Abstract
Airway epithelial-derived nitric oxide (NO), through the activation of nucleotide cyclases and downstream kinases, stimulates ciliary beating, yet the precise locations of these enzymes are unknown. We hypothesized that these NO-activated enzymes are located within, or adjacent to, the ciliary axoneme. Immunohistochemistry of intact ciliated cells revealed that endothelial-type nitric oxide synthase (eNOS), the RII isoform of the cAMP-dependent protein kinase (PKA-RII), the type I isoform of the cGMP-dependent protein kinase (PKG-I), and guanylate cyclase β (GC-β) all colocalized with pericentrin to the basal body. In contrast, the PKA-RI isoform and the PKG-II isoform localized to ciliary axonemes. Western blot analysis of isolated demembranated ciliary preparations detected eNOS, GC-β, and both isoforms of PKA and PKG. An A-kinase-anchoring protein was also detected. Our findings suggest that these enzymes are sequestered close to their points of action into a discrete ciliary metabolon, enabling targeted phosphorylation and efficient upregulation of ciliary beating.
T
CBF can be stimulated through a variety of signaling mechanisms. Studies from our laboratory have shown that nitric oxide (NO), cAMP, and cyclic guanosine monophosphate (cGMP) are important second messengers capable of stimulating ciliary motility through the activation of downstream cAMP- and cGMP-dependent kinases (Jain et al. 1993; Sisson 1995; Wyatt et al. 1998b,2004a; Sisson et al. 1999). We have previously shown that activation of the cAMP-dependent protein kinase (PKA) increased CBF in ciliated bovine bronchial epithelial cells (BBECs) (Wyatt et al. 1998a). There are two different isoforms of PKA based on differences in the regulatory subunits, named RI and RII (Taylor et al. 2004). The cGMP-dependent protein kinase (PKG), which can exist in two major isoforms designated PKG-I and PKG-II (Hofmann 2005), is also involved in stimulation of CBF (Wyatt et al. 1998a). Biologically relevant concentrations of ethanol increase CBF via a dual-kinase activation pathway that requires the sequential activation of PKG followed by PKA (Sisson et al. 1999). Other studies have demonstrated that there is cross-talk between the PKA and PKG ciliary signaling pathways in airway epithelial cells (Wyatt et al. 2003).
NO, cyclases, cyclic nucleotides, and kinases are involved in the signal transduction pathway(s) of CBF stimulation, yet little is known about the location of these regulatory enzymes. We hypothesized that the enzymes involved in this cilia stimulatory pathway are located either directly on ciliary axonemes or near the point of ciliary attachment to the cell. in this study we examined the locations and activities of these enzymes on the ciliated epithelium and their association with ciliary A-kinase-anchoring proteins (AKAPs), which could anchor and potentially coordinate the regulatory enzymes on the bovine ciliary axoneme.
Materials and Methods
Cell Isolation
Ciliated bronchial epithelial cells were isolated from bovine bronchi using the method previously described by Sisson et al. (1991). Briefly, the bronchi were removed from the lung and incubated overnight in 0.1% protease in medium 199 (M199). The next day, bronchi were repeatedly washed with M199 containing 10% FBS. The collected cells were centrifuged at 100 X g and the number of ciliated epithelial cells determined using a hemocytometer.
Ciliary Axoneme Isolation
Bovine ciliary axonemes were isolated using a method previously described (Wyatt et al. 2004b). Briefly, bovine trachea were excised, and the excess tissue was removed and washed twice with PBS. Trachea were then washed with extraction buffer (20 mM Tris-HCl, 50 mM NaCl, 10 mM CaCl2, 1 mM EDTA, 7 mM 2-mercaptoethanol, 100 mM Triton X-100, and 1 mM dithiothreitol). One end of the trachea was clamped, 25 ml of extraction buffer was added to the trachea, and the other end of the trachea was then closed. Trachea were vigorously shaken for 90 sec and the resulting mix of extraction buffer and ciliary units were filtered through a 100-μm mesh, centrifuged at 17,250 X g for 7 min, and the supernatant removed. The pelleted ciliary units were resuspended to a concentration of 1 mg/ml in resuspension buffer (20 mM Tris-HCl, 50 mM KCl, 4 mM MgCl2, 0.5 mM EDTA, 1 mM dithiothreitol, 10 mM soybean trypsin inhibitor, and 25% sucrose) and stored at −80C.
SDS-PAGE and Western Blotting
SDS-PAGE was based on the method of Laemmli (1970) using precast 10% Tris-HCl gels (Bio-Rad Laboratories; Hercules, CA). Each well was loaded with 10 μg of ciliary protein. After SDS-PAGE, proteins were transferred to nitrocellulose. Following transfer, the membranes were incubated overnight in wash buffer (0.005 M Tris, 0.15 M NaCl, 0.005% Tween-20, pH 7.5) with 3% BSA. Membranes were incubated with primary antibodies diluted in wash buffer for 1 hr, shaking at room temperature. The membranes were then washed three times with wash buffer followed by incubation with secondary antibodies diluted in wash buffer with 3% BSA for 1 hr at room temperature with shaking. Proteins were identified using SuperSignal West Pico Substrate (Pierce; Rockford, IL) and exposing the membranes to autoradiography film.
Antibodies
Primary antibodies used were as follows: cAMP-dependent PK-RI and PK-RII (BioMol; Plymouth Meeting, PA), cGMP-dependent PK-I and PK-II (generous gift from K. Pryzwansky), guanylate cyclase β (Cayman Chemical; Ann Arbor, MI), acetylated tubulin (Sigma-Aldrich; St Louis, MO), eNOS (BD Biosciences Pharmingen; San Jose, CA), pericentrin (Covance; Berkeley, CA), and adenylate cyclase-7 (FabGennix Inc. International; Frisco, TX). Secondary antibodies for immunohistochemistry (IHC) were anti-rabbit Alexa 488, anti-mouse Alexa 488, anti-rabbit Alexa 594, anti-mouse Alexa 594, goat anti-mouse, and goat anti-rabbit (all from Molecular Probes; Eugene, OR). Secondary antibodies for Western blotting were horseradish peroxidase-labeled goat anti-mouse and goat anti-rabbit (Sigma).
IHC
IHC was based on a previously published method (Jurczyk et al. 2004). Briefly, slides were rinsed with ethanol, coated with concanavalin A (5 mg/ml; Sigma-Aldrich), and left to dry. Slides were washed twice with PBS and air dried. Ciliated epithelial cells were cytospun onto the slides (∼1 × 105 cells per slide, 1200 X g for 4 min) and then treated with a pre-permeabilization solution (80 mM Pipes, pH 6.8, 5 mM EDTA, 1 mM MgCl2, 0.5% Triton X-100) for no longer than 1 min. Cells were fixed with cold methanol at −20C for 15 min, washed 2 × 5 min in PBS, and washed 2 × 15 min in PBSAT (0.5% Triton X-100, 2% BSA, in PBS). The slides were then kept in a humidity chamber for the remaining steps. Cells were incubated with primary antibodies diluted in PBSAT for 1 hr, washed 10 times with PBSAT, incubated with secondary antibodies diluted in PBSAT for 1 hr, washed five times with PBS, and then washed five times with PBSAT. Cells were then mounted in mounting fluid (5 ml PBS, 5 ml glycerol, 0.1 g n-propylgallate), and coverslips were sealed with clear nail varnish.
NO Assay
Bronchial epithelial cell NO production was monitored via the detection of NO by a gas-phase chemiluminescent reaction between NO and ozone (model 280i; Sievers Instrument, Boulder, CO). Isolated axoneme fractions from airway epithelial cells were treated with 100 mM ethanol over varying time periods. The exposure medium was separated from the particulate fraction and flash frozen to halt metabolic reactions. The proteins present in the exposure medium were precipitated in equal volumes of 0.5 N NaOH and 10% ZnSO4 for 15 min prior to being centrifuged at 14,000 X g for 5 min at 4C. Supernatants (10 μl) were injected into a reflux column containing 0.1 M VCl3 in 1 M HCl at 80C to reduce any nitrates and nitrites into NO. NO then combined with O3 produced by the analyzer to form NO2. The resulting emission from the excited NO2 was detected by a photomultiplier tube and recorded digitally (mV). Values were then interpolated to a standard curve of NaNO2 concentrations concurrently determined. Sample measurements were made in triplicate for each cell treatment with each sample being injected a minimum of three times for a total of nine readings per data point. Significance was determined by ANOVA.
Protein Kinase Assays
PKA and PKG activity in extracted bovine axonemes was determined using a previously described assay (Wyatt et al. 2005). PKA levels were assayed using 130 μM PKA substrate heptapeptide (LRRASLG), 10 μM cAMP, 0.2 mM IBMX, 20 mM Mg-acetate, and 0.2 mM [Λ-32P] in a 40-mM Tris-HCl buffer (pH 7.5). Measurement of PKG activity was the same as above with the substitution of the peptide RKKRSRAE for the heptapeptide substrate, the addition of 10 μM cGMP, and the presence of the PKA inhibitor peptide (0.05 μg; Bachem California Inc., Torrance, CA). Axonemes (20 μl) were added to 50 μl of the reaction mix described above and incubated for 15 min at 30C. To halt the reaction, 60 μl of the assay mix was spotted onto P-81 phosphocellulose papers (Whatman; Hillsboro, OR), the papers were washed five times for 5 min in 75 mM phosphoric acid, and then washed once in ethanol. The papers were dried and activity was counted using a scintillation counter. Enzyme activity was expressed as pmol/min/mg, and significance was determined using one-way ANOVA.
RII Overlay
Axonemes were run on SDS-PAGE and proteins transferred by Western blotting as described above. The RII overlay method was as described by Bregman (Bregman et al. 1989). PKA-RII was radiolabeled using the following reagents: 15 μg PKA-RII (generous gift from J. Corbin), 2 μg PKA C-subunit (Sigma-Aldrich), 10 μM cAMP, [Λ-32P] ATP (3.3 × 108 cpm/ml), brought up to 500 μl with 25 mM KPO4 buffer (pH 7) containing 10 mM MgCl2 and 0.5 mM dithiothreitol. The mixture was incubated at 0C for 5 min, and then non-radioactive ATP was added to give a total ATP concentration of 1 μM. The mixture was further incubated for 50 min. Residual [Λ-32P] ATP was separated from [Λ-32-32P] ATP-RII using Microcon YM-30 spin columns (Millipore; Billerica, MA). To determine the presence of an AKAP, the transferred proteins on nitrocellulose were incubated with the following solutions for 4 hr: 5 × 105 cpm [Λ-32P] ATP-RII (positive control); 5 × 105 cpm [Λ-32P] ATP-RII + StHt31 (Molecular Probes); and 5 × 105 cpm [Λ-32P] ATP-RII + StHt31-P (negative control). Nitrocellulose papers were then washed 6 × 10 min in wash buffer and exposed to photographic film.
Confocal Microscopy
Fluorescent images were obtained using a Zeiss LSM 410 confocal laser-scanning microscope (Göettingen, Germany) utilizing an argon/krypton laser (488 nm/568 nm/647 nm). Images were collected as .tif files. Fluorochromes used were Alexa 488 and 540.
Results
NO-dependent Cilia Regulatory Enzymes Are Located in the Basal Body of Intact Ciliated Cells
NO is an early upstream signaling molecule that triggers the rapid and transient stimulation of ciliary motility (Jain et al. 1993; Tamaoki et al. 1995; Uzlaner and Priel 1999; Li et al. 2000) through a NOS ŕ NO ŕ GC ŕ cGMP ŕ PKG pathway. We hypothesized that the three enzymes involved in this signaling pathway are located in the same subcellular region of the ciliated cell. Using confocal microscopy, antibodies specific for acetylated tubulin and pericentrin were used to define the axoneme (Figure 1) and basal body (Figure 1) regions, respectively, within intact bovine bronchial ciliated cells. The endothelial isoform of NOS (eNOS), the soluble guanylyl cyclase β-1 isoform (GC-β), and both PKG isoforms (PKG-I and PKG-II) all intensely stained in the subapical basal body region (Figures 11-C1). In addition to the basal body, PKG-II was detectable in the axoneme region and GC-β was detectable in the cytoplasm.
NO is also the upstream signaling molecule for stimulation of cilia by β-agonists via the AC ŕ cAMP ŕ PKA pathway. Because NO-dependent PKA-triggered cilia stimulation requires PKG activation, we hypothesized that adenylyl cyclase (AC) and PKA are located near the basal body. AC-7 and the RII isoform of PKA intensely stained the subapical basal body region (Figures 1G and 1I); PKA-RII staining was also present in the cytoplasm. In sharp contrast, the PKA-RI isoform antibody strongly stained the axoneme and was weakly detectable in the cytoplasm (Figure 1H). Importantly, PKA-RI was not detectable in the subapical basal body region. A summary of the distribution of each of these cyclic nucleotide-related enzymes is presented in Table 1.
NO-dependent Cilia Regulatory Enzymes Are Present in Isolated Ciliary Axoneme Preparations
The key cilia regulatory enzymes identified by IHC of intact airway epithelial cells were primarily located in the cilia basal body and/or axoneme; therefore, we hypothesized that these enzymes would be detected in isolated demembranated tracheal cilia preparations. To test this hypothesis, we prepared isolated demembranated cilia axonemes from fresh bovine trachea using the method of Hastie (Hastie et al. 1986) and subjected them to Western blotting and detection with the same antibodies used for the IHC of intact cells. Both acetylated tubulin and pericentrin were detectable, indicating the presence of both cilia axoneme and cilia basal body components within the isolated demembranated cilia preparation, respectively (Figures 2A and 2B). eNOS and each of the other NO-dependent cilia regulatory enzymes including GC-β, PKG-I, PKG-II, AC-7, PKA-RI, and PKA-RII were also detectable in the preparation (Figures 22-C2).
NO-dependent Cilia Regulatory Enzymes Colocalize With Basal Body- and/or Axoneme-specific Proteins
Detection of ciliary regulatory enzymes within the isolated organelle preparation by Western blot suggested that both basal bodies and axonemes were present in the demembranated cilia preparation. We hypothesized that these regulatory enzymes colocalize to basal body- and/or axoneme-specific proteins. To test this hypothesis, we used confocal microscopy to view intact ciliated epithelial cells stained with different combinations of regulatory enzymes and basal body-specific and axoneme-specific antibodies. We found that eNOS colocalized with only the very proximal insertion of the axoneme at apical/basal body area of the cell (Figure 3). In addition, eNOS colocalized with pericentrin, a basal body-specific protein, just below the apical attachment region of cilia (Figure 4). No colocalization between pericentrin and eNOS was observed in basal non-ciliated cells. The other two downstream enzymes in the classic NO-mediated signaling sequence, guanylyl cyclase and PKG, were also examined for eNOS colocalization. Both GC-β (Figure 5) and PKG-I (Figure 6) demonstrated colocalization with eNOS in the apical region of ciliated cells. No such colocalization between cyclase, kinase, or eNOS was observed in the basal non-ciliated cells.
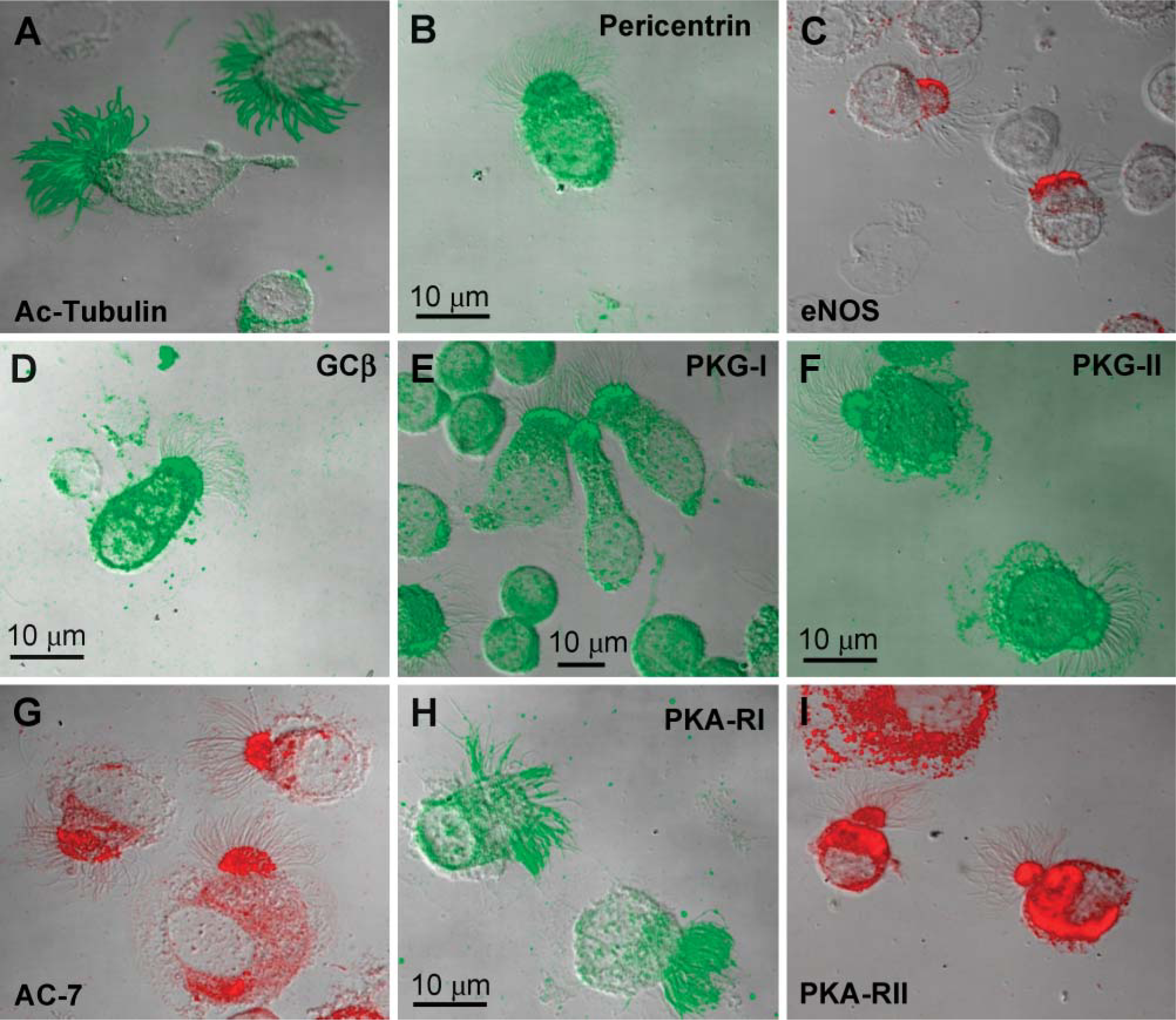
Identification of cilia motility-associated proteins by immunofluorescence confocal microscopy. Cytospin preparations of bovine ciliated airway epithelial cells were fixed and stained as described in Materials and Methods for the following proteins: cilia acetylated tubulin (
NOS, PKA, and PKG Are Active on the Ciliary Axoneme
We have previously shown that ethanol can activate NOS in intact ciliated epithelial cells (Wyatt et al. 2003). To determine if functional eNOS exists in the isolated cilia fractions, we assayed NO levels in response to L-arginine and ethanol exposure of axonemes. The addition of 1 μM L-arginine produced a baseline level of NO in axonemes (Figure 7). The addition of 100 mM ethanol significantly and immediately elevated the concentration of NO by 3- to 6-fold, with a maximum accumulation occurring at 20 min. Addition of 100 mM ethanol alone significantly elevated NO levels at 25 min, with the maximal accumulation of NO complete by 30 min.
Distribution of cyclic nucleotide-related enzymes in intact ciliated airway epithelial cells

eNOS, endothelial nitric oxide synthase; GC-β, guanylyl cyclase beta; PKG-I, type-I cGMP-dependent protein kinase; PKG-II, type II cGMP-dependent protein kinase; AC-7, adenylyl cyclase 7; PKA-RI, type I cAMP-dependent protein kinase; PKA-RII, type II cAMP-dependent protein kinase.
Because our current study demonstrated that PKA and PKG were localized on the ciliary axoneme, we hypothesized that these enzymes retain activity on the isolated axoneme. To test this hypothesis, PKA and PKG activities were measured using direct kinase catalytic assays. Enzyme activity was significantly increased (Figure 8) upon addition of the kinase second messengers (cAMP for PKA and cGMP for PKG). Following a 10-min incubation period at 37C, PKA activity increased 10-fold upon addition of 10 μM cAMP, and PKG activity increased 3-fold after the addition of 10 μM cGMP. Moreover, cAMP-stimulated PKA activity could be blocked by the addition of 1 μM KT5720, whereas cGMP-dependent PKG activity was inhibited by 100 nM Rp-cGMPS. These data suggest that the functional enzymatic activities of NOS, PKA, and PKG are retained on the isolated, cell-free cilia preparations.
PKA-RII Is Located on an AKAP on the Ciliary Axoneme
Based on the finding that PKA-RI was localized to the axoneme and PKA-RII was colocalized to the basal body, we hypothesized that an AKAP would be present in the bovine axoneme fraction that could act as a scaffolding protein to anchor protein kinases. To test this hypothesis, we performed an RII overlay assay on the ciliary axonemes and found that radiolabeled PKA-RII bound to a 30-kDa protein from the axoneme fraction (Figure 9). Axonemes were preincubated for 4 hr with St-Ht31 (10 mM), a peptide that competitively inhibits the binding of PKA-RII to an AKAP. In the presence of St-Ht31, no radiolabeled RII bound to the axonemal protein blot. Axonemes were also preincubated for 4 hr with St-Ht31P (10 mM), a control peptide that has a single amino acid substitution that prevents the peptide from binding to the RII-binding site of the AKAP. Radiolabeled RII binding was observed at 30 kDa in the presence of St-Ht31P. This overlay showed that an AKAP exists in the ciliary axoneme fraction that can bind PKA-RII, which likely keeps PKA-RII anchored at a site on or adjacent to the axoneme.
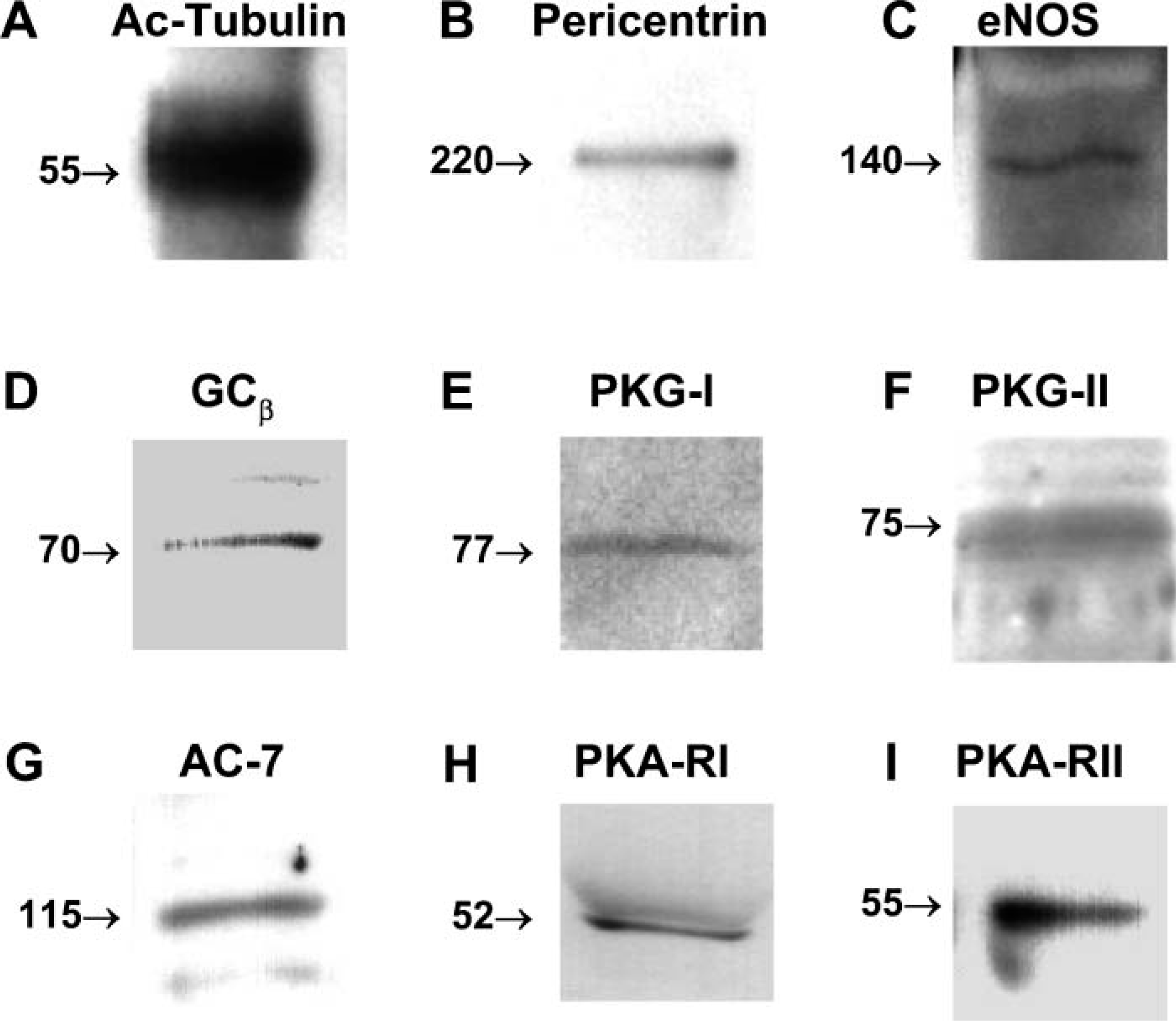
Identification of cilia motility-associated proteins by Western blot. Bovine ciliated axonemal fractions were prepared as described in Materials and Methods and the following proteins detected by Western blot: cilia-acetylated tubulin (
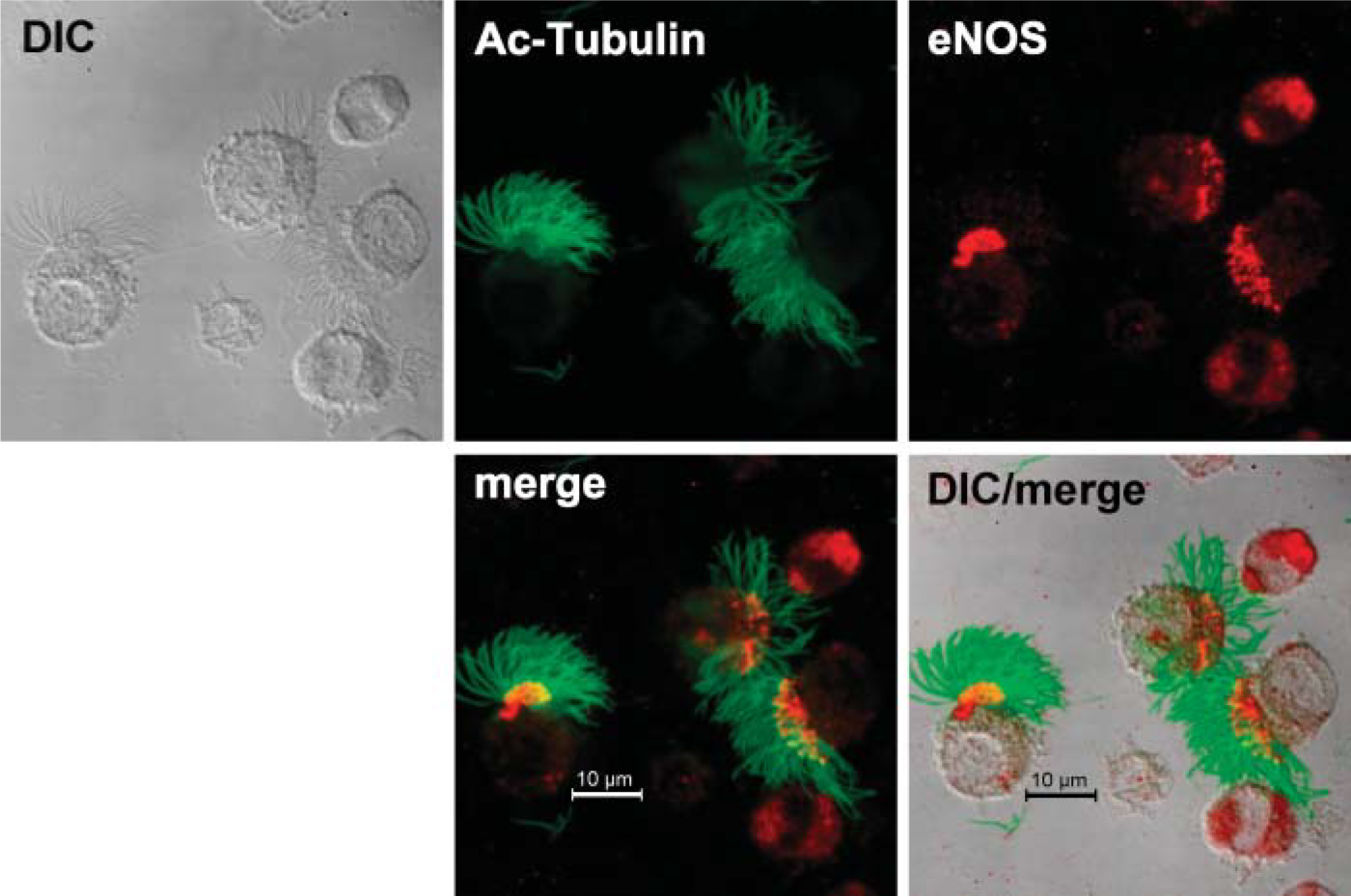
eNOS localizes to the apical surface of the bovine ciliated epithelial cell. Anti-acetylated tubulin (green) binds to cilia and anti-eNOS (red) is located in the apical surface of ciliated cells and throughout non-ciliated airway epithelial cells. eNOS and cilia colocalize (yellow) in the basal body region of the cilia.
Discussion
Protein kinases play an important role in the regulation of ciliary beat. Activation of PKA and PKG stimulates the ciliary beat of the mucociliary escalator and therefore the clearance of any particulate matter that could enter the respiratory tract. Although these protein kinases are found in epithelial cells, it has not previously been determined whether they are found on the ciliary axoneme, a site of action for these enzymes on the ciliated epithelium. Our data show that PKA-RII and PKG-I are predominantly located in the apical area of the cell where the basal bodies and eNOS colocalize. In contrast, PKA-RI and PKG-II are found both throughout the cell and localized directly on the ciliary axonemes. We have also demonstrated that PKA and PKG are active on the isolated ciliary fraction, can be stimulated by their respective cyclic nucleotide, and can be inhibited by their specific pharmacological inhibitors. The presence of these enzymes on the axoneme and in the apical area of ciliated epithelial cells suggests that they need to be located close to their site of action.
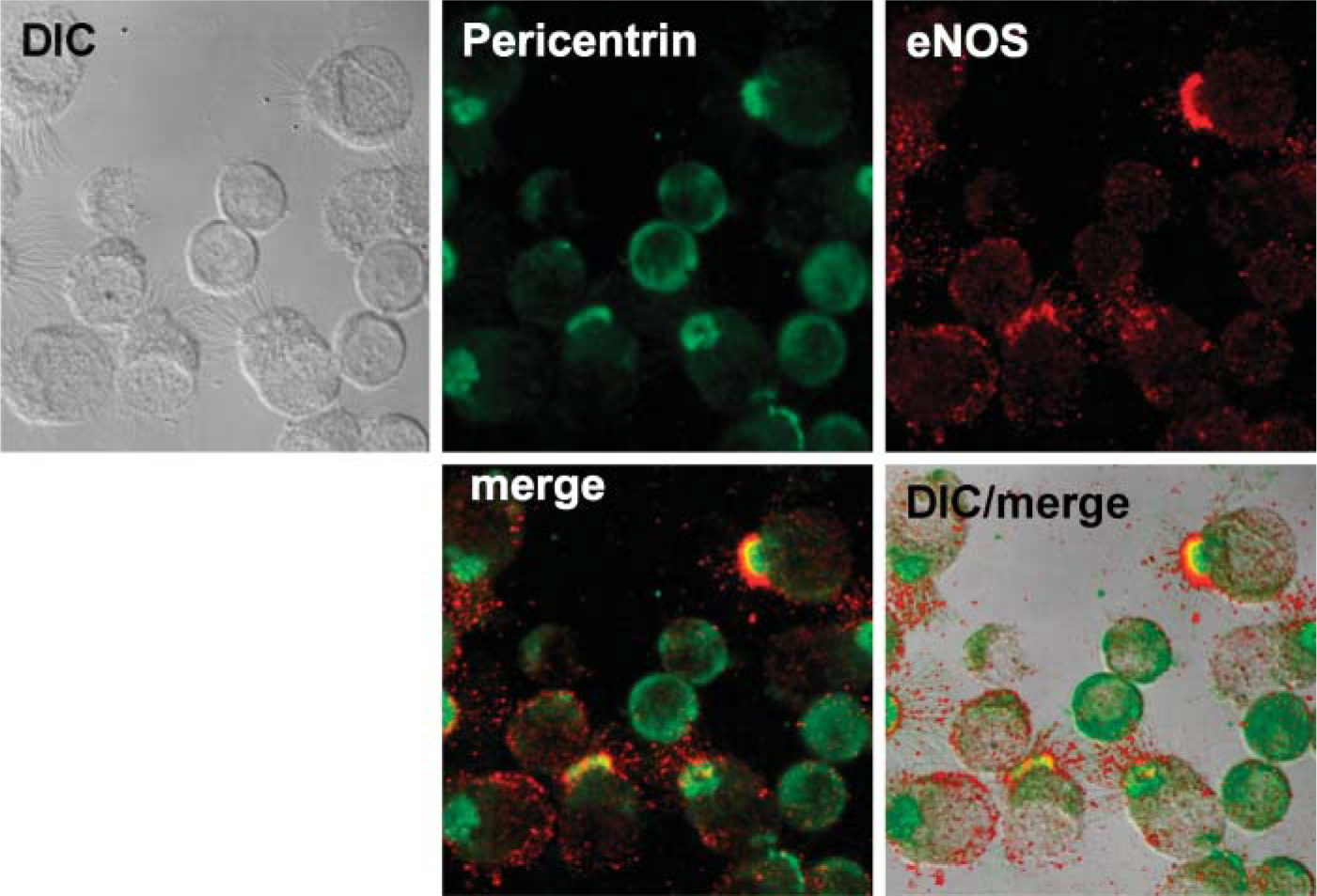
eNOS localizes to basal bodies in the bovine ciliated epithelial cell. Anti-pericentrin (green) and anti-eNOS (red) stain in the apical surface of ciliated cells. No colocalized staining is observed in non-ciliated airway epithelial cells. eNOS and pericentrin colocalize (yellow) in the apical region of the cell.
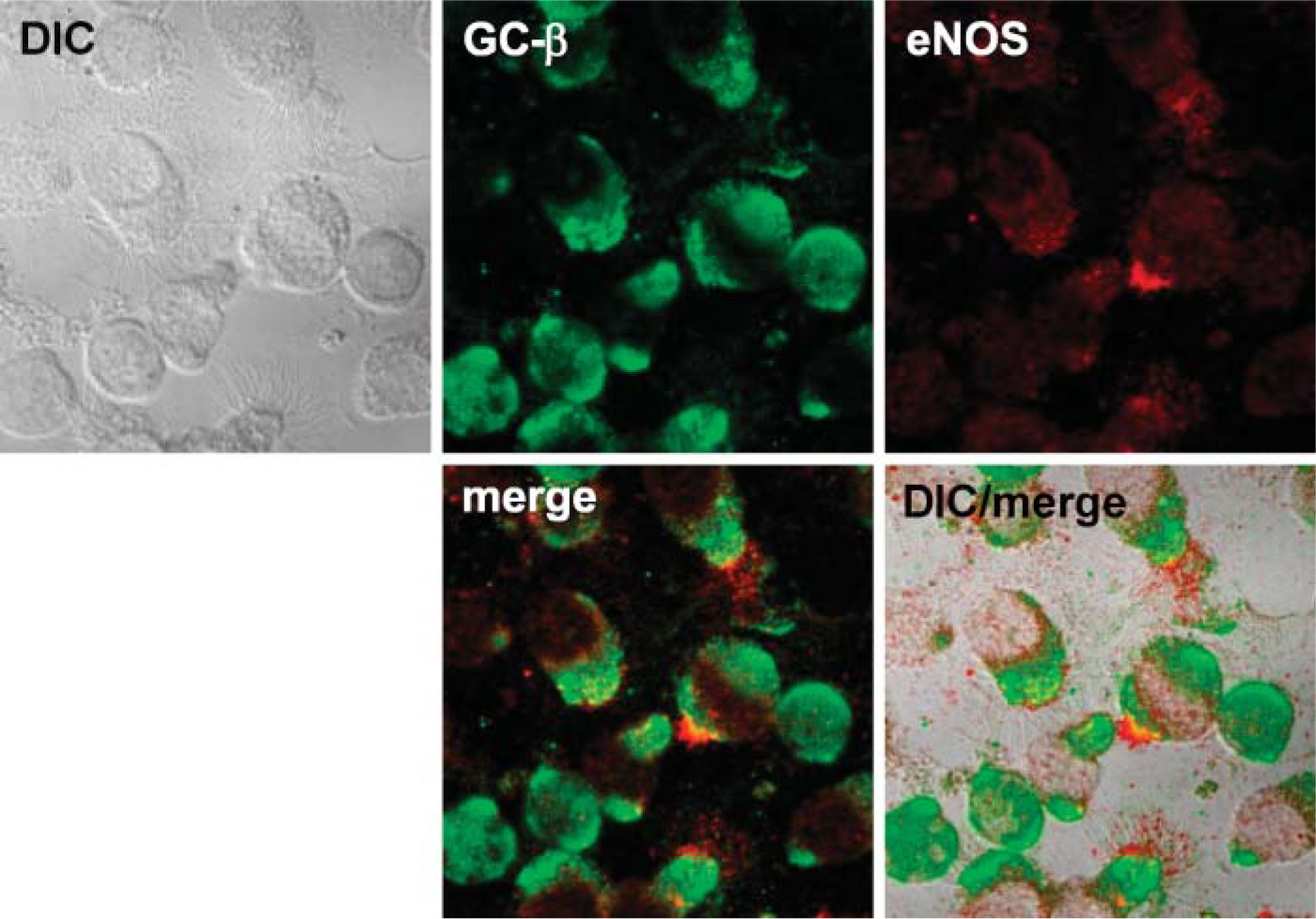
eNOS colocalizes with guanylyl cyclase in the bovine ciliated epithelial cell. Anti-GCβ (green) and anti-eNOS (red) stain in the apical surface of ciliated cells. No colocalized staining is observed in non-ciliated airway epithelial cells. eNOS and GCβ colocalize (yellow) in the apical region of the cell.
We have previously shown that there is an association between PKA, PKG, and CBF (Wyatt et al. 1998a,1999). Agents that activate PKA or PKG result in the stimulation of CBF in our BBEC model. In fact, ethanol stimulation of CBF requires the coordinated activation of both PKG and PKA (Wyatt et al. 2003). We have established that isolated axonemal fractions from BBEC can be stimulated to maximally “beat” upon the combined addition of both cAMP and cGMP (Wyatt et al. 2005). PKA regulation of axonemal movement has also been reported in Paramecium and sperm (Hamasaki et al. 1991; Inaba et al. 1998). Our findings that PKA and PKG are not only located on the ciliary axoneme but are also functionally active in the ex situ state give new insight into the compartmentalized regulation of ciliary motility.
PKG and its role in flagellar or ciliary beating have not been as widely studied as the other protein kinases; only PKG-Iβ has been detected on rat airway epithelial cells (Zhan et al. 1999). It has been reported that NO regulates CBF via activation of PKG (Li et al. 2000) and that NO is essential for maintaining ciliary motility (Zhan et al. 2003). Moreover, eNOS (required for the production of NO) has previously been identified on the ciliated epithelium (Zhan et al. 1999). In the current study we located eNOS at the base of the ciliary axoneme, further suggesting the importance of NO in controlling ciliary motility.
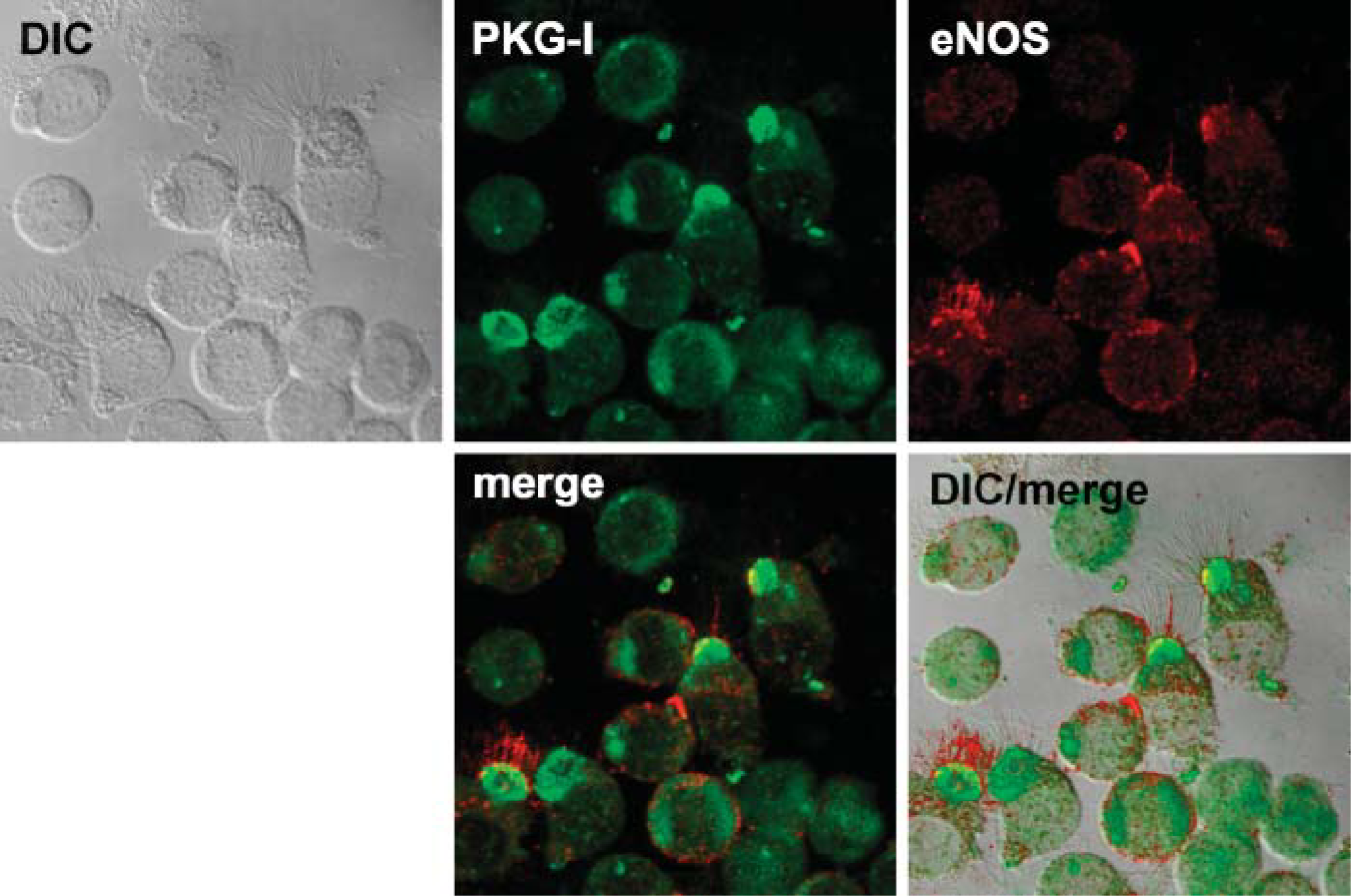
eNOS colocalizes with PKG-I in the bovine ciliated epithelial cell. Anti-PKGI (green) and anti-eNOS (red) stain in the apical surface of ciliated cells. No colocalized staining is observed in non-ciliated airway epithelial cells. eNOS and PKGI colocalize (yellow) in the apical region of the cell.
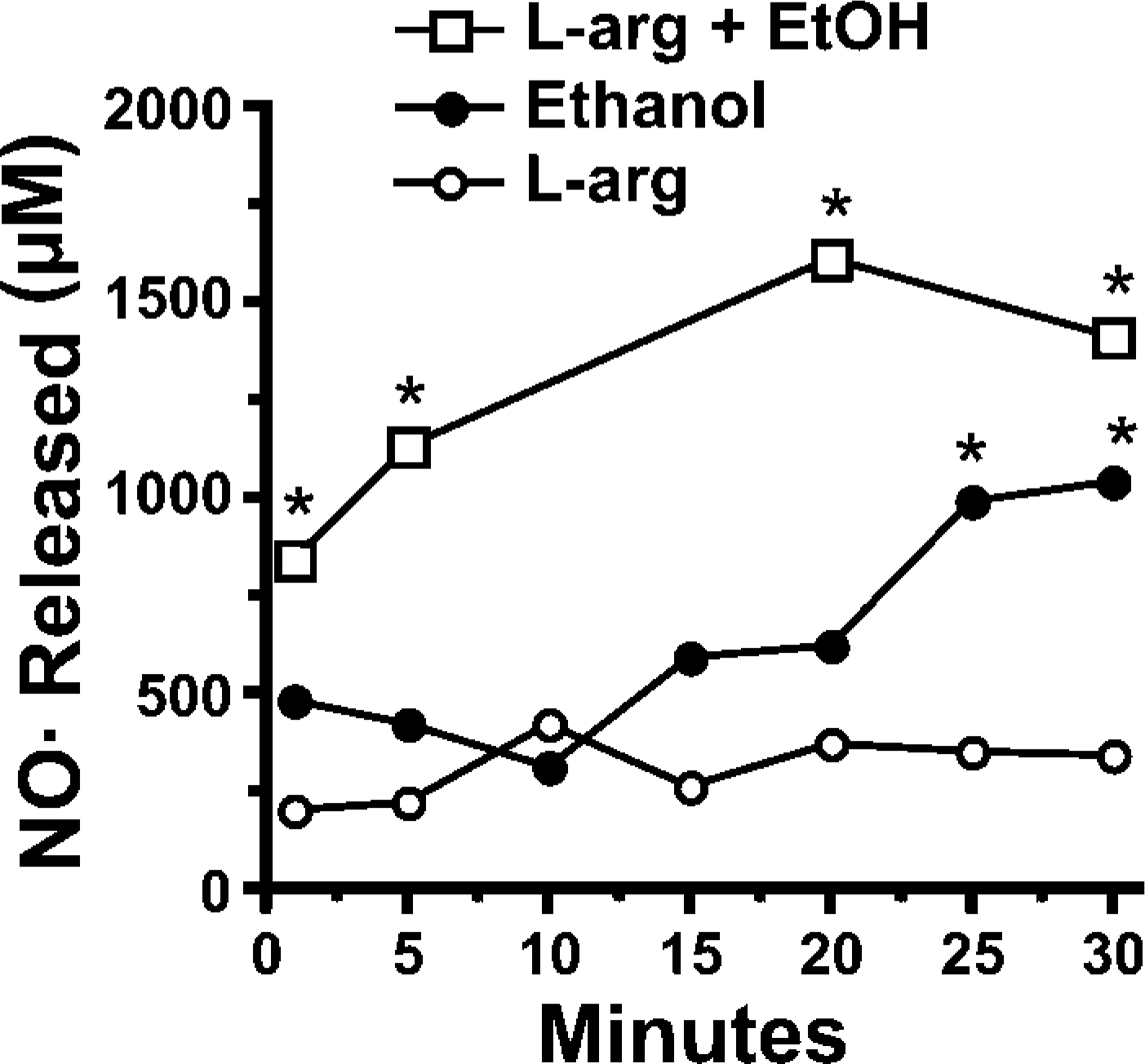
Nitric oxide (NO) levels in bovine ciliary axonemal fractions. Axoneme fractions were incubated with 1 μM L-arginine (L-arg), 100 mM ethanol (EtOH), or both and the change in media nitrite and nitrate (μM) measured. Ethanol-stimulated production of NO from the axoneme fraction was augmented by L-arg (∗ p<0.05).
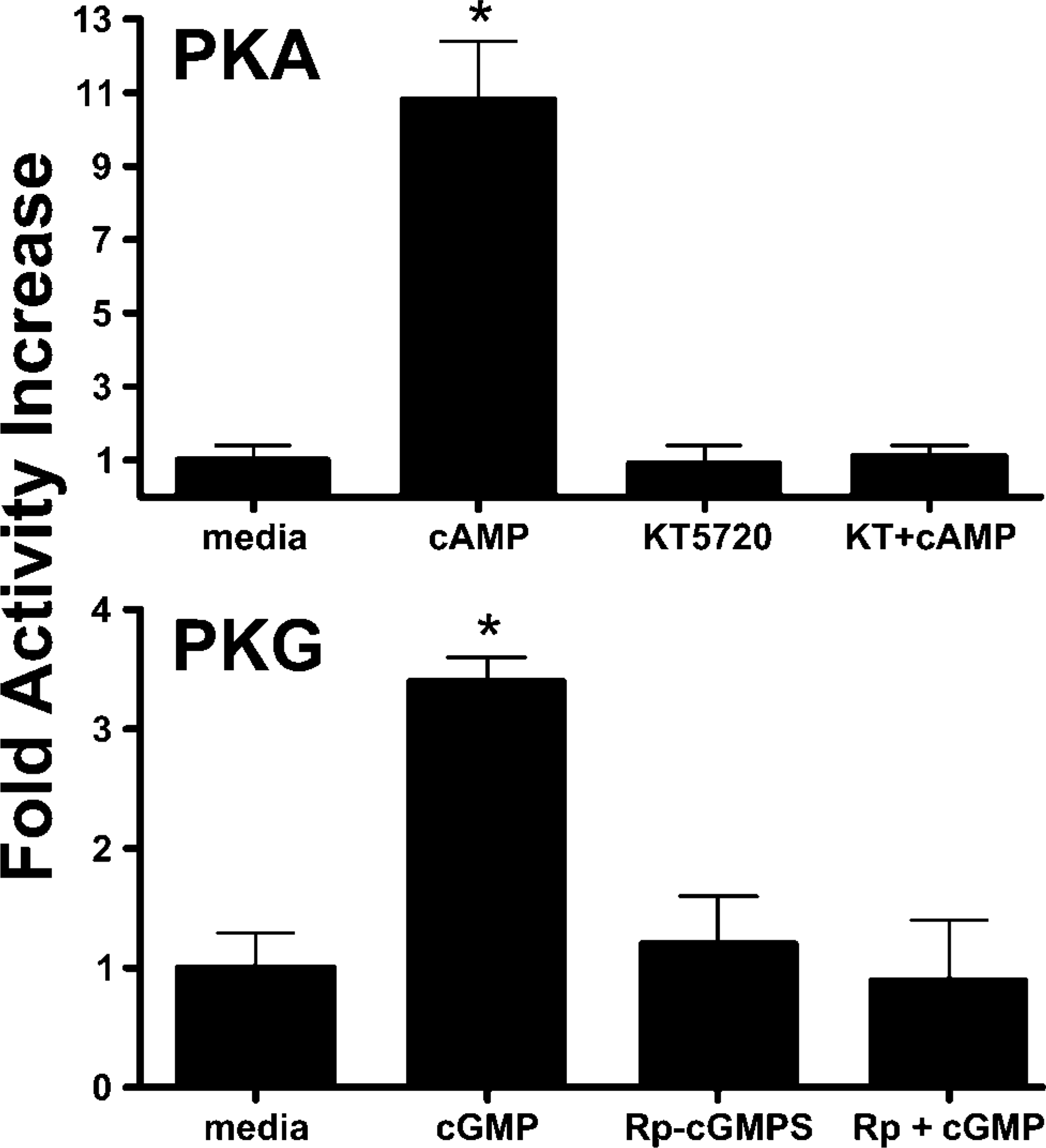
Cyclic nucleotide-dependent kinase activity in bovine ciliary axonemal fractions. Axoneme fractions were incubated with reaction buffer (media), 10 μM cAMP, 1 μM KT5720, or both cAMP + KT5720 for 10 min at 30C and assayed for PKA activity. Axoneme PKA was increased 11-fold by cAMP (∗ p<0.01) and blocked by KT5720 (top panel). Axoneme fractions were incubated with reaction buffer (media), 10 μM cGMP, 100 nM Rp-cGMPS, or both cGMP + Rp-cGMPS for 10 min at 30C and assayed for PKG activity. Axoneme PKG was increased 3.5-fold by cGMP (∗ p<0.05) and blocked by Rp-cGMPS (bottom panel).
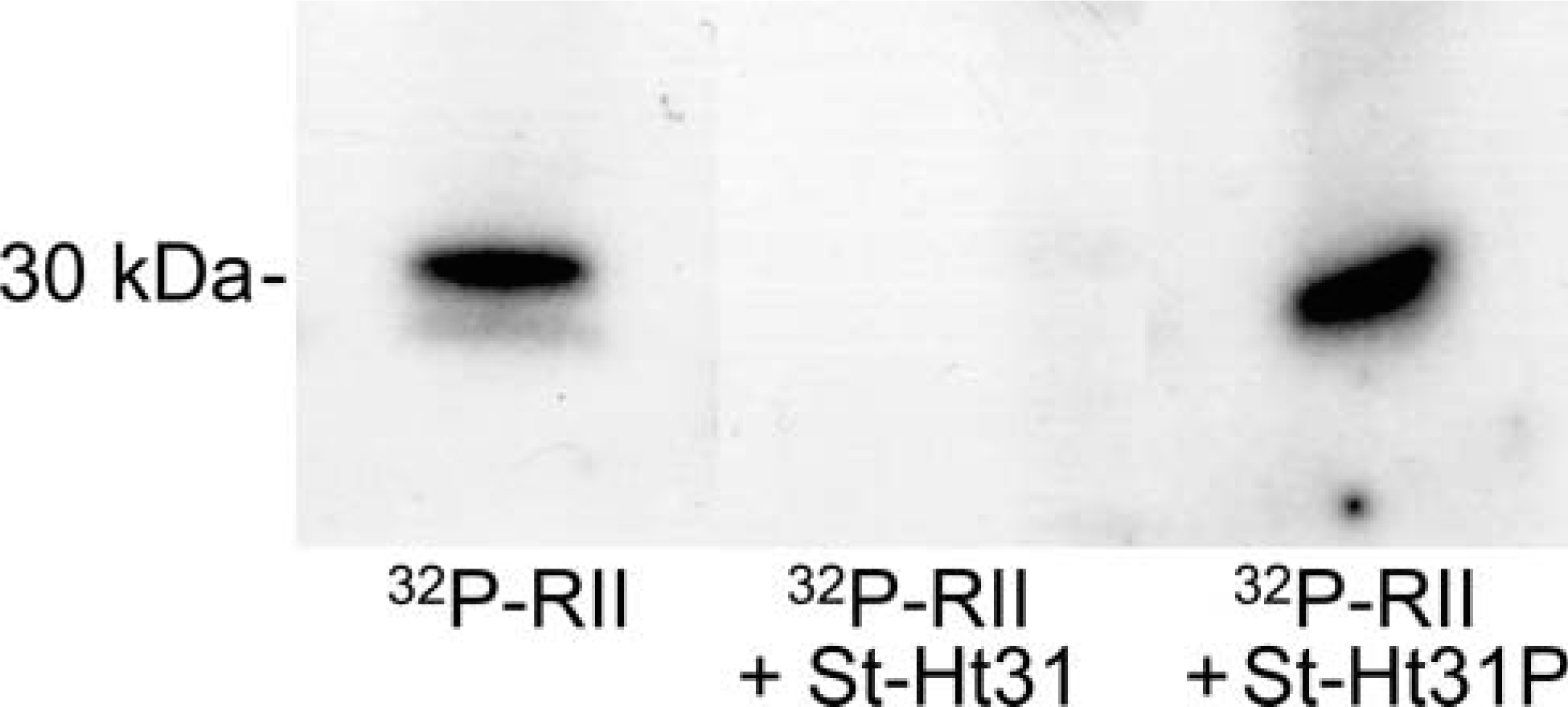
Identification of A-kinase-anchoring protein (AKAP) in axonemal fractions from ciliated bovine tracheal epithelial cells. Axonemal proteins were immobilized on blots as described in Materials and Methods and incubated with radiolabeled PKA-RII (32P-RII) in the presence or absence of AKAP-binding peptide (St-Ht31) or control peptide (St-Ht31P). PKA-RII binds to the axonemal blot only in the absence of the competitive AKAP-binding peptide.
Our findings suggest that stimulation of CBF involves a dual activation of both PKA and PKG (Wyatt et al. 1998a,2003); therefore, it would be logical that regulation of second messengers for these kinases would be efficiently orchestrated in a compartmentalized state near, or on, the axoneme (Figure 10). Indeed, our data demonstrate that AC-7 and GC-β are also localized at the apical surface of the ciliated cell in a colocalized state with eNOS. Agonist-stimulated production of NO would be poised to regulate cyclase activity leading to kinase activation at the same localized region of the cell for maximum, targeted efficiency. Colocalization of eNOS and PKG-I reported here also suggests that the location of these enzymes is essential to their coordinated function. These observations on the targeted localization of PKG to a functional regulatory unit at the apical surface of the ciliated cell support our previous observations of alcohol-mediated stimulation of CBF (Wyatt et al. 2003). Specifically, we have found that ethanol stimulation of CBF requires NO and PKG activation for the PKA-mediated increase in CBF. Likewise, chronic exposure to ethanol results in the uncoupling of this pathway (Wyatt and Sisson 2001; Wyatt et al. 2004a). We hypothesize that the phosphorylation of a putative PKG substrate facilitates this PKA-driven stimulation of cilia beating in response to alcohol. An orchestrated metabalon of these enzymes located at the base of the cilia would facilitate such regulation.
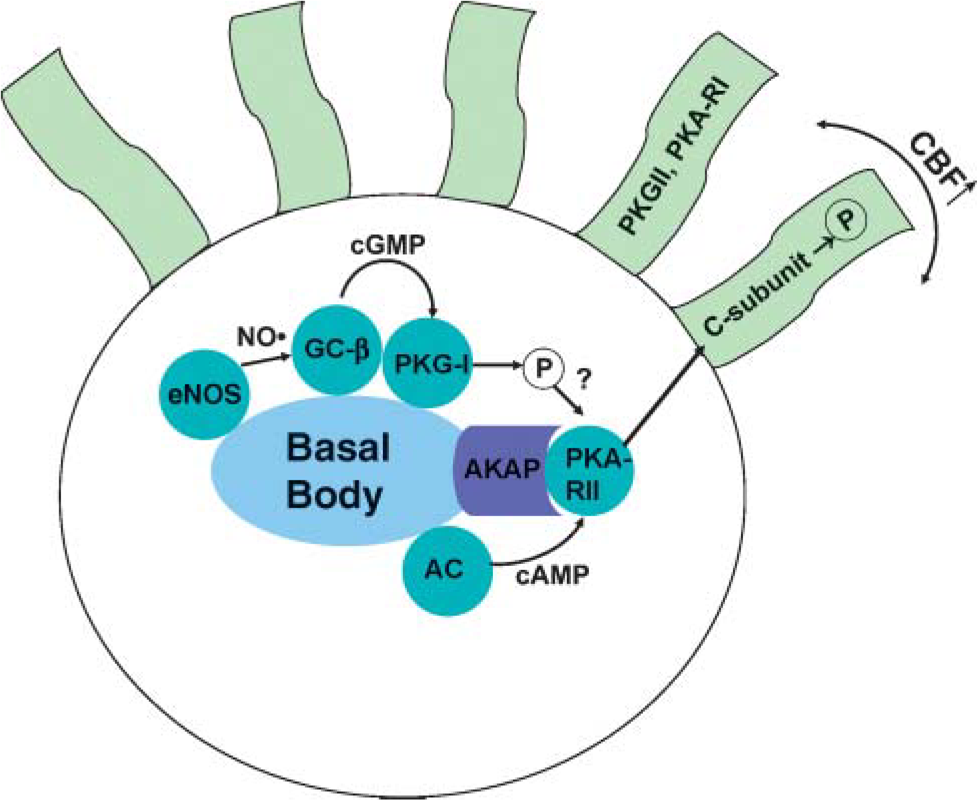
Model of localized cyclic nucleotide action in the apical region of ciliated airway epithelial cells. Several enzymes (blue dots) colocalize with the basal body in the apical region of the ciliated cell. Upon stimulation, constitutive type-2 nitric oxide synthase (eNOS) produces NO, which activates GC-β. Elevated cGMP then activates the PKG-I required for ethanol-mediated PKA-RII activity in response to AC-activated production of cAMP. PKA-RII is localized to an AKAP and releases the catalytically active subunit (C-subunit) upon activation. The C-subunit increases in vitro ciliary beat frequency. PKG-II and PKA-RI are localized directly on the axoneme.
Our results have shown that PKA-RI and PK-RII are located on the isolated bovine ciliary axoneme fraction. Immunolocalization suggests that PKA-RII is located in the apical region of the cell in association with the basal bodies and that PKA-RI is localized to the axoneme itself. The precise location of PKA-RI on the axoneme has yet to be determined. In many areas of the cell, PKA is bound to an AKAP. The AKAP keeps PKA anchored at a cellular location close to its site of action. AKAPs have been found on both the Chlamydomonas flagellar axoneme (Gaillard et al. 2006) and the human ciliary axoneme (Kultgen et al. 2002). The AKAP we found on the bovine ciliary axoneme is of a similar size to the human AKAP, which is 28 kDa and binds to PKA-RII. AKAPs are found associated with cellular membranes and organ-elles; some bind to both PKA-RI and PK-RII, and some also bind PKC and protein phosphatases. Whether the AKAP we identified binds more enzymes associated with ciliary motility has yet to be determined.
We have shown that isotypes of PKA and PKG are located at both the base of the ciliary axoneme, in the apical region of ciliated cells, and on the ciliary axoneme. Moreover, we have also demonstrated that these enzymes are active. PKA and PKG activity could both be stimulated and inhibited on the axoneme. Together these data suggest that all enzymes and precursors necessary for ciliary motility are located at the base of the ciliary axoneme close to their point of action, resulting in targeted phosphorylation and efficient regulation of ciliary beat.
Footnotes
Acknowledgements
This research was supported by the Department of Veterans Affairs MERIT grant (to TAW) and National Institute on Alcohol Abuse and Alcoholism, National Institutes of Health, Grant R01 AA-08769 (to JHS). TAW is an American Lung Association Career Investigator.
We thank Janice Taylor of the Confocal Laser Scanning Microscope Core Facility at the University of Nebraska Medical Center, which is supported by the Nebraska Research Initiative, for providing assistance with confocal microscopy.
