Abstract
Until now, significant differences in the neurochemical pattern of enteric neurons have been demonstrated in all species studied; however, some strong similarities also occur across species, such as the occurrence of nitric oxide synthase immunoreactivity (NOS-IR) in inhibitory motor neurons to muscle. In consideration of the insufficient data regarding the enteric nervous system (ENS) of sheep, we investigated the myenteric plexus and submucosal plexus of the ovine ileum. Since the pivotal role of the ENS in the early pathogenesis of sheep scrapie, the “prototype” of prion diseases, has been suggested, we have focused our observations also on the host's PrP genotype. We have studied the morphology and distribution of NOS-IR neurons and their relationships with the enteric glia in whole-mount preparations and in cryostat sections. NOS-IR neurons, always encircled by glial processes, were located in both plexuses. Many NOS-IR fibers were seen in the circular muscle layer, in the submucosa, and in the mucosa. In the submucosa they were close to the lymphoid tissue. No differences in the distribution and percentage of NOS-IR fibers and neurons were observed among sheep carrying different PrP genotype, thus making unlikely their contribution in the determinism of susceptibility/resistance to scrapie infection.
T
The site of entry for most TSE agents is believed to be the GIT. The infection initially targets follicular dendritic cells before spreading to the nearby nerves to reach the CNS (Mabbott et al. 2003; Prinz et al. 2003). In this respect, it is not clear how neuroinvasion takes place, although many data support ENS involvement. In fact, PrPSc, the pathological isoform of the “cellular prion protein” PrPC representing the hallmark of TSEs, has been detected in the ENS of spontaneously and experimentally scrapie-infected sheep (Van Keulen et al. 1999; Andréoletti et al. 2000; Heggebø et al. 2003; Mabbott and MacPherson 2006) and in experimentally infected rodents (Beekes and McBride 2000; McBride et al. 2001; Mabbott and MacPherson 2006). Moreover, the dorsal motor nucleus of the vagus nerve that marks the origin of the parasympathetic efferent fibers of the enteric plexuses innervating the subdiaphragmatic tract of the digestive apparatus, with the exception of the colon-rectum in sheep (Chiocchetti et al. 2003), and the intermedio-lateral nucleus of the spinal cord, represent the earliest PrPSc deposition sites in the CNS in both natural and experimental scrapie infection (Beekes et al. 1998; McBride et al. 2001; Van Keulen et al. 2002), suggesting an early involvement of the parasympathetic and sympathetic efferent fibers innervating the GIT.
Prion diseases are often considered to be models for other neurodegenerative disorders such as Alzheimer's disease, which lacks a “true” animal model because these diseases share a number of clinical and histopathological features such as astrogliosis/astrocytosis, spongiform degeneration of neurons, and amyloid deposition in the brain. In such neurodegenerative disorders, a significant perturbation in brain metal concentrations, especially in Cu, and elevated levels of nitric oxide occur (for review, see Gaeta and Hider 2005). As in Alzheimer's disease (Norris et al. 1996), in the scrapie-infected brains of mice and hamsters, neurons expressing neuronal nitric oxide synthase immunoreactivity (NOS-IR) are selectively affected (Ovadia et al. 1996; Keshet et al. 1999; Picançco-Diniz et al. 2004). It has been suggested that PrPC can interact with NOS, playing a role in the targeting of NOS to its proper subcellular localization (Keshet et al. 2000). The enteric glial cells (EGCs) constitute the major reservoir of L-arginine, the substrate required for nitric oxide (NO) synthesis within the ENS; L-arginine may be transferred to neurons whenever necessary. In fact, ultrastructural examination has shown that L-arginine immunoreactivity is present in EGCs but not in neurons (Nagahama et al. 2001). EGCs are involved in the control of perineuronal homeostasis, regulation of gastrointestinal motility, and control of the ENS neuro-chemical phenotype (Aube et al. 2006). Moreover, they can directly interact with immune cells via cytokines (Cabarrocas et al. 2003), as demonstrated by the fact that acute intestinal inflammation is usually associated with an increase in glial fibrillary acid protein (GFAP) expression and proliferation of EGCs (von Boyen et al. 2004). In prion diseases, neuroglial cells appear to be potential reservoirs of infectivity. The presence of PrPC in glial cells has been demonstrated both in vivo (Moleres and Velayos 2005) and in vitro (Archer et al. 2004). Additional data suggest that astrocytes are among the targets for the scrapie agent in the CNS (Manuelidis et al. 1987; Lazarini et al. 1994; Titeux et al. 2002), with EGCs likely playing a similar role in ENS plexuses from spontaneously and experimentally scrapie-infected sheep (Chiocchetti et al. 2005; Marruchella et al. 2006).
The aim of the present study was to characterize the morphology and distribution of neuronal subpopulations residing in the MP and the SMP of the sheep ileum. We have also carefully taken into account the host's PrP genotype. In sheep scrapie, in fact, the clinico-pathological disease phenotype is determined not only by the agent's strain, but also by the host's PrP genotype (Di Guardo and Marcato 2002; Ersdal et al. 2003). In this respect, the ovine PrP gene (PRNP) carries several non-silent polymorphisms, expressed as the triplet sequences present at codons 136,154, and 171 (Agrimi et al. 2003) that, in turn, modulate susceptibility/resistance to scrapie. Recent studies carried out on Sarda breed sheep have demonstrated a positive correlation between glutamine/glutamine homozygosity at the level of PrP codon 171 (171 Q/Q) and scrapie susceptibility (Vaccari et al. 2001), similar to what has previously been reported in Suffolk breed sheep (Westaway et al. 1994).
We have studied the NOS-IR neurons of the sheep ileum MP and SMP, as well as their relationships with the enteric glia in whole-mount preparations, whereas in cryostat sections histochemically stained by means of nicotinamide adenine dinucleotide phosphate-diaphorase (NADPH-d), the vertical projections of NOS fibers were investigated. This histochemical stain is usually used to identify NOS neurons and fibers, being co-localized with NOS-IR in many species such as dog (Ward et al. 1992) and guinea pig (Young et al. 1992). Extensive overlap between the two methods has also been demonstrated in sheep (Pfannkuche et al. 2003).
Materials and Methods
Sacrifice and Surgical Procedure
Twelve female Sarda breed ovines ranging from 5 to 30 months of age were used in this study, namely, two 171 ARQ/ARQ (Q/Q homozygote, scrapie-susceptible) animals along with two 171 ARR/ARQ (R/Q heterozygotes) and two 171 ARR/ARR (R/R homozygote, scrapie-resistant) animals, all 5 months of age, as well as two additional sheep for each genetic group, 26–30 months of age, included as healthy controls in an experimental scrapie infection study. Animals were deeply anesthetized using IV administration of sodium thiopental (10 mg/kg) and euthanized by means of an intracardiac injection of Tanax (0.5 ml/kg; Intervet Italia, Milano, Italy).
Thirty days before euthanasia, the six 5-month-old animals underwent a surgical procedure to inject the fluorescent retrograde tracer Fast Blue (FB) into the ileum wall to identify neurons with descending projections. Animals were without food the day prior to surgery. Anesthesia was induced by IV administration of sodium thiopental (10 mg/kg) and maintained with isoflurane, nitrous oxide, and oxygen via a cuffed endotracheal tube. Following a midline laparotomy, the ileum was exposed and intramurally slowly infiltrated with 100 μl of FB in aqueous solution (2%), injected at multiple sites by means of a Hamilton microsyringe. Each injection was performed carefully to avoid any leakage of the tracer from the injected site. Ten injections of FB were made into a segment of the ileum ∼10 cm in length, beginning from the ileocecal junction. Animals were allowed to recover from surgery and began to eat hay on the first postoperative day. After a survival period of 3 weeks, they were euthanized as described above.
All above-mentioned procedures were carried out in agreement with Italian legislation on experimental animals, after approval by the Ethic-Scientific Committee for Experiments on Animals of the university of Bologna.
Whole-mount Processing
As soon as 15-cm segments of the ileum were collected, beginning at ∼2 cm oral to the ileocecal orifice, they were immersed in PBS, 0.15 M NaCl in 0.01 M sodium phosphate buffer, pH 7.2, containing the type-l calcium-channel blocker nicardipine as a muscle relaxant (10−6 M; Sigma-Aldrich Chemie, Steinheim, Germany) for 15 min. Ileal segments were then cut open along the mesenteric border, and the tissues were vigorously flushed out with PBS and pinned tautly on balsa wood, the mucosal surface facing down. Specimens were subsequently fixed in 2% paraformaldehyde containing 0.2% picric acid in 0.1 M sodium phosphate buffer (pH 7.0) at 4C overnight. Afterwards, they were removed from the balsa wood and washed in dimethylsulfoxide (3 × 10 min), followed by washing in PBS (3 × 10 min). All tissues were stored at 4C in PBS containing sodium azide (0.1%) until they were processed to obtain whole-mount preparations of the MPs and the SMPs, as previously described in detail (Chiocchetti et al. 2004).
Cryosections and NADPH-d Staining
In the 5-month-old animals, additional cryosections of the ileum were prepared to determine the vertical projections of NO-synthesizing neurons and their presence in both muscle layers and the mucosa by means of the NADPH-d histo-chemical stain that, as stated elsewhere, marks NOS-IR enteric neurons (Chiocchetti et al. 2004). One-cm-wide ileal segments were collected orally to whole-mount samples and immersed overnight in 4% paraformaldehyde. Paraformaldehyde has been demonstrated to inhibit other diaphorase activities while enhancing formazan deposition in NADPH-d positive cells (Buwalda et al. 1995). On the following day, tissues were transferred to a mixture of PBS-sucrose-azide (PBS containing 0.1% sodium azide and 30% sucrose as cryoprotectant) and stored at 4C overnight. Tissues were then immersed in a mixture of PBS—sucrose-azide and optimal cutting temperature compound (OCT; Tissue Tek, Sakura Finetek Europe, Zoeterwoude, The Netherlands) at a ratio of 1:1 for an additional 24 hr before being embedded in 100% OCT and frozen in isopentane cooled in liquid nitrogen. Fifteen-μm-thick transverse and longitudinal sections were cut, placed onto poly-lysine-coated slides, and left to dry at room temperature before incubating the tissues for 45 min at 37C in a 0.1 M Tris-HCl buffer (pH 8) plus 0.3% Triton containing 0.6 mM nitroblue tetrazolium and 2.2 mM β-NADPH, according to Grozdanovic et al. (1992).
The NADPH-d reaction was also performed in some sections that were first immunohistochemically stained to reveal NOS-IR by an indirect immunofluorescence method (see below).
Immunohistochemistry
Double-labeling studies using the indirect immunofluorescence method were performed in whole-mount preparations. Table 1 summarizes the antibodies used and their dilutions. To count the neurons, an anti-Hu antibody was utilized. Hu antigens are the human members of the ELAV-like RNA-binding protein family (Szabo et al. 1991). We have previously shown that Hu is a pan-neuronal marker of sheep enteric neurons (Chiocchetti et al. 2004). Glial fibrillary acid protein (GFAP), a component of astroglial intermediate filaments, was utilized to identify EGCs.
List of antibodies used in this study
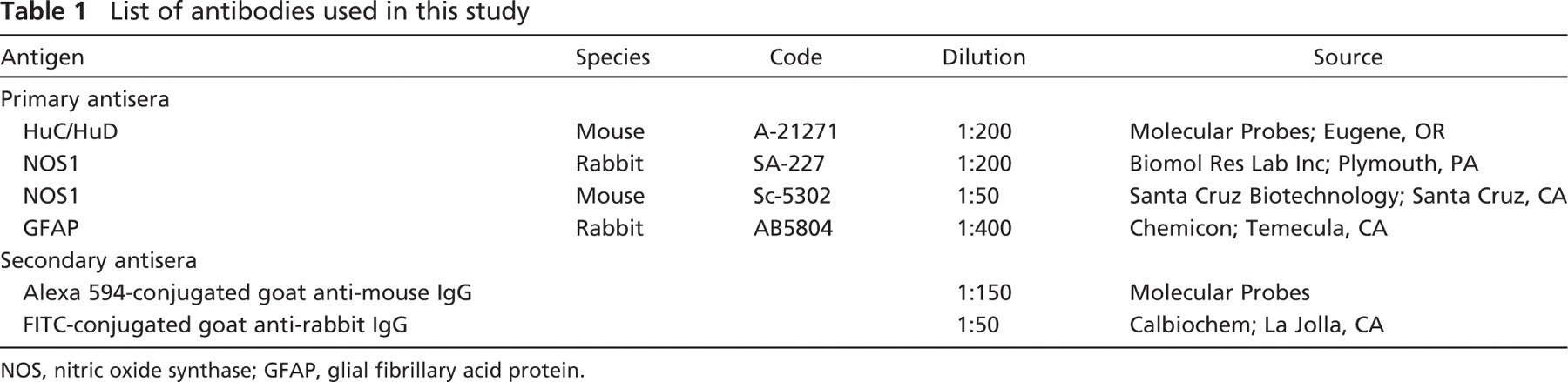
NOS, nitric oxide synthase; GFAP, glial fibrillary acid protein.
Tissues were incubated in 10% normal goat serum in PBS containing 1% Triton for 30 min at room temperature to reduce nonspecific binding of the secondary antibodies and to permeabilize the tissue to the antisera. Tissues were then incubated at 4C in a humid chamber for 2–3 days in a mixture of the following primary antibodies diluted in a suitable medium (1.8% NaCl in 0.01 M phosphate buffer containing 0.1% Na-azide): NOS/Hu, GFAP/Hu, GFAP/NOS. After washing in PBS (3 × 10 min), tissues were incubated for 3 hr at room temperature in a humid chamber in a mixture of the secondary antibodies diluted in PBS: goat anti rabbit-IgG fluorescein isothiocynate (FITC)-labeled and Alexa 594-labeled goat anti-mouse IgG. The tissues were then washed in PBS (3 × 10 min) and mounted in buffered glycerol (pH 8.6).
Two whole-mount preparations of the MP and SMP were taken from each animal and used for each double-staining protocol.
Specificity of the anti-NOS antibody was tested by Pfannkuche et al. (2003). Specificity of the anti-GFAP antibody has been investigated in other species (Hammerle et al. 2003; Jüngling et al. 2003) and, as it is raised against highly conserved peptides epitopes, it also recognizes the appropriate antigens in sheep. Specificity of the secondary antibodies was tested by applying these antisera without use of the primary antibodies. No stained neurons and fibers were seen after omitting the primary antisera.
Fluorescent Microscopy and Morphometrical Analysis
Preparations were examined on a Zeiss Axioplan microscope equipped with the appropriate filter cubes to discriminate between FITC and Alexa 594. Images were recorded with a Polaroid DMC digital photocamera (Polaroid; Cambridge, MA) and DMCV 2 software.
Images were further processed using Corel Photo Paint and Corel Draw software programs (Corel; Milan, Italy). Morphometrical analyses were undertaken with a × 40 objective lens using KS 300 image analysis software (Kontron Electronik; Munich, Germany) connected to the microscope. The cross-sectional areas (μm2) of 100 NOS-IR somata from the plexus of each animal were measured by outlining somata profiles on the monitor screen using a computer mouse. To determine the proportions of NOS-IR neurons, at least 1000 Hu-IR cells were counted in the whole-mount preparations of the MP and SMP from each animal. Percentages of neurochemically identified subpopulations, expressed as mean ± standard deviation, were calculated and expressed relative to the number of Hu-IR neurons, which was considered to represent 100% of the enteric neurons. Two whole-mount preparations of the MP and SMP taken from each animal were examined.
For statistical analysis, both the cross-sectional area of the somata and the percentage of the NOS-IR neurons were first tested for normality using the Shapiro-Wilk's W test. Because the data did not show a normal distribution, comparison among the three genetic groups and between the MPs and the SMPs of each genetic group was carried out using the Kruskall-Wallis ANOVA median test.
Analyses were performed using CSS software (Statsoft; Tulsa, OK) statistics, with a conventional 5% level being used to define statistical significance.
Results
NOS-IR in the MP
As previously reported elsewhere (Chiocchetti et al. 2004), Hu is a pan-neuronal marker and also in the present study all NOS-IR neurons always coexpressed Hu immunoreactivity. NOS-IR was detected in the cytoplasm and the axon of the neurons and, occasionally, some short lamellar processes arising from the irregular somata profile could also be seen.
A dense network of NOS-IR fibers with small varicosities occurred in the MP. Bundles of NOS-IR fibers were arranged within the primary, secondary, and tertiary nerve strands (Figures 1A-1C). The largest primary nerve strands were preferentially oriented parallel to the main axis of the longitudinal muscle layer and sometimes forked or bent without showing an apparent directionality. The primary nerve strands were joined by the secondary nerve strands, often oriented perpendicularly to the main axis of the primary nerve strands. The tertiary nerve strands were oriented both parallel and perpendicular to the secondary nerve strands and crossed to form a delicate interwoven meshwork (Figures 1B and 1C). NOS-IR fibers showed varicosities along the smallest secondary and tertiary interconnecting nerve strands and within the ganglia in which they made baskets around both NOS-negative and -positive neurons (Figure 2A).
In the sheep ileum, it was difficult to determine the boundaries of the individual large ganglia. They were apparently formed by the fusion of many ganglia, making it difficult to evaluate the number of neurons of the individual ganglia. Following Hu immunostaining, large ganglia appeared as polygonal or ring-like structures. Large elongated ganglia with their major axis perpendicular to the underlying longitudinal muscle bundles were also found. Generally, large ganglia containing up to 25 NOS-IR neurons were interconnected by primary and secondary nerve strands (Figures 1A and 1C), whereas small ganglia containing up to four to six NOS-IR neurons were generally interconnected by secondary and tertiary nerve strands (Figure 1B). Isolated neurons were occasionally observed along thin nerve strands (Figure 1B). NOS-IR neurons were observed in almost all the ganglia, and there was generally a direct correlation between the total number of neurons per ganglion and the number of NOS-IR neurons. The mean value of the percentage of NOS-IR neurons ranged from 31% in the 171 R/R group to 36% in the 171 R/Q group (see Table 2). Statistically significant differences could not be detected in the percentage of NOS-IR neurons from the MPs of sheep carrying different PrP genotypes.
NOS-IR neurons showed great variability both in their size and morphology; in the ganglia they were widespread or grouped in clusters (Figures 1A and 1C).
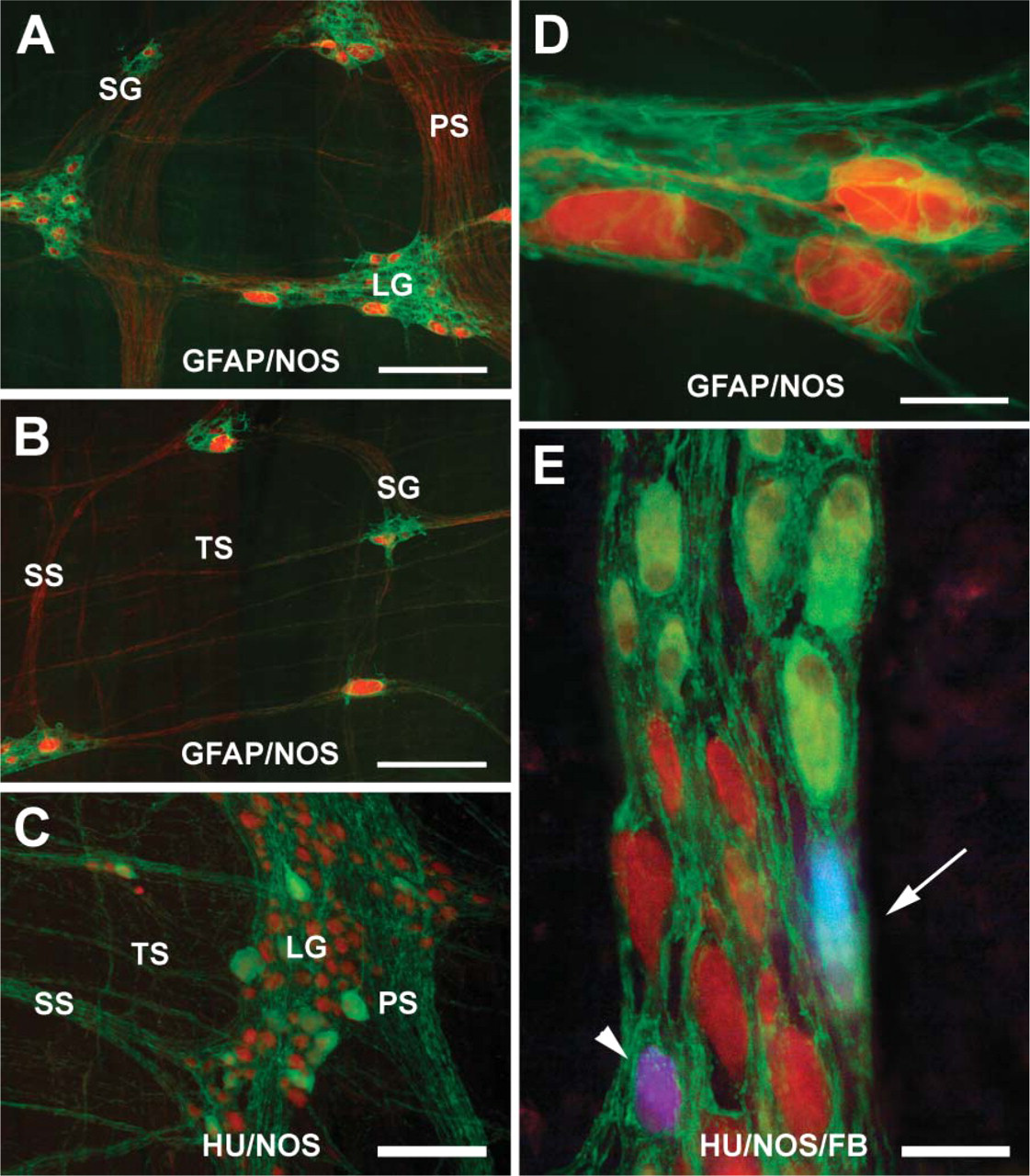
Photomicrographs illustrating the colocalization of particular antigens in the myenteric plexus (MP) of 26-month-old sheep (
Many neurons exhibited an irregular outline and a round NOS-negative nucleus, generally eccentrically located. The smallest cells (rarely the largest ones) showed a central nucleus (Figures 2A and 2B). The vast majority of neurons exhibited an ovoid shape and variability in size (Figures 2A-2C). In these neurons, a long process often arose from the side opposite the nucleus, being directed either anally or, rarely, orally. The process sometimes arose from the somata surface without showing a particular direction (Figure 2C). Round and polyhedral cells were also found intermingled with ovoid ones, generally showing low immunoreactivity and small size (Figures 2A and 2C). Elongated neurons with a narrow profile were observed either intermingled with the ovoid neurons or isolated along the smallest nerve strands, and a long process, directed anally, could often be seen starting from the side opposite the nucleus (Figure 2D)
NOS-IR in the MP of 5-month-old sheep. (
Neurons showed a wide variation in size, their cross-sectional area ranging between 140 and 1723 μm2 Sheep carrying the 171 R/R genotype exhibited a statistically significant greater mean value (p>0.01) when compared with the 171 R/Q genetic group, whereas no statistically significant differences were observed between the 171 R/Q and the 171 Q/Q genetic groups (Table 2). In all genetic groups, neurons exhibiting a cross-sectional area ranging from 150 μ to 600 μm2 were prevalent; however, in the 171 R/R genetic group there was a greater percentage of neurons with a cross-sectional area >450 μm2 (Figure 3A).
Percentage and cross-sectional area of NOS-IR neurons in the MP and SMP

Values are expressed as mean ± SD.
NOS-IR, nitric oxide synthase immunoreactivity; MP, myenteric plexus; SMP, submucosal plexus.
Glial cells and processes were numerous in the ganglia and in the connecting nerve strands of the plexus, with all NOS-IR neurons being encircled by glial processes (Figures 1A, 1B, and 1D); however, NOS-negative cells were sometimes found located on the borders of the ganglia without an apparent glial coat (data not shown).
NOS-IR in the SMP
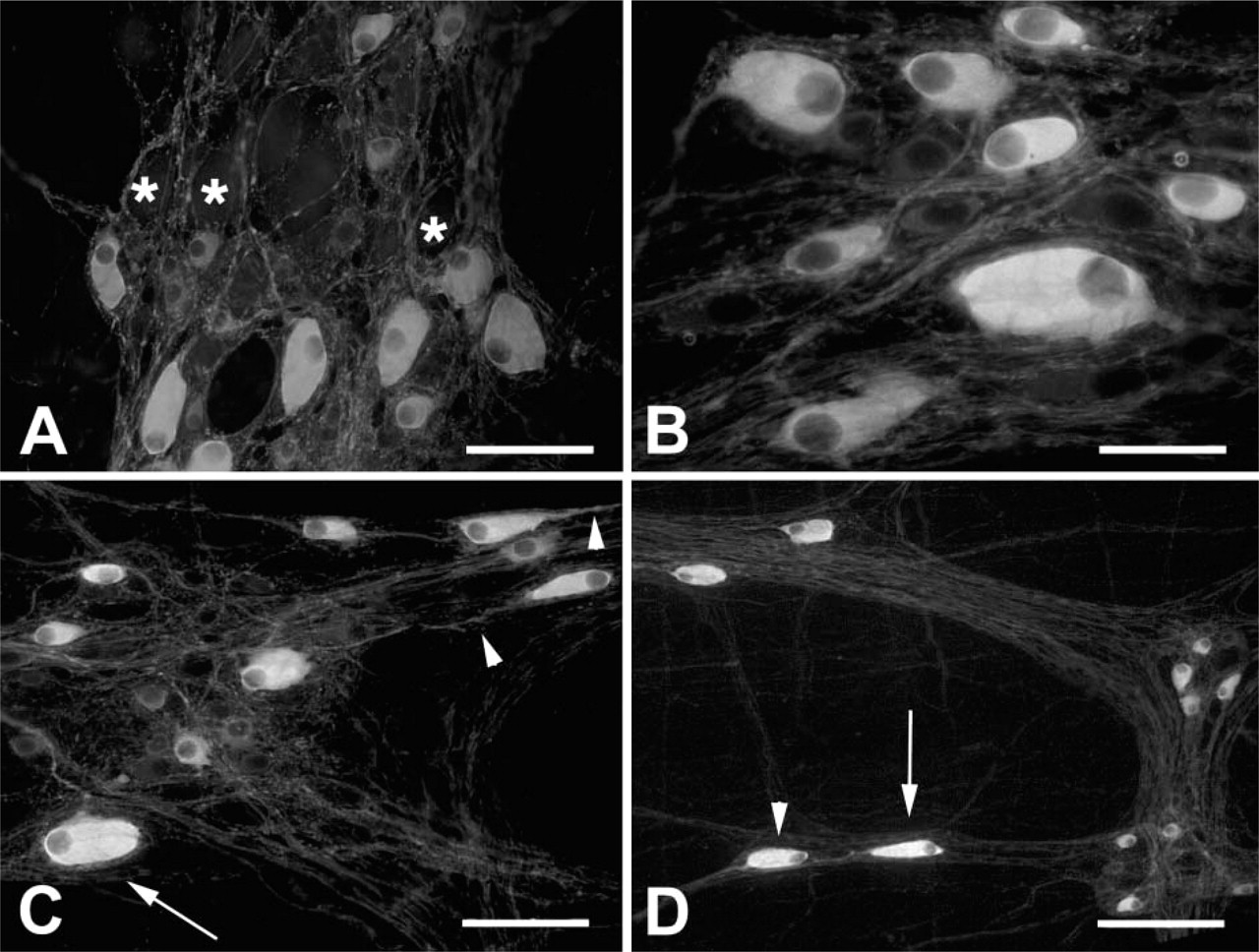
As in large animals, the SMP of the sheep ileum is composed of two separate but interconnected layers of the nerve network, separated by the submucosal vascular arcade. However, in the submucosa of younger animals, a large amount of lymphatic tissue coated by a thick layer of connective tissue makes it difficult to accurately separate the two SMPs. Counting the neurons, percentages in the whole-mount preparations from the SMPs of two adult animals in which the two SMPs were accurately separated revealed a topographically distinct distribution of NOS-IR neurons. In fact, they predominated in the ESP, whereas only 1–2% were counted in the ISP. For this reason, in the present study we did not consider the two plexuses separately.
NOS-IR fibers showing varicosities were immersed in a dense matrix of connective tissue, generally being arranged to form thin interconnected bundles, and few fibers were seen to be isolated. NOS-IR fibers with large varicosities often made baskets around the NOS-negative neurons (Figure 4A). Large ganglia were found at the intersections of the large nerve strands (Figure 4B) with up to 41 neurons being counted in the large ganglia. Small clusters of two to four neurons or isolated neurons were seen dispersed along the thin bundles. Many neurons showed an irregular profile (Figure 4C). Sometimes in the ovoid neurons, a long process arising from the side opposite to the nucleus could be followed anally for hundreds of microns (Figure 4D), and sometimes a long process arose without showing a particular direction. The mean value of the percentage of NOS-IR neurons ranged from 22% in the 171 R/Q genetic group to 24% in the 171 R/R and Q/Q genetic groups, with no statistically significant differences (see Table 2). More in detail, for each comparison four animals were included in every single study group, and on the basis of the results obtained by means of the Spearman's rank correlation test, it was shown that age does not influence the parameters under investigation.
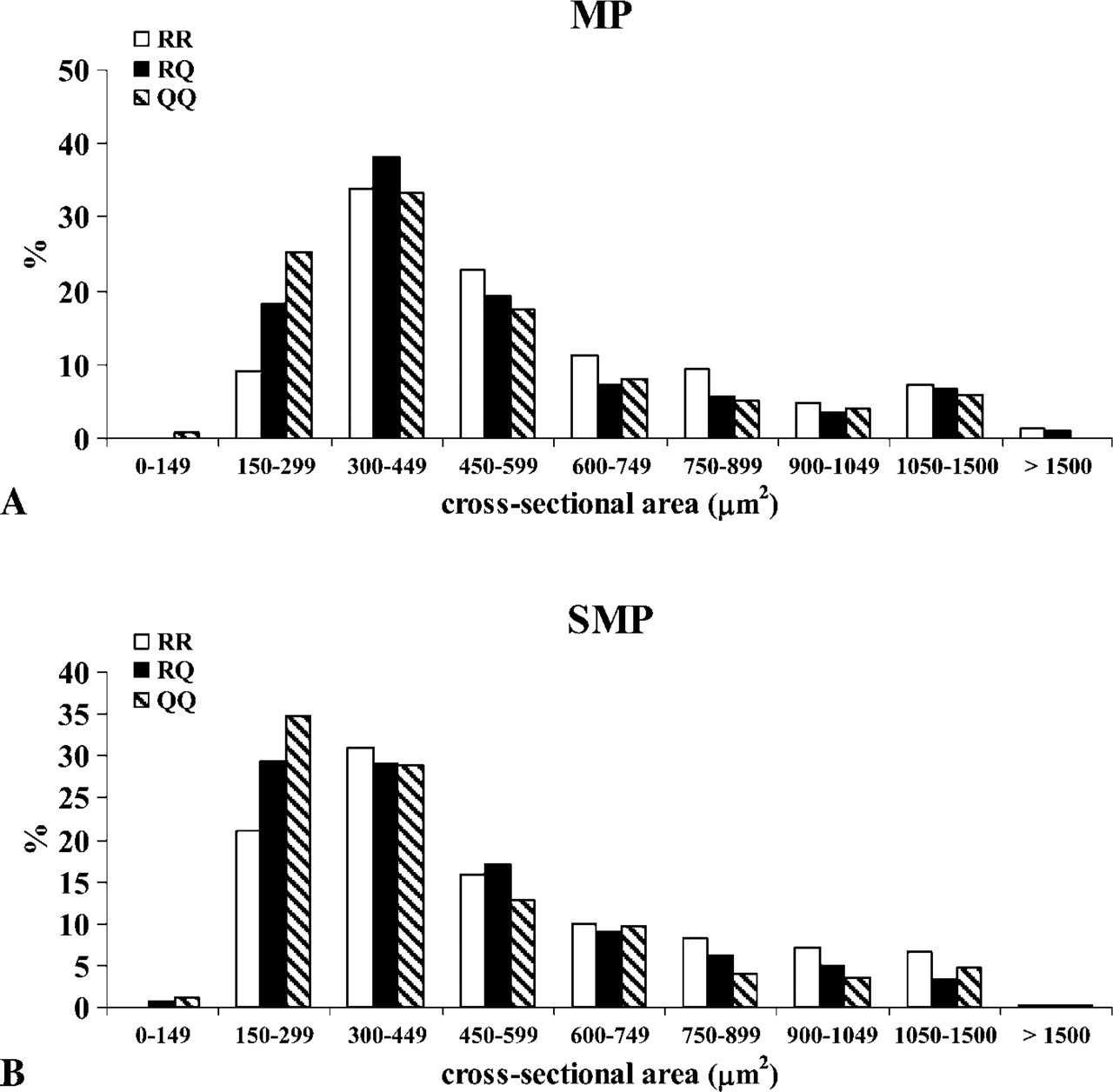
Histogram showing the distribution of the neuronal sizes (cross-sectional area) of NOS-IR neurons within the MP (
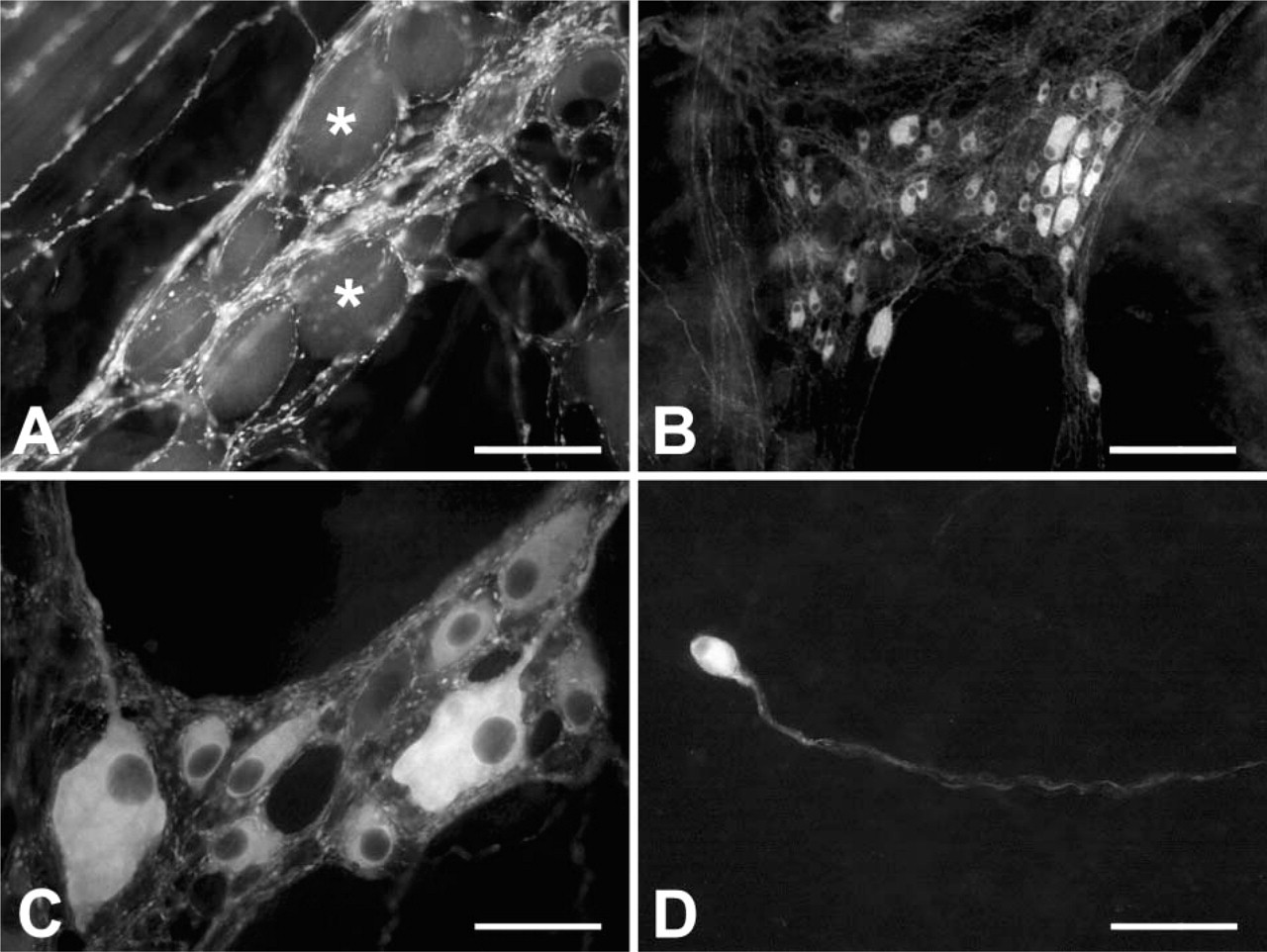
NOS-IR in the SMP of 5-month-old sheep. NOS-IR fibers with large varicosities make baskets around the NOS-negative neurons (asterisks in
Similar to what has been reported for the MP, in the SMP, NOS-IR neurons also showed a great variability both in size and morphology (Figures 4B and 4C). Ovoid neurons were prevalent and were intermingled with round, polyhedral, or fused neurons. Not all ganglia showed NOS-IR neurons that were unevenly widespread or grouped in clusters. In all genetic groups, the mean value of the cross-sectional area was smaller in a statistically significant manner (p<0.01) than that in the corresponding MP (Table 2). Sheep carrying the 171 R/R genotype exhibited a statistically significant greater mean value (p<0.01) when compared with the 171 R/Q and 171 Q/Q groups, whereas no statistically significant differences were observed between the 171R/Q and 171 Q/Q genetic groups. In all genetic groups, neurons exhibiting a cross-sectional area ranging from 150 μm2 to 450 μm2 were prevalent; however, in the 171 R/R group the percentage of neurons with a cross-sectional area >450 μm2 was greater in comparison to the 171 R/Q and 171 Q/Q groups (Figure 3B).
Glial processes were numerous in the ganglia and in the strands of the plexus, with all NOS-IR neurons constantly encircled by glial processes; however, it was not infrequent to see NOS-negative neuron cells without glial coat (data not shown).
NADPH-d Staining
Immunohistochemical stain to reveal NOS-IR and the NADPH-d histochemical stain marked the same neurons and fibers. However, with the immunohistochemical stain some background punctate immunoreactivity was observed in the mucosa and in the lymphatic submucosal tissue, probably related to macrophages exhibiting a natural fluorescence (Defaweux et al. 2005). Therefore, for detailed morphological studies only NADPH-stained sections were used.
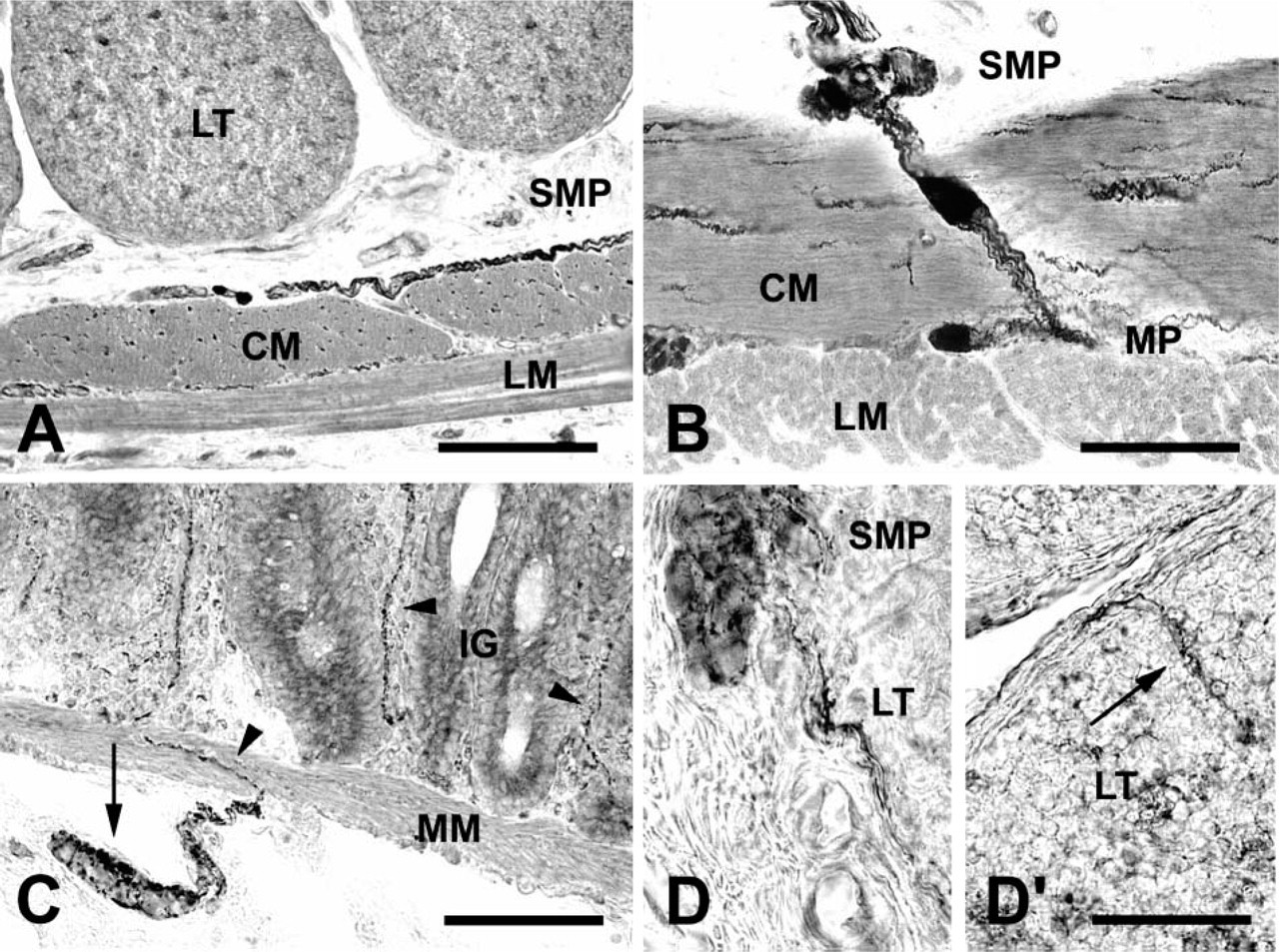
NADPH-d-positive fibers with prominent varicosities were numerous in the internal muscle layer where they were grouped into small thin bundles running parallel to the circular muscle cells. Few fibers were seen in the external muscle layer (Figures 5A and 5B). Bundles of NADPH-d-positive fibers crossing the internal muscle layer could be seen connecting the ganglia of the SMP and the MP, and it was also possible to see NADPH-d-positive neurons located in the middle of the circular muscle layer (Figure 5B). In the ISP, fibers formed baskets around clusters of NADPH-d-negative neurons located beneath the muscularis mucosae. Fibers then crossed the muscularis mucosae to reach the mucosa where they were found distributed around the intestinal glands and along the main axis of the villi (Figure 5C). Some fibers were also seen in the thin layer of connective tissue encircling and penetrating the gut-associated lymphoid nodules (Figure 5D).
NADPH-d-positive neurons were seen isolated or grouped in small clusters in the MP and in the ESP where they were preferentially adjacent to the circular muscle layer, whereas few neurons were located in the ISP.
No differences were observed among the animals under study in relation to their PrP genotype.
FB Labeling
In the six 5-month-old sheep, whole-mount preparations were obtained from ∼1cm to ∼4 cm oral to the FB fluorescent tracer injections. We avoided considering the whole-mount tissue in which FB spreading occurred, as revealed by FB marking of lymphoreticular cells. For this analysis, animals of the three genetic groups under study were considered together, and data were not evaluated statistically.
We found FB-labeled neurons up to 4 cm orally to the FB injection sites. In the MP, the mean value of the percentage of FB-labeled neurons was 3.5% of the total number of neuronal cell populations (174 FB-labeled neurons/5188 Hu-IR neurons were counted), ranging from 6.2% to 1.5%. In the SMP, the mean value of the percentage of FB-labeled neurons was 4% of the total number of neuronal cell populations (230 FB-labeled neurons/5706 HU-IR neurons were counted), ranging from 1.5% to 8.4% (Table 3). There was a direct correlation between the total number of FB-labeled neurons and the distance from the FB injection sites. In fact, the greatest values were found 1 cm from the FB injection sites, whereas the smallest values were found 4 cm orally to the FB injection sites. Approximately 67% of the FB-labeled neurons in the MP and 46% in the SMP showed NOS-IR (Table 3), and there was no apparent correlation between the distance from the injection sites of the FB tracer and the percentage of FB/NOS-IR neurons. The greatest majority of FB/NOS-IR cells exhibited an ovoid or fused shape in both plexuses (Figure 1E), with their cross-sectional area showing a greater variability as reflected in the large standard deviation (Table 3). SMP neurons, both FB and FB/NOS-IR, were smaller than MP-labeled neurons. We observed a direct correlation between the size of FB-labeled neurons and the distance from the FB injection sites. In fact, the greatest mean value of the cross-sectional area was observed at 4 cm (1147 ± 363 μm2 in the MP and 881 ± 329 μm2 in the SMP), whereas the smallest mean value (735 ± 280 μm2 in the MP and 467 ± 260 μm2 in the SMP) was observed at 1 cm. Analogously, the greatest mean value of the cross-sectional area of the FB/NOS-IR neurons was observed at 4 cm (1210 ± 330 μm2 in the MP and 899 ± 341 μm2 in the SMP), whereas the smallest mean value (685 ± 348 μm2 in the MP and 623 ± 231 μm2 in the SMP) was observed at 1 cm. In both plexuses, >50% of the neurons showed a cross-sectional area >600 μm2 (Figures 6A and 6B).
General appearance of the ileum of 5-month-old sheep in longitudinal (
Percentage and cross-sectional area of FB and FB-labeled neurons in the MP and SMP
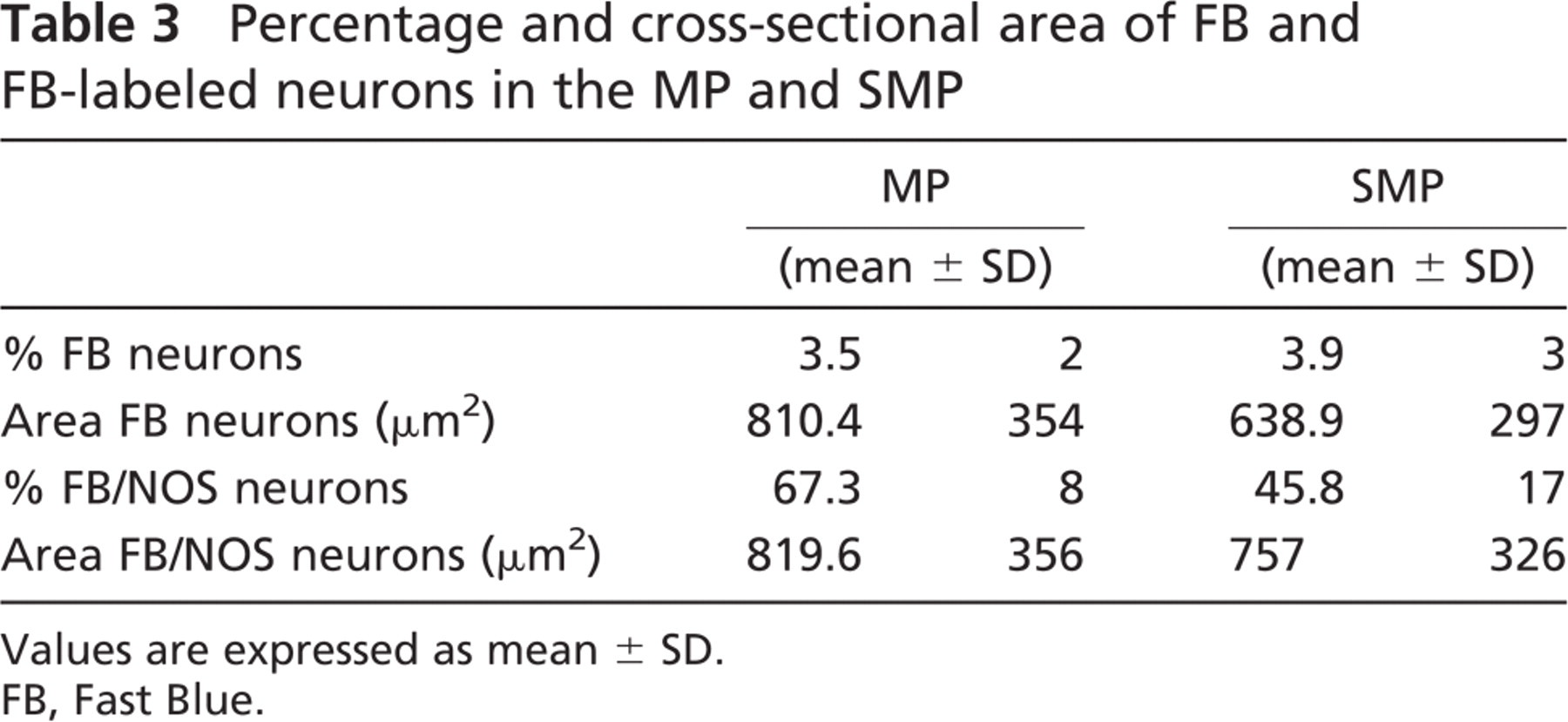
Values are expressed as mean ± SD.
FB, Fast Blue.
No differences were observed among animals under study in relation to their PrP genotype.
Discussion
This article describes the distribution of NOS-IR neurons and fibers in the ENS of Sarda breed sheep carrying different PrP genotypes. The plexuses were investigated by means of double-immunofluorescent stain in whole-mount preparations and in cryostat sections histochemically stained by NADPH-d. To date, no satisfactory description of ovine ENS plexuses is available, with the exception of the investigation of Gunn (1968) and of more recent studies by Chiocchetti et al. (2004, 2006). A greater knowledge is necessary because of the pivotal role of the ENS in functional and structural gut disorders as well as in the early pathogenesis of sheep scrapie and other prion diseases (Mabbott and MacPherson 2006). Data concerning the guinea pig and the rat are not interchangeable (Sayegh and Ritter 2003), and the differences between small laboratory animals and large mammals stress the importance of research in different species. The general organization of sheep ENS plexuses showed both similarities and differences between large and small laboratory animals. As in cattle (Balemba et al. 1999), in the sheep ileum, it was difficult to determine the boundaries of individual large ganglia that are often of variable shape with no apparent directionality; however, some elongated ganglia with the main axis perpendicular to the underlying longitudinal muscle layer and resembling those of pigs (Scheuermann et al. 1986) could be seen. The main axis of the primary nerve strands is generally oriented parallel to the longitudinal muscle layer as it occurs in the pig; however, primary nerve strands without showing an apparent directionality were also found. As in large mammals, the SMP is composed of two separate but interconnected layers, the ISP and the ESP, with a different distribution of neurons expressing NOS-IR, probably reflecting two distinct functions related to SMP plexuses.
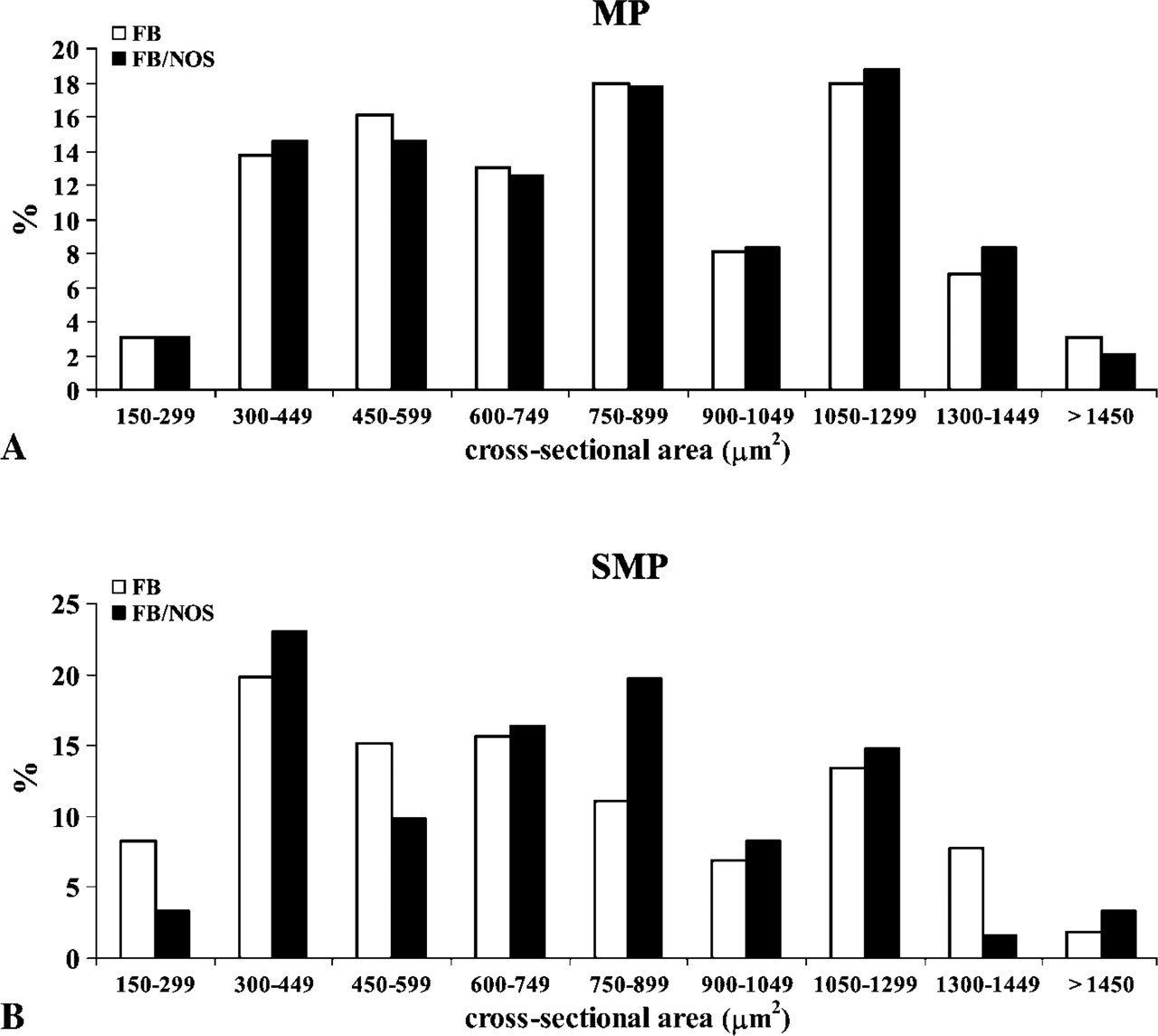
Histogram showing the distribution of neuronal sizes (cross-sectional area) of FB-positive and FB/NOS-IR neurons within the MP (
NO is considered the putative major mediator of non-adrenergic, non-cholinergic smooth muscle relaxation. In all mammals so far investigated, NO participates in inhibitory transmission and NOS (or NADPH-d) is always contained in inhibitory neurons innervating the muscle. In many cases it does not appear to be a primary transmitter and is often colocalized with other putative inhibitory neurotransmitters such as vasoactive intestinal peptide (Chiocchetti et al. 2006) or γ-aminobutyric acid (Furness 2000). NO has been found in both circular and longitudinal inhibitory muscle motor neurons, in interneurons, in intestinofugal neurons, and in submucosal projections controlling electrolyte secretion (Schemann and Neunlist 2004). We have found a consistent number of nitrinergic neurons in both the MP and SMP. Whereas in the guinea pig, NOS-IR neurons are absent from the SMP except for the large intestine (McConalogue and Furness 1993), in the sheep ileum a consistent percentage of nitrergic neurons was found in the SMP and, as in pigs and humans, were more numerous in the ESP than in the ISP (Timmermans et al. 1994; Porter et al. 1999). Thus, as in large animals, also in sheep, the ESP is more similar to the MP than to the ISP (Timmermans et al. 2001).
Although we have not used a cytoskeletal neuronal marker to best define cell morphology, many NOS-IR neurons exhibited an irregular profile and could be referred to as Dogiel type I. This assumption is strongly supported by previous data provided by our group (Chiocchetti et al. 2006) showing that the Dogiel type I-like neurons (identified in both the MP and SMP) do not express CGRP, a peptide typically contained in Dogiel type II-like intrinsic sensory neurons of the sheep enteric nervous system.
NOS-IR neurons showed a great variability both in size and morphology, probably reflecting different functional subclasses of neurons. A number of nitrinergic neurons within both plexuses could be considered descending neurons; in fact, a consistent number of FB-labeled neurons expressed NOS-IR. They were generally ovoid or fused, large-sized neurons that were more distant from the injection sites of FB into the ileum wall, having had a larger cross-sectional area than did neurons closer to the injection sites. By excluding that neurons projecting anally are Dogiel type II sensory neurons, these cells could be either interneurons or descending inhibitory motor neurons for the circular muscle layer. Interneurons and motor neurons have similar basic properties, having often been identified by excluding other known classes of cells (Bornstein et al. 2004). However, retrograde labeling techniques in organotypic cultures combined with intracellular recording and dye filling have revealed that motor neurons have shorter projections when compared with interneurons, both in guinea pigs and humans (for review, see Brookes 2001). Thus, it seems plausible that, also in sheep, neurons with longer projections having the largest somata cross-sectional area might be descending interneurons, whereas neurons with a shorter projection might be descending inhibitory motor neurons.
In sheep MPs nearly all Hu-IR neurons were encircled by glial processes, whereas in SMPs, some neurons located on the borders of the ganglia lacked a complete glial coat. EGCs represent the morphological and functional equivalent of CNS astrocytes, making a network with trophic and protective functions toward enteric neurons. EGCs act as an intermediary cell type between blood vessels not present in the ENS ganglia and enteric neurons (Gabella 1982). They also extend into the mucosal glands, blood vessels, and tips of the villi, sheathing the neuronal processes involved in the control of mucosal secretions (Cabarrocas et al. 2003). EGCs usually encircle neurons and their processes; however, the glial coat is sometimes incomplete and neurons can make direct contact with the extraganglionic connective tissue (Gabella 1972). Nevertheless, in our study, NOS-IR neurons always exhibited a glial coat in both plexuses, probably due to the role of EGCs as reservoirs of L-arginine required for NO synthesis.
In cryostat sections, neurons in the ESP sending their axons into the circular muscle layer could be seen. Based on these observations, one may reasonably assume that, in sheep, as in dogs, pigs, and humans, NOS-IR neurons of the ESP are involved in the inhibitory innervation of the circular muscle layer (Sanders and Smith 1986; Timmermans et al. 1997; Porter et al. 1999), in contrast to guinea pigs in which all motor neurons of the circular muscle layer are found exclusively in the MP (Wilson et al. 1987).
We detected many NADPH-d fibers in the inner circular muscle layer, in the submucosa, and in the mucosa, with very few fibers being observed in the longitudinal external muscle layer. NADPH-d fibers could also be seen connecting ganglia of the SMP with ganglia of the MP, suggesting an interneuronal function, or otherwise crossing the muscularis mucosae and being directed toward the mucosa after making baskets around the NADPH-d negative neurons located in the ISP. Not all NADPH-d (or NOS-IR) fibers had an intrinsic origin. In fact, after fluorescent tracer injection in the sheep ileum we observed in the dorsal root ganglia some fluorescent-labeled afferent neurons that were NOS-IR (unpublished data). Such primary afferent neurons may represent the source of some intramural NADPH-d (or NOS-IR fibers). NADPH-d fibers could also be seen connecting ganglia of the SMP with ganglia of the MP, suggesting an interneuronal function, or otherwise crossing the muscularis mucosae and being directed toward the mucosa after making baskets around the NADPH-d negative neurons located in the ISP. In the mucosa, NADPH-d positive fibers encircled the intestinal glands and were distributed along the lamina propria of the villi, indicating their involvement in the control of the mucosal function. The role of NO as an important mediator involved in the regulation of the electrolyte intestinal transport system has been discussed by Izzo et al. (1998) in many animals models including mice, rats, and guinea pigs. The above-mentioned authors have demonstrated mixed secretory and absorptive roles, with both species-specific and anatomic variations under physiological and pathophysiological conditions. King et al. (2004) demonstrated that NO is the neurotransmitter in the rat colon, which induces a chloride secretory response to serotonin.
In ileal cryostat sections obtained in the 5-month-old sheep, a large amount of lymphoid tissue was still present. In this respect, it is well known that in the gut of young ruminants there are long, continuous lymphoid tissue aggregates, Peyer's patches (PPs), which extend from the distal jejunum to the ileocecal junction. Whereas jejunal PPs persist into adult life and are involved in mucosal immunity, ileal PPs function as a primary lymphoid tissue, being responsible for the generation and maturation of the majority of B cells, and undergo involution about the time of sexual maturity (for review, see Press et al. 2004). We observed NADPH-d fibers located both in the thin layer of surrounding connective tissue and penetrating individual lymph nodules, which might facilitate contact between nerve components and lymphoid cells such as dendritic cells and follicular dendritic cells, thus supporting prion neuroinvasion (Aguzzi and Polymenidou 2004; Defaweux et al. 2005; Mabbott and MacPherson 2006). In sheep scrapie, in fact, the entry site of the infectious agent into the peripheral nerve fibers may be the mantle zone of the lymphoid follicles, because in this region follicular dendritic cell processes are in close contact with nerve fibers (Glatzel et al. 2000; Heggebø et al. 2003). In healthy and scrapie-infected sheep, electron microscope studies carried out by Heggebø et al. (2003) have identified fibers within the lymphoid nodules of jejunal and ileal PPs, usually associated with follicular dendritic cells. However, the neurochemical code of these fibers, which may be of relevance for prion neuroinvasion, has not been defined. Nevertheless, recent evidence has been provided that, in ARQ/ARQ homozygous Sarda breed sheep with natural scrapie, quite a relevant percentage of animals may develop infection without an apparent involvement of their lymphoreticular tissues, including PPs (Ligios et al. 2006). Furthermore, an apparent lack of lymphoreticular tissue involvement has been constantly reported also in the course of “atypical” scrapie (Benestad et al. 2003), which has also recently been described in ARR/ARR homozygous sheep, a “scrapie-resistant” PrP genotype included in the present study (Le Dur et al. 2005).
Despite what has been previously stated, to the best of our knowledge this is the first time that NOS fibers have been described in sheep PPs. We cannot state, at the moment, whether they originate from dorsal root ganglia and, if they do, could represent a route to directly transport the infectious agent to the CNS during the early phases of scrapie infection.
We did not observe in any case, as well as by any of the staining methods used in the study animals, statistically significant differences in the percentage of NOS-IR neurons among sheep carrying different PrP genotypes, although in the 171 R/R genetic group NOS-IR neurons exhibited a significantly larger somata cross-sectional area. At present, we cannot explain the relationships, if any, between a resistant PrP genotype and a larger somata cross-sectional area in the animals investigated here. We cannot, however, rule out a functional significance of the latter finding. Nevertheless, the limited number of animals investigated here prevents us from drawing firm conclusions from the above findings.
As far as the host's PrP genotype is concerned, recent studies carried out on Sarda breed sheep have clearly shown a positive correlation between glutamine/glutamine homozygosity at the level of PrP codon 171 (171 Q/Q) and scrapie susceptibility. This also demonstrates a direct correlation between arginine/arginine homozygosity (171 R/R) and resistance to the disease (Vaccari et al. 2001; Agrimi et al. 2003), similar to what has been previously reported in Suffolk breed sheep (Westaway et al. 1994). Currently, there is an open debate on the biological determinants upon which genetic susceptibility and resistance to scrapie are based, with the levels of endogenous PrPC expression on behalf of target cells being a crucial prerequisite (Halliday et al. 2005; Thackray et al. 2007), although exceptions to this rule have been reported (Aguzzi 2003). The complex sequence of pathogenetic events occurring throughout the long journey of the infectious agent from peripheral tissues to the CNS, so called “neuroinvasion”, has been only partially elucidated (Aguzzi and Polymenidou 2004; Mabbott and MacPherson 2006). In this respect, lack of evidence still exists on the possibility that the host's PrP genotype may influence a more or less efficient uptake of the agent from the intestinal lumen, as well as on the possibility, for instance, that genetic susceptibility/resistance may be dependent upon the rate of PPs innervation. At this time, this has been hypothetically linked to PPs colonization on behalf of the scrapie agent (Heggebø et al. 2003). The demonstration, for the first time, of NOS-IR fibers within PPs of the sheep investigated here adds an important piece of information with reference to the potential use of the above anatomic structures on behalf of the scrapie prion during neuroinvasion.
In conclusion, in addition to the presence of NOS-IR fibers in the gut-associated lymphatic tissue, which have been documented for the first time in Sarda breed sheep carrying different PrP genotypes, our data suggest that more than one subclass of NOS-IR neurons is detectable in the sheep ileum. Finally, albeit NOS-IR neurons residing within ileal ENS plexuses have been recently shown by our group to harbor more or less consistent PrPSc deposits in the course of natural scrapie infection in Sarda breed sheep (Marruchella et al. 2006), the fact that no differences were observed in the percentage and distribution of NOS-IR neurons and fibers among sheep carrying different PrP genotype makes unlikely the contribution of the above factors in the determinism of susceptibility/resistance to sheep scrapie infection.
Footnotes
Acknowledgements
This study was supported by a grant (PRIN 2004) from Ministero dell'Istruzione, dell'Università e della Ricerca and by an additional grant from Fondazione Del Monte di Bologna e Ravenna.
We are grateful to Prof. Giuseppe Sarli, DVM, PhD, for valuable assistance with statistical analysis.
