Abstract
As early as the beginning of the twentieth century some data indicated that macromolecules are able to cross the intestinal mucosa to reach the blood. Further evidence was added over the years; however, pathways for this transport still remain to be established. We report here the transfer of two pancreatic enzymes, amylase and lipase, from the intestinal lumen to the blood. Both are present in higher concentrations in the intestinal mucosa and in blood of fed rats. Upon cholinergic stimulation of pancreatic secretion, there was not only an increase in blood enzyme concentrations, but evidence for internalization by duodenal enterocytes was obtained. Following insertion of fluorochrome-tagged amylase and lipase into the duodenal lumen of fasting rats, blood and intestinal tissues were sampled at different time points. Serum activities for both enzymes clearly increased with time. Light microscopy established internalization of both proteins by duodenal enterocytes, and immunogold outlined the pathway taken by both proteins across the enterocytes. From the intestinal lumen, enzymes are channeled through the endosomal compartment to the Golgi apparatus and to the basolateral membrane reaching the interstitial space and blood circulation. Transcytosis through the intestinal mucosa thereby represents an access route for pancreatic enzymes to reach blood circulation.
P
Both pancreatic amylase and lipase are found in blood (Janowitz and Dreiling 1959; Yacoub et al. 1969; Rohr and scheele 1983). Their presence in circulation is a well-established fact, and their levels fluctuate under several conditions. Pancreatitis, a pathological situation often diagnosed through plasma levels of amylase and lipase, is characterized by dramatic increases in the level of circulating pancreatic enzymes (Ujihira et al. 1965; Yacoub et al. 1969; Rohr and Scheele 1983; Tietz and shuey 1993). This is due to an abnormal discharge of zymogen granules at the level of the basolateral membranes of acinar cells (Gaisano et al. 2004). More interestingly, in diabetes, the pancreatic secretion of amylase and lipase undergoes major changes influencing circulating levels. These changes take place without any apparent alteration or discharge at the basolateral pole of the acinar cells (Barneo et al. 1990). In fact, in diabetes, production of both pancreatic amylase and lipase fluctuate; amylase secretion decreases dramatically whereas lipase secretion increases 2-fold (Bazin and Lavau 1979; Gregoire and Bendayan 1986; Bendayan and Gregoire 1987; Bendayan and Levy 1988; Barneo et al. 1990). Blood levels closely follow those changes (Ujihira et al. 1965; Barneo et al. 1990). Moreover, variations in circulating amylase and lipase also occur under normal conditions. A relationship actually exists between circulating levels of digestive enzymes and the feeding state of the animal (Isenman et al. 1999). Digestive activity, intestinal motility, and pancreatic secretory activity being regulated by the same hormonal and neural mediators are directly interconnected (Keller and Layer 2002); therefore, blood levels of amylase and lipase are higher in fed animals than in fasted animals.
Even though a correlation clearly exists between pancreatic amylase and lipase exocrine secretion and their levels in circulation, the pathways by which these digestive enzymes reach the blood remain largely unknown (Isenman et al. 1999). Several hypotheses were put forward over the years. Among these, an endocrine secretion of the exocrine pancreas at the basolateral membrane of the acinar cells (Janowitz and Dreiling 1959; Tietz and Shuey 1993; Isenman et al. 1999) and a leakage across the pancreatic duct wall were proposed (Isenman et al. 1999). A third possibility would be a paracellular passage of intestinal luminal content through leaking junctional complexes, a possibility suggested by in vitro studies using Caco-2 cells (Bock et al. 1998). However, conclusive evidence for such pathways remains to be demonstrated
The present study proposes an alternative route of access for these enzymes to reach the blood circulation. This pathway would consist of the absorption of lipase and amylase by the intestinal mucosa followed by transcytosis through the enterocyte to reach the intestinal subepithelial space. The capability of intestinal enterocytes to absorb and transport intact macromolecules has been considered as the characteristic of the intestinal tissue of embryo and newborn (Sandborn et al. 1975; Udall et al. 1981). It is also a characteristic of pathologies affecting the digestive tissue integrity (Weiner 1988). Nowadays, however, the absorption of proteins by enterocytes and their transport to the basolateral pole could be considered as a rather normal physiological process that takes place in intestinal tissue of adult animals (Cornell et al. 1971; Warshaw et al. 1971; Bendayan et al. 1990,1994; Bendayan 2000; Ziv and Bendayan 2000; Bruneau et al. 2003a). Indeed, we have previously demonstrated that insulin (Bendayan et al. 1990,1994) and the pancreatic bile salt-dependent lipase (BSDL) (Bruneau et al. 1998,2001,2003a) are internalized by enterocytes and transferred without degradation to the blood circulation. The first evidence for the intestinal absorption of insulin was reported in 1987 (Ziv et al. 1987). The pathway undertaken by insulin to get from the intestinal lumen, through the enterocyte, to the blood circulation was established later (Bendayan et al. 1990, 1994; Ziv and Bendayan 2000). BSDL, a pancreatic lipase, was also demonstrated to gain entry to the blood circulation through enterocytes (Bruneau et al. 1998,2003a)
The present study shows that this physiological internalization by enterocytes is a more general phenomenon also occurring for pancreatic amylase and lipase under normal in vivo conditions. We have demonstrated that circulating levels of enzymes are related to their presence in the intestinal lumen, that internalization of amylase and lipase by enterocytes takes place, and that progression of the absorbed enzymes along a transcytotic pathway allows them to reach the blood circulation.
Materials and Methods
Antibodies
Primary antibodies were rabbit anti-human salivary α-amylase (Sigma-Aldrich; Oakville, Canada), rabbit anti-fluorescein isothiocyanate (FITC) (Dakopatts; Glostrup, Denmark), and sheep anti-human lipase (United States Biological; Swampscott, MA). Secondary antibodies for light microscopy were anti-goat IgG conjugated to crystalline tetramethylrhodamine isothiocyanate (TRITC) (Sigma-Aldrich) and anti-rabbit IgG-FITC (Chemicon International; Temecula, CA). For the immunoperoxidase we used the rabbit anti-goat IgG conjugated to peroxidase (Pierce Biotechnology; Rockford, IL). For electron microscopy, we used anti-goat IgG-gold complex (Sigma-Aldrich) and protein A-gold complex prepared with 10-nm gold particles as described previously (Ghitescu and Bendayan 1990). For immunoblots, an anti-rabbit IgG-horseradish peroxidase (Roche Molecular Biochemicals; Laval, Canada) was used.
Amylase and Lipase-FITC Experiments
Enzymes were tagged with FITC (fluorescein isothiocyanate, Isomer I; Sigma-Aldrich) according to methods described previously (Bendayan and Londoño 1996). Sixty mg of α-amylase (Type II-A,
Animal Experimentation
Male Sprague Dawley rats (Charles River Canada; St. Constant, Canada) weighing between 200 and 250 g were used for all experiments. Animals were housed and handled according to the guidelines from the Canadian Council on Animal Care. They were kept on a standard diet with free access to food and water. Rats were fasted for 12 hr before the experiments and were anesthetized by an IP injection of urethane (1 g/kg body weight).
Tissue Processing
Immunohistochemistry
For the direct detection of FITC-tagged enzymes, tissue sections were deparaffinized, rehydrated, and washed in 0.01 M phosphate-buffered saline (PBS). Tissue sections were counterstained with Evans Blue (0.01% in PBS), mounted with a coverslip using a 50% glycerol in PBS solution, and examined using a Leitz DMRB light microscope (Leica; St-Laurent, Canada).
For the immunodetection of amylase antigenic sites, tissue sections were rehydrated and incubated for 2 hr at room temperature (RT) with the anti-amylase antibody (dilution: 1/300), washed in PBS, and incubated 1 hr with the anti-rabbit IgG-FITC (dilution: 1/250). For lipase immunodetection, the anti-lipase antibody was used at 1/100 dilution, followed by 1 hr with the anti-goat IgG-TRITC (dilution: 1/200).
Specificity of both antibodies was assessed by immunoblot as well as by immunocytochemistry, adsorbing them with their corresponding antigens (24 hr at 4C) prior to performing the immunostainings. Adsorption led to absence of labeling. Omitting the primary antibodies also resulted in absence of specific labeling. In addition, both the anti-amylase and anti-lipase antibodies yielded specific stainings on pancreatic tissue sections (results not shown).
For the immunoperoxidase technique, lipase detection was carried out by a 2-hr incubation with the anti-lipase antibody (dilution: 1/100) followed by a 1-hr incubation with anti-goat IgG-peroxidase (dilution: 1/200) and a 2-min incubation with the DAB peroxidase substrate, all at RT. Omission of the primary antibody resulted in absence of staining.
For electron microscopy, anti-amylase (dilution: 1/300), anti-FITC (dilution: 1/250), and anti-lipase (dilution: 1/100) were used as primary antibodies. Incubations were for 2 hr at RT followed by a 30-min incubation with protein A-gold (dilution: 1/10) or with the anti-goat IgG-gold (dilution: 1/15) (Bendayan 1995). Tissue sections were pretreated with sodium metaperiodate (Bendayan 1995). Tissue sections were stained with uranyl acetate and examined using a Philips 410 electron microscope (Philips; Montreal, Canada). Adsorption of the antibodies with their corresponding antigen resulted in absence of labeling.
Evaluation of the Labelings
Morphometric evaluation of the labelings obtained by immunogold using anti-FITC antibody was performed using Videoplan 2 image processing system (Carl Zeiss; Toronto, Canada). Immunogold densities over cell compartments for both amylase-FITC and lipase-FITC at each time point were evaluated as described previously (Bendayan 1995). At least 50 micrographs at X12,000 magnification were analyzed for each experiment at each time point. Results are reported as mean values; number of gold particles per μm2 ± SEM. Microvilli as well as basolateral membranes are tightly associated among themselves. Basolateral membranes in particular make several foldings and interdigitations. Thus, their labelings were evaluated in reference to their area rather than to their length.
Western Blot Analysis
Presence of lipase-FITC and amylase-FITC in sera was demonstrated by migration of serum samples in 7% SDS polyacrylamide gel electrophoresis (PAGE), followed by immunoblotting for 2 hr at RT with the primary antibody, either anti-amylase (dilution: 1/10,000) or anti-FITC (dilution: 1/35,000), and 1 hr incubation at RT with anti-rabbit IgG-horseradish peroxidase antibody (dilution: 1/20,000). Detection was carried out after 1 min incubation with Lumina-LightPLUS substrate (Roche Molecular Biochemicals).
Biochemical Analysis
The Intersect System, Direct Amylase Reagent (Intersect Systems, Inc.; Longview, WA) was used to assess serum amylase activity. The Intersect System is intended for the quantitative kinetic determinations of serum α-amylase activity at 405 nm. Twenty five μl of serum was added to 1 ml of Direct Amylase Reagent. Sixty sec later the absorbance was registered followed by a second reading 60 sec later. Incubations were carried out at 37C. Serum lipase activity was measured using Lipase-PS Kit (Trinity Biotech; Jamestown, NY). The Lipase-PS system is intended for the quantitative kinetic determination of serum pancreatic lipase activity at 550 nm. Fifteen μl of serum was added to 900 μl of the substrate solution. Three hundred μl of the activator reagent was added 3 min later. An initial absorbance reading was taken 3 min later and a final one, 2 min after. Incubations were carried out at 37C. All controls for enzyme assays were carried out using Accutrol-TM Normal (Sigma Diagnostics; Oakville, Canada). Results are reported as mean values of serum activities ± SEM. All statistics for morphological and biochemical data were carried out using Student's
Results
Amylase and Lipase in Fasted and Fed Rats
Amylase and Lipase in Rats that Underwent Cholinergic Stimulation of Pancreatic Secretion
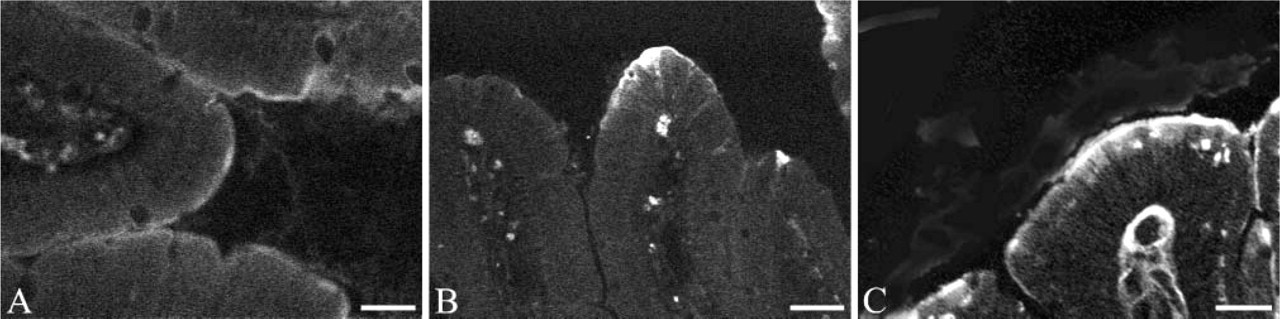
Light microscopy. Immunofluorescence detection of amylase and lipase using corresponding specific antibodies in the intestinal mucosa of fasted and fed rats. (
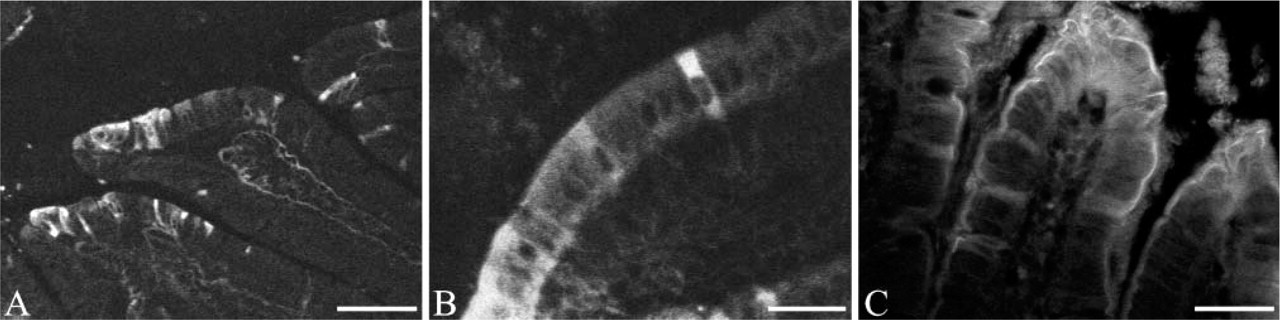
Light microscopy. Immunofluorescent detection of amylase and lipase using corresponding specific antibodies in the duodenal mucosa of carbachol-treated rats. (
Insertion of FITC-tagged Pancreatic Enzymes in the Intestinal Lumen
To further establish amylase and lipase internalization by enterocytes, solutions of exogenous amylase and lipase tagged with FITC were inserted into the duodenal lumen. Tissues were sampled at different time points and examined by light and electron microscopy.
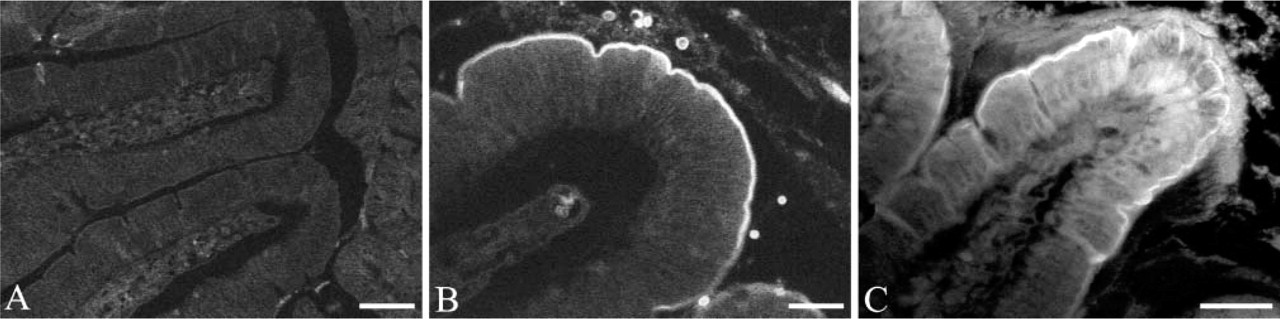
Light microscopy. Direct detection of FITC on duodenal tissue before and after the insertion of amylase-FITC in the intestinal lumen. (
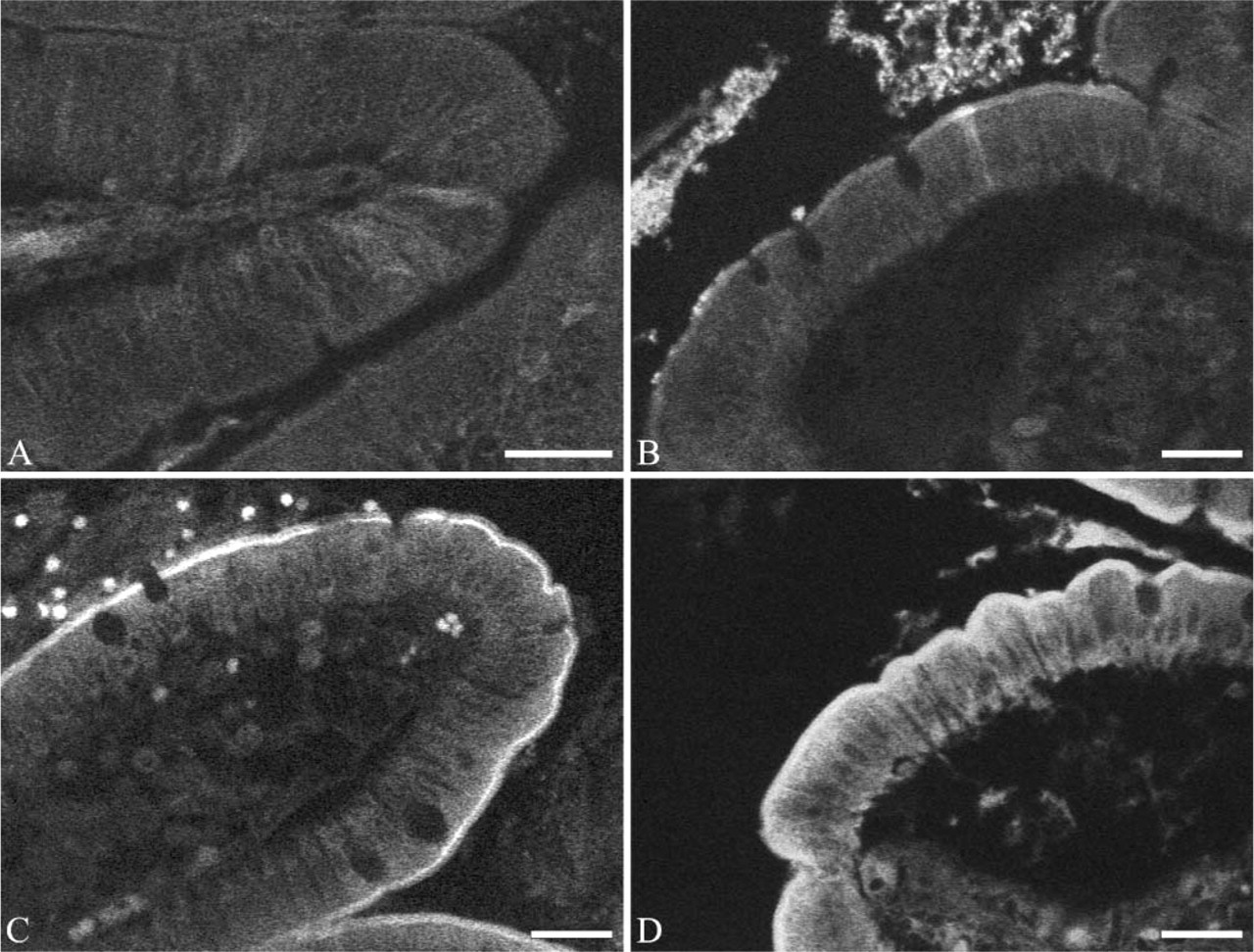
Light microscopy. Direct detection of FITC in duodenal tissue before and after the insertion of the lipase-FITC complex. (
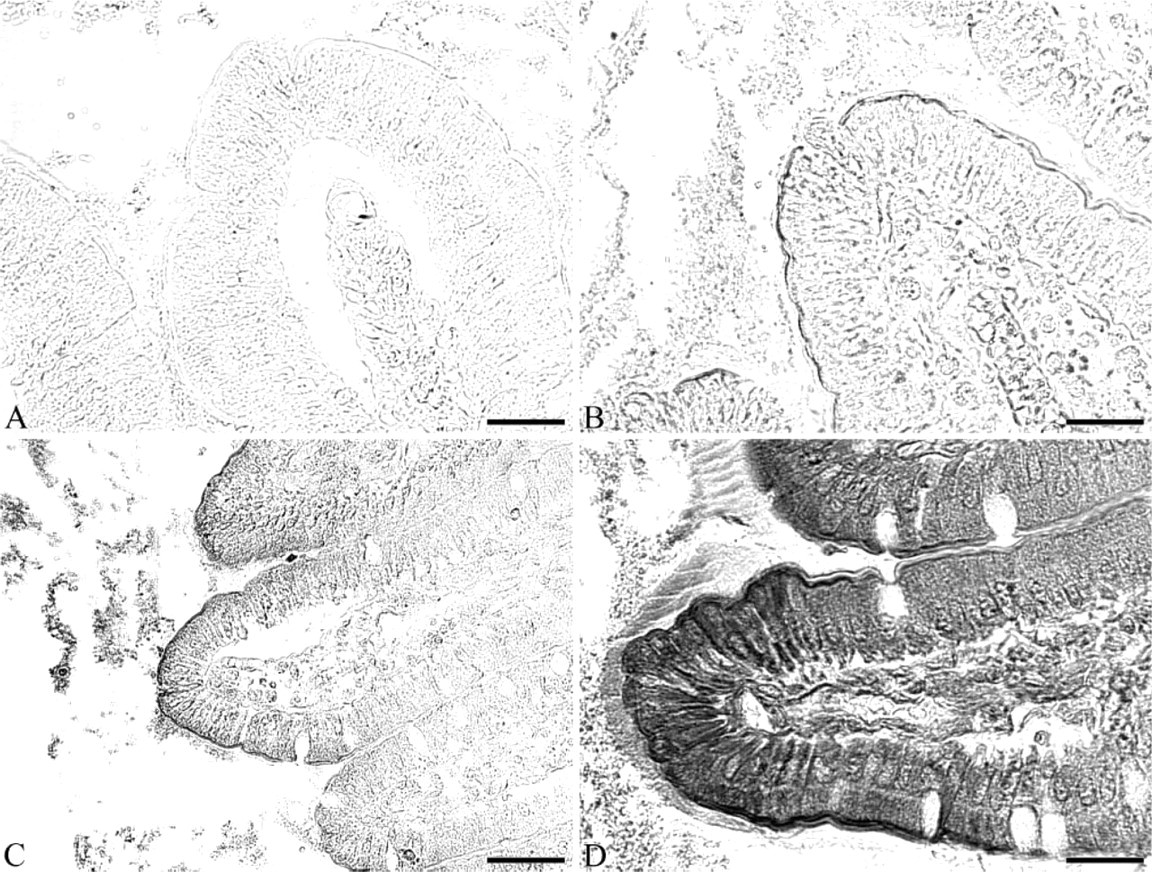
Light microscopy. Immunoperoxidase staining revealing lipase in duodenal tissue using the anti-lipase antibody before and after the insertion of lipase-FITC into the duodenal lumen. (
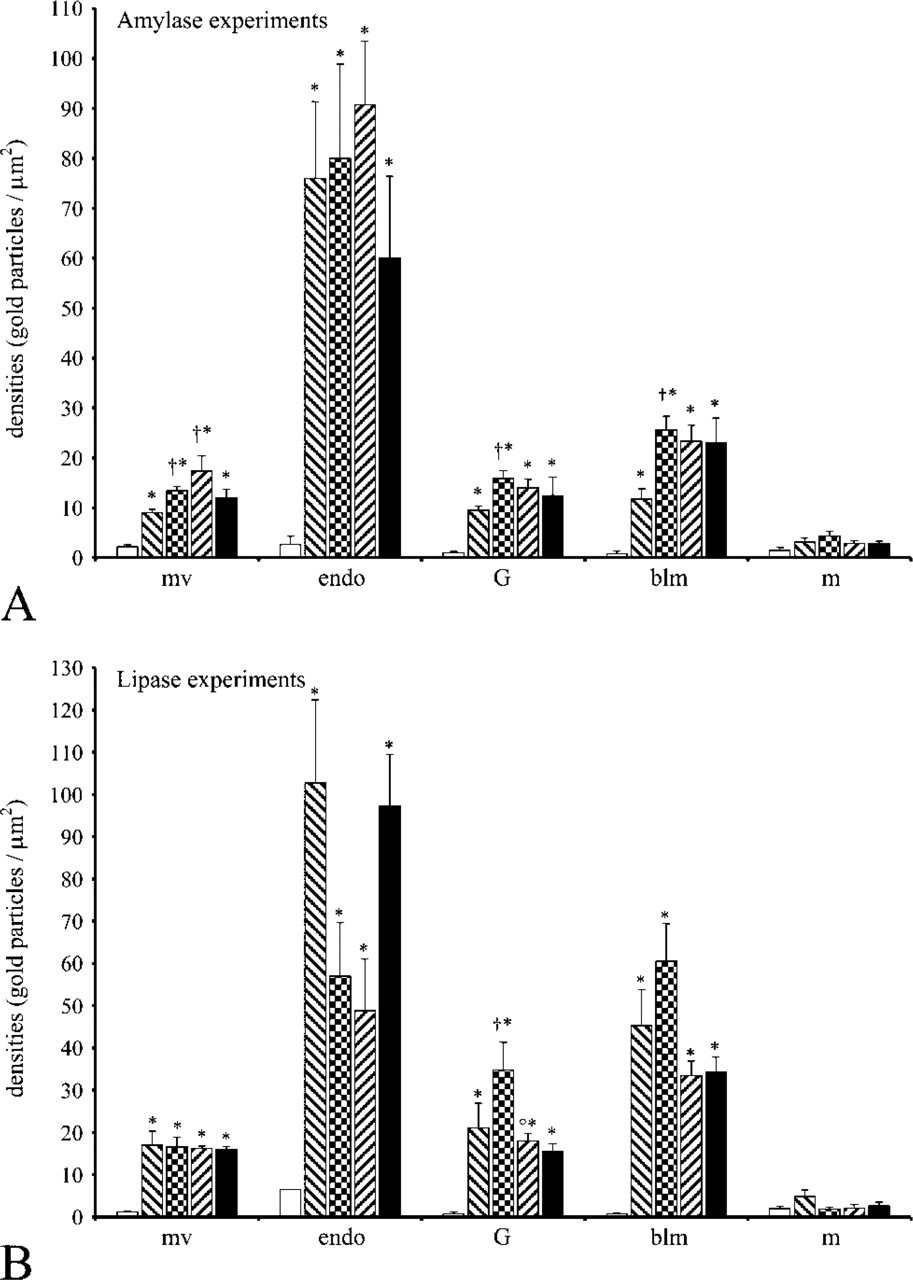
Quantitative evaluations confirmed the subjective observations. Figure 9 illustrates the labeling densities obtained for amylase-FITC and lipase-FITC experiments. Although values at time 0 remain very low and reflect background staining, labeling densities obtained in all other compartments in addition to mitochondria at all time points are significantly higher than background. The endosomal compartment appears to be the one displaying the highest density values at all time points, although some fluctuations were noted among time points for lipase (Figure 9B). However, deviations were large. Such large deviations might be due to heterogeneity in absorbing capacities among cells. Labeling densities of the Golgi apparatus remained constant and high, starting at 15 min. Similarly, labeling densities of the basolateral membranes became significant at 5 min and increased afterwards. This corroborates results obtained by biochemical determinations on amylase and lipase serum activities, which increased starting at 5 min as described in the next paragraphs. In contrast, labeling densities over mitochondria remained very low and showed no significant fluctuations over time. These values reflect background staining and are comparable to those obtained at time point 0.
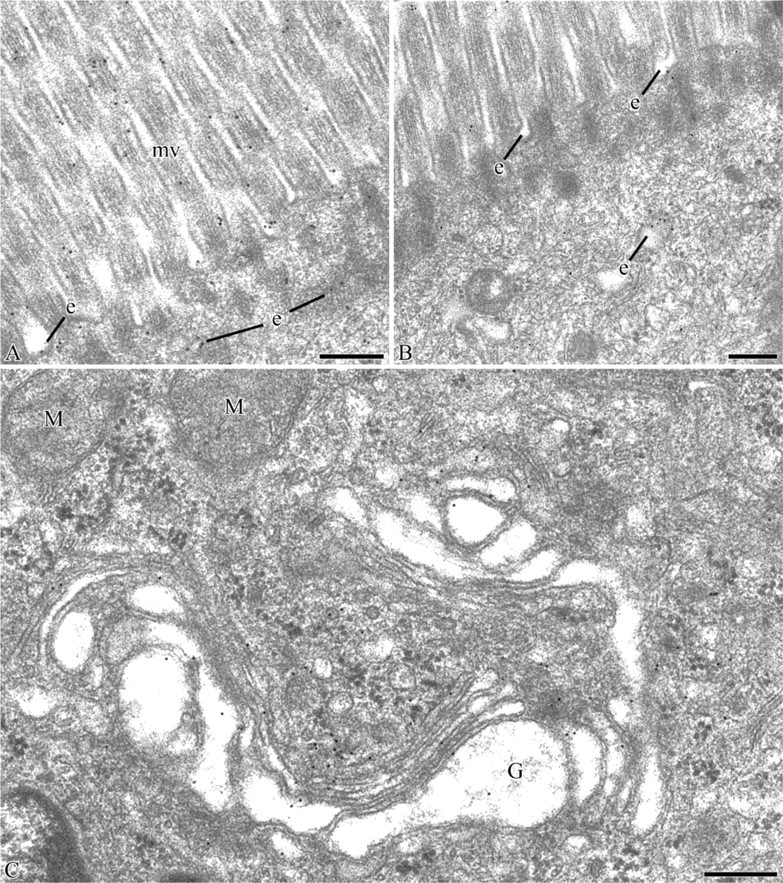
Electron microscopy. Immunocytochemical detection of amylase-FITC in the duodenal tissue using the anti-FITC/protein A-gold approach. (
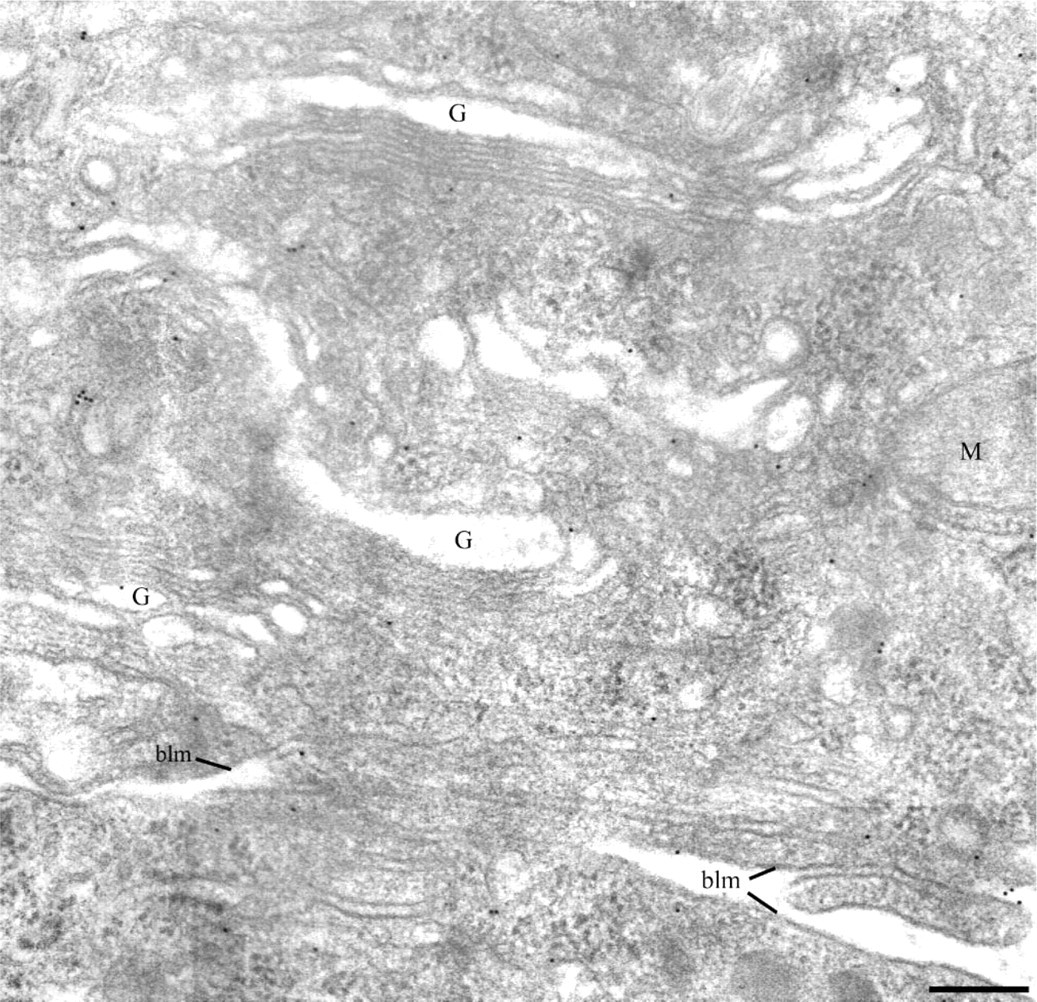
Electron microscopy. Immunocytochemical detection of lipase-FITC in the duodenal tissue using the anti-FITC/protein A-gold approach. At t = 30 min, gold labeling is present over the Golgi apparatus (G) and associated with the basolateral membrane (blm). No labeling is present over mitochondria (M). Bar = 0.25 mm.
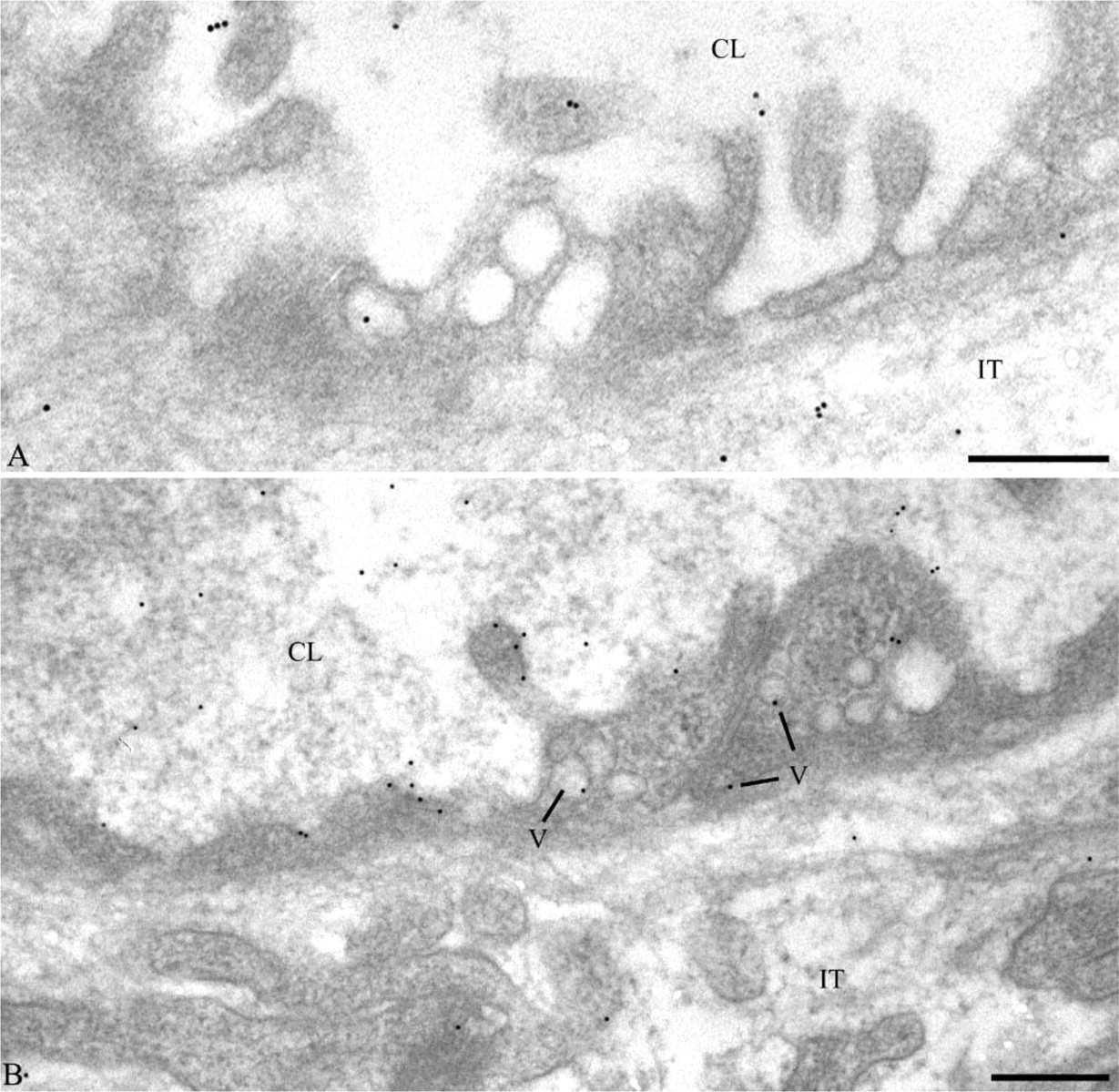
Electron microscopy. Immunocytochemical detection of amylase-FITC (
Amylase-FITC (
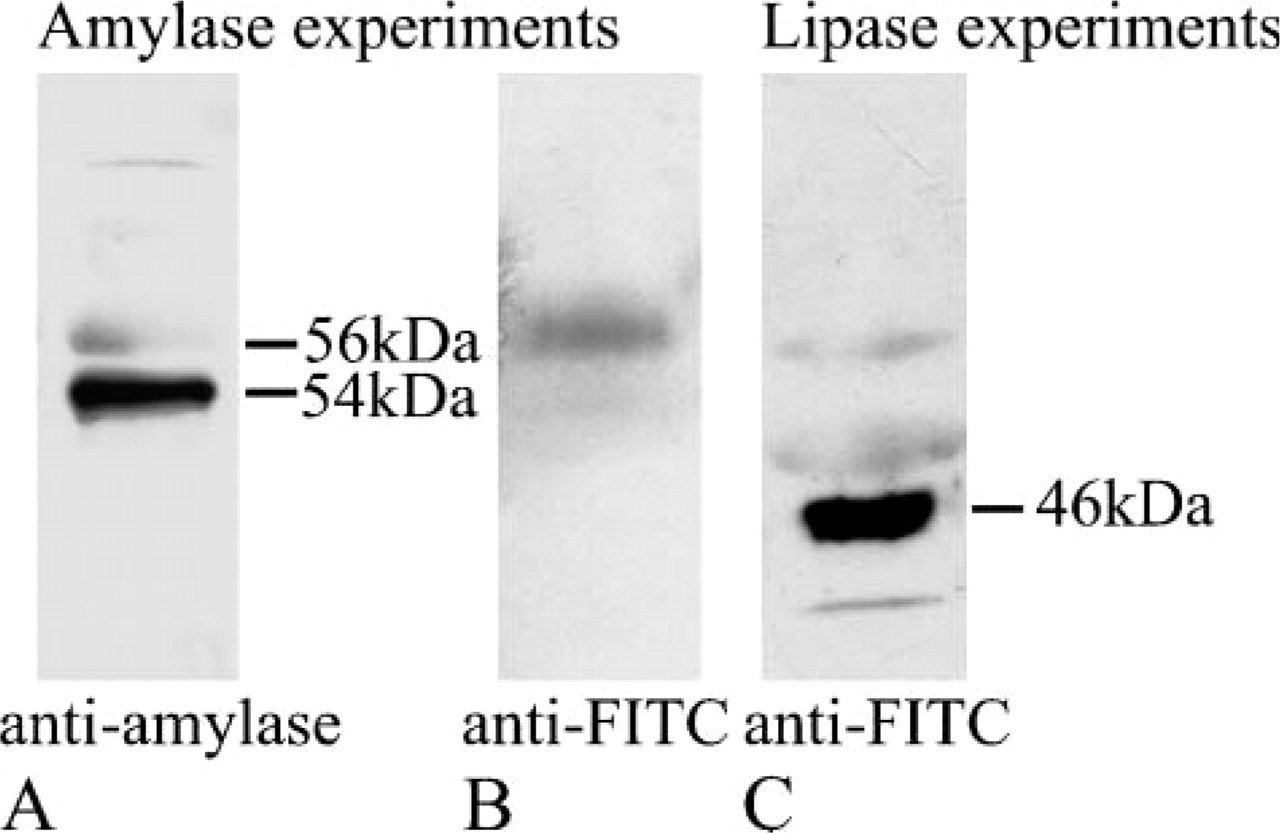
Immunoblot detection of amylase-FITC (
In addition, for the control experiments the levels of amylase and lipase before and 30 min after insertion of the solution not containing amylase or lipase were 61 ± 4.1 IU/liter and 57 ± 8 IU/liter for serum amylase and 66.1 ± 10.5 IU/liter and 67 ± 10.6 IU/liter for lipase, respectively.
Discussion
The present study demonstrates that the intestinal mucosa constitutes a route of access for amylase and lipase to the blood circulation. This may represent the main or alternative route to others previously proposed. We first confirmed the correlation existing between digestive activity and appearance of those enzymes in blood. Pancreatic secretion is stimulated by feeding, and this results in an increase in pancreatic enzyme activities in serum. Those results are in accordance with data published previously demonstrating that levels of serum amylase double upon feeding, suggesting the existence of a correlation between feeding, pancreatic function, and levels of circulating amylase (Schneyer and Schneyer 1960). The question that remained to be elucidated is the pathway by which pancreatic enzymes reach the blood circulation.
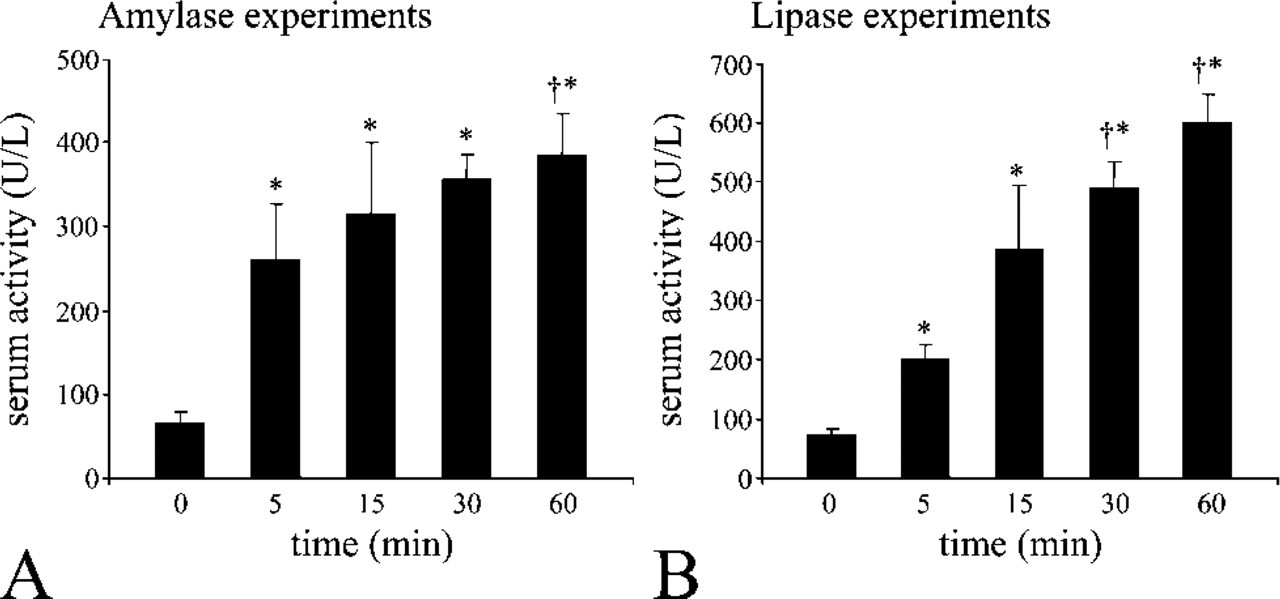
Enzyme activities of serum samples from duodenal insertion experiments. (
Over the years, several propositions were put forward, among them a potential endocrine secretion of the exocrine pancreatic acinar cells (Janowitz and Dreiling 1959; Tietz and Shuey 1993; Isenman et al. 1999) and a leakage of pancreatic enzymes across the pancreatic duct wall (Isenman et al. 1999). However, no direct evidence has established those pathways in normal conditions. Based on morphological and biochemical results, we propose here an alternative pathway through the intestinal mucosa by which pancreatic enzymes present in the duodenal lumen reach the circulation. Indeed, in addition to pancreatic cells and pancreatic ducts that are in contact with pancreatic enzymes and, therefore, always pinpointed as responsible for the transferring of those enzymes to the blood, the intestinal lumen is another site exposed to pancreatic enzymes but rarely fully considered as a partaker in this matter.
We demonstrate here the capability for the duodenal mucosa to transfer amylase and lipase from the intestinal lumen to the connective tissue. The presence of both amylase and lipase in the duodenal mucosa was higher in enterocytes of fed animals when compared with fasted ones. This indicates internalization of the enzymes by some enterocytes under normal conditions. To strengthen those observations we increased pancreatic enzyme concentrations in the duodenal lumen by stimulating pancreatic secretion. Serum activities of both amylase and lipase were found to increase under those conditions. Upon immunodetection, we found numerous, but notall, duodenal enterocytes displaying a strong cytoplasmic signal for both lipase and amylase as compared with tissues from control rats. These experiments strengthen the concept that enterocytes may be able to internalize pancreatic enzymes in relation to levels of pancreatic enzymes in the duodenal lumen. Morphological evaluation enabled us to affirm that the pancreatic cells remained intact upon stimulation of secretion. Indeed, several studies over the years have stated that pancreatic stimulation by cholinergic agonists such as carbachol increases circulating levels of amylase through an endocrine basolateral secretion by the pancreatic acinar cells (Janowitz and Dreiling 1959; Tietz and Shuey 1993; Isenman et al. 1999). It was demonstrated that multiple supramaximal doses of cholinergic agonists generate a reorientation of secretory granules from the apical pole of the acinar cells to the basolateral one, leading to secretion into the connective tissue (Scheele et al. 1987; Gaisano et al. 2001,2004) or an elevated presence of lysosomes in acinar cells due to increased lysosomal activity (Adler et al. 1983). In our experiments, we used a single non-maximal cholinergic stimulation that did not trigger reorientation of secretory granules, alteration in cellular polarization, or cellular integrity.
To establish the transcytotic pathway in enterocytes, we exposed the duodenal mucosa in situ to high concentrations of enzymes tagged to FITC molecules. Tagging amylase and lipase to FITC allowed us to discriminate between exogenous and endogenous enzymes as well as enabling us to follow their progression within the intestinal cells. It also allowed us to detect the enzyme-FITC complex in the serum, establishing the fact that intact enzyme molecules are internalized by the intestinal mucosa and discharged into the connective tissue to reach the blood circulation. Furthermore, the fact that the enzyme-FITC complex is found in circulation establishes that the increase in circulating enzymes originates from the intestinal mucosa. A substantial and extended association of the enzymes to the brush border was detected within the first 15 min and the signal progressed from the apical toward the basolateral region of the cells. In a previous study we demonstrated that BSDL, another pancreatic enzyme, is also transported from the intestinal lumen to the blood circulation through the intestinal mucosa (Bruneau et al. 2003a). In that same study we confirmed that the transport is quite specific because radiolabeled BSA was not internalized by enterocytes and did not reach the circulation (Bruneau et al. 2003a).
Electron microscopy immunocytochemistry confirmed and established the transcellular pathway. Gold immunolabeling was associated with the microvilli, forming endosomes, Golgi apparatus, and basolateral membranes. Those observations were underlined by assessing the labeling densities over the different compartments at different time points. Labeling was also found in the interstitial space and blood capillaries. Such transcytotic pathway matches the one previously established for BSDL, another pancreatic lipase, and insulin (Bendayan et al. 1990,1994; Ziv and Bendayan 2000; Bruneau et al. 1998,2003a). The reason for the Golgi apparatus to be involved in transcytosis is an interesting fact. Passage through the Golgi apparatus during their transcytotic pathway across the enterocytes may well allow for some modifications and glycosylation of the transported enzymes, enabling them to gain entry into the blood circulation. Golgi involvement in this transcytosis remains, however, to be established.
During the in vivo experiments, either feeding or stimulation of pancreatic secretion, some enterocytes were labeled more intensely than neighboring ones, suggesting a possible heterogeneity in cellular capability for enzyme internalization. This heterogeneity may also be responsible for fluctuations in our quantitative results particularly evident for lipase over the endosomal compartment (Figure 9B). Previous studies on BSDL have identified a receptor, LOX-1, located at the ente-rocyte luminal membrane responsible for its internalization (Bruneau et al. 2003b). This finding entails internalization of BSDL as a rather specific event, and some cells may be more prone than others. Also, lipase internalization was found to be more efficient than that of amylase by morphological as well as by biochemical means.
Following the appearance in the extracellular space, amylase and lipase were found associated with the endothelial plasmalemmal vesicles and present within the capillary lumen. Even though presence of pancreatic enzymes in blood has been established for quite some time, their function and role in circulation is still poorly defined (Isenman et al. 1999). Studies have shown that the half-life of lipase in the blood varies from 6.9 to 13.7 hr and that of amylase is a little shorter (Tietz and Shuey 1993). The two enzymes appear to interact with circulating molecules. Amylase interacts with albumin (McGeachin and Lewis 1959; Dreiling et al. 1963) as well as with the γ-globulins (Ujihira et al. 1965), whereas lipase is known to interact with γ-globulins and to some extent to α-globulins (Tietz and Shuey 1993). Similarly, pancreatic BSDL was shown to interact with circulating lipoproteins, being mostly associated with chylomicrons, VLDL, and LDL (Bruneau et al. 2003a).
In conclusion, in a matter of minutes, lipase and amylase present in the intestinal lumen are transported by enterocytes from their apical to their basolateral poles. Both enzymes transit through endosomal compartments and Golgi apparatus before being transferred to the basolateral membrane where they are released into the interstitial space to reach the blood circulation. Furthermore, we noticed a relation between availability of those enzymes in the intestinal lumen and their concentrations in blood, suggesting that this pathway is physiologically relevant for the normal presence of those enzymes in circulation.
Footnotes
Acknowledgements
This work was supported by a grant from Canadian Institutes of Health Research.
The authors thank Dr. Irene Londoño, Elizabeth Gervais, and Denis Rodrigue for their assistance. This article represents part of the work required for the fulfillment of the M.Sc. programme of M.C.
