Abstract
Considerable research has been focused on the use of bone marrow-derived mesenchymal stem cells (MSCs) for the repair of non-unions and bone defects. To date, the question of whether transplanted MSCs survive and engraft within newly formed tissue remains unresolved. The development of an easy and reliable method that would allow cell fate monitoring in transplant recipients is a pressing concern for the field of tissue engineering. To demonstrate the presence of transplanted cells in newly formed bone, we established a xenograft nude rat model allowing the detection of murine LacZ MSCs in vivo. MSCs were isolated from transgenic
T
Among the different techniques that have been proposed to follow the fate of implanted cells in vivo, only the transduction of detectable genetic markers, such as those encoding for β-galactosyltransferase (LacZ) and green fluorescent protein gene (GFP) allow stable and reliable long-term labeling of transplanted cells. To date, attempts to develop an easy, efficient, and reliable method to monitor genetically modified MSCs in a bone defect have had limited success, mainly because the label was not compatible with the specific procedures used for bone histology. GFP, from the jellyfish
The aims of the study were (1) to optimize the histological procedure for LacZ staining of bone explants harvested from
Materials and Methods
Transgenic Mice
Experiments were carried out using a transgenic line bearing
LacZ Staining
Fixation
Cells were rinsed twice in PBS and fixed for 12 hr at 4C before staining. Four different fixatives were evaluated in this study: (a) 10% neutral-buffered formaldehyde, (b) 2% formaldehyde/0.2% glutaraldehyde (F/GTA), (c) Schaffer's solution (containing 700 ml methanol, 300 ml 37% formaldehyde, 20g CaCO3, 10 mg MgCO3, pH 7.2), and (d) Neo-fix fixative, a formalinfree fixative (Ref. 65035-75; Merck, Darmstadt, Germany).
After fixation, tissue specimens were washed three times in PBS, gradually dehydrated in an acetone bath for 16 hr, and then soaked in xylene for 2 hr at 4C. Samples were then infiltrated for 6 hr with three changes of the resin solution (2 hr each) containing 10 ml of MMA with 50 mg benzoyl peroxide (Sigma), 2 ml of
In Vivo Tracking of Murine LacZ MSCs Into Nude Rats
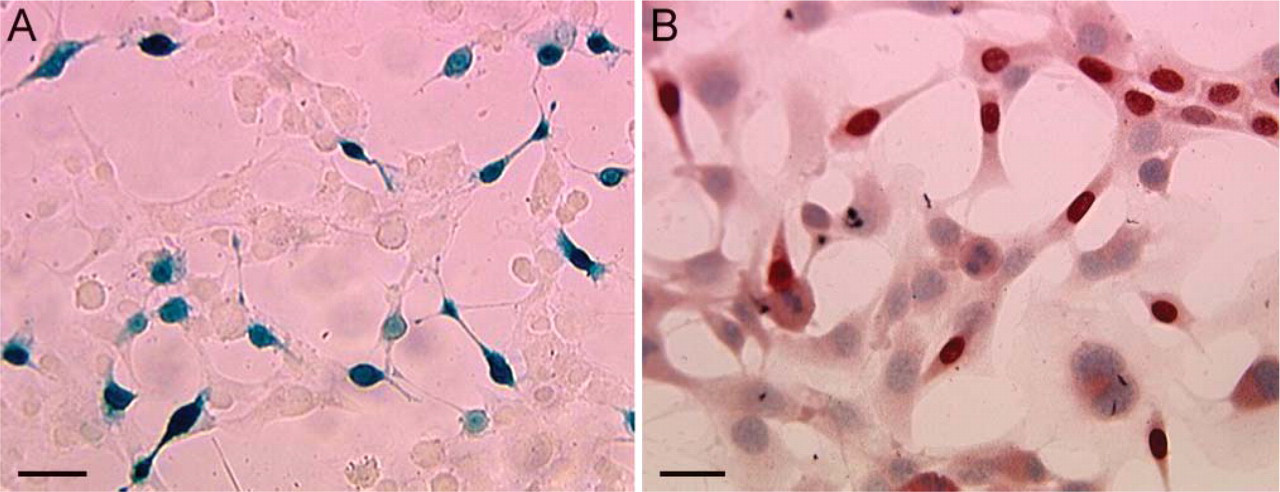
After 12 hr of fixation at 4C, higher staining intensity was obtained with 2% formaldehyde/0.2% glutaraldehyde (F/GTA) fixative for β-gal activity (
Results
Influence of Different Fixatives on LacZ Staining
As a preliminary step, we tested several fixatives for an incubation time of 12 hr on cell cultures (Table 1) and 2 to 21 days on bone explants harvested from LacZ mice (Table 2). Among all the fixatives tested, F/GTA at 4C for 4 days gave the best results for X-gal staining on both cell cultures (Figure 1) and bone explants (Figure 2). The use of Neo-fix fixative (Merck) led to a significant decrease in X-Gal staining after 2 days of fixation of bone samples when compared with the use of F/GTA. After 12 hr of fixation with Schaffer's solution and 10% formaldehyde, β-gal activity could not be detected on cell cultures or on bone specimens.
For immunodetection of β-gal with the primary rabbit polyclonal antibody, the influence of the different fixatives on the intensity of the reaction was more subtle (Table 1). The best performance was achieved with Schaffer's solution at 4C (Figure 1B). Negative immunostaining controls (control specimens and secondary antibody stainings) did not show any false positive binding (data not shown).
Visualization of LacZ Cells After Implantation In Vivo in a Bone Defect
Given a rate of β-gal expression of 45%, examination of LacZ cells must be interpreted with caution because β-gal-expressing cells may not be representative of the whole cell population. However, LacZ-positive cells plated on plastic did not show any difference in cell morphology as compared to non-labeled cells, thus reducing the possibility of a selective tracking of a subpopulation of MSCs (Figure 1).
Influence of different fixatives on LacZ staining in vitro

−, no staining; +, mild staining; ++, moderate staining; +++, intense staining. β-gal, β-galactosidase; F/GTA, 2% formaldehyde/2% glutaraldehyde.
Cells were fixed at 4C for 12 hr. For β-gal activity, X-Gal was used as staining substrate, and pH of the solution was set at 7.4
Influence of different fixatives on β-gal activity in MMA-embedded bone explants

−, no staining; +, mild staining; ++, moderate staining; +++, intense staining. MMA, methylmethacrylate.
Fixation was performed at 4C. For β-gal activity, X-Gal was used as staining substrate, and pH of the solution was set at 7.4
The embedding procedure described in this study was successful and reliable. Thin sections of 5-μm thickness could be performed, even several months after polymerization of the blocks. MMA embedding allowed optimal tissue preservation for morphology assessment. Standard stainings could be applied on acetone-deplastified sections without modification of the histological protocols. Bone formation occurred within the defect and was predominantly present at the periphery of the lesion (Figure 3A). Histological examination revealed extracellular matrix formation with an important neovascu-larization. The predominant type of bone observed was woven bone. Standard X-Gal staining showed evidence of integration of bone marrow-derived MSCs in the newly formed tissue (Figures 3B and 3C). Blue staining was uniform and mainly localized in the medullary spaces along bone trabeculae (Figure 3B) and within vascular walls (Figure 3C). No evidence of enchondral ossification was observed at the different endpoints. Nucleated cells expressing LacZ were occasionally detected in peripheral blood of transplanted animals (Figure 3D).
Discussion
The aim of the present study was to optimize LacZ staining in bone specimens embedded in MMA and to apply this protocol for the detection of MSCs transplanted in bone defects. The rapid embedding procedure for MMA described in this study allowed detailed visualization of LacZ cells in bone tissue embedded in MMA with high morphological preservation. This shortened embedding method was originally designed to alleviate time-consuming steps during the processing of bone marrow biopsies (Lebeau et al. 1995). It is reliable and can be done with commercially available methacrylates. It relies on the use of a low amount of benzoylperoxide as initiator and
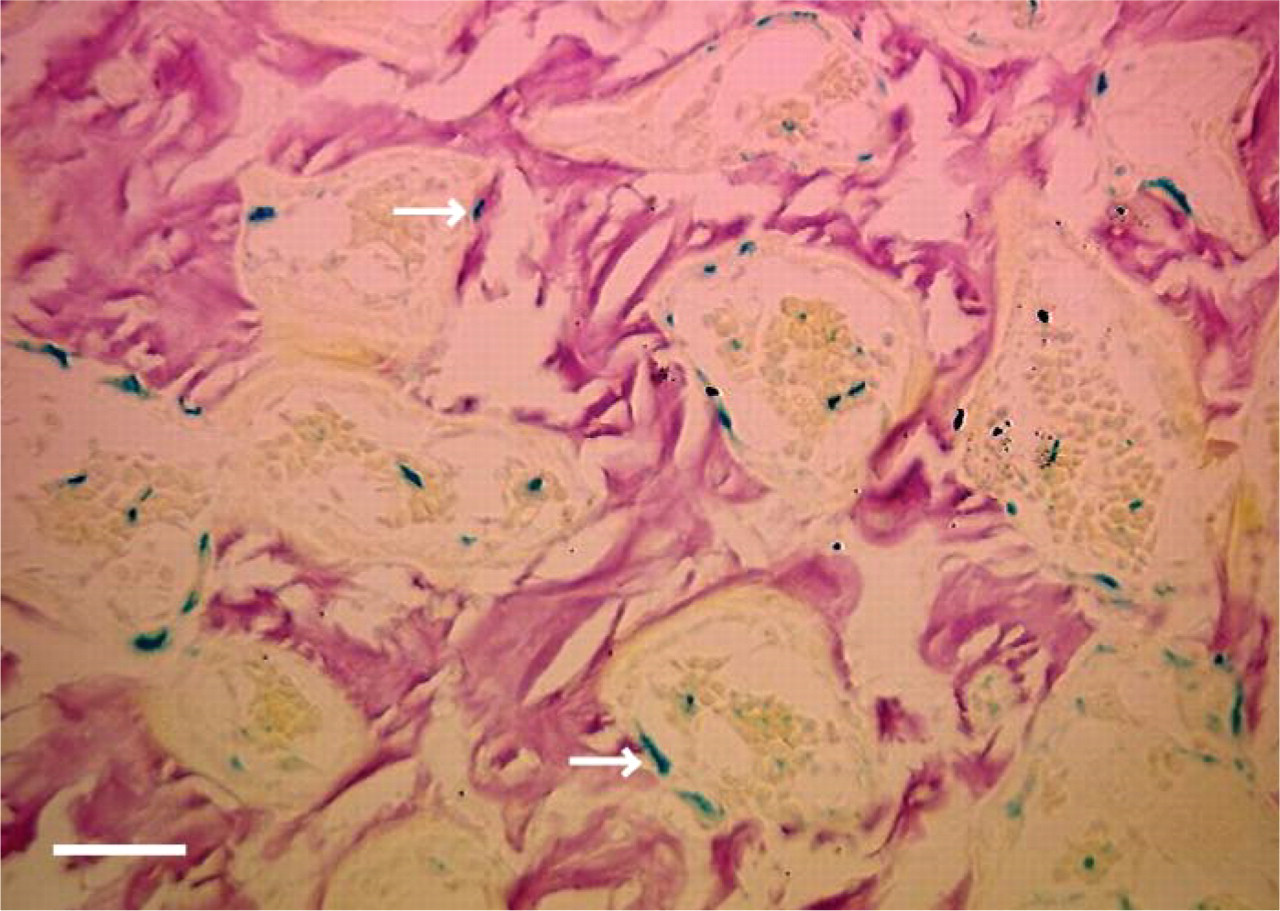
Tissue section of a femoral bone explant harvested from a lacZ transgenic mice and embedded in methylmethacrylate. X-gal staining was performed at 4C, pH 7.4. Among all the fixatives tested, F/GTA for 4 days gave the best results. Blue cells were observed within the medullary space and along the walls of bone trabeculae (white arrows). Picrofuschin staining. Bar = 100 μm.
In regard to β-gal detection in embedded tissues, the major problem seems to be the fixation step, which must be adapted to the hardness of the tissue and the size of the samples examined. In this study, sensitivity of β-gal detection by X-Gal staining was closely dependent on the nature of the fixative used and duration of fixation, which corroborates with previous reports (Ma et al. 2002; Bell et al. 2005). After 12 hr of fixation at 4C, β-gal activity decreased dramatically in 10% formaldehyde and Schaffer's solution and could only be detected in 0.2% glutaraldehyde containing fixative and Neo-fix fixative (Merck). Higher staining intensity was obtained after 4 days of fixation at 4C with F/GTA fixative. Similar results were reported by Ma et al. (2002) in kidney tissue sections with 0.2% glutaraldehyde, but could not be reproduced after 8 hr of fixation, which is not sufficient for the processing of bony specimens (Lebeau et al. 1995; Mueller et al. 2000). Skeletal tissues usually require longer times of fixation, which may impair tissue reactivity in histochemical and immunohistochemical investigations. Our results emphasize the importance of performing a test battery to optimize the main parameters (nature of the fixative, duration, and temperature for tissue preparation) that influence the detection of β-gal activity in the tissue examined and avoid invalid conclusions when evaluating the in vivo fate of LacZ cells.
Despite the number of research papers reporting the effectiveness of tissue engineering strategies for the repair of massive bone defects in animal models, very few studies have so far assessed the exact fate of implanted cells in vivo, mainly because of technical limitations during the histological procedure (Oshima et al. 2005). Whether implanted cells differentiate and incorporate into newly formed bone or simply interact with the host environment to initiate and promote the repair process is not well known. We have focused on tracking MSCs because these adult, autologous stem cells are excellent candidates for the engineering of several tissues including bone, cartilage, vessels, myocardium, and brain (Gregory et al. 2005). MSCs can easily and reliably be isolated from bone marrow aspirates and can maintain their growth potential through many cell divisions in vitro while at the same time retaining their capacity to differentiate along different lineages after their implantation in vivo (Caplan 2005). The
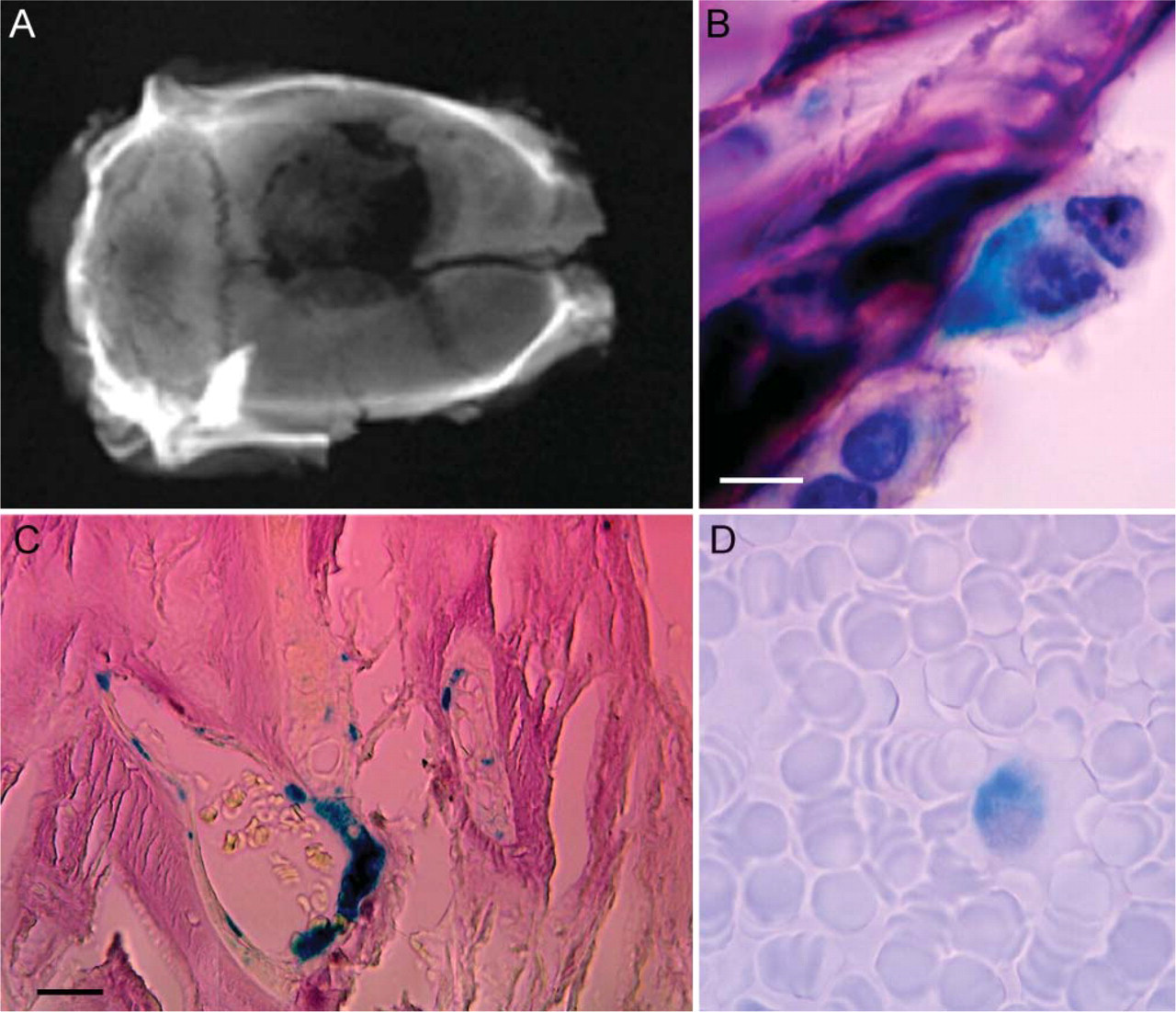
(
In this study we established a xenograft nude rat model allowing the detection of murine LacZ MSCs in tissue-engineered bone. To distinguish bone marrow-derived cells from host cells, murine LacZ MSCs were seeded onto a bioabsorbable collagen sponge and transplanted to repair a calvarial defect in nude rats. A non-transgenic background strain of mice could have been used to distinguish LacZ cells from host cells. However, the 8-mm rat craniotomy is a well-established intraosseous wound model that has been used by a number of authors to evaluate potential bone-healing materials including growth factors and cell-containing scaffolds. For reproducibility reasons, the craniotomy is performed with a cylindrical low-speed bur without breaching the dura. When performed in mice, this may damage the superior sagittal sinus located at the inner surface of the skull, resulting in a high animal mortality rate. Such damage is less likely to occur when performed in rats. Using the xenograft nude rat model, LacZ-labeled MSCs were detected in vivo for up to 4 weeks after implantation and contributed to the repair of the bone defect. After 4 weeks, the defect was partially filled with bone, which was predominant at the periphery of the defect. The short period of implantation in this study may account for the lack of osteogenesis in the middle of the defect. In a previous study performed in the same animal model, we showed a statistical increase in bone formation between 1 and 2 months when defects were filled with MSCs and BP (Arnaud et al. 1999). Moreover, bone healing after bone grafting involves a sequence of dynamic events that usually occurs in five phases: inflammation, microrevascularization, osteoinduction, osteoconduction, and osteoremodeling. Osteoinduction is largely controlled by growth factors, which induce migration, proliferation, and differentiation of non-committed mesenchymal cells in the fracture site. In this study, the scaffolds used were biodegradable bovine collagen sponges containing a mixture of BP that may have elicited new bone formation by the recruitment of cells migrating from the adjacent host bone marrow cavity. Although data from this study were only qualitatively evaluated without determining cell number per area, over time they gave important information on the fate of implanted cells. To the authors' current knowledge, this is the first time the lacZ gene was successfully used to track MSCs in skeletal tissues embedded in MMA. Using this chimeric animal model, we ascertained that the newly formed tissue contained cells derived from the transplanted LacZ MSCs. Histological examination revealed extracellular matrix formation and active osteoblastic cell rimming, but osteocytic cells could not be seen within bone tissue. Despite the use of BP, no enchondral ossification was observed in any specimens, which is consistent with previous reports (Arnaud et al. 1999) and is explained by the intramembranous ossification process that occurs in the calvaria. Interestingly, LacZ cells could be seen in the medullary spaces of recovered implants. In this study, MSCs were generated by a standard selection method based on plastic adhesion of bone marrow stromal cells. The proportion of hematopoietic cells in cultured cells is reduced from ∼11% of the total cell number at passage number 2 to <5% at passage 10 (Krebsbach et al. 1997). Therefore, it is not entirely clear whether LacZ-positive cells located in the bone marrow originated from the stromal or the hematopoietic fraction of bone marrow. This study also demonstrates that MSCs incorporate into the vasculature of newly formed bone and differentiate into endothelial cells. The neovascularization process, which occurs within a bone defect or at the fracture site, is of paramount importance to achieve bone healing. It supplies oxygen, nutrients, and growth factors that induce migration, proliferation, and differentiation of an appropriate subset of cells in the fracture site (Einhorn 1998). In bone marrow transplant studies, the contribution of MSCs to the formation of growing vessels in adult organisms remains controversial. Although there is increasing evidence that endothelial cells may originate from bone marrow-derived precursors (Takahashi et al. 1999; Hess et al. 2002; Zhang et al. 2002b), some authors failed to identify circulating GFP-labeled bone marrow-derived cells in vascular walls of growing vessels (Ziegelhoeffer et al. 2004). Here we demonstrate that the neovascularization that occurs within the defect not only depends on the sprouting of new vessels from a preexisting network (angiogenesis), but also on the contribution of implanted or circulating endothelial progenitors (postnatal vasculogenesis) contained in the bone marrow stroma. This study, however, did not permit us to identify the subpopulation of bone marrow-derived cells that are specifically involved in vasculogenesis. Further studies with rigorous quantification of cells are certainly warranted to clearly address this issue.
In conclusion, a rapid procedure for MMA embedding allows for both precise morphological analysis and histochemistry. This technique provides a unique tool for in vivo detection of β-gal-expressing cells incorporated into all types of tissues including undecalcified bone and cartilage. The present study provides irrefutable evidence that LacZ-labeled MSCs can be detected with the standard X-Gal staining in skeletal tissues embedded in MMA. LacZ still represents a useful phenotypic marker in cell-tracking studies and may provide important information on cell fate and relationships of cells to the different biomaterials investigated. Transplanted MSCs participated in the repair of a calvarial bone defect and contributed to the neovascularization of the lesion.
Footnotes
Acknowledgements
The authors are grateful to Cindy Blanchat (Laboratoire de Recherches Orthopédiques; CNRS, Paris, France) and Michel Soudière (UFR Biomédicale des Saints-Pères; Paris, France) for technical support.
