Abstract
From October to November 2001, the inhalational and cutaneous anthrax cases that occurred in the U.S. underscored the importance of recognizing the clinical and pathological features of infectious agents that can be used in acts of terrorism. Early confirmation of bioterrorist acts can only be performed by making organism-specific diagnosis of cases with clinical and pathologic syndromes that could be caused by possible bioterrorism weapons. Recognition and diagnosis of these cases is central to establish adequate responses. This review will examine the events that occurred during the anthrax bioterrorist attack with specific emphasis on the role of pathology and immunohistochemistry and will describe the histopathologic features of category A bioterrorism agents (anthrax, plague, tularemia, botulism, smallpox, and viral hemorrhagic fevers).
Keywords
F
A bioterrorism-related anthrax investigation started in early October 2001, when Bacillus anthracis was isolated from the cerebrospinal fluid of a patient who worked as a photo editor for a media company in Florida (Jernigan et al. 2001). Public health officials, together with federal and law enforcement authorities, launched a bioterrorism investigation even though the source or possible motive were unknown. The investigation included the search for other anthrax cases and used the following case definition: a confirmed case of anthrax was defined as a clinically compatible case of cutaneous, inhalational, or gastrointestinal illness that was laboratory confirmed by isolation of B. anthracis from an affected tissue or site or by other laboratory evidence of B. anthracis based on at least two supportive laboratory tests (ELISA serology, IHC, PCR) (CDC 2001a). Despite adequate antibiotic and supportive treatment, the index patient died, and autopsy results confirmed the cause of death as inhalational anthrax (Bush et al. 2001; Guarner et al. 2003).
List of category A, B, and C biological agentsa

IHC assays are available at CDC for the underlined agents.
The story became more complex when a cutaneous lesion compatible with anthrax was found on the neck of the secretary who handled mail for a leading news anchor in New York city. A dermatologist had prescribed antibiotics, but the lesion continued to enlarge. The patient went to the New York State Health Department when news of the anthrax case from Florida was linked to mail containing “white powder.” The patient had handled several threat letters at work. Skin biopsy samples were obtained for culture, PCR, and histopathologic examination. Cultures resulted in no growth because the patient had received antibiotics for 9 days. PCR did not yield anthrax nucleic acids; however, a formalin-fixed skin biopsy showed B. anthracis antigens by IHC (Figure 1) (Shieh et al. 2003). Eventually, this case was confirmed as cutaneous anthrax by serologic testing (CDC 2001d; Jernigan et al. 2001, 2002).
During this attack, there were 11 patients with inhalational anthrax and 11 with cutaneous anthrax (Jernigan et al. 2001, 2002). Five of the inhalational anthrax patients died, and B. anthracis was cultured from samples from all five patients before autopsies were performed. Autopsy findings were important for identifying the route of transmission and for obtaining medico-legal evidence of homicide. Of the six inhalational anthrax patients who survived, three had surgical pathology specimens (pleural fluid blocks and/or transbronchial biopsies) that were confirmed positive for anthrax by IHC (Figure 2) and at least one other test. The other three surviving inhalational anthrax patients had blood culture samples that grew B. anthracis. Of the 11 cutaneous anthrax cases, 3 were considered suspect because only one test showed evidence of B. anthracis (two by serology and one by IHC) but were never confirmed by a second test. Of the eight remaining cutaneous anthrax cases, three were confirmed by cultures of B. anthracis from skin or blood, whereas in five cases IHC played a pivotal role in the diagnosis.
The anthrax bioterrorism attack of 2001 exemplified the prominent role of pathology in the diagnosis of cases and provided important information that allowed public health officials and law enforcement agents to define potential sources of infection. As pathologists, we will be asked to evaluate clinical, surgical, and autopsy specimens for the presence of bioterrorism agents. Thus, pathologists need to know which are the specimens and tests that are most adequate for determining a definitive, organism-specific diagnosis.
Cultures are still considered the reference standard for diagnosis of viral and bacterial diseases, and it is crucial to place tissue samples in transport media that will allow bacterial and viral isolation. However, as could be observed in the anthrax bioterrrorism attack, growth of the microorganism may not occur for multiple reasons, the most frequent being the use of antibiotics prior to obtaining cultures (Guarner et al. 2003; Shieh et al. 2003). Hence, other specimens should be collected and assays performed; for example, serum is indispensable for serological and biological assays, tissue samples need to be frozen for PCR, and samples should be placed in electron microscopy fixative (glutaraldehyde) and formalin for morphologic examination. Performing a complete autopsy with multiple organ sampling will help determine the distribution of the agent and the portal of entry (Nolte et al. 2004). Microscopic examination of formalin-fixed, paraffinembedded tissues with hematoxylin and eosin (H&E) stains is indispensable because it will characterize the patterns of tissue damage defining a syndrome, which will establish a list of possible microorganisms in the differential diagnosis (Table 2). Special stains, such as tissue Gram's and silver impregnation stains (Steiner's or Warthin-Starry), can be helpful in identifying bacterial agents. In addition, specific IHC and direct fluorescent assays (DFA) for category A bioterrorism agents are necessary. Due to the high national profile of cases possibly related to bioterrorism, it is important to remember that samples from these patients will not only need to be tested at the site where they occur, but also by a variety of state and federal agencies, including law enforcement and state health departments. Consequently, organizations such as the CDC have taken the lead in establishing diagnostic procedures (e.g., IHC and PCR) that can be performed on formalin-fixed tissues.
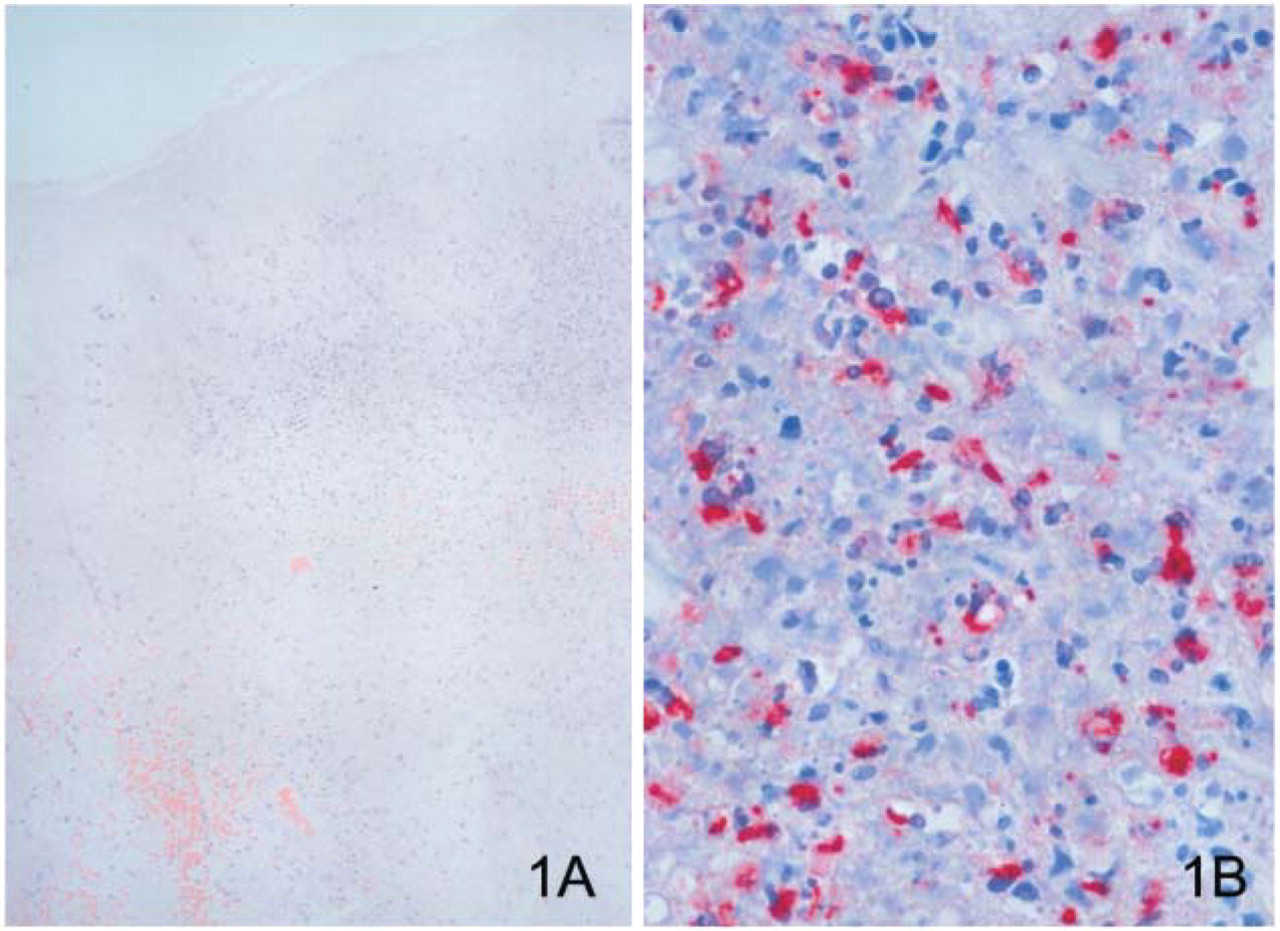
Cutaneous anthrax lesion shows necrosis of dermis and epidermis, edema, mild inflammatory infiltrate (
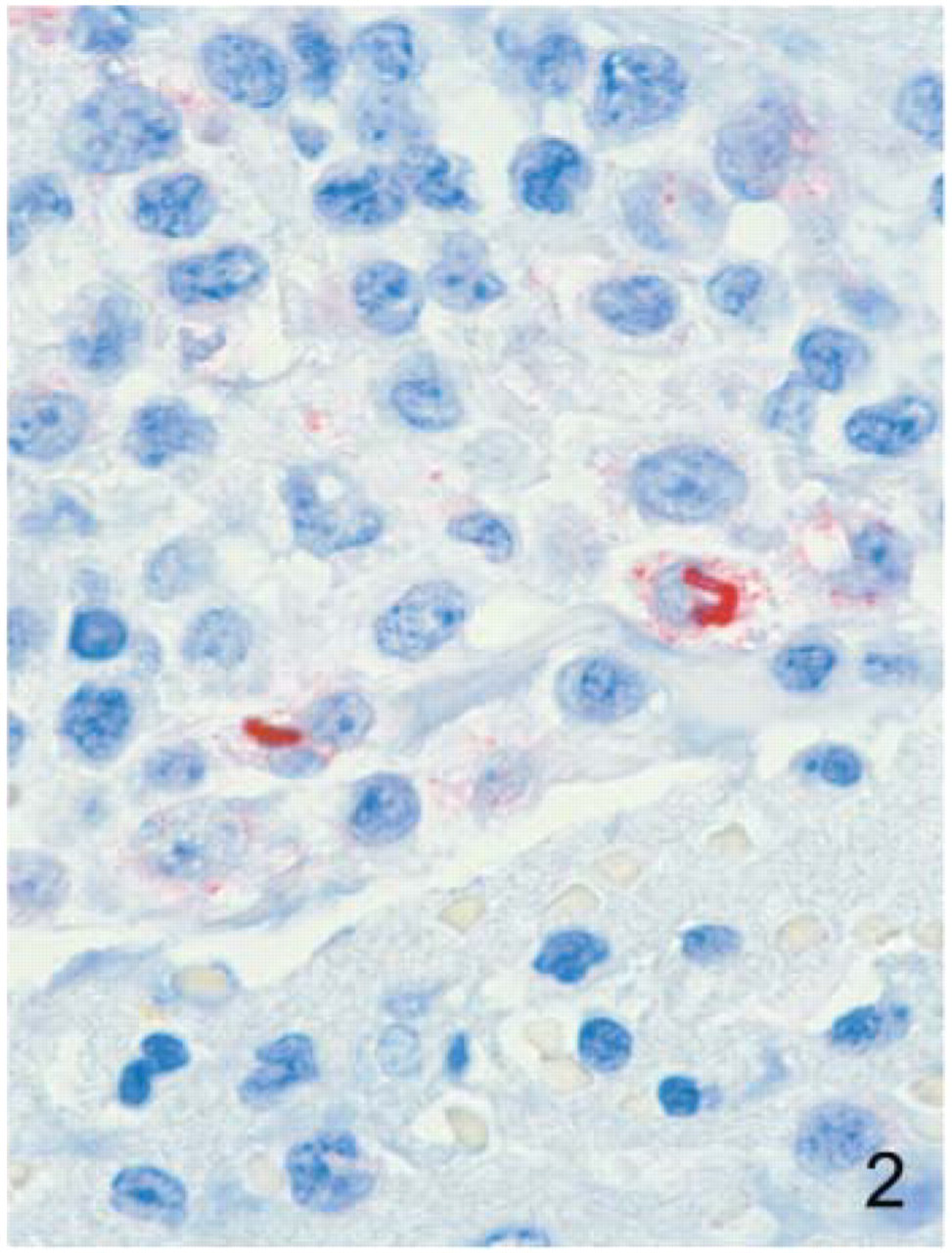
Cell block prepared from pleural cell fluid of a patient with inhalational anthrax showing bacilli and bacillary fragments. Results from immunohistochemical assay using a monoclonal antibody against B. anthracis cell wall (original magnification ×250).
(
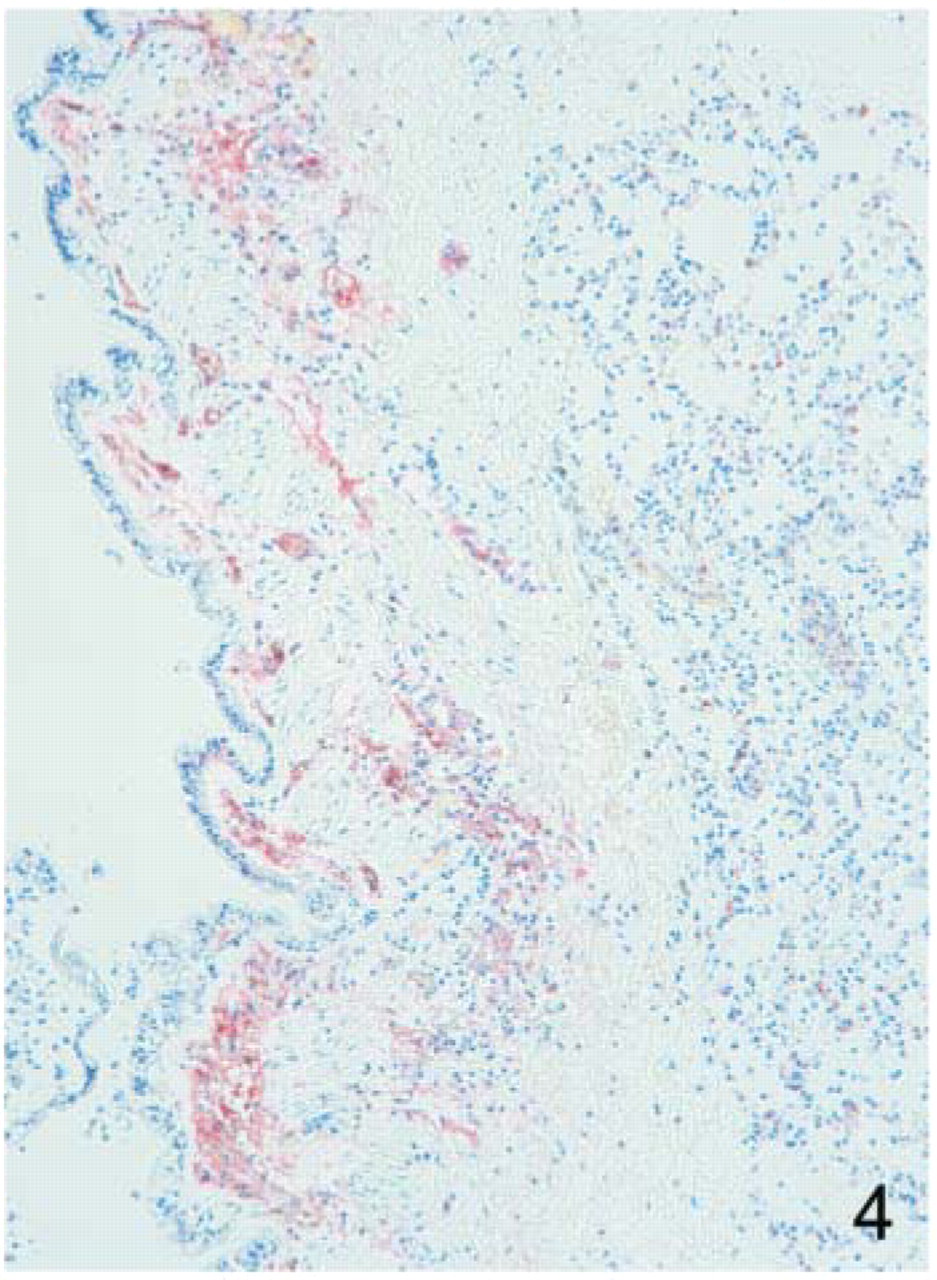
Lung showing secondary pneumonic plague. Note the presence of Y. pestis antigens in the interstitial tissue around bronchi but not in the alveolar spaces or bronchus. Results from immunohistochemical assay using a monoclonal antibody against Y. pestis (original magnification ×25).
Pathology-based syndromic surveillance
Development of IHC techniques has included development and testing of multiple polyclonal and monoclonal antibodies directed against the bioterrorism agents and validation of the assays in naturally occurring human or animal cases, as well as in animal models. IHC assays have important advantages over other assays because they combine localization of the microorganisms in the context of the host response, allowing a better interpretation of results. For example, in anthrax patients who had been treated with antibiotics, B. anthracis did not grow and intact bacilli were difficult to find with Gram and silver impregnation stains. However, presence of granular antigen IHC staining inside phagocytic cells in the areas of necrosis and edema suggested the diagnosis of anthrax in several cases. This diagnosis was later confirmed by other methods.
We will present a brief description of the pathologic features of category A agents as follows.
Anthrax
Humans can acquire B. anthracis when spores are introduced via the respiratory or gastrointestinal tracts or through skin abrasions (Perl and Dooley 1976; Jaax and Fritz 1997; Dixon et al. 1999). Once the spores are in an appropriate environment, they transform into bacilli (vegetative stage), produce toxins (edema and lethal), and divide (Dixon et al. 1999). The edema toxin induces cyclic AMP, which results in damage to water homeostasis. The lethal toxin cleaves MAP kinases, releasing oxygen radicals and proinflammatory cytokines that result in cell death.
Certain key clinicopathologic features serve to classify anthrax infection into cutaneous, gastrointestinal, or inhalational forms (Perl and Dooley 1976; Jaax and Fritz 1997; Dixon et al. 1999). Cutaneous anthrax is characterized by an eschar that corresponds to the area where the bacteria entered the skin. Microscopically, the epidermis shows necrosis and crust formation, whereas the dermis demonstrates necrosis, edema, hemorrhage, perivascular inflammation, and vasculitis. If untreated, the lymph nodes that drain the skin site will eventually become enlarged, necrotic, and hemorrhagic. Gastrointestinal anthrax should be suspected when hemorrhagic ulcers in the terminal ileum and cecum are accompanied by mesenteric hemorrhagic lymphadenitis and peritonitis. Inhalational anthrax is characterized by hemorrhagic mediastinal lymphadenitis accompanied by pleural effusions, but a pneumonic process will not be observed (Albrink et al. 1960; Abramova et al. 1993; Walker et al. 1994; Grinberg et al. 2001; Guarner et al. 2003). On microscopic examination, lymph nodes show abundant edema, hemorrhage, and necrosis with little inflammatory infiltrate. As any of the three anthrax forms progresses, the bacteria spread to abdominal organs, producing petechial hemorrhages and to the central nervous system, producing hemorrhagic meningitis (cardinal's cap).
The specimens that will harbor the highest load of B. anthracis will depend on the form of anthrax. For example, diagnosis of cutaneous anthrax requires skin biopsies from the center and periphery of the eschar, whereas for inhalational anthrax, pleural effusion cell blocks, pleural biopsies, and mediastinal lymph nodes have the highest amounts of bacilli and antigens. If the patient has not received treatment, bacilli can be observed in tissues with H&E, Gram's, and silver impregnation stains; however, when antibiotic treatment has been instituted, silver stains will highlight the bacilli and H&E and Gram's stains will not. On Gram's stains, the bacteria appear as large gram-positive bacilli. IHC assays for B. anthracis can demonstrate bacilli, bacillary fragments, and granular bacterial fragments in formalin-fixed tissues even after 10 days of antibiotic treatment (Guarner et al. 2003; Shieh et al. 2003).
Plague
Yersinia pestis is a bipolar, small, gram-negative bacillus that can be acquired through contact with infected mammals or through flea bites (Smith 1976; Smith and Reisner 1997). Once plague enters the human population through the cutaneous route, human-to-human transmission can occur by inhaling infected aerosolized droplets disbursed by coughing.
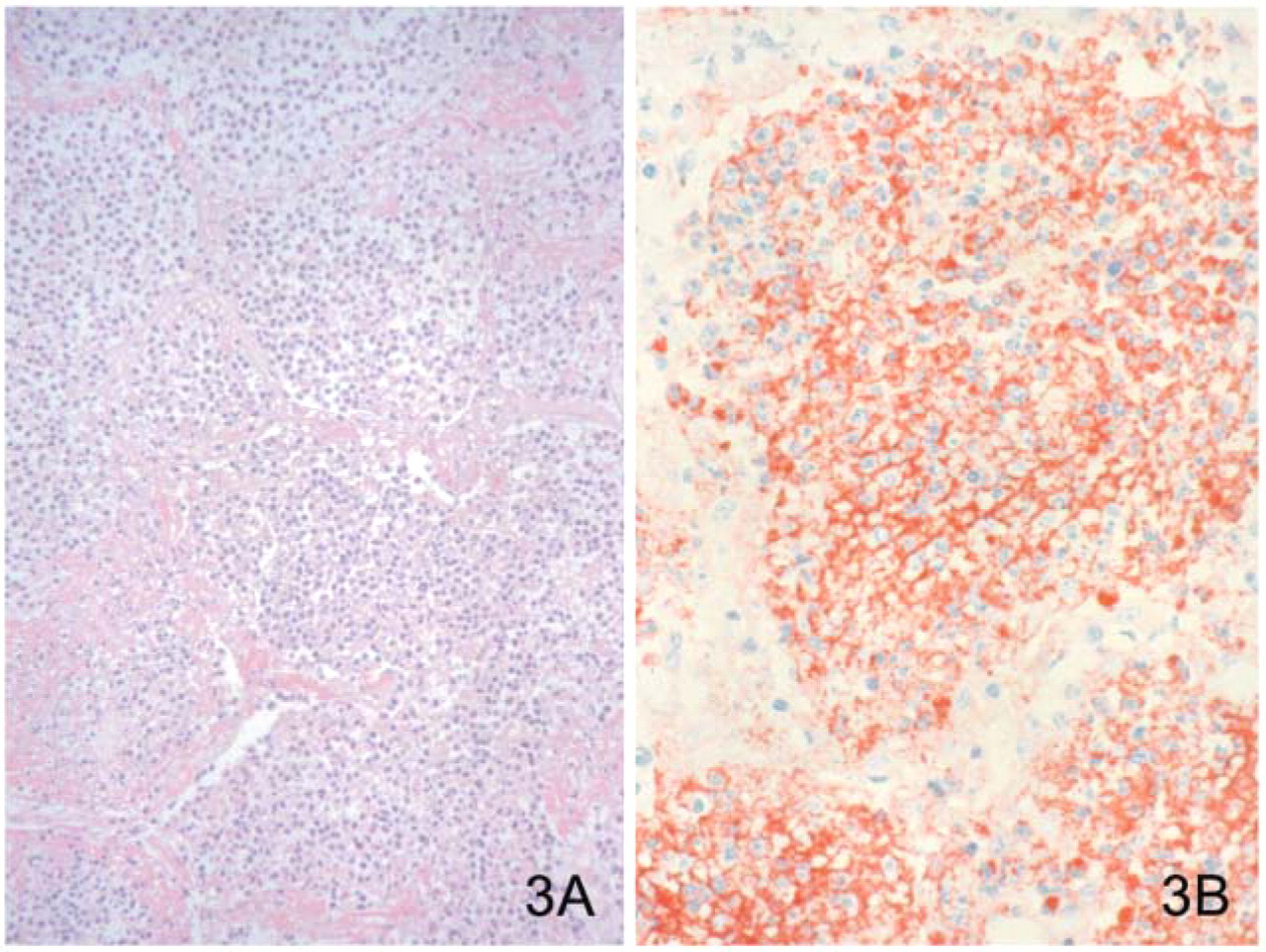
Similar to anthrax, plague is classified according to the portal of entry of Y. pestis (Smith 1976; Smith and Reisner 1997). Bubonic plague refers to an acute lymphadenitis (bubo) that occurs after the bacteria penetrate the skin. The skin lesion usually is inconspicuous or may present as a small vesicle or pustule that may or may not be evident at the time the bubo appears. Microscopic examination of the draining lymph node shows edema, hemorrhage, necrosis, and a groundglass amphophilic material that represents masses of bacilli. Primary pneumonic plague refers to inhalation of bacteria that produce intra-alveolar edema accompanied by various amounts of polymorphonuclear inflammatory infiltrate and intra-alveolar ground-glass amphophilic material (corresponding to bacteria and necrotic debris) (Figures 3A and 3B) (Finegold et al. 1968; Finegold 1969; Jones et al. 1979; Guarner et al. 2002). Primary septicemic plague occurs when Y. pestis enters through the oropharyngeal route; thus, the bubos correspond to cervical lymph nodes draining the bacilli from the throat. As the disease progresses, bacteria spread systemically producing shock, disseminated intravascular coagulation, and secondary pneumonic plague with bacteria primarily in the interstitium but not in the alveolar air spaces (Figure 4) (Smith 1976; Guarner et al. 2002).
Plague can be diagnosed by demonstrating the presence of Y. pestis in enlarged, soft, hemorrhagic lymph nodes by using culture, IHC (Figure 3B and Figure 4), DFA, or PCR (Guarner et al. 2002). The lungs should be sampled to determine whether there is a primary or a secondary pneumonic plague. The distinction between primary and secondary pneumonic plague has important implications because it will determine the mode of transmission and potential sources of the bacteria. Y. pestis can be visualized in formalin-fixed tissues using H&E, Gram's, silver impregnation, and Giemsa stains; however, specific identification of the bacilli in formalin-fixed tissues can be performed using only specific IHC, DFA, or PCR assays.
Tularemia
Tularemia, a zoonotic infection also known as rabbit or deerfly fever, is caused by the small, gram-negative, pleomorphic coccobacillus, Francisella tularensis (Schmid et al. 1983; Evans et al. 1985; Geyer et al. 1997). This infection is endemic in the south central region of the U.S. and peaks during the summer months. Tularemia is considered a possible bioterrorism agent because of its potential to cause life-threatening pneumonia when the bacteria are aerosolized.
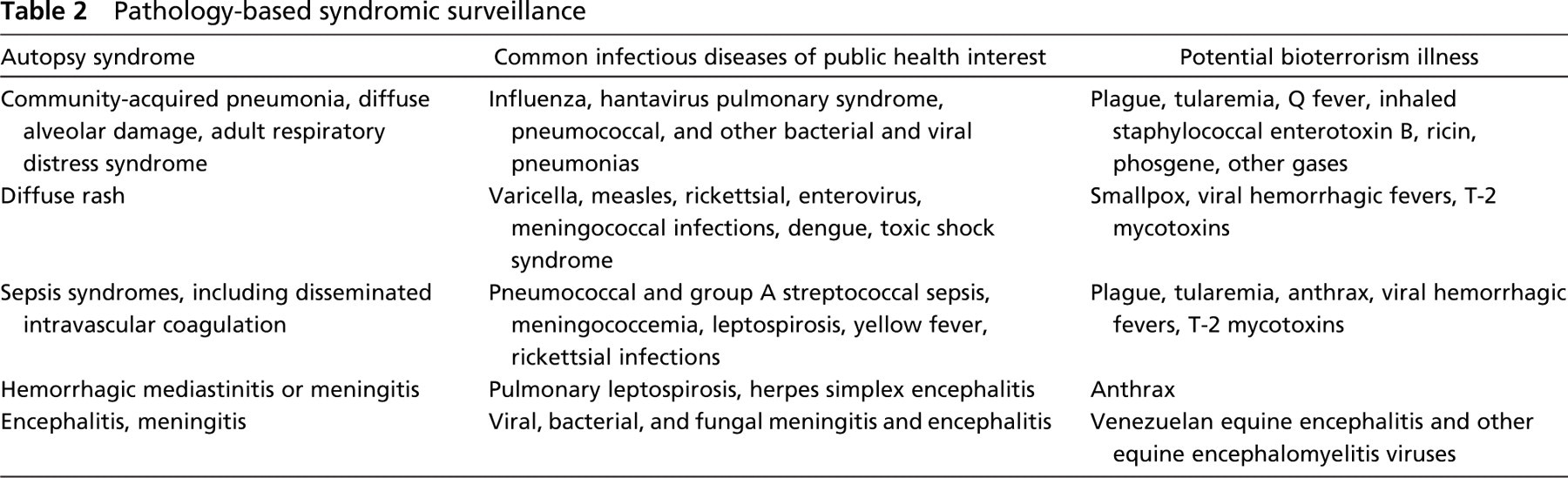
F. tularensis can have several presentation forms, depending on the portal of entry, including ulceroglandular, oculoglandular, glandular, pharyngeal, typhoidal, and pneumonic tularemia (Gallivan et al. 1980; Geyer et al. 1997; Guarner et al. 1999). In all forms, the primary draining lymph nodes show a lymphadenitis characterized by geographic areas of necrosis surrounded by a palisading, neutrophilic, and granulomatous inflammatory infiltrate. In the ulceroglandular form, a skin ulcer/eschar is noted, whereas skin lesions are absent in the glandular form. In the oculoglandular form, the eye shows conjunctivitis with ulcers and soft tissue edema. The pharyngeal form shows pharyngitis or tonsillitis with ulceration. The lungs in pneumonic tularemia show abundant fibrinous necrosis accompanied by various amounts of mixed inflammatory infiltrate (Figures 5A and 5B). Typhoidal tularemia refers to systemic involvement, with focal areas of necrosis in the major organs and disseminated intravascular coagulation, but does not involve a group of primary draining lymph nodes.
Diagnostic samples for tularemia include the enlarged, necrotic lymph nodes and swabs or biopsy samples from the potential portals of entry (conjunctiva, throat). These samples should be cultured and studied histopathologically. Microorganisms are difficult to demonstrate with special stains; however, IHC (Figure 5B) and DFA have been used successfully in formalin-fixed tissues to demonstrate the bacteria (Guarner et al. 1999).
Botulism
Botulism is an acute systemic toxemia, not an infection (Schwartz and Geyer 1997). It is acquired when the potent neurotoxin produced by Clostridium botulinum is ingested or enters through a wound. The most important diagnostic feature of botulism is the clinical history because the histopathologic changes are nonspecific (central nervous system hyperemia and microthrombosis of small vessels). When botulism is suspected because of a symmetrical, descending pattern of weakness and paralysis of cranial nerves, limbs, and trunk, it is essential that a serum sample be obtained for botulinum toxin mouse bioassay. Samples from the suspect entry sites (wound, gastrointestinal) should be obtained for anaerobic cultures, and an IHC can demonstrate the bacilli. Identification of clostridia at an entry site is a sign that the toxin was produced by microorganisms in the host and probably acquired naturally rather than being acquired through mass-produced and intentionally released toxin.
(
(
(
Smallpox
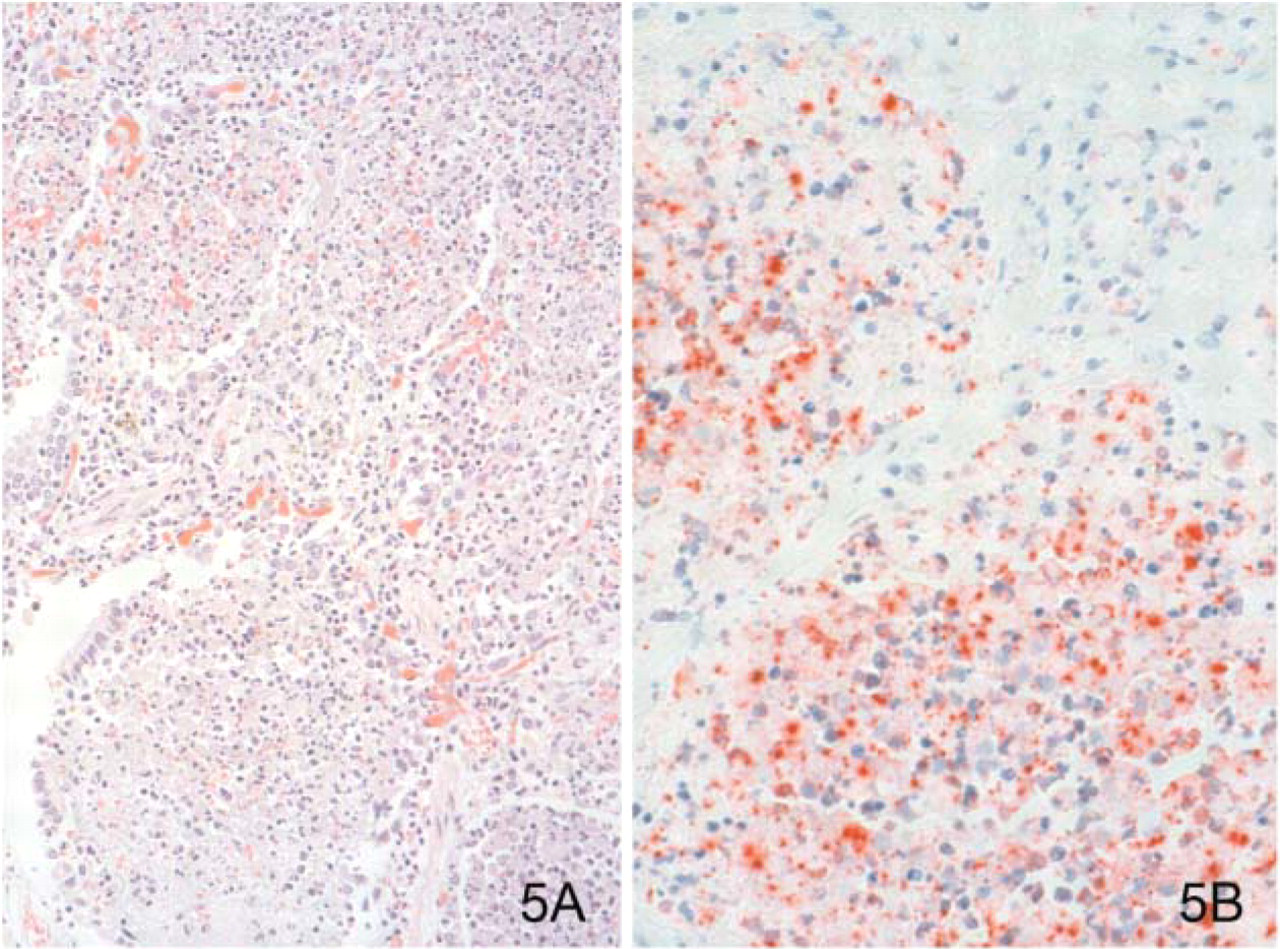
Smallpox is an acute, highly contagious exanthema caused by a member of the Poxviridae family (Strano 1976; Cockerell 1997). Variola major refers to the form with a higher mortality rate, whereas variola minor or alastrim is a milder variety. Before smallpox virus was eradicated, most human infections were acquired through the respiratory tract. The disease starts with fever, headache, vomiting, and low back pain. As the fever subsides, skin lesions develop at approximately the same time and rate, starting in the palms and soles, and spreading centrally. They progress through stages: first macules and papules, then vesicles and umbilicated pustules, followed by scabs and crusts, and ending with pitted scars. When viewed under a microscope, the skin shows multiloculated, intraepidermal vesicles, ballooning degeneration of epithelial cells, intracytoplasmic, paranuclear, eosinophilic viral inclusions (Guarnieri's bodies) (Figure 6A), and, occasionally, intranuclear viral changes (Councilman et al. 1904; Bras 1952; Strano 1976; Cockerell 1997). Secondary infections such as bronchitis, pneumonia, and encephalitis may complicate the clinical picture.
Cutaneous lesions are the most important sample for smallpox. Samples should include fluid from vesicles for electron microscopy study and skin biopsy samples fixed in formalin for histopathology, IHC, and DFA (Murray 1963; Cruickshank et al. 1966). IHC studies have demonstrated the virus in a variety of cells (epidermis, hepatocytes [Figure 6B], and others) and in adjacent fibroconnective tissues.
Viral Hemorrhagic Fevers
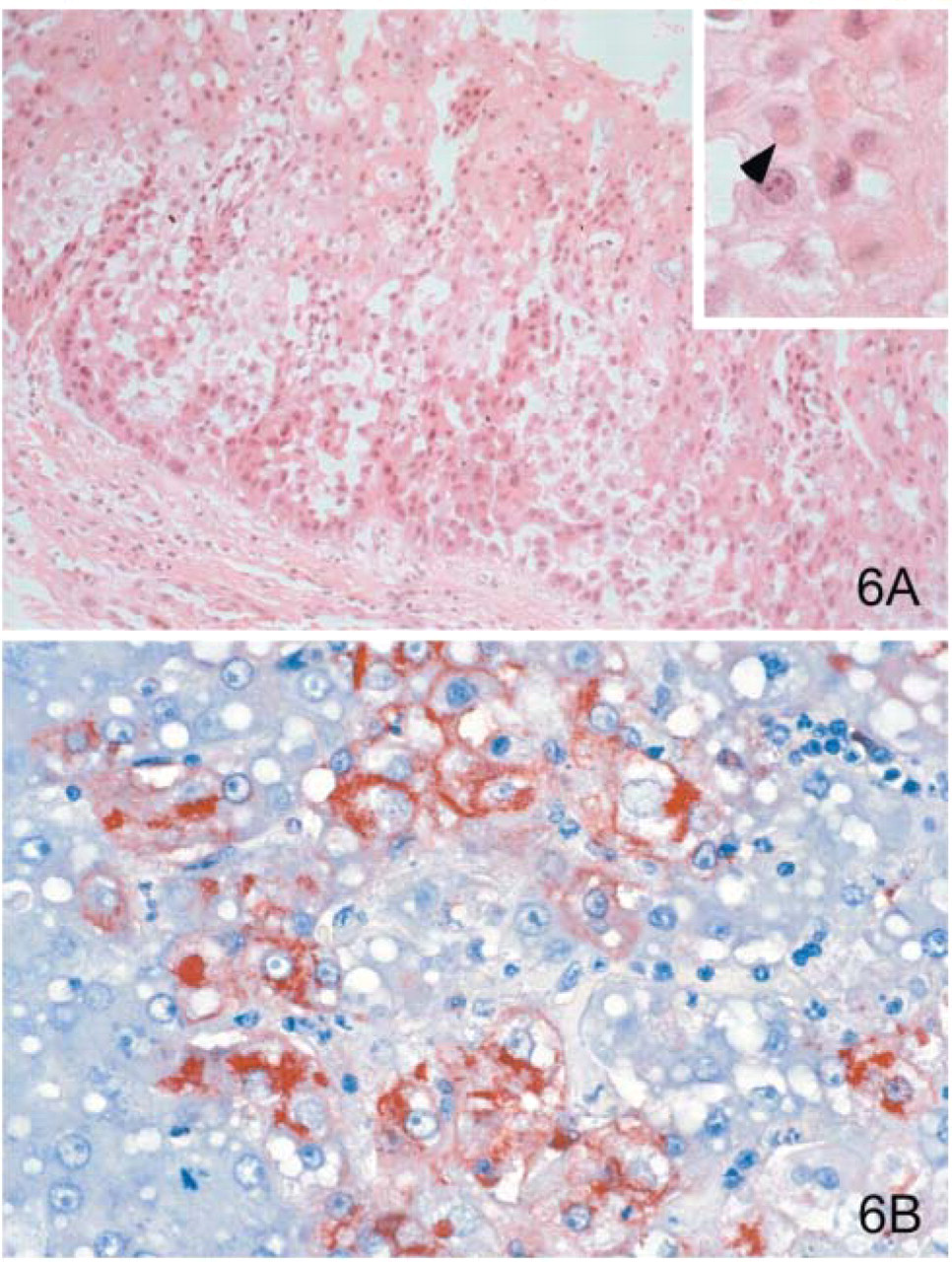
Viruses that can cause hemorrhagic fevers are considered category A bioterrorism agents and belong to two families: Filoviridae (Ebola and Marburg viruses) and Arenaviridae (Junin, Machupo, Guanarito, and Lassa viruses) (Child 1976; Zaki and Goldsmith 1998; Zaki and Kilmarx 1997; Zaki and Peters 1997). The term viral hemorrhagic fever is reserved for febrile illnesses associated with abnormal vascular regulation and vascular damage. Common pathologic findings at autopsy include petechial hemorrhages and ecchymoses of skin, mucous membranes, and internal organs. Necrosis of liver and lymphoid tissues and diffuse alveolar damage are frequent pathologic features of most viral hemorrhagic fevers (Gubler and Zaki 1998). Viral inclusions can be seen in hepatocytes with Ebola (Figure 7A) or Marburg infections by using light and electron microscopy (Zaki and Goldsmith 1998).
Although the clinical end stage of these viral infections consists of systemic hemorrhages, some have distinct distribution to specific cells; thus, histopathologic features may be different. kin samples can be tested using PCR, IHC, and electron microscopy. In addition, serum can be inoculated into experimental animals or culture cells for viral isolation or tested by using specific PCR assays. Once a specific etiologic agent has been isolated/diagnosed from the index case, targeted sampling of additional cases can decrease the exposure to hazardous agents during autopsy and still yield diagnostic material. For example, during outbreaks of Ebola hemorrhagic fever in Africa, the use of IHC on skin punch biopsy samples (Figure 7B) has provided a diagnosis for large numbers of fatal cases (Zaki et al. 1999).
Differential diagnosis for category A agents includes a variety of diseases from the clinical and pathological point of view. This brings us to the definition of terror: to create fear as a means of coercion. This was exemplified during the 2001 attack when any skin lesion or recurrent, hemorrhagic pleural effusion was considered a suspect case of anthrax, and the public demanded testing for the bacillus. This created an environment of fear disproportionate to the number of cases that occurred. During that time, CDC received over 100 skin biopsy samples to be tested for anthrax; these samples included an array of diseases and lesions such as Rickettsiapox and herpes simplex and spider bites. Even though cell blocks prepared from hemorrhagic pleural effusions are an important part of the differential diagnosis of inhalational anthrax, it must be remembered that the most frequent cause of recurrent, hemorrhagic pleural effusions is invasion of the pleural space by cancer. Similarly, the most frequent cause of an enlarged mediastinum is a neoplastic process. Thus, clinical, epidemiological, and pathologic correlation is indispensable for adequate diagnosis that will allow practice of medicine without fear.
Surveillance for bioterrorism category A agents is ongoing. For example, skin biopsy samples have been available through a prospective surveillance program for febrile vesicular skin rashes geared at detecting possible smallpox cases. During the time the program has been in place, the most frequent differential diagnosis has been varicella-zoster infection. This program was instrumental in studying a cluster of monkeypox infections that occurred during the summer of 2003 in the U.S. and proved the beneficial public health effect of having in place systems that can detect bioterrorism agents (CDC 2003; Sejvar et al. 2004)
The differential diagnoses of patients who present with fever, rash, and bleeding from mucous membranes will include category A agents such as Ebola, Marburg, and other viruses, some in category C, such as yellow fever and hantaviruses, and bacteria such as leptospirosis, which is not considered a possible bioterrorism agent. Hence, clinical, epidemiologic, and pathological data are required to guide diagnostic tests and treatment of patients with fever and skin rash.
The differential diagnosis of pneumonias due to tularemia and plague includes a large variety of bacteria that can cause intra-alveolar inflammatory infiltrate (i.e., Streptococcus pneumoniae, Legionella pneumophila). For diagnosis of pneumonias related to bioterrorism agents it is essential that an organism-specific diagnosis be made, and that requires the use of culture and/or techniques mentioned above. To confirm bioterrorismrelated pneumonia cases, a stride to make organismspecific diagnosis should be embraced by clinicians and pathologists. Currently, a sizeable number of patients who die of pneumonias will not have an organismspecific diagnosis (Marston et al. 1997). The most frequent cause of pneumonias will continue to be the usual community- and hospital-acquired infectious microorganisms. Specific testing for bioterrorism agents in patients with this syndrome will require a high level of clinical, epidemiological, and pathological suspicion.
From the clinicopathologic standpoint, it is practical to categorize cases by syndrome. Table 2 presents the different autopsy syndromes that can be caused by bioterrorism agents and a differential diagnosis of infectious agents of public health interest. The syndromic approach allows directed testing for a variety of agents, including category A, B, and C bioterrorism agents. Finally, it should be remembered that, although we need to be prepared for another bioterrorist attack, this will hopefully never occur again or will be a once in a lifetime occurrence in the pathology practice and that the vast majority of pneumonia cases, vesicular skin rashes, and mucosal hemorrhages occur because of naturally acquired infectious processes.
