Abstract
Immunohistochemical distribution and cellular localization of neurotrophins was investigated in adult monkey brains using antisera against nerve growth factor (NGF), brain-derived neurotrophic factor (BDNF), neurotrophin-3 (NT-3), and neurotrophin-4 (NT-4). Western blot analysis showed that each antibody specifically recognized appropriate bands of ∼14.7 kDa, 14.2 kDa, 13.6 kDa, and 14.5 kDa, for NGF, BDNF, NT-3, and NT-4, respectively. These positions coincided with the molecular masses of the neurotrophins studied. Furthermore, sections exposed to primary antiserum preadsorbed with full-length NGF, BDNF, NT-3, and NT-4 exhibited no detectable immunoreactivity, demonstrating specificities of the antibodies against the tissues prepared from rhesus monkeys. The study provided a systematic report on the distribution of NGF, BDNF, NT-3, and NT-4 in the monkey brain. Varying intensity of immunostaining was observed in the somata and processes of a wide variety of neurons and glial cells in the cerebrum, cerebellum, hippocampus, and other regions of the brain. Neurons in some regions such as the cerebral cortex and the hippocampus, which stained for neurotrophins, also expressed neurotrophic factor mRNA. In some other brain regions, there was discrepancy of protein distribution and mRNA expression reported previously, indicating a retrograde or anterograde action mode of neurotrophins. Results of this study provide a morphological basis for the elucidation of the roles of NGF, BDNF, NT-3, and NT-4 in adult primate brains.
Keywords
N
Neurotrophins (Distefano et al. 1992; Mufson et al. 1994,1996) are generally known to be target derived and are retrogradely transported from production sites to the responsive neuronal cell bodies where they bind to cell surface receptors to initiate a series of cellular reactions for biological functions. The action mode has been demonstrated in the peripheral nervous system (PNS) where NGF is transported from the periphery to support the survival and functions of sensory and sympathetic neurons (Levi-Montalcini and Angeletti 1968; Friedman et al. 1998; Bibel et al. 1999). Also, in the central nervous system (CNS), NGF has been shown to be retrogradely transported from the hippocampus to the large cholinergic neurons in the medial septum and the nucleus of the diagonal band of Broca and from the neocortex to large cholinergic neurons in the nucleus basalis in the rat (Seiler and Schwab 1984). Transport modes of BDNF and NT-3 are similar in both the PNS and CNS (Davies and Wright 1995). Furthermore, mRNAs for NGF (Ayer-LeLievre et al. 1988; Ernfors et al. 1990b), BDNF (Ernfors et al. 1990b; Hofer et al. 1990; Wetmore et al. 1990), NT-3 (Ernfors et al. 1990b; Friedman et al. 1991), and NT-4 (Timmusk et al. 1993) have been detected in the hippocampus and neocortex, which are target regions of several known neurotrophin-dependent populations including basal forebrain cholinergic neurons, dopamninergic neurons of mesencephalon, and noradrenergic neurons in the locus ceruleus. The above studies all support a target-derived mode of action of the neurotrophins.
Aside from the traditional target-derived action mode, studies of the sites of neurotrophic factor synthesis sustain the possibility of alternative action modes of neurotrophic factors (Davies and Wright 1995; Davies 1996). Whether in the CNS or PNS, neurotrophins and their receptors can coexpress in individual neurons of specific subpoplations (Kokaia et al. 1993; Miranda et al. 1993). Furthermore, some neuronal populations and their target cells may express NGF, BDNF, NT-3, or NT-4 simultaneously (Scarisbrick et al. 1993), suggesting the possibility of autocrine and paracrine actions and other local influences. Therefore, knowledge of the regional distribution and cellular localization of neurotrophins has become increasingly important as this contributes to the understanding of their function and mechanism.
Although there are many reports on neurotrophins, these data are derived mostly from in vitro studies (Danzer et al. 2002; Martin et al. 2002; Seifer et al. 2002; Takano et al. 2002; Ghinelli et al. 2003; Price et al. 2005). From rodents to primates and in humans, in situ hybridization has provided useful information regarding the endogenous expression of mRNAs (Shelton and Reichardt 1986; Huntley et al. 1992; Hayashi et al. 1993,1997; Hashimoto et al. 1999) and tyrosine kinase receptors (Schatteman et al. 1988; Mufson et al. 1991; Zhou and Rush 1993; Sandell et al. 1998; Hashimoto et al. 1999; Zhou and Yang 1999; Yamuy et al. 2000) for members of the neurotrophin family. However, immunohistochemical (IHC) localization of neurotrophin proteins is important in delineating potential sites and modes of action. Until now, in vivo studies to explore the expression of neurotrophin proteins, their distribution, and their functions in the CNS are limited to rats and mice (Shelton and Reichardt 1986; Conner and Varon 1992; Zhou and Rush 1994a; Dugich-Djordjevic et al. 1995; Kawamoto et al. 1996; Conner et al. 1997; Yan et al. 1997b; Zhou and Yang 1999). Although a few studies have reported on the distribution of single neurotrophic factors in primate brains (Kawamoto et al. 1999; Takuma et al. 2002), systemic distribution of neurotropins in primate brains has not been reported. We therefore undertook this study to investigate by IHC the distribution of NGF, BDNF, NT-3, and NT-4 in adult monkey brains.
Materials and Methods
Characterization of Antibodies
Antisera directed against NT-3 were gifts from Xin-Fu Zhou (Centre for Neuroscience, Department of Physiology, Flinders University of South Australia, Adelaide, Australia) and have been reported earlier (Zhou and Rush 1994b). Antibodies were affinity purified on a peptide-Affi-Gel 10 Column (Bio-Rad; Hercules, CA) and shown to be specific by recognition of recombinant human and endogenous rat NT-3 but not mouse NGF or recombinant BDNF by immunoblot analysis. Anti-NGF (Cat. #AB1526P; Chemicon, Temecula, CA), anti-BDNF (Cat. #AB1779; Chemicon), and anti-NT-4 (Cat. #AB1781; Chemicon) polyclonal antibodies used in this experiment have been extensively characterized in rat and mice. Antisera were specific for the appropriate neurotrophin and did not crossreact with other neurotrophins. As little as 10 ng of each neurotrophin can be visualized using these antisera. Control of immunostaining specificity was performed by omitting the primary antibody or antibodies preadsorbed with the appropriate neurotrophins. These controls did not exhibit any specific immunostaining. In this study, Western blot analysis also gives further support for antibody specificity in monkey.
Animals and Tissue Preparation
Seven adult male rhesus monkeys (6-9 years old) weighing ∼5-6.5 kg were obtained from the Animal Experimental Center of Kunming Medical College and were the subjects for the study. They were housed individually in clear steel cages under standard conditions of humidity and temperature with 12-hr light/dark cycle and ad libitum access to food and water as approved by the Association for Assessment and Accreditation of Laboratory Animal Care International. All monkeys were deeply anesthetized by IM injection of chloramines (0.25 ml/kg; Sigma Chemical Co., St Louis, MO). Experimental procedures were done in accordance with the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health (1996). Every effort was made to minimize both suffering and number of animals used.
For Western blot analysis, skulls of two monkeys were rapidly opened after anesthesia, and dissection for fresh samples (∼0.5 cm2) was performed on ice. Dissected samples were stored at −80C until use.
For IHC study, animals were perfused through the ascending aorta with 500 ml of normal saline followed by 1 liter of Zamboni's fixative: 2% paraformaldehyde + 15% saturated picric acid. After perfusion, brains were quickly removed and stored at −20C until use.
Western Blotting
Fresh monkey brain samples were lysed on ice for 10 min in a lysis buffer (400 mM KCl, 50 mM HEPES, 1.5 mM EDTA, 20% glycerol, 0.5% NP4O, 20 mM NaF, 10 mM Na2 molybdate, 10 μM Na3 ortho, and 1 mM dithiothreitol) containing a mixture of protease inhibitors (1 mM PMSF, 0.2 mg/ml bacitracin, 0.2 mg/ml aprotin, 5 μg/ml leupeptin, and 5 μg/ml pepstatin A) (Peiris et al. 2004). Tissues were then homogenized and centrifuged at 1000 × g for 30 min. Protein concentrations were determined by the Bradford method. The supernatant was boiled for 8 min in 2X SDS sample buffer (62.5 M Tris-HCl, 10% glycerol, 2% SDS, 0.05% bromphenol blue, 1% β-mercaptoethanol) and centrifuged at 1000 × g for 1 min at 4C. Samples were run on 15% SDS polyacrylamide gel electrophoresis. Gel was transferred onto nitrocellulose membrane using a semi-dry system at 24 V for 435 min. Membranes were stained by Ponceau S, and nonspecific bindings were blocked by 0.02 mM PBS containing 5% skim milk and 0.05% Tween-20 for 1 hr, followed by overnight incubation with affinity-purified rabbit polyclonal antibody for NGF (1:2000 dilution), BDNF (1:500 dilution), NT-3 (1:400 dilution), and NT-4 (1:500 dilution) in the same additive. After three 10-min PBS washes, the membrane was incubated with a horseradish peroxidase-conjugated goat anti-rabbit immunoglobulin G (HRP, 1:400 dilution; Vector Laboratories, Burlingame, CA) for 1 hr. Following a 30-min rinse with buffer, immunoreactive bands were dried and visualized by enhanced chemiluminescence in the dark for 30 min, using a developing solution (mixture of contents in tube 1:1 M Tris-HCl, pH 8.0, 30% H2O2 and in tube 2:1 M Tris-HCl, pH 8.0, 90 mM p-coumaric acid, 250 mM luminol; Vector Laboratories).
IHC Procedure
Brain sections were transferred to 20% sucrosed 0.1 M PBS. When they sank to the bottom of the container, they were removed, frozen, and sectioned at 30-μm thickness in a freezing microtome (CM1900; Leica, Wetzlar, Germany), and every tenth section was processed for IHC demonstration of NGF, BDNF, NT-3, and NT-4.
Free-floating sections of monkey brains were washed three times, 5 min each, in 0.1 M PBS and were then incubated at room temperature in 0.2% hydrogen peroxide for 20 min to block the action of any endogenous peroxidase. This was followed by a 30-min immersion in PBS containing 0.3% Triton X-100 and 5% normal goat serum at 37C. Afterwards, sections were incubated for 48 hr at 4C in one of the primary antibody solutions (polyclonal neurotrophin antisera for NGF, BDNF, NT-3, or NT-4, diluted 1:1000) containing 2% normal goat serum and 0.3% Triton X-100. Following three washes in 0.1 M PBS for 5 min each, sections were incubated with secondary antibodies (biotinylated anti-rabbit IgG, 1:400 dilution; Vector Laboratories) for 1 hr at room temperature. After washing with 0.1 PBS three times for 15 min, sections were incubated with an avidin-biotin-peroxidase reagent (1:250 dilution, ABC Elite; Vector Laboratories), and the immunoreaction products were visualized by placing the sections in a staining solution containing 0.04% 3,3′-diaminobenzidine, 0.06% nickel sulfate, and 0.06% hydrogen peroxide for 10 min. They were mounted, dehydrated, coverslipped, and observed under a light microscope (Olympus; Tokyo, Japan).
Results
Specificity of the Antisera
Specificity of the antisera for the individual neurotrophins was initially tested by Western blot analysis. Western blot analysis showed that each antibody specifically recognized appropriate bands of ∼14.7 kDa, 14.2 kDa, 13.6 kDa, and 14.5 kDa, for NGF, BDNF, NT-3, and NT-4, respectively. These positions coincided with the molecular masses of the neurotrophins studied (Figure 1).
IHC control was also used to detect specificity of the antisera. Moderate to scarce labeling of NGF, BDNF, NT-3, and NT-4 was observed in the inferior olivary nucleus (Figures 2A, 2D, 2G, and 2J). This pattern of immunostaining could be eliminated by exclusion of the primary antibodies (Figures 2B, 2E, 2H, and 2K) or by incubation with the preabsorbed primary antibodies (Figures 2C, 2F, 2I, and 2L).
Immunoreactive Localization of Neurotrophins in Adult Monkey Brains
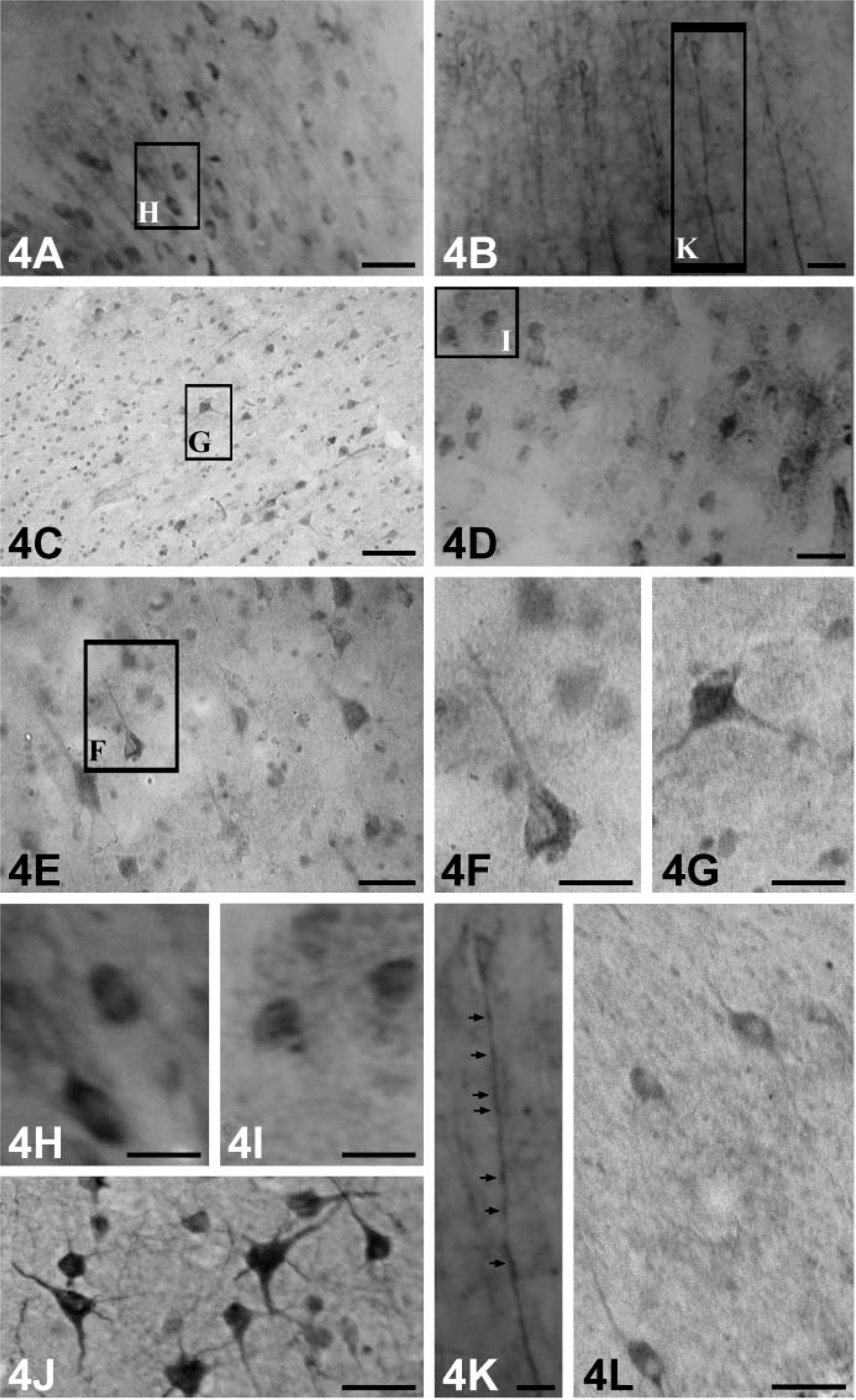
Cerebral Cortex. Within the gray matter, distributions of NGF-, BDNF-, NT-3-, and NT-4-immunopositive neurons were similar (Figure 3). They occurred in all layers of the cortical regions examined in this study except layer I. Intensely labeled neurons were scattered in layers III (external pyramidal layer) and V (internal pyramidal layer): a few strongly positive ones were also observed in layers II (external granular layer) and IV (internal granular layer) (Figures 4A-4E). Morphologically, they were mainly pyramidal neurons (Figure 4F), and most were large- and medium-sized cells. Additionally, the large pyramidal cells (Betz cells) were most intensely stained in the internal pyramidal layer of the motor cortex (Figure 4G). Immunoreaction products of NGF, BDNF, NT-3, and NT-4 were mainly observed in the perikarya of these neurons and seemed to be more abundant in the temporal (Figure 4J) and insular cortex (Figure 4L).
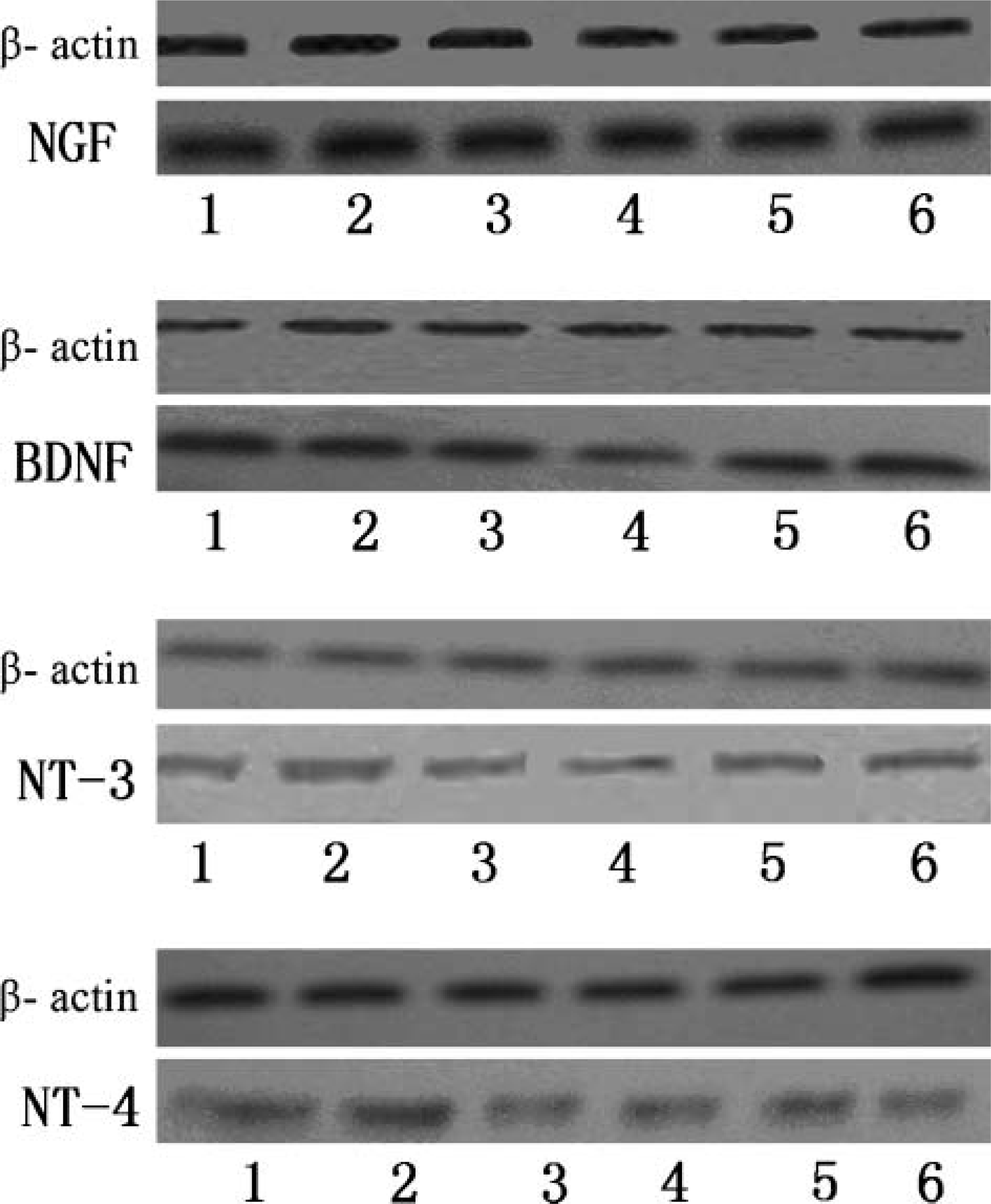
Autoradiograph of a blot developed using enhanced chemiluminescence to detect the molecules of NGF, BDNF, NT-3, and NT-4. Dilutions are as follows: anti-NGF (diluted 1:1000 in 0.02 mM PBS containing 5% skim milk, 0.05% Tween-20), anti-BDNF (1:1000), anti-NT-3 (1:1000), and anti-NT-4 (1:1000). Goat anti-rabbit immunoglobulin G (1:400 dilution) was used. Lane 1: cerebrum. Lane 2: midbrain. Lane 3: cerebellum. Lane 4: pons. Lane 5: hippocampus. Lane 6: medulla oblongata. Western blot analysis showed that each antibody specifically recognized appropriate bands of ∼14.7 kDa, 14.2 kDa, 13.6 kDa, and 14.5 kDa for NGF, BDNF, NT-3, and NT-4, respectively.

Inferior olivery nucleus was moderately to hardly stained by the anti-NGF
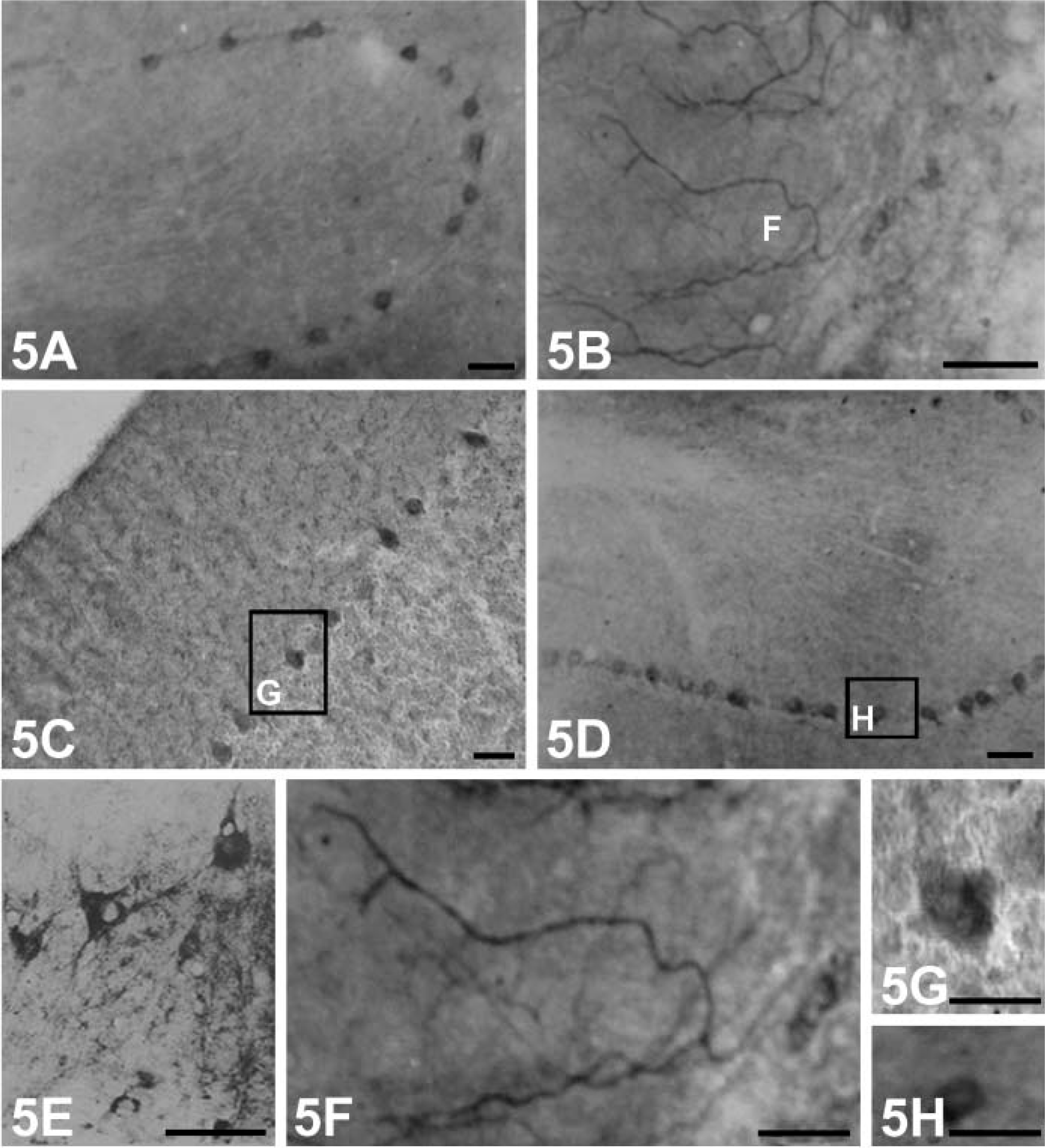
Compared with other neurotrophins, immunoreaction products of BDNF were more obvious in the processes of the stained neurons (Figure 4K). Robust positive products containing NGF or NT-4 accumulated mainly in the cytoplasm of the stained neurons (Figures 4H and 4I), and this could not be seen in BDNF-producing cells. None of the immunopositive neurons showed staining in their nuclei. When sections were incubated in preabsorbed NGF, BDNF, NT-3, and NT-4, respectively, immunoreactions of NGF, BDNF, NT-3, and NT-4 were substantially reduced (results not shown). Cerebellum. In the cerebellar cortex, immunoreaction products of NGF (Figure 5A), NT-3 (Figure 5C), and NT-4 (Figure 5D) showing varying staining intensities were present within the somata of Purkinje cells. They appeared as strongly stained dot-like structures covering the surfaces of the cell bodies and proximal processes of these cells (Figures 5G and 5H). No immunoreactivity of NGF, NT-3, and NT-4 could be found in the granule cell layer. Intriguingly, in our experiment, weak immunolabeling of BDNF was observed within only a few Purkinje cells. A stronger labeling was present in granular cells and in axon-like fibers in the molecular cell layer (Figure 5B). They were most likely sections of varicosities (Figure 5F). A few scattered immunopositive neuronal cell bodies and processes were present in the fastigial and dentate nucleus (Figure 5E). Preabsorbed antibodies failed to stain any tissues in the cerebellar cortex.
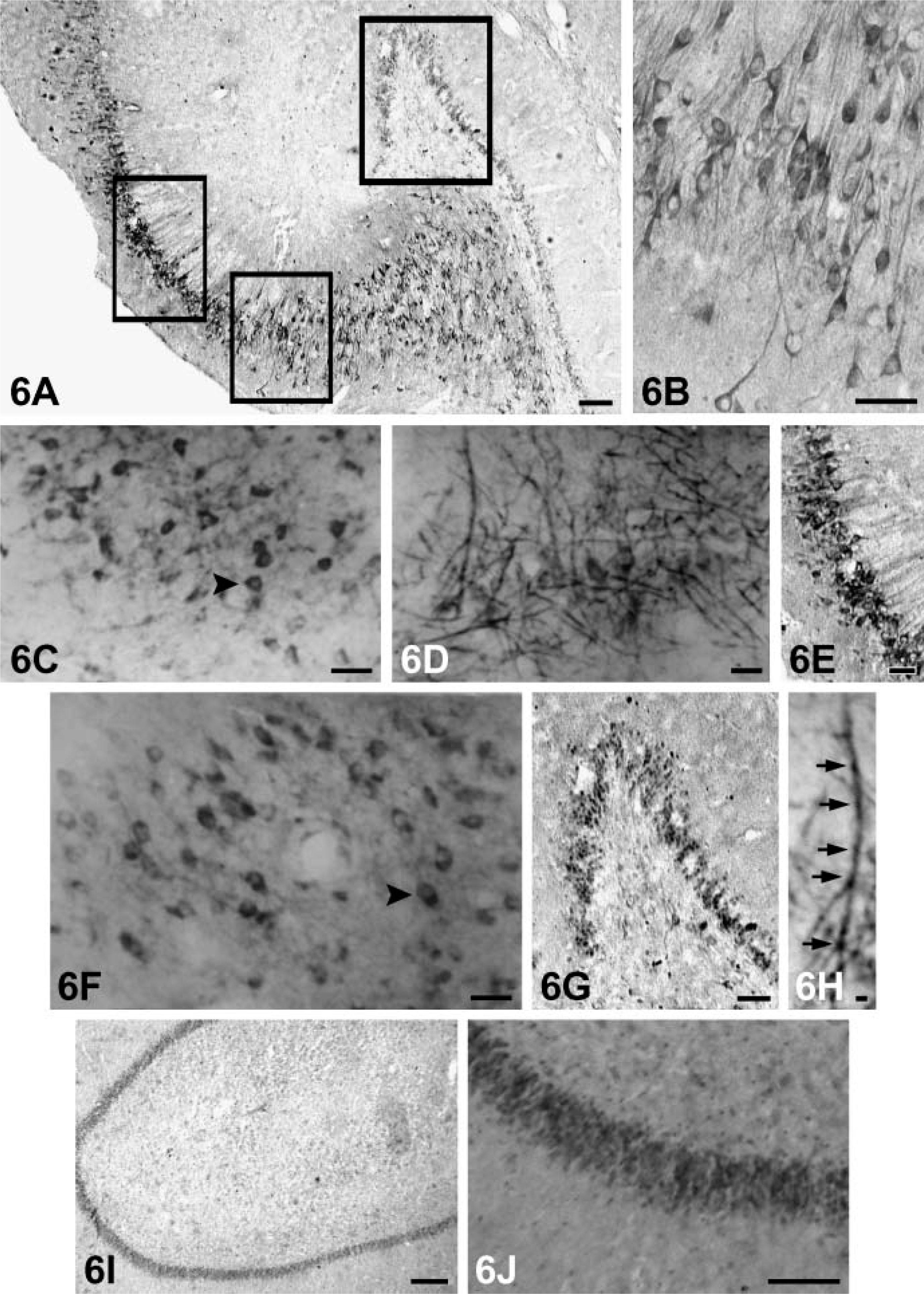
Hippocampus. Varying staining intensities of NGF, BDNF, NT-3, and NT-4 reaction products were observed in the cytoplasm and proximal dendrites of some pyramidal cells in some regions of subfields CA1-CA4, respectively. NGF-positive neurons with moderate staining were observed mainly in CA2-CA4 subfields; the cytoplasm of some granular cells in the dentate gyrus was also stained (Figures 6C, 6I, and 6J).
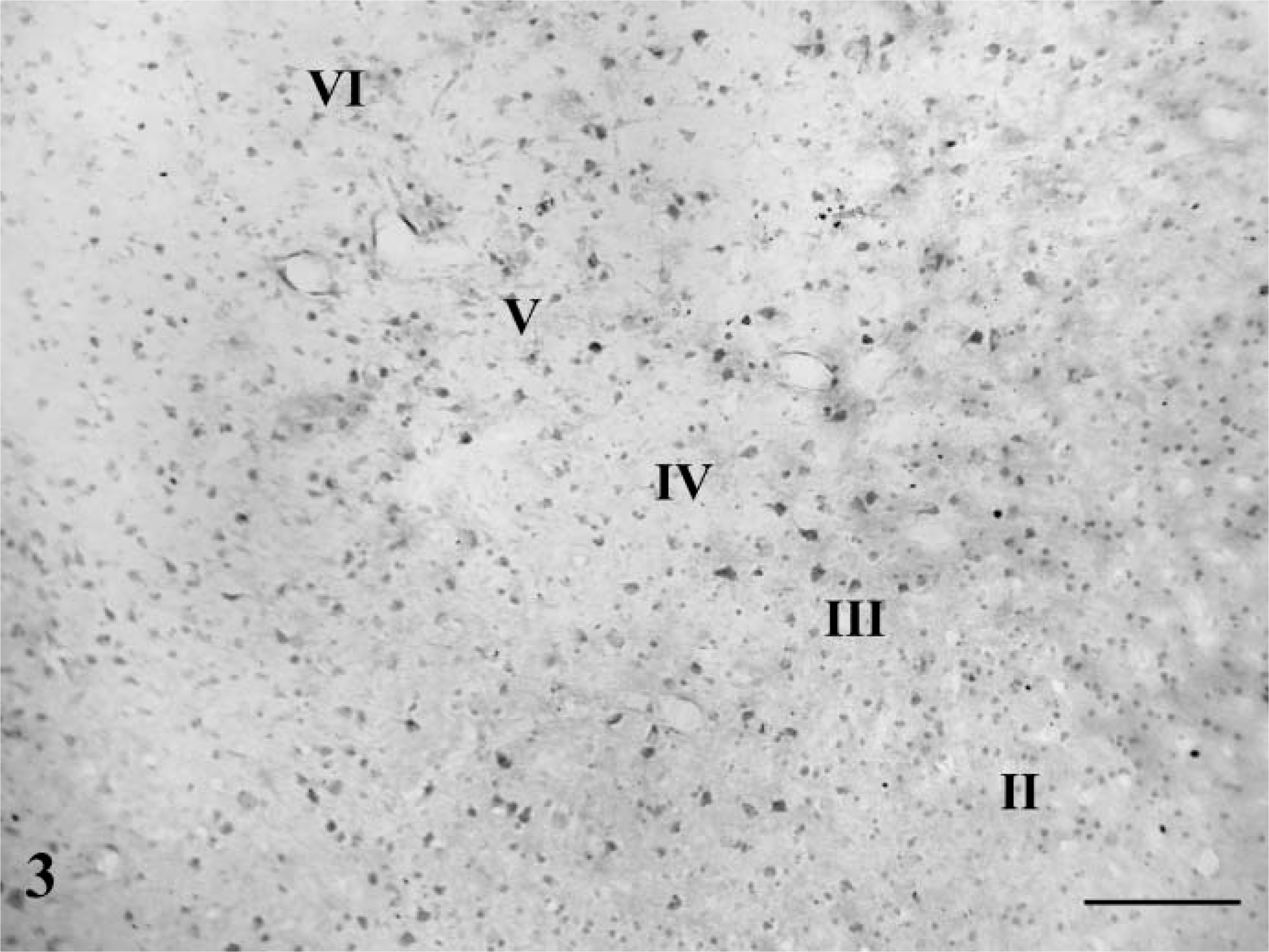
Low-power magnification of the temporal cortex showing the localization of NT-3 immunoreaction products. Intensely labeled neurons are scattered in layers III (external pyramidal layer) and V (internal pyramidal layer). A few strongly positive ones are also seen in layers II (external granular layer) and IV. Bar = 100 μm.
Strongly labeled BDNF immunoreactivity could be observed in the cytoplasm and proximal dendrites of the pyramidal neurons in subfields CA2-CA4. In addition, moderate BDNF-positive medium-sized and small granular cells and a dense plexus of fibers were observed in the polymorph layer and hilar region (Figure 6D). Occasionally, axon-like BDNF-positive fibers were also present in these fields (Figure 6E). Moderate NT-3-positive neurons were detected mainly in CA2 and CA3 regions (Figures 6A, 6B, and 6E), and immunopositive fibers corresponding to the mossy fibers were present in the granular cells and hilar region of the dentate gyrus (Figure 6G). Weak to moderately stained NT-4-positive cells were found throughout Ammon's horn, whereas CA1 and CA2 pyramidal neurons showed mild immunoreactivity (Figure 6F). Staining of BDNF and NT-3 was more obvious in the neuronal processes. Control of immunostaining specificity was performed by antibodies preadsorbed with purified recombinated human NGF, BDNF, NT-3, or NT-4. These controls did not exhibit any specific immunostaining in the hippocampus.
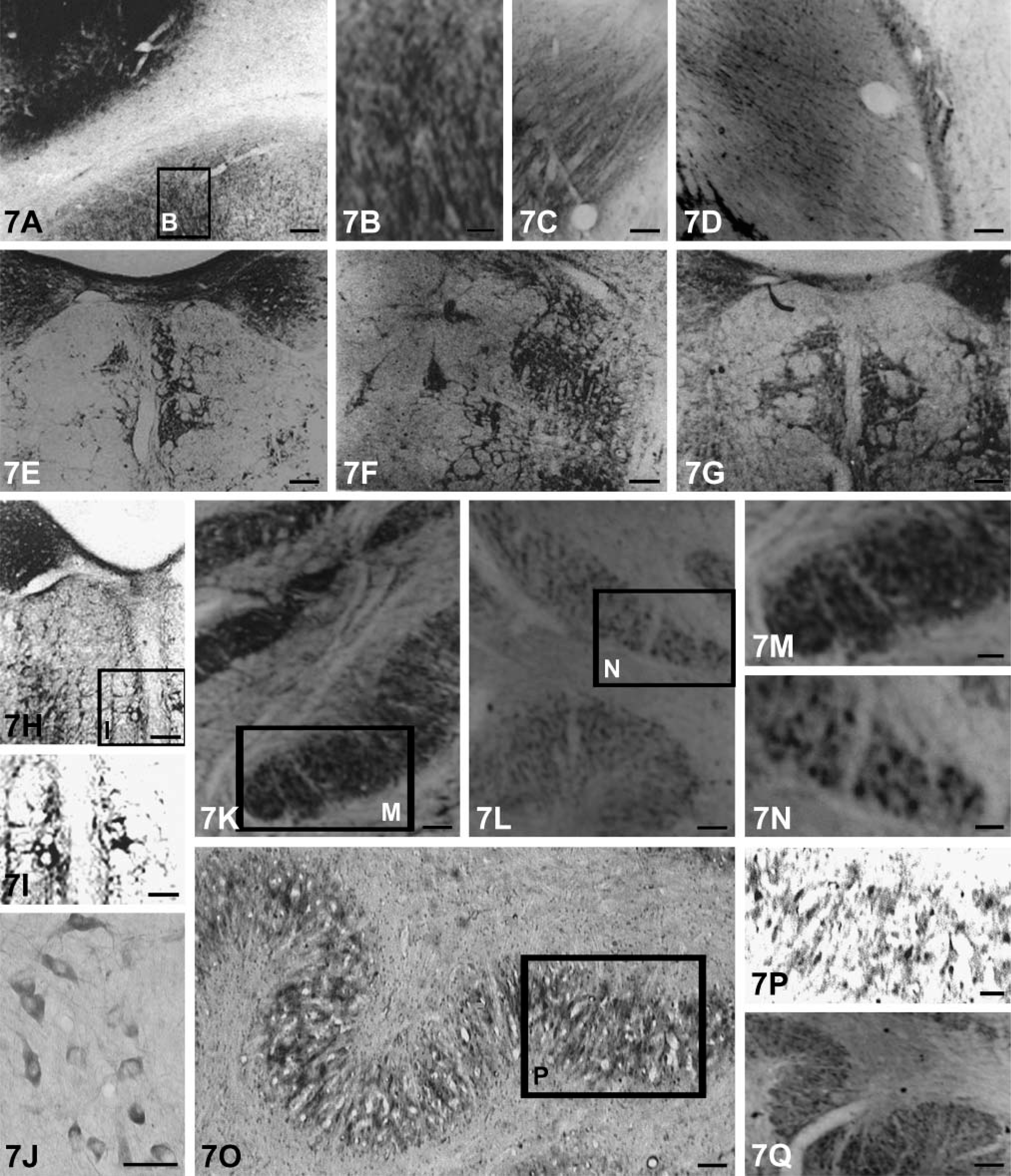
Basal Ganglia. In the basal ganglia, distribution of NGF-, BDNF-, NT-3-, and NT-4-immunopositive products were similar but the staining intensity differed somewhat. Immunoreactivities of NGF and BDNF were more robust than those of NT-3 and NT-4. A few neurons and numerous fibers that formed a meshwork showed moderate labeling in the lateral segment of the putamen (Figures 7A-7D). The globus pallidus did not display BDNF immunoreactivity. Patches of strongly intensive labeling of the four kinds of neurotrophins were clearly visible in the caudate nucleus (Figure 7B). There were no obvious differences in the immunolabeling of the four neurotrophins in all subdivisions of the amygdaloid nuclear complex. Pre-absorbed antibodies failed to stain any tissues of the basal ganglion.
Thalamus and Hypothalamus. NGF- and BDNF-immunopositive neurons were situated in several thalamic nuclei including the ventral anterior, ventral lateral, ventral medial, paraventricular, parafascicular, and centromedian nuclei, and the arcuate nucleus and lateral hypothalamic nuclei were also moderately stained. However, no immunoreactive signals of NT-3 and NT-4 were found in most nuclei of thalamus and hypothalamus.
Brain Stem and Other Regions. In addition to the above locations, NGF-, BDNF-, NT-3-, and NT-4-labeled neurons were also present in various nuclei of the brain stem (Table 1). Neurons devoid of immunoreactive reaction were intermingled with immunostained neurons in all explored structures. It was noted that the majority of the labeled neurons were motor neurons like those in the oculomotor nuclei, trochlear nuclei, etc., and the most intensively labeled neurons were located in the dorsal nucleus of the vagus (Figures 7E, 7G, and 7H), hypoglossal and vestibular nucleus (Figure 7F), facial nucleus (Figure 7I), spinal trigeminal nucleus (Figure 7J), and inferior olivary nucleus (Figures 7K-7Q).
Distribution of immunoreaction products of NGF
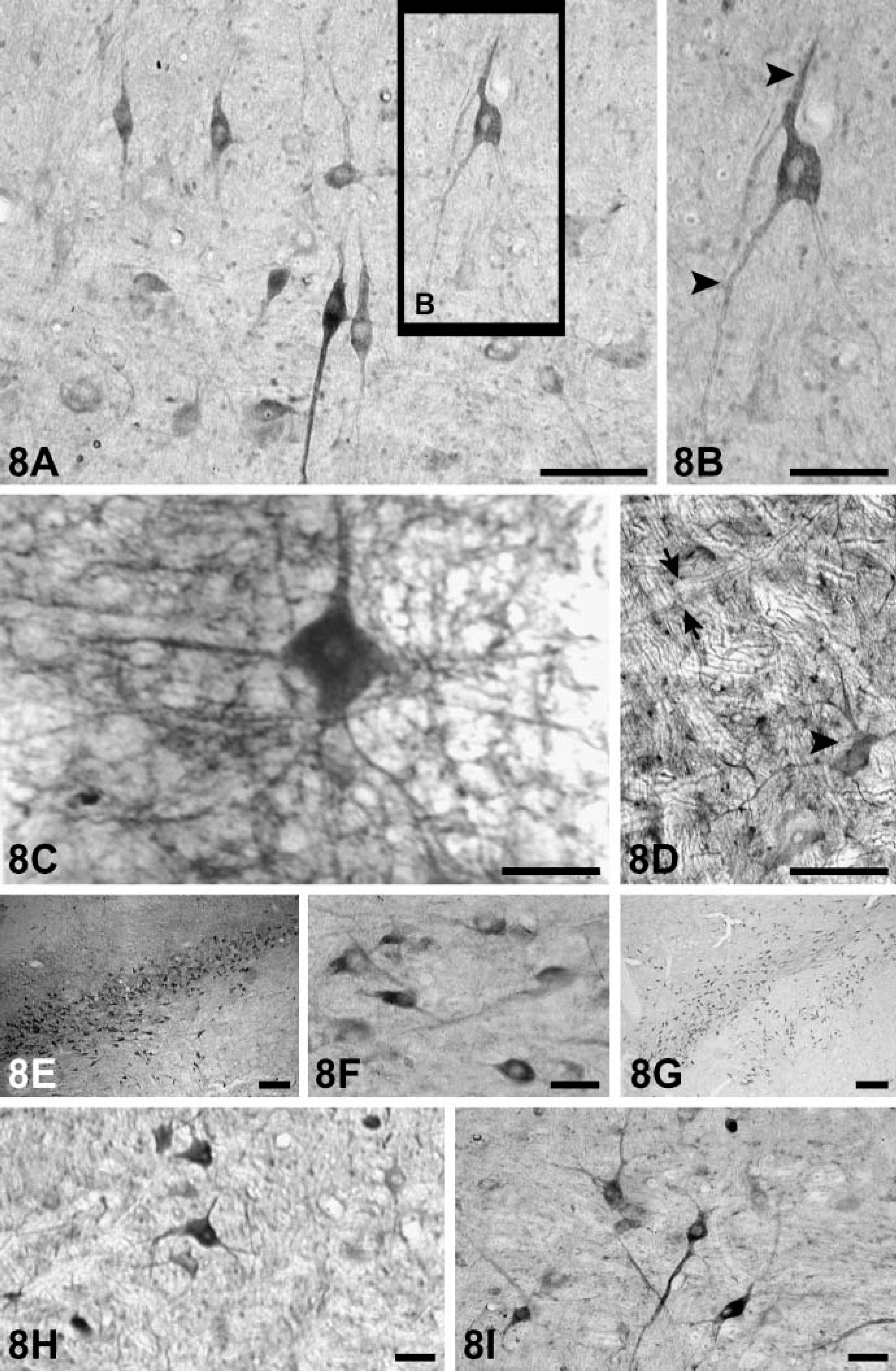
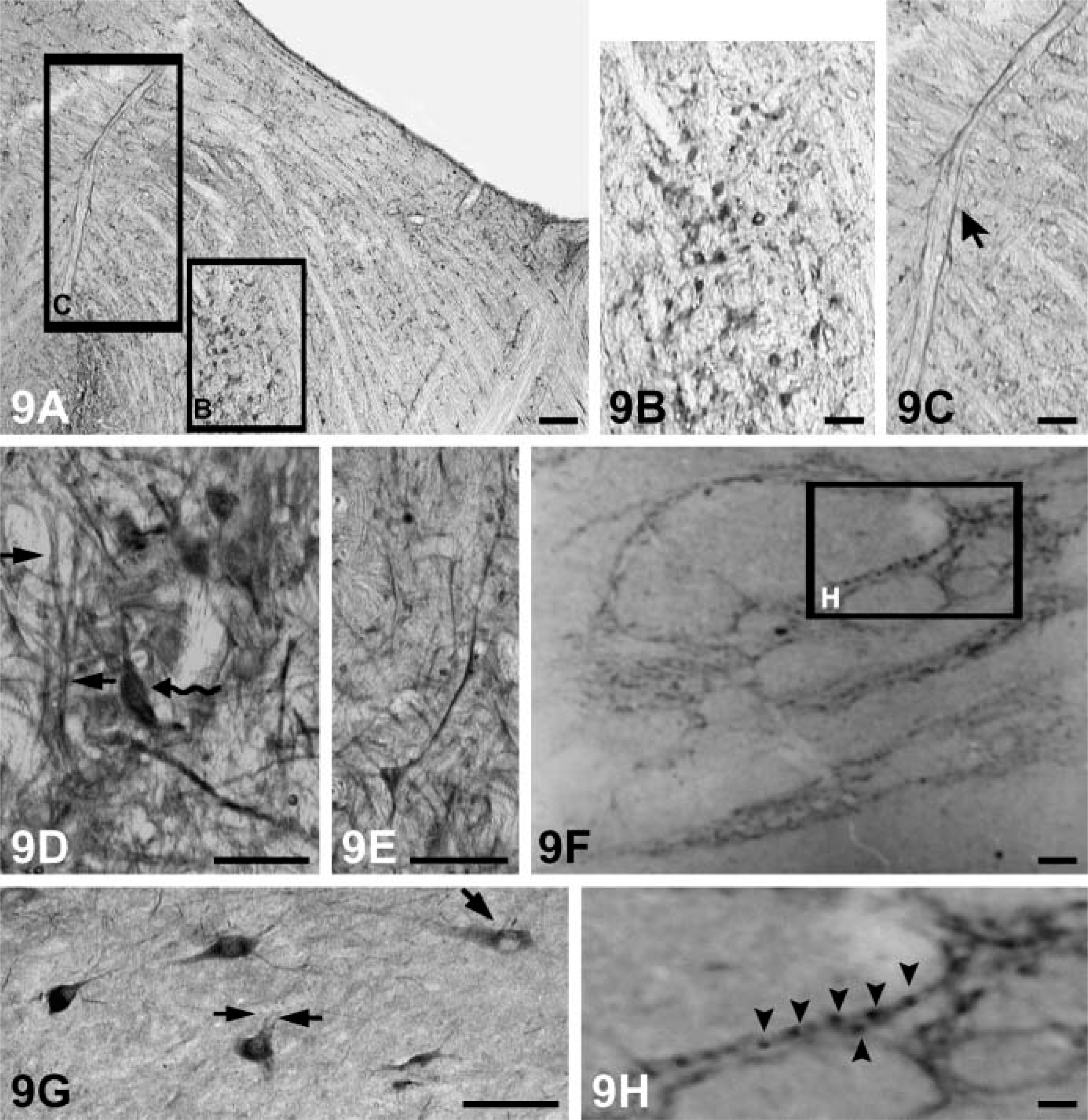
The somata and proximal dendrites with NGF, BDNF, NT-3, and NT-4 immunoreactivity can be observed in different subpopulations of neuronal cells in the brain stem (Figures 8A-8I). Nuclei of these cells were not stained. Immunoreactive products of NT-3 and BDNF were more obvious than NGF and NT-4 (Figures 8A–8C). Neuronal fibers also stained in some regions such as the spinal trigeminal tract and the peripeduncular nucleus (Figures 9A, 9C-9E). However, there are some regions (solitary tract, pontine nucleus) where fibers failed to be stained (Figures 9B and 9G). Immunoreactive labeling for all neurotrophins also can be found in the reticular formation (Figures 9F and 9H).
Immunoreaction products of NGF
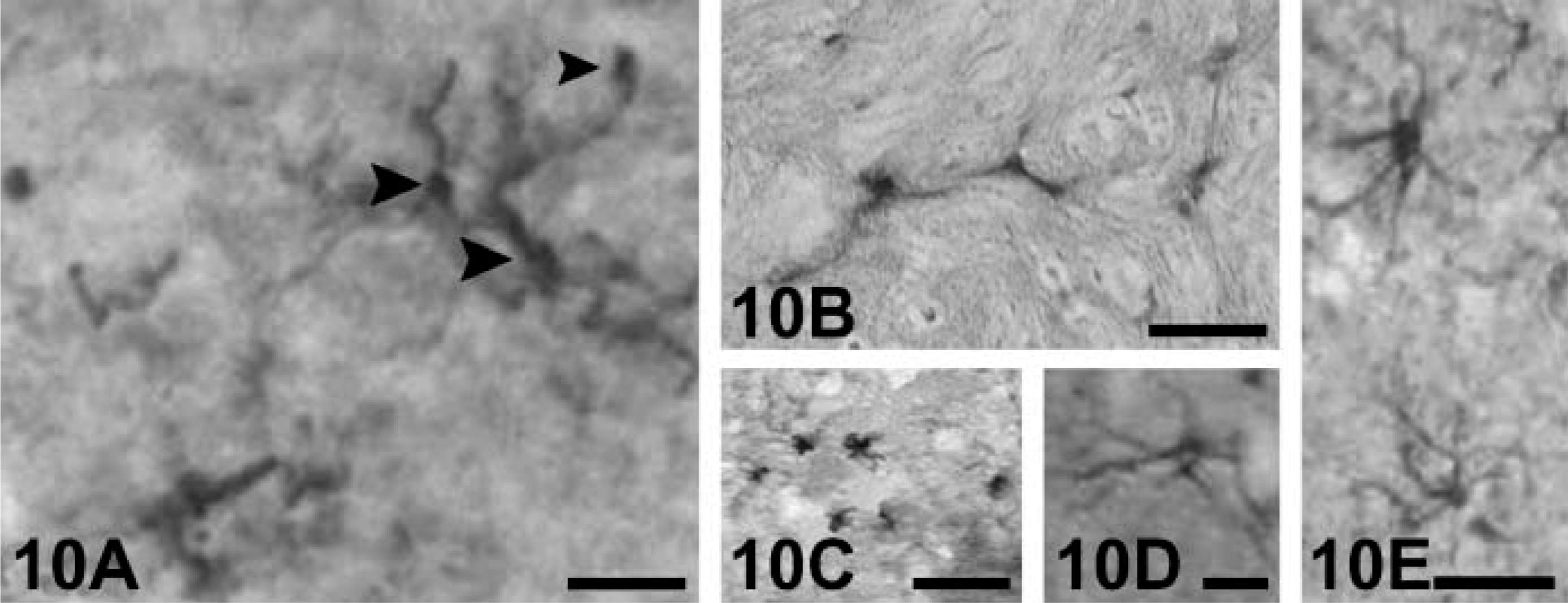
Glia. Glia in the white matter of the cerebrum and cerebellum (Figure 10A), the areas immediately beneath the central gray in the brain stem (Figure 10B), and corpus callosum all expressed weakly positive NGF, BDNF, NT-3, and NT-4 immunoreaction. Glial immunoreactive cells in the brain of the rhesus monkey showed different morphologies. The most frequently labeled glial cells resemble the morphological features of microglia (Figure 10A) or astrosytes (Figures 10C-10E).
Discussion
Antiserum Specificity
In this study, polyclonal antibodies against monkey NGF, BDNF, NT-3, and NT-4 were characterized. Analysis by Western blot showed that each antibody recognized specific domains of the neurotrophin proteins NGF, BDNF, NT-3, and NT-4 without cross-reaction with other neurotrophic factors. The antibodies recognized four bands of ∼14.7 kDa, 14.2 kDa, 13.6 kDa, and 14.5 kDa for NGF, BDNF, NT-3, and NT-4, respectively. Furthermore, sections exposed to primary antiserum preadsorbed with full-length NGF, BDNF, NT-3, and NT-4 exhibited no detectable immunoreactivity. This study first demonstrated that the antibodies used showed specificities to tissues prepared from rhesus monkeys.
Expression of Neurotrophins in the Brains of Adult Monkeys–Overview
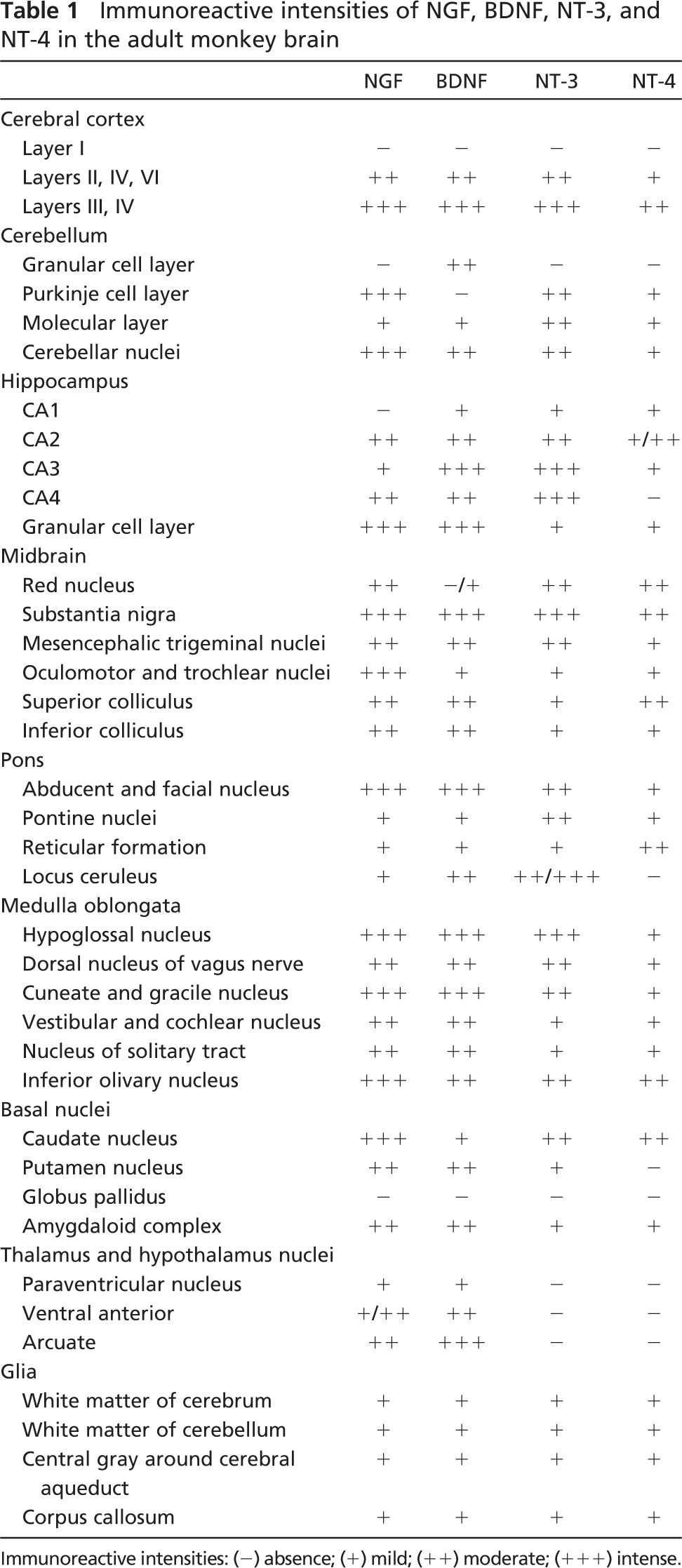
IHC localization of neurotrophins in the adult monkey brains, summarized in Table 1, is the first description and comparison of the expression and cellular localization of NGF, BDNF, NT-3, and NT-4 in adult primates.
Immunopositive products of NGF, BDNF, NT-3, and NT-4 were demonstrated mainly in the cytoplasm and proximal processes of neurons in the cerebrum, cerebellum, and hippocampus. The nuclei were not stained. This differed somewhat from the results of several authors (Kawamoto et al. 1996; Katoh-Semba et al. 1997; Furukawa et al. 1998) who reported nuclear staining of neurotrophins within different sub-populations of neurons. The discrepancy may be attributed to the use of different antibodies and different fixation methods, as has been demonstrated (Lee et al. 1998). Our results are in agreement with experiments performed by other research groups such as Dugich-Djordjevic et al. (1995) and Conner et al. (1997) on rat, Kawamoto et al. (1999) on monkey, and Murer et al. (1999) on human brains. Significantly, recent studies using methods to tag the translated products for intracellular trafficking of neurotrophins failed to reveal neurotrophin protein targeting to the nucleus (Carstén Möller et al. 1998; Haubensak et al. 1998; Mowla et al. 1999; Murer et al. 2001). In vitro studies (Goodman et al. 1996; Haubensak et al. 1998) provide further evidences that neurotrophin-like immunoreactivity was localized in the somatodendritic compartment of cultured neurons but not in the nucleus.
Distribution of immunoreaction products of NGF
Immunoreactive intensities of NGF, BDNF, NT-3, and NT-4 in the adult monkey brain
Immunoreactive intensities: (−) absence; (+) mild; (++) moderate; (+++)intense.
Varying staining intensities of the immunoreactive products of the four neurotrophins can be observed in specific neuronal populations in monkey brains. All in all, staining intensities of NGF and BDNF were stronger than those of NT-3 and NT-4. In agreement with the corresponding studies on rodents (Zhou and Rush 1994a; Dugich-Djordjevic et al. 1995; Kawamoto et al. 1996; Conner et al. 1997; Yan et al. 1997b; Zhou and Yang 1999), we detected intensive staining of all four neurotrophins in the gray matter of the cerebral cortex and hippocampus, indicating that these two areas were the important action targets of neurotrophins. Also, as shown in Table 1, there were several areas that lacked neurotrophin immunoreactivity, like the CA1 subfield of hippocampus for NGF, Purkinje cell layer of the cerebellum for BDNF, and most nuclei of the thalamus and hypothalamus for NT-3 and NT-4. Other than the possibility that proteins were not formed in these areas, they could be formed therein but soon afterwards were transported rapidly to other regions.
In the cerebral cortex of the rat and monkey, robust immunoreactivity of neurotrophins was principally seen in the cell bodies and primary dendrites of the medium to large pyramidal cells in the external pyramidal layer (layer III) that receives the project fibers from the diencephalon and internal pyramidal layer (layer V) and projects to subcortical regions to control the visceral efferent and somatic efferent functions. This suggests that neurotrophins may play crucial roles in maintaining specific connections between the cortex and other regions of the brain.
In the white matter of the cerebrum, cerebellum, central gray around the cerebral aqueduct, and the corpus striatum, scattered glial cells were immunopositive for NGF, BDNF, NT-3, and NT-4. This agrees with previous data from the rat and human (Murer et al. 1999). The low-affinity NGF receptor and trkB (high-affinity receptors of BDNF and NT-4) and trkB mRNA have all been detected in rat C6 glioma and type I astrocytes in culture (Acheson et al. 1991; Hamel et al. 1993). Furthermore, specific BDNF binding points and trkB mRNA were found in a glioma cell line and in Schwann cells (Hutton et al. 1992). We demonstrated further evidence that neurotrophins were also expressed in the glia of monkey brains. Hence, in vivo and in vitro studies on rodents, primates, and humans all point toward some crucial roles of neurotrophins in the physiological mechanism of glial cells. However, how these cells synthesize or take up the neurotrophins needs further study. According to several lines of evidence, NGF has also been detected in cerebrospinal fluid (Laudiero et al. 1992; Patterson 1996). It is possible that some glia may serve as a vehicle for the transportation of neurotrophins from the cerebrospinal fluid to specific regions of the CNS, as proposed by Zhou and Rush (1994b).
Because neurotrophic molecules can be observed in both neurons and glial cells, we speculate that by autocrine and paracrine actions, glia could regulate itself and the neurons nearby, suggesting a functional vinculum role of neurotrophins in neuron-glial interactions.
NGF-, BDNF-, NT-3-, and NT-4-immunopositive neurons were present in many parts of the adult monkey brain stem. As there are few reports on the distribution of neurotrophins in the primate brain stem, it is difficult to give a clear definition for the role of neurotrophins and their receptors in these regions. However, the following are noteworthy: (1) Distribution of neurotrophins in the primate brain stem mostly correspond to that in the rat brain (Hafidi 1999; Katoh-Semba et al. 2003), indicating that from rodents to primates, the neurotrophin family acts preferably on this part of the brain. Incidentally, the high-affinity neurotrophic factor receptors trkB and trkC and their mRNAs have been localized to most areas of the brain stem of rat (Merlio et al. 1992; Tessarollo et al. 1993; Zhou and Rush 1993,1994a). (2) The majority of the labeled neurons in the brain stem are motoneurons. Available evidence (Koliatsos et al. 1993) indicates that neurotrophic factors are survival factors for motoneurons. For example, BDNF mRNA and its protein increased 1 day after facial nerve axotomy (Vassrs and Michelle 1994). After sectioning the facial nerves in neonatal rats, local application of NT-4 prevented the death of 45% of the facial motor neurons. The survival number of corticospinal motor neurons cultured in vitro depended on the content of NT-4 in the culture medium (Junger and Varon 1997). Additionally, NT-4 can promote the differentiation of dopaminergic neurons in the midbrain (Katoh-Semba et al. 2003), and motoneurons can specifically transport 125I NT-3 from the sciatic nerve. Finally, Zhou and Rush (1994b) found that NT-3-immunoreactive products accumulate distal to a ligature on a sciatic nerve. Because NT-3 mRNA was also demonstrated in these motoneurons, a retrograde transport of an endogenous NT-3 was suggested.
Immunoreaction products of NT-3
Comparative Study of the Expression of Neurotrophins in Adult Monkey Brains
In the monkey brain, there were some dissimilarities of the immunoreactivity of the four existing neurotrophins. Some subtle differences of neurotrophin staining were noted in the somata of neurons in the cerebral cortex and the cerebellum. The interesting aspects of the NGF immunostaining pattern as seen here was that the presence of strongly positive punctate NGF-immunoreactive products on the surface of the somata and proximal processes of dendrites or in the vicinity of immunopositive neurons is consistent with what has been observed in the cerebral cortex of the rat (Kawamoto et al. 1999). NGF immunoreactivity shown here was presumably the result of its accumulation transported retrogradely from distant sites of synthesis. Staining of accumulated immunopositive products of NT-3 and NT-4 was similar to that of NGF. However, no BDNF-immunoreactive products accumulated in the somata in this manner. BDNF in the neurons of the cerebral cortex and the cerebellum may be carried to other regions persistently through anterograde transport following synthesis. Punctate varicose fibers with BDNF labeling gave further evidence.
High-power microphotographs display NGF
In comparison with NGF, NT-3, and NT-4, immunoreactivity of BDNF was more prominent in the processes of the labeled neurons. This suggests that one of the most important facets of BDNF actions in the CNS beyond cell survival was in the regulation of synaptic transmission and synaptogenesis. During the last decade, the role of BDNF in long-term synaptic plasticity was elucidated by numerous studies (Patterson et al. 1996; Martinez et al. 1998; Chen et al. 1999; Pozzo-Miller et al. 1999); however, in the case of primates, it remains to be determined. The difference in the distribution patterns of BDNF and the other three neurotrophins could mean that BDNF might work by different pathways to modulate normal physiological functions of the cerebral cortex.
Intriguingly, we did not find any strong BDNF-immunopositive products within the cell bodies in the Purkinje cell layer of the cerebellum as detected with the other three neurotrophins. But the staining of BDNF in axon-like fibers in the molecular cell layer was stronger than the other three. Intense BDNF-immunoreactive materials could also be found in the inferior olivary complex. A previous study (Ohira and Hayashi 2003) showed that the axons of basket cells situated above the deeper part of the molecular layer extended some of their branches to the cell bodies of Purkinje cells. The terminals of these branches formed an intricate network around the Purkinje somata (Lo 1995). With these findings we can presume that these fibers were climbing fibers. It appears that for proper physiological functions, Purkinje cells need the trophic support of BDNF, and as they do not synthesize BDNF themselves, they obtain through retrograde transport from other regions.
There were no detectable signals of NT-3 or NT-4 in most nuclei of the thalamus and hypothalamus. Only BDNF and NGF immunostainings were detected in the fibers and neurons. Presence of NGF mRNA and BDNF mRNA in vasopressin-positive neurons has been demonstrated in the rat hypothalamic nucleus (Goedert et al. 1986; Castron et al. 1995), suggesting that NGF and BDNF could be synthesized in these neurons and take part in water balance regulation of the body. A possible role for NGF and BDNF in neuroendocrine function can also be assumed.
Some Differences of Neurotrophin Expression in Adult Rat and Monkey Brains
Compared with previous IHC studies in the rat brain, we noted some differences in the expression of NGF, BDNF, NT-3, and NT-4 in adult rat and monkey brains.
In the rat there was little or no BDNF immunoreactivity detected in the cerebellum, most of the brain stem and the granular cell soma of the dentate gyrus, and the pyramidal neurons of the hippocampus in normal rats (Yan et al. 1997b). In the monkey, BDNF protein was detected in all these regions. The pattern of BDNF staining in monkey brains as reported by Kawamoto et al. (1999) conforms to that of the present study. It is possible that the actions of BDNF in these regions are different between the rodent and the primate. In the rat, the granule cell layer of the hippocampus demonstrated strong NT-3 immunostaining, but no NT-3 staining was observed in the locus ceruleus (Friedman et al. 1998). In the monkey, moderate NT-3 staining was seen in the same subfields of the hippocampus and also in the locus ceruleus.
On the other hand, NT-4 immunoreactivity observed in the rat hypothalamus was not seen in the monkey brain in this study. Not taking into consideration differences in technical details, it appears from the above analysis that the neurotrophins mentioned could exert different functions in some regions of the adult brains in different species of animals.
Comparison of Neurotrophin Protein, mRNAs, and Their Receptor Expressions
Previous studies showed the presence of mRNAs and receptor expressions of neurotrophins in specific regions of adult brains. Combining these with our data would shed some light on their sites and modes of action (Tables 2–5).
Both NGF protein and NGF mRNA have been reported strongly expressed in the cerebral cortex, cerebellum, and hippocampal formation of the monkey (Maisonpierre et al. 1990; Hayashi et al. 1993). RNA blot analysis (Korsching et al. 1985; Korsching 1986) in rat brain also showed that hippocampus and neo-cortex have the highest NGF mRNA levels. Despite the fact that distribution of TrkA-like immunoreactive fibers is much more restricted than that observed for TrkB, TrkA immunoreactivity was prominent in the hippocampus and cerebellum (Kordower et al. 1994). Colocalization of protein, mRNA, and their receptors indicates that NGF synthesis occurs in these neurons, and NGF might act in an autocrine and paracrine manner to re-allocate the protein. However, no immunoreactive signals of TrkA can be found in the cerebral cortex, and this has been demonstrated in the rat (Lee et al. 1998) and monkey (Schatteman et al. 1988). NGF synthesis in this region is possibly transported to other regions where NGF receptor immunoreactivity can be examined, such as thalamus and hypothalamus.
Expression of NGF, NGF mRNA (Hayashi et al. 1993), and NGF receptor (Schatteman et al. 1988; Koh et al. 1989; Talamini and Aloe 1993; Mufson et al. 1996; Lee et al. 1998) in the adult brain

Levels of NGF determined by Northern blot.
During development.
Data from rat brain.
Data from human brain.
After colchicine treatment.
Immunoreactive densities: (−) absence; (+) mild; (++) moderate; (+++) intense.
Expression of BDNF, BDNF mRNA (Huntley et al. 1992; Hayashi et al. 1997; Conner et al. 1997), and TrkB (Zhou and Rush 1993; Yan et al. 1997a; Okuno et al. 1999; Ohira and Hayashi 2003) in the adult brain
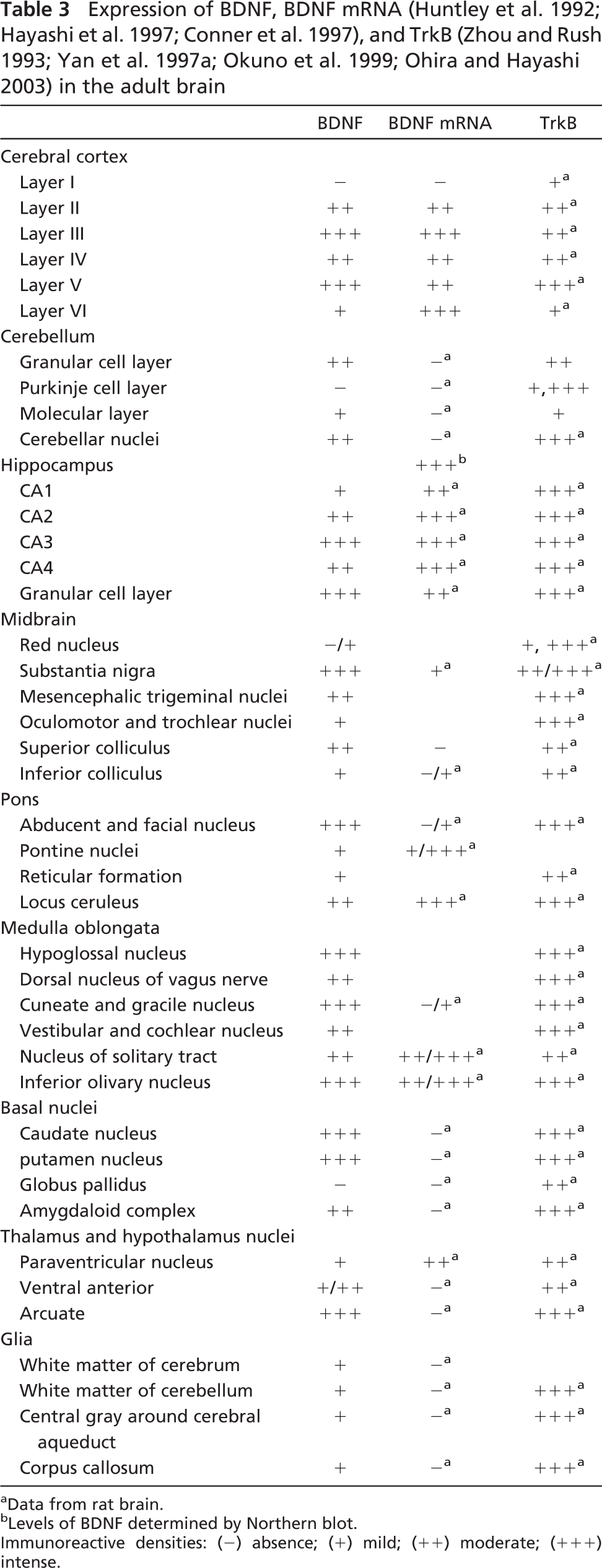
Data from rat brain.
Levels of BDNF determined by Northern blot.
Immunoreactive densities: (−) absence; (+) mild; (++) moderate; (+++) intense.
Expression of NT-3, NT-3 mRNA (Ernfors et al. 1990b; Hohn et al. 1990; Maisonpierre et al. 1990; Ceccatelli et al. 1991), and TrkC (Sandell et al. 1998) in the adult brain

Data from rat brain.
After colchicine treatment.
Immunoreactive densities: (−) absence; (+) mild; (++) moderate; (+++) intense.
Expression of NT-4, NT-4 mRNA (Timmusk et al. 1993; Okuno et al. 1999), andTrkB (Yan et al. 1997a; Ohira and Hayashi 2003; Zhou and Rush 1993) in the adult brain

Levels of NT-4 determined by RNAase protection assay.
Data from rat brain.
Immunoreactive densities: (-) absence; (+) mild; (++) moderate; (+++) intense.
There was an abundant expression of BDNF mRNA in various parts of the cerebral cortex and hippocampus of the adult rat and monkey (Ernfors et al. 1990b; Hofer et al. 1990; Wetmore et al. 1990; Hayashi et al. 1997). BDNF-positive neurons and fibers were observed in the same regions in the present study, indicating that these neurons themselves could synthesize BDNF. However, BDNF had a limited distribution in the rat hippocampus, as there was little or no BDNF immunoreactivity in the granular cell bodies of the dentate gyrus and the pyramidal neurons of the hippocampus. This suggests different action modes of BDNF in the hippocampus between rats and monkeys. TrkB-like immunoreactivity in the monkey brains has not been determined, but it has been reported to be present in neurons throughout the entire rat brain (Zhou and Rush 1993; Yan et al. 1997a), especially in many of the regions in which BDNF protein was localized in the monkey brain. The support from the finding in the rat brain suggests an autocrine and paracrine action mode of BDNF in some sites of the brain.
NT-3 immunoreactivity was widely and intensely distributed in the rat and monkey cerebral cortex, cerebellum, and hippocampal formation. Distribution of NT-3 mRNA has not been described in the monkey brain, but previous in situ hybridization studies (Ernfors et al. 1990b; Ceccatelli et al. 1991) failed to find NT-3 mRNA in the Purkinje cells of the rat cerebellum, and a low level of NT-3 mRNA could be detected in the cerebral cortex of the rat (Hohn et al. 1990; Maisonpierre et al. 1990). Immunoreactive trkC receptors were detected in the Purkinje cells of the adult monkey and human cerebellum and in the pyramidal cells of the developing and adult human cerebral cortex (Muragaki et al. 1995), showing that these cells need the trophic sustenance of NT-3, and the NT-3 protein may be synthesized from other regions. In adult rats, in situ hybridization showed an abundant distribution of NT-3 and NT-3 mRNA in the hippocampus (Ernfors et al. 1990b; Zhou and Rush 1994a), suggesting that NT-3 was synthesized within these neurons. Whether such is also the case in adult monkeys requires further elucidation. Interestingly, in experimental ischemia and hypoglycemia, NT-3 mRNA was downregulated, whereas BDNF mRNA and NGF mRNA were upregulated (Ernfors et al. 1991; Lindvall et al. 1992). In the developing rat brain, NT-3 mRNA concentrations are reduced, but the level of NGF mRNA and BDNF mRNA are increased (Maisonpierre et al. 1990; Friedman et al. 1991). Furthermore, electrical stimulation of the hippocampus induced a significant increase in NGF mRNA and BDNF mRNA but had no effect on NT-3 mRNA (Ernfors et al. 1991). Together this could mean that NT-3 plays a different biological role from NGF and BDNF in the hippocampus.
NT-3 is the only neurotrophin known to bind with high affinity to TrkC. Recently, TrkC-like immunoreactivity was detected by Sandell et al. (1998) in the brain of rhesus monkey. In many regions there was NT-3-like immunoreactivity in areas that also expressed TrkC, indicating that NT-3 acts in an autocrine and paracrine manner in some localizations. However, unlike the robust expression of NT-3 protein in cingulated cortex, hippocampus, and cerebellum, TrkC-like immunoreactive fibers were not common in these areas. Sandell et al. (1998) were particularly interested in the locus ceruleus because high concentrations of TrkC-like immunoreactive fibers and varicosities were examined in this area, and TrkC is the only neurotrophin receptor detected in the locus ceruleus by in situ hybridization. We also detected moderately labeled NT-3-like immunoreactivity in the locus ceruleus, suggesting the special role of NT-3 in this region. NT-3 may participate in sleep control.
To our knowledge, no corresponding reports are available on the systemic localization of NT-4 mRNA in the cerebral cortex of adult primates. In rodents, NT-4 mRNA is widely expressed and developmentally regulated in the rat brain during embryonic development (Timmusk et al. 1993; Hafidi 1999). In contrast, NT-4 mRNA levels are extremely low in the adult rat brain, and the staining intensity of NT-4 was detected lower than other neurotrophins in our study. Therefore, NT-4 content may be very limited in the monkey brain, and CNS is not the main target region of NT-4. Despite the fact that NT-4 and BDNF act through the same type of receptor, TrkB, BDNF was more widely distributed in CNS. BDNF knockout mice presented decreases of the number and intensity of neurons expressing neuropeptide or parvalbumin in the cortex (Ernfors et al. 1994; Jones et al. 1994); however, in NT-4 knockout mice, important defects were noted in the PNS but not in the CNS (Conover et al. 1995; Liu et al. 1995). BDNF and NT-4 have a different interaction with TrkB. BDNF, but not NT-4, is able to interact with the mutant form of TrkB (Ip et al. 1993). Moreover, a splice variant of TrkB, called ctrkB-Short, interacts strongly with BDNF but poorly with NT-4 (Strohmaier et al. 1996). BDNF was shown to down-regulate expression of TrkB in hippocampal neurons, whereas NT-4 did not exert any effect. Therefore, BDNF may have higher affinity to TrkB than NT-4 and may exert more important effects in CNS than NT-4.
Roles of Neurotrophins in the Adult Brain
Different populations of neurons became atrophic or lost in the course of neurodegenerative diseases. For example, degeneration and subsequent loss of neurons in the substantia nigra compacta occur in Parkinson's disease; of motoneurons in the cerebral cortex in amyotrophic lateral sclerosis; of basal forebrain cholinergic neurons, cerebral motoneurons, and hippocampal neurons in Alzheimer's disease; and of striatal neurons in Huntington's disease (Hefti et al. 1989). Neurotrophin expression in these neurons is also altered by these neurodegenerative diseases. Conceivably, modulation of neurotrophin levels in these regions may produce some clinical benefits. In fact, some exciting advances have been achieved in experimental animals, and these have contributed to some understanding in the mechanisms producing neuronal death and degeneration.
Glia, especially astrocytes, also play a very important role in the repair of CNS after injury. Astrocytes are known to sustain the survival of neurons and to advance the growth of neurites and have been proved to produce neurotrophins in culture. Following the transplantation of embryonic spinal cord into the adult spinal cord, there was increased expression of NGF, BDNF, and NT-3 (Zhou and Yang 1999). The presence of neurotrophin-immunopositive products in the glia indicates that neurotrophins may take part in the regulation of glial functions and play some role in the development of neurons and the repair of injured nerve cells.
Finally, changes in the expression of neurotrophins and their mRNA have been detected in many other diseases such as epilepsy (Mathern et al. 1997; Takahashi et al. 1999), ischemia, and hypoglycemia (Ernfors et al. 1991; Lindvall et al. 1992). Morphological demonstration of the correlation between these diseases and neurotrophins, their mRNAs, and receptors will provide the basis for a deeper molecular understanding of the topic and lead to effective therapeutic approaches to human neurodegenerative diseases.
Footnotes
Acknowledgements
This work was supported by a grant from the National Science Foundation of China (Grant No: 30000227). We thank Lang-Li and Ming-Li for technical assistance and Professor Seng-Kee Leong for very useful comments on the manuscript.
