Abstract
The poly(ADP-ribose) polymerase 1 (PARP-1) enzyme has received much attention in the last decade due to its promising role in cancer therapeutics. Despite the expanding use of PARP inhibitors in cancer therapy, little is known about PARP-1 tissue distribution. Our study provides a detailed survey of PARP-1 tissue and cellular distribution using well-preserved cynomolgus monkey organs and a well-characterized, highly specific monoclonal PARP-1 antibody. Overall, PARP-1 was detected in most organs, but its distribution was restricted to specific cells within each tissue, suggesting that PARP-1 expression is tightly regulated. The strongest expression was in the pituitary, the ovary, the male adrenal gland, and the thymus. One of the key findings of this study was the stronger expression of PARP-1 in proliferating cells rather than mature cells. This observation not only provides clues to the importance of PARP-1 in processes such as DNA replication and transcription in these cell types, but it also provides the basis for further investigation into the effects of its inhibition in the context of malignancy. Overall, this study greatly expands the current knowledge of PARP-1 tissue expression, enabling the identification of tissues where PARP inhibition may be most efficacious.
Introduction
Poly(ADP-ribose) polymerase-1 (PARP-1) is a nuclear enzyme that is best understood for its functions in stress response pathways, 1 namely, the DNA damage response (DDR). PARP-1 also participates in many other cellular events such as chromatin modification,2,3 transcription,4,5 regulation of inflammatory mediators,6,7 cell death pathways, 8 mitotic apparatus function, 9 and sex hormone signaling.10,11 Increased understanding that PARP-1 is a key signaling factor in the DDR has led to its clinical exploitation in the setting of cancer therapeutics. In addition, its multitude of roles in a wide variety of physiological processes has also led to the investigation of PARP-1 inhibition as therapy for non-oncologic conditions in which PARP’s functions are central. Such conditions include inflammatory processes, 7 cardiovascular and neurological diseases, 12 and age-related illnesses.13,14
This clinically promising activity derives from the synthetic lethality mechanism of PARP inhibition in cells with defects in homologous recombination (HR) DNA repair. Synthetic lethality describes two genetic lesions, which are individually not lethal, but become lethal when combined in a single organism.15,16 This effect was first recognized in tumors bearing mutations in breast cancer–related genes, BRCA1 and BRCA2, which encode proteins crucial to the successful resolution of HR.17,18 Thus, cells deficient in HR are hypersensitive to the inhibition of PARP enzymatic activity by specific PARP inhibitors (PARPi). Overall, the critical role of PARP-1 in the DDR 19 has made it one of the most important targets for chemotherapies targeting breast,20,21 ovarian,20,21 prostate,22,23 and pancreatic cancers,24,25 whose DNA repair weaknesses render them responsive to PARPi.
Despite the expanding clinical use of PARP-1 inhibitors and their broad functions in the biology of normal cells, the tissue distribution of this enzyme has received little attention. 26 Thus, the goal of this study was to extensively survey PARP-1 tissue and cellular distribution in the cynomolgus monkey, a valuable animal model for protein distribution and expression studies, due to its physiological similarity to humans. 27 We reveal that PARP-1 is expressed in all organs examined. However, its cellular and subcellular distribution suggests a tightly regulated expression in each tissue, because PARP-1 is not ubiquitously detected in all cell types, and strong expression is only present in specific subsets of cells of certain tissues.
Materials and Methods
Cell Line
HeLa cells, an epithelial cell line isolated from a cervical adenocarcinoma, were purchased from the American Type Culture Collection (ATCC; Manassas, VA) and cultured according to ATCC instructions. For immunostaining experiments, HeLa cells were formalin-fixed and paraffin-embedded as previously described. 28
Monkey Tissues
Paraffin blocks of cynomolgus monkey (Macaca fascicularis) tissues, from a library of two male (5 and 7 years old, weighing 5.6–8.6 kg) and two female (4 years old, weighing 3.1–4.0 kg) adult monkeys sacrificed for previous studies, were utilized for this research. 28 These animals were in good health, as verified by a complete veterinary examination, serum biochemistry, and complete blood count. They were treated per the guidelines and policies of the Canadian Council on Animal Care and the National Institutes of Health Guide for the Care and Use of Laboratory Animals. Briefly, animals were housed individually or in pairs in standard stainless-steel cages in a room maintained at 23 ± 3C with a 12-hr dark, 12-hr light cycle. Water was available ad libitum, and animals were fed four primate cookies twice daily at 6-hr (±1-hr) intervals. Fruits and/or vegetables were distributed twice weekly. This protocol has been approved by the Ethical Committee for Animal Protection of the Center Hospitalier Universitaire de Québec. The monkeys were sacrificed by an overdose of isoflurane anesthesia, followed by intracardiac perfusion with 10% neutral buffered formalin. Organs were collected and immersed in 4% paraformaldehyde (PFA) neutral buffer for 12 hr and embedded in paraffin for immunofluorescence and immunohistochemistry analysis. Two consecutive serial sections were cut (4 μm) from each tissue block, in which the first was immunostained and the second served as a control.
Immunohistochemistry
The highly specific anti-PARP-1 monoclonal antibody (clone C2-10) used in this study was produced in-house as previously described. 29 The immunogenic peptide sequence used to produce clone C2-10 (P273SGESAILDR282) is unique to PARP-1 and entirely conserved in several primates, including the cynomolgus monkey (Table 1). Immunostaining was performed using Zymed SP kits (San Francisco, CA). Paraffin sections were deparaffinized in toluene and gradually rehydrated through graded ethanols. Endogenous peroxidase activity was eliminated by preincubation of sections with 3% H2O2 in methanol for 20 min. A microwave antigen retrieval technique using citrate buffer was applied.28,30 Nonspecific binding sites were blocked using 10% goat serum for 30 min at room temperature, and sections were incubated for 2 hr at room temperature with anti-PARP-1 antibody (clone C2-10) at a concentration of 1:2000 for HeLa cells and 1:1000 for all tissues, except the bladder, seminal vesicle, and prostate (1:500). Sections were then washed in PBS buffer and incubated with biotinylated anti-mouse secondary antibody, followed by streptavidin–peroxidase as described by the manufacturer (Zymed SP kits). Under microscope monitoring, 3,3′-diaminobenzidine (DAB) was used as the chromogen to visualize the biotin/streptavidin–peroxidase complex. Counterstaining was performed using #2 Gill’s hematoxylin for 1 min. To assess nonspecific binding (control sections), anti-PARP-1 antibody was preincubated with an excess of the antigenic peptide (10 µg/ml) for 3 hr at room temperature before application to matched serial sections. Tissue sections were visualized on a Nikon Eclipse E400 microscope (Tokyo, Japan) and captured using a Coolpix s10 Nikon camera (Abingdon, VA), objectives 20× and/or 40× (CFI Plan Achromat; NA 0.40 and 0.65, respectively).
PARP-1 Peptide Sequence Alignment Between Human and Monkey Species.

High sequence conservation between human and monkey PARP-1 sequences. Alignment of the human PARP-1 sequence comprising the immunogenic peptide (highlighted in yellow) used to produce the C2-10 antibody, with homologous PARP-1 monkey sequences. Asterisks denote identical residues and colons denote conservative substitutions. PARP-1 sequences were taken from the UniProt database, and alignment was performed using the UniProt Blast tool. UniProt PARP-1 accession numbers: Human = P09874; Cynomolgus monkey = G7NTV2; Chimpanzee = K7C230; Rhesus macaque = F7EMW7; Gorilla = G3SBV5; Sumatran orangutan = H2N3J1.
Abbreviation: PARP-1, poly(ADP-ribose) polymerase-1.
Data Analysis
The organs (n=21) analyzed in this study include the brain, pituitary, mammary gland, skin, ovary, testis, epididymis, efferent ductules, seminal vesicle, prostate, adrenal, pancreas, stomach, heart, lung, salivary glands, spleen, thymus, urinary bladder, uterus, thyroid. The kidney, liver, duodenum, colon, and vagina could not be assessed. Antibody staining was analyzed by two individuals (LB and MTF). Results were described as a sum of the analysis of n=4 animals for each given organ, except for sex-specific organs and the adrenal, for which the results represent n=2 for each sex. In many tissues, the staining of a given cell type varied in intensity. Thus, the scoring of labeling was divided into three subcategories: staining intensity (unlabeled, weak, moderate, and strong), percentage of staining intensity (majority: >60%, some: >60%–20%, and few: <20%), and subcellular localization (nuclear and/or cytoplasmic). In the figure section, the images chosen for each tissue are representative of the average labeling.
Results
Specificity of the Anti-PARP-1 Monoclonal Antibody for IHC Staining
The specificity of the monoclonal anti-PARP-1 (C2-10) antibody has been previously validated by Western blot using whole cell extracts of different cell lines, including human (HeLa) and monkey (COS-7) cell lines,31 –35 where a unique protein of the expected molecular mass (113 kDa) is detected. Further validation has also been shown by PARP-1 knockdown cell lines.36,37 Here, we have established that the detection of PARP-1 with C2-10, using an immunohistochemical method, is also specific. Immunohistochemically stained HeLa cells, which are a model cell line for the study of PARP-1, 8 revealed specific nuclear labeling (Fig. 1A). This staining is abolished with a preadsorption of the C2-10 antibody with the immunogenic peptide before cell labeling (Fig. 1A’). In addition, the predominantly nuclear staining of HeLa cells is consistent with the previously reported nuclear localization of PARP-1 (Fig. 1A).37,38 Taken together, C2-10 specifically recognized PARP-1 by immunohistochemistry in cells.
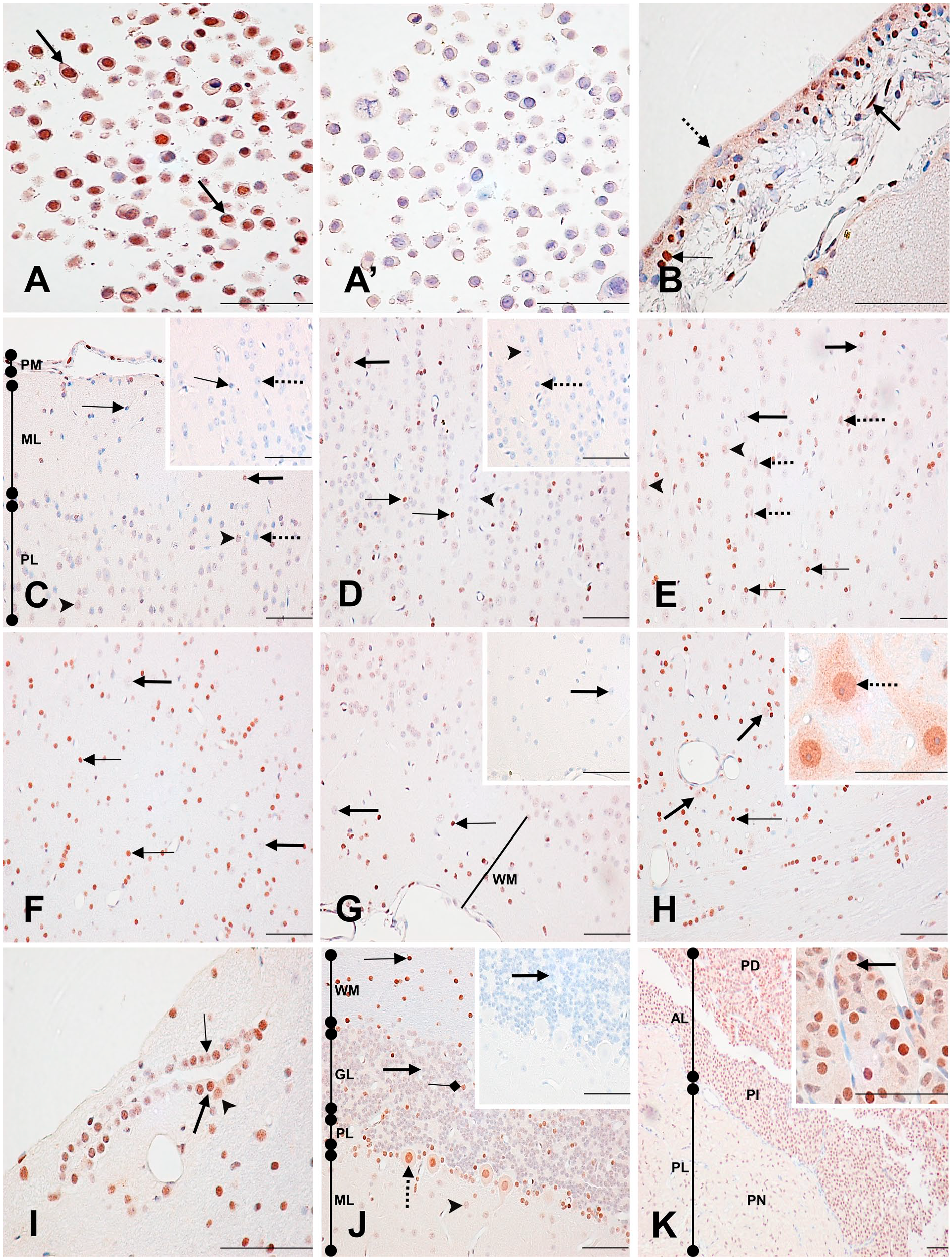
Immunodetection of PARP-1 in HeLa cells, normal monkey brain, and pituitary gland. (A) Specificity of the anti-PARP-1 antibody C2-10 in the human HeLa cell line. Strongly labeled nuclei (arrows). (A’) Preincubation of the PARP-1 antibody with an excess of the immunogenic peptide abolishes the HeLa nuclear staining. (B) Brain: pia mater (PM). Strongly labeled (thin arrow) and unlabeled (dotted arrow) epithelial cell nuclei with weak cytoplasmic staining. Strongly stained fibrocyte nucleus (thick arrow). (C) Brain: cerebral cortex. Weakly labeled (arrowhead) and unlabeled (dotted arrow) neuronal nuclei in the pyramidal layer (PL). Moderately labeled (thick arrow) and unlabeled (thin arrow) neuroglial nuclei in the molecular layer (ML). Control section of the PL (inset) shows unlabeled neuronal (dotted arrow) and neuroglial nuclei (thin arrow). PM. (D) Brain: granular layer. Weakly labeled (thick arrow) and unlabeled (arrowhead) neuronal nuclei. Strongly labeled (thin arrows) neuroglial nuclei. Control section (inset) shows unlabeled neuroglial (dotted arrow) and neuronal (arrowhead) nuclei. (E) Brain: polymorphic layer. Unlabeled (thick arrows) and weakly labeled (arrowheads) neuronal nuclei. Strongly stained neuroglial nuclei (thin arrows). Weakly labeled pyramidal neuronal nuclei (dotted arrows). (F) Brain: transition layer that connects to the white matter. Unlabeled neuronal nuclei (thick arrows). Strongly labeled neuroglial nuclei (thin arrows). (G) Brain: white matter (WM). Unlabeled neuronal nucleus (thick arrow). Strongly labeled neuroglial cell nucleus (thin arrow). Control section of the WM shows an unlabeled neuronal nucleus (thick arrow). (H) Midbrain: Weakly stained neuronal nuclei (thick arrows) and strongly stained neuroglial nucleus (thin arrow). The inset shows strongly labeled neuronal nuclei (dotted arrow) with a weak stippled cytoplasmic and nucleolus staining. (I) Midbrain: Strongly (thick arrow), moderately (arrowhead), and weakly (thin arrow) stained ependymal cell nuclei. (J) Cerebellum: Strongly stained neuroglial nucleus (thin arrow) in the WM. Unstained (thick arrows) and strongly stained (squared arrow) neuronal nuclei in the granular layer (GL). Weakly labeled neuronal nucleus (arrowhead) in the ML. Strongly stained Purkinje cell nucleus (dotted arrow) in the Purkinje cell layer (PL) with a weakly stained cytoplasm. Control section shows an unstained (thick arrow) neuronal nucleus in the GL. (K) Pituitary: The anterior lobe (AL) shows strongly labeled nuclei in the glandular cells of the Pars distalis (PD) and in the follicular epithelial cells of the Pars intermedia (PI). The Pars nervosa (PN) of the posterior lobe (PL) shows moderately labeled pituicyte nuclei. The inset shows a closer view of the PD glandular cell nuclear staining and their weak cytoplasmic staining (arrow). Scale bar = 50 µM. Abbreviation: PARP-1, poly(ADP-ribose) polymerase-1.
Distribution of PARP-1 in Tissues of the Cynomolgus Monkey
PARP-1 expression was found in all tissues surveyed. However, PARP-1 expression was not ubiquitous to all cell types of each tissue. This distribution is summarized in Supplementary Tables 1–24. In most of the monkey tissues where PARP-1 was detected, the expression was preferentially nuclear, including the nucleolus. Interestingly, staining intensity varied among individual cells of a given cell type. Although some nuclei were strongly labeled, adjacent cell nuclei were unlabeled. This was best seen in the sweat glands (Fig. 2I), in the epididymis head (Fig. 4A), in the pancreatic acini (Fig. 5C), and in the fundic glands of the stomach (Fig. 5H). A specific cytoplasmic staining was also seen in most tissues. Levels of PARP-1 expression also varied between tissues. Those with the strongest expression included the pituitary, mammary gland, pancreas, ovary, uterus, testis, seminal vesicle, epididymis, male adrenal, and the thymus. On the contrary, the weakest expression was seen in the cerebral cortex, the prostate, and the efferent ductules. Unless otherwise mentioned, all control tissue sections were devoid of staining.
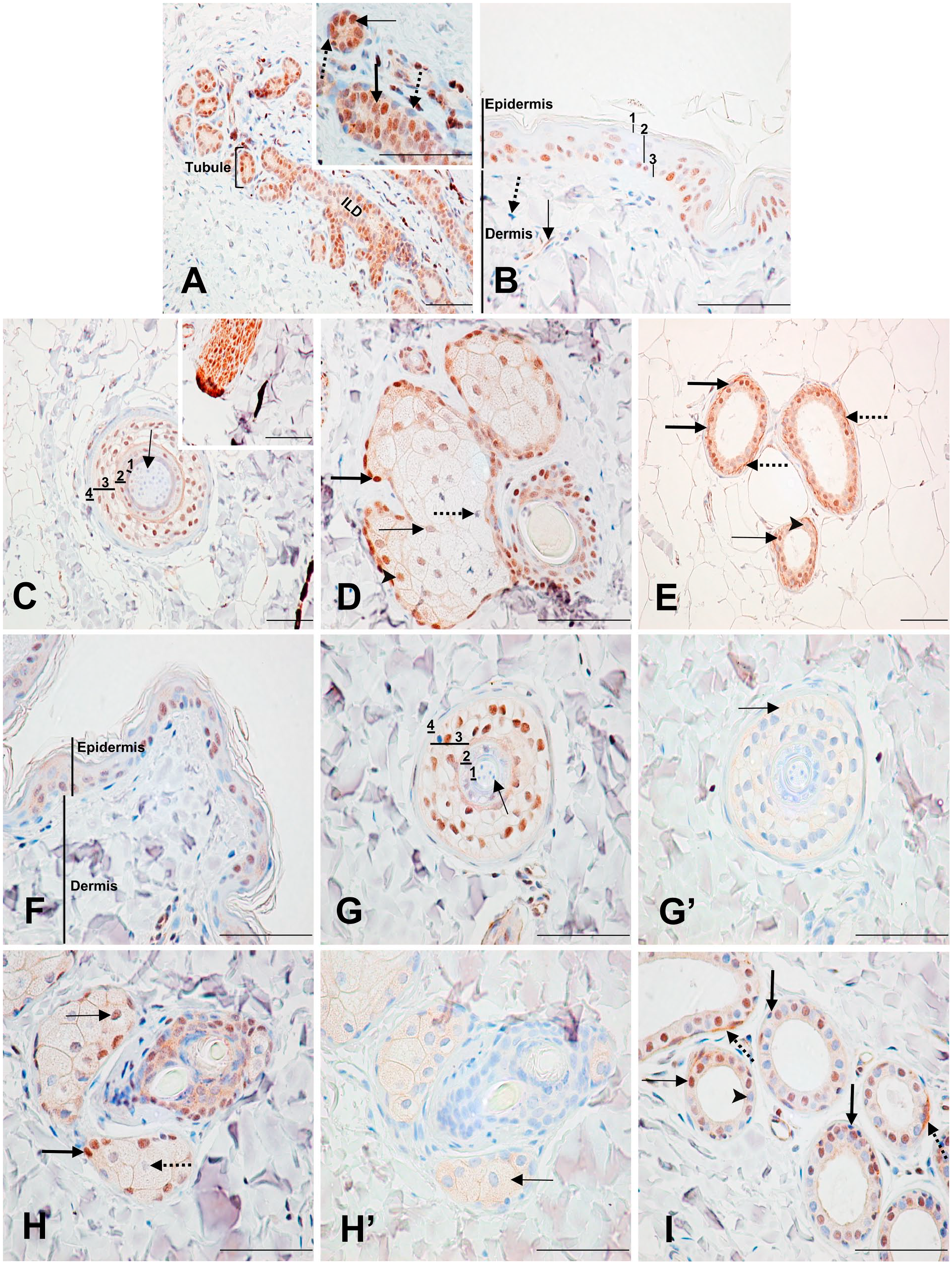
Immunodetection of PARP-1 in the mammary gland and skin. (A) Mammary gland: Strongly stained columnar epithelial cell nucleus (inset, thin arrow) with moderate cytoplasmic staining. Moderately stained intralobular ductal (ILD) cell nucleus (inset, thick arrow) with moderate cytoplasmic staining. Unlabeled myoepithelial cells (inset, dotted arrows). (B) Breast skin: (1) unlabeled stratum corneum; (2) moderately labeled nuclei in the stratum spinosum and granulosum; (3) strongly labeled nuclei in the stratum basale. Dermis shows labeled (thin arrow) and unlabeled (dotted arrow) fibrocytes. (C) Breast skin dermis: Hair follicle. (1) Cuticle cells with unlabeled nuclei; (2, 3) root sheath cells with moderately to strongly stained nuclei; (4) connective tissue cells with unlabeled nuclei. Unlabeled hair shaft cell (arrow). Cells from Layers 1 to 3 have weak cytoplasmic staining. The inset shows an arrector pili muscle with all cell compartments strongly labeled. (D) Breast skin dermis: sebaceous glands. Strongly labeled basal cell nucleus (thick arrow) with moderate cytoplasmic staining. Moderately stained immature secretory cell nucleus (arrowhead) with weak cytoplasmic staining. Weakly stained mature secretory cell nucleus (thin arrow). Unlabeled degenerating secretory cell nucleus (dotted arrow). (E) Breast skin dermis: sweat gland. Unlabeled (arrowhead) and strongly labeled (thin arrow) secretory cell nuclei with moderate cytoplasmic staining. Unlabeled myoepithelial cell nuclei (thick arrows). Strong linear labeling at the base of the secretory cells (dotted arrows). (F), (G), (H), and (I) show the skin and have the same legend as (B), (C), (D), and (E), respectively, with the exception of the nonspecific cytoplasmic staining of the root sheath cells of the hair follicle and the secretory cells of the sweat gland, which are shown by (G’) and (H’) respectively (arrow). Scale bar = 50 µM. Abbreviation: PARP-1, poly(ADP-ribose) polymerase-1.
Brain
Pia Mater, Cerebral Cortex, and White Matter
In the pia mater, there were many strongly labeled epithelial and fibrocyte cell nuclei. A weak, but specific cytoplasmic staining was also present in these cells (Fig. 1B; Supplementary Table 1). The staining profile of the gray matter of the cortex is described from the most external to the most internal layer of the cortex—molecular layer, pyramidal cell layer, granular layer, and the polymorphic layer—ending with the white matter. Moving from the molecular layer to the white matter, a progressively increasing staining intensity was seen in the nuclei of the neuroglial cells, whereas the neuronal nuclei were weakly stained or unstained throughout the cortex and white matter. Pyramidal neuronal cells from the pyramidal cell layer also had weak nuclear labeling (Fig. 1C–G; Supplementary Table 1). Nucleoli of neuronal cells were mostly unlabeled, and a weak, but specific cytoplasmic staining was detected occasionally. Overall, throughout the cortex, nuclei of neuroglial cells expressed more PARP-1 than nuclei of neuronal cells.
Midbrain (Mesencephalon)
In the midbrain, neuroglial nuclei were predominantly strongly labeled, whereas neuronal nuclei were weakly labeled. Interestingly, a few neuronal nuclei were moderately to strongly labeled, although they appeared to be similar in morphology to those with no labeling (Fig. 1H; Supplementary Table 2). These neuron’s cytoplasm had weak but specific labeling with a moderately stained stippled pattern. Their nucleoli were also weakly labeled (Fig. 1H inset). Ependymal cell nuclei labeling varied from weak to strong, with no consistent pattern of staining intensity (Fig. 1I).
Cerebellum
In the cerebellum, strong nuclear staining was seen in most neuroglial cells. Neuronal cells, conversely, were predominantly unlabeled, with occasional strongly labeled neuronal nuclei, as seen in the midbrain. Purkinje cell nuclei were strongly to moderately labeled, whereas their cytoplasm was weakly, but specifically labeled (Fig. 1J; Supplementary Table 3).
Pituitary
In the anterior lobe of the pituitary, glandular and follicular epithelial cell nuclei were mostly strongly labeled, whereas, in the posterior lobe, pituicyte nuclei were moderately labeled. A weak, but specific cytoplasmic staining was diffusely present in the pars distalis (Fig. 1K inset; Supplementary Table 4).
Mammary Gland
Within the mammary lobules, cuboidal to columnar epithelial cells lining the small tubules had moderately to strongly labeled nuclei. Their outer layer of myoepithelial cells was mainly unlabeled. A specific, weak-to-moderate cytoplasmic staining was also seen in the glandular epithelium of the mammary gland (Fig. 2A; Supplementary Table 5). In the connective tissue, fibrocytes were mainly unlabeled. The glandular cell nuclei labeling seen in the intra- and interlobular ducts was weaker than that seen in the lobules. Nuclei in small intralobular ducts cells were mostly weakly to moderately labeled, whereas nuclei in cells of larger interlobular ducts were weakly labeled or unlabeled. Myoepithelial cells of both intra- and interlobular ducts were mainly unlabeled (Fig. 2A inset).
Breast Skin
In the epidermis, strongly labeled nuclei were abundant in the stratum basale, whereas the middle layers (stratum spinosum and stratum granulosum) had mostly moderately stained nuclei, and the apical layers (stratum corneum) had weak or unlabeled nuclei (Fig. 2B). Within the dermis, fibrocytes were generally unlabeled, whereas the internal and external root sheaths of the hair follicles had mainly moderately to strongly labeled nuclei, and a weak, but specific cytoplasmic staining. Hair shafts were unlabeled, and all components of the arrector pili muscles were strongly labeled (Fig. 2C; Supplementary Table 6). In the sebaceous glands, basal cell nuclei were strongly stained, immature secretory cell nuclei were weakly to moderately stained, mature secretory cell nuclei were weakly stained, and degenerating sebaceous cells were unlabeled. A specific, moderate-to-weak cytoplasmic staining was present in the basal cells and immature secretory cells, respectively (Fig. 2D). In the sweat glands, about half of the secretory cell nuclei were moderately to strongly labeled, whereas the other half were unlabeled. Moderate cytoplasmic staining was also seen in these cells. An unusual feature detected in these cells was the presence of a linear labeling at the base of the secretory cells, which most likely represents cytoplasmic labeling of the myoepithelial cells. The thin spindle-shaped myoepithelial cells surrounding the secretory cells were unlabeled (Fig. 2E).
Skin
The labeling pattern in the skin was comparable with that of the breast skin, with a few notable differences (Fig. 2F–I). In the epidermis, cell nuclei were more strongly labeled in the basal lamina compared with the apical layers, but the overall labeling was less prominent than in the breast skin tissue. Multifocally within the epidermis, there were occasional epithelial cells with specific weak cytoplasmic staining (Fig. 2F; Supplementary Table 7). Cytoplasm labeling of the sweat gland secretory cells was slightly weaker than in the breast skin (Fig. 2I). Contrary to the breast skin, the cytoplasmic staining found in the hair follicle and sebaceous glands of the skin was found to be nonspecific by the control section (Fig. 2G’ and H’).
Ovary
In the ovary, strong nuclear labeling was detected in most oocytes of the primordial follicles, whereas their nucleoli were weakly labeled. Moderate cytoplasmic staining was also present in these cells. The flattened squamous epithelial cell nuclei surrounding the follicles varied from strongly labeled to unlabeled (Fig. 3A and B; Supplementary Table 8). The oocytes of the primary and second primary (growing) follicles had similar staining to those of the primordial follicle oocytes (Fig. 3A, C, and D). Almost all nuclei of the single-cell layer of cuboidal granulosa cells surrounding the oocytes of the primary follicles were strongly labeled (Fig. 3A inset). The multiple layers of granulosa cells surrounding the second primary (growing) follicles also had a strong nuclear labeling and a weak, but specific cytoplasmic staining (Fig. 3C and D). In small second primary (growing) follicles, most of the theca interna cell nuclei were unlabeled, whereas in large second primary follicles, most nuclei were moderately to strongly labeled, and their cytoplasm was weakly but specifically stained (Fig. 3C and D, respectively). The secondary (vesicular) follicles, characterized by the presence of an antrum at their centers, had moderately to strongly labeled granulosa cell nuclei, and a specific, weakly stained cytoplasm. The theca interna and externa cell nuclei had the same staining pattern as the granulosa cells (Fig. 3E). The ovarian stroma staining varied highly, with fibrocyte nuclei ranging from strongly labeled to unlabeled (Fig. 3A, B, C, and E). In degenerating atretic follicles, strong nuclear staining, and a specific, weak-to-moderate cytoplasmic staining was seen in most granulosa cells. Nuclear staining intensity decreased moving toward the follicle’s center, where many cells were unlabeled (Fig. 3F). In the corpus luteum, around half of the granulosa lutein cell nuclei were moderately labeled. Their weakly labeled cytoplasm was found to be nonspecific staining by the control section (Fig. G and G’). Endothelial cell nuclei from capillaries in the corpus luteum were mostly unlabeled (Fig. 3G, inset).
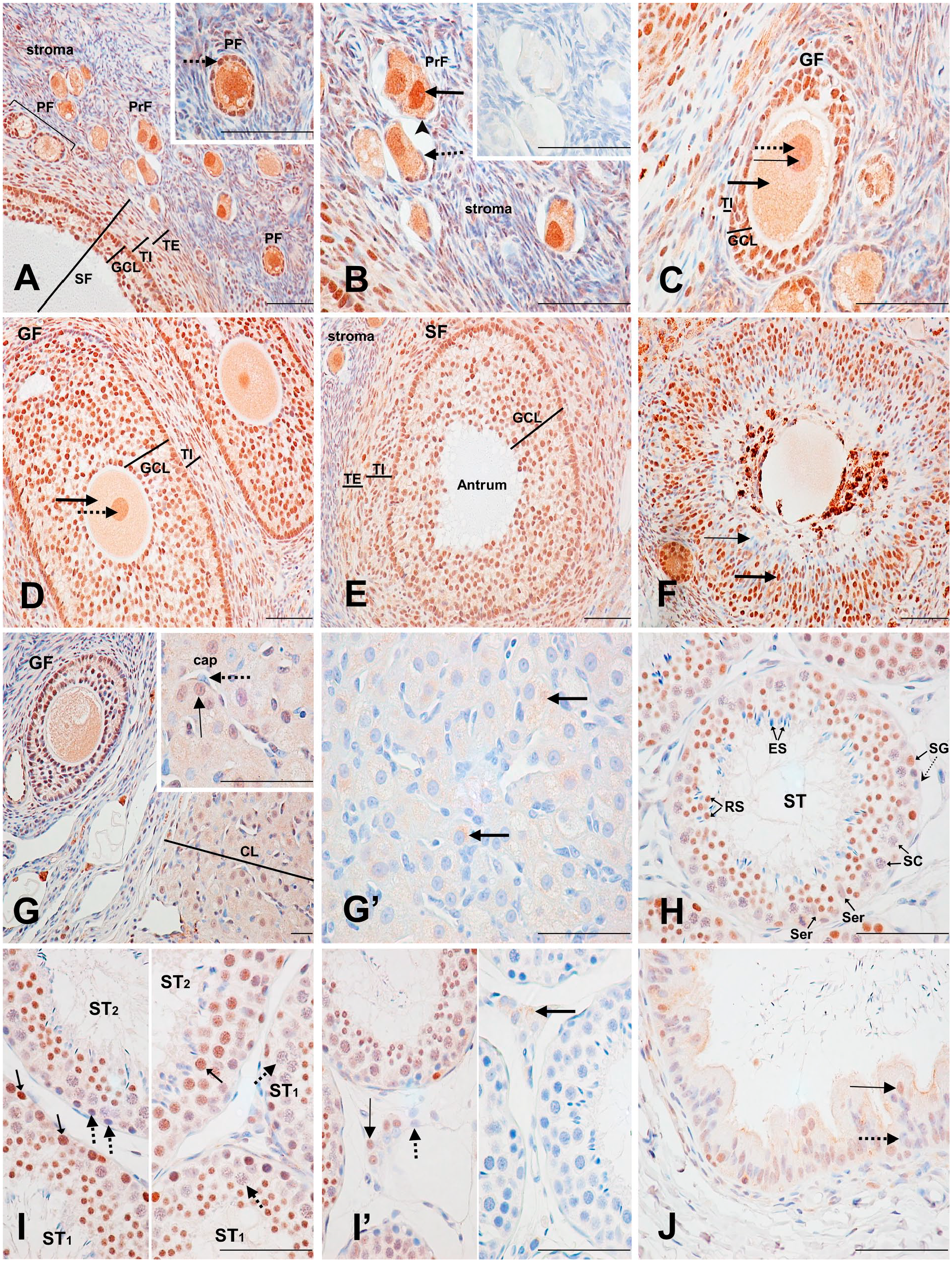
Immunodetection of PARP-1 in the ovary, testis, and efferent ductules. (A) Ovary: Overview of the staining patterns of the stroma and the different follicles found in the ovary: primordial follicle (PrF), primary follicle (PF), and secondary (vesicular) follicle (SF). The inset shows a PF with a strongly stained oocyte nucleus, weakly labeled nucleolus, and strongly stained cuboidal granulosa cell nuclei (dotted arrow). The staining of the PrF and the SF is described in (B) and (E), respectively. From (A–D): granulosa cell layer (GCL), theca interna (TI), theca externa (TE). (B) Ovary: primordial follicles. Strongly labeled oocyte nucleus (thick arrow), with a weakly labeled nucleolus. Unlabeled (dotted arrow) and moderately (arrowhead) labeled flattened squamous epithelial cell nuclei. The inset shows the control section. (C) Ovary: second primary (growing) follicle (GF). Strongly stained oocyte nucleus (dotted arrow), weakly labeled nucleolus (thin arrow), and moderately stained cytoplasm (thick arrow). The GCL shows strongly labeled nuclei, with weakly labeled cytoplasm, and the TI shows mainly unlabelled nuclei. (D) Ovary: large second primary (growing) follicle. Arrows show the same objects as in (B); however, the nucleolus is not observed. TI and GCL show mainly strongly labeled nuclei, with weakly stained cytoplasm. (E) Ovary: secondary (vesicular) follicle. The GCL, TE, and TI show mainly strongly labeled nuclei with weakly labeled cytoplasm. Antrum is unlabeled. (F) Ovary: atretic follicle: Strongly labeled (thick arrow) and unlabeled (thin arrow) granulosa cell nuclei. (G) Ovary: corpus luteum (CL). Moderately stained granulosa lutein cell nucleus (inset, thin arrow). Unlabeled endothelial cell nucleus (inset, dotted arrow) of a capillary (cap). (G’) Ovary: Nonspecific cytoplasmic staining of lutein cells cytoplasm (arrows). (H) Testis: seminiferous tubules (ST). Unlabeled elongated spermatids (ES). Strongly labeled round spermatids (RS). Unlabeled spermatocytes (SC). Strongly labeled and unlabeled spermatogonia (SG; small arrow and dotted arrow, respectively). Weakly labeled Sertoli cells (Ser). (I) Testis. The left inset shows strongly labeled (small arrows) and unlabeled (dotted arrows) spermatogonia cell nuclei. The right inset shows strongly labeled (small arrow) and unlabeled (dotted arrows) spermatocyte cell nuclei. ST1 (Seminiferous Tubule 1); ST2 (Seminiferous Tubule 2). (I’) Testis: The left inset shows moderately stained (thin arrow) and unstained (dotted arrow) Leydig cell nuclei. The control section (right inset) shows nonspecific cytoplasmic staining in Leydig cells (thick arrow). (J) Efferent ductules: Moderately labeled (thin arrow) and unlabeled (dotted arrow) pseudostratified columnar epithelial cell nuclei with weakly stained cytoplasm. Scale bar = 50 µM. Abbreviation: PARP-1, poly(ADP-ribose) polymerase-1.
Testis
Several dozens seminiferous tubules (STs) from two male monkeys were examined to assess the labeling at different meiosis stages (Fig. 3H; Supplementary Table 9). Depending on the analyzed ST, staining of the spermatogonia that line their basal lamina, varied in number and intensity, ranging from strongly stained to unstained (Fig. 3I, left inset). The labeling pattern of primary spermatocytes differed according to the simultaneous presence of round (early) or elongated (mature) spermatids in each ST. Spermatocyte nuclei were generally unlabeled when round spermatids were present, even if elongated spermatids were simultaneously present. In contrast, most spermatocyte nuclei were strongly stained when round spermatids were absent (Fig. 3I, right inset). Most round spermatids were strongly labeled, whereas elongated spermatids and Sertoli cells were unlabeled (Fig. 3H). The labeling of Leydig cells nuclei ranged from moderately labeled to unlabeled (Fig. 3I’, left inset). Weak cytoplasmic staining seen in some Leydig cells was found to be nonspecific (Fig. 3I’, right inset).
Efferent Ductules
The number of labeled cells and nuclear staining intensity of the ductuli efferentes’ pseudostratified epithelium increased from weak to moderate with increasing proximity to the ductus epididymis area. These cells also presented a weak, but specific, cytoplasmic staining (Fig. 3J; Supplementary Table 10).
Epididymis
At the entrance of the epididymal head, about half of the principal cell nuclei were moderately labeled. Moderate cytoplasmic staining was also seen in these cells (Fig. 4A; Supplementary Table 11). Toward the middle of the epididymal head, almost all principal cell nuclei were strongly labeled, and their cytoplasm was specific and weakly labeled. Occasionally, cytoplasmic staining was strongly stippled (Fig. 4B). In the epididymal body, principal cell nuclei and their cytoplasm were strongly labeled (Fig. 4C). The staining pattern in the epididymis tail was similar to that found in the epididymal body. However, closer to the ductus deferens, the staining of the tail was more similar to that of the entrance of the epididymal head (Fig. 4D and E, respectively). Throughout the epididymis, basal cells were mostly weakly labeled or unlabeled, with rare strongly stained nuclei (Fig. 4A–D). The smooth muscle cell layer surrounding the luminal epithelial cells was primarily unlabeled throughout the epididymis, except for in the tail, where areas of strong cytoplasmic staining and, to a lesser extent, nuclear staining were seen (Fig. 4A–E). Blood vessels in the epididymis had mostly unlabeled endothelial cells in the tunica intima, unlabeled smooth muscle cell nuclei in the tunica media, and unlabeled fibrocytes in the tunica adventitia. Strong cytoplasmic staining was also seen in the tunica media (Fig. 4F).
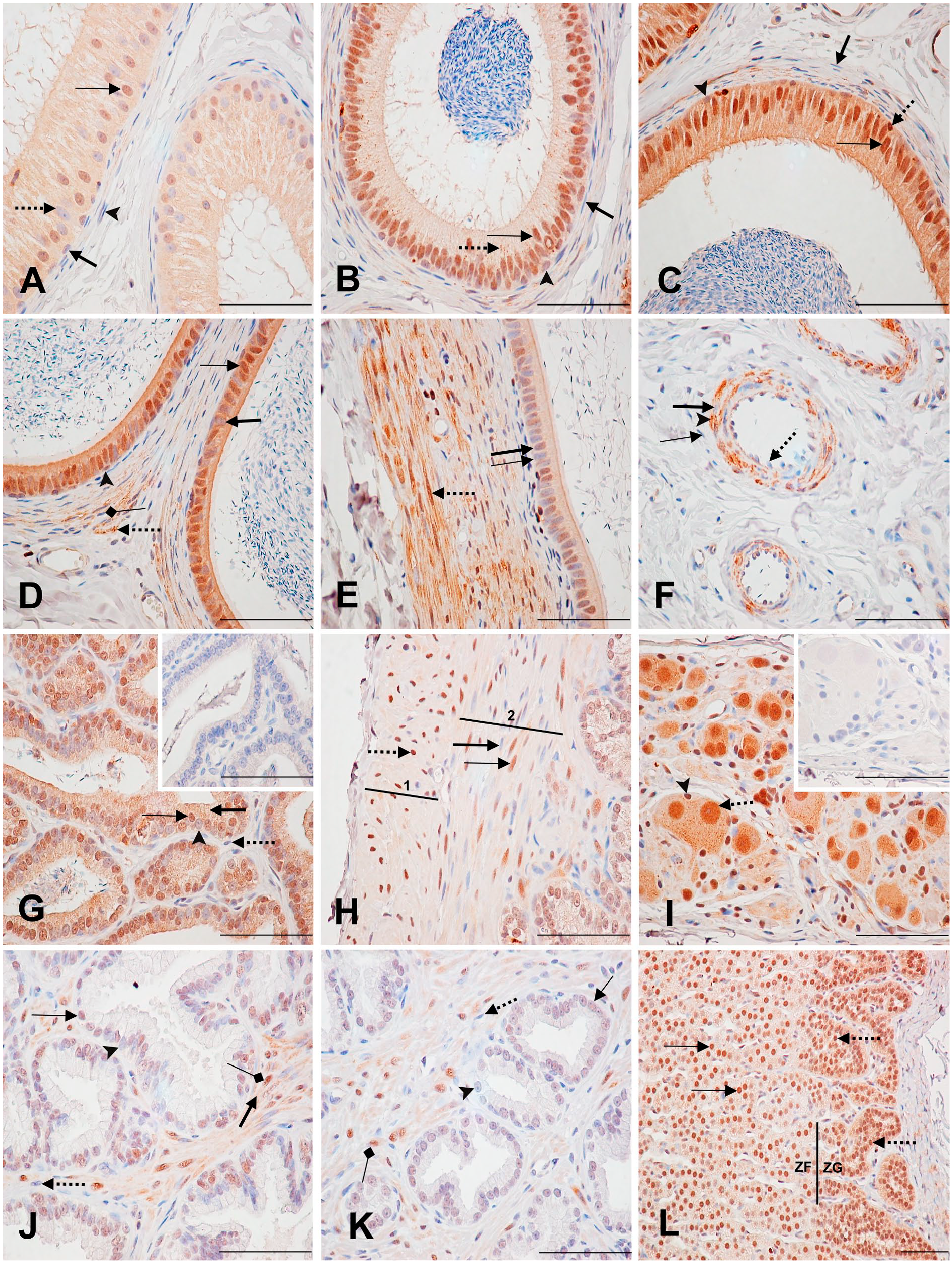
Immunodetection of PARP-1 in the epididymis, seminal vesicle, prostate, and adrenal. (A) Epididymis head: Moderately labeled (thin arrow) and unlabeled (dotted arrow) principal cell nuclei with moderately stained cytoplasm. Unlabeled (thick arrow) basal cell nucleus. Unlabeled smooth muscle cell nucleus (arrowhead). (B) Epididymis head (middle): Strongly stained principal cell nucleus (thin arrow) with a weakly stained cytoplasm. Weakly labeled basal cell nucleus (arrowhead). Stippled cytoplasmic staining (dotted arrow). Unlabeled smooth muscle cell nucleus (thick arrow). (C) Epididymal body: Strongly stained principal cell nucleus (thin arrow) with a strongly stained cytoplasm. Weakly (arrowhead) and strongly labeled (dotted arrow) basal cell nuclei. Unlabeled smooth muscle cell nuclei (thick arrow). (D) Epididymal tail: Strongly stained (thin arrow) and weakly stained (thick arrow) principal cell nuclei with strong cytoplasmic staining. Weakly labeled basal cell nuclei (arrowhead). Unlabeled smooth muscle cell nucleus (squared arrow) with a strongly labeled stippled cytoplasmic (dotted arrow). (E) Epididymal tail: Unlabeled (thin arrow) and moderately labeled (thick arrow) principal cell nuclei. Strongly labeled smooth muscle cell nucleus and cytoplasm (dotted arrow). (F) Epididymis: Blood vessels. Unlabeled nucleus (thick arrow) and strong stippled cytoplasmic staining (arrowhead) of a smooth muscle cell. Unlabeled endothelial cell nucleus (dotted arrow). Unlabeled fibroblast (thin arrow). (G) Seminal vesicle: Strongly stained (thin arrow) and weakly labeled (arrowhead) glandular epithelial nuclei with a moderately stained cytoplasm (thick arrow). Unlabeled stromal cell nucleus (dotted arrow). Inset shows the control section. (H) Seminal vesicle: Strongly stained outer longitudinal (1) smooth muscle cell nucleus (dotted arrow). Strongly stained (thin arrow) and unstained (thick arrow) inner circular (2) smooth muscle cell nuclei. Both smooth muscle cell layers (1 and 2) show a weakly labeled cytoplasmic layer. (I) Seminal vesicle: terminal ganglia. Strongly stained neuronal nucleus (dotted arrow), with a moderately labeled stippled cytoplasmic staining and a weakly labeled nucleolus. Strongly labeled satellite cell nucleus (arrowhead). Inset shows the control section. (J) Prostate: cranial lobe. Moderately stained (thin arrow) and unstained (arrowhead) epithelial cell nuclei. Unstained (dotted arrow) and strongly stained (squared arrow) stromal cell nuclei with a weakly stained cytoplasm (thick arrow). (K) Prostate: caudal lobe. Weakly labeled (thin arrow) and unlabeled (arrowhead) epithelial cell nuclei. Unstained (dotted arrow) and strongly stained (squared arrow) stromal cell nuclei, with weak cytoplasmic staining. (L) Male adrenal: Zona glomerulosa (ZG) with strongly labeled nuclei (dotted arrows) and a moderately labeled cytoplasm. Zona fasciculata (ZF) with strongly labeled nuclei (thin arrows) and a weakly labeled cytoplasm. Scale bar = 50 µM. Abbreviation: PARP-1, poly(ADP-ribose) polymerase-1.
Seminal Vesicle
In the seminal vesicle, approximately half of the glandular epithelial cell nuclei and cytoplasm were strongly stained (Fig. 4G; Supplementary Table 12). Stromal cells were generally unlabeled, and most smooth muscle cells of the muscular layer had strongly labeled nuclei and a weak, but specific cytoplasmic labeling (Fig. 4G and H). Of note, while most of the outer longitudinal smooth muscle cell nuclei were strongly labeled, a lower proportion of the inner circular smooth muscle cells were labeled (Fig. 4H). Most neuronal nuclei in terminal ganglia found in the seminal vesicle were strongly labeled, whereas their nucleoli were weakly labeled. Moderate cytoplasmic staining was also present in these cells. Most of the satellite cell nuclei were also strongly labeled (Fig. 4I).
Prostate
In the cranial portion of the prostate, epithelial cell nuclei varied from moderately labeled to unlabeled. About half of the stromal cell nuclei were strongly labeled (Fig. 4J; Supplementary Table 13). In the caudal portion of the prostate, epithelial cell nuclei were weakly labeled or unlabeled. Stromal cells had the same staining as in the cranial portion, along with a specific, weak cytoplasmic staining in both the cranial and caudal portions (Fig. 4J and K). Overall, there was stronger staining in the epithelial cells of the cranial area compared with the caudal area.
Adrena
The adrenal is the only organ in this study for which results are presented separately for male and female monkeys, due to significant sex differences in nuclear and cytoplasmic staining intensities (Supplementary Table 14). In the zona glomerulosa of the male, almost all nuclei were strongly labeled, whereas in the female nuclei were more moderately labeled. A stippled pattern characterized the nuclear staining (not shown), and moderate cytoplasmic staining was seen in the male zona glomerulosa. In the female, on the contrary, cytoplasmic staining was nonspecific. The zona fasciculata nuclei had a similar staining pattern as the zona glomerulosa, and the cytoplasmic staining was weak, but specific (Figs. 4L and 5A). In the zona reticularis of the male, most nuclei were strongly labeled with a stippled pattern (not shown). The remaining were unlabeled. A specific, weak cytoplasmic staining was diffusely present in these cells. In the female, the staining pattern of the zona reticularis was similar to the male, but the staining intensity was much weaker (Fig. 5B and B’). Their moderately stained cytoplasm was found to be nonspecific (5B’ inset). Last, in the male adrenal medulla, almost all chromaffin cells had a strong nuclear and cytoplasmic staining (Fig. 5B). Depending on the zone, a few areas of the medulla had very weak or absent nuclear and cytoplasmic labeling (not shown). In the female, chromaffin cell nuclei were mainly moderately to strongly labeled, occasionally with a stippled pattern (not shown), and their cytoplasm was very weakly but specifically labeled (Fig. 5B’). Overall, nuclear and cytoplasmic staining intensity was stronger in the male tissues relative to the female; however, proportions of stained cells were similar.
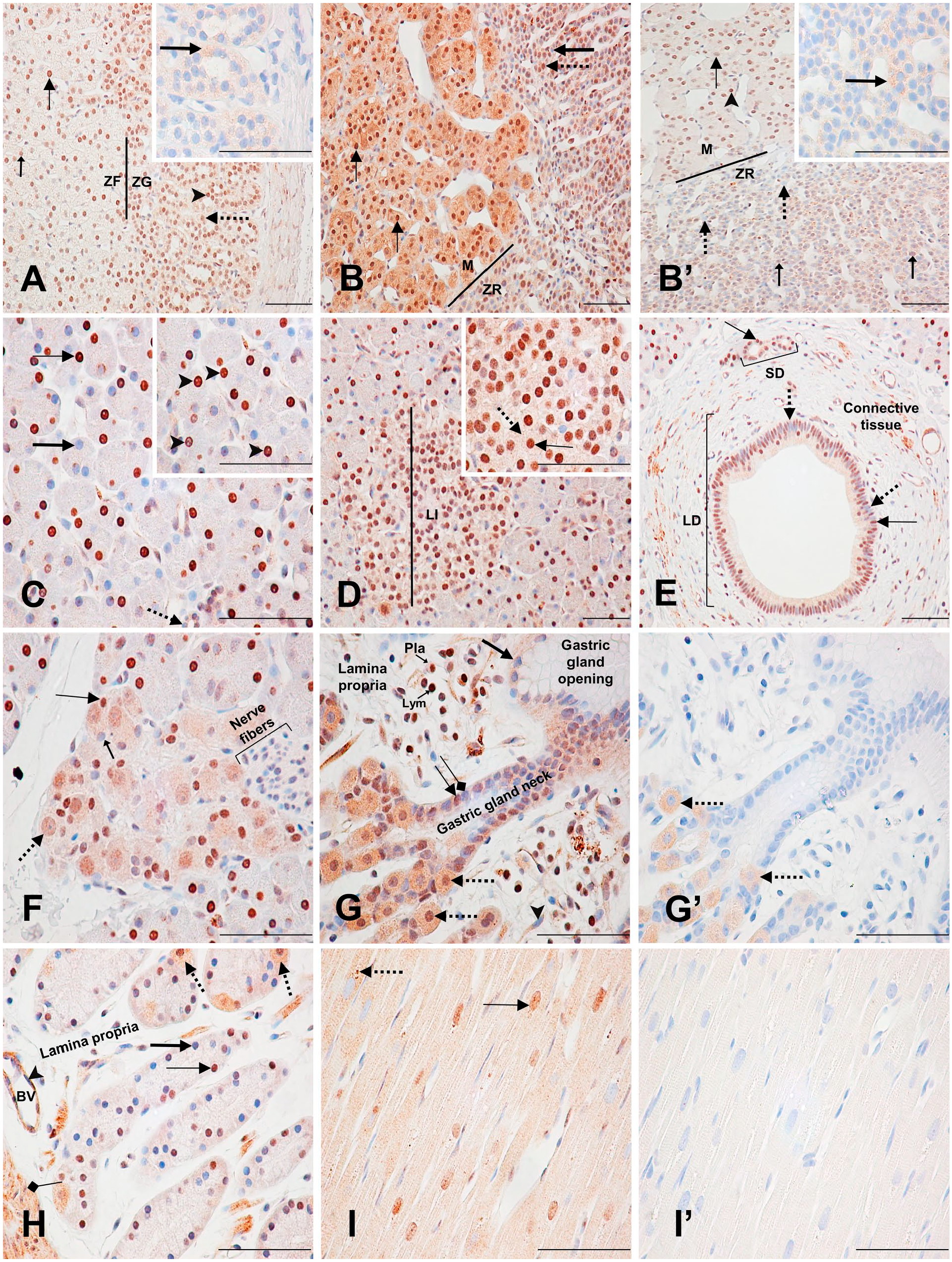
Immunodetection of PARP-1 in the adrenal, pancreas, stomach, and heart. (A) Female adrenal: Zona glomerulosa with moderately (dotted arrows) and strongly (arrowhead) labeled nuclei and a weakly labeled cytoplasm. Zona fasciculata with moderately (small arrow) and strongly (thin arrow) labeled nuclei with a very weakly labeled cytoplasm. Inset shows the nonspecific cytoplasmic staining (thick arrow) of the zona glomerulosa. (B) Male adrenal: Zona reticularis (ZR) with strongly labeled (dotted arrow) and unlabeled (thick arrow) nuclei with a weakly stained cytoplasm. Medulla (M) with strongly stained (thin arrows) nuclei and cytoplasm. (B’) Female adrenal: Zona reticularis with weakly labeled (small arrows) and unlabeled (dotted arrows) nuclei and a very weakly stained cytoplasm. Medulla with strongly stained (arrowhead) and weakly stained (thin arrow) nuclei with a very weakly stained cytoplasm. Inset shows the nonspecific cytoplasmic staining of the zona reticularis (thick arrow). (C) Pancreas: Unlabeled (thick arrow) and strongly labeled (thin arrow) secretory cell nuclei. Unlabeled small cuboidal intralobular ductal cell nucleus (dotted arrow). Inset shows small circular nucleolus-like unlabeled areas in secretory cell nuclei (arrowheads). (D) Pancreas: Strongly labeled Langerhans’s islet (LI) cell nucleus (inset, thin arrow) with a weakly stained cytoplasm (inset, dotted arrow). (E) Pancreas: Small (SD) and large (LD) interlobular ducts. Strongly labeled (thin arrows) and unlabeled (dotted arrows) nuclei with a weakly labeled cytoplasm. (F) Pancreas: terminal ganglia. Moderately labeled neuronal nucleus (dotted arrow) with a weakly stained cytoplasm and an unlabeled nucleolus. Strongly labeled (thin arrow) and unlabeled (small arrow) satellite cell nuclei. Nerve fibers are unlabeled. (G) Fundic stomach: Unlabeled foveolar cell nucleus (thick arrow) with a weakly labeled cytoplasm. Unlabeled (squared-end arrow) and strongly labeled (thin arrow) mucous neck cell nuclei with a moderately labeled cytoplasm. Strongly labeled parietal cell nuclei (dotted arrows). Strongly labeled plasmocyte (Pla) and lymphocyte (Lym). Weakly labeled fibrocyte nucleus (arrowhead) with a weakly labeled cytoplasm. (G’) Fundic stomach: Nonspecific cytoplasmic staining of the parietal cells (dotted arrows). (H) Fundic stomach: fundic glands. Strongly labeled (thin arrow) and unlabeled (thick arrow) epithelial chief cell nuclei. Parietal cells (dotted arrows) as described in (G) and (G’). Unlabeled endothelial cell nucleus (arrowhead) of a blood vessel (BV). Weakly labeled smooth muscle cell nucleus (squared arrow) with a moderately labeled stippled cytoplasm. (I) Heart: ventricle. Moderately labeled (thin arrow) longitudinal cardiac myocyte nucleus with a weakly labeled cytoplasm. Strongly labeled stippled cytoplasmic pattern (dotted arrow). (I’) Control section of (I). Scale bar = 50 µM. Abbreviations: PARP-1, poly(ADP-ribose) polymerase-1; ZF, Zona fasciculate; ZG, Zona glomerulosa.
Pancreas
In the exocrine pancreas, about two thirds of the secretory cells forming the serous acini of the pancreatic parenchyma had a strong nuclear labeling and an unlabeled nucleolus (Fig. 5C; Supplementary Table 15). In the endocrine pancreas, the Langerhans’s islet cell nuclei were mainly strongly labeled, and their cytoplasm was weakly but specifically labeled (Fig. 5D). Staining of the nuclei of the small cuboidal intralobular duct cells varied from weakly labeled to unlabeled, whereas nuclei of both small and large interlobular duct cells were mainly strongly labeled. Both cell types had a specific, weak cytoplasmic staining, and the large interlobular ducts pattern was also stippled (not shown) (Fig. 5C and E). In the peripheral ganglions localized within the pancreatic parenchyma, the neuronal nuclei were moderately labeled, their nucleoli were unstained, and their cytoplasm was specifically and weakly labeled. Satellite cell nuclei staining varied from strongly labeled to unlabeled. Neither the nerve fibers located next to the ganglions nor their protective satellite cell nuclei were labeled (Fig. 5F).
Stomach
In this study, only the stomach fundus was examined (Supplementary Table 16). The cardiac and pyloric stomach were not available for review. The gastric surface foveolar cell nuclei were mostly unlabeled with a weak but specific cytoplasmic labeling, whereas the labeling of fundic glands mucous neck cell nuclei ranged from strongly labeled to unlabeled and their cytoplasm was moderately labeled. Throughout the fundic gland, parietal cell nuclei were nearly universally strongly labeled, but their moderate cytoplasmic labeling was found to be nonspecific (Fig. 5G and G’). Less than half of the fundic gland epithelial chief cell nuclei were strongly stained, whereas the rest was mostly unlabeled (Fig. 5H). Strongly labeled lymphocytes and plasmocytes were also seen in the lamina propria, along with mostly unlabeled fibrocytes (Fig. 5G). Endothelial cell nuclei of blood vessels in the stomach were mainly unlabeled, whereas their cytoplasm was moderately labeled. Similarly, smooth muscle cell nuclei of the muscularis mucosa were mostly weakly labeled, whereas their cytoplasm was moderately labeled with a stippled pattern (Fig. 5H).
Heart
In the ventricle, most striated cardiac muscle cell nuclei were moderately labeled with a specific, weak cytoplasmic staining (Figs. 5I and 6A; Supplementary Table 17). In the atrium, striated muscle cell nuclei were mainly strongly labeled, and their cytoplasm was moderately stained (Fig. 6B). A strong, stippled cytoplasmic labeling was detected throughout the heart tissue sections (Figs. 5I, 5I’, 6A and B).
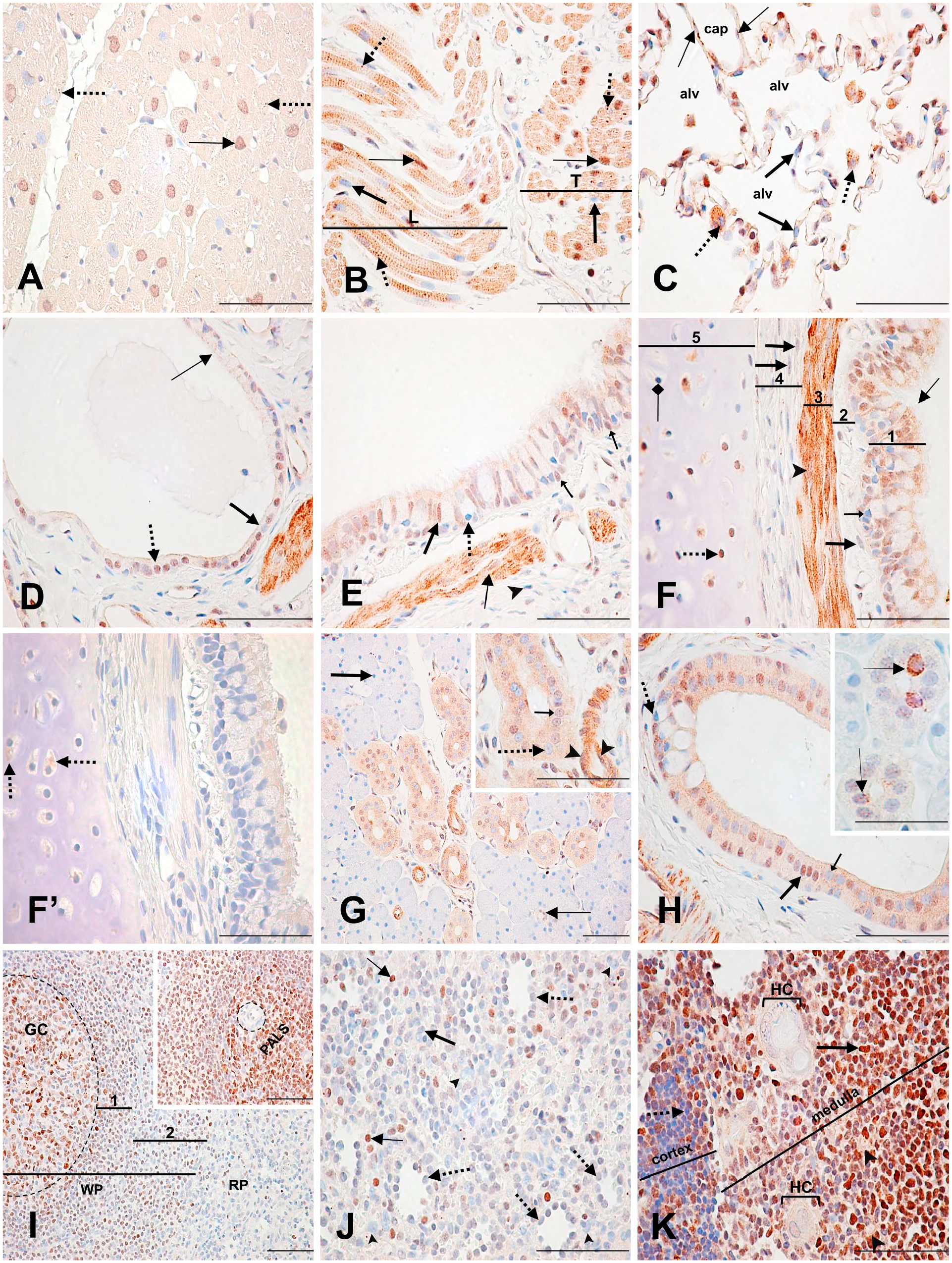
Immunodetection of PARP-1 in the heart, lung, salivary gland, spleen, and thymus. (A) Heart: ventricle. Moderately labeled transverse cardiac myocyte nucleus (thin arrow) with a weakly labeled cytoplasm and a moderately labeled stippled pattern (dotted arrows). (B) Heart: atrium. Strongly labeled (thin arrows) and unlabeled (thick arrows) longitudinal (L) and transverse (T) striated muscle cell nuclei. Strong, stippled cytoplasmic labeling (dotted arrows). (C) Lung: alveoli (alv). Unlabeled type I pneumocytes (thick arrows). Unlabeled macrophage nuclei (dotted arrows) with a strongly stippled cytoplasm. Weakly labeled endothelial cell nuclei (thin arrows). (D) Lung: high respiratory bronchiole. Moderately labeled (dotted arrow) and unlabeled (thick arrow) cuboidal epithelial cell nuclei. Unlabeled ciliated epithelial cell nuclei (thin arrow). (E) Lung: low (distal) bronchus. Moderately stained pseudostratified ciliated columnar epithelial cell nucleus (thick arrow) with weakly stained cytoplasm. Unlabeled goblet cell nucleus (dotted arrow). Moderately labeled basal cell nuclei (small arrows). Weakly stained smooth muscle cell nuclei with a strongly stained cytoplasm (thin arrow). Unlabeled fibrocyte nucleus (arrowhead). (F) Lung: bronchus. (1) Moderately labeled pseudostratified ciliated columnar epithelial cell nucleus and cytoplasm (thin arrow), and unlabeled goblet cell nucleus (small arrow) in the epithelium. (3) Moderately labeled smooth muscle cell nucleus (arrowhead) with a strongly labeled cytoplasm in the smooth muscle cell layer. Unlabeled fibrocyte nuclei with moderately stippled staining (thick arrows) in (2) the lamina propria and (4) the submucosa. (5) Unlabeled (square-shaped arrow) and strongly labeled (dotted arrow) chondrocyte nuclei in the cartilage. (F’) Lung: Nonspecific cytoplasmic staining of the chondrocyte cytoplasm (dotted arrows). (G) Salivary gland: serous acini and intralobular ducts. Unlabeled serous acini cell nucleus (thick arrow). Unlabeled (inset, dotted arrow) and weakly labeled (inset, small arrow) striated duct cell nuclei. Weakly labeled intercalated duct cell nucleus (thin arrow). Moderately labeled endothelial cell nuclei with a strongly stained cytoplasm (inset, arrowheads). (H) Salivary gland: interlobular duct. Strongly stained (thick arrow) and unstained (small arrow) tall columnar epithelial cell nuclei. Unlabeled goblet cell nucleus (dotted arrow). Inset shows intralobular striated duct cells with a strongly stained stippled or ring-like nuclear pattern (thin arrows). (I) Spleen: White pulp (WP) shows a germinal center (GC) with mostly strongly to moderately labeled lymphocyte nuclei. (1) Mantle zone and (2) marginal zone show mostly unlabeled or weakly labeled B lymphocytes. Red pulp (RP) shows unlabeled lymphocyte nuclei. Inset shows a trabecular arteriole (dotted circle) with unlabeled endothelial and trabeculae cells, surrounded by strongly labeled T lymphocyte nuclei of the periarteriolar lymphatic sheath (PALS). (J) Spleen: red pulp. Unlabeled endothelial cell nuclei of the splenic sinusoids (dotted arrows). Unlabeled macrophages (arrowheads). Strongly labeled (thin arrow) and unlabeled (thick arrow) lymphocytes. (K) Thymus: Unlabeled cortical lymphocyte nucleus (dotted arrow). Strongly stained medullary lymphocyte nucleus (thick arrow). Unlabeled epithelial reticular cell nuclei (arrowheads). Unlabeled flattened epithelial cell nuclei of the Hassall’s corpuscles (HC). Scale bar = 50 µM. Abbreviation: PARP-1, poly(ADP-ribose) polymerase-1.
Lung
In the lung alveoli, type I pneumocyte nuclei were mainly unlabeled. Macrophage nuclei were unlabeled, whereas their cytoplasm was strongly stippled. Occasionally, type I pneumocytes also had strong cytoplasmic staining. Capillaries in the alveoli had unlabeled or weakly labeled endothelial cell nuclei (Fig. 6C; Supplementary Table 18). In the respiratory bronchiole, about half of the cuboidal epithelial cell nuclei were moderately labeled, whereas ciliated epithelial cell nuclei were mostly unlabeled (Fig. 6D). In the low (distal) bronchus area, the pseudostratified ciliated columnar epithelium nuclei and cytoplasm were weakly to moderately labeled, whereas goblet cell nuclei were not labeled. Basal cell nuclei were weakly to moderately labeled and fibrocytes were mostly unlabeled. Smooth muscle cell nuclei were weakly labeled, whereas their cytoplasm was strongly labeled (Fig. 6E). In the thick pseudostratified ciliated columnar epithelium of the bronchus, most cell nuclei and cytoplasm were moderately labeled. Goblet cells were unlabeled, as well as the majority of the fibrocytes. Some fibrocyte nuclei were moderately stippled. The layers of smooth muscle cells surrounding the lamina propria had strong cytoplasmic staining and moderately labeled to unlabeled nuclei. In the cartilage, about half of the chondrocyte nuclei were strongly labeled, but their moderately stained cytoplasm was found to be nonspecific by the control tissue section (Fig. 6F and F’).
Salivary Glands
In the parotid salivary gland, almost all serous acini cell nuclei were unlabeled. The small intralobular intercalated duct cell nuclei were mainly weakly labeled. Most of the intralobular striated duct columnar cell nuclei and cytoplasm were weakly but specifically labeled. The capillaries surrounding the striated ducts had moderately labeled endothelial cell nuclei and strongly stained cytoplasm (Fig. 6G; Supplementary Table 19). Some striated duct cells had a strongly labeled perinuclear ring-like or stippled pattern (Fig. 6H, inset). About half of the tall columnar epithelial cell nuclei of the interlobular ducts were moderately to strongly labeled, and their cytoplasm was weakly, but specifically labeled. Goblet cells were mainly unlabeled (Fig. 6H).
Spleen
The splenic capsule was not available for review in this study. In the white pulp, splenic follicles had varied staining. In their germinal centers, B lymphocytes (centrocytes and centroblasts) were mostly strongly to moderately stained, whereas follicular dendritic cells were weakly labeled. Tingible body macrophages were unlabeled (Fig. 6I; Supplementary Table 20). The periarteriolar lymphatic sheath (PALS) surrounding the central arterioles in the splenic follicles had most of their densely packed T lymphocyte nuclei strongly labeled. Neither the endothelial cells of the central arterioles nor the trabeculae surrounding them were labeled (Fig. 6I inset). Surrounding the germinal centers, the rings of small and densely packed B lymphocytes that make up the mantle zone were generally unlabeled or weakly labeled. Between the mantle zone and the red pulp, the lymphocytes of the marginal zone were similarly labeled to the mantle zone, but with an increasing number of unlabeled lymphocyte nuclei (Fig. 6I). Depending on the splenic follicle, the staining intensity of the mantle and the marginal zones collectively varied from weak to moderate (not shown). In the red pulp, almost all lymphocyte nuclei were unlabeled. Endothelial cells surrounding the splenic sinusoids in the red pulp were mostly unlabeled. Macrophages found in the marginal zone and red pulp were also unlabeled (Fig. 6I and J).
Thymus
In the thymus, the thin connective tissue layer that makes up the capsule and septa had mostly unlabeled fibrocytes. The cortical area of the thymus lobules was densely packed with mainly unlabeled small lymphocyte nuclei; however, there were some exceptions of strongly stained nuclei. In contrast, most lobules of the medullary region had strongly stained large and small lymphocyte nuclei. The medullary epithelial reticular cell nuclei were unstained, similar to the flattened epithelial cells of the Hassall’s corpuscles (Fig. 6K; Supplementary Table 21).
Urinary Bladder
In the urinary bladder, cell nuclei of the transitional epithelium were weakly labeled, and their cytoplasm was moderately to strongly labeled. The strong intensity of the cytoplasmic staining at the epithelial surface prevented a clear analysis of the labeling of the nucleus. In the lamina propria, some fibrocyte nuclei were moderately labeled (Fig. 7A; Supplementary Table 22). The nuclei of myocytes in the smooth muscle cell layers that form the muscularis externa were strongly labeled, and occasionally stippled. A weak, but specific cytoplasmic staining was also seen in these cells (Fig. 7B). The adventitia of the urinary bladder was not available for review.
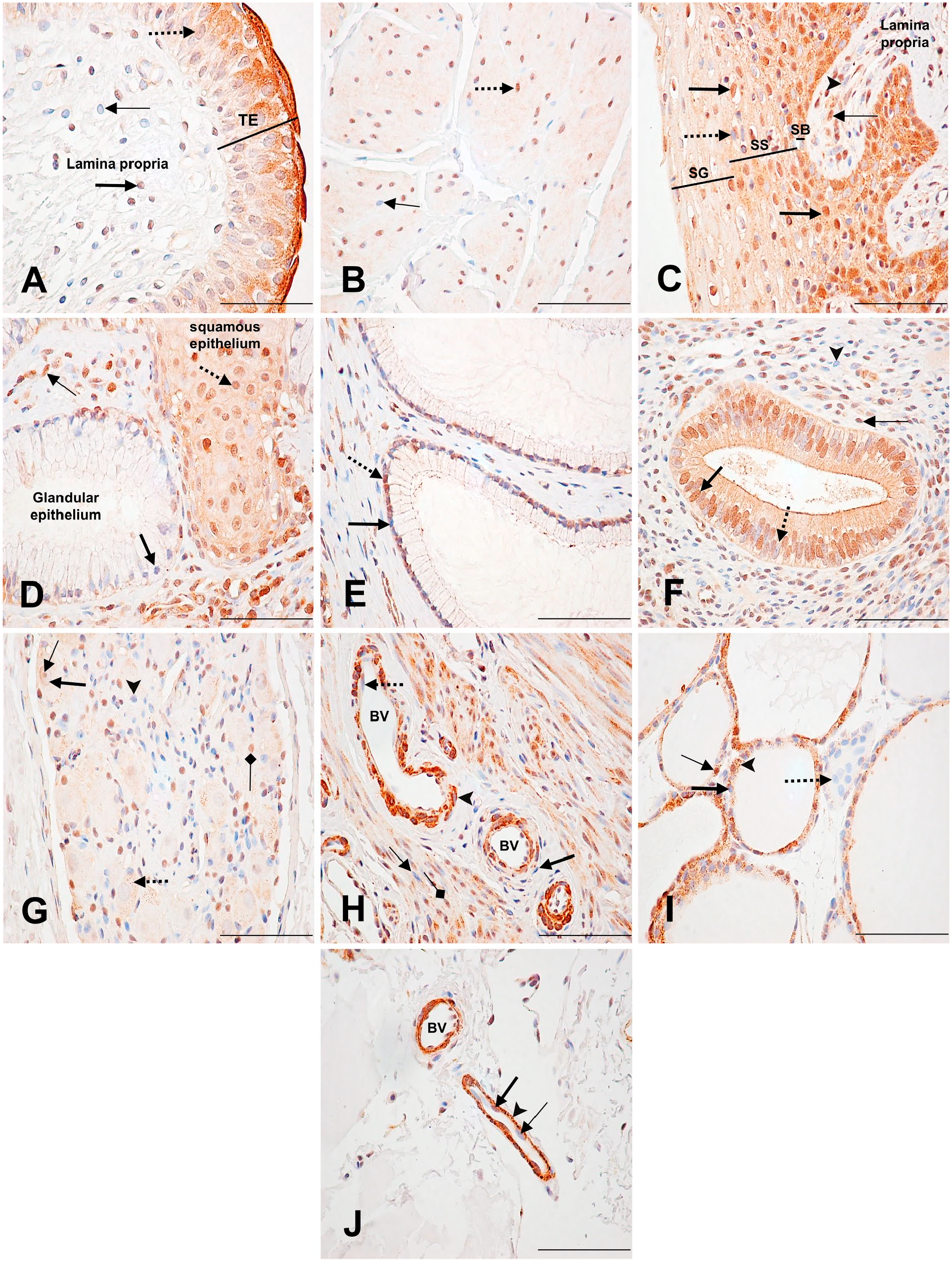
Immunodetection of PARP-1 in urinary bladder, uterus, and thyroid. (A) Urinary bladder: Weakly labeled transitional epithelial (TE) cell nucleus (dotted arrow) with a moderately labeled cytoplasm. Moderately labeled (thick arrow) and unlabeled (thin arrow) fibrocyte nuclei in the lamina propria. (B) Urinary bladder: inner longitudinal layer of the muscularis externa. Strongly stained (dotted arrow) and unstained (thin arrow) smooth muscle cell nuclei, with weak cytoplasmic labeling. (C) Uterus: ectocervix. Strongly labeled (thick arrow) and unlabeled (dotted arrow) stratified squamous epithelial cell nuclei. Strongly labeled (thin arrow) and unlabeled (arrowhead) fibrocyte nuclei. SB (stratum basale); SS (stratum spinosum); SG (stratum granulosum). (D) Uterus: cervix transformation zone. Moderately labeled squamous epithelial cell nucleus (dotted arrow) and cytoplasm. Unlabeled glandular epithelial cell nucleus (thick arrow). Strongly labeled fibrocyte nucleus (thin arrow). (E) Uterus: endocervix. Strongly stained (dotted arrow) and unstained (thick arrow) columnar epithelial cell nuclei. (F) Uterus endometrium: uterine gland. Strongly stained (thick arrow) and unstained (dotted arrow) columnar glandular epithelial cell nuclei. Strongly stained (thin arrow) and unstained (arrowhead) stromal cell nuclei. (G) Uterus: Terminal ganglia. Weakly labeled neuronal nucleus (thick arrow), with a weakly labeled nucleolus (thin arrow). Weakly stained cytoplasm with a moderately stained stippled pattern (dotted arrow). Moderately labeled (square-shaped arrow) and unlabeled (arrowhead) satellite cell nuclei. (H) Uterus myometrium: Blood vessel (BV) showing an unlabeled endothelial cell nucleus (dotted arrow), a weakly labeled smooth muscle cell nuclei (arrowhead) with a strongly labeled cytoplasm, and an unlabeled fibroblast nucleus (thick arrow). Unlabeled (thin arrow) and moderately labeled (squared arrow) uterine smooth muscle cell nuclei. (I) Thyroid: Unlabeled follicular (thick arrow) and parafollicular (dotted arrow) cell nuclei. Strongly labeled follicular cell nuclei (thin arrow). Strong, stippled cytoplasmic staining (arrowhead). (J) Thyroid: blood vessels. Unlabeled (thin arrow) and weakly labeled (thick arrow) endothelial cell nuclei. Strong, stippled cytoplasmic staining (arrowhead). Scale bar = 50 µM.
Uterus
In the cervix, the stratified squamous epithelium cell nuclei of the ectocervix were mainly strongly labeled in the stratum basale. Staining intensity became gradually weaker with apical movement through the stratum spinosum and stratum granulosum layers. The cytoplasmic staining followed the same pattern. In the subepithelial lamina propria, fibrocyte nuclei were mostly strongly labeled. Some also had a weak but specific cytoplasmic staining (Fig. 7C; Supplementary Table 23). In the transformation zone, where the squamous epithelium of the ectocervix meets the mucus-secreting columnar epithelium of the endocervix, most squamous epithelial cell nuclei and cytoplasm were moderately labeled. On the contrary, most glandular epithelial cell nuclei were not labeled. In this area, many fibrocyte nuclei in the lamina propria were also strongly labeled (Fig. 7D).
In the endocervix, about half of the columnar epithelial cell nuclei were strongly labeled (Fig. 7E). The glandular columnar epithelial cells also had similar staining. In the endometrium, most columnar glandular epithelial cells had moderate nuclear labeling, as well as moderate, stippled cytoplasmic staining. While most glands had this staining pattern, some only had weakly labeled or unlabeled nuclei (not shown). Close to half of the stromal cells surrounding these glands had strongly labeled nuclei (Fig. 7F). In contrast to other tissues, most of the neuronal nuclei in the terminal ganglia were weakly labeled or unlabeled. Their cytoplasmic staining was also weak but specific, and some also had a stippled pattern. Some satellite cell nuclei were moderately to strongly labeled (Fig. 7G). The myometrium had mainly moderately labeled muscle cell nuclei with a specific, weak-to-moderate cytoplasm staining. Blood vessels in this layer mainly had unlabeled endothelial cells in the tunica intima, unlabeled smooth muscle cell nuclei in the tunica media, and unlabeled fibroblasts in the tunica adventitia. The cytoplasm of the smooth muscle cells of the tunica media was strongly stained (Fig. 7H).
Thyroid
The thyroids’ follicular cells generally had a strong, stippled cytoplasmic staining, and unlabeled nuclei. However, rarely were nuclei strongly stained. Parafollicular cells had no staining as well (Fig. 7I; Supplementary Table 24). Endothelial cells of the capillaries surrounding the follicles had a strong, stippled cytoplasmic staining, and their nuclei were unlabeled or weakly labeled (Fig. 7J).
Discussion
This study extensively describes PARP-1 distribution and expression profiles in well-preserved monkey organs, using the thoroughly validated monoclonal antibody C2-10. 29 We report that all organs analyzed express PARP-1, but not all cell types. The strongest expression was seen in several endocrine tissues, including the pituitary, ovary, testis, and male adrenal; in some reproductive organs, including the seminal vesicle, epididymis, and uterus; and in lymphoid tissues like the thymus. The weakest expression was seen in the cerebral cortex, prostate, and efferent ductules.
The Macaca genus is closely related to humans. Gene maps of human and non-human primates are highly conserved, and therefore support a high degree of physiological similarity between species.27,39 This understanding has made the cynomolgus monkey one of the most common animal models for drug development.40,41 So, given the increased phylogenetic proximity of humans and macaques, leveraging our library of monkey tissues to assess and examine PARP-1 tissue distribution comes as an advantage, while the usage of human tissues to assess protein distribution by IHC may come with disadvantages regarding tissue handling, which may result in suboptimal specimen quality. 42
The human protein atlas (HPA) has recently provided the in situ distribution of PARP-1 in human tissues (protein score available from v18.www.proteinatlas.org). 26 Based on assessments with the PARP-1 antibody HPA045168, for which some validation of specificity is provided, we were able to compare our monkey IHC data with human PARP-1 expression. Overall, tissue protein expression is very similar between species; however, there are some noticeable differences. In the cerebral cortex of the human, neuronal cells show strong expression of PARP-1, whereas in the monkey, neuronal PARP-1 expression is almost absent. Another noticeable difference is in the follicular cells of the thyroid. In the monkey, PARP-1 expression is mostly absent, whereas in the human PARP-1, it is strongly expressed. This is also seen in several other cell types such as macrophages in the lung, Leydig cells of the testis, myoepithelial cells of the mammary gland, and the serous acini cells of the salivary glands. Moreover, the prostate and the lung also show an overall weaker expression in the monkey compared with the human. These differences may reflect species-specific distribution of PARP-1; however, they could also arise from distinct affinities of anti-PARP-1 antibody used in each study. Of note, not all cell types for each tissue of the monkey could be compared with their human counterparts because of HPA’s restricted sample size. Nevertheless, these observations suggest a relatively well-conserved distribution of PARP-1 in primates.
Moreover, our reported distribution and expression of PARP-1 in the monkey tissues are also relatively consistent with mRNA expression of PARP-1 in human tissues, which suggests PARP-1 is expressed in all tissues at least at a low level.26,43 –45 For instance, the strong PARP-1 protein expression in the pituitary and the testis, as well as in lymphoid tissues such as the thymus, is paralleled by medium-to-strong human PARP-1 mRNA expression. Exceptions to this distribution include the prostate and the cerebral cortex where we did not detect any significant PARP-1 protein expression in the monkey, whereas human PARP-1 mRNA shows moderate expression in these tissues. Our IHC data are also in agreement with PARP-1 enzymatic activity assessed in other mammals, including mice and sheep, where high activity was noted in the testis and lymphatic tissues.46,47
Interestingly, although PARP-1 is found to be expressed in all macaque tissues surveyed, it is not ubiquitously expressed in all cell types of a given tissue. Thus, we report a specific PARP-1 expression at the cell type level within each tissue. In the monkey, examples include the strong expression of PARP-1 in the glial cells of the cerebral cortex and not in neuronal cells. In addition, PARP-1 expression also varies within a cell type. This is best seen in several organs, including the skin (Fig. 2I), testis (Fig. 3I), epididymis (Fig. 4A), seminal vesicle (Fig. 4G), and pancreas (Fig. 5C). While some nuclei are strongly labeled, adjacent cell nuclei are unlabeled. This pattern is also seen in the human pancreas and seminal vesicle (HPA, www.proteinatlas.org). 26 Last, our findings further suggest a differential PARP-1 expression in specific organ’s cell types. We report that the level of PARP-1 expression in the spermatocytes of the testis depends on the presence or absence of round spermatids that the level of PARP-1 expression in the splenic follicle’s PALS depends on the follicle’s status itself—also seen in the human adrenal (HPA, www.proteinatlas.org) 26 —and that the intensity of PARP-1 expression in the muscularis of the seminal vesicle varies according to the orientation of the muscle tissue. Altogether, our observations support a regulated expression of PARP-1, the complexities of which remain to be explored.
PARP-1 subcellular expression is primarily nuclear, consistent with observations in cultured cell lines and PARP-1 known functions in the DDR.8,37,48 Notable exceptions include the follicular and parafollicular cells of the thyroid, as well as most of the endothelial cells composing blood vessels, where cytoplasmic expression predominates (Fig. 7I and J).
In line with this cytoplasmic expression, most tissues also present specific cytoplasmic PARP-1 expression. This expression pattern is at times strongly stippled, reminiscent of Nissl bodies, which consist of rough endoplasmic reticulum covered with free ribosomes. 49 This stippled cytoplasmic pattern was also seen in the human salivary glands (HPA, www.proteinatlas.org). 26 Although comprehensive research of PARP-1 cytoplasmic expression has not been carried out in the human, several studies have addressed possible reasons for the presence of PARP-1 in the cytoplasm. For example, in the adult human central nervous system, it was proposed that neurons with intense cytoplasmic PARP immunoreactivity could represent cell groups exposed to a higher degree of metabolic stress. 50 Rat lung fetal development research has also demonstrated that at late gestational stages, as well as postnatally, PARP-1 staining was primarily cytoplasmic and restricted to a subset of cells, mainly bronchial epithelial and smooth muscle cells. 51 In oncology, cytoplasmic PARP-1 has been associated with an aggressive breast tumor form.52 –54 Overall, our results raise the possibility that PARP-1 may have preferential cytoplasmic expression in some tissues or cell types, an observation that deserves further investigation, as PARPi are currently being investigated for the treatment of several tissue-specific malignancies.20 –25,55
In contrast to our previous findings for PARP-3 protein expression, PARP-1 is strongly expressed in less differentiated cells compared with mature cells. 28 For instance, within the epidermis of the breast skin, PARP-1 expression is much stronger in the basal layers composed of immature cells compared with the apical layers composed of more mature cells. Similarly, round spermatids (immature) have strong PARP-1 expression compared with elongated spermatids (mature), which have none. This expression pattern is also paralleled by a similar distribution in corresponding human tissues and cells (HPA, www.proteinatlas.org). 26 The proliferative nature of cells expressing high PARP-1 levels is further supported by the stronger expression of proliferation markers such as MCM-2 and Ki67 in these cells.56 –59 For instance, while the immature cells of the basal layers of the human skin epidermis strongly express PARP-1, MCM-2, and Ki-67, mature cells of the apical layers do not. Similarly, in the human testis, mature elongated spermatids have no PARP-1 expression while proliferating round spermatids show strong expression of MCM-2 and Ki-67 (HPA, www.proteinatlas.org). 26 These observations raise the hypothesis that PARP-1 may be preferentially expressed in proliferating cells rather than in mature cells. Although this notion remains underexplored, it is tempting to speculate that this differential expression may reflect a significant role for PARP-1 during this process, supported by its reported functions in the surveillance of the genome, DNA replication, and transcription.60 –68 This notion is also in line with previous reports showing an inverse correlation between the level of cell maturation and PARP-1 expression levels, and its enzymatic activity during spermatogenesis.69 –71 Cell-based assays have also correlated maximal PARP activity and poly(ADP-ribose) levels with the replicative and division phases of the cell cycle.67,68,72 One exception to this pattern in the monkey would be the strong PARP-1 expression in lymphocyte nuclei of the thymus medulla (mature lymphocytes) compared with its very weak expression in the lymphocyte nuclei of the cortex (immature lymphocytes). 73 Nevertheless, the strong protein expression of PARP-1 in lymphoid tissues emphasizes the clinical potential of the inhibition of PARP. In line with our observations, recent findings have revealed PARP-1 regulates gene transcription of immune cells such as lymphocytes 74 to participate in the resolution of DNA double-strand breaks that occur during class switch recombination, 75 and to take part in the development of inflammatory responses.
In this study, except for the adrenal, most male and female tissues are similarly labeled. The overall stronger PARP-1 expression in the male adrenal relative to the female suggests that there may be sex differences in PARP-1 expression in this tissue. The adrenal gland is responsible for the secretion of adrenal androgens which are produced in the zona reticularis and zona fasciculata of the adrenal cortex. This production progressively increases from the beginning of puberty until late puberty. 76 It is therefore interesting to see that in tissues from monkeys that have reached sexual maturity (age 5–7), PARP-1 expression is stronger in male androgen-producing zones compared with female. 77 Although sexual dimorphism in PARP expression and activity remains underexplored, it has been reported that in peripheral blood monocytic cells from healthy individuals, men had nearly a 2-fold higher PARP activity compared with women and that this difference could be due to sex hormones. 78 Overall, the influence of androgens in the regulation of PARP-1 expression in both sexes is worthy of further investigation.
Based on the results of this study, we conclude that PARP-1 is expressed in all the cynomolgus monkey organs surveyed. However, its variable expression levels within most tissues suggest a complex regulation of PARP-1. Given the expanding use of PARPi in chemotherapeutic strategies, a better understanding of this regulated expression is warranted. An important aspect of this therapeutic strategy is the presence of PARP-1 itself to create toxicity in cancer cells, 79 highlighting the need to understand PARP-1 distribution and expression in tissues and cell types. The observation that the ovary is one of the highest PARP-1 expressing tissues from our IHC analysis strengthens the current clinical responsiveness of ovarian cancer to the FDA approved PARPi Olaparib (Lymparza). In addition, because PARPi also target PARP-2 and PARP-3,80,81 which are also involved in DDR,82–84 there is a necessity to understand the tissue distribution of these PARPs as well. Our group has previously reported the distribution of PARP-3 expression in the same monkey tissues. Results show that tissue and cellular distribution of PARP-3 is relatively distinct from that of PARP-1. For instance, high expression of PARP-1 in the testis, particularly in spermatogenic cells, markedly contrasts with the low expression of PARP-3 in the testis. Thus, this study, together with our previously reported PARP-3 expression, will help further characterize and highlight tissues that will most likely be affected by PARPi. Moving forward, additional PARP-2 protein expression studies would add tremendous value to our current protein expression database, by providing a clearer understanding of the potential impact of PARP inhibition in different tissues.
Supplemental Material
2019-00024R4_Production_Supplemental_Table_online_supp – Supplemental material for Assessment of PARP-1 Distribution in Tissues of Cynomolgus Monkeys
Supplemental material, 2019-00024R4_Production_Supplemental_Table_online_supp for Assessment of PARP-1 Distribution in Tissues of Cynomolgus Monkeys by Maria Tedim Ferreira, Louise Berger, Michèle Rouleau and Guy G. Poirier in Journal of Histochemistry & Cytochemistry
Footnotes
Acknowledgements
We would like to thank Johanne Ouellet (Histology Platform, Laval University Hospital Research Center) for her immunohistochemistry work.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this article as follows: planning (LB, MR, GGP), interpretation and the description of the immunohistochemistry results (LB, MTF), manuscript drafting (MTF, LB, MR), and all authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Canadian Institutes of Health Research to GGP (FRN-133430).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
