Abstract
The function of actin cytoskeleton in mesangial cells (MCs) during the recovering process of injured glomeruli is not fully understood. MCs in injured glomeruli express α-smooth muscle actin (α-SMA), which is not detected in normal glomeruli. We focused on the localization of α-SMA in MCs of Thy-1. 1 nephritic rat. Expression of α-SMA in the injured glomeruli peaked at day 5 after antibody injection and then declined gradually. At day 5, MCs, where α-SMA was localized at their cytoplasmic processes situated in various positions, occupied the expanded mesangium. MCs expressing α-SMA tended to be located at the peripheral region close to the glomerular basement membrane (GBM) or endothelial cells at day 8. Localization of α-SMA within the peripheral MCs was restricted to the cytoplasmic processes radiating toward the GBM and touching it with their tips at day 8. These α-SMA-containing processes are suitable to transmit the contractile force to GBM and may contribute to normalize the expanded glomerular volume. In addition, an actin-binding protein, drebrin, was localized in all MC processes extending toward various directions throughout the course of nephritis, suggesting that drebrin is involved in the formation of MC processes.
Keywords
M
MCs sustain the convoluted folding of glomerular capillaries by their contractile force to pull the GBM (Kriz et al. 1995). Laminin, a major component of GBM, is important for such an interaction. In laminin α5-null mice, glomerular capillaries were distended and exhibited a ballooned appearance due to the lack of MC adhesion to GBM (Kikkawa et al. 2003). A similar phenomenon is also inducible by a single injection of monoclonal antibody against Thy-1.1 molecule to rats. Administered antibodies bind to the surface of MCs and induce a complement-dependent mesangial injury followed by severe damage of the glomerular capillary network, but the interaction between podocytes and GBM is maintained in the damaged glomeruli. In spite of the drastic changes of the glomerular structure, most of the damaged glomeruli are restored in architecture at various degrees (Kriz et al. 2003). Consequently, Thy-1.1 nephritis is a useful model for investigating the restorative remodeling of glomerular structure. Using Thy-1.1 nephritis, we have demonstrated that intussusceptive angiogenesis characterized by tissue pillar formation is involved in the postinjury glomerular remodeling (Notoya et al. 2003).
In normal kidney, MCs extend the tongue-like processes containing actin filament bundle underneath the capillary endothelium toward mesangial angles where they are attached to the GBM (Sakai and Kriz 1987). The subtype of actin protein expressed in normal differentiated MCs is limited to non-muscle cell types: β- and γ-cytoplasmic actins. However, in human and experimental glomerulonephritis, MCs express smooth muscle cell-type actin, α-smooth muscle actin (α-SMA), which is also expressed in specialized interstitial cells, so-called myofibroblasts, of various tissues under physiological and pathological conditions (Grinnell 1994; Tomasek et al. 2002). α-SMA within myofibroblasts is incorporated in stress fibers that allow the retractile movement producing wound contraction through its N-terminal domain Ac-EEED (Hinz et al. 2002). Although de novo expression of α-SMA is known as a molecular marker for phenotypical changes of MCs in glomerular disease (Johnson et al. 1991), the subcellular localization of α-SMA in MCs is still undefined. Also, the role of α-SMA expressed transiently in MCs is not known.
The multiplicity seen in structures and functions of actin cytoskeletons is ensured by various kinds of actin-binding proteins. Our recent study showed that α-SMA is temporally expressed in the alveolar interstitial cells and colocalized with an actin-binding protein, drebrin, in the cellular protrusion (Yamada et al. 2005). Drebrin (developmentally regulated brain protein) was originally identified as a protein related to brain development (Shirao et al. 1988,1989). In neuronal cell types, drebrin is involved in the formation of dendrites and dendritic spines (Shirao and Sekino 2001). Furthermore, drebrin is also expressed in the podocytes of particular mammals including human and is involved in the formation and maintenance of the foot processes (Peitsch et al. 2003). Although drebrin is expressed in normal MC (Peitsch et al. 1999), expression and localization of this molecule under the diseased condition have not yet been elucidated.
In the present study we demonstrate the roles of MCs and their actin cytoskeletons containing α-SMA in the restorative remodeling process of glomeruli injured by Thy-1.1 nephritis. Furthermore, we also show the involvement of drebrin in the formation of MC processes during glomerular remodeling.
Materials and Methods
Reagents and Antibodies
Antipain, benzamidine, leupeptin, pepstatin A, PMSF, BSA (fraction V), diaminobenzidine (DAB), and saponin were obtained from Sigma-Aldrich (St Louis, MO). Mouse monoclonal anti-proliferating cell nuclear antigen (PCNA), clone PC10, was from Novocastra Laboratories (Newcastle, UK). Rabbit polyclonal anti-desmin antibody, mouse monoclonal anti-α-SMA antibody (ASM-1), mouse monoclonal anti-drebrin antibody (clone MX823), and guinea pig polyclonal anti-drebrin antibody (GP823) were from Progen (Heidelberg, Germany). The monoclonal anti-drebrin antibody was used for immunoelectron microscopy, whereas the polyclonal one was used for immunofluorescence staining and immunoblot analysis. Mouse monoclonal anti-β-cytoplasmic actin antibody (clone AC-74) was from Sigma-Aldrich. TRITC-conjugated donkey anti-mouse IgG F(abî)2 fragment, FITC-conjugated donkey anti-rabbit and anti-guinea pig IgG F(abî)2 fragments, and horseradish-peroxidase (HRP)-conjugated goat anti-mouse IgG F(abî)2 fragment were from Jackson ImmunoResearch Laboratories (West Grove, PA). FITC-conjugated Lycopersicon esculentum (tomato) lectin and Vectastain ABC kit (containing avidin DH, biotinylated HRP, and biotinylated anti-mouse IgG antibody) was from Vector Laboratories (Burlingame, CA).
Induction of Thy-1.1 Nephritis
All procedures performed on laboratory animals were approved by the Institutional Animal Care Committee of Juntendo University School of Medicine. All animal experiments were in compliance with the guidelines for animal experimentation of Juntendo University School of Medicine.
Six-week-old male Wistar rats were obtained from Charles River Japan (Kanagawa, Japan). Animals were kept in an airconditioned room and given free access to standard rat chow and tap water. To induce Thy-1.1 nephritis, mouse monoclonal anti-Thy-1.1 antibody E30 (100 μl/rat) was intravenously injected under anesthesia with diethyl ether (Shinosaki et al. 2002). Kidneys from control and Thy-1.1 nephritis rats (days 1, 2, 3, 4, 5, 8, 14, and 30 after the antibody injection) were prepared for immunoblot and immunohistochemical analyses.
Immunoblot Analysis
Kidneys of control and Thy-1.1 nephritic animals were perfused with PBS, pH 7.4, including protease inhibitors (1 mM each antipain, benzamidine, leupeptin, pepstatin A, and PMSF) under anesthesia with Nembutal (Abbott Laboratories; Abbott Park, IL). Rat glomeruli were isolated by graded sieving at 4C in the presence of protease inhibitors. Glomerular fractions were collected by centrifugation at 800 × g for 5 min and washed with PBS including protease inhibitors. Isolated glomeruli were solubilized in PBS including protease inhibitors, 1% SDS, and 5 mM EDTA. We loaded 50 μg (Thy-1.1 and α-SMA) or 200 μg (drebrin) of protein from each lysate onto a 7.5% (drebrin) or 12% (Thy-1.1 and α-SMA) SDS-polyacrylamide gel and subjected these gels to electrophoresis. Proteins were then transferred onto nitrocellulose membranes. Membranes were incubated with anti-Thy-1.1 (E30, 1:1000), anti-drebrin (GP823, 1:500), or anti-α-SMA (ASM-1, 1:1000) antibodies and then with the adequate HRP-conjugated secondary antibodies (1:2500). Bound antibodies were detected by ECL Western blotting detection system (Amersham Biosciences; Arlington Heights, IL).
Measurement of Proliferating Cells
Kidneys of control and Thy-1.1 nephritic animals were fixed in methacarn solutions overnight at 4C and embedded in paraffin. Specimens were cut into 4-μm-thick sections and mounted on glass slides. Sections were deparaffinized and incubated for 1 hr at room temperature with mouse monoclonal anti-PCNA antibody diluted 1:100 with PBS. Subsequently, sections were incubated with biotinylated anti-mouse IgG (1:100) for 30 min at room temperature, washed with PBS, and then placed in methanol containing 0.3% H2O2 for 20 min to neutralize endogenous peroxidase. Sections were adequately washed with PBS and visualized by reaction with avidin-biotin-peroxidase using Vectastatin ABC kit (Vector Laboratories). Sections were then incubated with DAB solution (0.7 mg/ml in Tris-buffered saline containing 0.02% H2O2) for 10 min, and the reaction was stopped. The number of PCNA-positive cells was evaluated in 30 glomerular sections per each animal. Value was expressed as the mean number of positive cells per one glomerular section from three animals in each group.
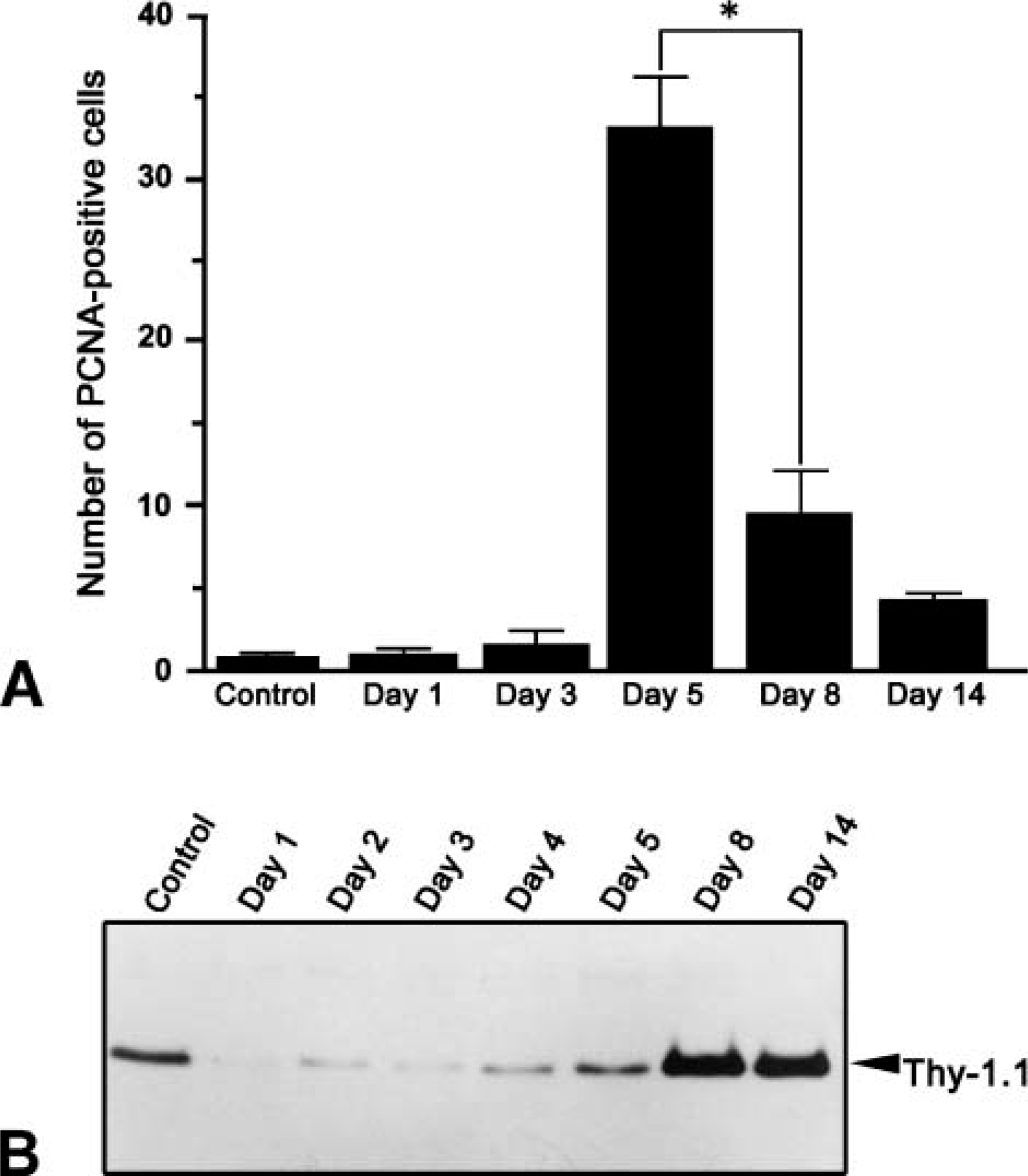
Number of PCNA-positive cells and protein expression of Thy-1.1 in Thy-1.1 nephritic glomeruli.
Confocal Laser Scanning Microscopy
Kidneys of control and Thy-1.1 nephritic animals were perfused with 4% paraformaldehyde fixative buffered with 0.1 M phosphate buffer (PB, pH 7.4) under anesthesia with Nembutal (Abbott Laboratories). The extirpated kidneys were cut into small pieces and immersed in the same fixative for ~30 min. After washing with PBS, tissue was immersed successively in PBS including 10%, 15%, and 20% sucrose, respectively, for 4, 12, and 4 hr. After the tissue was embedded in optimal cutting temperature compound and frozen, 7-μm-thick cryosections were cut using a Jung Frigocut 2800E (Leica; Wetzlar, Germany) and then mounted on silane-coated glass slides (Matsunami Glass Industries; Osaka, Japan). Sections were rinsed with PBS and blocked in blocking solution (0.1% BSA in PBS). Sections were incubated for 2 hr at room temperature with primary antibodies diluted with 1% BSA at 1:100. Subsequently, sections were incubated for 1 hr at room temperature with adequate secondary antibodies diluted with 1% BSA at 1:100 to 1:200. Fluorescence specimens were viewed with confocal laser scanning microscope (LSM510; Carl Zeiss, Oberkochen, Germany). Confocal images were obtained at 1.6-μm optical thickness.
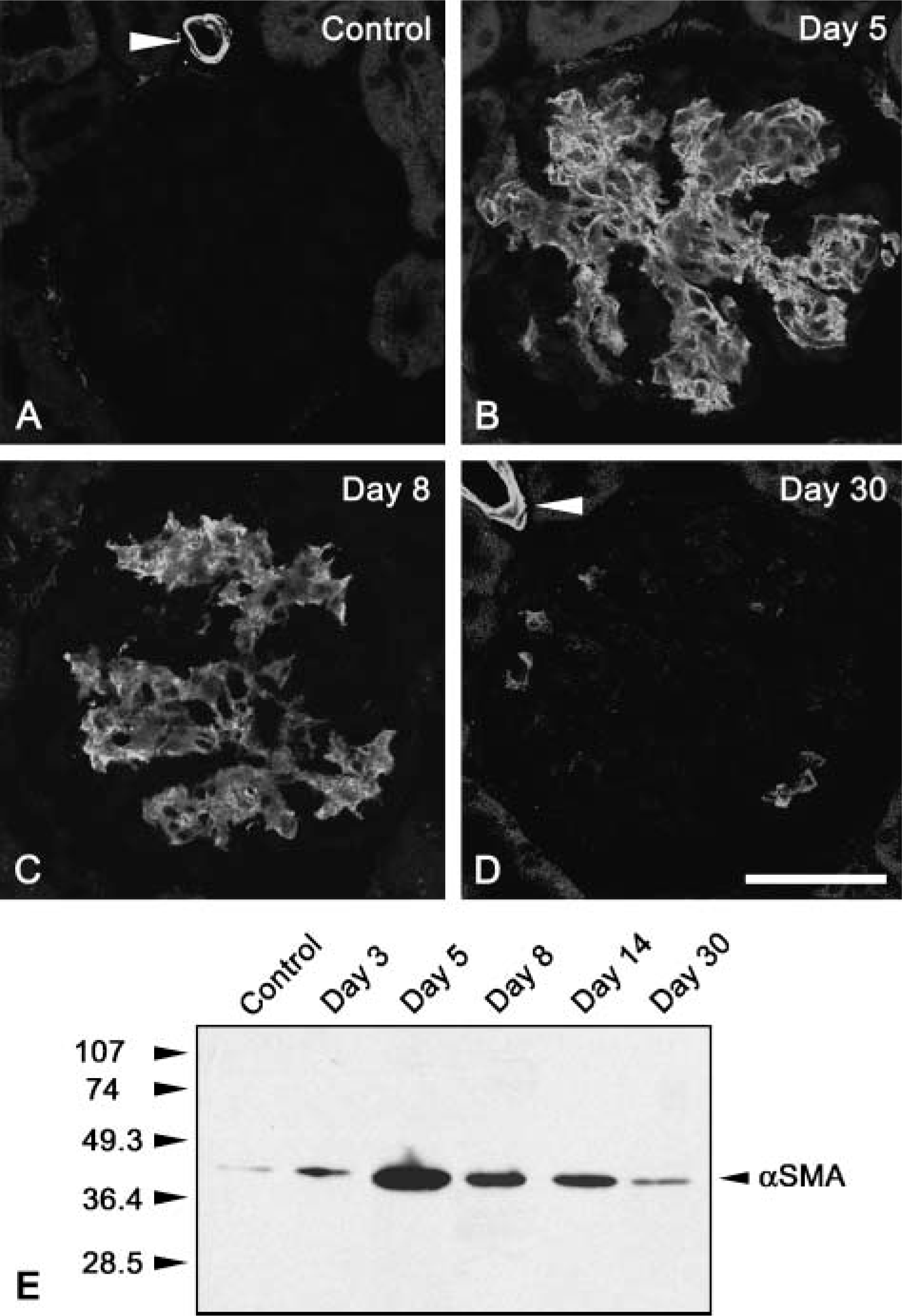
Protein expression of α-SMA in Thy-1.1 nephritic glomeruli. Expression of α-SMA in glomeruli of control and Thy-1.1 nephritis rats (days 5, 8, and 30) was shown by confocal laser scanning microscopy. No immunoreactivity for α-SMA was detected in control glomerulus
Specimens for Z-scanning analysis were processed by another protocol described below. Frozen kidney was cut into 50-μm-thick slices, and sections were then floated and washed with PBS and blocked in blocking solution (0.1% BSA/0.05% saponin in PBS). Sections were incubated for 12 hr at room temperature with anti-α-SMA antibody (1:100) in 1% BSA/0.05% saponin solution. Subsequently, sections were incubated for 6 hr at room temperature with both TRITC-conjugated anti-mouse IgG (abî)2 fragments (1:200) and FITC-conjugated tomato lectin (1:100) in 1% BSA/0.05% saponin solution. Fluorescence specimens were mounted on silane-coated glass slides and then Z-scanned at 1.6-μm optical thickness.
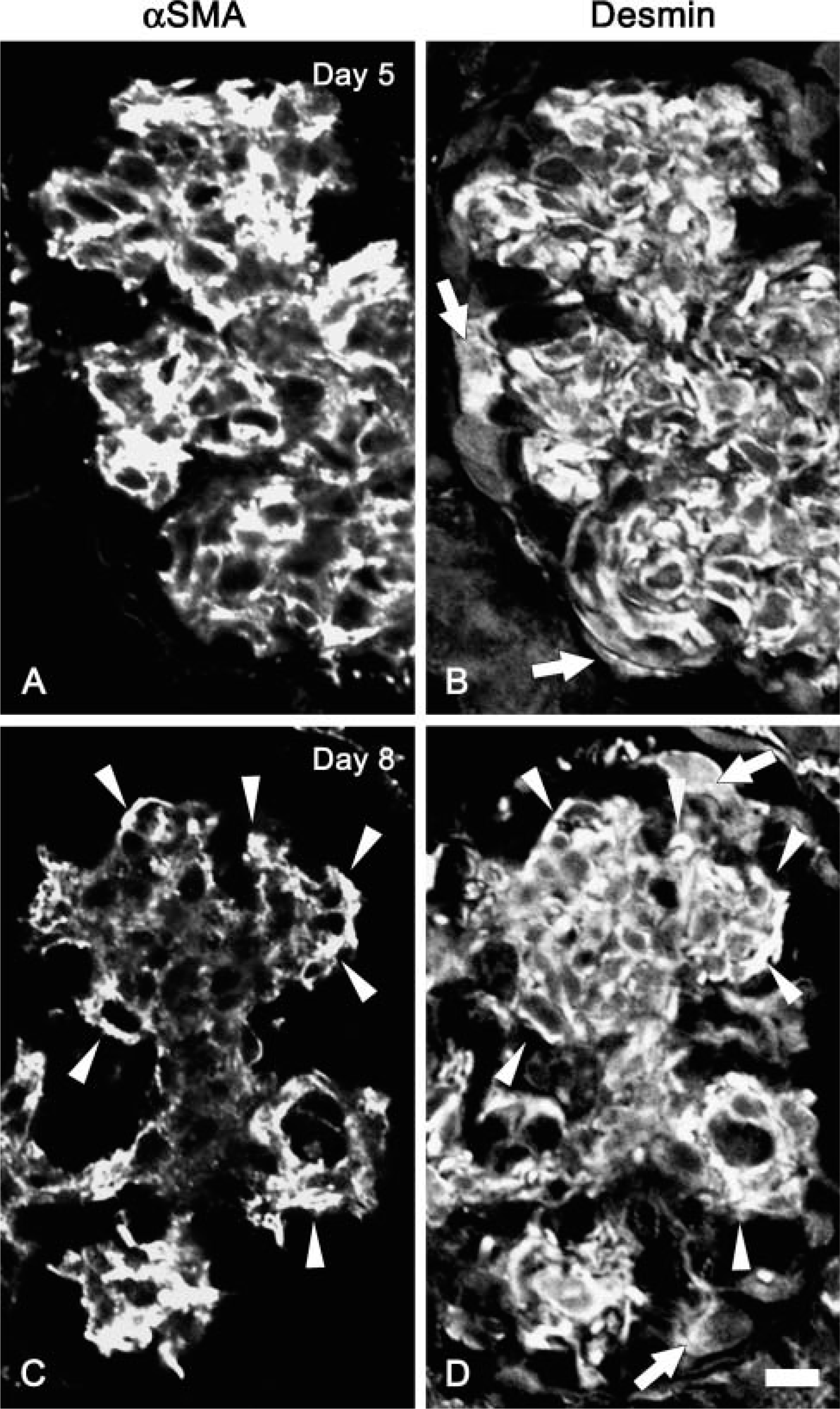
Double immunofluorescence staining for α-SMA and desmin in Thy-1.1 nephritic glomeruli at day 5 and day 8. Localization of α-SMA and desmin in glomeruli of Thy-1.1 nephritis rats at day 5
Immunoelectron Microscopy
Frozen samples of kidneys were prepared from Thy-1.1 nephritic animals as mentioned above. The 50-μm-thick cryosections were floated and washed with PBS and then blocked in blocking solution (0.1% BSA/0.05% saponin in PBS). Sections were incubated for 12 hr at room temperature with anti-α-SMA antibody (1:50) or anti-drebrin antibody (GP823, 1:50) in 1% BSA/0.05% saponin solution. Subsequently, the sections were incubated with HRP-conjugated anti-mouse IgG F(abî)2 fragments (1:100) for 6 hr, with DAB solution (0.7 mg/ml in TBS including 0.02% H2O2) for 10-15 min, and with 1% OsO4 in 0.1 M cacodylate buffer for 45 min, in that order. After dehydration with a graded series of ethanol, sections were embedded in epoxy resin. Ultrathin gold sections were processed with a diamond knife, transferred to copper grids (50 mesh) with Formvar membrane, and observed in a transmission electron microscope (JEM1230; JEOL, Tokyo, Japan).
Statistics
Data are shown as the mean ± standard error. Significance of the difference from each value was calculated by Student's t-test.
Results
Characteristics of E30-induced Thy-1.1 Nephritis
A single injection of mouse monoclonal antibody E30 induced mesangiolysis within a day. Prominent proliferation of MCs and enlargement of mesangium including collagen fibrils were observed at day 5. The injured glomeruli, however, had returned to almost normal in architecture within a month as described previously (Shinosaki et al. 2002). The number of proliferating cells in the glomerulus was evaluated by staining with anti-PCNA antibody in E30-treated rats (Figure 1A). PCNA-positive glomerular cells drastically increased by day 5 and then significantly decreased at day 8 and day 14. Immunoblot data showed that Thy 1.1 protein recognized with E30 was dramatically decreased due to the mesangiolysis 1 day after E30 injection. The signal for Thy-1.1 remained reduced until day 5 and then increased markedly at day 8 and thereafter (Figure 1B).
Expression and Localization of α-SMA in E30-induced Thy-1.1 Nephritic Glomeruli
Immunofluorescence signal for α-SMA was not detected in normal glomeruli of control rats, except for smooth muscle cells of efferent and afferent arterioles (Figure 2A). At day 5 after E30 injection, prominent signals for α-SMA were seen in the mesangial area (Figure 2B). Thereafter, the signal for α-SMA gradually decreased from day 8 to day 30 (Figures 2C and 2D). Furthermore, we examined expression of α-SMA in the isolated glomeruli from control and Thy-1.1 nephritic rats using immunoblot analysis. α-SMA expression increased remarkably by day 5, gradually decreasing from day 8 to day 30 (Figure 2E).
Distribution of α-SMA-positive cells in the glomerulus altered between day 5 and day 8 after E30 injection. At day 5, α-SMA-positive MCs were found throughout the expanded mesangial area (Figure 3A). However, at day 8, α-SMA-positive MCs were mainly found at the peripheral region of the expanded mesangial area, with no or faint signals at the central region (Figure 3C). We detected immunoreactivity for desmin, an intermediate filament protein expressed by MCs throughout the expanded mesangial area including the central region, both at day 5 and day 8 (Figures 3B and 3D). This fact indicated that the MCs also existed in the central region at day 8. Immunoreactivity for α-SMA was segregated from that for desmin within the MCs. Desmin was localized mainly at the perinuclear cytoplasm of the MCs, whereas α-SMA was localized at the periphery of the cells including the cytoplasmic processes (data not shown). Both at day 5 and day 8, desmin was also localized in the podocytes, which were positioned at the periphery of the glomerulus (arrows in Figures 3B and 3D), whereas this molecule was not expressed in normal rat podocytes (data not shown).
To investigate detailed localization of α-SMA, we performed immunoelectron microscopy by using specific antibody to α-SMA. At day 5, immunoreactivity for α-SMA was recognized along the entire portion of the cell periphery, especially at the cytoplasmic processes, in almost all of the MCs (Figure 4A). However, at day 8, MCs at the peripheral region of the expanded mesangium predominantly exhibited immunoreactivity for α-SMA, but not at the central region of the mesangium. Peripheral MCs were situated close to the GBM or endothelial cells, and the central MCs were separated from the GBM (Figure 4B). Localization of α-SMA within the peripheral MCs was restricted to the site facing the GBM or endothelial cells, especially to the cytoplasmic processes radiating toward the GBM, which was attached with their tips (Figures 4B and 4C). As seen in Figure 4C, some α-SMA-containing processes that were ~1-3 μm wide and bound to the GBM were often positioned between two capillary lumina.
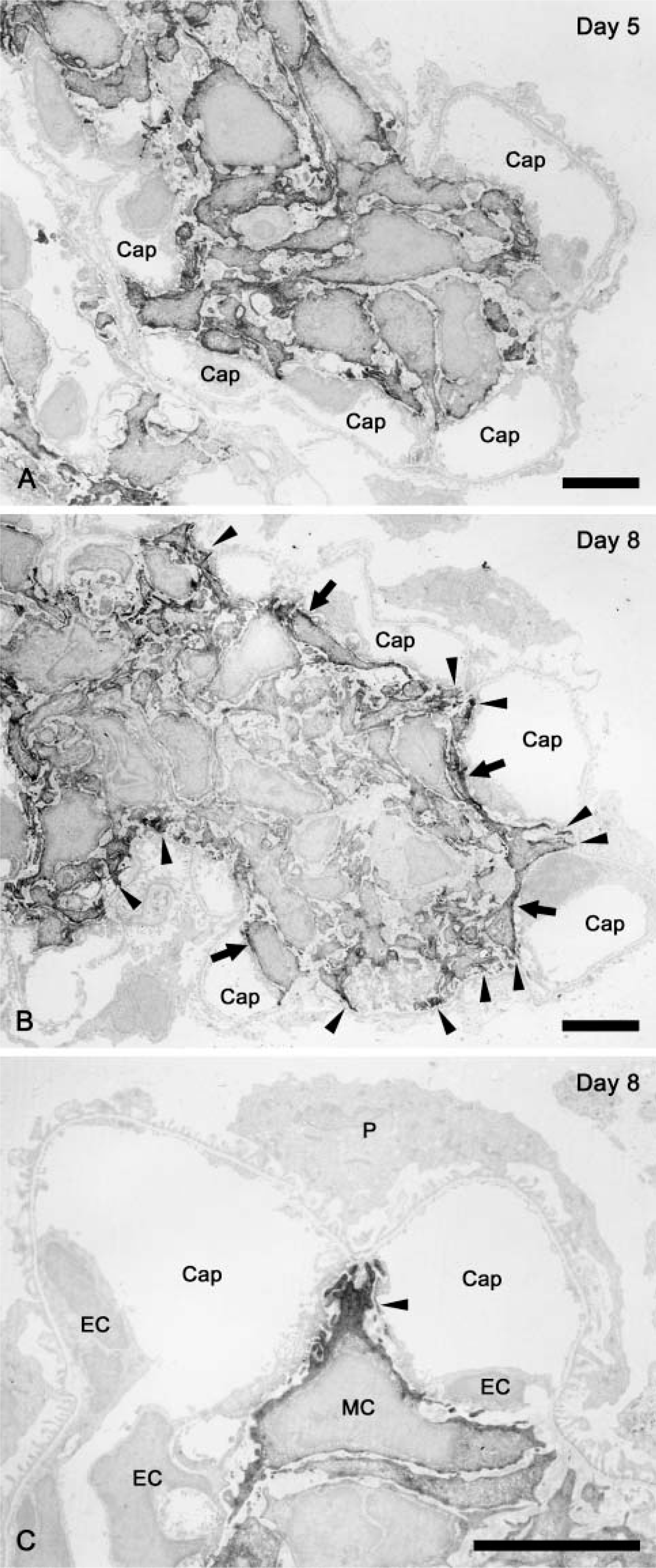
Immunoelectron microscopy for α-SMA in Thy-1.1 nephritic glomeruli at day 5 and day 8. Immunoreactivity for α-SMA in glomeruli of Thy-1.1 nephritis rats was shown as DAB chromogen. At day 5, almost all MCs exhibited immunoreactivity for α-SMA, and the reactivity was recognized along the entire portion of the cell periphery, especially in the cytoplasmic processes
We have previously demonstrated that intussusceptive capillary growth occurs during the reconstruction of glomerular vasculature (Notoya et al. 2003). In this angiogenesis process, tissue pillar formation is a key event. By using vascular casts and scanning electron microscopy, the tissue pillar is identified as a tiny hole (~1 μm in diameter). To clarify the contribution of MC processes to tissue pillar formation, we obtained the serial Z-scanning images of the thick sections labeled with the anti-αSMA antibody and an endothelial marker, tomato lectin, which binds specifically to the apical surface of rat endothelial cells (Porter et al. 1990). We could then recognize that an α-SMA-containing cell protrusion penetrated the glomerular capillary canal through a tissue pillar of ~1 μm in diameter and traversed the capillary lumen at several portions, mainly at day 8 (Figure 5).
Expression and Localization of Drebrin in E30-induced Thy-1.1 Nephritic Glomeruli
Formation of cytoplasmic processes is one of the characteristics of MCs. The above-mentioned data indicate that α-SMA is concerned with the process formation in MCs during restorative glomerular remodeling. Recently, it has been shown that drebrin, a kind of actin-binding protein expressed in MCs, is involved in the formation of cytoplasmic processes in some cell types including neuron and podocytes. Therefore, we next examined the expression and localization of drebrin in MCs in Thy-1.1 nephritic rat glomerulus.
Immunoreactivity for drebrin was detected in the mesangial area of both normal (Figure 6A) and Thy-1.1 nephritic (Figures 6B-6D) glomeruli. We found prominent dot-like signals for drebrin in the proliferated MCs, especially at day 5 and day 8 (Figures 6B and 6C). Immunoblot analysis using anti-drebrin antibody revealed the higher expression of drebrin in the glomerulus from day 3 to day 14 after E30 injection. Protein expression of drebrin returned to control level at day 30 (Figure 6E). Immunoelectron microscopy using anti-drebrin antibody revealed that drebrin was concentrated at the MC processes in Thy-1.1 nephritic glomeruli (Figure 7). Judging from the findings of immunoelectron microscopy, dot-like signals for drebrin at day 5 and day 8 probably represented the MC processes.
Drebrin was detected in the MCs not only at the peripheral region of the mesangium but also at the central region at both day 5 and day 8 (Figures 8A, 8D, 8G, and 8J). Most dot-like signals for drebrin were colocalized with α-SMA at day 5; however, dot-like signals were colocalized with α-SMA only in the peripheral MCs at day 8 (Figures 8A-8F). Furthermore, dot-like signals were also colocalized with β-cytoplasmic actin (βCPA) at both day 5 and day 8 (Figures 8G-8L). Glomerular cells (MCs, podocytes, and endothelial cells) predominantly expressed βCPA in normal and diseased glomeruli. Unlike α-SMA, βCPA was expressed both in the peripheral and the central MCs throughout the recovery course of Thy-1.1 nephritis (Figures 8H and 8K).
Discussion
Several reports have demonstrated that α-SMA is specifically expressed in proliferated MCs in injured glomerulus (Johnson et al. 1991; Alpers et al. 1992). Therefore, this protein is regarded as an appropriate indicator for mesangial proliferative glomerulonephritis. However, the biological significance of α-SMA expressed in MCs remains to be elucidated. In the present study, we demonstrated the dynamic reorganization of α-SMA-containing actin cytoskeleton of the MCs in the Thy-1.1 nephritic glomeruli. Furthermore, we discuss the functional significance of α-SMA-containing actin cytoskeleton during restorative remodeling in Thy-1.1 nephritic glomeruli.
Studies on the function of α-SMA using myofibroblasts and smooth muscle cells suggest that α-SMA plays a direct role in the contractile activity of those cells. Myofibroblasts possessing α-SMA exert a strong contractile force to pull the surrounding extracellular matrices compared with fibroblasts that express only non-muscle cell type(s) of actin (Hinz et al. 2002). Furthermore, impaired vascular contractility is shown in the α-SMA-null mice, in which α-skeletal muscle actin is irregularly expressed in vascular smooth muscle cells (Schildmeyer et al. 2000). Based on these facts, it is possible that the contractile apparatus containing α-SMA can generate stronger tensile force in fibroblasts and smooth muscle cells than that constructed by other types of actin. According to our results, peripheral MCs, not central ones, in expanded mesangium predominantly expressed α-SMA at day 8 after E30 injection. At that time, α-SMA was concentrated at their cytoplasmic processes projecting toward and adhering to the GBM. Therefore, it is strongly suggested that peripheral MCs efficiently pull the expanded GBM centripetally through the α-SMA-containing processes and contribute to the normalization of the expanded glomerular volume (Figure 9). Moreover, the superficial actin bundle along a capillary in a main body of peripheral MCs may interconnect the actin bundles in the mesangial processes that project toward the GBM and are located next to a capillary. It is thought that the contraction of the unit of these actin bundles contributes to refolding of the GBM. Kriz et al. (2003) have also reported a similar idea: MCs in the expanded glomerulus establish an interconnection between the GBM and the newly formed network of collagen fibers (or between opposing points of the GBM), and thus the contraction of MCs may lead the expanded glomerulus to normal condition in volume, as seen in wound closure.
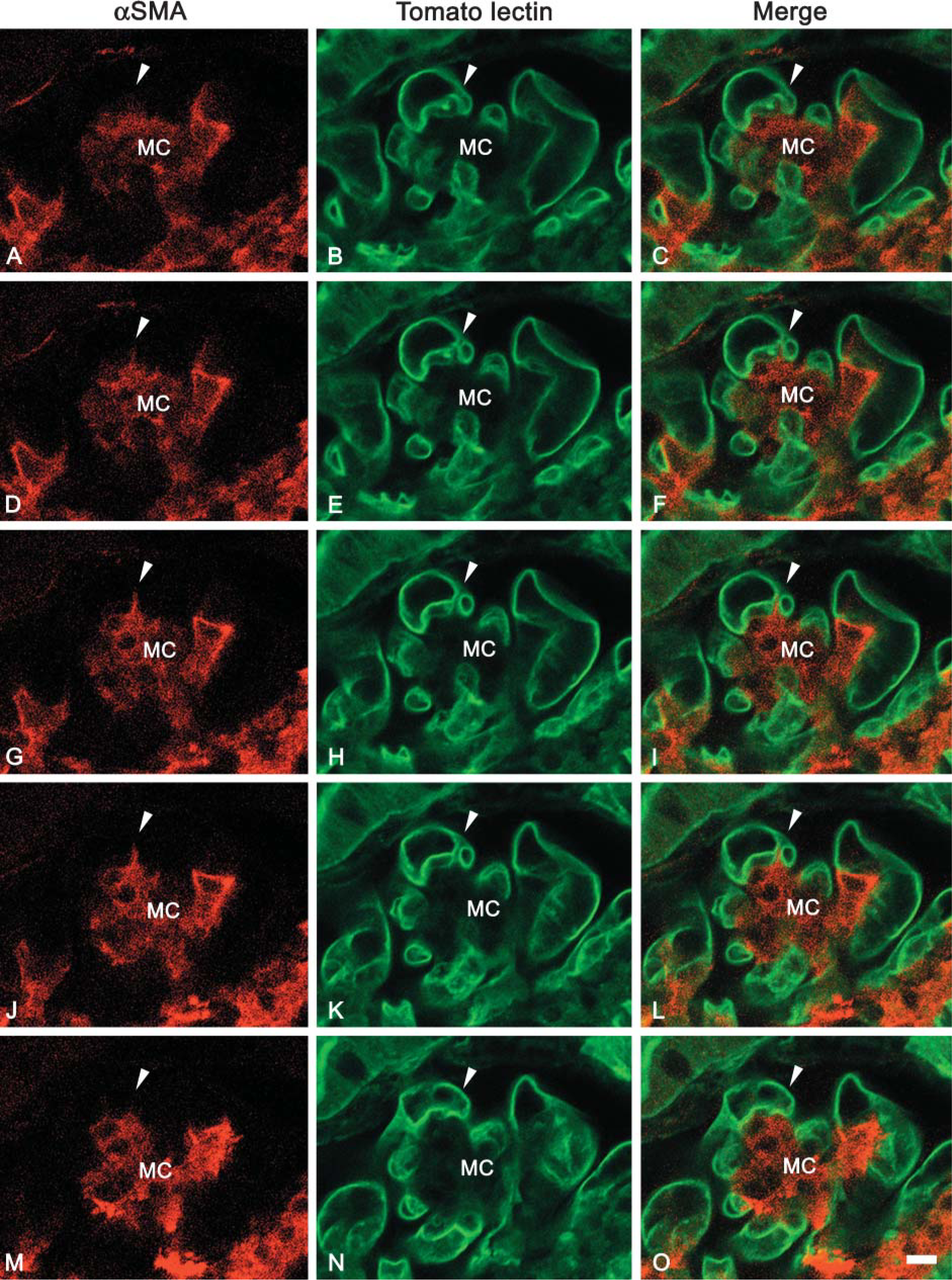
Serial confocal images showing the spatial relation between α-SMA-containing MC processes and endothelial cells in Thy-1.1 nephritic glomerulus at day 8. α-SMA (red in
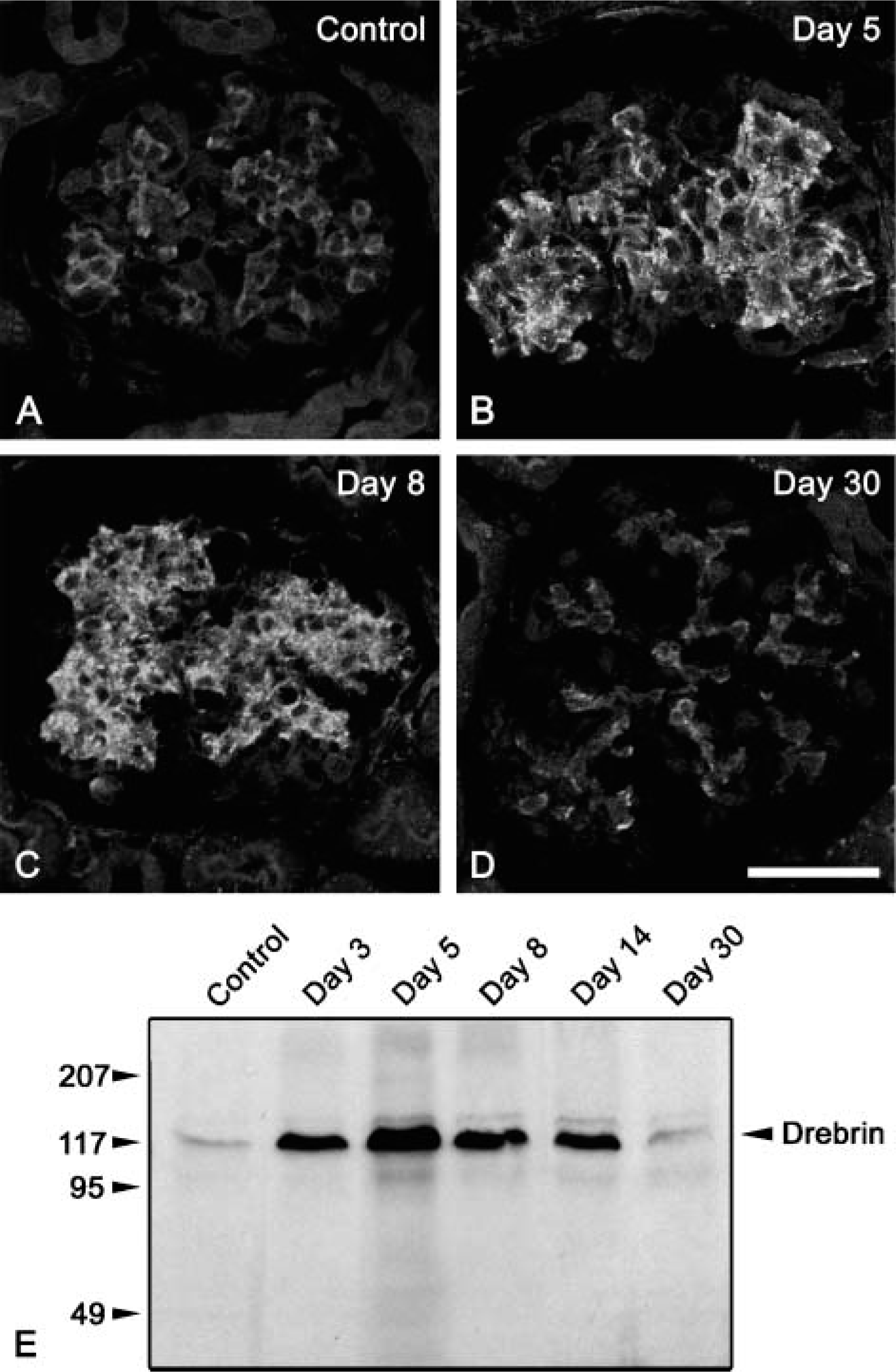
Protein expression of drebrin in Thy-1.1 nephritic glomeruli. Expression of drebrin in glomeruli of control (
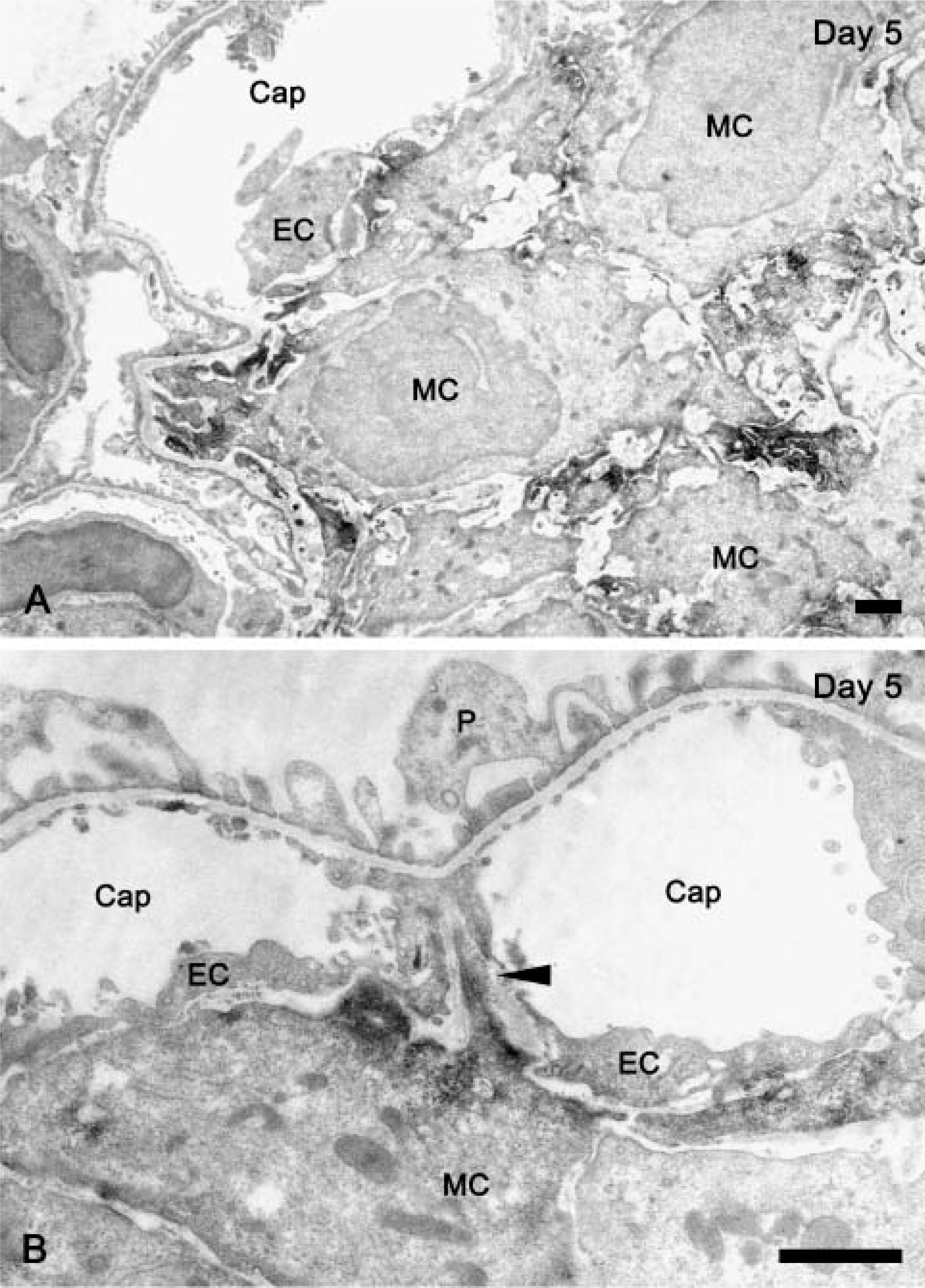
Immunoelectron microscopy for drebrin in Thy-1.1 nephritic glomeruli at day 5. Immunoreactivity for drebrin in glomeruli of Thy-1.1 nephritis rats at day 5 was shown as DAB chromogen. Signals were predominantly recognized in MC processes (
Although the majority of the α-SMA-containing processes of peripheral MCs were located next to the capillary loops, a part of these processes penetrated the capillary canal via the tissue pillar involved in the intussusceptive capillary growth at the recovery phase of Thy-1.1 nephritis. This style of capillary growth has also been reported in various developing tissues (Burri and Tarek 1990; Patan et al. 1993; Djonov et al. 2000). In general, the growing capillary network by the intussusceptive fashion represents a transluminal tissue pillar running along the vertical axis against the blood flow. The tissue pillar forms two daughter capillary loops and contributes to a rapid increase in capillary density of existent capillary network. Our results indicate that a part of α-SMA-containing MC processes also contribute to the formation of a tissue pillar in Thy-1.1 nephritic glomerulus.
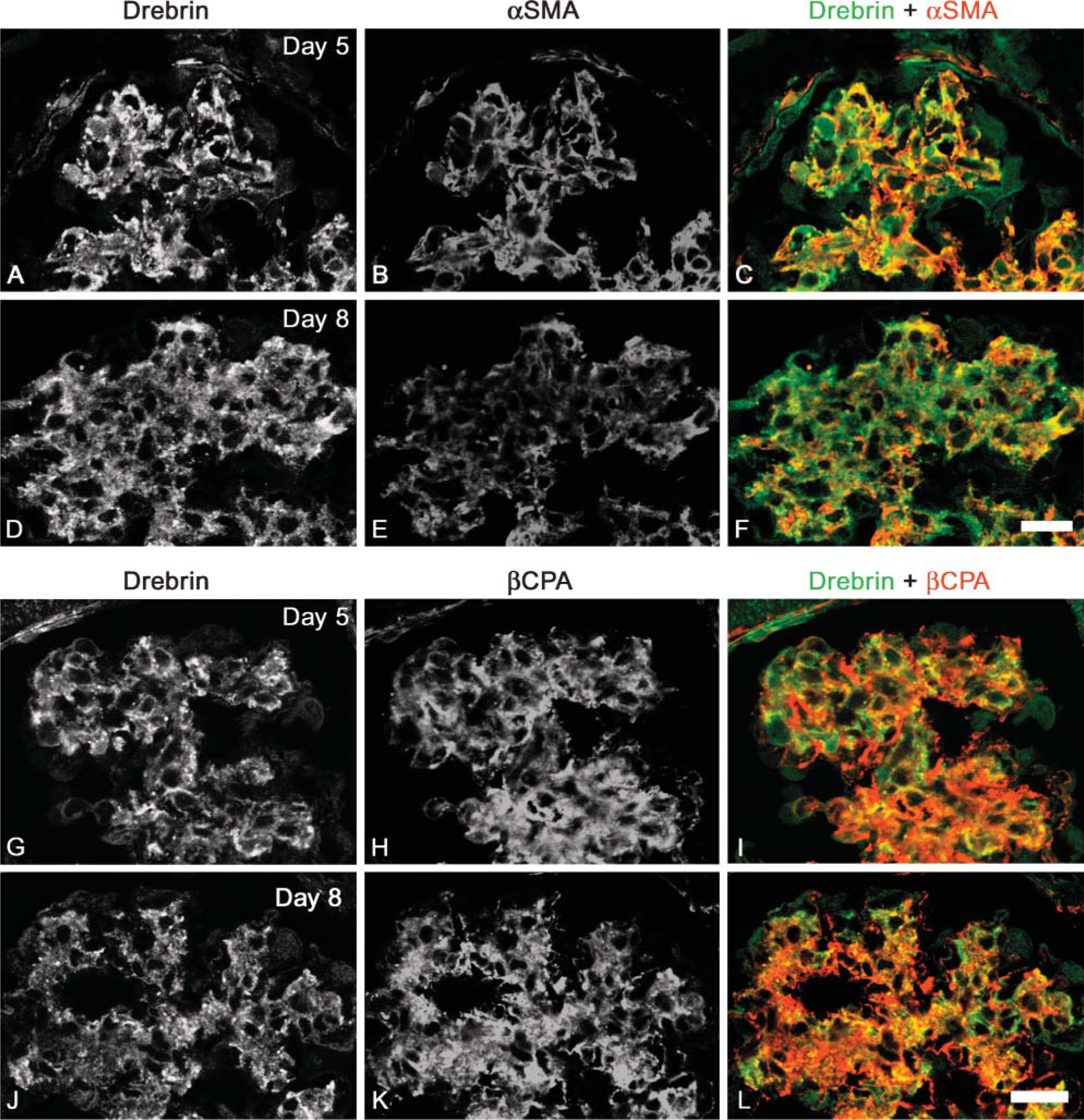
Double immunofluorescence stainings for drebrin and actin isoforms in Thy-1.1 nephritic glomeruli at day 5 and day 8. Localization of drebrin and actin isoforms (α-SMA,
In E30-induced Thy-1.1 nephritis, both protein expression of α-SMA and number of PCNA-positive cells in the glomeruli showed a peak at day 5 and declined at day 8. On the contrary, protein expression of Thy-1.1, which is a marker of rat MCs and E30 antigen, was faint at day 5 but dramatically increased at day 8. Therefore, the present findings suggest that the changes of mesangial phenotype were recognized between day 5 and day 8 in Thy-1.1 nephritis. A similar reciprocal phenomenon on the expression of Thy-1 and α-SMA is observed in lung fibroblast (Hagood et al. 2005). Normal lung fibroblasts express Thy-1 molecule; however, the proliferated fibroblasts in the bleomycin-induced pulmonary fibrosis lose Thy-1 expression and show myofibroblastic transformation involving de novo expression of α-SMA. Because both MCs and lung fibroblasts lose Thy-1 expression when they show proliferative activity, the Thy-1 molecule can be regarded as a differentiation marker for these cell types.
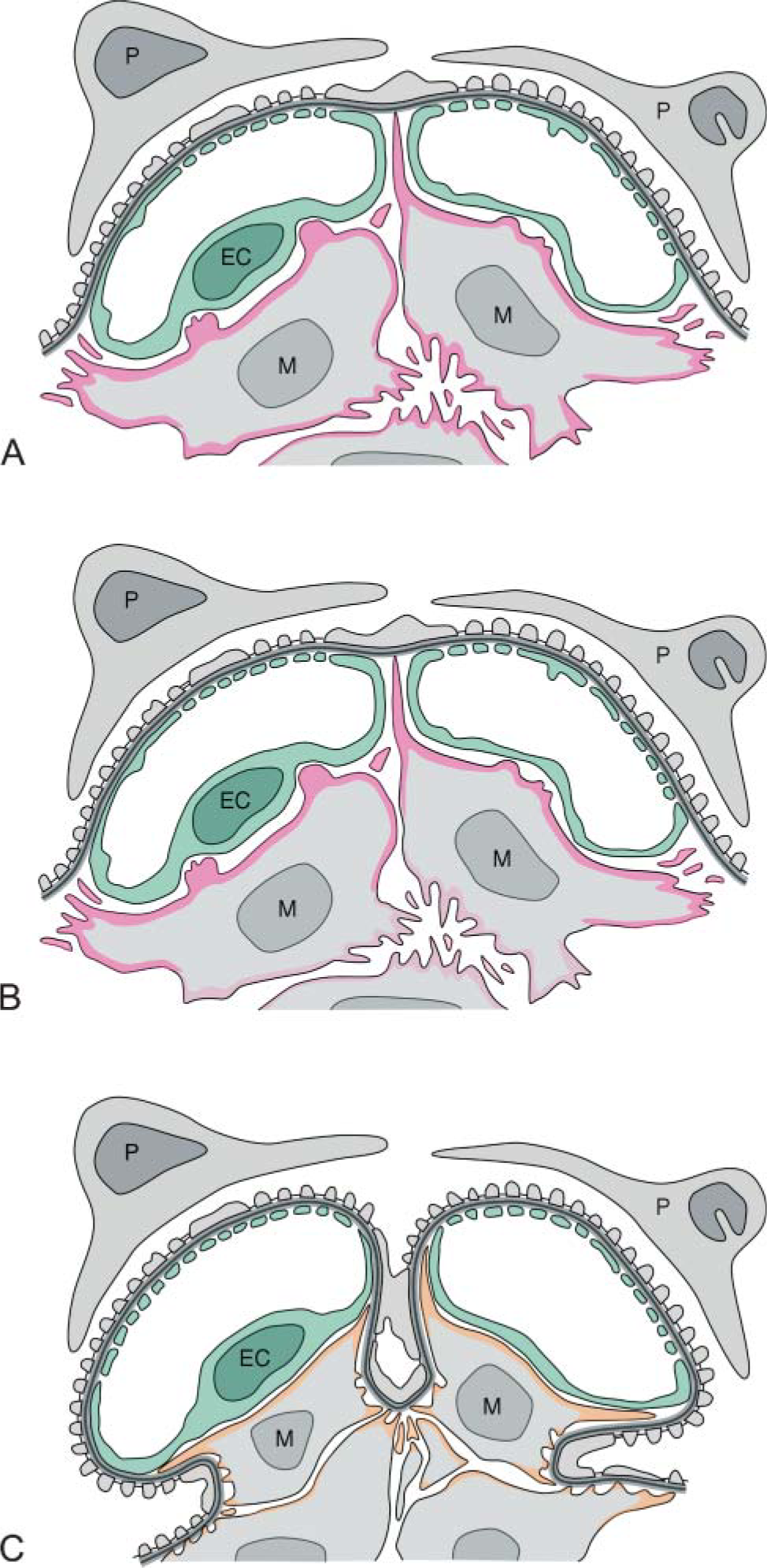
Schematic drawings showing the contribution of α-SMA-containing MC processes to glomerular remodeling in repairing phase of Thy-1.1 nephritis. The remodeling process of injured glomerulus in Thy-1.1 nephritis was depicted in due course. In the initial phase of the nephritis, α-SMA (pink) was expressed in all MCs and was localized in the cell periphery, especially the cytoplasmic processes (
Previous works reported that apoptosis of MCs is a major cell clearance mechanism in the resolution of glomerular hypercellularity caused by rat Thy-1.1 nephritis model (Baker et al. 1994; Shimizu et al. 1995). There are no data on the cell signaling contributing to apoptotic cell death and survival in MCs; however, release from mechanical tension by reorganization of the actin cytoskeleton triggered an apoptotic response in human fibroblasts (Grinnell et al. 1999). If the release from mechanical tension also induces apoptosis of MCs, the unloaded central MCs, which are separated from the GBM and do not adhere to it, might be excluded by this mechanism. On the other hand, peripheral MCs, which adhere to the GBM through their α-SMA-containing processes, may survive during the process of apoptotic cell clearance.
In the present study we also examined expression and localization of drebrin in Thy-1.1 nephritic glomeruli. Drebrin was localized in the cytoplasmic processes of the proliferated MCs in Thy-1.1 nephritic glomeruli. Furthermore, drebrin remains in all MC processes after the decrement of α-SMA. Therefore, it is suggested that drebrin is involved in the formation and maintenance of MC processes during the restorative remodeling of Thy-1.1 nephritic glomeruli. Increased expression of drebrin in the injured glomeruli of E30-treated rats from day 3 to day 14 suggests that the MC processes are formed vigorously during the repairing phase of Thy-1.1 nephritis. Moreover, our data indicate that drebrin is extensively distributed in MCs with the actin isoforms.
In conclusion, the α-SMA-containing processes of MCs are suitable to effectively pull the GBM, and these processes contribute to re-folding of the GBM and to normalization of expanded glomerular volume during restorative remodeling of Thy-1.1 nephritic glomeruli. Moreover, drebrin is involved in the formation and maintenance of MC processes in glomerular remodeling.
Footnotes
Acknowledgment
This study was supported by Grants-in-Aid from the Ministry of Education, Science, Sports and Culture of Japan (grant #16390247 to HK).
