Abstract
A large number of studies have established the mitochondrial uncoupling protein UCP1 as a specific marker of brown adipocytes, where it controls energy dissipation of fatty acid oxidation as heat in response to physiological requirements. Following the recent report of the detection of UCP1 in thymocytes of rats and mice, we reinvestigated its presence in thymus. Light microscopy and immunohistochemical analysis demonstrated that the UCP1 signal in thymus is entirely explained by the presence of typical brown adipocytes around the gland. Staining for UCP1 was not observed in thymocytes. Similarly, UCP1 failed to be observed in rat spleen, skeletal muscle, stomach, intestine, or uterus, even after exposure of animals to the cold. These data confirm the specificity of UCP1 expression in the thermogenic brown adipocytes and argue against a direct role for this mitochondrial transporter in immune cells. Whether brown adipocytes adjacent to thymic lobes play a role in thymus physiology remains to be investigated.
N
Materials and Methods
Animals and Tissues
Four-week-old male and female Wistar rats (75-100 g) and 4-month-old rats (225-375 g) were obtained from Charles River Italia (Lecco, Italy). They were caged singly and randomly assigned to different experimental groups. Three 4-week-old males and three females were acclimated at 6C for 8 days, whereas six age- and sex-matched rats (control group) were kept at room temperature (22C) for 8 days before sacrifice. During cold adaptation, the cages were cleaned every other day. Four-month-old Wistar rats (three males and three females) were kept at room temperature. Finally, we used three 2-month-old adult and three 6-day-old newborn wild-type C57BL-6J mice. Animals were treated and experimental procedures were performed according to protocols approved by the Marche Polytechnic University Institutional Animal Care and Use Committee.
For immunohistochemical studies, rats and mice were anesthetized with 100 mg/kg ketamine (Ketavet; Aprilia, Italy) in combination with 19 mg/kg xylazine (Rompum; Bayer AG, Leverkusen, Germany) and transcardially perfused with 4% formaldehyde in 0.1 M phosphate buffer (PB), pH 7.4. Several tissues were carefully dissected under a surgical microscope (thymus, BAT, white adipose tissue, spleen, liver, kidney, skeletal muscle, intestine, stomach, and uterus) and postfixed by overnight immersion in the same fixative at 4C. After a brief wash in PB, they were dehydrated with gradual steps of ethanol, cleared in xylene for 2 × 60 min, and then paraffin embedded overnight at 60C. The next day, after two paraffin changes, tissues were orientated in embedding mold.
For immunofluorescence and confocal microscopic analysis, animals were perfused as described above and tissues were postfixed by overnight immersion. After a brief wash in PB, they were cryoprotected in a solution of 30% sucrose in PB for 24 hr at 4C. A glass beaker containing 2-methylbutane (isopentane) was cooled in liquid nitrogen, and specimens were embedded in optimal cutting temperature medium (OCT, Tissue-Tek; Sakura Finetek Europe, The Netherlands), frozen, and stored at −80C.
Immunohistochemistry
Three-μm-thick paraffin sections were obtained with a microtome and dried overnight at 37C. The sections to be studied were collected at different levels to obtain representative samples of organs. Immunoreactivity was visualized using the avidin-biotin-peroxidase (ABC) method. Sections were deparaffinized with xylene (2 × 10 min), hydrated to water with decreasing alcohol concentrations, incubated with 0.3% H2O2 in methanol for 30 min at room temperature to block endogenous peroxidase, washed twice in 0.05 M PBS, pH 7.4, and incubated in 1:75 v/v normal rabbit serum or normal goat serum in PBS for 20 min at room temperature to block nonspecific sites. Sections were subsequently incubated overnight at 4C with the primary antibody raised in sheep against rat UCP1 (Ricquier et al. 1983) at dilutions ranging from 1:4000 v/v to 1:6000 v/v. A commercial antibody raised in rabbit against mouse UCP1 (cat. #AB3038, rabbit antimouse uncoupling protein-1 polyclonal antibody; Chemicon International, Hampshire, UK) was also used (1:3000 v/v dilution). The following day, sections were washed twice with PBS for 15 min and incubated in 1:200 v/v biotinylated IgG, either anti-sheep rabbit serum or anti-rabbit goat serum in PBS for 30 min at room temperature. They were washed twice in PBS for 15 min, incubated in ABC reagent (Vector Laboratories; Burlingame, CA) in PBS for 1 hr at room temperature, washed twice with PBS for 15 min, incubated in 0.02% H2O2 and 0.075% diaminobenzidine (Sigma-Aldrich; St Louis, MO) in 0.05 M Tris buffer (pH 7.6), and kept for 5 min in a dark room. Sections were rinsed in distilled water, counterstained with hematoxylin, dehydrated, mounted in Entellan, and finally viewed using an Eclipse E600 microscope (Nikon; Sesto Fiorentino, Florence, Italy) equipped with a digital camera and software acquisition.
Immunofluorescence and Confocal Microscopy
For the double-labeling experiments, 7-μm-thick cryosections obtained with a CM1900 cryostat (Leica Microsystems; Vienna, Austria) were collected and air dried overnight at room temperature. After two 15-min washes in PB, sections were incubated in 1:75 v/v normal donkey serum (Jackson ImmunoResearch; West Grove, PA) in PB for 20 min at room temperature to block nonspecific sites and then incubated overnight at 4C with a mixture containing the primary antibodies against UCP1 (Ricquier et al. 1983) and the thymocyte marker Thy-1 (cat. #CBL1500, mouse anti-rat Thy-1 monoclonal antibody; Chemicon) at 1:8000 and 1:1600 v/v, respectively. They were washed twice with PB (2 × 15 min) and incubated in a cocktail of secondary antibodies made by TRITC donkey anti-sheep 100 v/v and FITC donkey antimouse (Jackson ImmunoResearch) in PB for 30 min at room temperature. Sections were subsequently washed twice with PB, air dried, and coverslipped using Vectashield mounting medium (Vector Laboratories). Sections were viewed under a motorized Leica DM6000 microscope using a X60 plan apochromat objective and 1.4 numerical aperture. Fluorescence was detected with a Leica TCS-SL confocal microscope equipped with an Argon and He/Ne mixed gas laser. FITC and TRITC were excited with the 488 nm and 543 nm lines, respectively, and imaged separately. Images (1024 × 1024) were obtained sequentially from two channels using a confocal pinhole of 1.1200 and stored as TIFF files. Brightness and contrast of the final images were adjusted using Photoshop 6 software (Adobe Systems; Mountain View, CA).
Results
In agreement with the observations of Carroll et al. (2005), we detected UCP1 in rat thymus by Western blot analysis using a very large amount of thymus mitochondrial protein (30 μg) from eight rats kept at room temperature or exposed to cold. UCP1 was observed in thymic mitochondria in variable amounts ranging from almost 0 to 1% of the level measured in BAT mitochondria (data not shown). Because Western blotting does not allow identification of the cell type expressing protein in a tissue, we performed an immunohistochemical study (representative images are shown in Figure 1 and Figure 2). Similar results were obtained with the commercial antibody and the one made in the laboratory (Ricquier et al. 1983). Data presented were obtained using the latter antibody. The interscapular portion of the anterior subcutaneous depot of the adipose organ is mainly composed of brown adipocytes and is generally considered as BAT (Cinti 1999,2005). Experiments were performed on a section containing brown adipocytes taken as positive control (Figure 1A, right) as well as white adipocytes (Figure 1A, center) and muscle cells (Figure 1A, left). Intense UCP1 immunoreactivity was observed exclusively in brown adipocytes and never in white adipocytes or muscle cells.
Light microscopic analysis of thymuses from rats (Figures 2A, 2C, and 2D) and mice (Figure 2B) clearly evidenced a significant amount of BAT around the gland, especially in the area closest to its parenchyma, despite their careful dissection. Immunohistochemical analysis of UCP1 in these sections revealed intense reactivity only in brown adipocytes, whereas the parenchyma was consistently negative.
Given that UCP1 expression increases after cold exposure, thymuses from cold-exposed rats were also analyzed. All were negative for UCP1 (Figures 2C and 2D). A varying amount of BAT was present in all sections, yielding intense UCP1-positive reactions in brown adipocytes. For a more exhaustive investigation of the UCP1 immunoreactivity of the gland, immunohistochemical experiments were conducted at different section levels of the thymus, and the slides were examined at the highest magnification allowed by the microscope (X100). Again, thymocytes displayed a consistently negative signal (Figure 2D, upper right corner), whereas brown adipocytes exhibited strongly positive reactions (Figure 2D, left-hand side). Gastrointestinal tract organs and uterus were also investigated and found to be negative (Figures 1C and 1E). Similarly, white adipose tissue, skeletal muscle, liver, pancreas, spleen, ovary, and kidney from cold-acclimated rats gave negative results (Figures 1B-1E), whereas strong UCP1 immunoreactivity was observed in BAT from the same animals (Figures 1A-1F).
Further confirmation of these results was sought by double labeling of thymocytes and observing them under a confocal microscope. Similar results were obtained from both cold-acclimated (Figure 3) and control animals. Thymocytes, labeled by Thy-1 (Figure 3A, green stain), were always negative for UCP1. On the same histological section, BAT surrounding the thymus was intensely positive (Figure 3B, red stain). Merging of the two images (Figure 3C) confirmed that the two proteins do not colocalize. Further analysis of several histological preparations at higher magnification failed to evidence UCP1 in thymocytes.
Discussion
Despite the broad consensus on the unique expression of mitochondrial UCP1 in brown adipocytes, in relation to their specific ability to perform regulatory thermogenesis, its presence in organs or tissues where it has never been sought cannot, of course, be ruled out. Several studies have conclusively demonstrated that UCP1 is not found in spleen or in the immune system (Jacobsson et al. 1985; Boyer and Kozak 1991; Cassard-Doulcier et al. 1993; Negre-Salvayre et al. 1997; Arsenijevic et al. 2000). Recent reports by Porter's team (Carroll et al. 2004,2005) of the presence of UCP1 in thymocytes of mice and rats were, therefore, a surprise. This finding, and the authors' surmise that the presence of UCP1 in thymocytes, suggest an intriguing role for respiration uncoupling in immune cells, prompted us to reinvestigate the matter. Biochemical studies performed by Carroll and coworkers using mitochondrial respiration measurement, mass spectrometry analysis of purified UCP1, and immunological analysis of Ucp1−/− mice clearly established the identity of UCP1, demonstrating that the protein they detected was not a related carrier, such as UCP2, which is found in immune cells and thymus (Arsenijevic et al. 2000; Krauss et al. 2002).
In the present work, Western blot analysis of mitochondria isolated from the thymus of cold-exposed and control rats did evidence a varyingly small amount of UCP1. For these experiments we used a large amount of thymus protein (up to 300-fold the amount allowing UCP1 detection in BAT). Our findings agree with those of Carroll et al. (2005) to the extent that UCP1 can be detected in thymus. However, our histological, immunohistochemical, and confocal microscopy double-labeling studies failed to confirm that thymic UCP1 is found in thymocytes, but rather pointed to a different conclusion as to its localization: brown adipocytes. These experiments were not performed by Carroll et al. (2005). It is has been shown that although most brown adipocytes are found in specific depots of the adipose organ in rodents, a small amount of brown fat may exist inside white fat depots or in thoracic and pericervical areas (Cinti 2005; Murano et al. 2005). In addition, small amounts of brown fat can be found around the thymus and can be detected using a binocular magnifying glass (Frontini A, et al., unpublished data). However, even after careful dissection of the thymuses, light microscopic examination still revealed varying amounts of residual mediastinic BAT around the gland, indicating that the positive results obtained with Western blotting can be ascribed to the presence of residual brown adipocytes. Interestingly, small fragments of brown fat around rodent thymus were already noted by Aronson and Schwartzman (1956).
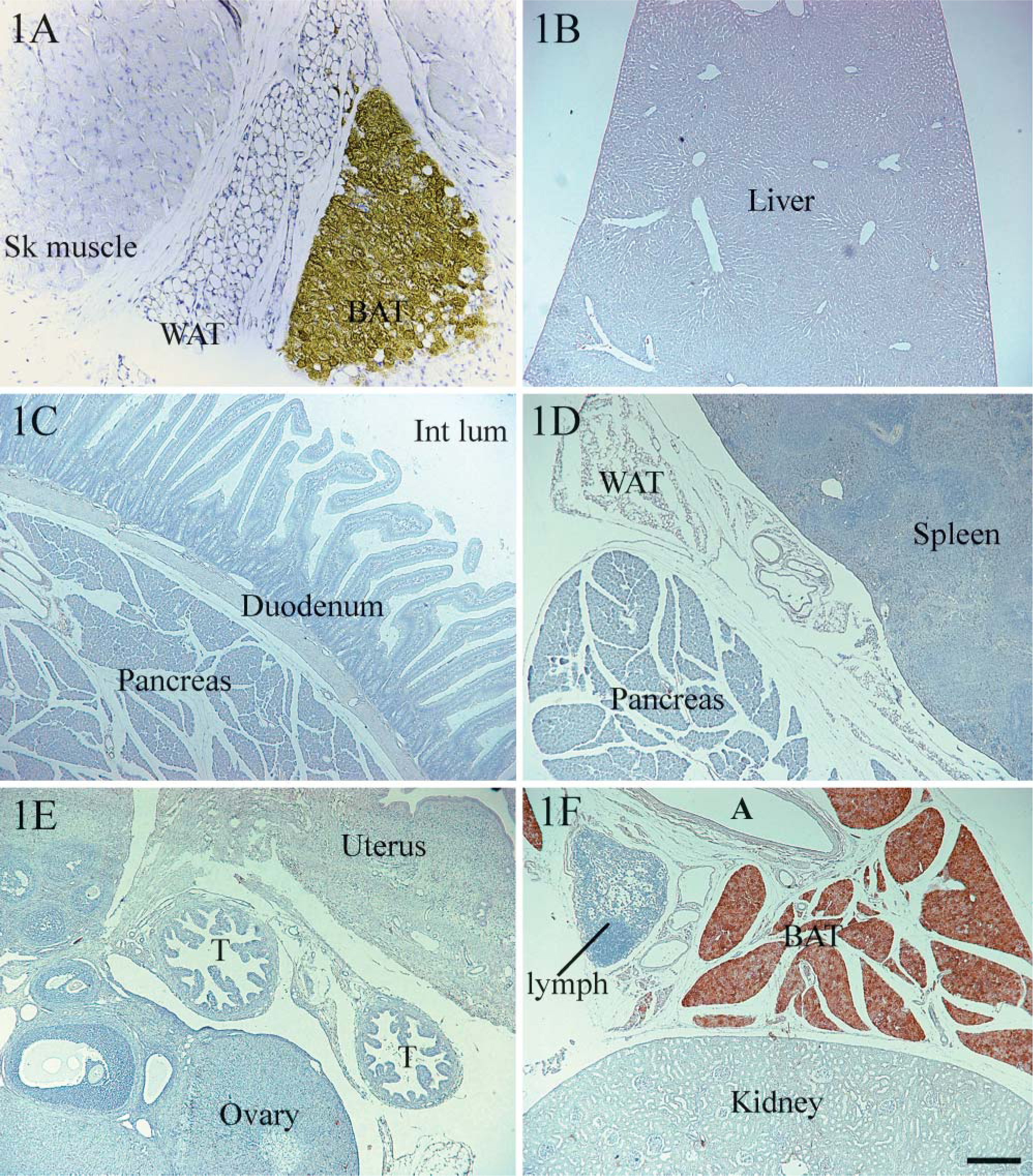
Uncoupling protein (UCP1) immunohistochemical analysis of tissues from cold-acclimated rats. Interscapular brown adipose tissue (BAT) was used as a positive control for UCP1 immunoreactivity (
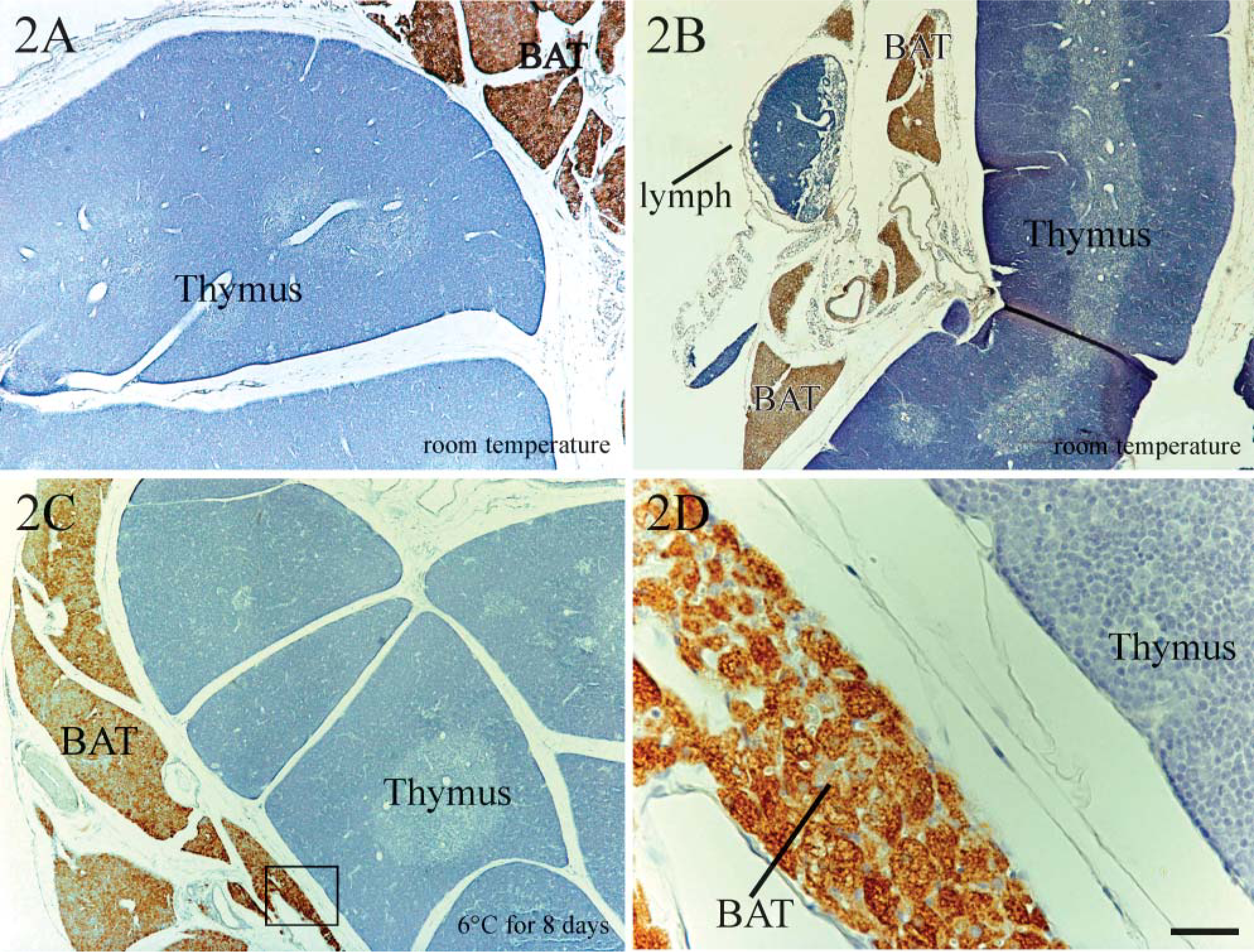
UCP1 immunohistochemical analysis of mouse and rat thymus. Mouse (
Immunohistochemistry is the most widely used technique to establish protein expression in situ and allows the identification of the immunoreactive cell type in tissues. UCP1 antibody used in our immunohistochemical experiments can detect UCP1 in the mitochondria of brown adipocytes even at a dilution of 1:10,000 in paraffin blocks (Cancello et al. 1998). The brown adipocyte mitochondria of UCP1-knockout mice were negative for the antibody (Rousset et al. 2003; Cannon B and Nedergaard J, unpublished data). Immunohistochemical experiments presented here confirm the presence of UCP1 uniquely in brown adipocytes and its absence in thymocytes. UCP1 has also been described in skeletal muscle of mice treated with β3-adrenoceptor agonist (Nagase et al. 1996; Yoshida et al. 1998) and in smooth muscle cells of the gastrointestinal tract and uterus (Nibbelink et al. 2001). In a previous study using the UCP1 antibody used here, we showed that the skeletal muscle of rats treated with the CL-316243 β3-adrenoceptor agonist is negative for UCP1 (Cinti et al. 2002). The presence of UCP1 in uterus was then disproven by Rousset et al. (2003). In the present work, the skeletal muscle of rats maintained at room temperature as well as that of cold-exposed animals contained no trace of UCP1. The technique also failed to confirm the results reported for gastrointestinal tract and uterus (Figures 1C and 1E). Our group and others previously demonstrated UCP1 in white adipose depots, but we also provided evidence that in these tissues any expression of UCP1 was associated with the number of brown adipocytes dispersed in white fat and was entirely confined to brown adipocytes (Cousin et al. 1992; Cancello et al. 1998; Cinti 1999,2001; Himms-Hagen et al. 2000; Cinti et al. 2002; Murano et al. 2005).
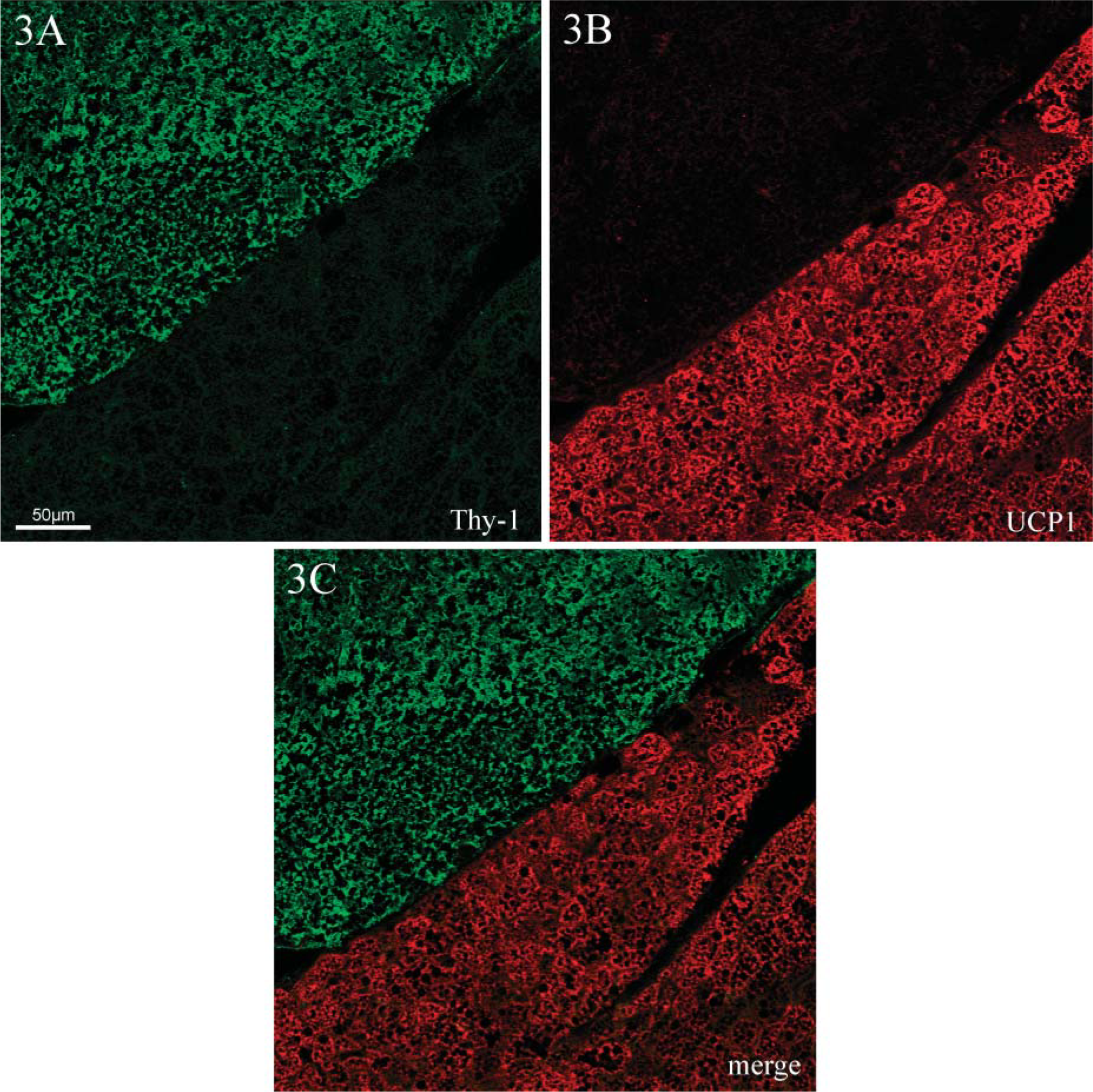
Indirect immunofluorescence and confocal microscopic analysis of the thymus of cold-acclimated rats. Double labeling for Thy-1 (green) and UCP1 (red) was performed on thymus samples from cold-acclimated rats. Confocal microscopy proved that the two proteins are not colocalized in thymocytes (
In conclusion, the present findings confirm that UCP1 is uniquely expressed in brown adipocytes and show that any UCP1 signal detected in thymus is accounted for by the brown adipocytes that remain attached to the thymus even after careful dissection. Whether brown adipocytes adjacent to thymic lobes play a role in thymus physiology remains to be investigated. At least for the present time the search for a role for UCP1 in thymocytes can thus be called off.
Footnotes
Acknowledgements
This work was supported by the Italian Ministry of Research (Grant No. FIRB-2001 to SC), the Centre National de la Recherche Scientifique (France), and the EU (Grant ECFP6 ‘Diabesity’ Contract No. LSHM-CT-2003-503041 to DR).
