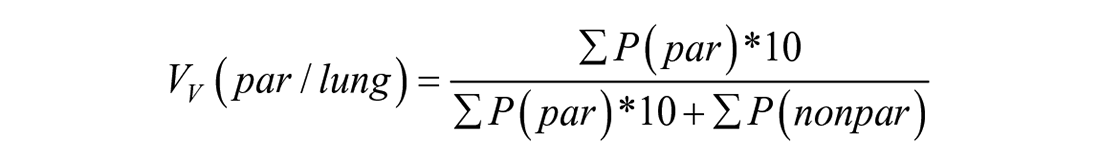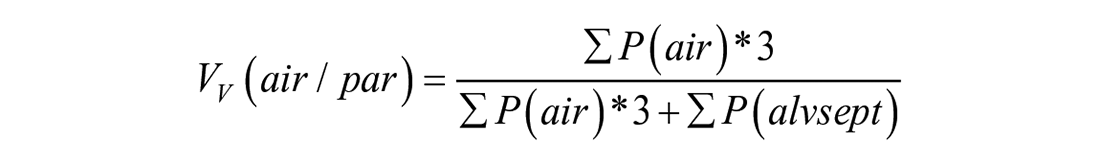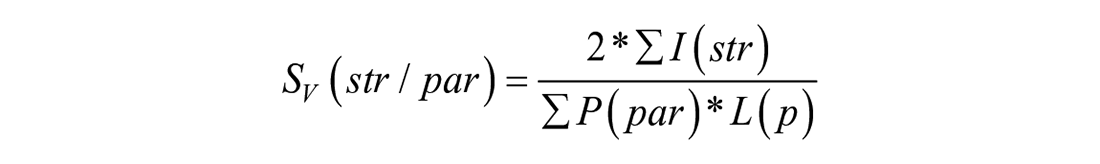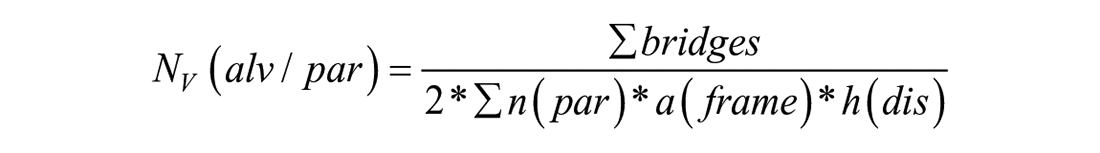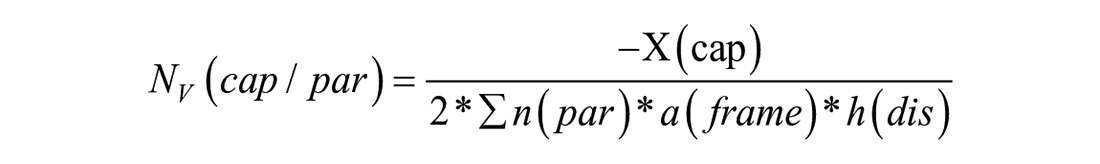Abstract
Bronchopulmonary dysplasia (BPD) is a developmental disorder of infants born prematurely, characterized by disrupted alveolarization and microvascular maturation. However, the sequence of alveolar and vascular alterations is currently not fully understood. Therefore, we used a rabbit model to evaluate alveolar and vascular development under preterm birth and hyperoxia, respectively. Pups were born by cesarean section 3 days before term and exposed for 7 days to hyperoxia (95% O2) or normoxia (21% O2). In addition, term-born rabbits were exposed to normoxia for 4 days. Rabbit lungs were fixed by vascular perfusion and prepared for stereological analysis. Normoxic preterm rabbits had a significantly lower number of alveoli than term rabbits. The number of septal capillaries was lower in preterm rabbits but less pronounced than the alveolar reduction. In hyperoxic preterm rabbits, the number of alveoli was similar to that in normoxic preterm animals; however, hyperoxia had a severe additional negative effect on the capillary number. In conclusion, preterm birth had a strong effect on alveolar development, and hyperoxia had a more pronounced effect on capillary development. The data provide a complex picture of the vascular hypothesis of BPD which rather seems to reflect the ambient oxygen concentration than the effect of premature birth.
Introduction
Bronchopulmonary dysplasia is a disease of the preterm-born infant that remains a significant problem in neonatology, increasingly shaped by extreme prematurity as advanced medical care improves survival.1–3
Diverse efforts were made to determine the factors that predestine or directly cause the characteristic changes of the lungs perceived in BPD infants. Considering previous research, causes are multifactorial and complex involving oxidative stress, inflammation, and mechanical stress.4,5 As the disease manifests during the early saccular stage of human lung development, understanding the underlying impairment in alveolar and vascular development is essential to discover adequate treatments for affected patients.1,6
For this purpose, different animal models have been used. Exposing newborn animals to high levels of hyperoxia is a frequently used model resulting in pulmonary tissue changes similar to those observed in infants suffering from BPD, namely an “alveolar simplification” and structurally abnormal microvasculature. 7 Term rodent models are cheap and easy to handle but show important differences in antenatal development, both temporally and structurally, when compared with humans. An advanced model is that of preterm rabbits exposed to hyperoxia, recently used by Deprest, Toelen and colleagues,8–12 Mühlfeld et al., 13 and Loi et al. 14 While rodents are born naturally in the early saccular stage of lung development and start alveolarization days later, rabbits and humans are born in the alveolar stage with their alveolarization starting before birth.11,15
Most studies using a rabbit BPD model have either made a comparison between normoxia and hyperoxia or have compared term-born rabbits with preterm-born rabbits without an hyperoxic intervention. Therefore, we included a term control group in our study to differentiate the effects of mere prematurity and hyperoxia as independent factors.
Increasing evidence hints to the “vascular hypothesis” of BPD, where errand microvascular development is thought to be a pivotal precondition for pathological changes in alveolar structure. 16 In human BPD patients,17,18 preterm infants, 7 and baboon BPD models,19,20 alveolar capillary networks (ACNs) were conspicuously dysmorphic. In mice, capillary changes seem to become apparent before impaired alveolarization. 21
For quantitative morphometry of the lung, design-based stereology is widely accepted to be the gold standard. In addition to methods quantifying total alveolar numbers, a recently established method allows the quantification of the microvasculature 22 by determining the number of septal capillary loops (SCLs). Unlike the capillary volume and surface area, this parameter is not affected by the perfusion and inflation pressure, changes in vessel diameter, or resolution during analysis. There has been no stereology-based numerical characterization of the microvasculature in BPD models so far.
Therefore, the present study used a stereological approach to analyze the effects of prematurity and hyperoxia on both the alveolar and microvascular systems of the rabbit lung.
Material and Methods
BPD Rabbit Model
All experimental procedures were approved by the local animal ethics committee (Ethics committee for Animal Experimentation of KU Leuven, Belgium) and complied with the standard European regulations on animal research (no. 744/2017) and the animal research: reporting of in vivo experiments (ARRIVE) guidelines.
Time-mated pregnant rabbits (New Zealand White and Flemish Giant cross-breed) underwent a cesarean section at 28 days of pregnancy (early saccular stage of lung development corresponding to a human developmental stage susceptible to BPD, term = 31 days) to extract neonatal pups through hysterotomy. After delivery, the pups were dried, stimulated, and placed in an incubator at 32C with 50% of humidity. Pups were fed twice daily via an orogastric tube with increasing quantities of a milk replacer approaching the protein/fat ratio of rabbit milk, supplemented with vitamins, probiotics, and immunoglobulins. Furthermore, intramuscular vitamin K was administered. For experimental conditions, all animals were randomly assigned to two different groups: a normoxia group (21% oxygen; NOX) and a hyperoxia group (≥95% oxygen; HYX). An additional control group (CO) consisted of animals that were delivered vaginally at term (31 days of gestation). CO pups were transferred to an incubator within 2 hr after birth and exposed to normoxia (all other interventions such as feeding or medical treatment were identical).
Deeply anesthetized animals (35 mg/kg ketamine and 6 mg/kg xylazin) were euthanized on day 7 of the experiment for all preterm pups and day 4 for term controls (same corrected gestational age). Lungs were fixed by vascular perfusion. In short, the trachea was exposed and cannulated with a blunt needle. In addition, the pulmonary trunk was cannulated. The lungs were fully inflated and afterwards deflated to approximately two third of the total capacity and maintained at a pressure of 5 cm H2O using a standard recruitment maneuver. Afterward, the chest was opened using a bilateral thoracotomy. The left atrial appendage was opened, and the pulmonary trunk was cannulated. Then, the perfusion of the vasculature was started at a pressure of 25 cm H2O. After shortly flushing the lungs with sodium chloride to remove blood cells from the vasculature, fixation was performed by perfusion of a medium containing 1.5% glutaraldehyde and 1.5% paraformaldehyde in 0.15-M N-2-hydroxyethylpiperazine-N ’-2-ethanesulfonic acid buffer for 10 minutes. After that the lungs were removed from the body and kept in toto in the same fixative for at least 24 hr.
Lung Volume Estimation, Sampling, and Embedding Procedures
The lungs were separated, and the left lung was stored for other analyses. Before sampling and tissue embedding, the volume of the right lung was estimated by volume displacement based on Archimedes’ principle. 23 The right lung was sectioned from apex to base into approximately equally thick slabs using a scalpel. The slices were subjected to a systematic uniform random sampling (SURS), i.e., starting randomly with the first or second slice, every second slice was assigned to glycol methacrylate embedding (Technovit 7100, Kulzer GmbH, Hanau, Germany) for light microscopy (LM). Samples for LM were osmicated, stained en bloc with uranyl acetate, dehydrated, and embedded in glycol methacrylate as described previously. 24 The other slices were further subjected to SURS into smaller tissue blocks (1 mm³) for epoxy resin embedding.
From the glycol-methacrylate-embedded tissue blocks, 1.5-μm-thick sections were cut at a similar distance from the block face to make sure that deformations at the outer part of the tissue were removed. The sections were stained with toluidine blue (Toluidin-Blau; Merck KGaA, Darmstadt, Germany) and used for LM estimation of standard volume parameters such as volume of parenchyma, nonparenchyma, septum, and alveolar airspace. Toluidine staining was performed by incubating the sections with 0.1% toluidine blue for 2 min. Afterwards, sections were rinsed with distilled water for a few seconds, dehydrated in an ascending isopropanol series, and covered with a cover slip. In addition, the first and third sections from a consecutive row of toluidine blue–stained sections were used as a disector to estimate the number of alveoli. 25
From the epoxy resin–embedded tissue blocks, the first and third sections from a consecutive row of semi-thin sections (0.5 μm each) were used as a disector to estimate the number of capillary loops in the interalveolar septa with LM. 22
All histological slides with tissue sections were scanned and digitalized using a microscopic slide scanner (AxioScan.Z1; Zeiss, Göttingen, Germany) at a 10× objective lens magnification for glycol methacrylate and 40× objective lens magnification for epoxy resin sections.
Design-Based Stereology
All stereological analyses were performed according to the American Thoracic Society/European Respiratory Society’s recommendations for quantitative assessment of lung structure 26 and based on established standards of design-based stereology27,28 and newly established methods. 22
For this study, only the right lung was investigated. Total values are therefore referring to the right lung as the reference space. All stereological analyses were performed using the newCAST software (Visiopharm, Hørsholm, Denmark). We applied automated SURS 29 to generate random, nonoverlapping fields for each parameter described below. Sampling fractions were individually set at a varying level between 10% and 30% that ensured registration of at least 100–200 counting events for each parameter per animal.
Estimation of volumes was carried out at 10× objective lens magnification. Fields of view were superimposed with a test system consisting of point grids. The first system used four points for parenchyma and 40 points for nonparenchyma. The second system used six points for septum and two points for airspace. In addition, atelectatic areas were counted using the septum test points. Volume densities
The multiplication of the points hitting the parenchyma by 10 in the upper formula relates to the fact that the coarse point grid used for counting the parenchyma consisted of one tenth of the points that were used to count the nonparenchyma (fine grid). Similarly, in the lower formula, the factor of three is included because the point grid used to count the alveolar septa (fine grid) had three times more points than the grid used for counting the airspace (coarse grid). Such combinations of fine and coarse grids are often used when the volume fractions of the structures of interest differ greatly to enhance the efficiency.
Estimation of alveolar epithelial and endothelial surface areas was carried out at 40× objective lens magnification. Fields of view were superimposed with a test system of a 52.9-μm-long probe line. The number of intersections with the desired structure
with
Estimation of alveolar numbers was carried out at 10× objective lens magnification. Two consecutive sections were aligned within the Visiopharm stereology software to form a physical disector. The image pairs with a known disector height of 3 μm were superimposed with a counting frame with an area of 200,000 μm2. The number of alveoli was acquired based on the Euler–Poincaré characteristic (EPC) as previously described.25,30 Briefly, the EPC can be used to characterize a complex network by determining the total number of components based on the connectivity of the elements composing the network. The EPC or Euler number considers three different topological phenomena, visible when comparing two consecutive sections in a physical disector.
22
For the acquisition of the total number of alveoli, the Euler number is reduced to one phenomenon called “bridges,” representing the number of alveolar openings into alveolar ducts. The numerical densities of alveoli were calculated by relating the sum of observed bridges to the disector volume (counting frame size
Estimation of the numbers of SCL was carried out at 40× objective lens magnification. Two consecutive sections were aligned within the Visiopharm stereology software to form a physical disector. The image pairs with a known disector height of 1 μm were superimposed with a counting frame with an area of 10,000 μm2. As described for the alveoli, the EPC measures the connectivity of a network. For the microvasculature of the lung, the EPC cannot determine the smallest unit, i.e., capillary segments, but rather the unit of SCLs, defined as the shortest cycle of capillary segments around a tissue pillar.
22
For the microvasculature, connectivity is observed in a physical disector by counting three topological phenomena ascribed as “bridges” (new connections between two capillary profiles), “islands” (new isolated capillary profiles), and “holes” (new enclosed cavities within capillary profiles).22,31 The numerical densities of SCL were calculated by relating the Euler number to the disector volume (counting frame size
with the Euler number
The total numbers of alveoli and SCL were calculated by multiplying densities with their corresponding reference space volume. The mean linear intercept (Lm) as an estimate for the size of acinar air spaces was calculated using the ratio of total airspace volume to the total epithelial surface area, multiplied by four. 32
Number and Exclusion of Animals and Blinding of Investigator
Before exclusion of individual animals, the number of pups per group was n=9 for CO, n=10 for NOX, and n=10 for HYX. Four animals were excluded from the study, namely HYX-3 and CO-6 with large areas of collapsed capillaries that could not be used for analysis, as well as NOX-1 and CO-2 with more than 5% volume density of atelectatic regions related to the overall parenchyma volume. We set this cutoff value of 5% a priori for all probes to ensure reliability of our acquired data. This led to a final number of animals of n=7 for CO, n=9 for NOX, and n=9 for HYX for statistical analysis. The number of animals used in this study was determined by prestudy power calculations based on a previous publication using stereology in this rabbit model. 13 During the volume estimation and sampling, as well as during the stereological analysis, the investigators were blinded to the group identity of the samples.
Statistical Analysis
Data are presented in scatter plots as means. Differences between the pairs CO/NOX and NOX/HYX were each evaluated using Wilcoxon Mann–Whitney rank sum tests. Results were considered significant if p≤0.05. Analysis was conducted in R version 3.5.2, 33 and plots were produced using the packages ggplot2, 34 ggpubr, 35 and ggsignif. 36
Results
Figure 1 shows representative LM images of the investigated groups. All groups showed well-fixed lungs, with opened septal capillaries containing only a few red blood cells. Thus, with the exception of three animals, where the lungs contained too many collapsed capillaries or alveoli, all lungs could be analyzed. Qualitatively, it was hard to observe differences between the lungs of the different groups. No clear differences in alveolar or capillary size or numerical density were observed, but secondary septal crests were much more frequent in the term control than in the preterm groups, indicating a higher degree of alveolarization.
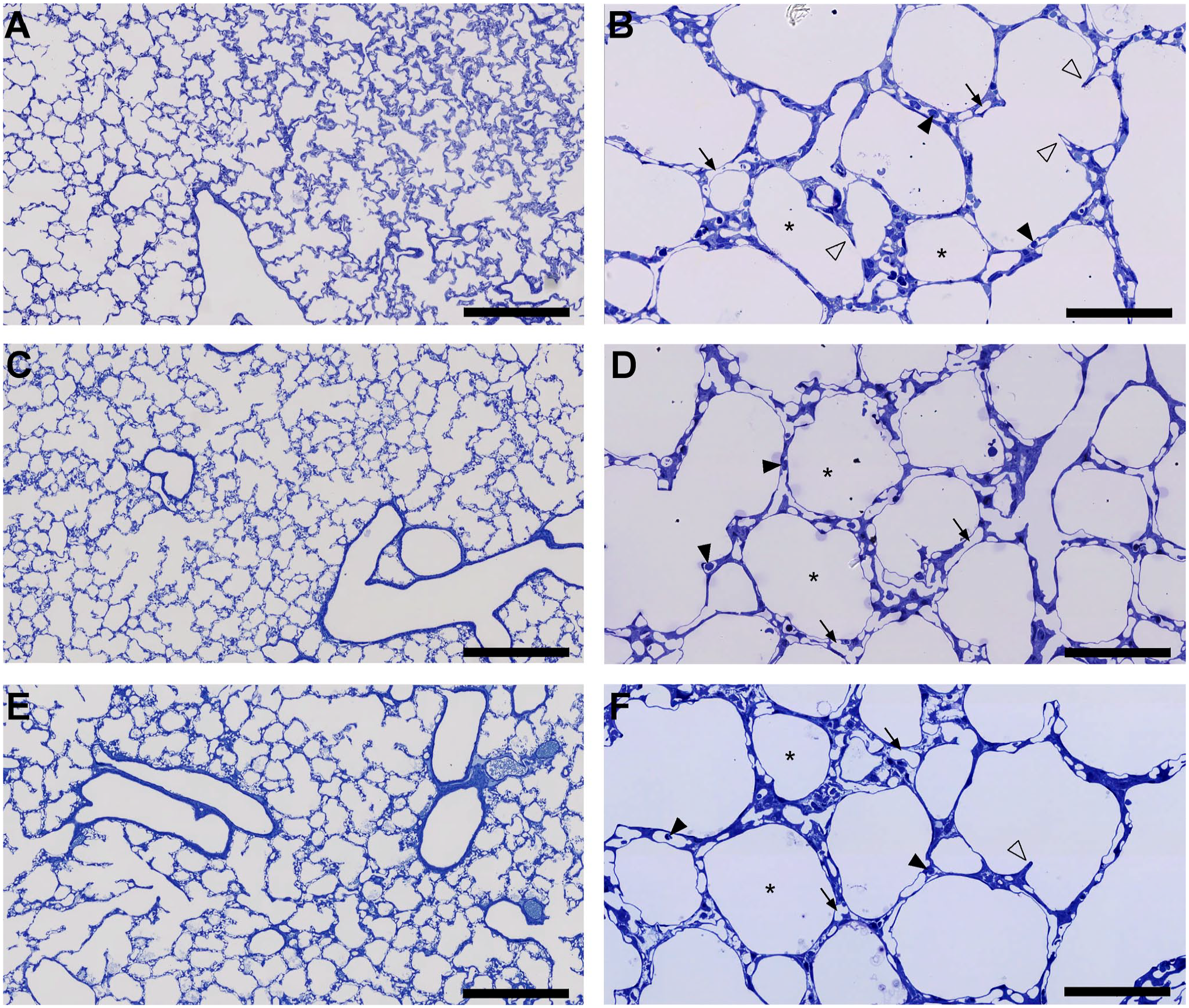
Histopathology of alveolar and septal structures in term and normoxia- and hyperoxia-exposed preterm rabbit lungs.
All acquired stereological data are summarized in Table 1. Animals of all groups were euthanized at the same age in days after conception (CO: 31-day gestational age + 4 days; NOX, HYX: 28-day gestational age + 7 days). At this point, survival was reduced in both preterm groups (CO 100%, NOX 77.8%, HYX 88%). At the end of experiments, the mean body weight in preterm pups under normoxia was reduced by 30.9%, and the mean lung volume reduced by 37.6% compared with the CO (Fig. 2A and B). Reduction in lung volume was primarily due to reduced parenchymal volume (Fig. 2C), along with an equal reduction of its components—ductal/alveolar airspace and septal volume (Table 1). Nonparenchymal volume (conducting airways, vasculature, peribronchiolar and perivascular tissues, pleura) was not significantly affected by prematurity (Fig. 2D). Between the preterm groups NOX and HYX, there was no statistically significant difference in mean body weight (p=0.69) or lung volume (p=0.135) at the end of the experiments (Fig. 2A and B). Lung volumes of parenchymal and nonparenchymal compartments remained unchanged by hyperoxia (Fig. 2C and D).
Acquired Lung Parameters of CO, NOX, and HYX.
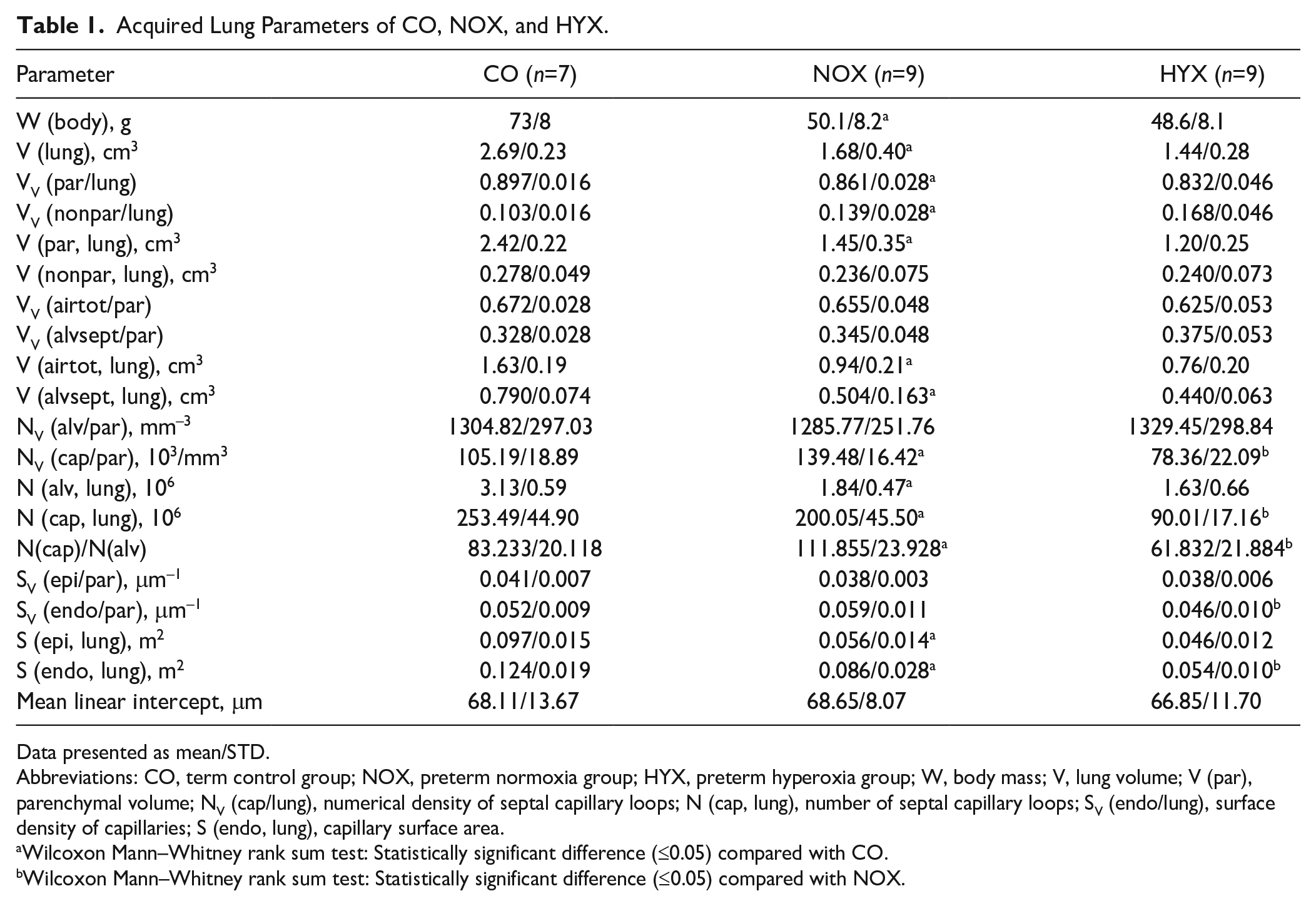
Data presented as mean/STD.
Abbreviations: CO, term control group; NOX, preterm normoxia group; HYX, preterm hyperoxia group; W, body mass; V, lung volume; V (par), parenchymal volume; NV (cap/lung), numerical density of septal capillary loops; N (cap, lung), number of septal capillary loops; SV (endo/lung), surface density of capillaries; S (endo, lung), capillary surface area.
Wilcoxon Mann–Whitney rank sum test: Statistically significant difference (≤0.05) compared with CO.
Wilcoxon Mann–Whitney rank sum test: Statistically significant difference (≤0.05) compared with NOX.
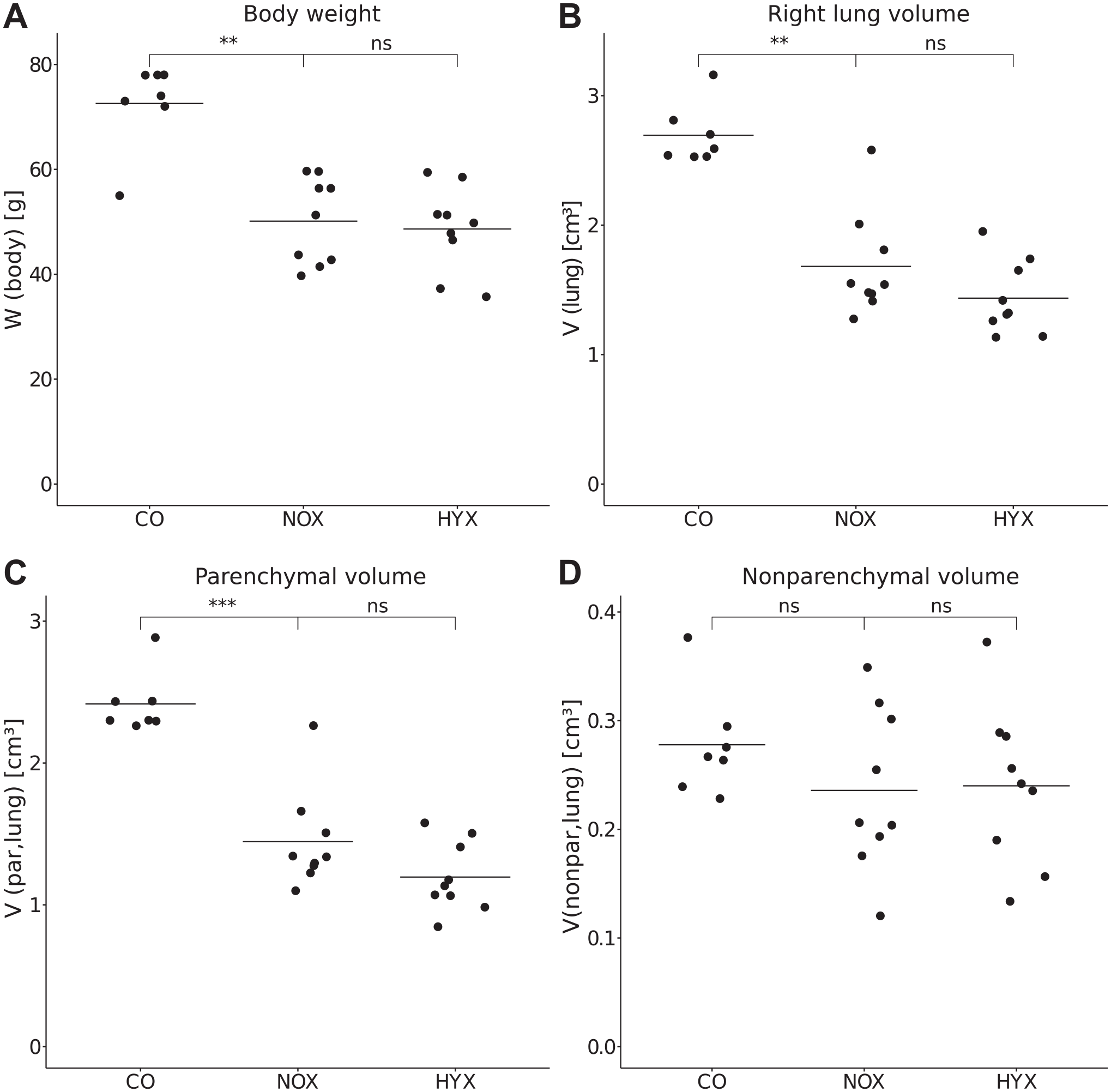
Effects of weight and volume.
Regarding the effect of prematurity alone, we compared term controls with preterm normoxic rabbits. The stereological data revealed a highly significant decrease by 41% in the total alveolar number in the NOX compared with CO (Fig. 3A). The NOX also showed a statistically significant 42.9% decrease in total epithelial surface area of alveoli (Fig. 3B). Regarding the additional effect of hyperoxia, we compared preterm normoxic and hyperoxic rabbits. We did not detect a statistically significant difference in either alveolar number or epithelial surface area between the NOX and HYX (p=0.2973 for both) (Fig. 3A and B). Numerical and surface densities of alveoli remained unaffected by both premature birth and postnatal hyperoxia (Fig. 3C and D). There was no statistically significant difference in Lm between the groups (p(CO/NOX)=0.8371, p(NOX/HYX)=1) (Table 1).
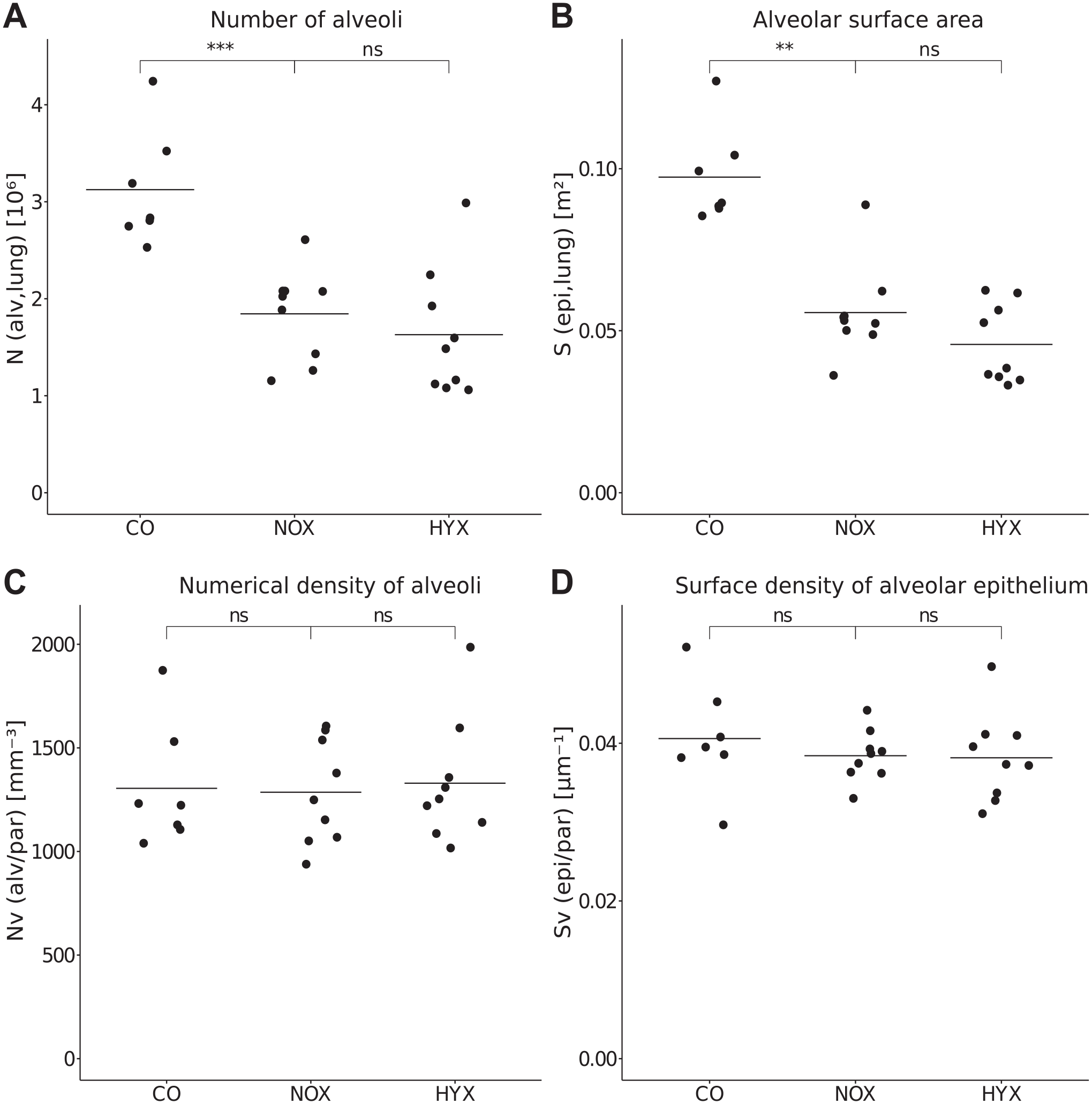
Stereological analysis of alveolar characteristics.
Both prematurity and hyperoxia led to a significant decrease in total number of SCL and endothelial surface area of capillaries. In detail, prematurity decreased the mean SCL number by 21.1% (CO vs NOX), and hyperoxia led to a decrease in SCL number by 55% (NOX vs HYX) (Fig. 4). The endothelial surface area was 31% lower in NOX than that in CO and 37.5% lower in HYX than that in NOX (Fig. 4B). The average numerical density of SCL in NOX was higher than that of CO, while hyperoxia led to a lower numerical density in HYX (Fig. 4C). Capillary surface densities were not significantly different between CO and NOX (p=0.4079) but significantly lower in HYX (Fig. 4D). The ratio between the numbers of SCL and alveoli (N(cap)/N(alv)) was calculated to compare the processes of alveolarization and microvascular development. The ratio was higher in NOX than that in CO and lower in HYX than that in NOX (Fig. 4E).
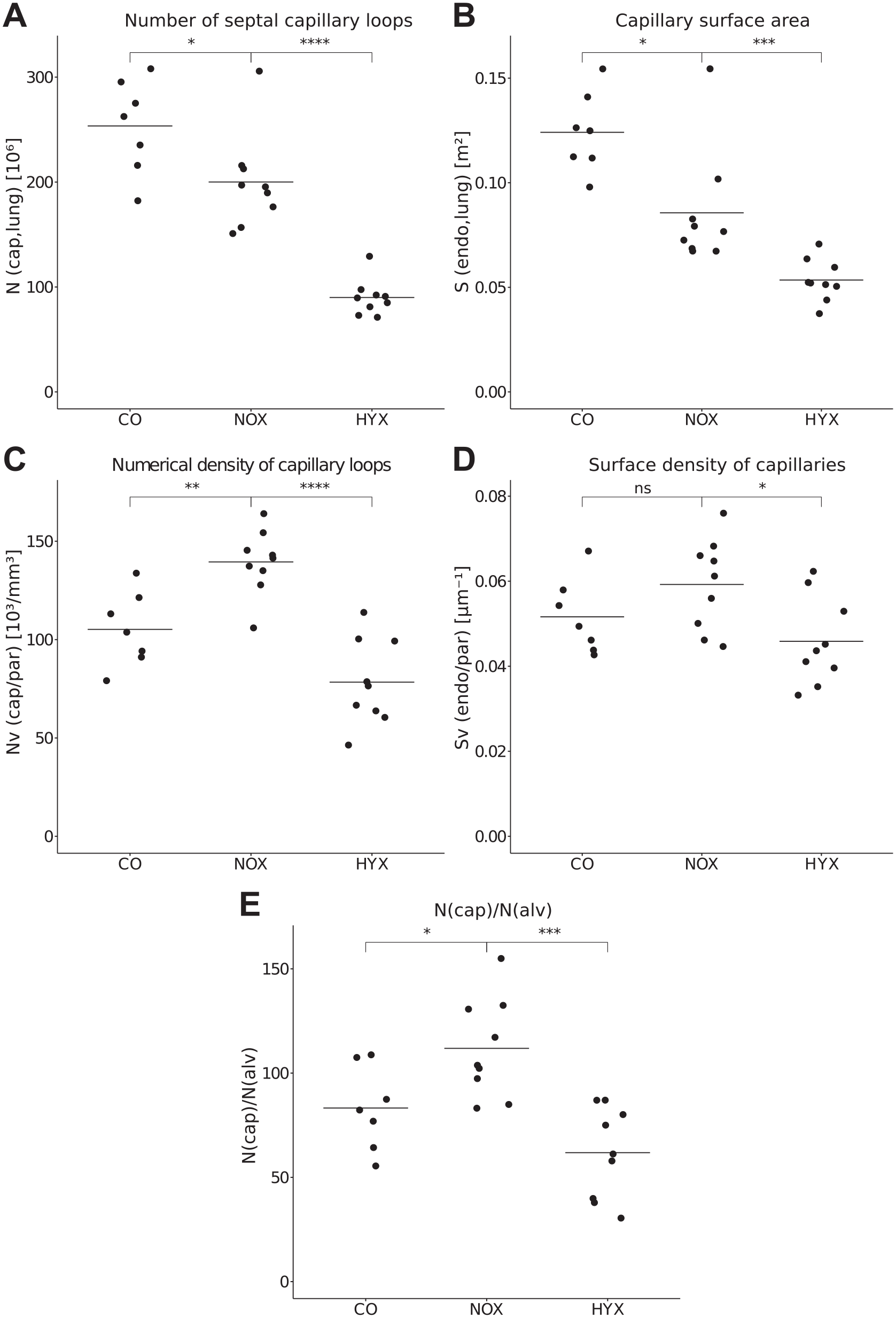
Stereological analysis of microvascular characteristics.
Discussion
In this study, we investigated the effects of preterm birth and hyperoxia on alveolar and microvascular development of the lung in a rabbit BPD model. We used design-based stereological methods to characterize volume, surface area, and number of alveoli and septal capillaries. Design-based stereology is the gold standard of lung morphometry providing unbiased or at least minimally biased data at high precision and is superior to other morphometric approaches like radial alveolar counts and capillary density per high-power field. 37
The present study demonstrated that prematurity and hyperoxia had distinct and diverse effects in alveolar and vascular development. Between the term and preterm normoxia-exposed pups, a negative effect of preterm birth on both alveoli and capillaries was detected, but the alveolar decrease was more pronounced. Importantly, preterm pups were smaller and had lower lung volumes. The decrease in lung volume was mainly in the parenchymal compartment. Interestingly, the decrease in total number of alveoli was proportional to the decrease in total alveolar surface area and parenchymal volume (around 40–43%). It can be speculated that there is a dependency between total number of alveoli and total lung volume. The lack of statistically significant differences in Lm between groups suggests that the proportions of individual alveoli were not affected by the developmental impairments of prematurity and hyperoxia in this set of experiments. Comparing normoxia- and hyperoxia-exposed preterm pups, there were differences neither in lung volumes nor in alveoli number and surface area in the present study. Previous investigations in rabbits showed the same close relationship between the lung volume and alveolar proportions. Salaets et al. 8 found a proportional decrease in body weight, lung volume, and alveolar surface area in preterm pups compared with term control while Lm did not differ. In contrast to the current study, Mühlfeld et al. 13 observed a proportional decrease in lung volume, alveolar surface area, and alveolar number in hyperoxia-exposed preterm pups compared with normoxia control while Lm did not differ. In healthy human lungs, a linear correlation of lung volume and number of alveoli has been described. 25 Moreover, it has been proposed that alveolar development and total number may be limited by the lung volume expansion rate. 38
Changes of the ACN had no correlation to lung volumes. Both the number of SCL and the endothelial surface area were lower in NOX than those in term control, although not to an equal extent as the reduction of parenchymal volume. Also, the surface area decreased more strongly than the absolute number of SCL. Exposure to hyperoxia had a more pronounced effect on the ACN than prematurity, without altering the lung volumes. In contrast to the effect of prematurity, hyperoxia lowered the total number of SCL to a higher degree than the endothelial surface area. There seems to be no clear correlation between the number of capillary loops and the total endothelial surface area. This could be due to different structural changes in the microvascular system in prematurity versus hyperoxia.
Preterm rabbits at day 28 are born in the saccular stage of lung development and forced to breathe with saccules and develop the first alveoli while already breathing. This is physiological for rodents but unnatural for both rabbits and humans.11,15 In humans, the saccular stage begins around the 26th gestational week, and alveolarization starts around week 36. In rabbits, the last 2 days of prenatal development (day 30 and 31) are known to comprise what is known as “bulk alveolarization,” the first phase of massive secondary septa formation.39,40 This crucial developmental process is largely completed at term birth, giving the newborn an initial number of alveoli to breathe with. Disturbance of bulk alveolarization by preterm birth could impede proper increase in alveolar numbers (and surface area) and, therefore, limit volume expansion of the lung as observed in our preterm groups. In addition, prematurity per se means relative degrees of supraphysiological oxygen concentration compared to the natural environment at 28 days of gestation, i.e., in utero. 8 Oxygen is toxic for endothelial cells, 18 and vascular endothelial growth factor expression has been repeatedly reported to be reduced in hyperoxia experimental groups10,41 and in the lungs of deceased human infants with BPD,17,18 along with other signaling components important for endothelial homeostasis and angiogenesis.10,42 Mild relative hyperoxia and other potential factors introduced by prematurity (e.g., tendencies for inflammatory processes)43,44 could be sufficient to cause a maximal disturbance in alveolarization and a significant impairment of microvascular development. The addition of hyperoxia might not have further effects on the already arrested alveolarization but poses an extra threat for capillary development and maturation. Artificially high oxygen levels like 95% in our HYX could lead to a maximal, perhaps unrealistic, disturbance of the microvasculature.
With 41% less alveoli but only 21% less SCL, prematurity led to more capillary loops per alveolus in NOX than in CO. Contrarily, without demise in alveoli number, the addition of hyperoxia produced 55% less SCL, leading to much fewer capillary loops per alveolus (Fig. 4). Epithelial cells of the ACN are known to induce neo-alveologenesis in regenerating lungs45,46 and influence myofibroblasts, which are important for septation. 47 With a higher ratio of SCL per alveolus, it seems that the preterm lung under normoxic conditions retains good potential for regeneration and catch-up. A more severe damage to the ACN under extreme hyperoxic conditions (>95% O2) or a second hit may limit the potential for regenerative alveolar processes.
The main limitation of this study is that only a single time point in postnatal development was analyzed, and therefore, no conclusions about the temporal effects can be drawn. Although we did not observe changes in the alveolar compartment through hyperoxia, our time point may be too early in the development of BPD-like impairment, and further distortion of alveolar structures might follow. In a hyperoxia-exposed mouse model, there were no epithelial surface area changes present at day 7 but started to be significant from day 14 onwards, whereas changes in endothelial surface area were present at day 7. 21 In a rabbit model, hyperoxia was reported to show first significant effects from day 7. 12 It would be interesting to learn if changes to the alveolar compartment occurred after day 7. This would fit the “vascular hypothesis” that alveolar impairment follows microvascular changes.
On the other hand, changes might have been present between birth and day 7 or 4 but succeeded to compensate during ongoing postnatal development. Salaets et al. 8 noticed a partial catch-up of preterm rabbits in body weight after 7/4 days and reported the initially altered alveolar structure had returned to a comparable state as in term-delivered pups although without improvement in lung function.
In the present study, we did not distinguish between female and male rabbits but investigated mixed groups. There are indications, at least for mice, that male animals have more alveoli in total because of higher lung volumes.48,49 Studies in rabbits could not detect a sex difference, 8 yet they were not powered for this kind of comparison. Male sex is an independent predictor for BPD 50 and may influence response to hyperoxia. 37 However, a strong effect as presented in this study would be unlikely to originate entirely from different ratios of female and male pups in different experimental groups.
In conclusion, the present study has demonstrated with methods of design-based stereology that prematurity and hyperoxia have differing effects in rabbits. Prematurity had a negative effect on capillaries and, more pronounced, on alveoli and parenchymal lung volume. Hyperoxia had no further effect on alveoli but affected the microvascular compartment more severely. Changes in the ACN must involve an underlying mechanism that alters its spatial arrangement and connectivity. The specific changes remain speculation until further 3D investigation has been performed. To further investigate the “vascular hypothesis” of BPD, it is crucial to differentiate the effects of prematurity and hyperoxia and avoid their overlay, potentially masking or amplifying one another. These results may help to better understand the pathophysiology of BPD, and new therapeutic approaches may address the adverse effects of high oxygen ventilation to support alveolar capillary development.
Footnotes
Acknowledgements
The authors thank Susanne Faßbender and Rita Lichatz for excellent technical assistance. This work is part of the M.D. thesis of Giacomo Rößler.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
GR and CM contributed to the conception and design of the work, as well as to the acquisition, analysis, and interpretation of data, and drafted the manuscript. JL, YR, TS, AG, and JT contributed to the acquisition and analysis of data and revised the manuscript for important intellectual content. All authors approved the final version of the manuscript and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Bundesministerium für Bildung und Forschung (BMBF) via the German Center for Lung Research (DZL) and the Deutsche Forschungsgemeinschaft (MU 3118/8-1). The study was in part supported by The Research Foundation—Flanders (FWO Flanders) (grant G0C4419N) and KU Leuven (C2 grant: C24/18/101).