Abstract
ABCA3 is a member of the ATP-binding cassette (ABC) family of transport proteins and is required for perinatal respiratory adaptation. Monoclonal and polyclonal antibodies were generated against a recombinant human ABCA3 peptide and used to assess its expression in the developing lung and adult tissues. Immunostaining for ABCA3 was detected at highest levels in type II epithelial cells of the lung but was also noted in other organs including liver, stomach, kidney, adrenal, pancreas, trachea, and brain. In the fetal lung, ABCA3 staining and mRNA increased prior to birth. Like other surfactant protein genes, ABCA3 expression was induced by thyroid transcription factor-1 in vitro. ABCA3 was coexpressed with SP-B and proSP-C in type II epithelial cells. ABCA3 staining was detected surrounding large, intracellular organelles consistent with its association with lamellar bodies. In the human fetal lung, ABCA3 staining was not detected prior to 22-23 weeks of gestation, except in the presence of pulmonary inflammation. ABCA3 was detected in type II epithelial cells of the human lung from 28 weeks of gestation and thereafter. Postnatally, intense ABCA3 staining was observed in hyperplastic epithelial cells relining injured airways in infants with chronic lung disease. Localization and regulation of ABCA3 in the respiratory epithelium is consistent with its proposed role in surfactant homeostasis. The role of ABCA3 in extrapulmonary tissues and organs remains to be elucidated. This manuscript contains online supplemental material at
Keywords
ABCA3 is a 1704 amino acid member of the ATP-binding cassette (ABC)-containing family of transport proteins associated with the translocation of various substrates across cellular membranes (Higgins 1992; Klugbauer and Hofmann 1996; Connors et al. 1997; Dean et al. 2001). ABCA3 was identified in type II epithelial cells of the lung where it was associated with lamellar body membranes (Zen et al. 1998; Yamano et al. 2001; Mulugeta et al. 2002). Structurally similar members of the ABC family of proteins transport various lipid substrates (Dean et al. 2001). The association of closely related ABC family members with inherited disorders including Tangier disease (ABCA1) (Brooks-Wilson et al. 1999), Stargardt's retinopathy (ABCA4) (Allikmets et al. 1997), ichthyosis (ABCA12) (Lefevre et al. 2003; Akiyama et al. 2005), Dubin-Johnson Syndrome (ABCC3) (Elferink and Groen 2002), and cystic fibrosis (ABCC7) (Riordan et al. 1989), and its expression in type II cells that produce pulmonary surfactant provides support for the concept that ABCA3 may play an important role in lung function. Recently, mutations in the human ABCA3 gene were associated with lethal respiratory distress in newborn infants (Shulenin et al. 2004). Most infants with ABCA3-related lung disease developed respiratory failure in the first hours after birth, generally succumbing from surfactant deficiency and respiratory failure in the first months of life. Electron microscopic evaluation of lung tissue from infants with ABCA3 deficiency demonstrated abnormally small, dense, cytoplasmic inclusions with the absence of normal lamellar bodies and tubular myelin, findings that have been useful for the diagnosis of ABCA3-related disease (Shulenin et al. 2004; Edwards et al. 2005). Abnormalities in lipid content and function were observed in surfactant from patients with ABCA3-related pulmonary disease (Garmany et al. 2006). Inhibition of ABCA3 expression in type II cells impaired lamellar body formation in vitro (Cheong et al. 2006). A number of distinct mutations in the ABCA3 gene have now been associated with severe lung disease in newborns (Shulenin et al. 2004; Bullard et al. 2005). The disorder is inherited as an autosomal recessive gene that generally causes respiratory failure in newborns. Hereditary ABCA3 deficiency presents with clinical features similar to diseases of alveolar homeostasis caused by mutations in SP-B (SFTPB) and, in some cases, SP-C (SFTPC) (Whitsett and Weaver 2002 for review; Nogee 2004a,b,2006; Whitsett et al. 2004,2005; Whitsett 2006).
The pulmonary surfactant system matures in the last trimester of gestation in most mammals, being influenced by developmental, humoral, and environmental factors. Levels of surfactant proteins (A, B, C, and D) and surfactant phospholipids (phosphatidylcholine) increase prior to birth and are useful markers of pulmonary maturation and function in preterm infants (Pryhuber et al. 1991; Khoor et al. 1993,1994; Stahlman et al. 2002). Expression of surfactant proteins is dependent upon a number of transcription factors expressed in respiratory epithelial cells including thyroid transcription factor-1 (TTF-1) and Forkhead ortholog a2 (Foxa2) that influence the expression of groups of genes mediating perinatal lung adaptation including SFTPB (Bohinski et al. 1994; deFelice et al. 2003; Wan et al. 2004).
In the present study we generated polyclonal and monoclonal antibodies (MAbs) to a recombinant peptide derived from the human ABCA3 protein to determine the temporal-spatial distribution of ABCA3 during murine and human lung development and following neonatal lung injury. ABCA3 was detected in close association with lamellar bodies in type II epithelial cells, and its expression increased prior to birth in concert with other components of the surfactant system. In both mouse and humans, ABCA3 mRNA and/or immunostaining was detected in a number of organs, supporting its potential role in non-pulmonary tissues.
Materials and Methods
Procurement and Preparation of Tissues
Human Tissue. This was a retrospective study approved by the Committee for the Protection of Human Subjects, Vanderbilt University Medical Center, Nashville, TN. Fetal tissues were obtained immediately after hysterotomy, hysterectomy, or spontaneous abortion. Infant samples were obtained at surgical biopsy or by postmortem sampling, usually within 2 hr of death. Child and adult samples were obtained postmortem. Tissues were fixed in 10% phosphate-buffered formalin, dehydrated through graded ethanols, and embedded in paraffin. Four-μm-thick serial sections were cut and mounted separately on SuperFrost Plus glass slides (Fisher; Atlanta, GA) for immunohistochemistry (IHC).
Mouse Tissue. FVB/N mice were housed in pathogen-free conditions in accordance with institutional guidelines at Cincinnati Children's Hospital Research Foundation. Timedpregnant dams (E0.5 = 12 hr after fertilization, as determined by detection of a vaginal plug) were killed by a 0.25-ml, IP injection of anesthetic (ketamine, xylazine, or acepromazine). Fetuses were removed from the uterine sac, weighed, and crown-to-rump lengths determined to assess developmental age (Rugh 1993). Non-pregnant adult female and male mice were anesthetized and then exsanguinated by severing the inferior vena cava. The trachea was cannulated, and the lungs were inflation fixed at 25 cm of water pressure with 4% paraformaldehyde formulated in PBS (pH 7.4). Fetal and adult tissues were then immersion fixed overnight in the cold, washed in PBS, dehydrated through a series of alcohols and xylene, and embedded in paraffin. Six-μ-thick sections were cut and loaded onto polysine-coated glass slides (Thermo Electron Corp.; Pittsburgh, PA) for IHC.
Antibody Production
Polyclonal and mouse MAbs to ABCA3 were generated against a recombinant peptide corresponding to the first loop (amino acids 43-233) of the human ABCA3 peptide. Polyclonal antibodies were produced in both rabbit and guinea pig. The sequences contained in this region of the protein differ significantly from that of other members of the ABCA subfamily. Recombinant ABCA3 peptide was produced in E. coli and purified from bacterial lysates by Ni-NTA chromatography before injection. MAbs were produced and screened essentially as previously described (Holzinger et al. 1996). Likewise, guinea pig (GP985) and rabbit antisera (R28, R29) recognized the 24-kDa recombinant ABCA3 peptide by ELISA, by Western blot against the immunizing antigen, and by IHC. Seventeen MAbs were isolated, and MAbs 12 and 13 were chosen for further study on the basis of IHC, Western blotting, and ELISA. MAb 12 (IgG) and MAb 13 (IgA) were further studied in both mouse and human lung. Immunostaining of both human and mouse lung was completely blocked by preincubation of the antisera with the recombinant antigen. Extracts were prepared from adult mouse kidney and mouse type II epithelial cells that were isolated as previously described (Jacobs et al. 1982; Rice et al. 2002). Proteins were separated by SDS-PAGE under non-reducing conditions followed by transfer to nitrocellulose membranes and then subjected to Western blotting.
IHC
For human tissues, the sections were deparaffinized in three changes of xylene (10 min each) and then rehydrated through 100%, 95%, and 80% ethanol. The sections were transferred into TBS plus 0.1% Tween 20. Immunostaining was performed using a very sensitive, biotin-free, peroxidase-conjugated polymer attached to the secondary antibody (anti-rabbit, Envision + HRP/DAB Kit; Dako, Carpinteria, CA) according to the manufacturer's directions. Rabbit ABCA3 antibodies were applied to the sections at a dilution of 1:1000 for 1 hr, and guinea pig antibody was used at a dilution of 1:2500. Slices were washed three times in TBS with 0.1% Tween 20, and then the labeled polymer complex was applied to the sections for 30 min. After washing in TBS plus 0.1% Tween 20, the peroxidase activity was localized by reaction with 3,3′-diaminobenzidine (DAB) in 0.01% H2O2 and counterstained with hematoxylin. Substitution of preimmune sera at a dilution of 1:1000 was used as a control for specificity of the sera. MAbs 12 and 13 were utilized at 1:500 dilution of the ascites fluid and developed with isotype specific secondary antibodies.
For mouse tissues, the sections were deparaffinized through three changes of xylene (10 min each) and rehydrated in a descending series of ethanol solutions (100%, 95%, 70%). Endogenous peroxidase was quenched for 15 min with 3% H2O2 in methanol and then blocked for 2 hr at room temperature in 4% normal goat serum in PBS with 0.2% Triton. Both rabbit and guinea pig ABCA3 antibodies were then applied to the sections at a dilution of 1:500 and incubated overnight in the cold. The slides were washed five times (5 min each) in PBS with 0.2% Triton X-100 and then incubated for 30 min at room temperature with biotinylated goat anti-rabbit IgG diluted 1:200 in the blocking solution (Vector Laboratories; Burlingame, CA). After washing, the sections were incubated for 30 min at room temperature in peroxidase-conjugated avidin-biotin-complex diluted in 0.1 M PBS with 0.2% Triton X-100 (Vectastain Elite ABC kit; Vector Laboratories). The enzymatic reaction product was detected by using nickel-DAB as a substrate in 0.1 M acetate buffer with 0.01% H2O2. The precipitation reaction was enhanced with Tris-cobalt, and the sections were counterstained with 0.1% of nuclear fast red in 5% aluminum sulfate. Omission of the primary antibody and substitution of both preimmune and non-immune sera at a dilution of 1:500 for the primary antibody were used as controls to check for endogenous biotin and peroxidase activity, as well as for nonspecific binding of the secondary antibody. Preincubation of the working antibody solution (1:500) with the antigenic peptide (5, 10, 20, 50, 100, and 200 μg/ml) blocked specific staining for ABCA3 in murine tissues. MAbs 12 and 13 were utilized at a dilution of 1:500 for IHC. A mouse-on-mouse blocking kit (Vector Laboratories) was used with the mouse MAbs.
Colocalization of ABCA3, SP-B, and ProSP-C
Dual immunofluorescence labeling was performed using monospecific, polyclonal guinea pig antibody generated to the human ABCA3 peptide as described above for the rabbit antisera. Rabbit polyclonal antibodies to SP-B and proSP-C were generated as previously described and characterized (Khoor et al. 1993,1994; Vorbroker et al. 1995a,b; Zhou et al. 1996). Staining was assessed by using Alexa Fluor 488-conjugated anti-guinea pig IgG (green for ABCA3) and Alexa Fluor 594-conjugated anti-rabbit IgG (red for either SP-B or proSP-C) (Molecular Probes; Eugene, OR). Dual immunostaining was performed on sections of fixed, paraffin-embedded adult mouse and human lung. Briefly, the sections were deparaffinized and rehydrated as described above and then blocked for 2 hr at room temperature in 4% normal donkey serum in PBS with 0.2% Triton X-100. A cocktail containing the guinea pig anti-ABCA3 antibody at a dilution of 1:200 and either rabbit anti-SP-B or rabbit anti-proSP-C antisera diluted 1:200 was then applied to the sections and incubated overnight in the cold. Sections were washed the next day in PBS with 0.2% Triton X-100 and then incubated sequentially for 1 hr each with the appropriate Alexa Fluorlabeled secondary antibody diluted 1:200 in the blocking solution. After rinsing in PBS for 5 min and then in phosphate buffer (0.1 M PB 2X for 5 min each and 0.05 PB 2X for 5 min each), the sections were coverslipped with Vectashield mounting medium (Vector Laboratories). Omission of the primary antibody and both the primary and secondary antibodies were used as controls to check for autofluorescence as well as for nonspecific binding of the secondary antibodies. Fluorescent labeling was visualized and photographed using a Leica TCS NT confocal laser-scanning microscope (Leica Microsystems; Mannheim, Germany) equipped with a Leica DM IRBE inverted microscope, an Ar-laser (488 nm), a Kr-laser (568 nm), and a HeNe-laser (633 nM).
ABCA3 mRNA Analysis
RNA was isolated from MLE-13, MLE-15, HeLa cells, mouse alveolar macrophages, purified mouse type II cells (Rice et al. 2002), mouse lung, and other organs using Trizol (Invitrogen; Carlsbad, CA). RNA was treated with DNase at room temperature for 15 min before cDNA synthesis. RNA (4 μg) was reverse transcribed and then analyzed by PCR for ABCA3 and β-actin mRNA. Mouse ABCA3 and β-actin were identified with the following primers: ABCA3 forward (5′-GCA TTG CCC TCA TTG GAG AGC CTG-3′) and reverse (5′-TCC GGC CAT CCT CAG TGG TGG G-3′), β-actin forward (5′-TGG AAT CCT GTG GCA TCC ATG AAC-3′) and reverse (5′-TAA AAC GCA GCT CAG TAA CAG TCC G-3′). PCR parameters used were as follows: for ABCA3, 95C for 5 min, followed by 22 cycles at 95C for 30 sec, 62C for 30 sec, and 72C for 40 sec, followed by a 7-min extension at 72C; for β-actin, 95C for 5 min, followed by 20 cycles at 95C for 30 sec, 58C for 30 sec, and 72C for 50 sec, followed by a 7-min extension at 72C. Samples lacking reverse transcriptase and β-actin mRNAs were assessed as controls. RT-PCR products were separated by electrophoresis and stained for image analysis.
Transcription Assays
A 2.6-kb fragment of the 5′ region of the mouse Abca3 gene was isolated by PCR, sequenced, and ligated into pGL3. The Abca3 reporter gene was cotransfected with pRc-CMV-TTF-1, a plasmid expressing rat TTF-1, into immortalized mouse lung epithelial cells (MLE-15) (Wikenheiser et al. 1993) and HeLa cells as previously described (Bohinski et al. 1994). Forty eight hr after transfection, luciferase was assessed using a luminometer. Transfection efficiency was assessed by cotransfection with pCMV-β-galactosidase.
Expression Data
The expression distribution of human ABCA3 mRNA and cDNA in different tissues and cell lines was obtained via interrogation of the Genomics Institute of Novartis Research Foundation gene expression database (available at
Results
ABCA3 Expression in Mouse Tissues and Cells
Abca3 mRNA was detected by RT-PCR in mouse lung, liver, kidney, cerebellum, and gastrointestinal tract (Figure 1A). Abca3 mRNA was also detected in SV40 large T-immortalized mouse lung epithelial cell lines (both MLE-13 and MLE-15 cells), alveolar macrophages, and purified type II epithelial cells isolated from the adult mouse lung (data not shown). In the fetal mouse lung, Abca3 mRNA was present at relatively low levels at E14, its abundance increasing prior to birth (Figures 1B and 1C).
Reactivity of ABCA3 Antibodies in Mouse Tissues
Rabbit, guinea pig antisera, and mouse MAbs recognized the immunogen purified from the bacterial lysates expressing the human ABCA3 peptide (amino acids 43-233) by ELISA and Western blot (Figures 2A and 2C). MAbs 12 and 13 detected a single band with a molecular mass = 191,000 in mouse lamellar body fractions (Figure 2B) and were highly active in ELISA and Western blot against the recombinant ABCA3 immunogen (Figure 2A). ABCA3 was also detected by Western blot of adult mouse kidney homogenates, migrating at molecular mass = 191,000. In fetal mouse lung, ABCA3 staining was not detected at E15.5 or E16.5. ABCA3 staining was first detected at E17.5 in the early saccular stage of lung development in subsets of epithelial cells in the peripheral lung (Figure 3), consistent with the ontogenic changes in ABCA3 mRNA. ABCA3 staining was observed primarily in alveolar type II epithelial cells in the adult mouse lung (Figure 3), where identical patterns of staining were observed with rabbit, guinea pig, and mouse MAbs (Figures 3E and 3F), the cellular sites of staining being consistent with that of proSP-C, a type II cell-specific marker. Mouse alveolar macrophages did not stain with any of the ABCA3 antibodies, in spite of detectable Abca3 mRNA by RT-PCR. ABCA3 staining was also detected in Purkinje cells of the mouse cerebellum with rabbit (Supplemental Figure 1) and guinea pig antisera (not shown). Detection of ABCA3 in mouse brain with GP985 required the use of antigen retrieval. In spite of the presence of Abca3 mRNA in various tissues, ABCA3 staining was not detected in adult mouse liver, pancreas, salivary glands, esophagus, stomach, intestine, colon, kidney, adrenal, spleen, heart, thymus, thyroid, trachea, spinal cord, cerebral cortex, medulla, brain stem, or in female or male reproductive organs including ovary, oviduct, uterus, prostate, vesicular gland, coagulating gland, epididymis, or testes. Staining was completely blocked in fetal and adult mouse lung and the cerebellum by preincubation of the antiserum with increasing concentrations (5-200 μg/ml) of recombinant ABCA3 peptide (Supplemental Figure 1). MAbs 12 and 13 strongly stained type II epithelial cells in the mouse lung but did not stain other mouse tissues. Together all ABCA3 antibodies were strongly reactive with type II cells in the lung. IHC staining in tissues other than the lung was variable, most likely dependent upon levels of expression, accessibility of the ABCA3 antigenic epitopes in various tissues, and the affinity of each antibody for the protein.
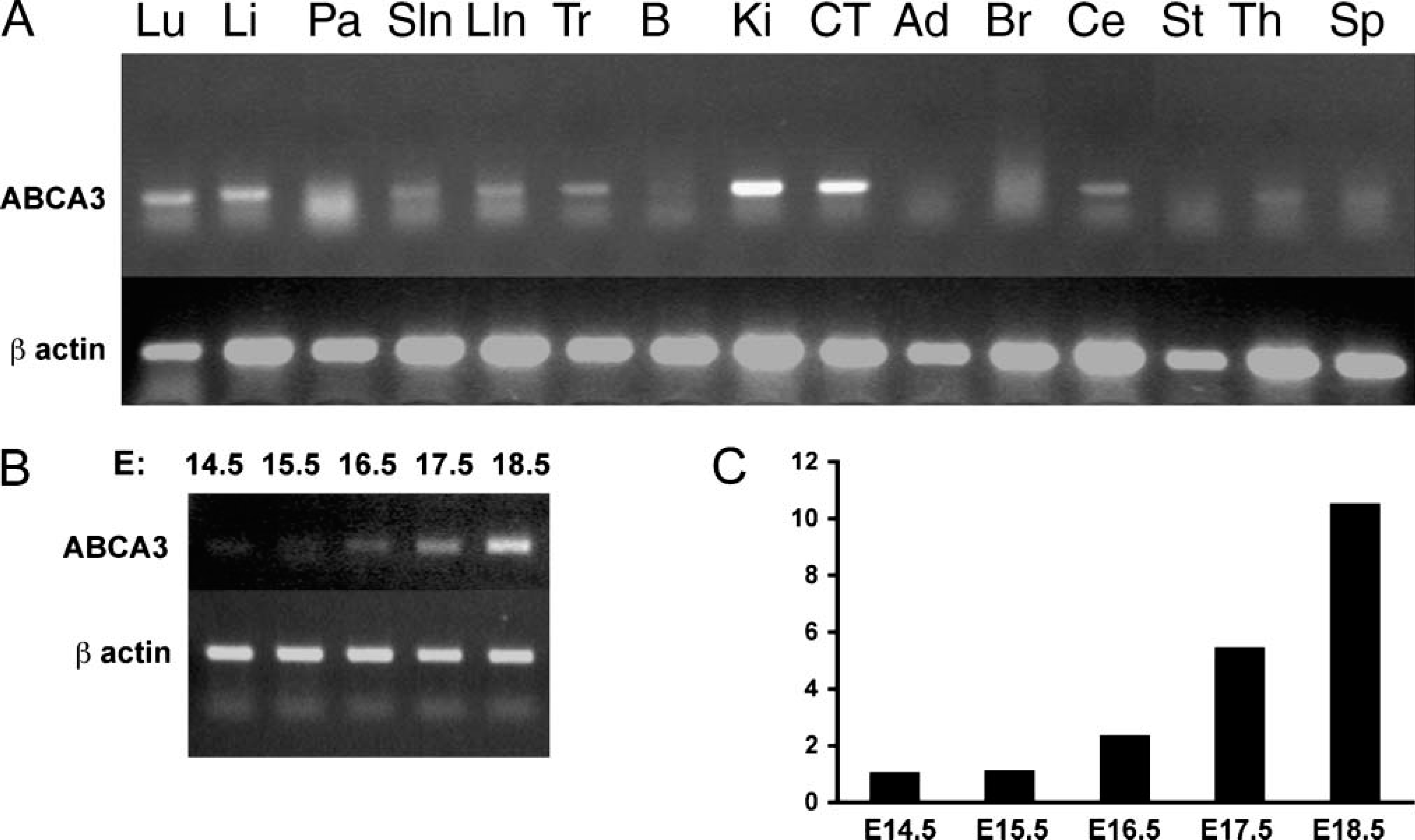
Distribution of Abca3 mRNA in mouse tissue. Abca3 mRNA was assessed by RT-PCR of mRNA isolated from adult mouse organs (
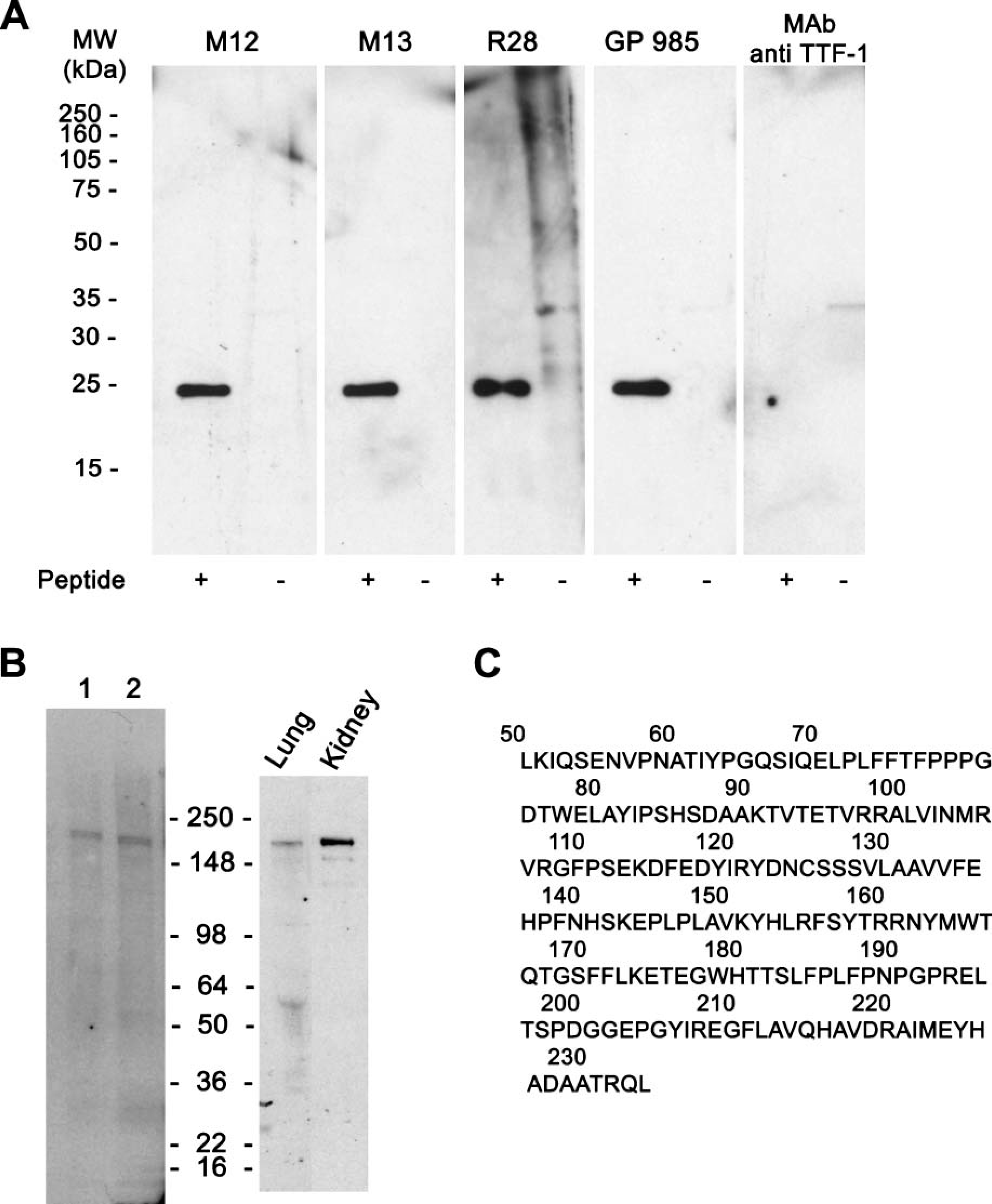
Western blot analysis with ABCA3 antibodies and antisera. Proteins were separated by SDS-PAGE under non-reducing conditions. (
ABCA3 expression increasing during mouse lung development. ABCA3 was not detected at E16.5 (
Distinct Intracellular Distribution of ABCA3, ProSP-C, and SP-B
Dual immunofluorescence microscopy was performed on type II epithelial cells from postnatal human (not shown) and mouse lung (Figure 4). Whereas all three proteins were present in alveolar type II cells, ABCA3 was detected as cytoplasmic staining outlining large and small intracellular organelles, consistent with its localization near lamellar bodies as well as other intracellular organelles. In contrast, SP-B staining was detected as large, dense, cytoplasmic inclusion bodies, which did not directly colocalize with ABCA3 but appeared to be surrounded or outlined by ABCA3 staining. Likewise, proSP-C staining did not colocalize with ABCA3, being detected in perinuclear inclusions within type II cells, consistent with localization in the endoplasmic reticulum and Golgi apparatus, intracellular sites of synthesis, and proteolytic processing of the peptide prior to and during transport to the lamellar body. Together, ABCA3 staining was most intense in membranes surrounding lamellar bodies within type II cells, being detected primarily in a compartment distinct from proSP-C or SP-B. Consistent with the findings in murine type II cells, dual immunostaining for SP-B and ABCA3 demonstrated staining of ABCA3 surrounding, but not within, lamellar bodies in human adult type II cells, whereas SP-B staining was localized to the interior of the lamellar body (not shown). The cellular sites of ABCA3 expression in the lung are also consistent with that of surfactant protein C, a specific marker of type II epithelial cells (Wert et al. 1993; Khoor et al. 1994; Zhou et al. 1996).
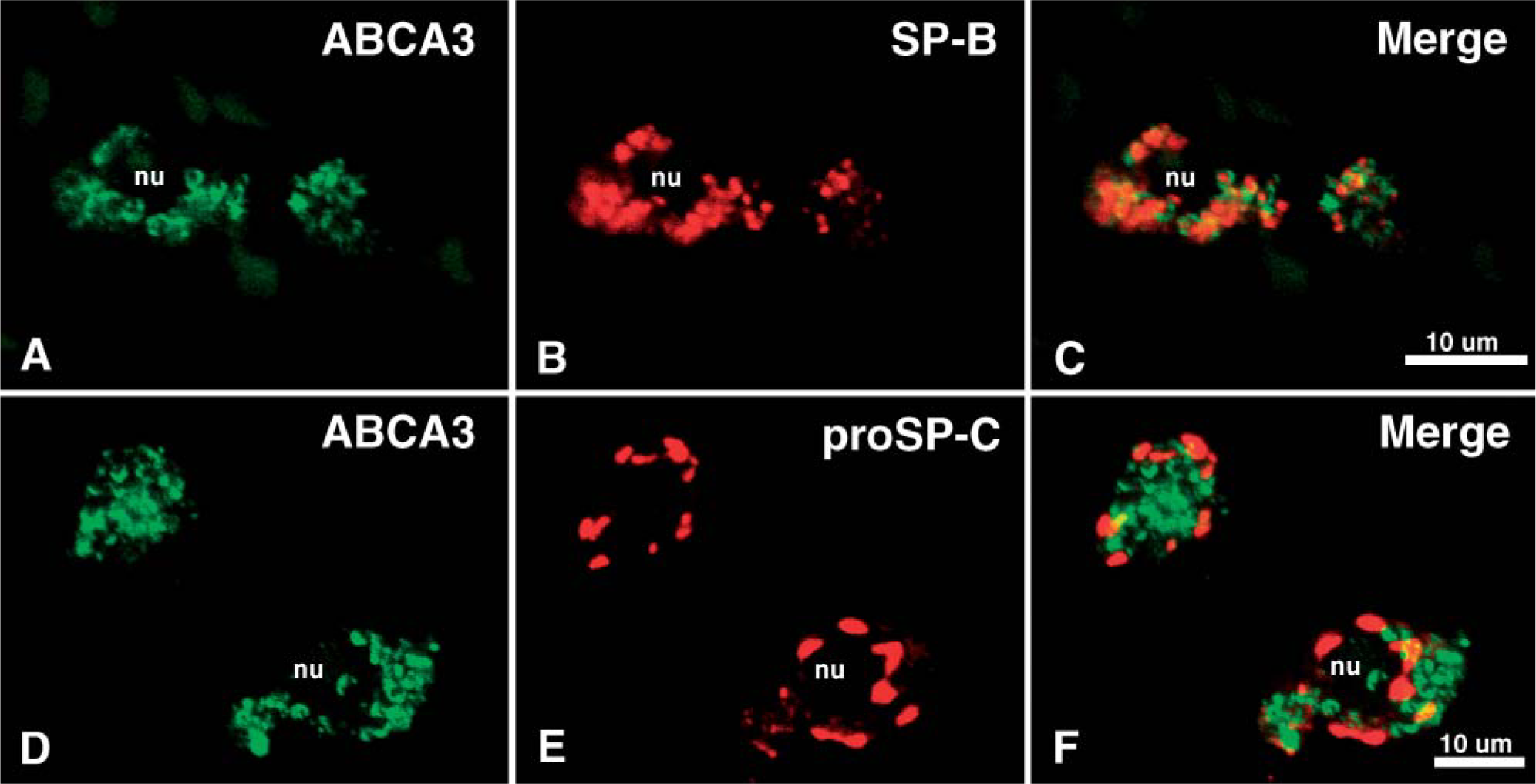
Dual immunolocalization of ABCA3 and SP-B or proSP-C in adult mouse type II cells. Immunofluorescent staining for ABCA3 (green in
TTF-1 Activates ABCA3 Gene Expression In Vitro
TTF-1 is a homeobox-containing transcription factor that is a member of the Nkx family of nuclear proteins. TTF-1 regulates and is required for the expression of a number of genes whose expression in the respiratory epithelium increases prior to birth, including the surfactant proteins SP-A, SP-B, SP-C, and the Clara cell secretory protein (Bohinski et al. 1994). To assess whether ABCA3 was regulated in a manner similar to the surfactant proteins, the effect of TTF-1 on murine Abca3 gene promoter activity was assessed in vitro. TTF-1 enhanced expression of a reporter construct, consisting of 2.6 kb from the start of transcription of the Abca3 gene, in MLE-15 cells (Figure 5A) and in HeLa cells (data not shown). Consistent with this observation, a number of potential TTF-1 binding sites were identified in the promoter region of the mouse Abca3 gene (Figure 5B).
Ontogenic Changes in ABCA3 Staining in the Human Lung
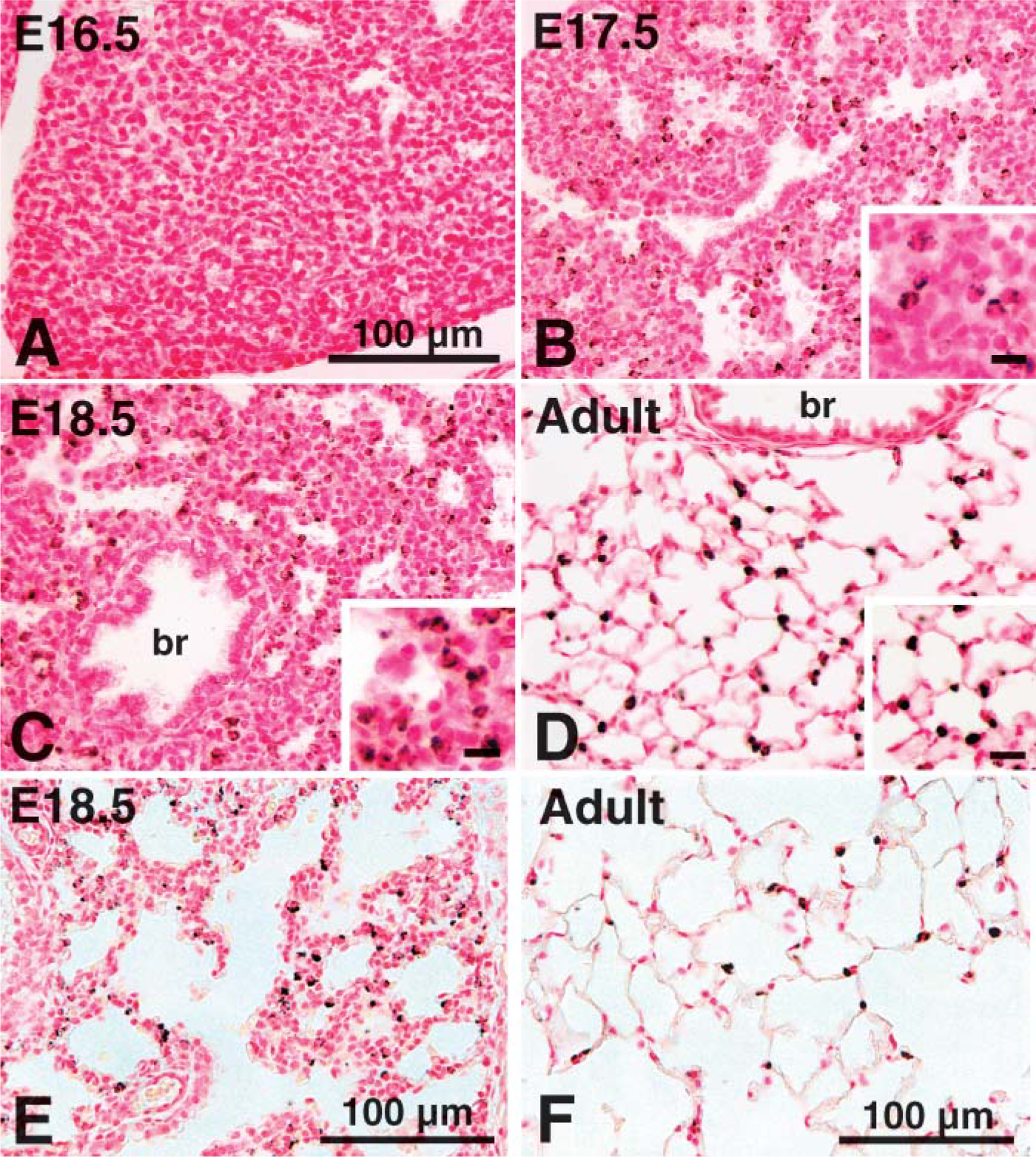
ABCA3 staining was assessed in normal human lung from 12 to 24 weeks gestation and thereafter (Figure 6) using the rabbit polyclonal antiserum. Similar findings were observed with both MAbs 12 and 13 and guinea pig antiserum. ABCA3 was generally absent in human fetal lung from 14 to 23 weeks (Figure 6B). ABCA3 staining was detected in the cytoplasm of epithelial cells lining the peripheral lung tubules in epithelial cells from several samples of lung tissue from 23 to 24 weeks gestation with evidence of inflammation, as indicated by alveolar neutrophilic and monocytic infiltrates (Figures 6E and 6F). ABCA3 was not detected in the inflammatory cells. These findings suggest that pulmonary infection caused precocious ABCA3 expression in respiratory epithelial cells in the human lung. ABCA3 staining was readily detected in normal lung from 26 to 27 weeks gestation and thereafter (Table 1; Figure 6A). ABCA3 staining was detected surrounding large and small cytoplasmic organelles, consistent with its close association with lamellar bodies or other organelles. Thus, ABCA3 staining was detected in type II epithelial cells and was not detected in non-epithelial cells in the human lung. Staining in alveolar macrophages was observed with both rabbit and guinea pig antibodies but was variable from sample to sample.
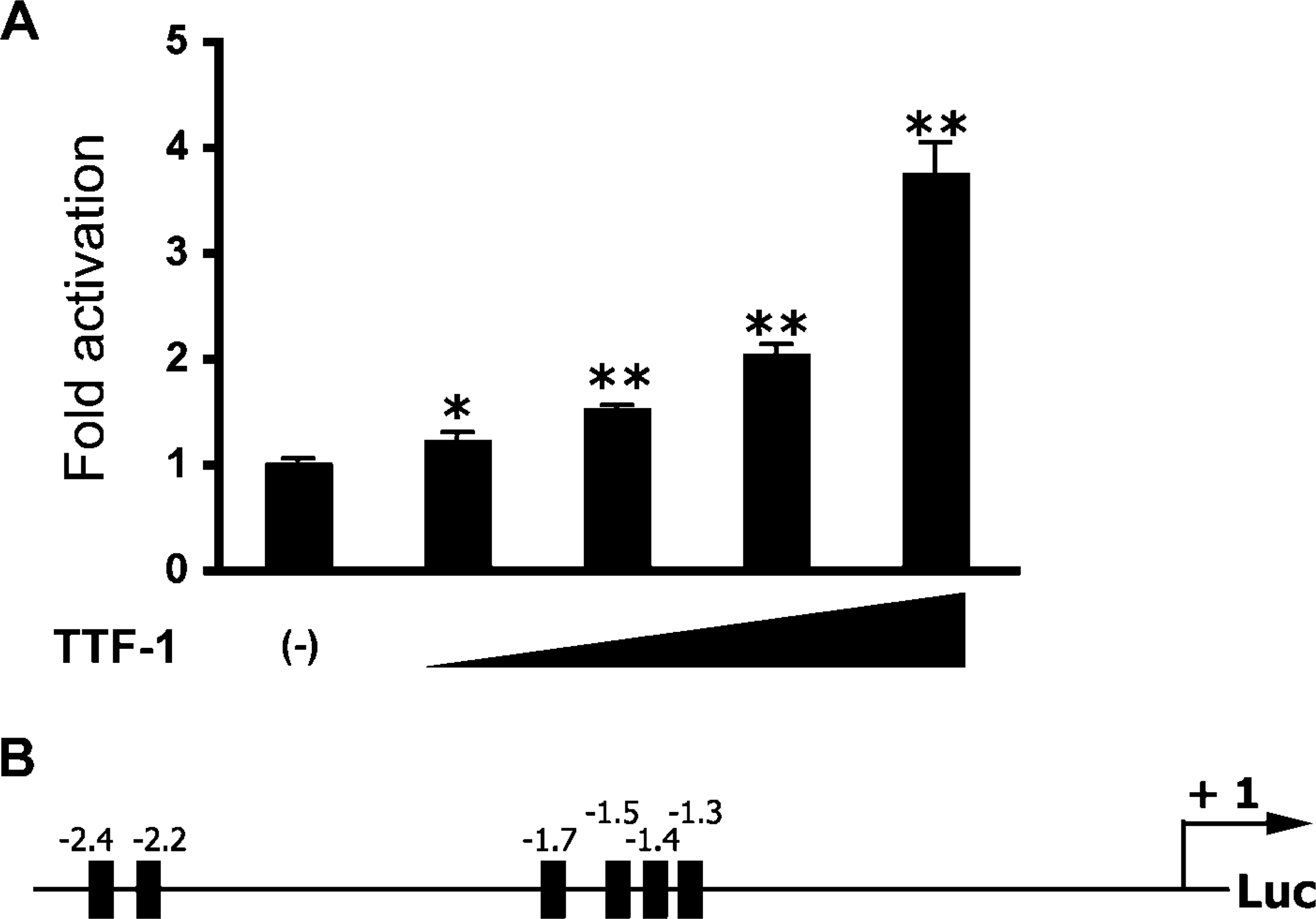
TTF-1 activates the mouse Abca3 gene promoter in vitro. MLE-15 cells were cotransfected with increasing amounts (0-2 μg) of a plasmid expressing rat TTF-1 and a luciferase construct consisting of 2.6 kb of the 5′ region of the mouse Abca3 gene (
ABCA3 Staining After Lung Injury
ABCA3 was generally expressed at high levels in the injured lung and regenerating epithelial tubules in infants with bronchopulmonary dysplasia (BPD) (Figures 6C and 6D). In most of these infants, peripheral lung tubules were lined by hyperplastic/dysplastic epithelial cells containing numerous, large intracellular organelles surrounded by ABCA3 staining. Previous studies demonstrated that these cells express proSP-C, a specific marker of type II cells (Khoor et al. 1994). Thus, ABCA3 is a useful marker for hyperplastic epithelial cells lining the lungs of infants with BPD.
Detection of ABCA3 in Non-pulmonary Human Tissues
ABCA3 staining with the rabbit antibody (R28) was detected in subsets of epithelial cells in the renal tubules, adrenal cortex, parietal cells of the stomach, hepatocytes, pancreatic islet cells, lachrymal glands, and submaxillary glands in adult human tissue. In fetal tissue, ABCA3 was detected in meninges, Purkinje cells of the cerebellum, and trachea, wherein ABCA3 was noted with the cell cytoplasm outlining intracellular organelles (Figure 7). ABCA3 staining in both lung and other tissues was also completely blocked by preincubation with ABCA3 peptides (Supplemental Figure 2). Consistent with the staining of ABCA3 in non-pulmonary tissues, a search of expression databases derived from human mRNA microarray and cDNA analyses (Genome Institute of Novartis Research Foundation, Gene Expression Omnibus Database, GeneCards encyclopedia, and UniGene's EST Profile Viewer) revealed detectable ABCA3 expression in kidney, brain, intestine, thyroid, and other tissues, albeit at lower levels than those present in lung, indicating that ABCA3 may play roles in other tissues. A similar pattern of ABCA3 staining in the neonatal brain was also observed with GP985.
Discussion
In this study, ABCA3 protein was detected in alveolar type II cells of the lung, at cellular sites coexpressing surfactant proteins SP-B and proSP-C. TTF-1, a transcription factor expressed in alveolar type II cells (Stahlman et al. 1996; Zhou et al. 1996), enhanced the expression of the Abca3 gene promoter in vitro, consistent with previous studies demonstrating its critical role in the regulation of surfactant protein genes (Bohinski et al. 1994). ABCA3 expression increased in association with morphological maturation of the mouse and human lung. Increase in ABCA3 expression correlated with appearance of lamellar bodies (Hilfer 1983; Stahlman et al. 1992) and with the expression of surfactant proteins including SP-A, SP-B, and SP-C in human and mouse lung (Khoor et al. 1994; Stahlman et al. 1996; Zhou et al. 1996). At the subcellular level, ABCA3 was associated with large intracellular organelles in type II cells, consistent with its association with lamellar bodies and other cytoplasmic organelles (Zen et al. 1998; Yamano et al. 2001; Mulugeta et al. 2002). Abca3 mRNA or staining was also detected in a number of non-pulmonary tissues, being most abundantly expressed in kidney and cerebellum.
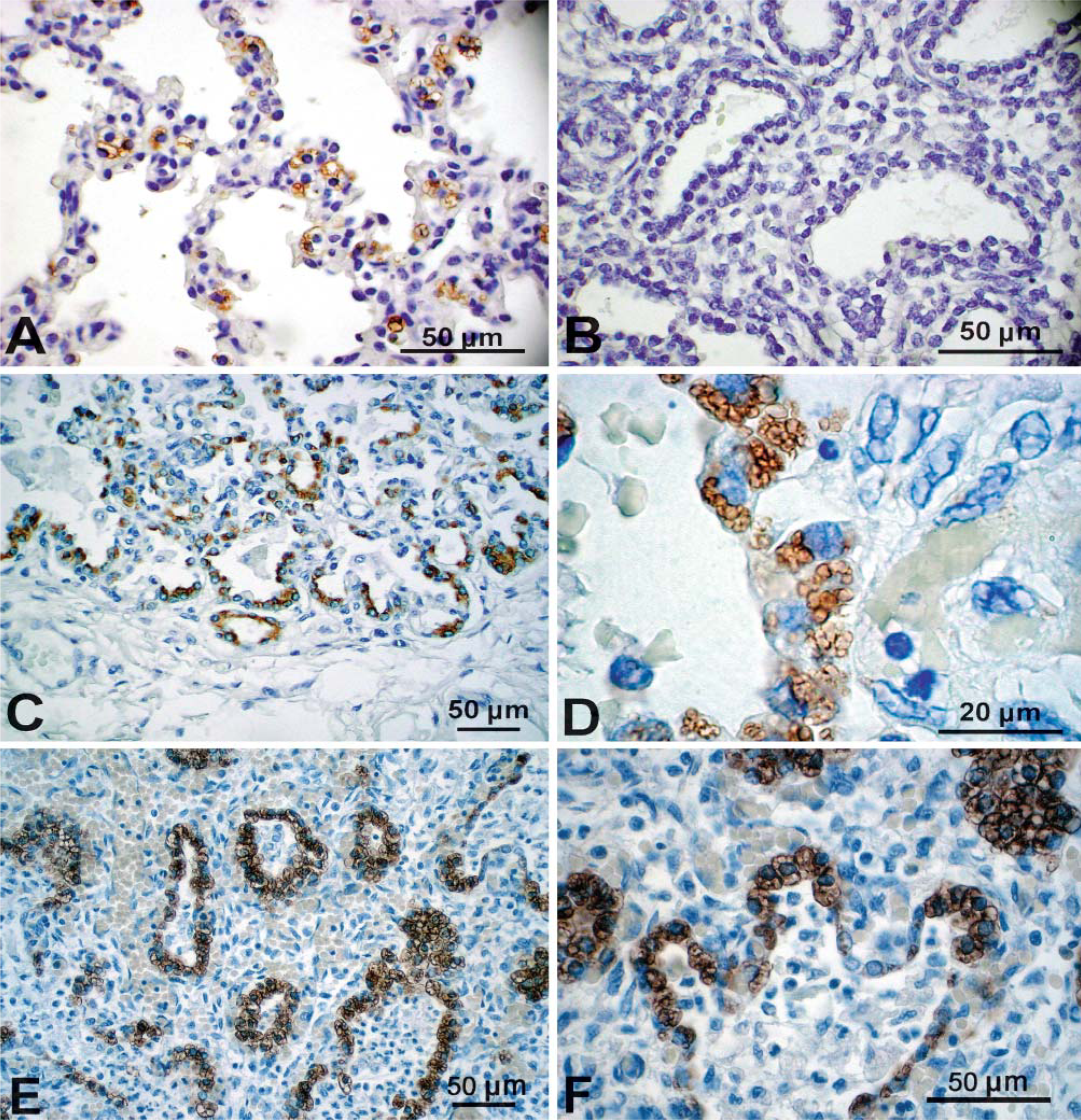
Immunostaining of ABCA3 in human fetal lung tissue. ABCA3 was assessed with anti-rabbit ABCA3 in human lung at 37 weeks gestation (
ABCA3 antisera and MAbs generated for the present study were generated against a 24-kDa fragment of the protein, a site consistent with the first non-membrane-associated loop in the unpredicted structure. ABCA3 protein with a molecular mass of ∼191,000 was detected in non-reduced SDS-PAGE gels of mouse type II cell lysates and kidney homogenates. Both rabbit anti-serum and mouse MAbs stained the ABCA3 protein (molecular mass = 191,000), consistent with the predicted size of the ABCA3 monomer. The antiserum was highly selective, its activity being entirely blocked by preincubation with the recombinant antigenic peptide. This peptide was chosen for its conservation across mouse and human species as well as for its lack of homology with other ABCA family members. ABCA3 staining was most intense in pulmonary tissues in both human and mouse lung but was also detected in other tissues and cells. With the rabbit polyclonal antibody, immunostaining was detected primarily in epithelial cells of various human tissues including kidney, pancreas, stomach, adrenal cortex, lachrymal gland, liver, and brain. ABCA3 was expressed most abundantly in type II cells in the developing and mature lung. GP985 and MAbs 12 and 13 were highly selective and reactive with type II epithelial cells in the lung but were less reactive or non-reactive in other tissues. These differences in staining may be related to differences in affinity, sensitivity, and accessibility of the antigen in various tissues or to differences in protein levels in each tissue. The functional role of ABCA3 in other tissues is unknown at present. The present analysis of ABCA3 mRNA in human and mouse cDNA and mRNA databases and previous studies (Klugbauer and Hofmann 1996; Connors et al. 1997; Mulugeta et al. 2002) was consistent with its expression in mouse tissue and that reported in lung and its presence in many non-pulmonary tissues. Whereas immunostaining of ABCA3 was most intense in type II cells, Abca3 mRNA was readily detected by RT-PCR in various organs in the mouse.
Summary of human lung tissue samples a
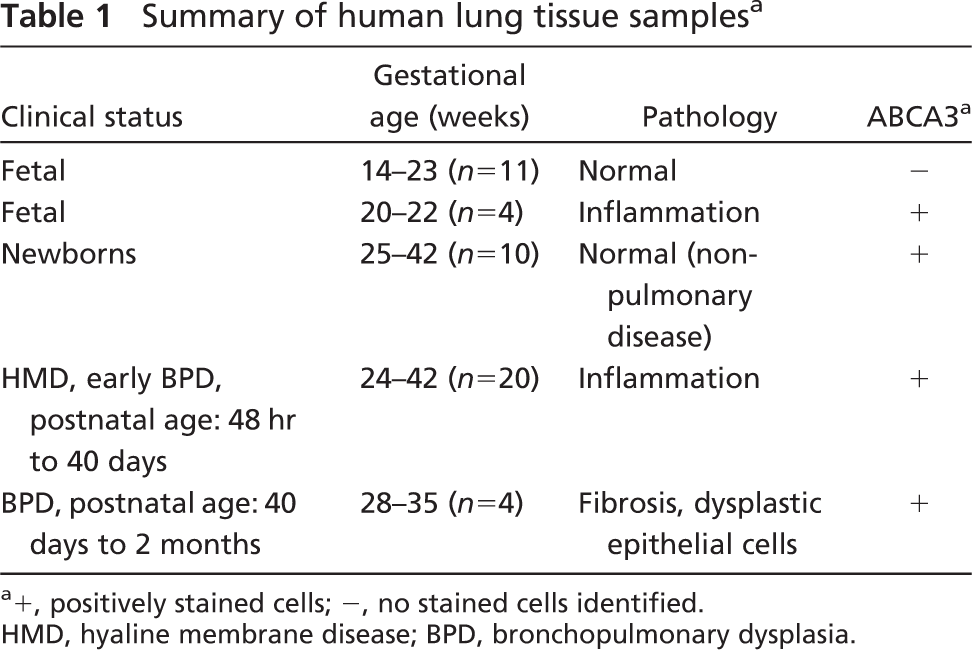
+, positively stained cells; -, no stained cells identified. HMD, hyaline membrane disease; BPD, bronchopulmonary dysplasia.
The temporal-spatial distribution of ABCA3 staining and its subcellular distribution in the lung are consistent with previous findings that support an important role for ABCA3 in formation of lamellar bodies and in surfactant homeostasis. ABCA3 was expressed at increasing levels in association with the increased expression of surfactant proteins and lipids and the formation of lamellar bodies that occurs during perinatal maturation of the lung, consistent with previous studies (Mulugeta et al. 2002; Yoshida et al. 2004). In the present study, TTF-1 enhanced the expression of Abca3 in vitro, likely mediated by its binding to regulatory elements in the Abca3 gene promoter. TTF-1 is a nuclear transcription factor that is expressed in subsets of respiratory epithelial cells within the lung and is known to directly bind and activate transcription of surfactant protein genes (Bohinski et al. 1994). TTF-1 also influences the expression of groups of genes regulating surfactant homeostasis, innate host defense, and fluid and electrolyte transport in the lung, indicating its important role in lung maturation (deFelice et al. 2003). Previous studies of the ABCA3 gene indicated its induction by glucocorticoids (Yoshida et al. 2004), which are also known to play an important role in the maturation of the lung and in the enhancement of surfactant synthesis prior to birth. In the human lung, ABCA3 expression increased in association with increased surfactant protein expression and type II cell maturation, as indicated by the formation of lamellar bodies (Stahlman et al. 1992). Mutations in the human ABCA3 gene cause fatal respiratory distress in the newborn period associated with surfactant dysfunction; surfactant lipid abnormalities; and the formation of small, dense lamellar bodies (Shulenin et al. 2004; Edwards et al. 2005; Garmany et al. 2006). Recent studies demonstrating that inhibition of ABCA3 in type II cells disturbs lamellar body synthesis and lipid transport provide further support for the importance of ABCA3 in surfactant homeostasis (Cheong et al. 2006). Together these findings support a critical role for ABCA3 in surfactant synthesis and/or packaging.
ABCA3 staining was noted surrounding lamellar bodies in type II cells but was primarily localized in intracellular membranous compartments distinct from those of the active SP-B peptide and proSP-C. Direct colocalization of SP-B, proSP-C, and ABCA3 was not observed under these imaging conditions. Although proSP-B and proSP-C are cotrafficked through the endoplasmic reticulum, Golgi apparatus, and multivesicular bodies, both are proteolytically processed prior to their insertion into the lamellar bodies. The active SP-B and SP-C peptides found in secreted surfactant are colocalized with lipids within lamellar bodies (Voorhout et al. 1992,1993; see Weaver 1998 and Weaver et al. 2002 for review). ProSP-C marks the ER/Golgi/multivesicular bodies in type II epithelial cells. Because the active SP-C peptide is poorly antigenic, its IHC localization has not been possible. The distinct localization of ABCA3 suggests that it resides in discrete subcellular sites, distinct from those of the surfactant proteins. It remains possible that ABCA3, proSP-B, and SP-C are co-routed earlier in the synthetic pathway prior to their insertion into the lamellar bodies, at levels below the levels of sensitivity of the fluorescence assays used in the present study.
ABCA3 was expressed at high levels in the respiratory epithelial cells of infants with BPD where it outlined abundant cytoplasmic organelles. The presence of ABCA3 staining in cuboidal cells lining injured airways suggests that ABCA3 deficiency does not play a direct role in the pathogenesis of BPD. Intense staining of ABCA3 surrounding the large, abnormal inclusions, however, may indicate abnormalities in surfactant synthesis, packaging, or secretion that might play a role in the pathogenesis of BPD. Whether increased expression or activity of ABCA3 contributes to the cellular abnormalities typical of BPD or other forms of lung injury remains to be assessed.
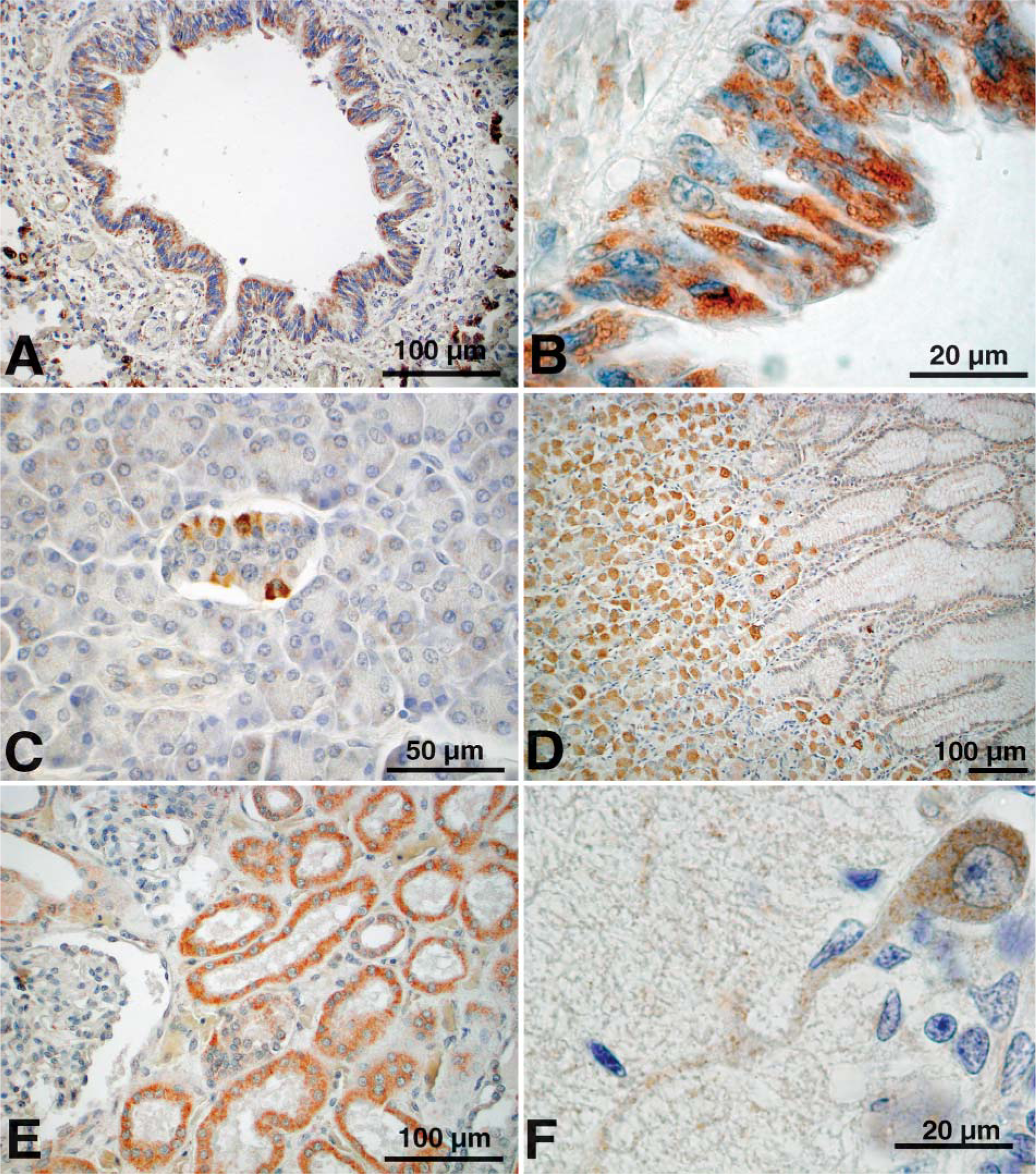
Immunostaining of ABCA3 in human non-pulmonary tissues. ABCA3 was detected with rabbit anti-ABCA3 in bronchi (
ABCA3 immunostaining was detected in a number of non-pulmonary human tissues from adults, children, and fetuses including liver, pancreas, stomach, trachea, lachrymal duct, adrenal cortex, cerebellum, and proximal tubules of the kidney. In mouse and human brain, ABCA3 was selectively expressed in Purkinje cells in the cerebellum. ABCA3 staining generally was associated with epithelial cells in those tissues, whereas underlying stromal, vascular, and tissues of hematopoietic origin were generally unstained. Consistent with the presence of ABCA3 in non-pulmonary tissues, ABCA3 cDNAs or mRNAs were detected in various mouse tissues and in gene expression databases, confirming the presence of ABCA3 mRNAs in non-pulmonary tissues. Although patients bearing mutations in their ABCA3 genes develop disease that is confined to the lung (Shulenin et al. 2004), the severity of lung disease and poor survival of such patients does not exclude the possibility that ABCA3 plays a role in other tissues, which has not been appreciated clinically.
In summary, ABCA3 was expressed at high levels in type II cells in the developing and mature lung. Pulmonary ABCA3 expression increased prior to birth, consistent with its proposed role in the formation of lamellar bodies critical for surfactant homeostasis. ABCA3 was also detected in a variety of non-pulmonary tissues in mouse and human where its function remains unknown. Polyclonal and monoclonal antibodies produced for this study are likely to be useful for the study of ABCA3 in the lung and other tissues. Likewise, variations in ABCA3 expression, routing, or function that may be associated with inherited or acquired lung disease may also be studied at the histochemical and ultrastructural level using these reagents.
Footnotes
Acknowledgements
This work was funded by the National Institutes of Health, Grant HL-53687 (to JAW, SEW, MTS, and TEW), Grant HL-33863 (to JAW and SEW), Grant HL-38859 (to JAW), and Grant HL-61646 (to JAW, TEW, and SEW).
We acknowledge the technical assistance of Paula Blair, David Loudy, and William Hull; secretarial assistance of Ann Maher; and photographic assistance of Terry Johnson.
