Abstract
Pseudoachondroplasia (PSACH) is an autosomal dominant disease characterized by dwarfism, morphological irregularities of long bones and hips, and early-onset osteoarthritis. This disease has been attributed to mutations in a structural protein of the cartilage extracellular matrix (ECM), cartilage oligomeric matrix protein (COMP), which result in its selective retention in the chondrocyte rough endoplasmic reticulum (ER). Accumulation of excessive amounts of mutated COMP might reflect a defect in protein trafficking by PSACH chondrocytes. Here we identify the matricellular protein SPARC as a component of this trafficking deficit. SPARC was localized to the hypertrophic chondrocytes in the normal human tibial growth plate and in cultured control cartilage nodules. In contrast, concentrated intracellular depots of SPARC were identified in nodules cultured from three PSACH patients with mutations in COMP. The accumulated SPARC was coincident with COMP and with protein disulfide isomerase, a resident chaperone of the rough ER, whereas SPARC and COMP were not coincident in the ECM of control or PSACH nodules. SPARC-null mice develop severe osteopenia and degenerative intervertebral disc disease, and exhibit attenuation of collagenous ECM. The retention of SPARC in the ER of chondrocytes producing mutant COMP indicates a new intracellular function for SPARC in the trafficking/secretion of cartilage ECM.
Keywords
P
Mutations in cartilage oligomeric matrix protein (COMP, also known as thrombospondin 5), a large extracellular matrix (ECM) protein (Mr 524,000), cause PSACH and the characteristic chondrocyte cellular pathology (Cooper et al. 1973; Hecht et al. 1995; Hecht et al. 2004). PSACH chondrocytes have a distended rough endoplasmic reticulum (ER) complex that has a lamellar appearance and is filled with COMP, type IX collagen, and matrilin-3, as well as other ECM proteins (Cooper et al. 1973; Hecht et al. 1995; Hecht et al. 2004). The abnormal retention of otherwise secreted structural proteins of the ECM is reminiscent of other connective tissue diseases (e.g., Ehlers Danlos Syndrome IV, in which collagen accumulates in a dilated rough ER) (Byers et al. 1981). In the case of PSACH, the presence of excessive intracellular mutated COMP has recently been characterized as a chondrocyte-specific defect in protein trafficking (Chen et al. 2004). Of the numerous mutations in COMP identified from PSACH patients, most (>95%) occur in the Ca+2-binding type 3 repeats adjacent to the C-terminal, collagen-binding globular domain, itself a site of mutations that produce severe phenotypes (Chen et al. 2000; Hou et al. 2000; Thur et al. 2001; Unger and Hecht 2001; Briggs and Chapman 2002). Delayed secretion or retention of COMP in the ER likely results from aberrant protein folding, abnormal posttranslational modification, and defective chain association (oligomerization). PSACH chondrocytes, however, do not exhibit defects in the trafficking or secretion of aggrecan and collagen type II, both major structural components of the territorial cartilaginous ECM, whereas collagen type IX, a nonfibrillar collagen associated with collagen type II fibrils, and the ECM protein matrilin-3, are retained with COMP in cellular compartments identified with several chaperone proteins including protein disulfide isomerase (PDI) (Hecht et al. 2001; Vranka et al. 2001; Chen et al. 2004; Hecht et al. 2005). The significance of this selective retention of ECM proteins in PSACH chondrocytes is poorly understood.
An understudied component of cartilage is the matricellular protein SPARC (secreted protein, acidic and rich in cysteine; also known as osteonectin and BM-40), an Mr 34,000 glycoprotein produced in many tissues during fetal development. In the adult, SPARC exhibits a more restricted pattern confined largely to remodeling organs with high cellular turnover, such as gut and bone, and to tissues responding to injury (Brekken and Sage 2001). Chondrocytes of both developing and adult bone, however, produce significant amounts of SPARC (Sage et al. 1989; Mundlos et al. 1992; Nakase et al. 1994; Porter et al. 1995). Mundlos et al. (1992) initially identified SPARC (as osteonectin) in human fetal bone and noted that its production was characteristic of cells exhibiting high rates of proliferation or ECM synthesis. Whereas SPARC mRNA was abundant in osteoblasts and chondrocytes of the upper hypertrophic and proliferative zones, SPARC protein was detected extracellularly only in bone and in the zone of mineralized cartilage. A similar distribution of SPARC mRNA was described in developing bone and cartilage of the mouse (e.g., in immature and hypertrophic chondrocytes) (Nakase et al. 1994). Other investigators have observed a substantial increase in the synthesis of SPARC by chondrocytes in human arthritic cartilage that was attributed in part to stimulation by transforming growth factor-α and interleukin-1α (Nakamura et al. 1996). Recent studies have established a cellular, but not extracellular, location of SPARC in the human intervertebral disc, as well as a marked diminution of SPARC in disc cells from older patients with disc degeneration (Gruber et al. 2004). These data are consistent with the phenotype of SPARC-null mice, which includes a significant degeneration of the intervertebral discs in aging animals, due in part to aberrant production and decreased assembly of collagen type I (Gruber et al. 2005). In summary, SPARC is a major noncollagenous product of differentiating chondrocytes and osteoblasts that has been associated with several types of skeletal diseases. Its role in the PSACH chondrocyte pathology, however, had not been investigated.
Several different mutations in COMP responsible for the PSACH phenotype have recently been shown to impair the secretion of COMP itself, as well as collagen type IX and matrilin 3, by cultured chondrocyte nodules (Hecht et al. 2001; Chen et al. 2004; Hecht et al. 2004). Hecht et al. (2005) have postulated that the retention of COMP leads eventually to chondrocyte death and thereby a reduction in cell number or ECM that predisposes PSACH patients to the deceleration of linear bone growth at 4–6 years of age. This hypothesis is consistent with the absence of overt skeletal abnormalities reported in COMP-null mice (Svensson et al. 2002) and emphasizes the role of protein folding, complex formation, and ECM assembly that are clearly affected by the mutations in COMP described in PSACH patients. We now show that SPARC is a component of the trafficking defect previously identified in chondrocytes from several patients with PSACH. These experiments support previous reports indicating an intracellular function of SPARC (Gooden et al. 1999; Yan et al. 2005a) and identify a potential role of SPARC as a chaperone for one or more proteins of the cartilage ECM.
Materials and Methods
Sample Preparation
Preparation of control costochondral chondrocytes and chondrocytes from PSACH patients with the mutations D469del, G427E, and D511Y, as well as culture of cartilage nodules, were as previously described (Hecht et al. 1998; Hecht et al. 2005). Briefly, the chondrocytes were expanded in monolayer and were subsequently cultured on polyHEMA matrix (Corning, Inc; Corning, NY) in Optimem (Invitrogen; Carlsbad, CA) containing 3% FBS, 25 μg/ml ascorbic acid, and 50 ng/ml bone morphogenetic protein 3 (BMP4) (Sigma Chemicals; St Louis, MO) for up to 8 weeks. The nodules were fixed in 4% paraformaldehyde, embedded in paraffin, and sectioned for light microscopy.
Immunostaining
Paraffin-embedded sections were deparaffinized and were incubated with pepsin (Sigma Chemicals) (1 mg/ml in 0.1 M HCl) for 20 min at room temperature (RT). All sections were washed in PBS/Tween20 between steps. Nonspecific binding was blocked with 10% normal donkey serum for 30 min at RT.
Horseradish Peroxidase—Single Label
Goat anti-human SPARC IgG (R & D Systems; Minneapolis, MN), was used at a dilution of 1:200 overnight at 4C. Signal for SPARC was amplified with biotinylated donkey anti-goat IgG (Chemicon; Temecula, CA) at a dilution of 1:500 for 30 min at RT and was detected with streptavidin-horseradish peroxidase (Chemicon) at 1:500 for 30 min at RT. Bound anti-SPARC antibody was visualized after reaction with diaminobenzidine Chromgen (DAKO; Carpinteria, CA) for 5 min at RT.
Fluorescence—Double Label
After incubation with goat anti-SPARC IgG overnight, the sections were washed in PBS/Tween20 and were incubated in either anti-human COMP (Kamiya Biomedical; Seattle, WA) at a dilution of 1:400 for 60 min at RT or rabbit anti-bovine PDI IgG (StressGen; San Diego, CA) at a dilution of 1:2000 for 60 min at RT. After washes, the signal for SPARC was amplified with biotinylated donkey anti-goat IgG as described previously. Sections were subsequently incubated in a cocktail consisting of streptavidin–Alexa Fluor 594 (Molecular Probes; Eugene, OR) and donkey anti-rabbit IgG conjugated to FITC (Chemicon) at a dilution of 1:500 for 60 min at RT. The slides were washed three times for 10 min each in PBS and were cover-slipped with Prolong Gold containing 4′,6-diamidino-2-phenylindole (Molecular Probes).
All images were collected with an Olympus BX51 microscope (Olympus America, Inc.; Melville, NY) equipped with a SPOT RT SLIDER digital camera and were edited by the use of Corel Draw (Corel Corp.; Ottawa, Canada).
Results
SPARC is Present in Normal Human Growth Plate
As shown in Figures 1A and 1B, human tibial growth plate was stained by anti-SPARC IgG within the hypertrophic zone, specifically around and in some cases within the hypertrophic chondrocytes. This staining pattern was recapitulated in cartilage nodules cultured from normal human donors (with differences in relative staining intensity) (Figure 1). In contrast, Figures 1D–F present an altered distribution of SPARC in cartilage nodules cultured from three PSACH patients, each of whom has been identified with different mutations in COMP. In these PSACH chondrocytes, SPARC was concentrated intracellularly, with apparently minimal levels in the cartilage ECM.
Cartilage nodules derived from normal (control) and PSACH patients were examined for potential colocalization of SPARC with a marker of the rough ER, PDI, a resident chaperone of this cellular compartment. As shown in Figures 2A–C, SPARC and PDI exhibited minimal colocalization in control nodules, despite the trafficking of SPARC through the ER-Golgi as a secretory product of these cells. In contrast, chondrocytes with COMP mutations D511Y (Figures 2D–F) and D469del (Figures 2G–I) showed clear and substantial levels of coincidence in their distribution of SPARC and PDI (indicated by the yellow fluorescence in Figures 2F and 2I). These data are consistent with those shown in Figure 1 and indicate that SPARC is retained in the rough ER, colocalizing with PDI, in PSACH chondrocytes with mutations in COMP.
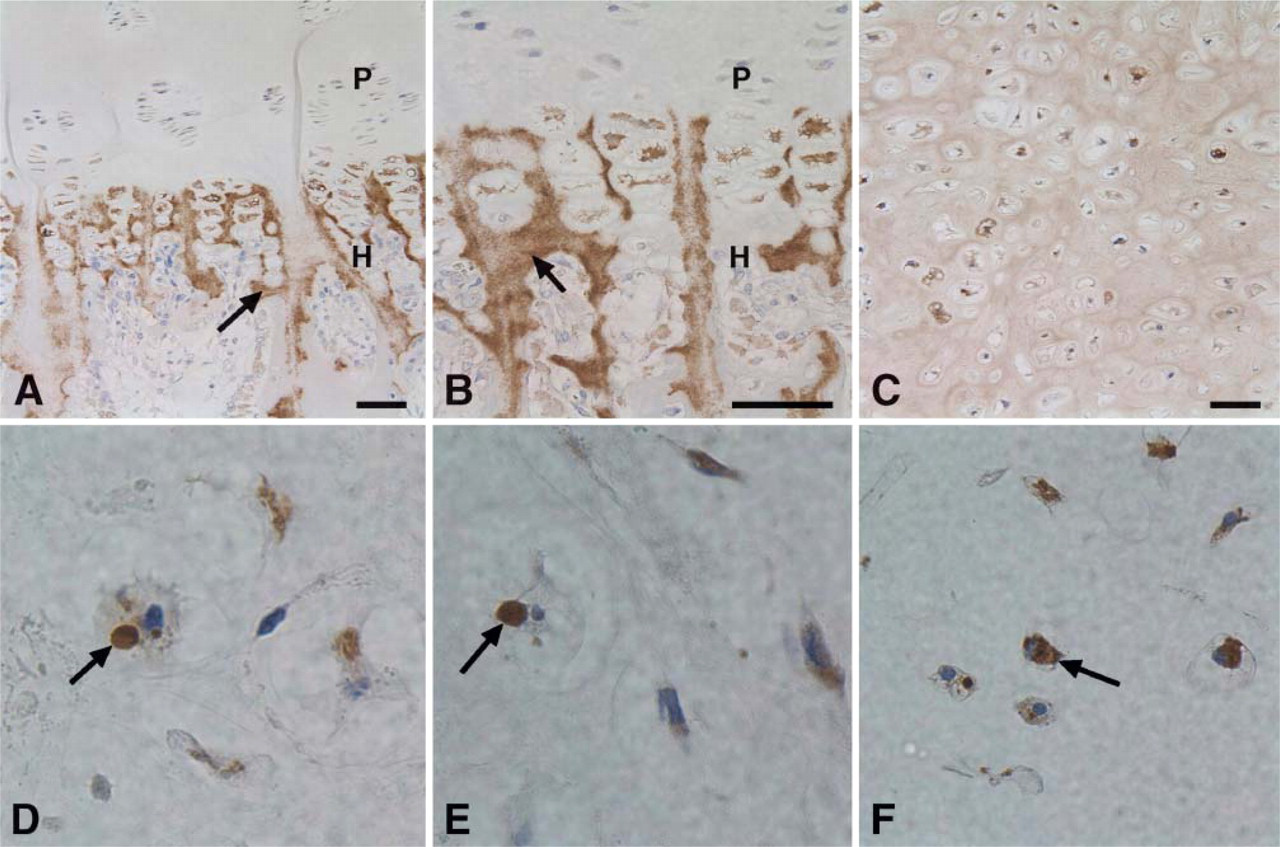
Distribution of SPARC in human growth plate and in cultured cartilage nodules. Low (
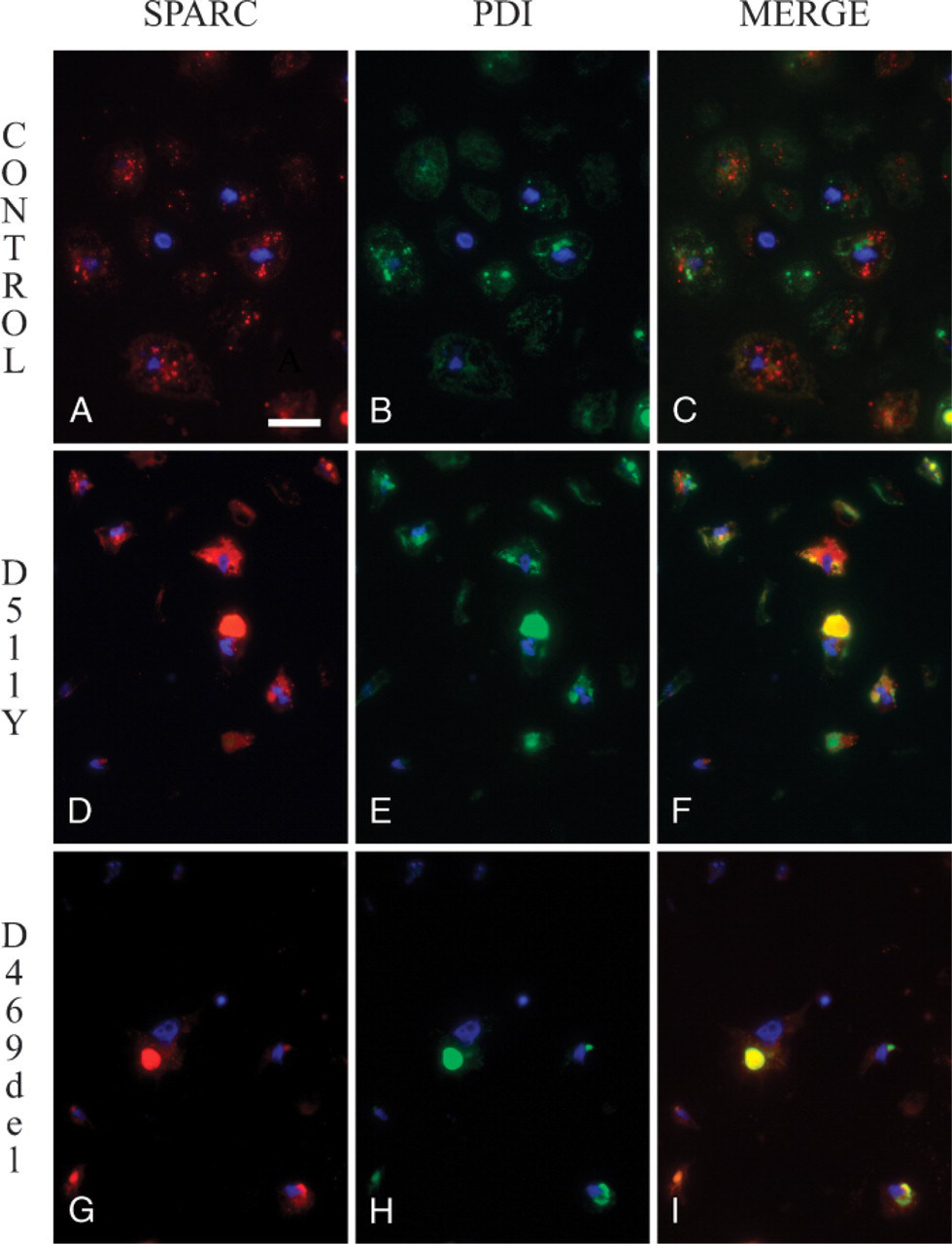
Localization of SPARC in cultured control and pseudoachondroplasia (PSACH) cartilage nodules. Cartilage nodules, as described in Figure 1, were derived from control (
The relative distribution of SPARC and COMP in cultured cartilage nodules is shown in Figure 3. In control nodules (Figures 3A–3C), SPARC was observed as punctate, intracellular granules, a distribution typical of a secreted protein (Figure 3). In contrast, COMP appeared primarily in the nodular ECM (Figure 3), and the merged image (Figure 3) reinforced the disparate distribution of these two secreted proteins. Coincidence between SPARC and COMP, however, was seen in chondrocytes from a PSACH patient with the COMP mutation D511Y (Figures 3D–F). Although the colocalization of these proteins was not seen in all cells, there were numerous merged images in which intracellular COMP and SPARC were coincident (Figure 3F). Extracellular COMP, however, was not coincident with SPARC in the ECM surrounding the D511Y chondrocytes (Figures 3D–3F). These images indicate that SPARC and a major structural protein of the cartilaginous ECM, COMP, are retained in the ER simultaneously in PSACH chondrocytes.
Discussion
Previous studies on cartilaginous tissues from PSACH patients have revealed defects in both the secretion and deposition of territorial ECM by resident chondrocytes (Hecht et al. 1998; Chen et al. 2004; Hecht et al. 2004; Hecht et al. 2005). Moreover, mutations in COMP, a protein not unique to cartilage, are responsible for the chondrodysplastic phenotype (Posey et al. 2004). Although a precise explanation for the retention of COMP in the ER has not been provided, it is known that certain of the mutations in COMP affect its binding of Ca+2 and its interaction with collagens I, II, and IX (Chen et al. 2000; Thur et al. 2001). Deficiencies in ECM affect not only the structural properties of the developing cartilage, but also affect directly on the proliferative, apoptotic, and differentiation-specific signals that regulate chondrocyte function (Beier 2005). Proteins that modulate the production, secretion, or assembly of tissue-specific ECM therefore become important to our understanding of connective tissue diseases in which ECM appears directly implicated.
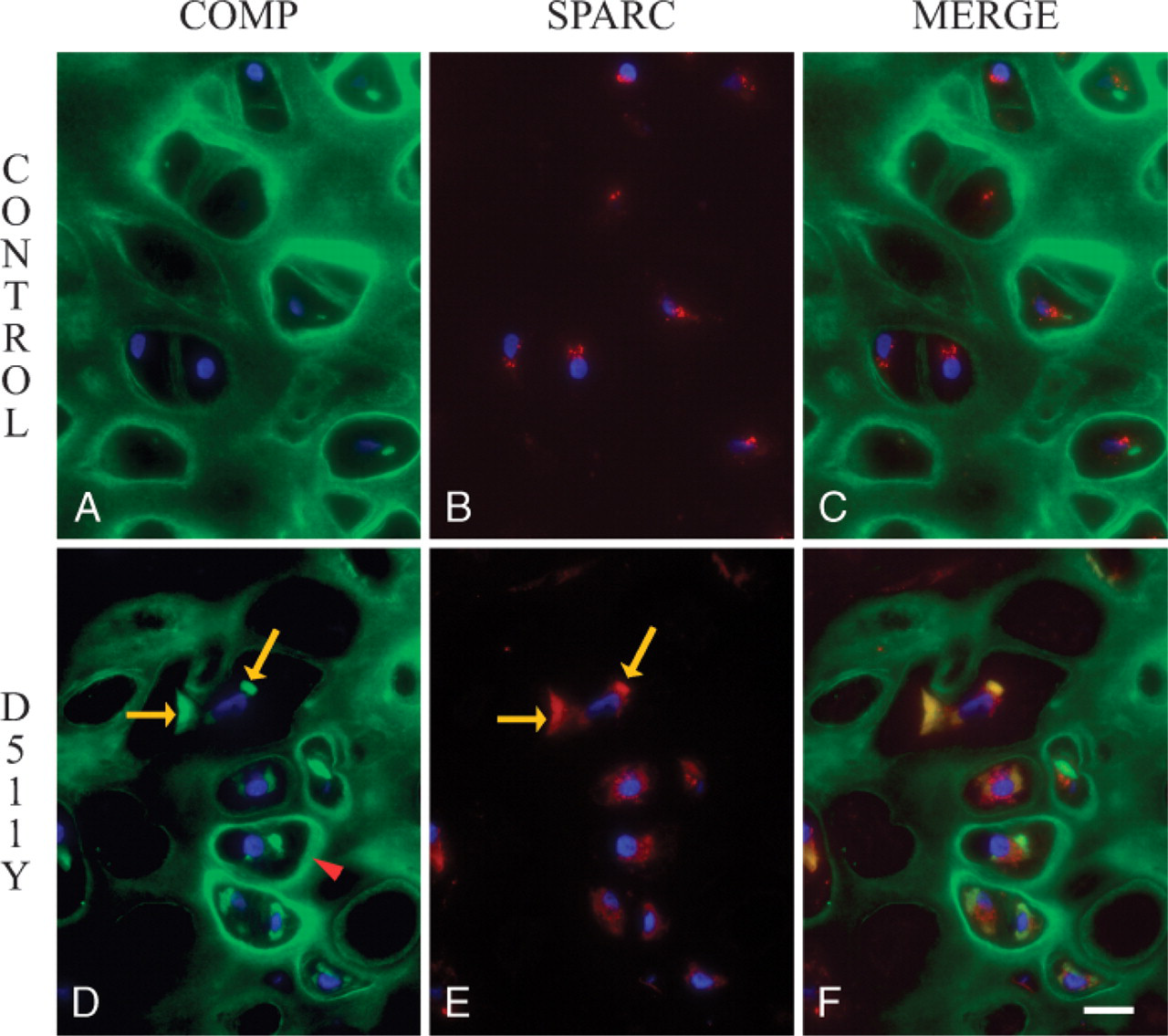
Distribution of cartilage oligomeric matrix protein (COMP) and SPARC in cultured cartilage nodules. Cartilage nodules were prepared as described in Materials and Methods, and sections were incubated with anti-SPARC IgG (reactivity shown in red) (
Certain of the matricellular proteins (e.g., osteopontin, thrombospondins 1 and 2, and SPARC) are produced by developing osseous tissues and are variously known to regulate aspects of ECM production, angiogenesis, and immune cell or macrophage function (Bornstein and Sage 2002). Their role in cartilage development and maintenance, however, is poorly understood. In this study, we therefore asked whether SPARC, a matricellular protein synthesized by chondrocytes, with demonstrated regulatory effects on collagen I production and assembly (Brekken and Sage 2001; Bornstein and Sage 2002; Bradshaw et al. 2003), might participate in the aberrant trafficking phenomenon previously observed with respect to COMP in PSACH chondrocytes (Chen et al. 2004).
The presence of SPARC in normal growth plate has been confirmed, and its preferential localization in the hypertrophic zone was recapitulated in organ cultures of human cartilage nodules. However, in cartilage nodules cultured from PSACH donors, SPARC was located primarily intracellularly, and coincidently with PDI, a resident chaperone of the rough ER. A further distinction was made between the distribution of SPARC and COMP, the latter observed largely in the nodular ECM. In contrast, SPARC and mutant COMP were coincident in a significant proportion of chondrocytes from a PSACH patient. The data therefore indicate that, in PSACH chondrocytes, the principal structural protein COMP and the matricellular protein SPARC, both of which are secreted proteins, are simultaneously retained in the ER.
SPARC-null mice develop osteopenia and exhibit a general reduction in collagenous ECM (Delany et al. 2000; Bradshaw et al. 2003). Within the dermis, the collagen I fibrils are smaller and more uniform in size, in comparison to those of age-matched, wild-type mice. That there are also structural defects in the lens capsule basement membrane indicates that the proposed regulatory effect of SPARC on ECM deposition/assembly is not limited to collagen I (Yan et al. 2005b). It is clear, however, that high levels of SPARC coincide with fibrosis and with secretion and remodeling of components of the ECM. As a typical matricellular protein, however, SPARC is not a resident structural ECM protein in that it is not generally found extracellularly in nonpathological tissues (Brekken and Sage 2001). In contrast, exogenous or endogenously produced SPARC has been reported to be translocated to the nuclei of endothelial and lens epithelial cells in vitro (Gooden et al. 1999; Yan et al. 2005a). The association of SPARC with mutated forms of COMP in PSACH chondrocytes suggests a new intracellular function for SPARC in the trafficking of COMP. Whether SPARC is acting in the capacity of a molecular chaperone or regulates the selective secretion of specific components are questions that should be resolved in future experiments.
Footnotes
Acknowledgements
This work was supported by National Institutes of Health Grant GM40711 (EHS) and a Shriners Hospital grant (JTH).
The authors thank Elizabeth Hayes for excellent technical assistance and Eileen Neligan for assistance with the manuscript.
