Abstract
Lichen planus (LP) is a chronic inflammatory mucocutaneous disease involving the oral mucosa and skin. Both oral LP (OLP) and cutaneous LP (CLP) are histopathologically characterized by dense subepithelial lymphocyte infiltrates; however, the mechanisms underlying lymphocyte recruitment to sites of LP lesions are not fully understood. Here, we assessed the induction of peripheral lymph node addressin (PNAd)-expressing high endothelial venule (HEV)-like vessels in 19 OLP and 17 CLP cases. To do so, we performed immunohistochemical staining for PNAd and CD34, followed by quantitative analysis. We also conducted triple immunohistochemistry for PNAd and either CD3 and CD20 or CD4 and CD8 to identify the lymphocyte subset preferentially recruited via HEV-like vessels. PNAd-expressing HEV-like vessels were induced in and around lymphocyte aggregates in all cases of OLP and in 10 of 17 CLP cases, and these vessels were more frequently observed in OLP relative to CLP. Although the number of T-cells attached per HEV-like vessel exceeded the number of B-cells in both OLP and CLP, the number of CD4+ T-cells attached was greater than the number of CD8+ T-cells only in OLP. These findings combined suggest that PNAd-expressing HEV-like vessels play a more important role in the pathogenesis of OLP compared with CLP.
Keywords
Introduction
Lichen planus (LP) is a chronic mucocutaneous inflammatory disease usually affecting middle-aged women.1,2 Histopathologically, LP is characterized by dense subepithelial band-like lymphocyte infiltration, liquefactive degeneration of the basal layer of stratified squamous epithelium, and keratinocyte apoptosis (Civatte bodies).1 –3 Although these histopathological features are common in oral LP (OLP) and cutaneous LP (CLP), their clinical courses differ. In general, CLP is self-limiting, and the mean duration is 1 to 2 years. 2 OLP, however, is chronic and non-remissive, with a much longer duration than CLP. 4 Such differences in clinical behavior are thought to originate from local differences in either immunopathological mechanisms3,5 or bacterial invasion of tissues 6 ; however, the etiopathogenesis of OLP and CLP is not fully understood.
LP is regarded essentially as an exaggerated T-cell-mediated immune response. It has been shown that lymphocytes infiltrating LP lesions consist mainly of CD4+ and CD8+ T-cells. 7 CD4+ T-cells reportedly infiltrate the lamina propria of oral mucosa in the case of OLP. 8 However, CD8+ T-cells infiltrating the mucosal epithelia of OLP are reportedly associated with disease remission. 9 Although T-cell characteristics and sites of infiltration have been demonstrated in OLP,7 –9 how T cells are recruited to OLP tissues remains unknown.
High endothelial venules (HEVs) present in secondary lymphoid organs play an important role in physiological lymphocyte migration known as lymphocyte “homing.” 10 Lymphocyte homing is tightly regulated by multistep adhesive interactions between lymphocytes and the endothelial cells that comprise HEVs. 11 For the first step, L-selectin on circulating lymphocytes binds loosely to peripheral lymph node addressin (PNAd), a group of glycoproteins decorated with sialyl 6-sulfo Lewis X-capped O-glycans, 12 expressed on the luminal surface of HEVs, resulting in rolling along that surface.13,14 Subsequently, lymphocytes are activated by chemokines, interact with immunoglobulin superfamily cell adhesion molecules on HEVs, such as intercellular adhesion molecule 1 and vascular cell adhesion molecule 1, and then transmigrate across HEVs. 11
Under pathological conditions, vessels morphologically similar to HEVs, which we call HEV-like vessels, are induced in non-lymphoid tissues. We previously demonstrated induction of HEV-like vessels in chronic inflammatory conditions, including chronic Helicobacter pylori gastritis,15 –17 ulcerative colitis,18,19 autoimmune pancreatitis, 20 chronic prostatitis associated with benign prostatic hyperplasia, 21 and eosinophilic chronic rhinosinusitis, 22 as well as in malignant tumors including bladder urothelial carcinoma 23 and oral squamous cell carcinoma. 24 In these studies, we detected PNAd on HEV-like vessels using the monoclonal antibody MECA-79, whose epitope is known to be 6-sulfo N-acetyllactosamine attached to extended core 1 O-glycans,25,26 which overlaps with sialyl 6-sulfo Lewis X, the L-selectin recognition determinant. Here, we performed quantitative immunohistochemical analysis of a total of 36 LP cases (19 OLP and 17 CLP) to assess induction of PNAd-expressing HEV-like vessels. We also conducted triple immunohistochemistry to evaluate which lymphocyte subset is most likely to be recruited via these vessels.
Materials and Methods
Tissue Samples
Formalin-fixed, paraffin-embedded tissue blocks of biopsy specimens of OLP (n=19) and CLP (n=17) were retrieved from the pathology archives of University of Fukui Hospital. Oral mucosa tissues without inflammatory cell infiltrates were used as controls. Analysis of human tissues was approved by the Ethics Committee of the Faculty of Medical Sciences, University of Fukui (Reference No. 20170117, approved on September 19, 2017).
Antibodies
The following antibodies served as primary antibodies: QBEND10 recognizing human CD34 (mouse monoclonal, cat. no. IM0786; Immunotech, Marseille, France), MECA-79 recognizing PNAd (rat monoclonal, cat. no. 553863; BD Pharmingen, San Diego, CA),25,26 rabbit polyclonal anti-human CD3 (cat. no. A0452; Dako, Glostrup, Denmark), L26 recognizing human CD20 (mouse monoclonal, cat. no. M0755; Dako), 1F6 recognizing human CD4 (mouse monoclonal, cat. no. NCL-CD4-1F6; Leica Biosystems, Newcastle Upon Tyne, UK), and C8/144B recognizing human CD8 (mouse monoclonal, cat. no. M7103; Dako).
Immunohistochemistry
Conventional CD34 immunohistochemistry was carried out using the Histofine system (Nichirei; Tokyo, Japan) according to the manufacturer’s instructions. Immunohistochemistry for PNAd was undertaken using an indirect method, as described. 27 Triple immunohistochemistry for PNAd and either CD3 and CD20 or CD4 and CD8 was conducted as described. 28
Quantification of HEV-like Vessels
We first scanned tissue sections immunostained for PNAd at 100× magnification and identified three areas with the highest density of PNAd-positive HEV-like vessels (“hot spots”). PNAd-positive HEV-like vessels in those areas were then counted at 200× magnification. In serial sections immunostained for CD34, the number of CD34-positive vessels in the three areas was also determined, and the PNAd+/CD34+ vessel ratio was calculated in each case.
Quantification of Lymphocyte Subsets
Using tissue sections triply immunostained for PNAd and either CD3 and CD20 or CD4 and CD8, we counted each lymphocyte subset attached to a total of 768 (in OLP) or 78 (in CLP) PNAd-positive HEV-like vessels and calculated the mean number of each lymphocyte subset attached per HEV-like vessel, as described. 18 For the quantification of lymphocyte subsets infiltrated outside the vessels, the number of each lymphocyte subset per high-power field (HPF) at 400× magnification was counted, and the mean number per HPF was determined.
Results
Induction of PNAd-expressing HEV-like Vessels in OLP and CLP
We first examined hematoxylin and eosin-stained tissue sections of OLP (n=19) and CLP (n=17) and found that all cases examined showed histological characteristics of LP. As shown in Fig. 1, we observed subepithelial band-like dense lymphocyte infiltrates, together with liquefactive degeneration of the basal layer of stratified squamous epithelium and occasional keratinocyte apoptosis (Civatte bodies), in both OLP and CLP. In and around these lymphocyte infiltrates, we observed induction of vessels morphologically similar to HEVs in secondary lymphoid organs. Immunohistochemically, a majority of these vessels were positive for both PNAd and the vascular endothelial marker CD34 (Fig. 2). It is of note that such PNAd-positive HEV-like vessels were not observed in intact oral mucosa.
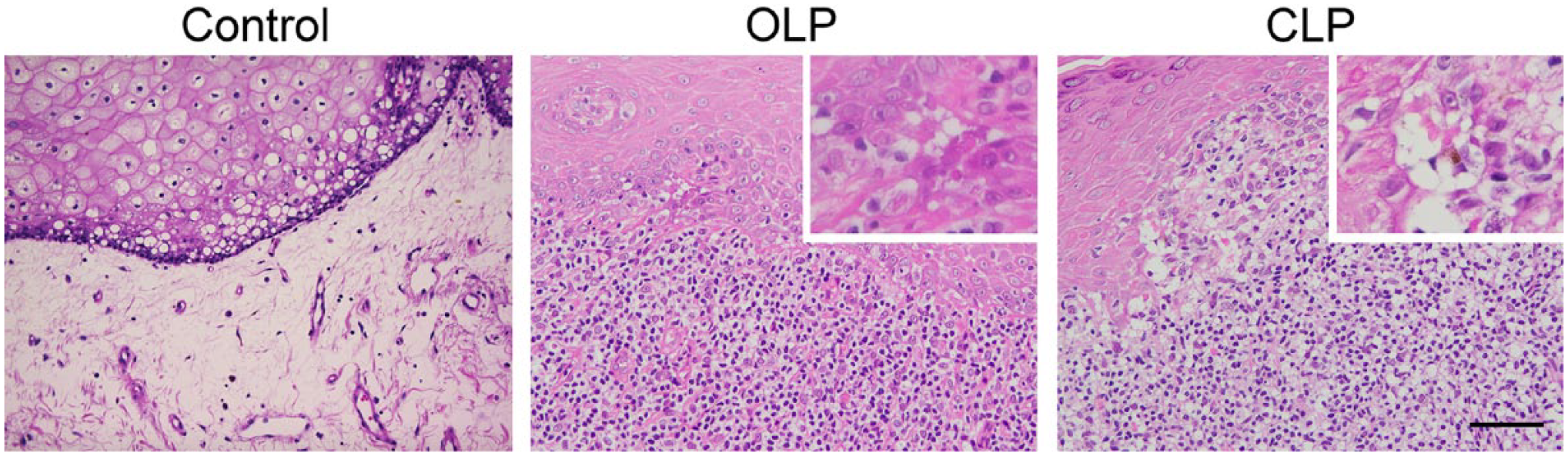
Histopathology of OLP (middle panel) and CLP (right panel). Intact oral mucosa tissues served as controls (left panel). Note that subepithelial band-like lymphocyte infiltrates are common in both types of LP. Insets indicate keratinocyte apoptosis (Civatte bodies). Scale bar = 100 µm for main pictures and 20 µm for insets. Abbreviations: LP = lichen planus; OLP = oral LP; CLP = cutaneous LP.
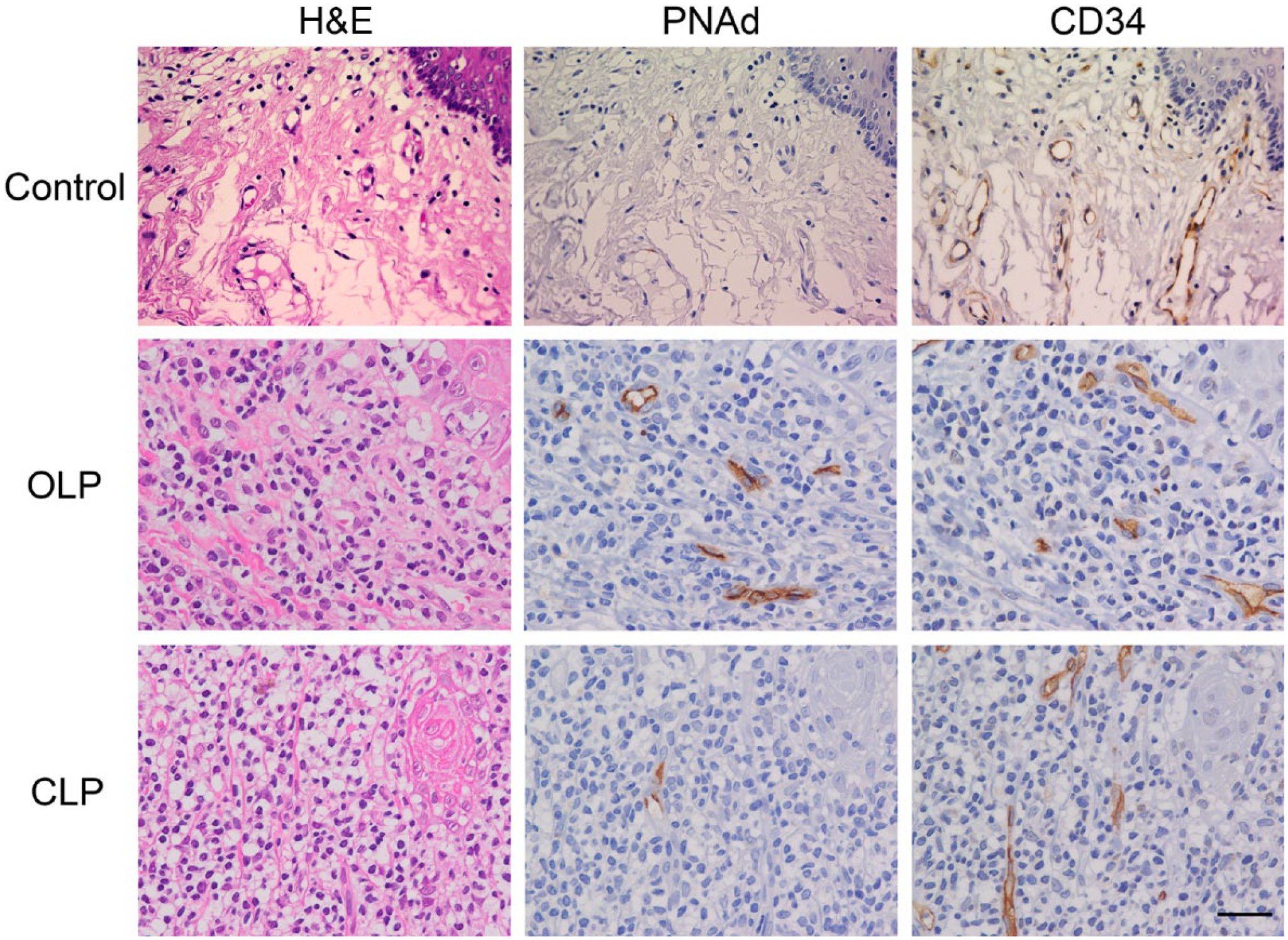
Induction of HEV-like vessels in OLP and CLP. Serial tissue sections were stained with H&E and immunostained for CD34 and PNAd. HEV-like vessels positive for PNAd are seen in and around lymphocyte aggregates formed in OLP and CLP. These HEV-like vessels are not observed in intact oral mucosa. Scale bar = 50 µm. Abbreviations: H&E = hematoxylin and eosin; HEV = high endothelial venule; LP = lichen planus; OLP = oral LP; CLP = cutaneous LP; PNAd = peripheral lymph node addressin.
Preferential Induction of PNAd-expressing HEV-like Vessels in OLP Relative to CLP
Although PNAd-expressing HEV-like vessels were induced in all OLP cases, such vessels were induced in only 10 of 17 CLP cases, suggesting that PNAd-expressing HEV-like vessels are preferentially induced in OLP relative to CLP. To confirm this hypothesis, we calculated the frequency of PNAd-expressing HEV-like vessels, namely the PNAd+/CD34+ vessel ratio, in each of 36 LP cases and compared that ratio between OLP and CLP. As shown in Fig. 3, the PNAd+/CD34+ vessel ratio in OLP was significantly greater than that seen in CLP (0.28 ± 0.07 vs 0.05 ± 0.07).
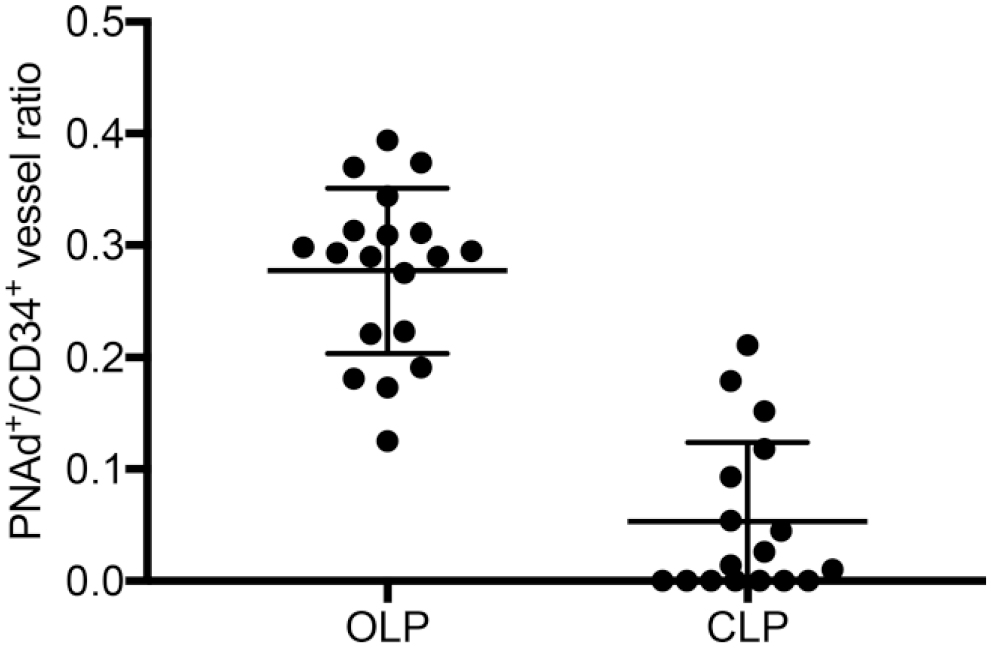
Differences in the PNAd+/CD34+ vessel ratio in OLP (n=19) and CLP (n=17). The PNAd+/CD34+ vessel ratio in OLP significantly exceeds that seen in CLP. Data are presented as means with SD. Abbreviations: LP = lichen planus; OLP = oral LP; CLP = cutaneous LP.
Close Association of T-cells With PNAd-expressing HEV-like Vessels in OLP
To identify the lymphocyte subset predominantly recruited via HEV-like vessels in OLP and CLP, we performed triple immunohistochemical staining for PNAd, CD3, and CD20. As shown in Fig. 4A, both T- and B-cells were attached to the apical aspect of endothelial cells that constitute HEV-like vessels. Moreover, the number of T-cells attached per HEV-like vessel was significantly greater than that of B-cells in both OLP (0.40 ± 0.81 vs 0.07 ± 0.32) (Fig. 4B, left panel) and CLP (0.37 ± 0.81 vs 0.04 ± 0.20) (Fig. 4B, right panel). We also carried out triple immunostaining for PNAd, CD4, and CD8 to determine the T-cell subpopulation preferentially recruited via HEV-like vessels. As shown in Fig. 5A, we observed both CD4+ and CD8+ T cells attached to the apical aspect of HEV-like vessels. The number of CD4+ T cells attached per HEV-like vessel in OLP was significantly greater than that of CD8+ T cells (0.31 ± 0.61 vs 0.08 ± 0.30) (Fig. 5B, left panel). However, in CLP, the number of CD4+ T cells attached per HEV-like vessel and that of CD8+ T cells did not differ significantly (0.19 ± 0.54 vs 0.12 ± 0.37) (Fig. 5B, right panel).
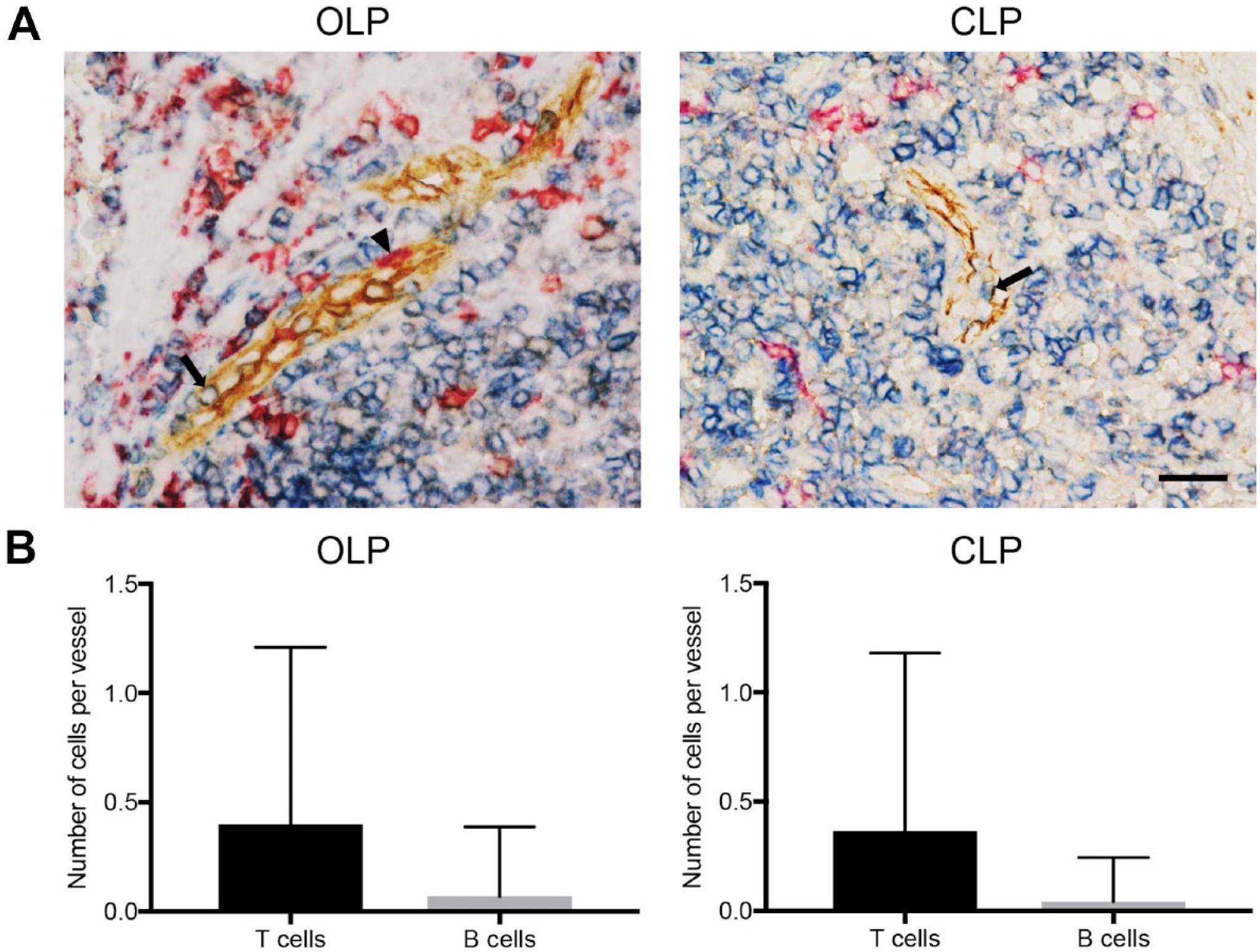
Lymphocyte subsets attached to the luminal aspect of HEV-like vessels induced in OLP and CLP. (A) Triple immunohistochemistry for PNAd (brown), CD3 (blue), and CD20 (red). Both T-cells (arrow) and B-cells (arrowhead) attach to the luminal aspect of HEV-like vessels. Scale bar = 50 µm. (B) The number of respective T and B cells attached per HEV-like vessel. Data are presented as means with SD. Abbreviations: HEV = high endothelial venule; LP = lichen planus; OLP = oral LP; CLP = cutaneous LP; PNAd = peripheral lymph node addressin.
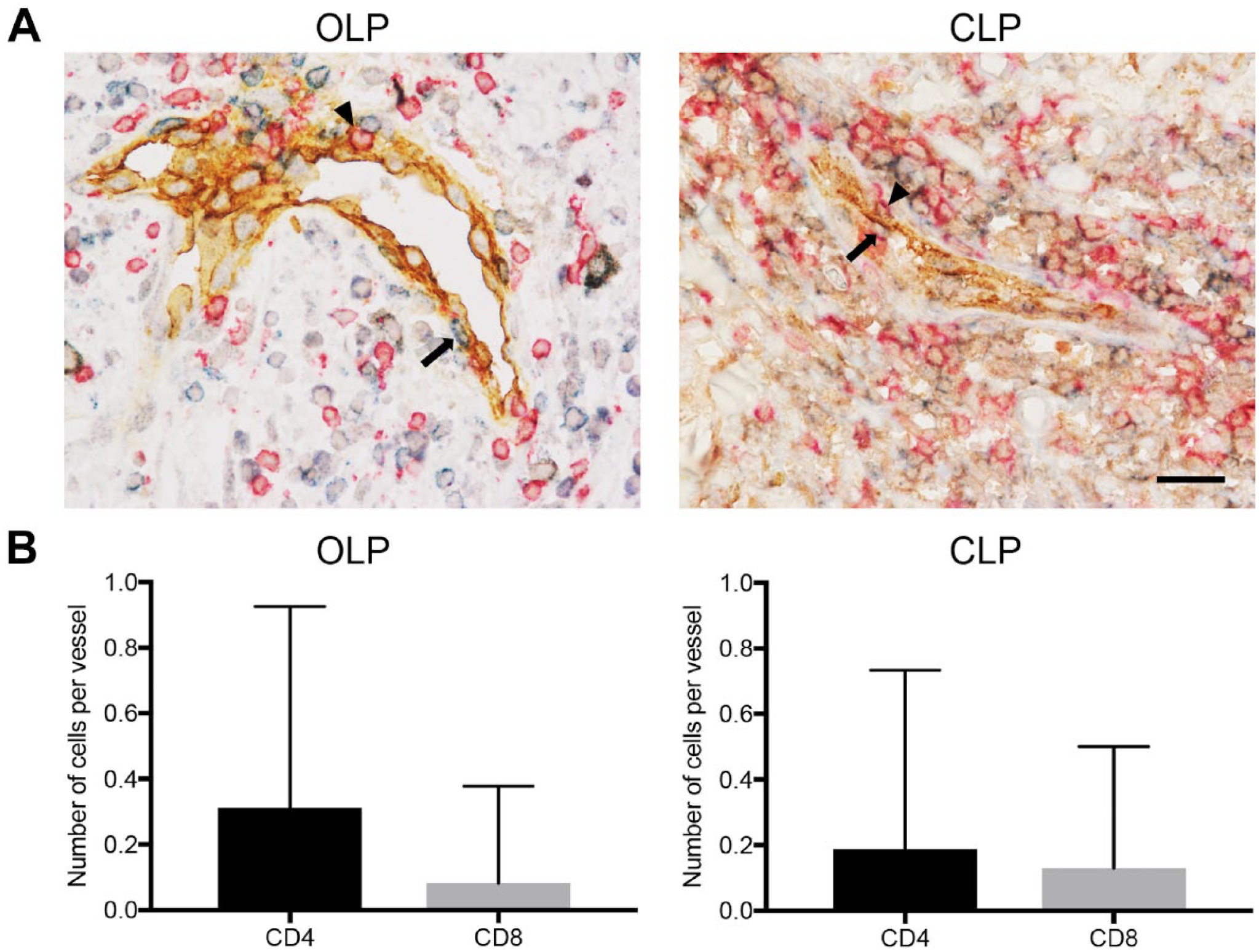
T-cell subsets attached to the luminal aspect of HEV-like vessels induced in OLP and CLP. (A) Triple immunohistochemistry for PNAd (brown), CD4 (blue), and CD8 (red). Both CD4+ (arrow) and CD8+ T cells (arrowhead) attach to the luminal aspect of HEV-like vessels. Scale bar = 50 µm. (B) The number of respective CD4+ and CD8+ T cells attached per HEV-like vessel. Data are presented as means with SD. Abbreviations: HEV = high endothelial venule; LP = lichen planus; OLP = oral LP; CLP = cutaneous LP; PNAd = peripheral lymph node addressin.
However, the number of T cells infiltrated outside the HEV-like vessels was greater than that of B cells in both OLP and CLP (119.0 ± 18.1 vs 20.1 ± 6.2 and 107.2 ± 19.5 vs 12.4 ± 7.9, respectively). Among the T cell population, in OLP, the number of CD4+ T cells infiltrated outside the HEV-like vessels was greater than that of CD8+ T cells (82.0 ± 10.7 vs 36.6 ± 8.3); however, in CLP, these two numbers did not differ significantly (32.3 ± 8.7 vs 33.2 ± 7.0).
Discussion
Here, we show that although PNAd-expressing HEV-like vessels are induced in both OLP and CLP, HEV-like vessels are more frequently observed in OLP than in CLP. In our previous studies, we observed HEV-like vessels, as detected by positive staining for PNAd, in and around lymphocyte aggregates formed in chronic inflammatory settings such as chronic H. pylori gastritis,15 –17 ulcerative colitis,18,19 autoimmune pancreatitis, 20 chronic prostatitis associated with benign prostatic hyperplasia, 21 and eosinophilic chronic rhinosinusitis. 22 All of these studies suggest that such vessels function in lymphocyte recruitment to sites of chronic inflammation. Thus, subepithelial band-like dense lymphocytic infiltrates seen commonly in both OLP and CLP are likely recruited by HEV-like vessels as well.
Although CLP is self-limiting, OLP is chronic and rarely undergoes spontaneous remission.1,4 It is well known that the oral mucosa is exposed to antigens originating from bacteria, viruses, or fungi. Bacteria such as periodontopathogenic microorganisms or Mycoplasma salivarium are more frequently detected in OLP than in non-OLP patients.30,31 Choi et al. 6 reported that bacteria invade the epithelium and lamina propria of OLP tissues and that such activity is significantly correlated with infiltration of CD3+, CD4+, and CD8+ T cells. Thus, it is reasonable to conclude that HEV-like vessels induced in OLP could be the portal of entry for these T cells.
In this study, we found that the number of T cells attached to the luminal aspect of PNAd-expressing HEV-like vessels was significantly greater than that of B cells in both OLP and CLP, and that the number of CD4+ T cells attached exceeded that of CD8+ T cells in OLP but not in CLP. We also found that the composition of lymphocytes infiltrated outside the vessels was essentially identical to that of lymphocytes attached to the apical aspect of HEV-like vessels in both OLP and CLP. In our previous studies, the number of T cells, particularly CD4+ T cells, attached per PNAd-expressing HEV-like vessel was significantly greater than that of B-cells in ulcerative colitis. 16 Furthermore, levels of CD4+ T cell infiltration are reportedly higher than those of CD8+ T cells in OLP.6,32 These observations strongly support our hypothesis that CD4+ T cells in OLP are likely recruited via PNAd-expressing HEV-like vessels. In addition, the chemokine CCL21 is reportedly expressed in endothelial cells of LP.33,34 CCL21 and CCL19 are expressed on the luminal surface of HEVs, and both bind to CCR7 presented on T cells.35,36 Thus, it is plausible that CD4+ T cells recruited via PNAd-expressing HEV-like vessels are activated by interaction of CCL21 with its cognate receptor CCR7.
In addition to PNAd (defined as 6-sulfo N-acetyllactosamine attached to extended core 1 O-glycans), a fraction of HEVs and HEV-like vessels express other carbohydrate structures recognized by a battery of anti-carbohydrate antibodies.15,16,18,27 –29,37 –39 Among these structures, 6-sulfo and non-sulfated sialyl Lewis X are recognized by the monoclonal antibody HECA-452,37,38 6-sulfo and non-sulfated sialyl Lewis X attached to core 2-branched O-glycans are recognized by the monoclonal antibody NCC-ST-439,15,16 and terminal N-acetyllactosamine with Gal-6-O-sulfation is recognized by our recently developed monoclonal antibody 297-11A. 39 Thus, the overall expression patterns of these carbohydrate structures on HEV-like vessels may differ between OLP and CLP.
Indeed, we previously showed that HEV-like vessels induced in gastric mucosa-associated lymphoid tissue (MALT) lymphoma exhibit different carbohydrate structures than those seen in its precursor lesion chronic H. pylori gastritis. 16 Non-sulfated sialyl Lewis X attached to core 2-branched O-glycans is not expressed in chronic H. pylori gastritis but is preferentially expressed in gastric MALT lymphoma, and this type of unsulfated glycan functions as an L-selectin ligand. 16 This finding is consistent with previous reports demonstrating that elimination of the two GlcNAc-6-O-sulfotransferases (GlcNAc6STs) responsible for L-selectin ligand biosynthesis in HEVs, namely, GlcNAc6ST-1 and GlcNAc6ST-2, resulted in partial (~75%) but not complete abolition of lymphocyte homing to peripheral lymph nodes.13,14 Thus, it is plausible that similar mechanisms may operate in HEV-like vessels in CLP. Further studies are, however, required to support this hypothesis.
In conclusion, PNAd-expressing HEV-like vessels are frequently observed in OLP relative to CLP. The number of T cells, particularly CD4+ T cells, attached to these vessels was significantly greater than that of B cells in OLP. These findings combined suggest that PNAd-expressing HEV-like vessels play an important role in the pathogenesis of OLP relative to CLP.
Footnotes
Acknowledgements
We thank Mr Hisataka Kato for technical assistance and Dr Elise Lamar for critical reading of the manuscript.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this article as follows: HYoshida designed and performed the research, analyzed the data, and wrote the manuscript; YI and HYoshimura performed the research; and MK conceived of and designed the research, analyzed the data, and wrote the manuscript. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by a Grant-in-Aid for Young Scientists 18K17196 (to H.Yoshida) and a Grant-in-Aid for Scientific Research 15K08343 (to M.K.) from the Japan Society for the Promotion of Science.
