Abstract
Serine proteases have been documented to play key roles in uterine matrix turnover and trophoblastic invasion during implantation. Roles of prostasin serine protease in these processes, however, are currently unclear. The present study was first conducted to investigate the colocalization of prostasin and its cognate serpin, protease nexin-1 (PN-1), in rhesus monkey endometrium and placenta on days 12, 18, and 26 of pregnancy by using in situ hybridization (ISH) and immunohistochemistry. With ISH, expression of prostasin mRNA was intensely localized in the glandular epithelium on days 12 and 18 and in the placental villi, trophoblastic column, trophoblastic shell, and fetal-maternal border on days 18 and 26. With the progress of pregnancy, expression level in the glandular epithelium was significantly decreased, and the accumulation in the placental compartments was further increased. In addition, the stroma and arterioles exhibited modest levels of prostasin signals. However, expression level of PN-1 in these compartments on adjacent sections in the three stages of early pregnancy was weak or below the level of detection. Prostasin protein expression in the endometrium was found to be consistent with the distribution patterns revealed in the ISH experiments. It may be suggested from these results that prostasin is involved in endometrial epithelial morphology establishment, tissue remodeling, and trophoblastic invasion during early pregnancy. The cognate serpin PN-1 was not coordinately expressed along with prostasin, creating a tissue environment favorable for proteolytic activities of prostasin during early pregnancy events.
E
Prostasin, a serine protease originally purified from human seminal fluid (Yu et al. 1994), is synthesized with a glycosylphosphatidylinositol (GPI) membrane anchor and can also be secreted (Chen et al. 2001a). At the amino acid level, prostasin shares 50-55% sequence identity with other serine proteinases such as α-tryptase, elastase 2A and 2B, chymotrypsin, acrosin, hepsin, plasma kallikrein, and coagulation factor XI (Yu et al. 1994). It is tryptic in specificity in that it displays arginine amidase activity preferentially at arginine residues on the carboxyl-terminal of target peptides (Yu et al. 1994). Prostasin is abundantly present at the protein level in the seminal fluid, urine, and prostate gland (Yu et al. 1994; Chen et al. 2001b) and also present at moderate levels in colon, lung, kidney, pancreas, salivary gland, liver, and bronchi (Yu et al. 1994). Prostasin has been identified as a potential regulator of epithelial sodium channel (ENaC) function in the kidney, lung, and airways (Donaldson et al. 2002; Narikiyo et al. 2002; Iwashita et al. 2003; Tong et al. 2004; Olivieri et al. 2005; Tuyen et al. 2005). Furthermore, it is counterintuitive that prostasin has been shown as an invasion suppressor of prostate cancer and breast cancer with downregulated expression in the invasive tumor cells by our and other laboratories (Chen et al. 2001b; Chen and Chai 2002; Takahashi et al. 2003), in sharp contrast to the conventional paradigm of the role of serine protease or expression behaviors in cancer. Expression and function of prostasin in the uterus that undergoes dramatic ECM degradation and remodeling, however, have not been addressed.
The serpin PN-1 can inhibit the activity of prostasin and bind to membrane-anchored prostasin (Chen et al. 2004). Distribution pattern of PN-1 has been described in human term placenta (White et al. 1993) and rat uterus (Kim et al. 2001). As reported in the latter study, expression level of PN-1 in rat uterus during preimplantation was extremely low, and the highest PN-1 expression occurred at the time of implantation and then decreased during postimplantation (Kim et al. 2001). These findings provide insight into the function of this protease inhibitor in implantation, most likely in the regulation of proteolysis through interaction with uterine serine proteases.
In the current study we used in situ hybridization (ISH) and immunohistochemistry (IHC) to localize prostasin and PN-1 transcripts and proteins in rhesus monkey endometrium during early pregnancy.
Materials and Methods
Animals and Tissue Collection
All procedures involving animals were carried out in accordance with the Guidelines on the Care and Use of Laboratory Animals of the Ethical Committee, State Key Laboratory of Reproductive Biology, Institute of Zoology, Chinese Academy of Sciences. Uterine tissues of pregnant rhesus monkeys (Macaca mulatta) were collected in the Center for Medical Primate, Institute of Medical Biology, Chinese Academy of Medical Sciences as detailed previously (Wang et al. 2001). In brief, female rhesus monkeys with regular menstrual cycles and histories of pregnancy were chosen for this study. Pregnancy was determined through sperm plug examination, ultrasonography, and mCG detection. Tissues were removed from monkeys under ketamine hydrochloride anesthesia on estimated days 12, 18, and 26 of pregnancy (n=3 for each time point). Uterine tissue samples containing the implantation sites were carefully excised, fixed in Bouin solution for 20 hr, and then embedded in paraffin.
Plasmid Constructs and Probe Synthesis
A 274-bp XhoI-BamHI fragment corresponding to bases 799-1072 of the human prostasin cDNA (GenBank accession number L41351) (Chen et al. 2001a) was inserted into the pSP73 vector (Promega; Madison, WI) and used for RNA probe labeling.
A full-length human PN-1 cDNA was amplified by PCR as described previously (Chen et al. 2004) and cloned into pGEM-T Easy vector (Promega). Authenticities of the cDNA product were confirmed by sequencing. cDNA fragment was then inserted into the pREP8 vector (Invitrogen; Gaithersburg, MD) at the NotI site. A SacI-HindIII fragment (corresponding to bases 804-1084 of A03911 in the GenBank) was subcloned into the pGEM-3Z vector (Promega).
Sense and antisense single-stranded RNA probes were synthesized using a digoxigenin (DIG) RNA labeling kit (SP6/T7) (Roche Diagnostics Ltd.; Indianapolis, IN). Yield of the riboprobes was determined using a spot test following the instructions of the manufacturer with a DIG-labeled RNA control, which is provided in the labeling kit. Spot intensities of the control and experimental probes were quantified using the Meta View image analyzing system version 4.50 (Universal Imaging Corp.; Downington, PA).
ISH
ISH protocol was described previously (Wang et al. 2001). Successive 5-μm-thick paraffin-embedded sections were deparaffinized and rehydrated in a decreasing series of ethanol solutions. Sections were rinsed with 1X PBS twice for 5 min each and were subjected to proteinase K digestion (10 μg/ml proteinase K in 50 mM EDTA, and 100 mM Tris, pH 7.5) at 37C for 10 min. After brief rinsing in 1X PBS, sections were fixed in 4% paraformaldehyde for 15 min and dehydrated in an increasing series of ethanol solutions. In a moist chamber, prehybridization was carried out at 50C for 2 hr in a prehybridization buffer containing 50% deionized formamide, 10% dextran sulfate, 0.5% SDS, 1X Denhardt, 10 mM Tris, pH 8.0, 10 mM dithiothreitol, 2X SSC (1X SSC is 0.15 M NaCl, 0.015 M sodium citrate), and 250 μg/ml salmon sperm DNA. Hybridization was then performed with 400 ng/ml denatured DIG-labeled probes in the prehybridization solution at 50C for 18 hr. Slides were washed at 42C stepwise in 2X SSC for 30 min, 1X SSC for 30 min, and 0.1X SSC for 30 min to remove nonspecific bindings. After brief rinsing in buffer I (100 mM Tris, 150 mM NaCl, pH 7.5) at room temperature, slides were incubated with the anti-DIG-alkaline phosphatase conjugate (diluted 1:500 in buffer I containing 0.5% blocking reagent; Roche) at room temperature for 2 hr, followed by further washes in buffer I three times at 10 min each. Hybridization signal was visualized with nitroblue tetrazolium/5-bromo-4-chloro-3-indolyl phosphate reagents (NBT/BCIP; Roche) in buffer II (100 mM Tris, pH 9.5, 100 mM NaCl, 50 mM MgCl2). Finally, slides were rinsed in 95% ethanol for 30 min and mounted for photography with a SPOT digital camera system (Diagnostic Instruments; Sterling Heights, MI). Sense probes were used on adjacent slides as negative controls for background levels.
IHC
Antiserum against human prostasin raised in rabbits was prepared as detailed previously (Chen et al. 2001b). A recombinant human PN-1 protein expressed as a fusion protein with the schistosomal glutathione-S-transferase in Escherichia coli was purified as described previously (Chen et al. 2004) and used as the antigen for raising rabbit anti-human PN-1 antiserum. Antiserum was produced by Cocalico Biologicals (Reamstown, PA) under contract. IHC was performed with a Vectastain biotin-streptavidin-peroxidase (SP) kit (Vector Laboratories; Burlingame, CA) according to the manufacturer's instructions. In brief, successive 5-μm-thick sections were deparaffinized completely and rehydrated in decreasing series of alcohol. Sections were subjected to antigen retrieval in citrate buffer (10 mM citrate sodium, 10 mM citric acid, pH 6.0) in a microwave oven at 92-98C for 15 min followed by treatment with 3% H2O2 in methanol at room temperature for 15 min. Sections were then incubated stepwise in 10% BSA (Sigma-Aldrich; Oakville, Ontario, Canada) in TBST (10 mM Tris-HCl, 150 mM NaCl, 0.1% Triton X-100, pH 7.6) for 30 min, with prostasin or PN-1 antiserum (diluted at 1:500 in the blocking buffer) for 2 hr, biotinylated goat anti-rabbit IgG (Vector Laboratories) for 30-60 min, and horseradish peroxidase (HRP)-labeled streptavidin (Vector Laboratories) for 1 hr. After each incubation, sections were washed with 1X PBS three times for 5 min each. Color development was performed with diaminobenzidine (DAB kit; Zhongshan Corp., Beijing, China). Finally, sections were counterstained with Harris hematoxylin and mounted. Negative controls were included where primary antiserum was substituted with normal rabbit serum.
Statistical Analysis
Densitometric readings for the transcripts and proteins of prostasin and PN-1 were performed by computer-aided laser-scanning densitometry (Personal Densitometer SI; Molecular Dynamics, Sunnyvale, CA). For each time point of early pregnancy, uteri from three different monkeys were used. For each section used in prostasin transcripts detecting or immunostaining, the adjacent section was used for PN-1 transcripts or protein detection. All experiments for ISH and IHC were repeated at least three times. To make the statistical significance of the quantitative difference credible, at least three slides from three monkeys of each group were examined for density scanning. In each compartment on a slide, at least 30 areas were randomly selected. Densitometric readings of 1-1.05, 1.05-1.3, 1.3-1.5, and >1.5 are assigned to areas with no, weak, moderate, and strong staining, respectively. Values are expressed as means + SEM for each group. Differences were examined by ANOVA (one-way ANOVA, defining p<0.05 as being statistically different), using the SPSS 10.0 software program (SPSS Inc.; Chicago, IL).
Results
In Situ Localization of Prostasin and PN-1 mRNA in Rhesus Monkey Endometrium and Placenta During Early Pregnancy
ISH experiments were performed to investigate mRNA localization of prostasin and PN-1 in pregnant macaque uterus and placenta. As shown in Figure 1 and Table 1, the glandular epithelium, placental villi, trophoblastic column, trophoblastic shell, and fetal-maternal border are the major sites of intense localization of prostasin mRNA in pregnant macaque uterus, whereas staining of PN-1 was relatively weak or hardly detectable in these compartments. In detail, prostasin mRNA was highly expressed in the glandular epithelium on day 12 of pregnancy (Figures 1A and 1B) (p<0.05) and modestly expressed in the stroma (Figures 1A and 1B) and arterioles (Figure 1B) (p<0.05). However, expression level of PN-1 on day 12 was below the level of detection (p>0.05) in the glandular epithelium and weak (p<0.05) in the stroma and arterioles (Figures 1F and 1G). Prostasin hybridization signals were abundantly localized in the placental villi (Figure 1C), trophoblastic column (Figures 1C and 1D), trophoblastic shell (Figure 1D), fetal-maternal border (Figure 1D), and glandular epithelium (Figure 1E) on day 18 (p<0.05) and were detected at moderate levels in the stroma and arterioles (Figure 1E) (p<0.05). Distinct from the expression pattern of prostasin, PN-1 exhibited faint signals in the trophoblastic column (Figures 1H and 1I), trophoblastic shell (Figure 1I), stroma (Figure 1J), and arterioles (Figure 1J) (p<0.05), with no discernible staining in the placental villi (Figure 1H), fetal-maternal border (Figure 1I), and glandular epithelium (Figure 1J) (p>0.05). On day 26, prostasin signals were dramatically intense in the placental villi (Figure 1K), trophoblastic column (Figure 1K and 1L), trophoblastic shell (Figure 1L), and fetal-maternal border (Figure 1L) (p<0.05), but the glandular epithelium (Figure 1M), stroma (Figures 1M and 1N), and arterioles (Figure 1N) exhibited only a moderate level of prostasin signals (p<0.05). In addition, signals in the placental villi, trophoblastic column, and trophoblastic shell on day 26 (Figures 1K and 1L) increased significantly (p<0.05) when compared with those on day 18 (Figures 1C and 1D). Intensities of prostasin mRNA in the glandular epithelium at three stages of pregnancy significantly (p<0.05) decreased with the progress of gestation (Figures 1A, 1B, 1E, and 1M). Only weak reactions for PN-1 were detected in the placental and endometrial compartments on day 26 (Figures 1P-1S) (p<0.05). Representative negative control results of sense-probe hybridization for prostasin and PN-1 mRNA, respectively, are shown in Figures 1O and 1T in the endometrium on day 18.
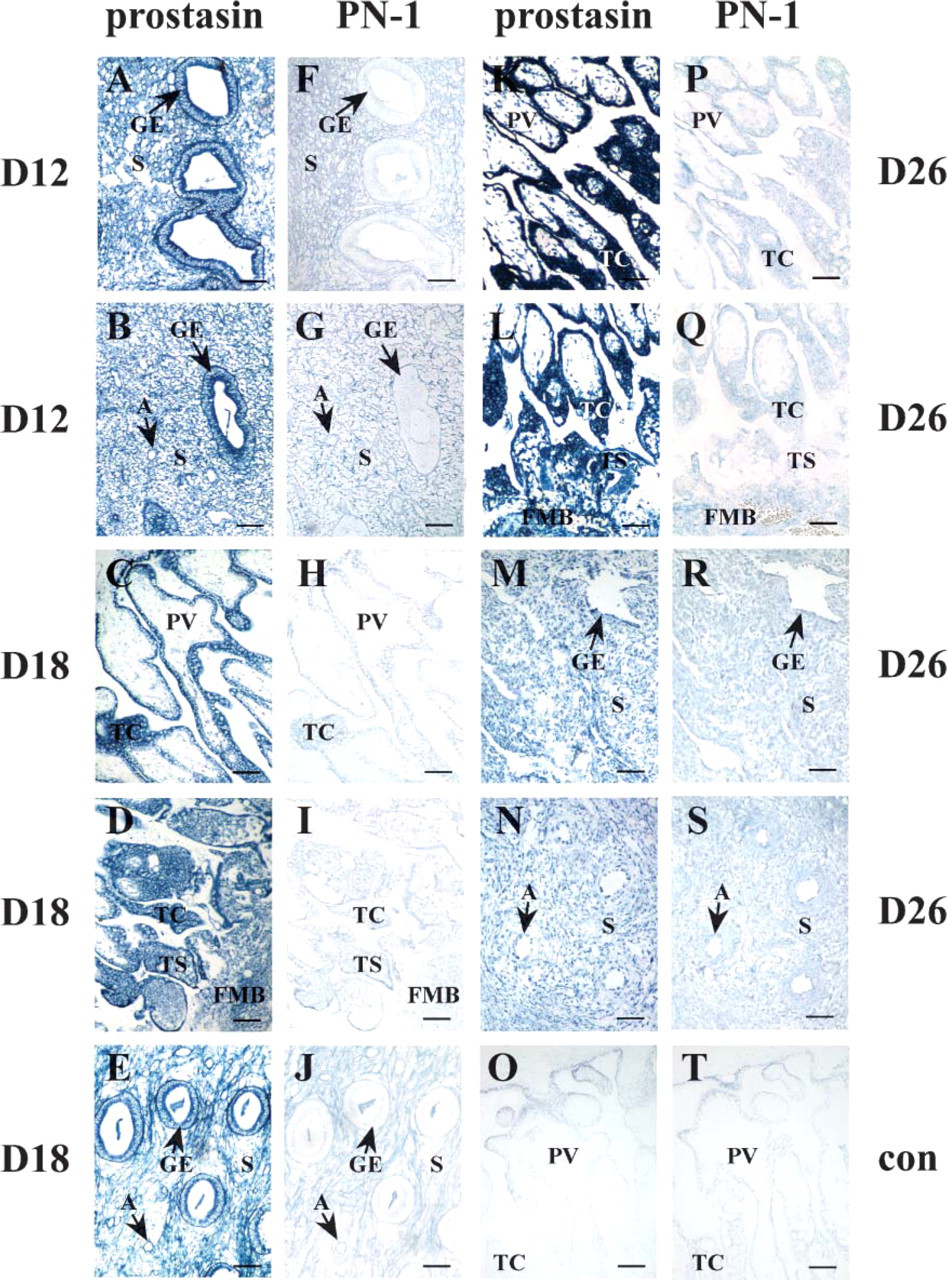
In situ localization of prostasin and PN-1 mRNA in rhesus monkey endometrium and placenta on days 12 (D12), 18 (D18), and 26 (D26) of pregnancy.
Densitometric quantification of prostasin and PN-1 transcripts in rhesus monkey uterus and placenta on days 12 (D12), 18 (D18), and 26 (D26) of pregnancy
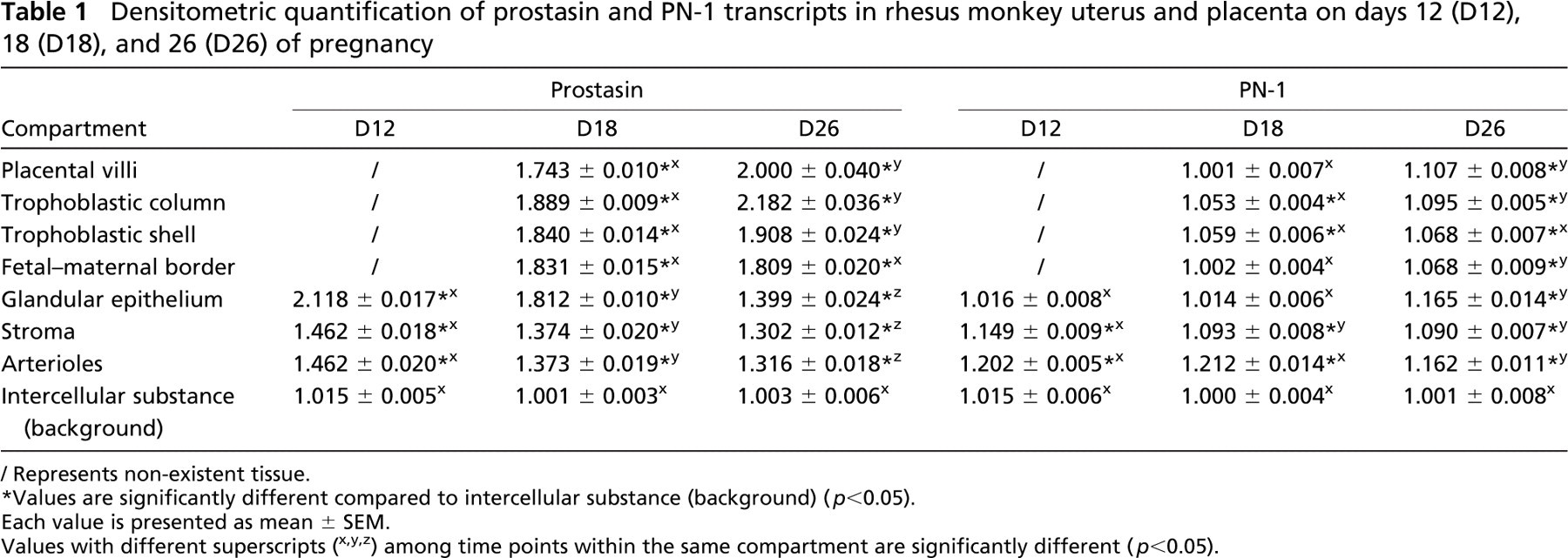
Represents non-existent tissue.
Values are significantly different compared to intercellular substance (background) (p<0.05).
Each value is presented as mean ± SEM.
Values with different superscripts (x,y,z) among time points within the same compartment are significantly different (p<0.05).
IHC Localization of Prostasin and PN-1 Protein in the Macaque Endometrium and Placenta During Early Pregnancy
IHC analysis was used to examine prostasin and PN-1 protein localization, which confirmed the data obtained by ISH. As shown in Figure 2 and Table 2, the antiprostasin antiserum produced strong staining in the glandular epithelium (Figures 2A and 2B) (p<0.05) on day 12 and in the placental villi (Figures 2C and 2D), trophoblastic column (Figures 2C and 2D), trophoblastic shell (Figure 2D), fetal-maternal border (Figure 2D), and glandular epithelium (Figure 2E) (p<0.05) on day 18. Furthermore, anti-prostasin immunoreactivity was moderate in the stroma and arterioles on days 12 (Figures 2A and 2B) (p<0.05) and 18 (Figure 2E) (p<0.05). On day 26, anti-prostasin immunoreactivities in the placental villi (Figure 2K), trophoblastic column (Figure 2K), trophoblastic shell (Figure 2L), and fetal-maternal border (Figure 2L) were intense (p<0.05). Specifically, signals in the placental villi and fetal-maternal border on day 26 (Figures 2K and 2L) increased significantly (p<0.05) when compared with those on day 18 (Figures 2C and 2D). However, prostasin signals in the glandular epithelium (Figure 2M), stroma (Figures 2M and 2N), and arterioles (Figures 2M and 2N) on day 26 were moderate (p<0.05).
IHC staining with anti-PN-1 antiserum revealed a general lack of expression of this protein. Only light immunopositive PN-1 reactions were detected in the endometrial compartments on day 12 (Figures 2F and 2G) (p<0.05) and in the placental and endometrial compartments on days 18 (Figures 2H-2J) (p<0.05) and 26 (Figures 2P-2S) (p<0.05).
Negative control staining with normal rabbit serum on day 18 produced negligible background (Figures 2O and 2T).
Discussion
Much evidence exists to show that serine proteases are involved in matrix degradation required for implantation. For instance, uPA and tPA are present in uterus or trophoblast (Hofmann et al. 1994; Harvey et al. 1995; Lockwood and Schatz 1996; Teesalu et al. 1996; Menino et al. 1997; Schatz et al. 1999), implicating their roles in embryo implantation. Rat and human endometrial kallikrein gene expression has also been demonstrated (Clements et al. 1997). In this report, we present the first evidence to demonstrate the localization of prostasin in primate endometrial compartments and trophoblast. We showed by both ISH and IHC that prostasin was highly expressed in the glandular epithelium of macaque endometrium on days 12 and 18 of pregnancy but was decreased to a rather moderate level on day 26. It has been well documented that secretions of endometrial glands serve as primary regulators of implantation and placentation, and interaction between glandular epithelium and stroma will lead to local control and coordination of morphogenetically important cell behaviors such as movement, adhesion, differentiation, and proliferation (Gray et al. 2001). Therefore, intense epithelial localizations of prostasin on days 12 and 18 raise the possibility that this serine protease is involved in the establishment of uterine epithelial morphology, which in turn contributes to tissue remodeling of the endometrium during macaque early pregnancy. Decreased expression on day 26 may be a hint that its epithelial function is reduced with the progress of pregnancy.
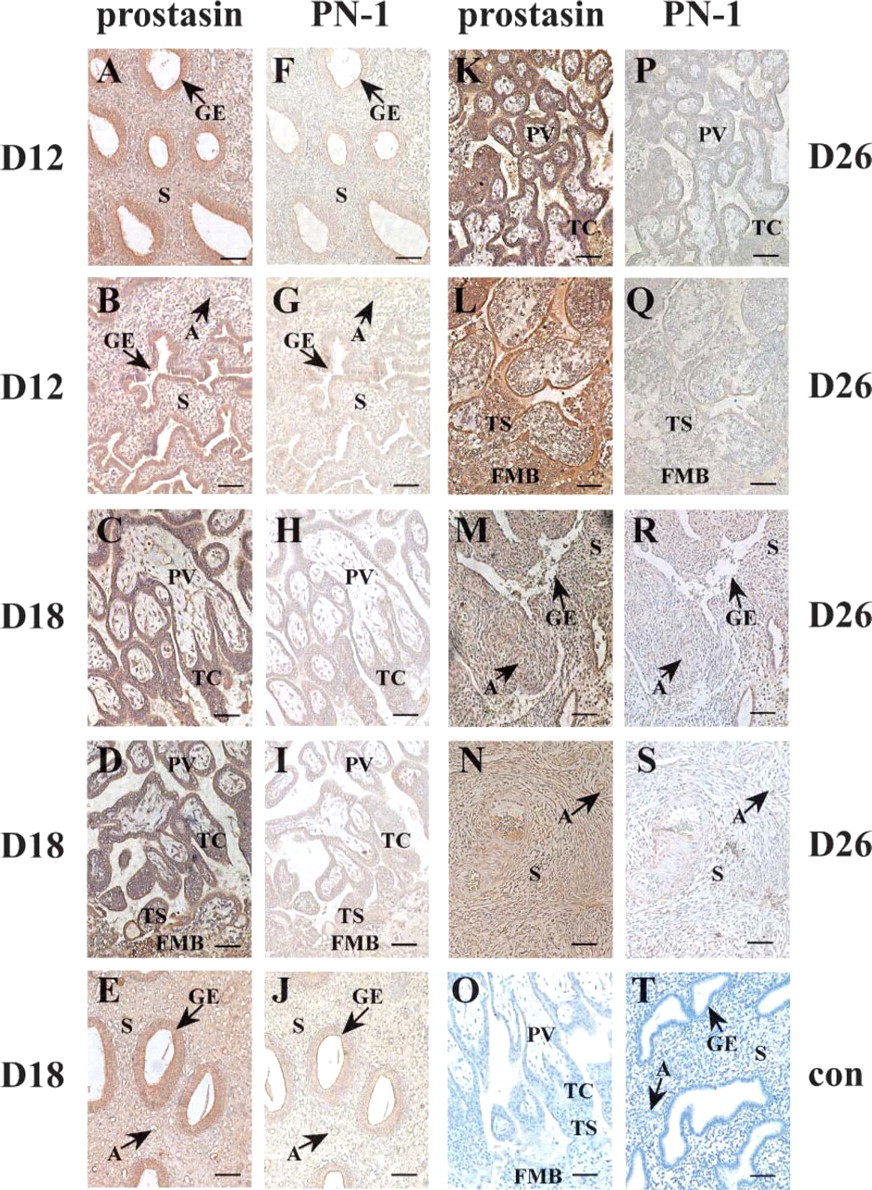
Immunohistochemical localization of prostasin and PN-1 in rhesus monkey endometrium and placenta on days 12 (D12), 18 (D18), and 26 (D26) of pregnancy.
Densitometric quantification of prostasin and PN-1 proteins in rhesus monkey uterus and placenta on days 12 (D12), 18 (D18), and 26 (D26) of pregnancy
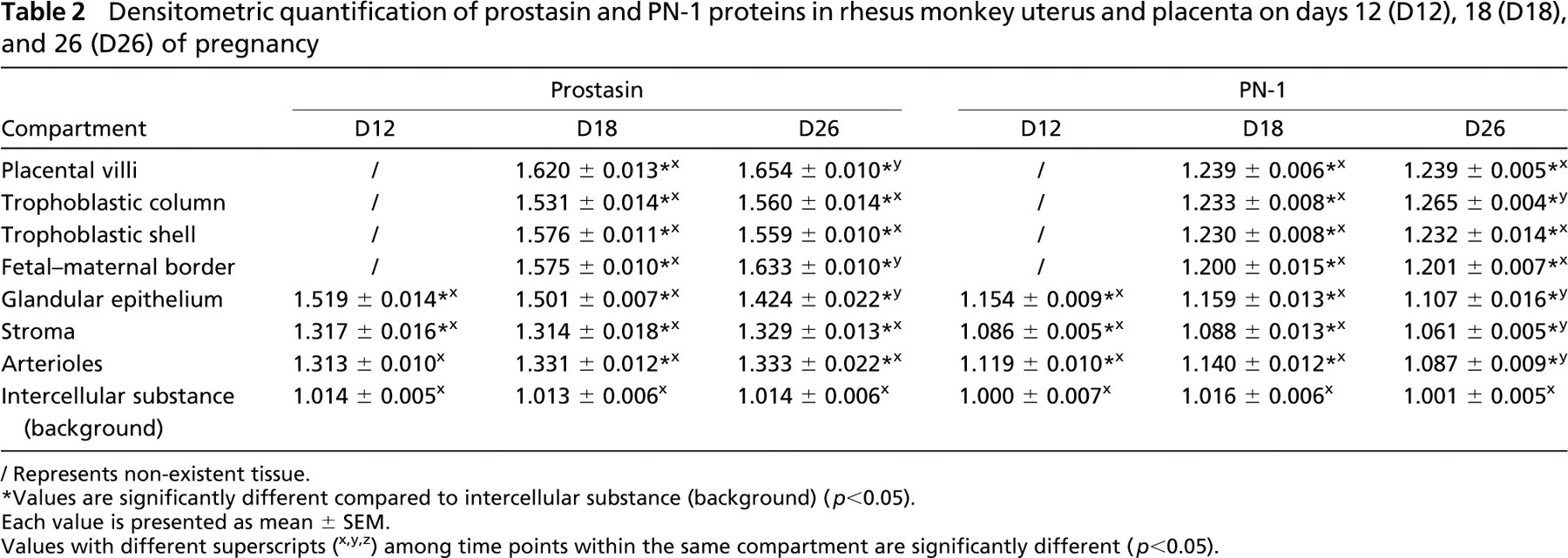
Represents non-existent tissue.
Values are significantly different compared to intercellular substance (background) (p<0.05).
Each value is presented as mean ± SEM.
Values with different superscripts (x,y,z) among time points within the same compartment are significantly different (p<0.05).
Embryo implantation occurs on day 9.5 of pregnancy in rhesus monkey (Enders et al. 1983). Selected uteri on days 12, 18, and 26 of pregnancy in our study represent tissues from lacunar stage, early villous stage, and villous stage, respectively, during the developmental process of the placenta (Carson et al. 2000; Ghosh et al. 2000). In our assay, prostasin was intensely localized in the placental villi, trophoblastic column, trophoblastic shell, and the fetal-maternal border on day 18 and was further enhanced in these placental compartments on day 26, as shown by ISH and/or IHC. These results suggest that prostasin serve to participate in the early development of the villus, rapid matrix remodeling in placental tissue, and invasion of the trophoblast cells during early villous stage and villous stage.
Subunits of the ENaC are expressed in the endometrial epithelium, and their expression is regulated by hormones and dietary sodium. However, a clearly defined role in the uterus for this ion channel is presently pending (Tsang et al. 2004; Yang et al. 2004; Salleh et al. 2005). ENaC had first been suggested as a physiological substrate for prostasin serine protease, which was also named as channel-activating protease 1 (CAP-1) and protease serine S1 family member 8 (Prss 8) by these authors (Vallet et al. 1997,2002; Leyvraz et al. 2005). One recent report presented updated evidence that the serine protease activity of prostasin/CAP-1 may not be required for its role in activating the ENaC (Andreasen et al. 2006). Whereas prostasin/CAP-1 may be involved in the activation events of the ENaC by an alternative non-proteolytic mechanism, its proteolytic functions may be required for modulating other cellular activities such as the response to inflammation (Chen et al. in press). In the latter context, timing and pattern of PN-1 expression regulation become very relevant.
As PN-1 has been proven to be the inhibitor of prostasin (Chen et al. 2004), we tried to investigate whether PN-1 is coordinately localized with prostasin to maintain a balanced presence of a serine protease and its inhibitor during endometrial matrix remodeling and controlled trophoblast intrusion in macaque early pregnancy. However, such was not the case because the expression of PN-1 at either the mRNA or the protein level in the compartments of endometrium and placenta on days 12, 18, and 26 of pregnancy was weak or hardly detectable, a pattern distinct from that in human term placenta (White et al. 1993). In another study recently completed by our group on macaque ovary during the menstrual cycle and pregnancy, PN-1 was coordinately localized with prostasin in preovulatory follicles and corpus luteal cells (unpublished data). It has been reported that PN-1 expression in rat uterus is at the highest level on day 6.5 postcoitum, the time of implantation, and decreases after day 13.5 of pregnancy, corresponding to the time point of postimplantation (Kim et al. 2001). Whether PN-1 is expressed at the highest level in macaque endometrium at the time of implantation (day 9.5 of pregnancy) and then decreased is a subject that requires further study. PN-1 is a broad-spectrum protease inhibitor, which also inhibits the proteolytic activity of plasminogen activators, plasmin, trypsin, and thrombin (Murer et al. 2001). Accordingly, the absence or weak expression of PN-1, contrary to the intense distribution pattern of prostasin, may contribute to the intense matrix remodeling in both endometrial and placental tissue, which needs the activity of prostasin. Alternatively, PN-1 may not function to control the proteolytic prostasin during macaque early pregnancy. Due to the fact that proteolysis in the endometrium and placenta during early pregnancy is spatially restricted and requires a fine-tuned balance between the level of protease and inhibitor, the question of whether there is another serine protease inhibitor functioning to control the activity of prostasin needs to be further investigated. A recent identification of hepatocyte growth factor activator inhibitor-1B as a potential inhibitor of prostasin (Fan et al. 2005) might provide the clue.
In conclusion, we have presented data that suggest a role for prostasin in the glandular secretion, endometrial matrix remodeling, early development of the villus, and trophoblastic invasion during macaque early pregnancy. PN-1 may not be involved in controlling prostasin activities during these events.
Footnotes
Acknowledgements
This work was supported by the Special Funds for Major State Basic Research Project of China, the Knowledge Innovation Program of the Chinese Academy of Sciences (KSCX3-IOZ-07), the Department of Defense Prostate Cancer Research Program, Grant #DAMD17-02-1-0032 (to KXC), and funds from National Institutes of Health Grant #HD-40241 (to L-MC).
