expect that a similar set of genes is upregulated in H. pylori-infected humans, the expressions of CD74, ENA-78, and SP-D are likely useful markers for disease progression in H. pylori-infected patients.
Footnotes
Acknowledgements
This work was supported by Grants CA-33000 and CA-71932 (to MF) from the National Institutes of Health and by Grants-in-Aid for Scientific Research on Priority Area 14082201 from the Ministry of Education, Culture, Sports, Science and Technology of Japan (to JN). The Consortium for Functional Glycomics was supported by Grant GM-62116 from the National Institutes of Health.
The authors thank Drs. Junya Mitoma, Hiroto Kawashima, Michiko Fukuda, James Paulson, and Pradman Qasba for useful discussion. We also thank Ms. Akiko Ishida and Ms. Matsuko Watanabe for preparing tissue sections, Ms. Aleli Morse for organizing the manuscript, and Dr. Elise Lamar for critical reading of the manuscript.
