Abstract
Live cell imaging methods were used to characterize goblet cells expressing a MUC5AC domain fused to enhanced green fluorescent protein that labels the granule lumen. Golgi complex and endosome/lysosome elements largely resided in the periphery of the granular mass. On the contrary, a tubular meshwork of endoplasmic reticulum (ER) was in close contact with the mucin granules. This meshwork could be identified in fixed native human bronchial goblet cells labeled with an anti-calreticulin antibody. The potential biological significance of this ER network for mucin secretion is discussed.
We report here the spatial relationship between mucin granules and other organelles, including the Golgi complex, endosomes/lysosomes and the endoplasmic reticulum (ER) in HT29-SHGFP-MUC5AC/CK mucous cells. These cells, which have been characterized previously (Perez-Vilar et al., 2005), were obtained by transduction of human colon adenocarcinoma HT29-18N2 cells (e.g., Phillips et al. 1995) with a retrovirus expressing a fusion protein comprising a signal peptide, a His-tag, GFP, and the CK domain of MUC5AC at the C termini. Morphological studies demonstrated formation of typical mucous/goblet cells with abundant mucin granules. The granules stored large quantities of the fusion protein (SHGFP-MUC5AC/CK) together with endogenous MUC5AC. Addition of ATP, an established mucin secretagogue, resulted in granule discharge within minutes (Perez-Vilar et al. 2005). These results strongly suggested that these cells differentiated into mucous cells and, accordingly, were a suitable model system to study mucous/goblet cells.
Live HT29-SHGFP-MUC5AC/CK cells grown for 6–14 days in serum-free media (Phillips et al. 1995) were observed on the microscope stage of a Zeiss LSM 510 (University of North Carolina M. Hooker Microscopy Facility; Chapel Hill, NC) at 37C using 488-nm laser excitation for GFP and, when needed, 510 nm for markers tagged/labeled with DsRFP2 or rhodamine (Perez-Vilar et al. 2005). For detecting trans-Golgi elements, HT29-SHGFP-MUC5AC/CK cells were transfected with plasmid pGOLR and fugene-6 (Boehringer; Ingelheim, Germany), selected in G418-containing medium (500 μg/ml) and positive cells isolated by fluorescence activated cell sorting (University of North Carolina Flow Cytometry Facility; Chapel Hill, NC). The expression vector pGOLR was made by replacing the enhanced cyan fluorescent protein-encoding sequence in pGOLGI-ECFP (BD Bioscience; Palo Alto, CA) with a red fluorescent protein (DsRFP2)-encoding DNA sequence obtained from pDsRFP2 (Invitrogen Co.; Carlsbad, CA) by standard PCR. The resulting cells coexpressed SHGFP-MUC5AC/CK and GOLR, which contained the N-terminal, sorting region of human galactosyltransferase and, accordingly, was retained in the trans-Golgi compartment. As shown in Figure 1A, GOLR-containing trans-Golgi elements (magenta signal) were clearly separated from the granules containing SHGFP-MUC5AC/CK (green signal). Although we have not yet succeeded in expressing a cis-Golgi protein marker in live HT29-18N2 cells, we have studied instead the intracellular location of GM130, a well-characterized cis-Golgi protein (Nakamura et al. 1997), in cells fixed with paraformaldehyde, permeabilized with triton X100 and sequentially stained with mouse monoclonal anti-human GM130 (BD Bioscience) followed by rhodamine-conjugated secondary antibody (Jackson Immunochemicals Inc.; West Grove, PA) (Perez-Vilar et al. 2005). GM130 was detected in tubulovesicular structures around the granular mass (Figure 1B), although it must be noticed that the immunofluorescence protocol affected mucin granule morphology, producing a loss of its characteristic round geometry.
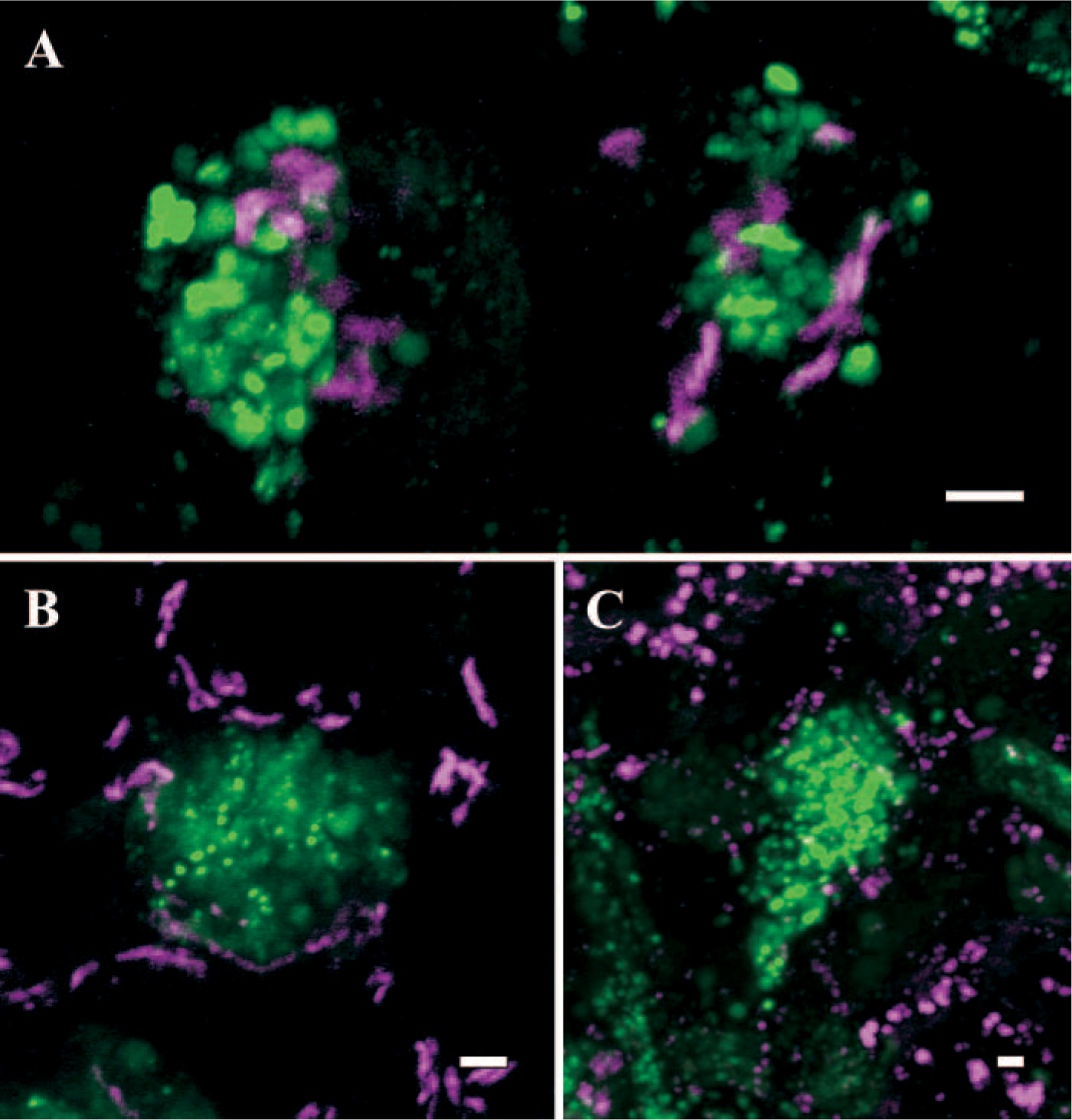
Spatial relationship between mucin granules and other organelles in HT29-SHGFP-MUC5AC/CK mucous cells.
To identify endosomes/lysosomes, live SHGFP-MUC5AC/CK mucous cells were incubated for up to 4 hr at 37C with wheat germ agglutinin-rhodamine (0.5 μg/ml; Vector Laboratories, Burlingame, CA) in regular media before changing the media to confocal buffer (HBSS supplemented with 20 mM HEPES, pH 7.2, 1% [v/v] FBS, 4.5 g/l glucose, essential and nonessential amino acids). As shown in Figure 1C, the internalized wheat germ agglutinin could not be detected in the SHGFP-MUC5AC/CK-labeled mucin granules, although it was found at the cell surface and in tubulovesicular structures scattered in the cytoplasm of goblet cells. Altogether, these results suggest that, in live mucous cells, Golgi complex elements and endosomes/lysosomes were largely excluded from the intergranular cytoplasm.
To identify the ER in live mucous cells, we generated HT29-SHGFP-MUC5AC/CK cells stably expressing the fluorescent protein DsRFP-ER using the expression plasmid pDsRFP2-ER (Invitrogen Co.) and the transfection agent fugene-6 as directed by the manufacturer (Boehringer Ingelheim). This red fluorescent fusion protein contains two ER retention signals, including the calreticulin N-terminal region and a C-terminal KDEL sequence. As shown in Figure 2A, DsRFP-ER was clearly distributed within the cytoplasm, but absent from the mucin granules, as expected for an ER luminal protein. At high magnification, however, a network of thin, DsRFP-ER-containing tubules encircling the granules was evident (Figures 2B and 2C). That only midsections of the cells were imaged for these studies indicated that these ER tubules were different from the cortical ER that surrounds the granular mass. The thin nature of the ER meshwork, however, made it difficult to prevent photobleaching of DsRFP-ER2 during imaging and, ultimately, its visualization when compared with the cortical ER. Independent evidence for the granule-associated ER network was obtained by fluorescence recovery after photobleaching analysis of live HT29-18N2-SHGFP-MUC5AC/CK cells (Perez-Vilar et al. 2005). Selected regions of cells maintained at 37C at the microscope stage were irreversibly photobleached at high laser power (80% laser power/100% transmittance) for 0.07–0.1 sec using the 488-nm line of the confocal microscope. Recovery of the fluorescent signal inside the bleaching region was imaged at 80% laser power/0.5% transmittance. Images were analyzed with Zeiss LSM software(Carl Zeiss; Verkauf, Germany). SHGFP-MUC5AC/CK diffuses throughout the whole ER with an apparent diffusion constant of ≃1.5 μm2/sec (Perez-Vilar et al. 2005). In contrast, mucin granules are independent structures and, accordingly, bleaching of an intact granule does not affect fluorescence of the surrounding granules. We took advantage of this difference to visualize the ER in the region occupied by granules.
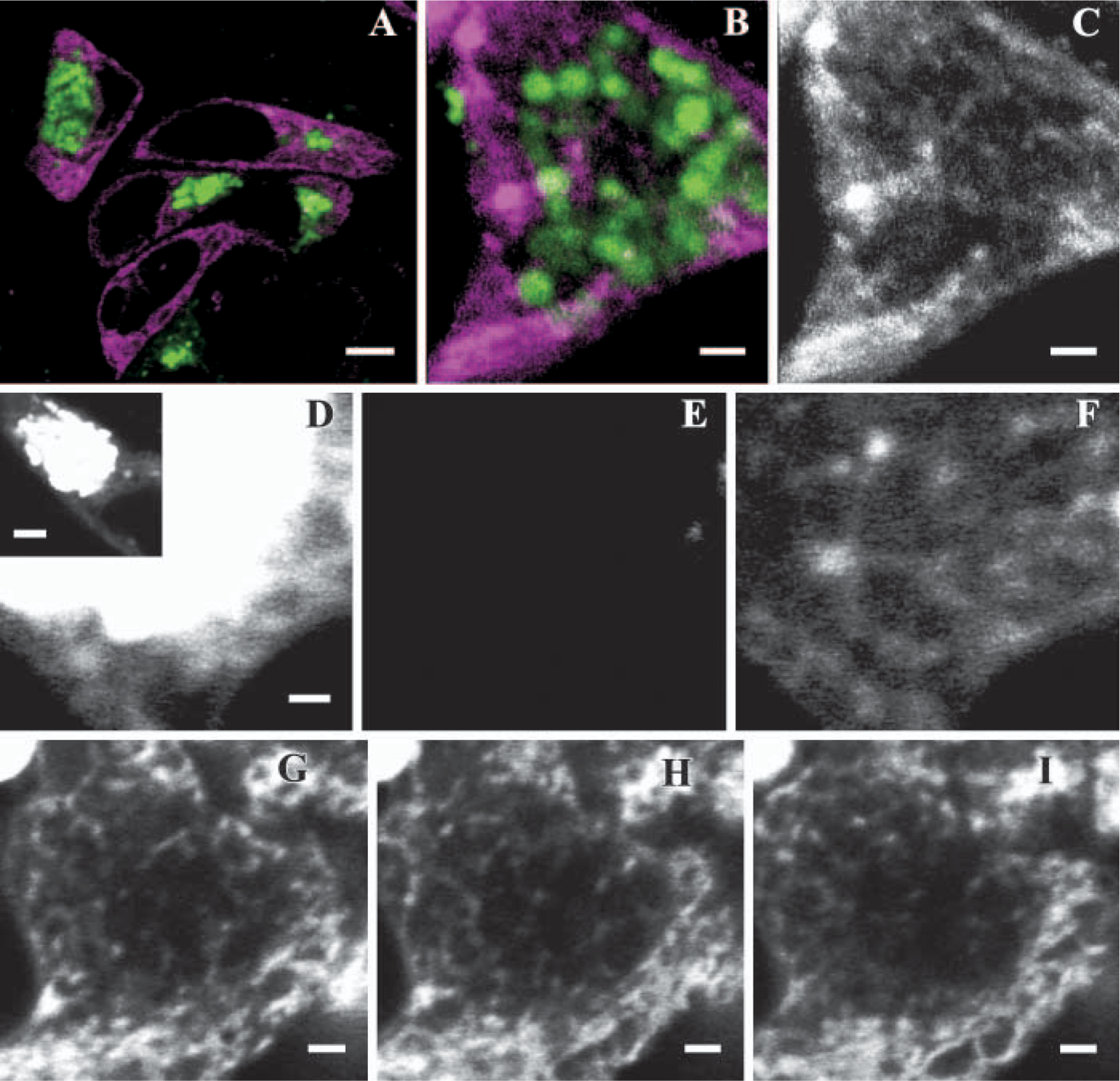
The mucin granule-associated endoplasmic reticulum (ER) tubular network in live HT29-SHGFP-MUC5AC/CK mucous cells. Low magnification xy-image of HT28-SHGFP-MUC5AC/CK cells coexpressing SHGFP-MUC5AC/CK (green signal) and DsRFP2-ER (magenta signal)
Thus midoptical sections of mucous cells were imaged in areas containing ER and mucin granules, with both organelles extending above and below the focal plane. A focal plane containing the nuclear envelope was also selected to better identify the ER after the bleaching. Because the intraluminal ER SHGFP-MUC5AC/CK signal was weaker than the granule signal, it was necessary to saturate the fluorescent signal in the granules to visualize the ER (Figure 2D). The entire granular mass was bleached at high laser power (Figure 2E), and the cell scanned every 30 sec afterwards. Within seconds after the bleaching pulse, a fluorescent network of interconnected tubules that was continuous with the nuclear envelope was visible in the region occupied by the bleached granules (Figure 2F). These results indicated that unbleached SHGFP-MUC5AC/CK from neighboring areas of the ER had mixed with the photobleached molecules within the ER tubular meshwork, as expected for connected regions. Figures 2G-2I show three consecutive, 0.55-μm apart, xy images of a live mucous cell 5 min after photobleaching the granular mass, showing the tubular nature of the ER associated with the granules.
To investigate whether the mucin-granule-associated ER network was present in native goblet/mucous cells, freshly isolated human bronchial epithelia from chronic obstructive pulmonary disease subjects, provided by the University of North Carolina Cystic Fibrosis Center under the auspices of protocols approved by the Institutional Committee on the Protection of the Rights of Human Subjects, were fixed with 4% (w/v) paraformaldehyde in PBS, pH 7.4, and embedded in paraffin. Sections (≃7-μm thick) were double labeled with a rabbit polyclonal anti-calreticulin antibody (Affinity Bioreagents; Golden, CO) and a mouse anti-MUC5AC monoclonal antibody (Lab Vision Co.; Fremont, CA) following procedures described earlier (Ribeiro et al. 2005). Images were captured on the DeltaVision RT Imaging System (Applied Precision, LLC; Seattle, WA) as a z-series with 0.15-μm focal steps through the entire specimen with a 64 X 1.4 numerical aperture objective and standard FITC and rhodamine filters. The entire dataset was deconvolved for each wavelength using DeltaVision's contrained iterative three-dimensional deconconvolution algorithm to improve image contrast. MetaMorph software (Universal Imaging Co.; Downington, PA) was used to assemble the figures using a single representative plane from the deconvolved z-series. Using this system, the mucin granule-associated ER tubular meshwork in superficial goblet cells was clearly revealed (Figure 3). Similarly to cultured goblet cells, the calreticulin-containing meshwork (Figures 3B, 3D, 3E and 3G) was visualized as thin tubules that appeared to encircle the mucin granules stained with anti-MUC5AC antibodies (Figures 3C, 3D, 3F, and 3G).
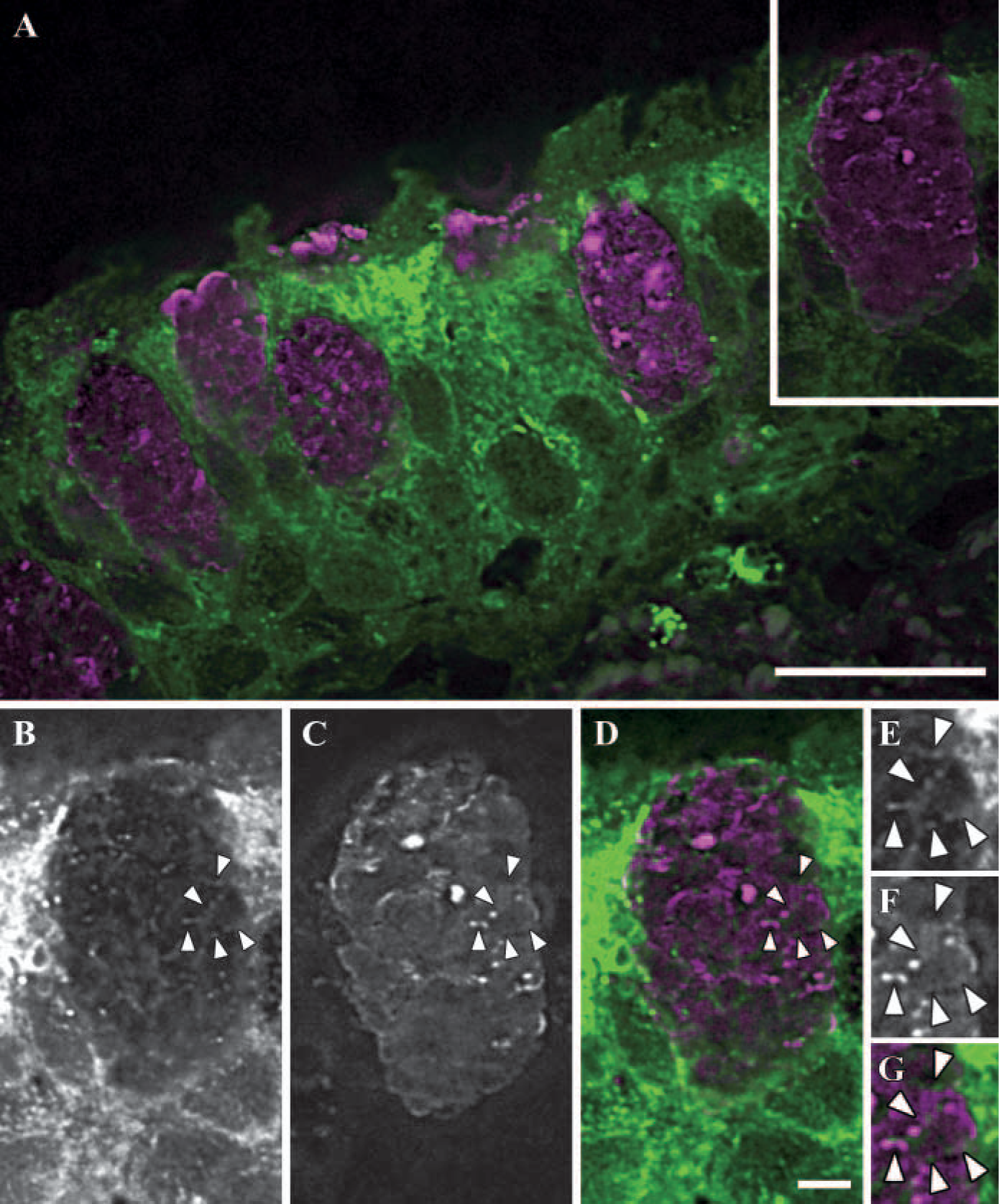
The mucin granule-associated endoplasmic reticulum (ER) tubular network in fixed human bronchial goblet cells. Representative histological section of human bronchial tissue, immunostained with anti-calreticulin (green color) and anti-MUC5AC (magenta color), showing numerous goblet cells
The close association of an ER network with mucin granules in goblet cells from native human bronchial epithelia is in agreement with the studies with live cultured mucous cells. Because HT29 cells were originally derived from a human colon adenocarcinoma, these results suggest that the mucin granule-associated ER meshwork is a general characteristic of mucous/goblet cells irrespective of the tissue these cells were derived from. In addition, the existence of ER structures closely associated with secretory granules in pancreatic acinar cells (Gerasimenko et al. 2002) suggests that such a morphological feature is not exclusive of mucous/goblet cells.
Although the function of this ER meshwork is not known, its close association with the mucin granules might indicate a role for this specialized ER compartment in granule homeostasis or exocytosis. The requirement for locally generated Ca2+ signals during granule-regulated secretion has been recently suggested by two independent studies. Thus Nguyen et al (1998) provided evidence that the mucin granule lumen could supply the Ca2+ required for its own exocytosis, assuming the existence of an inositol 1,4,5-triphosphate (InsP3)-activated Ca2+ channel in the granular membrane. Studies in rat SPOC cells by Rossi et al. (2004) also suggested the existence of an InsP3-independent release of Ca2+ from granules, or in close proximity, the ER. The close association between ER meshwork and granules found in the present study supports the view that the primary source of intracellular Ca2+ mobilization on mucin secretagogue signaling is the ER meshwork surrounding the granules.
In conclusion, our studies have revealed a close spatial association between mucin granules and thin extensions of the ER that appear to form a tubular meshwork surrounding the granules in live cultured mucous cells and fixed native human bronchial goblet cells. Future studies are warranted to address the functional role of this specialized ER network on Ca2+-dependent mucin secretion.
Footnotes
Acknowledgements
These studies were supported by the Cystic Fibrosis Foundation, grants PEREZ3I0 and PEREZV04G0 (to JPV), and the National Institutes of Health Grant DK-63030 (to JPV).
We thank Dr. L. Arnold (University of North Carolina Flow Cytometry Facility) and Mrs. K. Burns (UNC CF Histology Core) for fluorescence cell-sorting and histological procedures, respectively.
