Abstract
Expression of five zinc transporters (ZnT1, 4, 5, 6, and 7) of the Slc30 family in the mouse gastrointestinal tract was studied by immunohistochemical analysis. Results demonstrated unique expression patterns, levels, and cellular localization among ZnT proteins in the mouse gastrointestinal tract with some overlapping. ZnT1 was abundantly expressed in the epithelium of the esophagus, duodenum of the small intestine, and cecum of the large intestine. ZnT4 was predominantly detected in the large intestine. ZnT5 was mainly expressed in the parietal cell of the stomach and in the absorptive epithelium of the duodenum and jejunum. ZnT6 was predominantly detected in the chief cell of the stomach, columnar epithelial cells of the jejunum, cecum, colon, and rectum. Lastly, ZnT7 was observed in all epithelia of the mouse gastrointestinal tract with the highest expression in the small intestine. Expression of ZnT proteins in the absorptive epithelial cell of the gastrointestinal tract suggests that ZnT proteins may play important roles in zinc absorption and endogenous zinc secretion.
Z
All ZnT proteins are predicted to have similar protein structures with six transmembrane domains and a histidine-rich cytoplasmic loop between transmembrane domains IV and V (Palmiter and Huang 2004) except for ZnT5, which has 16 potential transmembrane domains predicted by the SOSUI system (
Although tissue distributions of ZnT message RNAs and rat small intestinal distributions of ZnT1 and ZnT4 proteins have been demonstrated by Northern blot analysis, the Expressed Sequence Tag (EST) clone collections (
Materials and Methods
Animals
Five-week-old male C57BL/6 adult mice were purchased from the Jackson Laboratory (Bar Harbor, ME). Mice were housed in a temperature-controlled room at 22-24C with a 12 hr light:dark cycle and were fed a standard laboratory chow diet and double-distilled water ad libitum. At 6 weeks old, mice were fasted for 16 hr and killed by cardiac puncture while anesthetized. Animal experiments were conducted in accordance with NIH Guidelines for the Care and Use of Experimental Animals and were approved by the Institutional Animal Care and Use Committee of the University of California at Davis.
Plasmids
EST clones, AA252544 and BC008198, containing partial cDNA sequences of human Znt1 and Znt5, respectively, were purchased from ATCC (Manassas, VA). A 522-bp DNA fragment encoding 174 amino acids from the C-terminal end of the ZnT1 protein was cloned into the BamHI and XhoI sites of a bacterial GST fusion protein expression vector, pGEX-4T-3 (Amersham Biosciences; Piscataway, NJ). In addition, a 1725-bp DNA fragment encoding 575 amino acids from the C-terminal end of the ZnT5 protein was cloned into the XhoI site (blunted) of pGEX-4T-3. The resulting plasmids, pGEX/ZnT1 and pGEX/ZnT5, were used for bacterial transformation and production of GST-ZnT1 and GST-ZnT5 fusion proteins for Western blot analysis.
Preparation of Tissue Sections
Tissues including esophagus, stomach, small intestine, and large intestine were rapidly harvested from killed mice. Harvested tissues were flushed twice with ice-cold 1X PBS, pH 7.4, and once with cold 4% paraformaldehyde. Tissues were then cut into the desired segments and sizes and fixed in 4% paraformaldehyde solution at 4C overnight. A 2-cm segment of the small intestine adjacent to the pyloric sphincter was taken as duodenum. A 2-cm segment of the middle portion of the small intestine was taken as jejunum. A 2-cm segment of the small intestine adjacent to the cecum was taken as ileum. All specimens were dehydrated in a graded series of ethanol, cleared in Clear-Rite 3, and embedded in paraffin at 58C (Richard-Allan Scientific; Kalamazoo, MI). Seven-μm tissue sections were cut, mounted on the positive-charged lysine-coated slides, and dried at 42C overnight before immunohistochemical staining.
Antibodies
Rabbit anti-ZnT4, anti-ZnT6, and anti-ZnT7 polyclonal antibodies were raised against synthetic peptides (amino acids 93-110 of mouse ZnT4, amino acids 446-460 of mouse ZnT6, and amino acids 299-315 of mouse ZnT7) (Huang et al. 2002; Kirschke and Huang 2003) and affinity purified (Pierce; Rockford, IL). Rabbit anti-ZnT1 and anti-ZnT5 polyclonal antibodies were raised against synthetic peptides (amino acids of 495-507 of human ZnT1, CKNMPNKQPESSL, and amino acids 750-762 of human ZnT5, CKQMESMKYCKDGT). Epitopes used in the generation of rabbit anti-ZnT1, anti-ZnT4, anti-ZnT5, anti-ZnT6, and anti-ZnT7 polyclonal antibodies were unique to the respective protein as determined by BLAST searches of the SWISSPROT database. Specificities of the antibodies against ZnT4, ZnT6, and ZnT7 were described previously by Western blot analyses (Huang et al. 2002; Kirschke and Huang 2003). Specificities of the antibodies against human and mouse ZnT1 and ZnT5 were examined using Western blot and immunohistochemical analyses shown in Figure 1. The monoclonal anti-GST antibody was purchased from Stressgen Bioreagents-Assay Design (Ann Arbor, MI).
Cell Culture
Human prostate epithelial cells (RWPE2) were purchased from ATCC and cultured in the keratinocyte serum-free medium containing 5 ng/ml human recombinant epithelial growth factor, 0.05 mg/ml bovine pituitary extract, 100 U/ml penicillin, and 100 μg/ml streptomycin (Invitrogen; Carlsbad, CA). RWPE2 grew adherently in tissue culture.
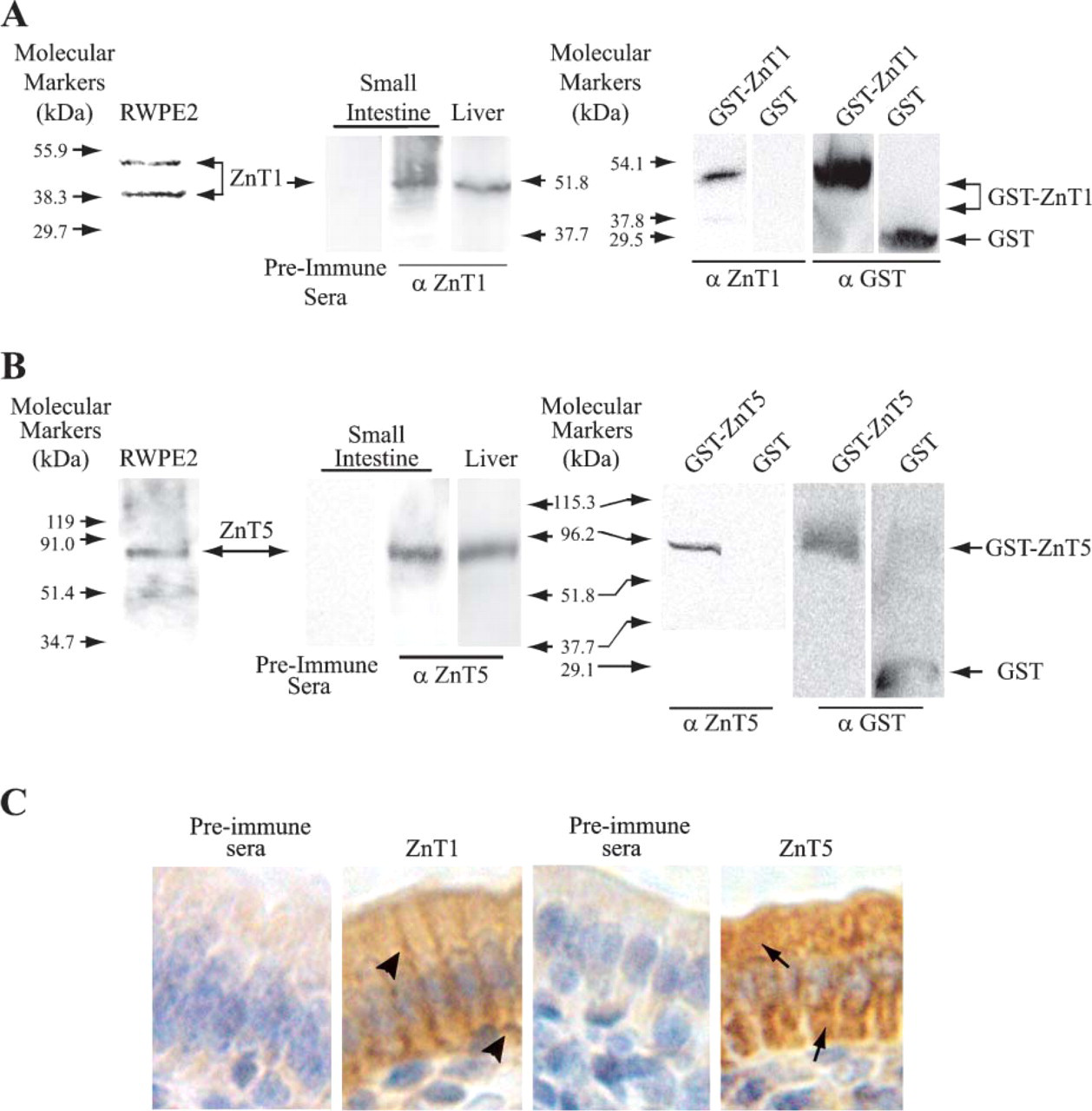
Expression of ZnT1 and ZnT5 proteins. (
Western Blot Analysis
RWPE2 were harvested after 48 hr culture at 37C. Protein lysate was prepared as previously described (Huang et al. 2002). Fifty-μg protein extracts were separated on a 4-20% Tris-HCl gel (Bio-Rad; Hercules, CA) and transferred to a nitrocellulose membrane (Bio-Rad). Blots were then probed with either an affinity-purified rabbit anti-ZnT1 antibody (1:250 dilution) or an affinity-purified rabbit anti-ZnT5 antibody (1:250 dilution) followed by a peroxidase-conjugated goat anti-rabbit secondary antibody (1:2500 dilution). ZnT1 and ZnT5 were visualized using SuperSignal West Femto kit (Pierce) and an Alpha Innotech Gel Documentation System (Alpha Innotech; San Leandro, CA).
The mucosa of the duodenum of the mouse small intestine and liver were isolated from C57BL/6J mice and homogenized in the lysis buffer containing 50 mM Tris-HCl, pH 7.5, 150 mM NaCl, 1% Triton X-100, 10% SDS, and 0.5% deoxycholate. One and one half Complete Mini-proteinase Inhibitor Tabs (Roche Applied Science; Indianapolis, IN), 375 μl of 20 mM PMSF, and 5 μl of 2 mg/ml pepstatin A were added to 7.5 ml of the lysis buffer just before use. Homogenized tissues were incubated on ice for 15 min, pasted through syringes with 25-gauge needles six times, heated at 100C for 10 min, and centrifuged at 20,800 × g at 4C for 10 min. Supernatants were collected and quantitated using Bio-Rad Protein Assay. Fifty μg of the lysates was resolved in a 4-20% Tris-HCl gel (Bio-Rad) and transferred to a nitrocellulose membrane (Bio-Rad). Blots were probed and proteins were detected as described above.
Bacterial lysates containing the GST-ZnT1, GST-ZnT5 fusion proteins or the GST protein alone were prepared according to the manufacturer's instructions (Amersham Biosciences). Fifty μg of the bacterial lysates was loaded onto each well of a 4-20% Tris-HCl gel (Bio-Rad) and transferred to a nitrocellulose membrane (Bio-Rad). Blots were probed with either anti-ZnT1 or anti-ZnT5 antibody, and proteins were detected as described above.
Immunohistochemical Analysis
Mounted sections were heated at 60C for 1 hr, cooled to room temperature, deparaffinized, and rehydrated. Antigen retrieval was performed by heating slides at 98C for 20 min in a 10-μM Tris-HCl buffer (pH 10) followed by 20-min cooling without changing buffer. Slides were then immersed for 15 min in a 5% H2O2 solution to inhibit endogenous peroxidase activity and washed twice with 1X PBS, pH 7.4, for 2 min. After washes, slides were treated with a Mouse Ig Blocking Reagent following manufacturer's instructions (Vector M.O.M. Immunodetection Kit; Vector Laboratories, Burlingame, CA). Endogenous biotin was blocked with an Avidin-Biotin Blocking kit purchased from Vector Laboratories. Affinity-purified primary antibodies were diluted in the M.O.M diluent (Vector Laboratories) as follows: ZnT1, 1:50; ZnT4, 1:25; ZnT5, 1:25; ZnT6, 1:50; and ZnT7, 1:750. Specimens were incubated with the diluted primary antibodies at 37C for 30 min in a humidified chamber. Control sections were incubated with preimmune sera diluted in the M.O.M diluent (1:750). Detection of the primary antibody binding was done using a rabbit ABC Staining Kit (Vector Laboratories) per manufacturer's instructions. Sections were counterstained with hematoxylin. Expressed ZnT proteins were indicated by the deposition of a brown product developed by using a DAB kit (Vector Laboratories).
Results
Specificities of Antibodies Against ZnT1 and ZnT5 Proteins
To investigate the distribution and localization of ZnT proteins in the gastrointestinal tract, we generated rabbit polyclonal antibodies against peptides derived from ZnT1, 4, 5, 6, and 7 amino acid sequences (Huang et al. 2002; Kirschke and Huang 2003) (see Materials and Methods). All antisera were affinity purified with the corresponding peptides used for immunization of the rabbits. Specificities of the antibodies against mouse ZnT4, 6, and 7 were described previously (Huang et al. 2002; Kirschke and Huang 2003). Anti-ZnT1 and anti-ZnT5 antibodies were raised against synthetic peptides from the carboxyl-terminal end of human ZnT1 and ZnT5. Selected regions of ZnT1 and ZnT5 used for antibody production display no homology to other members of the Slc30 family or any other proteins in the GenBank SwissProt database. Therefore, the raised antibodies are expected to specifically recognize corresponding human and mouse ZnT1 and ZnT5 proteins.
Specificities of anti-ZnT1 and anti-ZnT5 antibodies were confirmed by Western blot and immunohistochemical analyses (Figure 1). Calculated molecular masses for human and mouse ZnT1 and ZnT5 are 55 kDa and 84 kDa, respectively. As shown in Figure 1, the anti-ZnT1 antibody recognized two protein bands migrating at ∼54.7 and ∼39.9 kDa in the protein lysate isolated from human prostate epithelial cells and a protein band migrating at ∼56 kDa in protein extract isolated from mouse tissues (small intestine and liver). The anti-ZnT5 antibody recognized a single protein band migrating at ∼87 kDa in protein extracts isolated from human prostate epithelial cells and mouse tissues (small intestine and liver). These protein bands (ZnT1 and ZnT5) were not seen when preimmune sera were used (Figure 1). The 54.7 kDa of ZnT1 and 87 kDa of ZnT5 detected on the Western blots were consistent with the calculated molecular masses of human and mouse ZnT1 and ZnT5, respectively. The protein band of 39.9 kDa of ZnT1 detected in human prostate epithelial cells may be a degraded product as it has been consistently detected in many human and mouse tissues (McMahon and Cousins 1998; Sekler et al. 2002; Kelleher and Lonnerdal 2003).
Using protein extracts from bacteria expressing the GST-ZnT1 or GST-ZnT5 fusion protein or the GST protein alone further validated the specificity of the newly synthesized anti-ZnT1 and anti-ZnT5 antibody. In this experiment, 174 amino acids at the C-terminal end of ZnT1 or 575 amino acids at the C-terminal end of ZnT5 were fused in frame to the C-terminal end of the GST protein. Predicted molecular masses of the GST-ZnT1 and GST-ZnT5 fusion proteins were 45 kDa and 89 kDa, respectively. On the Western blots containing either the GST-ZnT1 or GST-ZnT5 fusion protein, protein bands migrating at ∼49 kDa and ∼86 kDa were detected by the anti-ZnT1 and anti-ZnT5 antibodies, respectively. Sizes of the GST-ZnT1 (49 kDa) and GST-ZnT5 (86 kDa) fusion protein bands were in agreement with the predicted molecular masses of the GST-ZnT1 and GST-ZnT5 fusion proteins. No protein band was detected by either the anti-ZnT1 or anti-ZnT5 antibody in the bacterial lysate containing the GST protein alone (Figure 1). The sizes of the GST-ZnT1 (49 kDa) and GST-ZnT5 (86 kDa) protein bands were further confirmed on the Western blots by using a mouse monoclonal anti-GST antibody (Figures 1A and 1B). On the same Western blots, the GST protein alone was detected ∼28 kDa that was consistent with its calculated molecular mass of 26 kDa (Figures 1A and 1B).
Immunohistochemical analysis revealed that ZnT1 was localized on the plasma membrane and ZnT5 was localized intracellularly (Figure 1C) as reported previously (Kambe et al. 2002; Kelleher and Lonnerdal 2003). Together, the results indicate that the newly synthesized anti-ZnT1 and anti-ZnT5 antibodies specifically react with the corresponding proteins.
Expression of ZnT Proteins in the Mouse Esophagus and Stomach
The differential expression of ZnT1, 4, 5, 6, and 7 proteins in the mouse gastrointestinal tract was examined by immunohistochemical analysis. The esophagus is the first part of the alimentary canal that delivers food from mouth to stomach. In mice, the esophagus is lined throughout its length with a keratinized stratified squamous epithelium. All ZnT proteins examined were expressed in the stratified squamous epithelium of the mouse esophagus (Figure 2A). ZnT1 was detected at the basal membrane of the basal cell layer of the squamous epithelial cells (Figure 2A). ZnT1, 5, and 6 were also detectable in the lamina propria of the esophagus (Figure 2A).
The distinctive features of the stomach are the gastric glands that secrete enzymes and acids that accelerate the digestive process. Gastric glands are simple tubular glands that contain mainly four types of cells: larger light-staining parietal cells that secrete concentrated HCl, smaller basophilic chief cells that secrete proenzymes such as pepsinogen, mucous neck cells that secret soluble mucus, and enteroendocrine cells that secrete gastrin. Immunostaining patterns of ZnT1, 4, 5, 6, and 7 in the gastric mucosa of the stomach that houses these secretory glands were examined. ZnT1 was not detectable in the gastric mucosa (Figure 2B). Both ZnT4 and ZnT5 were detected in the cytoplasm of the parietal cell (Figure 2B). ZnT6 was expressed in the chief cell in the perinuclear and peripheral regions of the cell (Figure 2B). ZnT7 expression was detected in the cytoplasm of the neck cell (Figure 2B). However, expression of ZnT proteins in the enteroen-docrine cells of the gastric gland of the stomach could not be determined.
Expression of ZnT Proteins in the Mouse Small Intestine
The small intestine is the major site for digestion of food and for absorption of the digested products. It can be divided into three segments (duodenum, jejunum, and ileum) based on their anatomic appearances. Essential features of the small intestine are villi, a lining epithelium composed of columnar absorptive cells interspersed with mucus-secreting goblet cells, and tubular glands (crypts of Lieberkühn) between the villi with an opening to the luminal surface of the intestine at the base of villi. Immunoreactivities for ZnT1, 4, 5, 6, and 7 in villi of the duodenum, jejunum, ileum, and intestinal glands (crypts of Lieberkühn) in the duodenum were studied. Our results showed that ZnT1 was strongly expressed in the duodenum with limited expression in the jejunum and ileum and was predominantly localized at the basolateral membrane of the absorptive epithelial cells in the duodenum (Figure 3A). ZnT4 was localized in perinuclear regions of the absorptive epithelium and the goblet cells in the duodenum and jejunum (Figure 3A). Protein expression patterns for ZnT5 and ZnT7 were similar to each other. Both were strongly expressed in the duodenum and jejunum and predominantly detected in the cytoplasm of absorptive epithelial cells and goblet cells with stronger immnuoreactivity at the basal side of both cell types (Figure 3A). Among the ZnT proteins examined, ZnT7 was the only one that expressed in the ileum (Figure 3A). Lastly, ZnT6 was mainly expressed in absorptive epithelial cells of the jejunum. It resided at the apical membrane with a punctate staining pattern as well as in the perinuclear region of the cell. ZnT6 was also seen intracellularly in the goblet cell of the jejunum and ileum (Figure 3A).
Crypts of Lieberkühn are largely composed of columnar epithelial cells, which are similar to those in the epithelium of small intestinal villi. Undifferentiated cells close to the bottom of the crypts regenerate the epithelium to replace the shed epithelial cells of villi. Sections containing the crypts of Lieberkhn in the duodenum were examined for ZnT protein expression. The results showed that ZnT5 and ZnT7 were heavily stained in the developing epithelial cells in crypts. ZnT5 appeared to reside along the membranes in cell-cell contacts, whereas ZnT7 was localized throughout the epithelial cell (Figure 3B). ZnT1 and ZnT6 were also expressed in the columnar epithelial cells in crypts. Immunoreactivity for ZnT1 was largely seen at the lateral membrane of the developing epithelial cells, whereas ZnT6 was detected in the cytoplasm close to the base (Figure 3B). ZnT4 was barely detectable in the columnar epithelial cells of crypts (Figure 3B).
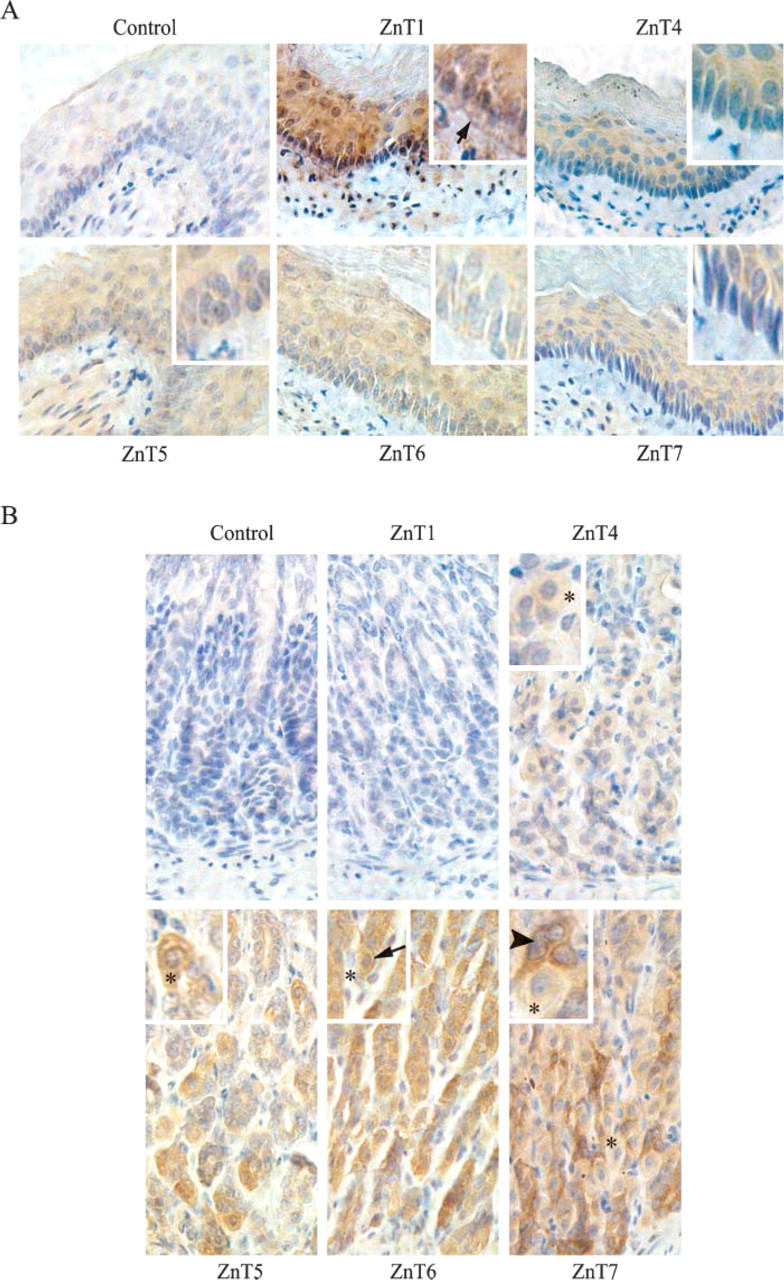
Expression of ZnT1, 4, 5, 6, and 7 proteins in the mouse esophagus and stomach. (
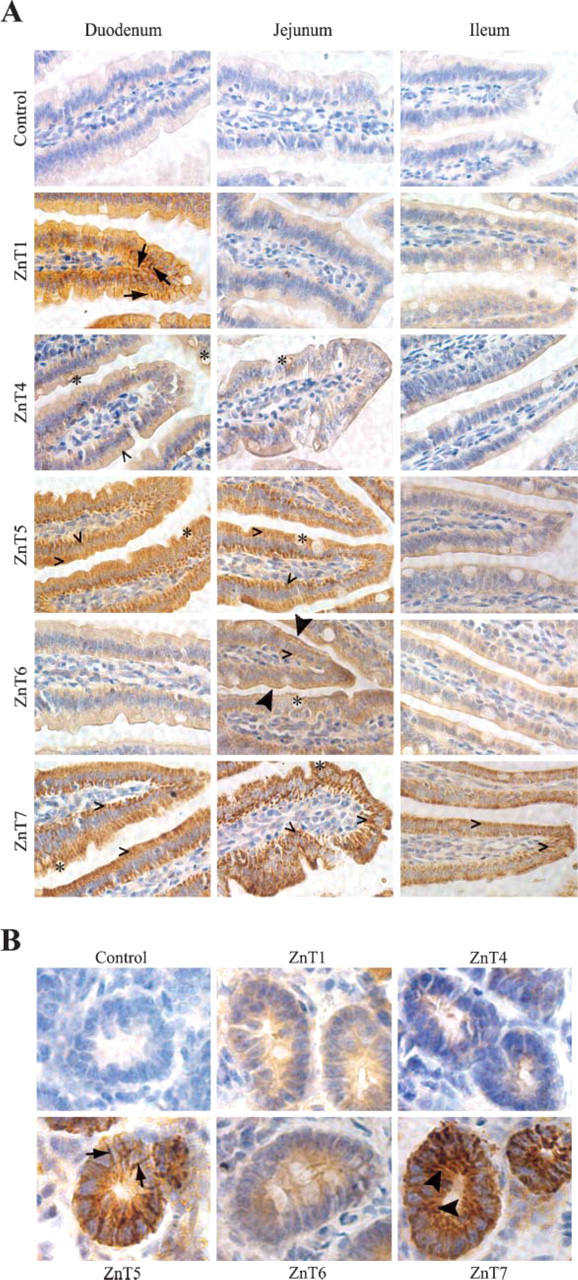
Expression of ZnT1, 4, 5, 6, and 7 proteins in the mouse small intestine. Immunohistochemical staining of ZnT1, 4, 5, 6, and 7 in the mouse small intestine (C57BL/6J) was performed as described in Materials and Methods. Control sections were incubated with preimmune sera isolated from the rabbit that the anti-ZnT7 antibody was raised from. Sections were counterstained with hematoxylin. Brown color shows immunoreactivity. (
Expression of ZnT Proteins in the Mouse Large Intestine
The large intestine is composed of the cecum, colon, rectum, and anal canal. Its principal functions are to receive the undigested food from the small intestine, reabsorption of electrolytes and water, and elimination of undigested food. Figure 4A shows the intense and uniform expression of ZnT1 at the lateral membrane as well as in the cytoplasm of epithelial cells in the cecum. ZnT1 was also observed in epithelial cells of the colon, which mainly resided in the cytoplasm of the cell (Figure 4B). Little to no ZnT1 was detected in epithelial cells of the rectum (Figure 4B). In contrast to the low expression in the small intestine, ZnT4 was strongly expressed in the colon and rectum with marginal expression in the cecum (Figures 4A-4C). Both epithelial and goblet cells stained intracellularly for ZnT4 in the epithelium of the colon and rectum (Figures 4B and 4C). Expression of ZnT5 in the epithelium of the cecum, colon, and rectum was much weaker than that in the duodenum and jejunum of the small intestine (Figures 4A-4C). Expression of ZnT6 was restricted to the apical membrane of epithelial cells in the cecum and colon, whereas in the rectum it was localized throughout the cytoplasm of epithelial cells (Figures 4A-4C). Following its strong expression pattern in the small intestine, ZnT7 was heavily expressed in epithelial and goblet cells in the cecum, colon, and rectum with cytoplasmic localization (Figures 4A-4C).
Discussion
In this study we have demonstrated the tissue distribution and subcellular localization of ZnT1, 4, 5, 6, and 7 proteins in the mouse gastrointestinal tract. We have shown that the overall expression levels of ZnT proteins are quite high in the mouse gut, and each ZnT protein presents a unique expression pattern along the tract. However, some overlapping expression of ZnT proteins also exists. High expression of these zinc transporters in the mouse gut implies that they may be actively involved in dietary zinc absorption, luminal zinc reabsorption, and intestinal zinc secretion processes in the gut. Because of the high degree of homology for ZnT proteins between human and mouse (
In mice, ZnT1 was observed to be highly expressed in epithelial cells of the esophagus, duodenum, and cecum. However, epithelial expression in the intestinal crypts was much weaker than in villi of the duodenum, indicating that ZnT1 expression is differentially regulated. ZnT1 is thought to be the only ZnT protein that functions in directly transporting the cytoplasmic zinc out of the cell (Palmiter and Findley 1995; Palmiter 2004). Therefore, ZnT1 is anticipated to transfer absorbed dietary zinc across the basolateral membrane of absorptive epithelial cells into the blood stream. The observation that ZnT1 is mainly expressed and localized at the basolateral membrane of the differentiated absorptive epithelial cell of the duodenum and cecum suggests that the duodenum of the small intestine and the cecum of the large intestine may be the major sites in the gastrointestinal tract for zinc absorption. Observation of the highest expression of ZnT1 in the duodenum is also in agreement with the previous observation that the proximal segment of the small intestine was the major site for dietary zinc absorption (Van Camen and Mitchell 1965). Higher ZnT1 expression in the cecum of the large intestine revealed in this study also supports the previous observation that zinc was absorbed in the large intestine (Meneely and Ghishan 1982; Seal and Mathers 1989; Suzuki et al. 2005b).
Zinc has been shown to be an important metal required for growth and repair of squamous tissue in the esophagus. Zinc deficiency increases esophageal squamous epithelial cell proliferation, which results in esophageal carcinoma (Abnet et al. 2005; Liu et al. 2005). Our finding that ZnT1 was expressed in the squamous epithelium of the esophagus suggests that ZnT1 may have an important role in maintaining esophageal zinc homeostasis, critical for preventing esophageal cancer development.
ZnT proteins were differentially expressed in different cell types in the gastric gland of the stomach in this study. Gastric glands contain highly differentiated cells including neck cells, parietal cells, and chief cells that all play specialized roles in the secretion of mucus, hydrochloric acid, and digestive enzymes into the stomach lumen. Zinc has been implicated to be involved in the synaptic transmission in brain and insulin secretion in pancreas (Palmiter et al. 1996b; Kambe et al. 2002; Chimienti et al. 2004). Following similar mechanism, ZnT4, 5, 6, and 7 may be the selective transporters that accumulate cytoplasmic zinc into secretory granules in the neck, parietal, and chief cells of the stomach for mucus, hydrochloric acid, and pepsinogen secretions. As for the cell type-specific expression in gastric glands, one possible explanation is that regulatory factors, which control expression of each ZnT protein either at transcriptional or translational level, are differentially present in each cell type of gastric glands.
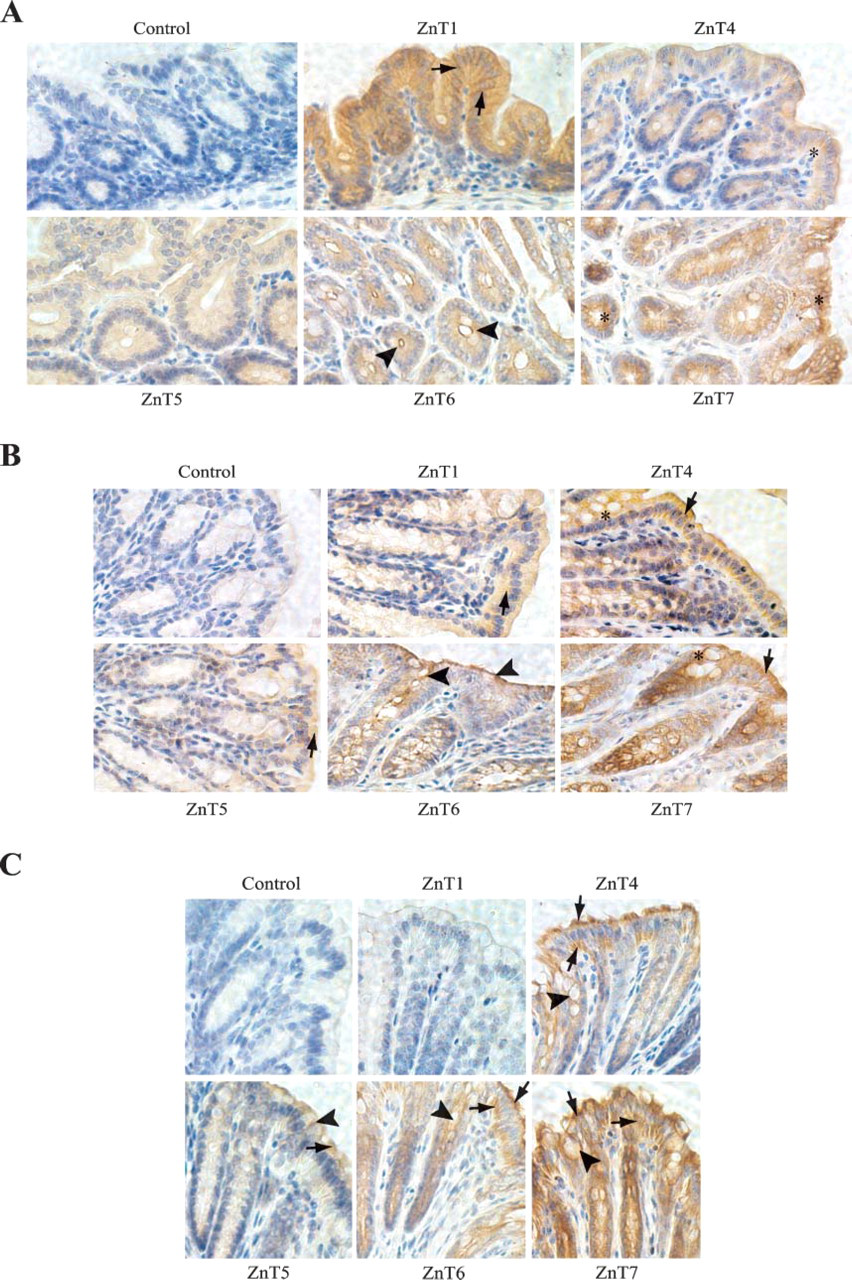
Expression of ZnT1, 4, 5, 6, and 7 proteins in the mouse large intestine. Immunolocalization of ZnT1, 4, 5, 6, and 7 in the cecum, colon, and rectum of the large intestine (C57BL/6J) is shown. Control sections were incubated with preimmune sera isolated from the rabbit that the anti-ZnT7 antibody was raised from. Sections were counterstained with hematoxylin. Brown color shows immunoreactivity. (
The primary site for dietary zinc absorption occurs in the proximal small intestine. Therefore, the higher ZnT5 and ZnT7 expression levels in absorptive epithelial cells of the duodenum and jejunum (Figure 3) may suggest that both transporters may actively participate in the metabolism of dietary zinc. ZnT5 and ZnT7 have the highest amino acid similarity among the mammalian Slc30 family members (Kambe et al. 2002; Kirschke and Huang 2003). Fluorescent immunostaining analysis has indicated that both ZnT5 and ZnT7 reside on the membrane of the Golgi apparatus and vesicular compartments (Kambe et al. 2002; Kirschke and Huang 2003; Suzuki et al. 2005b; Chi et al. 2006). Our immunohistochemical analysis demonstrated that ZnT5 and ZnT7 were expressed in both apical and basal cytoplasm of the small intestinal absorptive epithelial cell (Figure 3), a distribution pattern similar to alkaline phosphatase, a digestive enzyme requiring zinc for activation (Hasumi et al. 2003). Moreover, ZnT5 and ZnT7 are required for delivery of zinc to alkaline phosphatases in the secretory pathway (Suzuki et al. 2005b). Given the amino acid similarity between ZnT5 and ZnT7 and almost identical immunostaining patterns in the duodenum and jejunum of the small intestine, we propose that the molecular function of ZnT5 and ZnT7 in the dietary zinc absorption process in the small intestine is overlapping. This possibility is supported by the fact that disruption of either gene in the mouse genome did not affect the dietary zinc absorption process and, therefore, cause zinc deficiency in mice (unpublished data and Inoue et al. 2002). In addition, an in vitro study reported by Suzuki et al. (2005a) suggested that both ZnT5 and ZnT7 function in delivery of the cytoplasmic zinc into the secretory pathway but via different mechanism. Both ZnT5 and ZnT7 may be involved in moving dietary zinc from the apical cytoplasm of the absorptive epithelial cell to the basolateral cytoplasm of the cell for zinc deposition into the blood stream via transporting vesicles and in supplying zinc to zinc-requiring proteins via the Golgi apparatus. Moreover, ZnT5 and ZnT7 are the two genes found highly expressed in columnar epithelial cells in crypts of Lieberkhn along the small intestinal tract, indicating ZnT5 and ZnT7 play roles in absorptive epithelial cell differentiation.
Localization of ZnT4 in the epithelium of the rat small intestine was reported previously (Murgia et al. 1999). Our immunostaining results of ZnT4 confirm expression of ZnT4 and its localization in the mouse small intestine. Our results also indicate that the distribution patterns of ZnT4, 5, and 7 largely overlap each other in the epithelium of the small intestine isolated in young adult mice. In addition, higher expression of ZnT4 in the colon and rectum of the large intestine were revealed in this study. These data may explain why the young lethal milk mice with a defective ZnT4 gene have normal zinc absorption (Erway and Grider 1984). Homozygotes of 8-month-old lethal milk mice display progressive hair loss, dermatitis, and skin lesions, symptoms of zinc deficiency. This zinc deficiency phenotype observed in the old lethal milk mice indicates that the expression of ZnT5 and ZnT7 in the small intestine may be regulated with age in mice. It is known that zinc absorption efficiency decreases with age, which causes poor zinc status in human (Turnlund et al. 1986; August et al. 1989). The decrease in zinc transporter expression such as ZnT5 and ZnT7 may be responsible for the poor zinc absorption in the elderly.
We reported previously that the cellular localization of ZnT6 was regulated by extracellular zinc concentrations (Kirschke and Huang 2003). In the zinc-rich condition, ZnT6 is likely to localize to the peripheral region of the cell instead of the Golgi apparatus where it normally resides. This zinc-induced intracellular protein trafficking may underlie the function of ZnT6 as a zinc exporter via exocytosis (Kirschke and Huang 2003). In zinc-rich conditions, ZnT6 may transport the cytoplasmic zinc into the Golgi apparatus. Then zinc-rich transport vesicles bud off from the trans-Golgi network (TGN) and move toward the peripheral region of cell to deposit zinc out of the cell. This process may be directed by a special signal(s) within the ZnT6 protein. Moreover, this zinc export function of ZnT6 may require ZnT5 as a partner as ZnT6 and ZnT5 were found to form heterodimers in transporting zinc into the Golgi apparatus (Suzuki et al. 2005a). A similar mechanism of metal export has been described for the Menkes disease protein (MNK). MNK resides in the TGN when cellular copper concentration is low (Petris et al. 1996; Petris and Mercer 1999). However, under elevated cellular copper conditions, MNK redistributes to the plasma membrane to remove copper through an exocytosis mechanism.
It has been demonstrated that endogenous zinc is eliminated from the body primarily through the intestine (McCance and Widdowson 1942; Ballou and Thompson 1961; Methfessel and Spencer 1973). Moreover, an endogenous zinc secretion study in rodents has suggested that the jejunum of the small intestine may secrete most of the endogenous zinc in the intestinal tract (Methfessel and Spencer 1973). Observation of the highest expression level of ZnT6 in the jejunum of the small intestine in this study is in agreement with the results obtained from the above endogenous zinc secretion study (Methfessel and Spencer 1973), indicating that ZnT6 may mediate endogenous zinc secretion via exocytosis in intestinal epithelial cells. ZnT6 was also detected at the apical membrane of epithelial cells in the large intestine in this study. Localization of ZnT6 at the apical membrane of epithelial cells in the large intestine implies the presence of relatively high cellular zinc concentrations in these cells. Based on the expression patterns of ZnT1 and ZnT6 in the large intestine, we propose that different segments of the large intestine may have different roles in zinc absorption and secretion. In the cecum, ZnT1 was found highly expressed at the basolateral membrane of the epithelium, whereas ZnT6 was found at the apical membrane of the cell, suggesting that the cecum may be a site for both zinc absorption and secretion. On the other hand, in the colon, no basolateral localization of ZnT1 was detected in epithelial cells, whereas ZnT6 was highly expressed near the apical membrane of these cells, strongly indicating that the colon may not be a site for zinc absorption but for intestinal zinc secretion. In the rectum, ZnT1 was not detectable, whereas ZnT6 was distributed throughout the cytoplasm of the epithelial cell. These expression patterns point out that the rectum may not play an important role in either zinc absorption or zinc secretion.
Studies on the expression of ZIP proteins including ZIP1, 3, 4, and 5 in mouse small intestine have shown unique patterns of expression similar to ZnT proteins. For example, in adult mice, ZIP1 was prominently expressed in the connective tissue matrix surrounding the crypts and lamina propria of the villi with limited expression in the epithelium of the small intestine, whereas ZIP3 was mainly expressed in the epithelium of the crypts and small intestine (Dufner-Beattie et al. 2006). ZIP4 was predominantly expressed at the apical surfaces of enterocytes of the small intestines isolated from the mice fed with a zinc-deficient diet (Dufner-Beattie et al. 2003). Mutations in the ZIP4 gene in humans underlie acrodermatitis enteropathica, a genetic disease caused by a defect in dietary zinc absorption (Wang et al. 2002). On the other hand, the expression of ZIP5 in the mouse small intestine was detected at the basolateral membrane of enterocytes of the small intestine when the mice were fed with an adequate zinc diet (Dufner-Beattie et al. 2004). Based on unique ZIP protein expression patterns and cellular localization in the mouse small intestine during zinc deficiency or adequacy, it is proposed that ZIP1 and ZIP3 may play roles in distribution and/or retention of zinc, whereas ZIP4 and ZIP5 play key roles in dietary zinc acquisition and in zinc uptake for secretion from enterocytes back into the intestinal lumen, respectively (Dufner-Beattie et al. 2003,2004,2006).
In conclusion, the findings of the present study provide the first comprehensive study of ZnT protein expression in the mouse gastrointestinal tract. The study demonstrates that there appear to be dedicated ZnT proteins for each secretory cell type in the mouse stomach. The study also shows that the maximal expression of ZnT1 was found on the basolateral membrane in the epithelium of the esophagus, duodenum, and cecum. ZnT4 mainly expressed in the cytoplasm of epithelial cells in the large intestine. ZnT5 and ZnT7 present similar immunostaining in the duodenum and jejunum of the small intestine. ZnT7 was the only ZnT protein expressed along the length of the mouse gastrointestinal tract. Expression patterns and cellular localization of ZnT6 are different along the gastrointestinal tract.
Footnotes
Acknowledgements
This work was supported by the United States Department of Agriculture (Grant CRIS-5603-515-30-014-00D).
