Abstract
CD208 DC lysosomal-associated protein is a marker of activated human dendritic cells; however, recently it was described as a marker of adult type II pneumocytes in many species including humans and sheep. Our hypothesis was that CD208 is developmentally regulated in lung pneumocytes. Lamb lungs at varying stages of development were stained immunohistochemically for CD208 and with Nile red (a fluorescent stain for lamellar bodies of type II cells) along with pulmonary markers of maturation (glycogen stores and surfactant protein A [SP-A] expression) or proliferation (Ki-67). CD208 staining and Nile red were localized to rare pneumocytes in young fetal lambs (day 115), increasing in frequency and stain intensity with age. Periodic acid-Schiff staining of glycogen granules was most prominent in the young lambs (day 115) with reduced staining through advancing lung development. SP-A was detected in pulmonary epithelia and staining in alveoli increased through gestation with decreased staining at 2 weeks of age. Intranuclear Ki-67 staining decreased through late gestation but was increased in 2-week-old lambs. Ontogeny of CD208 staining and depletion of glycogen were correlated (p<0.0001) and consistent with the premise that CD208 is localized to developing lamellar bodies. The findings suggest that CD208 antigen expression may serve as a marker for pneumocyte maturation in the developing fetal lung.
I
It was reported recently that dendritic cell lysosomal-associated membrane protein (DC-LAMP or CD208) was expressed specifically in normal and transformed type II pneumocytes of adult humans and showed similar specificity in sheep and mice (Salaun et al. 2004). CD208 is classically described as a protein marker expressed in activated human, but not murine, dendritic cells (Salaun et al. 2003). In human dendritic cells, CD208 is associated with the intracellular major histocompatibility complex (MHC) II compartment, but following MHC II dissociation to the cell surface it is present in the perinuclear lysosomes (Liu et al. 1996; de Saint-Vis et al. 1998). Specificity of CD208 to type II pneumocytes in the healthy adult lung of many species is a significant finding; however, its application to perinatal models of developmental disease is not clearly defined. The late-gestational to perinatal lamb is commonly used as a model for pulmonary development/disease including surfactant regulation, congenital diaphragmatic hernia, ventilation-induced injury, and paramyxovirus infection (Tan et al. 1999; Luks et al. 2001; Ikegami and Jobe 2002; Meyerholz et al. 2004a,b). Alveolar type II pneumocytes are critical cellular components in the study of these pulmonary models. Recently, CD208 immunofluorescence has been successfully applied in laser capture microdissection retrieval of type II pneumocytes in perinatal lamb lung (Meyerholz et al. 2006); however, during ontogeny the detection and distribution of CD208 immunohistochemical (IHC) expression has not been determined. Our hypothesis is that CD208 expression is developmentally regulated in the young lamb, corresponding to maturation of pneumocytes. In this study, cellular CD208 staining during pulmonary development was determined and compared with markers of pulmonary maturation (glycogen depletion of epithelia and surfactant protein A [SP-A] expression) and proliferation (Ki-67).
Materials and Methods
Animals/Tissues
Date-mated ewes were acquired from Iowa State University Laboratory of Animal Resources, and all procedures were approved by the Animal Care and Use Committee of Iowa State University. Ewes were euthanized on appropriate days of gestation and lamb tissue surgically extracted, or following natural birth the lambs were euthanized on the appropriate day. Lamb lung tissues were collected for gestational age (ga) day 115 (n=5) and 130 (n=4) or within 6 hr of natural birth (day 145 ga, n=4) or 2 weeks postnatally (day 160 ga, n=4). Tissues were consistently taken from the right middle lung lobe and placed in 10% neutral-buffered formalin for 24 to 48 hr. These were further trimmed, placed in cassettes, and processed for routine paraffin embedding. Lung tissue lacked lesions.
IHC
For CD208 and Ki-67 IHC staining, 6-μm lung sections were cut onto slides and for the following steps the entire set of slides were processed together. Slides were deparaffinized through a series of xylene and alcohol baths (including a 2% hydrogen peroxide in methanol bath for 15 min). Microwave antigen retrieval (~1 min at 1000 W, then 3 min at 300 W) was performed with slides in a commercial buffer (AR-10; BioGenex, San Ramon, CA). Slides were washed with Bio-Genex Wash Buffer (PBS, pH 7.4, containing 0.1% Tween 20, hereafter referred to as BioGenex PBS) for 5 min, and a liquid wax pen was then used (PAP-pen; BioGenex) to mark each slide above and below the tissue to corral subsequent reagents over the tissue on each slide and also to prevent loss of reagent from each slide. Blocking serum was applied (20% normal goat serum in BioGenex PBS, 20 min), and then subject slides were incubated (4C for 3 days) with either primary mouse monoclonal anti-CD208 antibody (1:5000; IM3448, Beckman Coulter, Miami, FL) or mouse monoclonal anti-Ki-67 (1:25,000; M7420, DAKO, Carpinteria, CA) in a commercial diluent (Common Antibody Diluent, HK156-5K; BioGenex) supplemented with 5% normal goat serum (Sigma; St Louis, MO). Slides were allowed to warm to room temperature for 30 min, rinsed twice with BioGenex PBS, incubated with 3% normal sheep serum in BioGenex diluent (45 min), and followed by a preincubation for 30 sec with biotinylated secondary goat anti-mouse IgG antibody (1:200 for 45 min; KPL, Gaithersburg, MD) in BioGenex diluent. Slides were cleared of the first secondary application (without rinsing), and the same biotinylated secondary goat anti-mouse IgG antibody solution was again applied for 45 min and then washed twice with BioGenex PBS. Endogenous peroxidases were again blocked by application of 3% hydrogen peroxide (in BioGenex PBS) for 25 min followed by two BioGenex PBS rinses and a 5-min incubation in BioGenex PBS. Supersensitive streptavidin-conjugated horseradish peroxidase (SS-HRP; BioGenex) was then applied for 45 min followed by two BioGenex PBS rinses and a 5-min BioGenex PBS incubation. Next, chromogen (Nova Red for 5 min; Vector Laboratories, Burlingame, CA) was applied, and sections were washed five times with water. Slides were rinsed briefly in a fresh water bath and then counterstained with one-third strength Harris hematoxylin (Shandon-Lipshaw; Pittsburgh, PA) for 2 min, dehydrated through graded alcohol and xylene baths, and coverslipped with Permount (Sigma). Control slides (one lung section from each time point) lacked primary antibody. Additional sections from each lung not used for IHC were stained with periodic acid-Shiff (PAS) stain.
For SP-A IHC staining, tissues were treated as previously described (Grubor et al. 2004) using primary mouse IgM anti-human SP-A antibody (1:50, MAB3270; Chemicon International, Temecula, CA) at 4C for 3 days.
For staining of lamellar bodies with Nile Red, frozen sections from animals of each age of gestation were soaked in PBS for 5 min, then incubated with two to three drops of Nile red (5 μg dye/ml in 75% glycerol), coverslipped, and viewed with an Olympus fluorescent microscope (Olympus; Tokyo, Japan) with a 450- to 500-nm excitation filter (Fowler and Greenspan 1985).
Microscopic Scoring
For CD208, SP-A, and Ki-67, stained cells were counted in nine alveoli per tissue section with at least two sections per animal and from all animals per time point. Alveoli were of similar size and shape (between 150 and 300 μm in diameter) with the average per alveolus being recorded by a pathologist blinded to the specifics of the examined tissue. Target stain intensity was scored by a pathologist using the following scale: 0 = no detectable staining, 1 = first detectable staining, 2 = light to pale brown, 3 = deep, dark brown. Cytoplasmic glycogen stores of developing alveoli were scored as follows: 0 = no detectable staining, 1 = first detectable staining, 2 = cytoplasmic glycogen granules in >30% of cells, 3 = cytoplasmic staining in 30-60% of cells, and 4 = cytoplasmic granules in 60+% of cells.
Statistics
To assess stain expression during development, linear regression was used to calculate the sign (+/-) of the slope coefficient and its significance from zero. For comparison of detection among age groups, a one-way ANOVA was used to assess for group effects. Where significant group differences were detected, posthoc tests (with Bonferroni correction) were performed on groups that had scientific relevance. Spearman's rank order correlation coefficient test was applied to assess relationship between CD208 expression and target staining. Unless otherwise noted, statistical significance was set at p<0.05.
Results
CD208 Staining
CD208 staining was localized to multifocal cuboidal cells lining and within the alveolar septa consistent with type II pneumocytes. Staining was cytoplasmic, and intensity was prominent lining the periphery of small (~0.75-2.0 μm) clear vacuoles (Figure 1). These stained vacuoles were often diffuse to eccentrically (luminal side) located in the apical cytoplasm. CD208 staining was not observed in the epithelia of bronchioles or bronchi. Lamb developmental age affected the extent and intensity of cellular staining. Lungs at day 115 ga had rare, faint staining within cells of similar type to older lambs (Figure 2 and Figure 3). CD208 staining increased with advanced age in frequency (p<0.001, r = 0.81) and intensity (p<0.001, r = 0.88) from day 115 of gestation through 2-week-old lambs (Figure 4).
Detection of Lamellar Bodies With Nile Red
Staining distribution and intensity were similar to CD208 IHC staining. Staining was present in multifocal cuboidal cells lining and within the alveolar septa consistent with type II pneumocytes. Staining was cytoplasmic and intensity was prominent (Figure 5). Lamb developmental age affected the extent and intensity of cellular staining. Nile red staining increased with advanced age in frequency and intensity from day 115 of gestation through 2-week-old lambs.
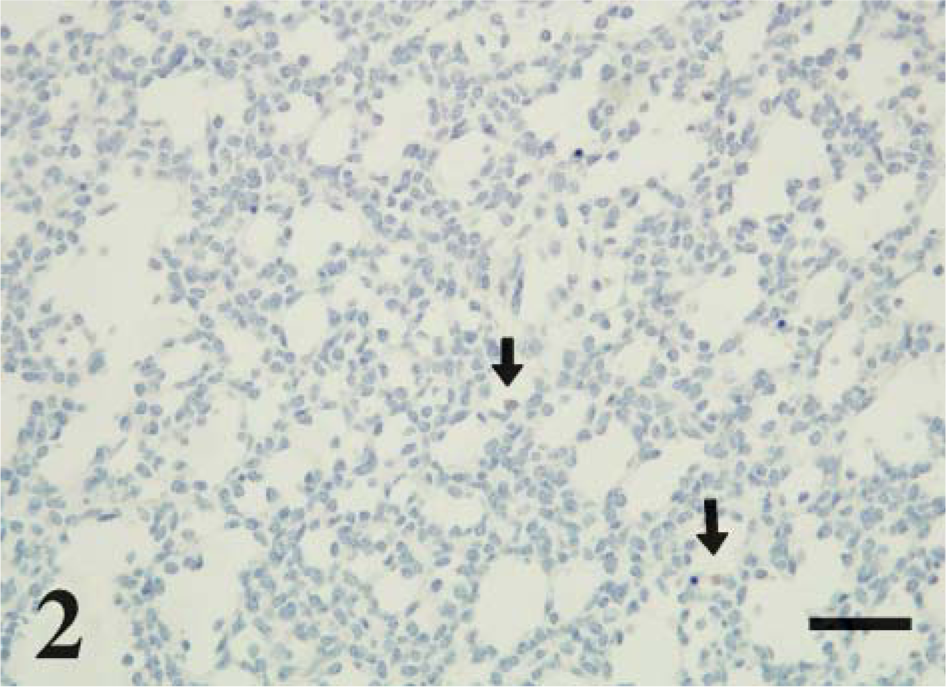
Day 115 ga, preterm lamb lung. CD208 IHC. CD208 staining is found in rare cuboidal cells (arrows) lining the developing alveoli. Bar = 100 μm.
Glycogen Storage
To compare CD208 staining to pulmonary maturation, the quantity of glycogen stores (PAS staining) were determined. In alveoli, PAS-stained sections from lambs at day 115 of gestation had abundant cytoplasmic magenta granules (~0.5-2.5 μm) in cuboidal cells, and the number of magenta granules decreased with advancing age (p<0.001, r =-0.94) (Figure 6-Figure 8). There was a significant correlation (p<0.0001, r = 0.80) during development between CD208 staining and the depletion of PAS granules in the pneumocytes.
SP-A Staining
In all age groups, SP-A cellular staining was present in epithelia of the alveoli (especially type II pneumocytes), bronchioles, and bronchi, including submucosal glands. Light granular to diffuse cytoplasmic staining was present in epithelia of the bronchi in day 115 lambs, less so in bronchioles, and rare in alveoli. With increasing ga, the distal airway epithelia (bronchioles and alveoli) increased in staining frequency and intensity. From day 115 to term, cellular frequency (p<0.001) and intensity (p<0.05) of SP-A staining increased but decreased from term to 2 weeks of age (p<0.001). Increases of CD208 and SP-A staining were correlated from late gestation to term (p<0.01, r = 0.70, Figure 9).
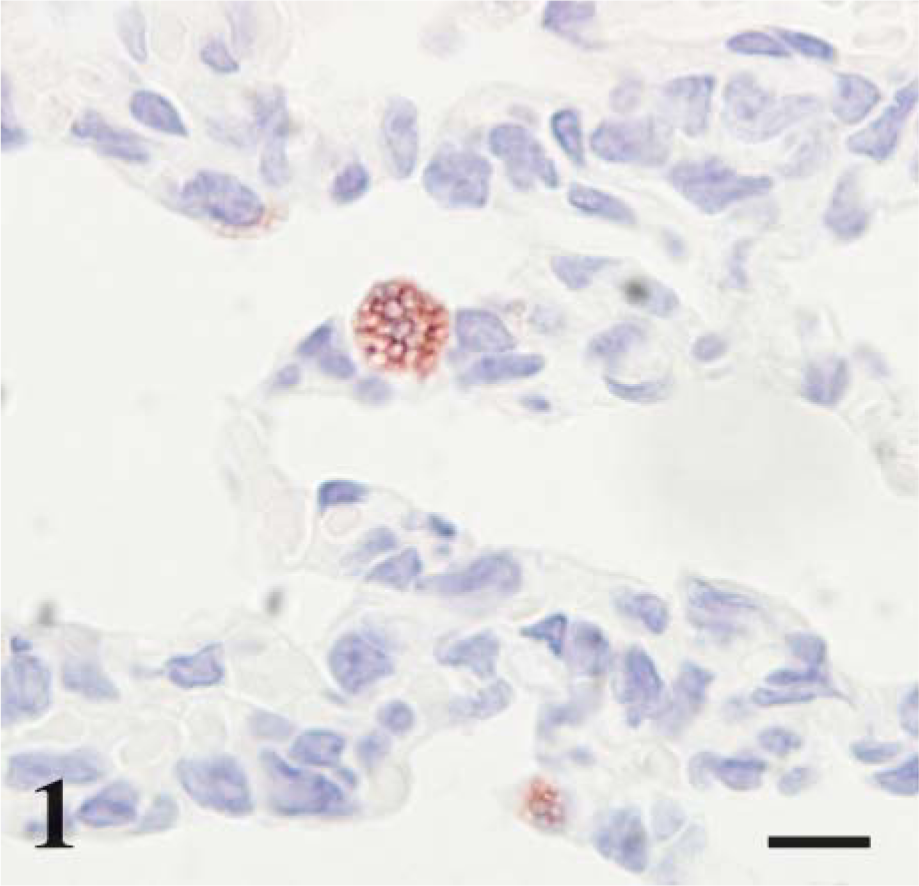
Day 160 gestational age (ga) postnatal lamb lung, CD208 immunohistochemistry (IHC). The alveolar epithelia retain stain along the peripheral borders of cytoplasmic vacuoles. Bar = 10 μm.
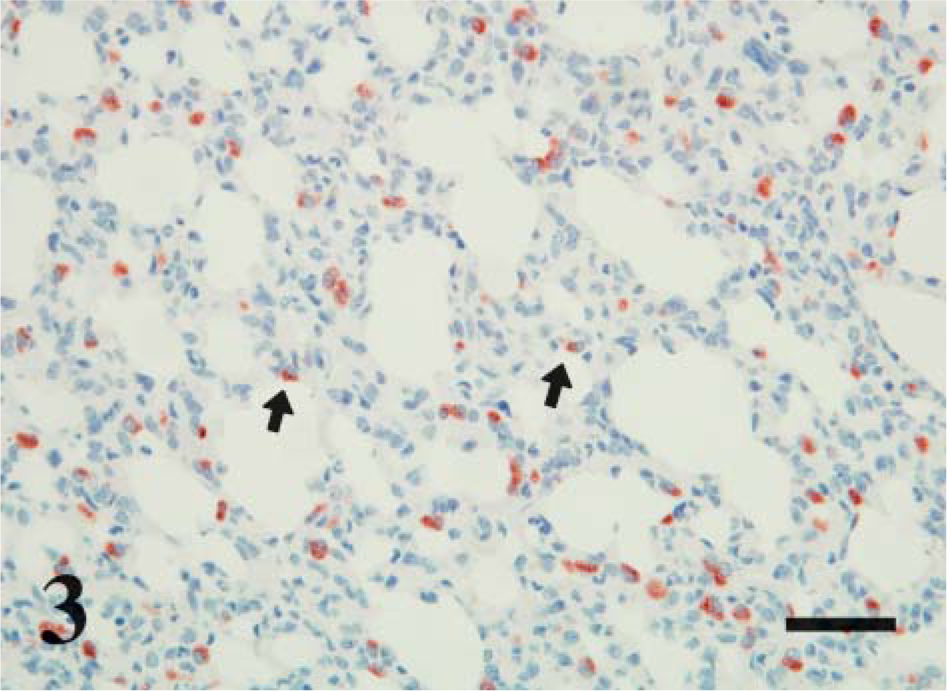
Day 160 ga, postnatal lamb lung. CD208 IHC. CD208 is in cuboidal cells scattered along the alveolar septa. Staining is prominent on the limiting membrane of vacuoles that are either diffuse or apically (arrows) located in the cytoplasm. Bar = 100 μm.
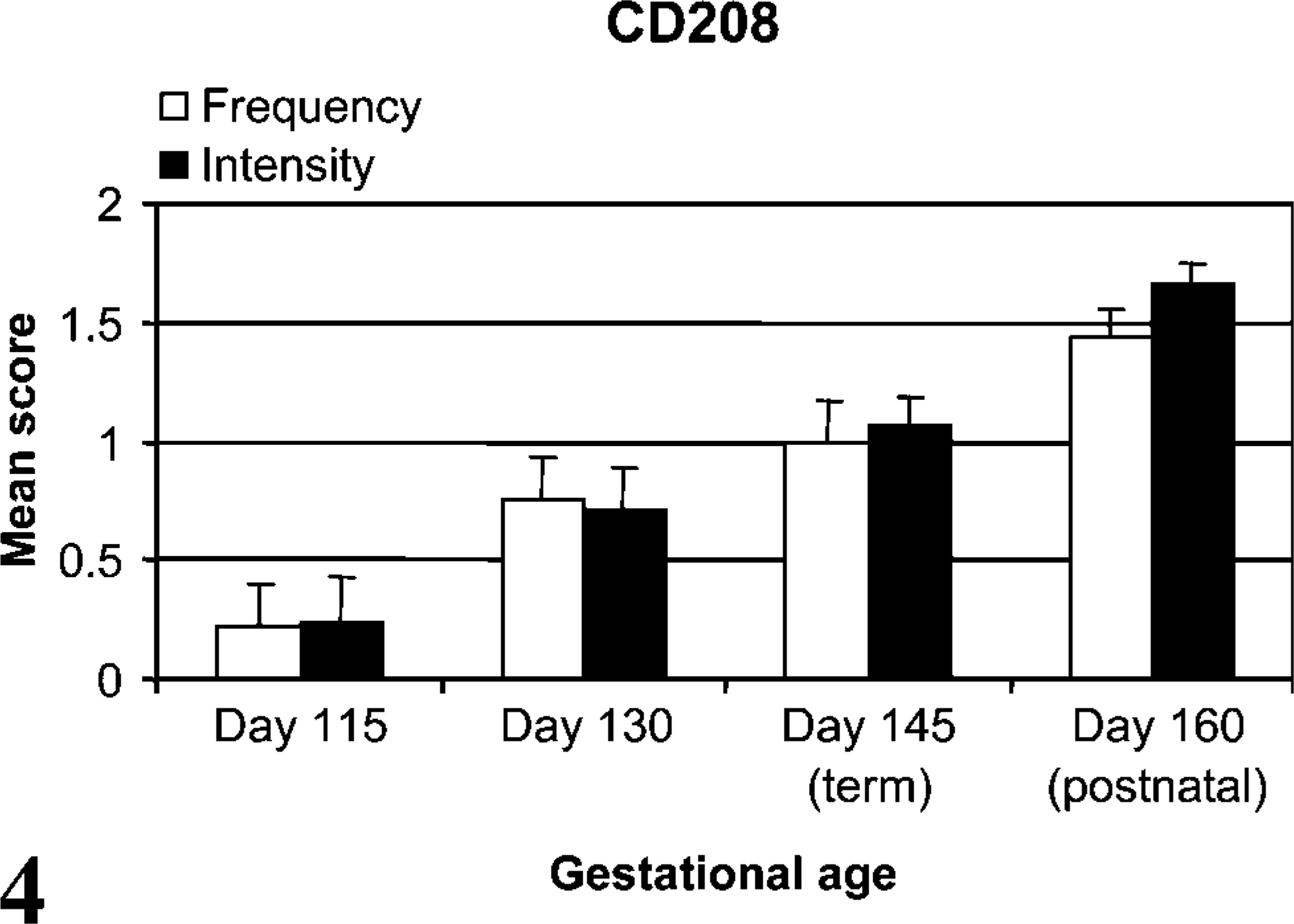
CD208 stain frequency and intensity. Both the frequency (p<0.001, r = 0.81) and intensity scores (p<0.001, r = 0.88) of CD208 stain increased throughout perinatal development.
Ki-67 Staining
Ki-67 staining included scattered cells of the bronchi, bronchioles, and alveoli with intranuclear localization characterized as granular to diffuse. Alveolar Ki-67 IHC staining decreased from day 115 to day 145 of gestation (p<0.05) with a significant increase from term to 2-week old lambs (p<0.01) (Figure 10). Although a decrease in Ki-67 staining was detected through gestation, this did not correlate statistically with the increased detection of CD208 antigen.
Discussion
In this study, CD208 staining (for all age groups) was limited to cuboidal cells of the alveolar septa that were anatomically and morphologically consistent with type II pneumocytes. This is similar to observations in adult lung where transmission electron microscopy and immunogold labeling were used to localize the CD208 antigen to the limiting membrane of lamellar bodies in type II pneumocytes with weak localization to multivesicular bodies and the surface plasma membrane (Salaun et al. 2004). Cellular and cytoplasmic localization of CD208 corresponds to the staining seen in the current study as apical to diffuse cytoplasmic vacuoles with principal staining seen along the periphery (i.e., limiting membrane) of the vacuoles. The vacuoles are consistent with lamellar bodies; the central clear-space represents lipid that was dissolved/extracted during routine paraffin embedding and IHC processing. A similar distribution of cells with lamellar bodies was seen with Nile red-stained frozen sections. Nile red is also a marker of type II cells (Fowler and Greenspan 1985).
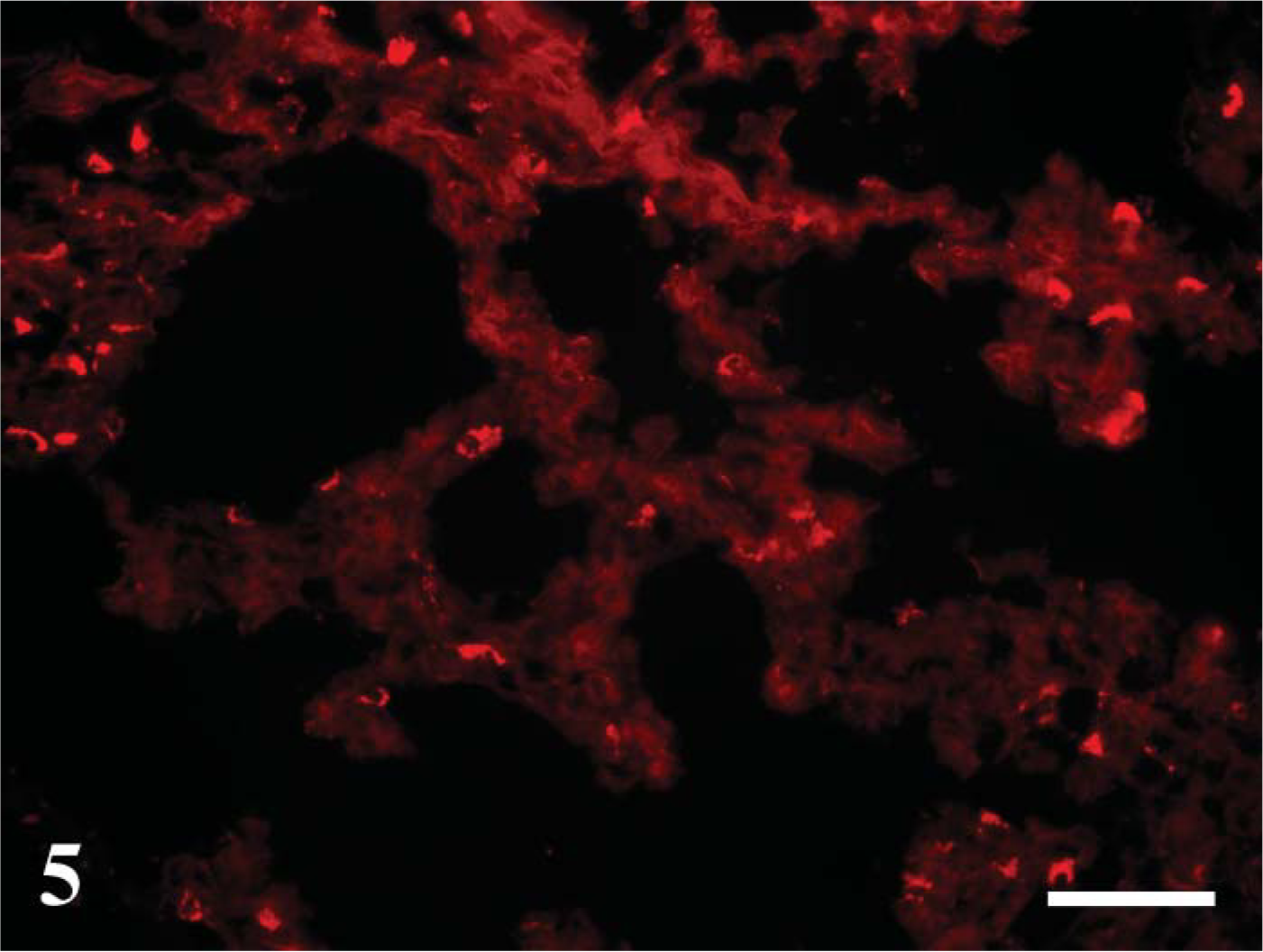
Day 145 ga (newborn). Nile red staining. Multifocal individual cells are stained, and stained cells have abundant fluorescence throughout the cytoplasm. At earlier gestational time points, only small areas of occasional cells stained (not shown). This pattern is similar to CD208 distribution. Bar = 100 μm.
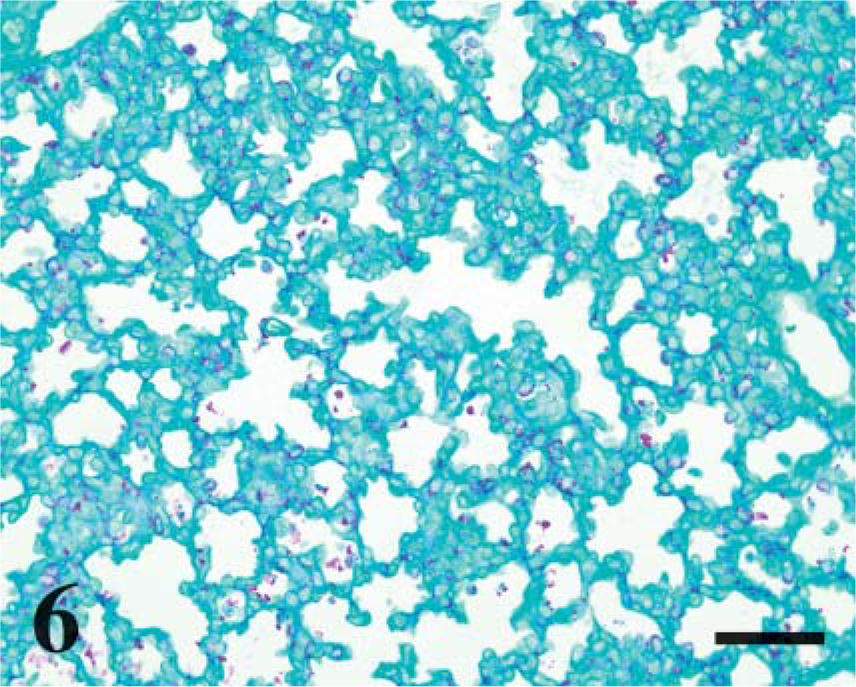
Day 115 ga, preterm lamb lung. Periodic acid-Shiff (PAS) stain. Numerous cytoplasmic aggregates of glycogen (magenta-colored granules) line the alveolar septa. Bar = 100 μm.
SP-A staining was detected in proximal (e.g., bronchi) to distal (e.g., alveolar type II pneumocytes) pulmonary epithelia. Intensity of SP-A staining was relatively uniform in bronchi for all age groups, whereas bronchiolar and alveolar staining generally increased through day 145 (term), which correlated to CD208 increased expression during the same time period. Increase of SP-A in the lamb is similar to SP-A mRNA and protein localization in the developing human lung. In humans, developmental SP-A expression is detectable earlier in the proximal airway epithelium (e.g., trachea, bronchi and bronchial glands), whereas distal airway epithelia (bronchioles and alveoli) did not express SP-A until later in gestation (Khoor et al. 1993). In this current study, SP-A staining in type II pneumocytes increased through gestation and was distinct in cellular distribution and morphology from CD208 staining. Whereas increasing SP-A mRNA and protein expression are associated with late-term gestational development, SP-A antigen is typically localized in intracellular sites apart from lamellar bodies (Ochs et al. 2002; Meyerholz et al. 2006). This is due to SP-A cellular trafficking pathways that bypass localization in the lamellar bodies, whereas CD208 resides in the limiting membrane of these specialized organelles (Ochs et al. 2002; Salaun et al. 2004). SP-A and CD208 both had developmental increases of cell staining through gestation, but CD208 staining continued to increase postnatally in frequency and intensity following birth. Increased CD208 staining is likely due to continued maturation and proportion of pneumocytes through at least 2 weeks of age as previously documented (Flecknoe et al. 2003). The postnatal decrease in SP-A antigen is uncharacteristic of what is typically seen through the postnatal period of many species, but it has been documented to decrease months after birth in the rat (Ohashi et al. 1994; Xu et al. 1998; Beresford and Shaw 2003).
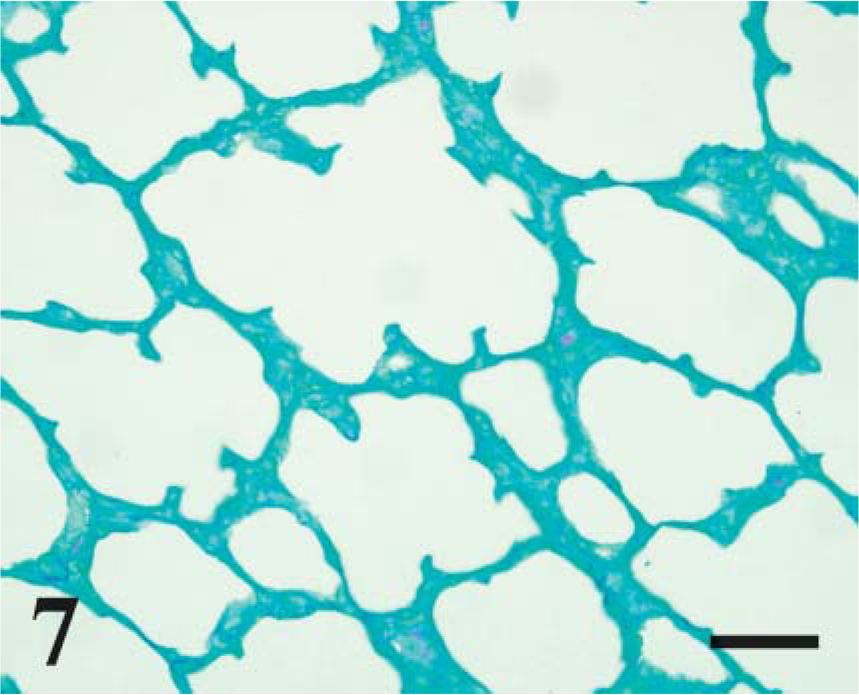
Day 160 ga, postnatal lamb lung. PAS. Granular cytoplasmic staining for glycogen is rare. Bar = 100 μm.
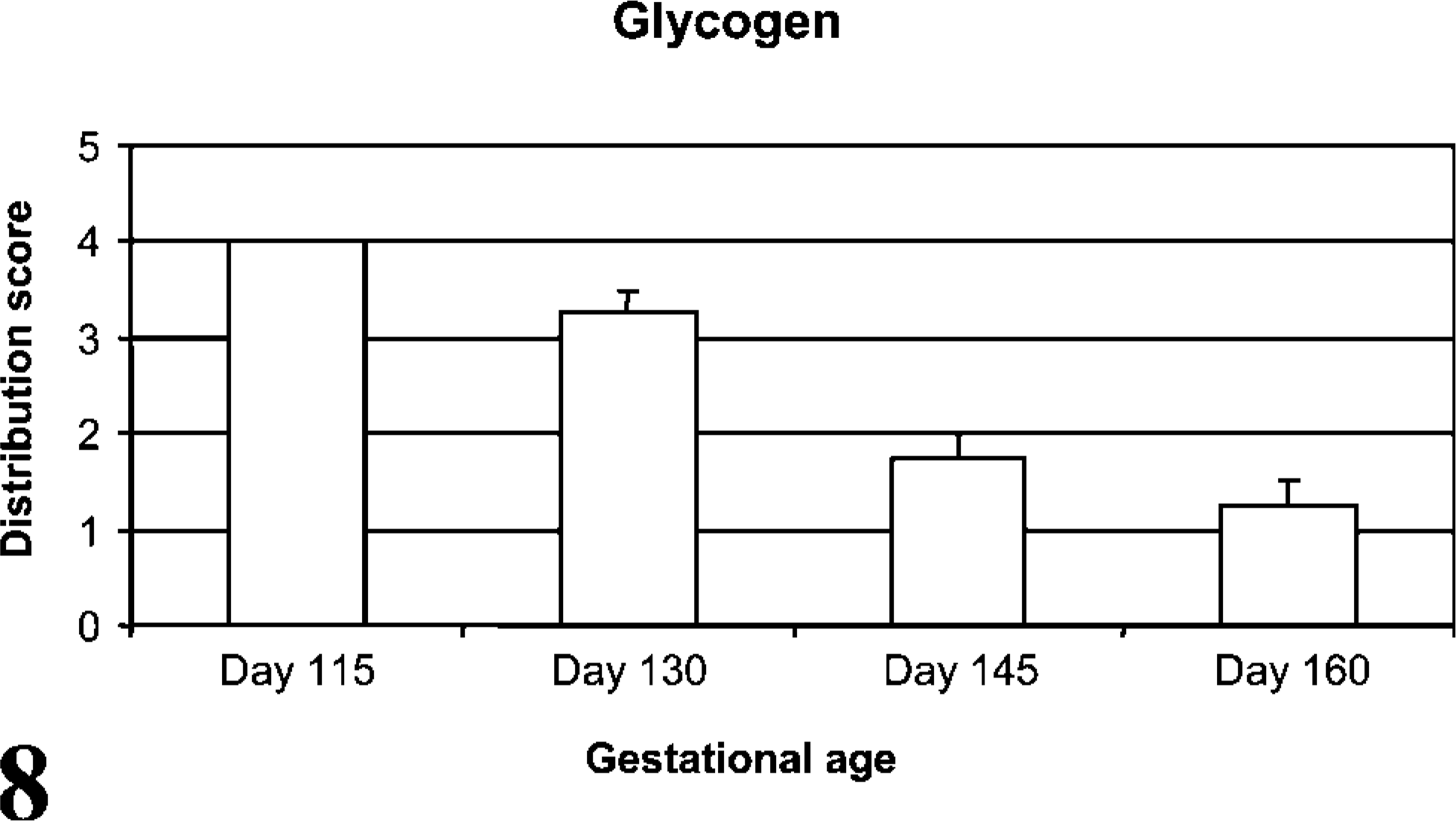
Cytoplasmic glycogen stores assessed by PAS stain. Distribution of granular glycogen staining in alveolar type II pneumocytes decreased through development (p<0.001, r =-0.94).
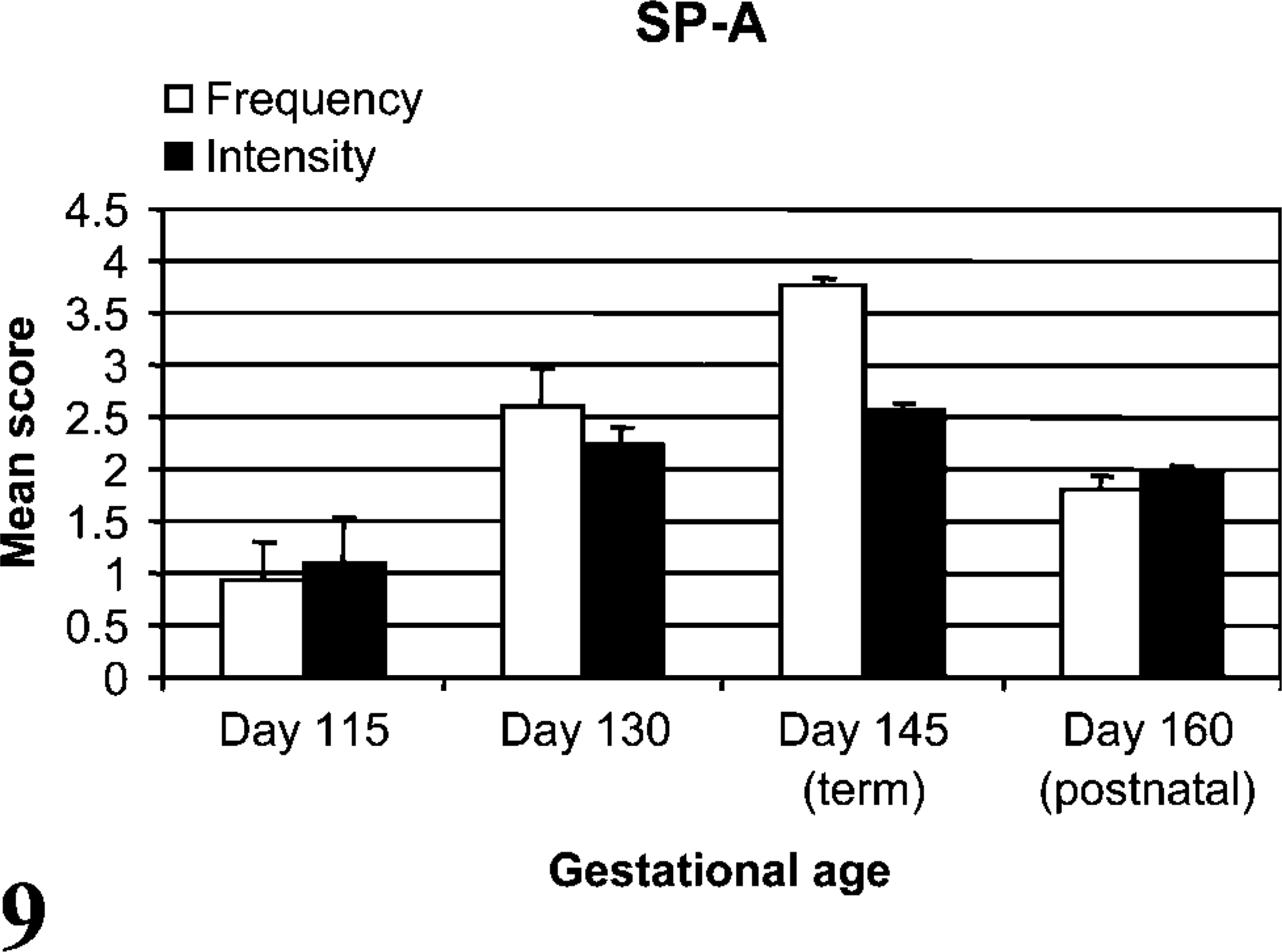
SP-A stain frequency and intensity. Both the mean frequency (p<0.001) and stain intensity (p<0.05) of SP-A increased from day 115 to day 145 with a subsequent decrease in frequency from term to 2 weeks of age (p<0.001). SP-A and CD208 cell IHC staining frequencies were correlated (p<0.01, r = 0.70) during late gestation through birth.
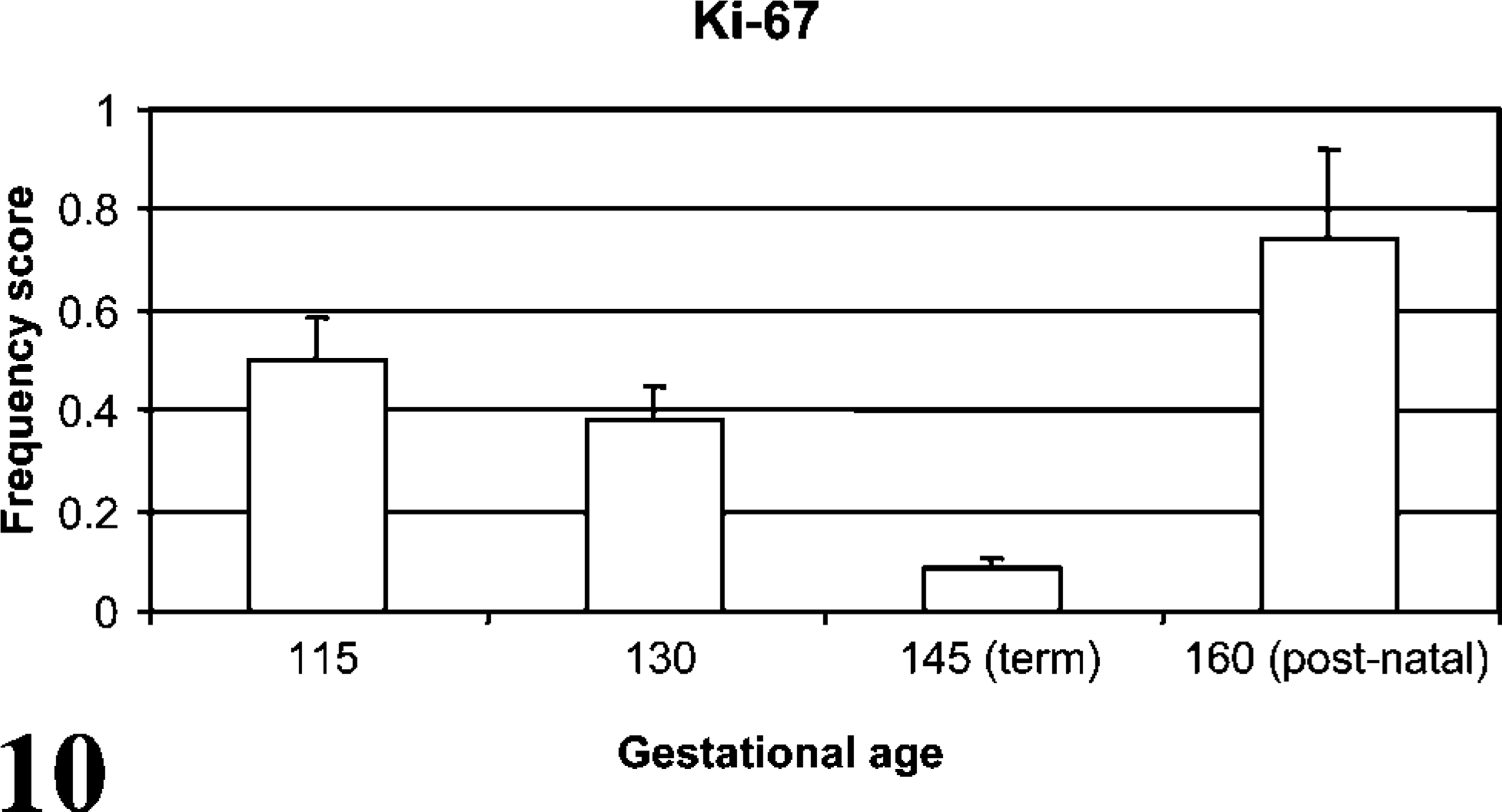
Frequency of Ki-67 staining during development. Ki-67 staining frequency decreased (p<0.05) from day 115 to day 145 with a subsequent increase from term to 2 weeks of age (p<0.01).
Ki-67 is a nuclear protein that is functional at all stages of the cell cycle except resting (G0) phase, and thus is an established IHC marker for proliferation and differentiation (Birner et al. 2001; Glunde et al. 2005). Ki-67 staining is useful in detecting cellular proliferation experimentally, and Ki-67 staining is clinically associated with less differentiated cells and more aggressive phenotypes in neoplasms (Alcorn et al. 1981; Carvalho et al. 2005). In this current study, alveolar cell proliferation decreased during gestation followed by a postnatal increase. Lambs and human infants both enter the alveolar stage of morphological development as early as 80% of gestation, continuing into the postnatal period (Alcorn et al. 1981; Davies et al. 1988; Langston et al. 1984). Previous investigators have found a decrease in pulmonary proliferation during late-term fetal development, attributed to declining type II pneumocyte proliferation and differentiation into type I pneumocytes during alveolar development (Adamson and King 1984; Maniscalco et al. 2002). The increased proliferation in lambs from day 145 to day 160 ga is consistent with previous work in rats, increasing pulmonary Ki-67 staining starting at 4 days of age and correlating with postnatal alveolar development in rodents (Luyet et al. 2002). Ki-67 gestational scoring did not correlate with CD208 during gestation and thus may not be physiologically linked to CD208 expression.
Of special interest in this study was the ontogeny of pulmonary CD208 antigen. Staining intensity and cellular frequency for CD208 was age dependent with a small number of pneumocytes that had weak staining in the young-age group (day 115 ga), which intensified with increased developmental age through day 160 ga of the postnatal period. Cellular staining for CD208 was inversely associated with glycogen staining (p<0.0001). Correlation between glycogen depletion and CD208 staining is consistent with maturation of pneumocytes and development of lamellar bodies from glycogen stores (Ridsdale and Post 2004). PAS stain induces a magenta coloration in glycogen stores of type II pneumocytes. As the type II pneumocyte matures, its glycogen stores are consumed to synthesize the phospholipids that comprise surfactant (Compernolle et al. 2002). The final surfactant product is stored in specialized intracellular inclusions called lamellar bodies that reside diffusely to apically in the cytoplasm of type II pneumocytes (Haagsman and van Golde 1991; Platt et al. 2002). In this study, IHC staining of CD208 and SP-A increased and Ki-67 decreased during gestation, suggesting developmental regulation. Interestingly, although CD208 and SP-A staining increased gestationally into term birth, postnatal detection was divergent with CD208 continuing to increase and SP-A decreasing. This may suggest alternative postnatal regulatory mechanisms for these markers in the sheep.
This study demonstrates that IHC detection of CD208 is developmentally regulated and is detected in cells anatomically and morphologically consistent with type II pneumocytes in the perinatal lamb. CD208 expression is temporally linked to depletion of glycogen stores in the maturing alveolar type II pneumocyte, whereas other markers (SP-A and Ki-67) were developmentally regulated during gestation only. These findings are important for cardiopulmonary, innate immunity, and other studies utilizing lamb lungs. Concerning innate immunity, CD208 lamb cells produce increasing levels of several innate immune genes during gestation including surfactant protein A, D, and sheep beta defensin-1, all of which have potent or potential antimicrobial activity (Meyerholz et al. 2006). Moreover, respiratory syncytial virus (RSV) alters expression of these genes (Kawashima et al. 2006). Increased proliferation and/or maturation of CD208 cells may protect against microbial agents such as RSV, which is as severe in preterm lambs as in preterm human infants.
Footnotes
Acknowledgements
The work was supported by Grants RAI-062787A and 5K08AI-055499-03 from the National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, MD and by Grant NRI/CGP 2003-35204-13492 from the U.S. Department of Agriculture/Agricultural Research Service, Cooperative State Research, Education, and Extension Service, Washington, DC.
