Abstract
The study of intermediate filament expression in the pancreatic epithelium has been previously focused almost exclusively on cytokeratins. Transient vimentin immunoreactivity has also been detected in duct cells of rat fetal pancreas. Here we report that, in rat pancreas, intense GFAP-like immunoreactivity is detectable in a subpopulation of endocrine cells located in the periphery of the islet of Langerhans. In addition, staining appeared to be preferentially localized to the apical pole of the cells. Two different polyclonal antibodies were employed in this study, with analogous results. Staining of consecutive sections with anti-GFAP, anti-glucagon, and anti-somatostatin antibodies demonstrates that GFAP-like immunoreactivity is present in glucagon-secreting cells. The relevance of this finding is discussed.
T
GFAP has been reported to be specifically expressed in mature astrocytes (Eng et al. 1971), but GFAP immunoreactivity has been also reported in enteric glia (Jessen et al. 1984), satellite cells of sympathetic and sensory ganglia (Jessen et al. 1984), and in non-myelin-forming Schwann cells (Yen and Fields 1981; Jessen et al. 1984; Fields and McMenamin 1985; Fields and Yen 1985). For this feature, GFAP has been also used as a marker of nerve fibers to follow their course and distribution (Krammer and Kühnel 1993). However, GFAP expression has also been reported in some cell types of mesenchymal origin scattered throughout different organs, such as perisinusoidal stellate cells from rat liver (Gard et al. 1985), Leydig cells from rat testis (Holash et al. 1993), chondrocytes from human elastic cartilage (Kepes and Perentes 1988) and, recently, periacinar stellate cells from rat pancreas (Apte et al. 1998). Here we report that GFAP-like immunoreactivity can be detected within islet cells as well. Further investigations enabled us to identify GFAP-immunoreactive cells as glucagonsecreting cells.
Materials and Methods
Antibodies
Two rabbit anti-GFAP polyclonal antibodies (PAbs) were used. One was purchased from Zymed Laboratories (San Francisco, CA). The other, from Sigma (St Louis, MO), was kindly provided by Dr. C. Nicoletti (Laboratory of Molecular Recognition, The Babraham Institute, Cambridge, UK). Rabbit anti-glucagon and anti-somatostatin PAbs were a generous gift from Dr. M. Bendayan (Department of Pathology and Cell Biology, University of Montreal, Montreal, PQ, Canada). Anti-vimentin MAb (clone V9) was kindly provided by Dr. C. Nicoletti. Alkaline phosphatase (AP)-conjugated anti-rabbit IgG was obtained from Roche Diagnostic (Monza, MI, Italy). AP-conjugated anti-mouse IgG was purchased from Sigma-Aldrich (Milan, Italy).
Animals and Tissue Processing
Four-month-old Wistar rats were used for this study. Rats were anesthesized with diethylether and decapitated. In some cases, the entire splenic portion of the pancreas was fixed in 50 ml of 10% formalin or Bouin's fixative overnight, for 2 days or for 5 days at room temperature (RT), and processed for routine embedding in paraffin. In other cases, the pancreas was trimmed in small blocks of ≈0.5 mm3, fixed for 3 hr at 4C in 5 ml of 4% paraformaldehyde dissolved in 0.1 M cacodylate buffer, and processed for ordinary embedding in Epoxy resin (Epon 812).
Immunocytochemistry
Deparaffinized sections (4-5 μm thick) were pretreated with 1% BSA in PBS for 30 min and then incubated with anti-GFAP PAb or anti-vimentin MAb (clone V9) for 4 hr at RT. After rinsing with 0.1% Triton X-100 in PBS, sections were treated with 1% BSA in PBS for 10 min and incubated with the appropriate AP-conjugated anti-rabbit or anti-mouse IgG. Slides were thoroughly rinsed with 0.1% Triton X-100 in PBS, and AP reaction was developed with NBT/BCIP stock solution (Roche Diagnostic) as chromogen diluted in 0.1 M Tris buffer, pH 9.5, 0.05 M MgCl2, and 0.1 M NaCl. Consecutive semithin sections (1 μm thick) from Eponembedded samples were also used. In this case, sections were mounted on well-degreased slides and Epon was removed according to the technique suggested by Maxwell (1978), with a solution of 2 g potassium hydroxide in 10 ml methyl alcohol and 5 ml propylene oxide for 5 min at RT. Slides were then rinsed twice in methanol, thoroughly washed in distilled water, and placed in TBS. Series of three sections were blocked with 1% BSA in TBS and subsequently incubated with the following antibodies: Section 1 with rabbit anti-glucagon PAb, Section 2 with rabbit anti-GFAP PAb, and Section 3 with rabbit anti-somatostatin PAb. Other experiments were conducted with anti-vimentin MAb. After rinsing with 0.1% Triton X-100 in modified TBS (0.05 M Tris, 150 mM NaCl, 20 mM MgCl2, 1% BSA), sections were treated with 1% BSA in TBS and then incubated with AP-conjugated anti-rabbit or anti-mouse IgG, depending on the primary antibody. Then slides were washed with 0.1% Triton X-100 in modified TBS and the AP reaction was developed with NBT/BCIP stock solution as chromogen diluted in 0.1 M Tris buffer, pH 9.5, 0.05 M MgCl2, and 0.1 M NaCl. Control experiments consisted of: omission of the primary antibodies or their replacement with rabbit or mouse nonimmune serum; anti-GFAP PAb liquid-phase absorption with glucagon (Sigma-Aldrich) (100 μg/ml); or anti-GFAP PAb liquid-phase absorption with poly-
Results
GFAP-immunoreactive cells were detected in rat pancreas as Schwann cells of nerve fibers. In the islets of Langerhans, however, strong staining with anti-GFAP Abs was obtained in peripheral islet cells regardless of the type of antibody employed (Figure 1). Moreover, observation of these cells at higher magnification allowed recognition of an asymmetrical distribution of staining, which was stronger at the apical (perivascular) pole of the cells (Figure 2). The intensity of the staining was dependent on fixation time regardless of the fixative employed: the longer the fixation, the weaker the reaction. Fixation for as long as 2 days abolished immunoreactivity.
To determine precisely the type of cell responsible for islet GFAP immunoreactivity, consecutive semithin sections were stained with anti-glucagon, anti-GFAP, and anti-somatostatin PAbs. By this procedure, it was evident that somatostatin-secreting cells did not stain with anti-GFAP Abs, whereas all glucagon-secreting cells expressed GFAP-like immunoreactivity as well (Figure 3). On the other hand, a limited number of cells expressing GFAP-like immunoreactivity scattered throughout the exocrine tissue were also identified as glucagon-secreting cells (Figure 4). All GFAP-immunoreactive cells detected in our series of experiments therefore appeared to be glucagon cells. Omission of primary antibodies or replacement with rabbit nonimmune serum abolished all the reactions. On the other hand, staining was not impaired on anti-GFAP Ab absorption with glucagon or with poly-
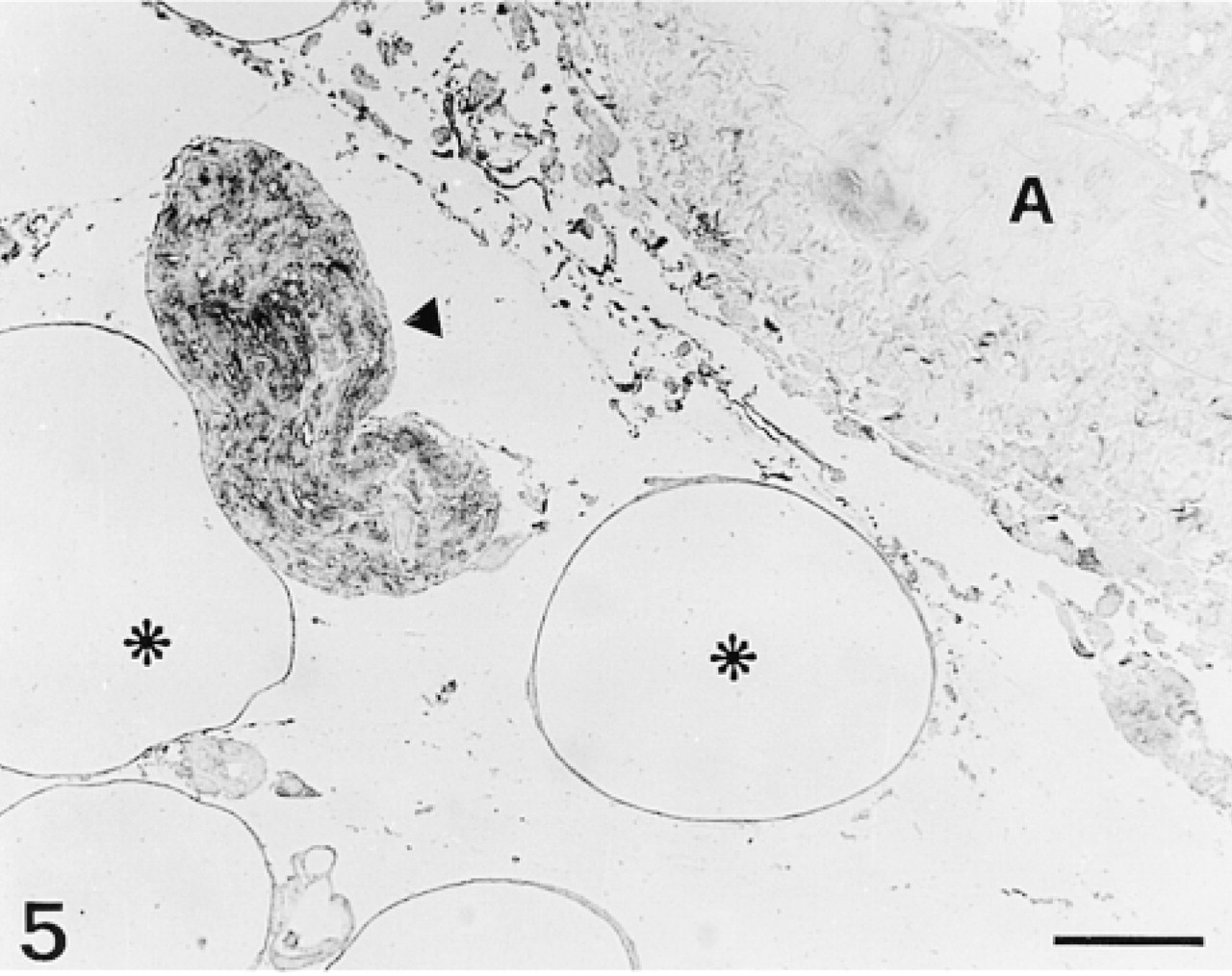
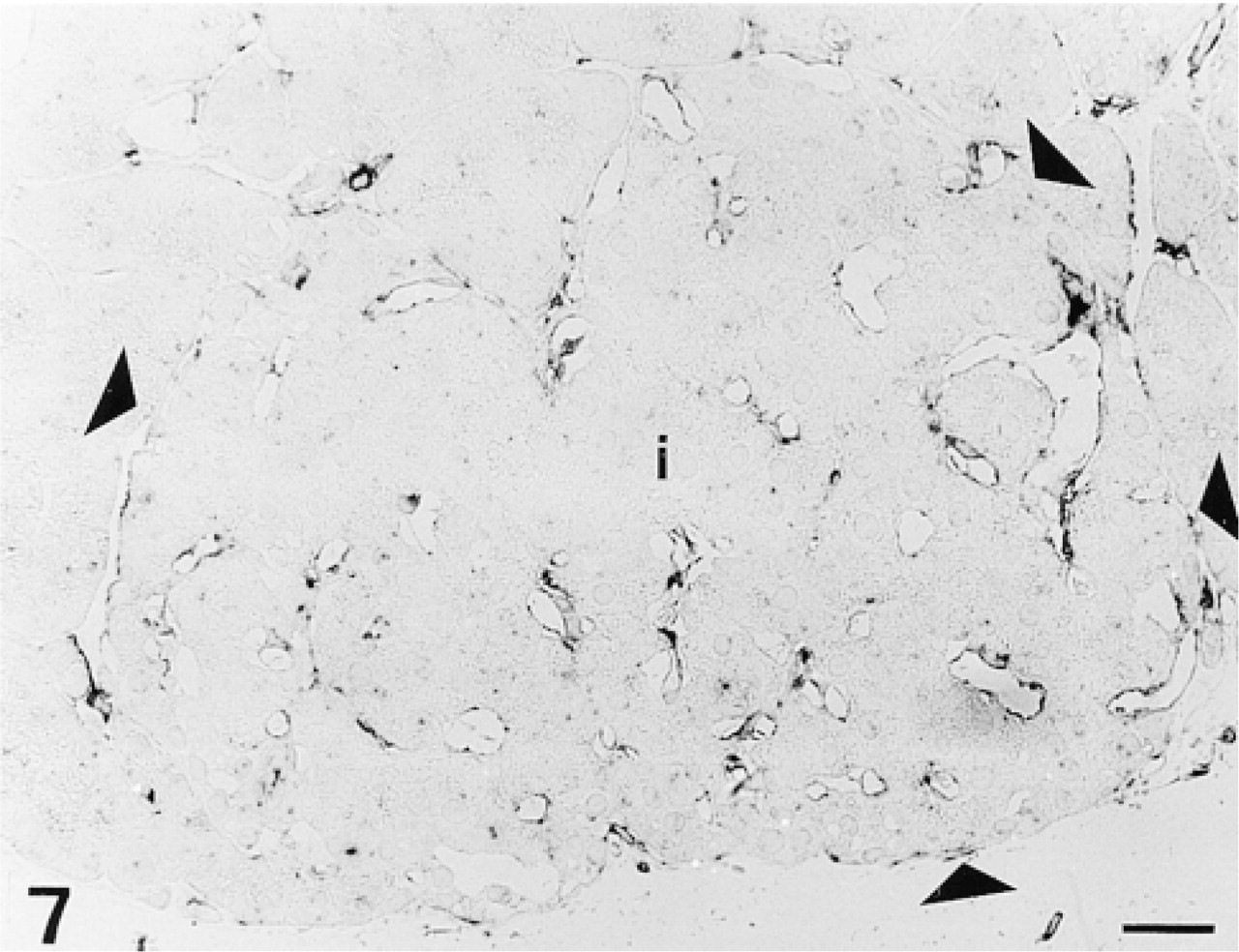
None of the anti-GFAP Abs employed stained blood vessels (Figure 5) either in endothelial or in muscle cell components. We also performed a series of positive controls with sections of rat brain tissue incubated with anti-GFAP Abs that stained exclusively glial cells (Figure 6). Incubation of sections with anti-vimentin MAb resulted in a completely different pattern of staining, with no labeling of islet cells but with strong staining of endothelial cells lining intra-islet blood capillaries (Figure 7).
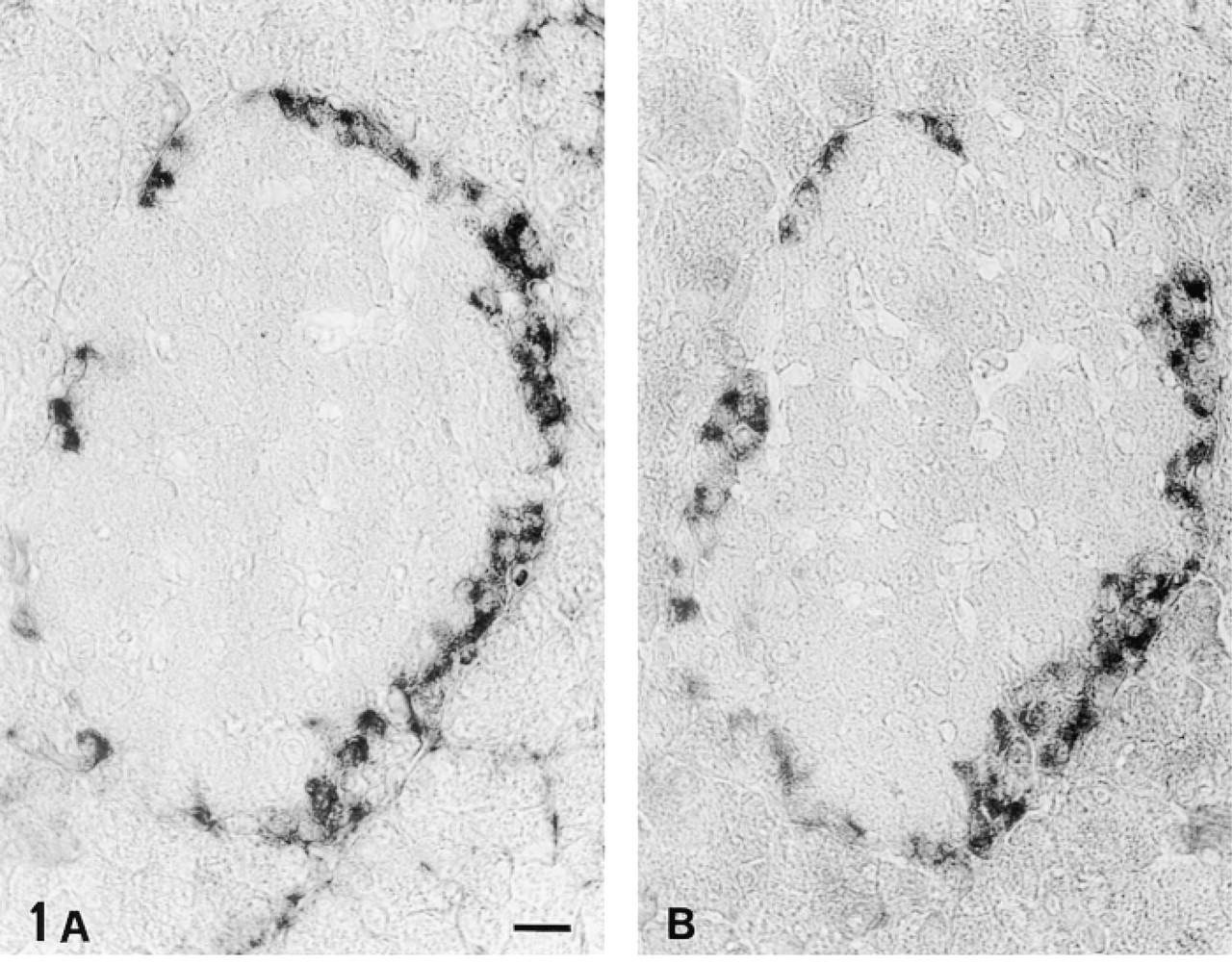
Two pancreatic sections passing through the same islet of Langerhans. Incubation with different anti-GFAP PAbs results in the same pattern of staining with several peripheral islet cells intensely labeled. (
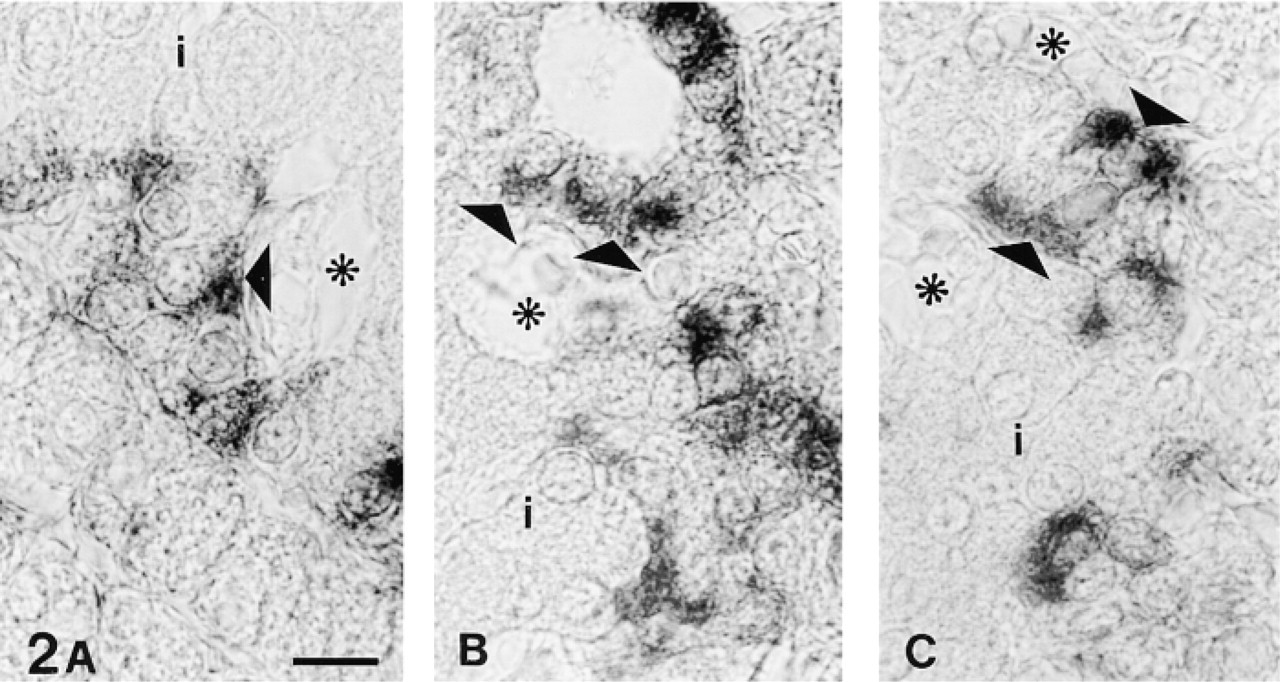
(A-C) High magnification of some GFAP-like immunoreactive islet cells. Staining appears to gather on the apical (perivascular) pole of the cells (triangles). Asterisks, intraislet blood capillaries; i, islet tissue. Fixation in Bouin's fluid; embedding in paraffin. Bar = 10 μm.
Discussion
Our immunocytochemical findings provide evidence that pancreatic glucagon-secreting cells exhibit GFAP-like immunoreactive antigens. Eventual crossreactions of anti-GFAP antibodies employed in the present study with other known IFs appear to be unlikely on the basis of the following considerations. The pattern of staining obtained in the pancreas and in other tissues does not match that expected if antibodies recognized desmin (unstained muscle cells), neurofilaments (unstained neurons), or CKs (no known CK has been reported to be expressed exclusively in a single islet cell type; for a review on CK expression in pancreas see Bouwens 1998). Regarding vimentin, we used a specific anti-vimentin MAb to exclude its presence in the endocrine pancreas and, accordingly, we did not detect any signal in pancreatic epithelial cells. Moreover, we obtained consistent results with two different anti-GFAP PAbs raised using different sources of antigen (human brain and bovine spinal cord). Nonspecific binding to basic peptides, occurring in A-cells as well (Buffa et al. 1979), was excluded by liquid-phase absorption experiments with poly-
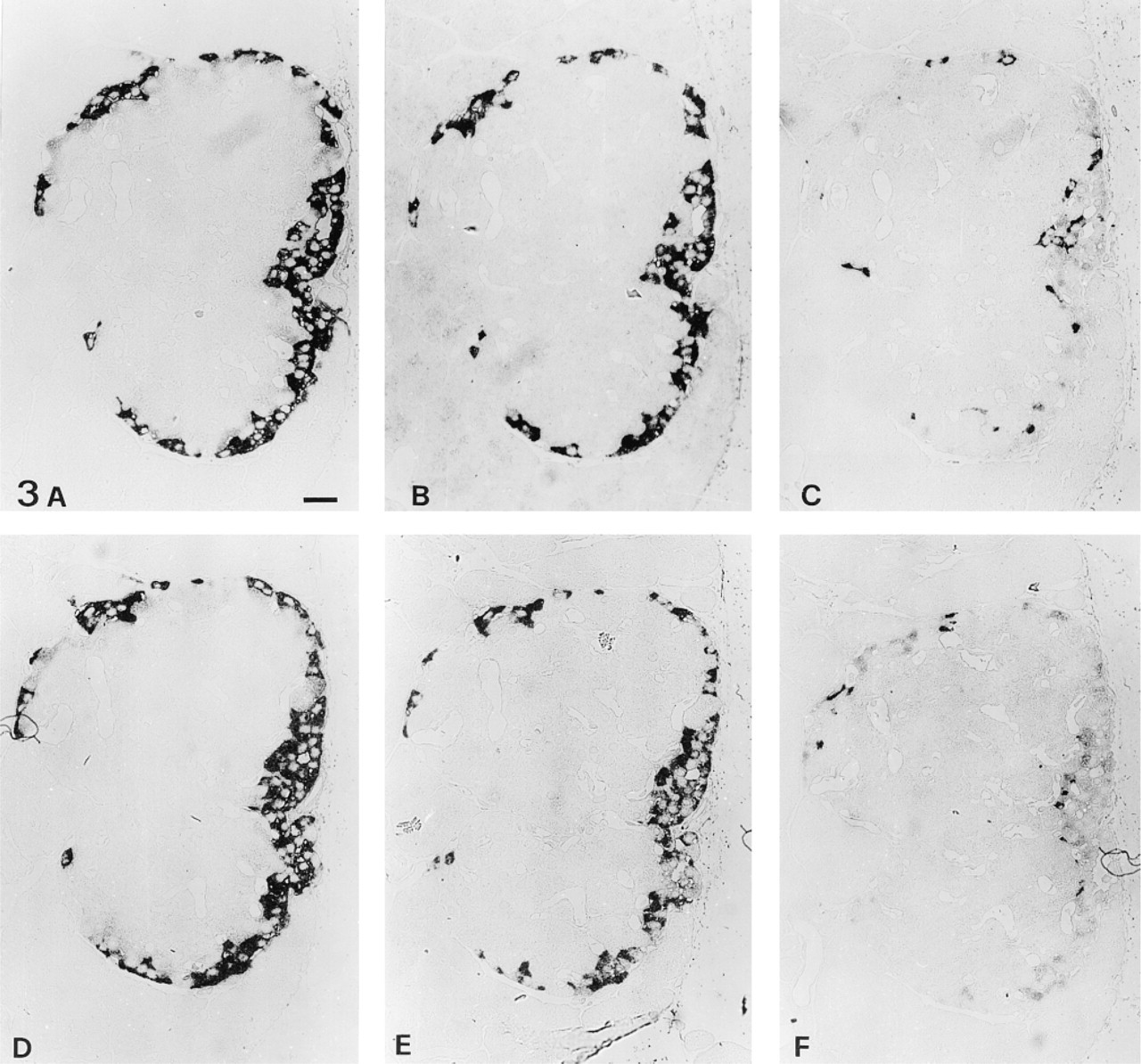
Two series (
Even though we can not completely exclude the detection of a peptide not strictly related to GFAP, we can affirm that GFAP-like immunoreactivity in these conditions should reveal actual GFAP (GFAPα) or a protein derived from an alternative splicing of GFAP mRNA. In this regard, in addition to GFAPβ located prevalently in the peripheral nervous system (Feinstein et al. 1992), an alternatively spliced GFAP mRNA (GFAPγ), lacking the entire exon 1, has been reported to be expressed in mouse bone marrow and spleen (Zelenika et al. 1995).
The presence of GFAP in the endocrine pancreas could prompt reconsideration in a new light of the theory of a neural crest origin of endocrine cells (Pearse 1977, 1982). A similar finding in other types of cells has previously induced other investigators to speculate on an ectodermal origin of Leydig cells (Holash et al. 1993) and folliculostellate cells in the adenohypophysis (Velasco et al. 1982). Moreover, even though restricted to glucagon-secreting elements, GFAP expression should be added to an already long list of markers shared by the endocrine and nervous systems (Le Douarin 1988; Teitelman 1991; Larsson 1998). In particular, glucagon cells constantly or transiently express molecules that have been also reported in the nervous system, such as glucagon-like peptide-1 and glucagon-like peptide-2 (Eissele et al. 1994; Larsen et al. 1997), tyrosine hydroxylase (Alpert et al. 1988), neuron-specific enolase (Lloyd et al. 1984), the 67-kD isoform of the glutamic acid decarboxylase (Li et al. 1995), chromogranins (Schmid et al. 1994), synaptophysin (Bouwens et al. 1997), insulin (Larsson 1998), and PYY (Larsson 1998). However, we emphasize that by the use of several different technical approaches, such as the removal of the entire ectoderm from embryonic rats (Pictet et al. 1976) or the generation of chimeric chick-quail embryos (Andrew 1976; Fontaine and Le Douarin 1977), the hypothesis that pancreatic endocrine precursors reside in the neural crest appears less likely (Le Douarin 1988). It is therefore generally accepted that endocrine and exocrine pancreatic epithelium derives from the epithelial lining of the embryonic gut, which is endodermal in origin (Le Douarin 1988). After the formation of a diverticulum from the primitive gut, a branched tree of ductules of protodifferentiated cells forms and gives rise to acinar and endocrine cells (Pictet and Rutter 1972; Pictet et al. 1972). The common embryological derivation of both the exocrine and endocrine components of pancreas explains how they share the same classes of IF proteins, i.e., Type I and Type II CKs (Bouwens 1998). Endocrine cells, however, display a slightly different pattern of CK expression. In addition to CK8 and CK18, which are found in all pancreatic epithelia (Bouwens 1998), rat islet cells have been shown to be CK7-immunoreactive as well (Bouwens et al. 1994). Thus far, other classes of IFs have not been reported in pancreatic epithelial cells, with the remarkable exception of a transient vimentin immunoreactivity detected in the fetal rat pancreatic ducts (Bouwens and De Blay 1996). GFAP immunoreactivity in adult rat glucagon-secreting cells therefore represents the first demonstration of the stable expression of an IF other than CKs in pancreatic epithelial cells. Moreover, the restriction of its pattern of expression to A-cells, which are the first endocrine cells immunocytochemically detectable in rat pancreas (Ghitens 1993), makes GFAP particularly interesting in view of its possible use as a differentiation marker. However, its validity for this purpose has to be assessed by determining whether its expression precedes glucagon synthesis, thereby identifying A-cell precursors, is simultaneous with A-cell differentiation, or follows glucagon synthesis in A-cells.
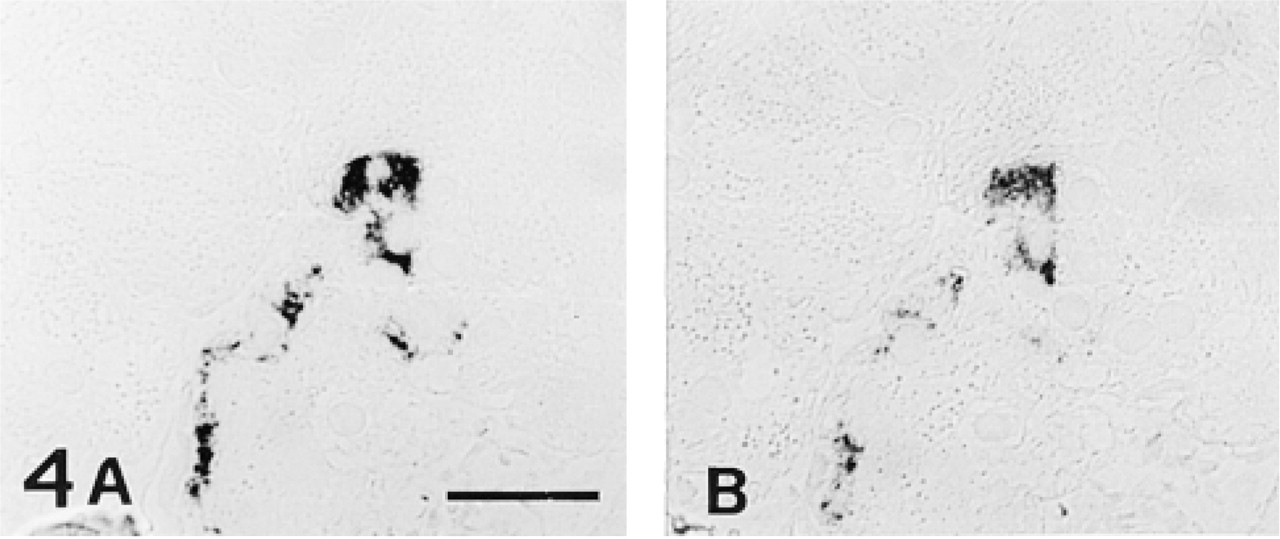
Consecutive semithin sections stained with anti-glucagon (
Semithin section stained with anti-GFAP PAb. A large bundle of nerve fibers is stained (triangle), whereas a large artery (
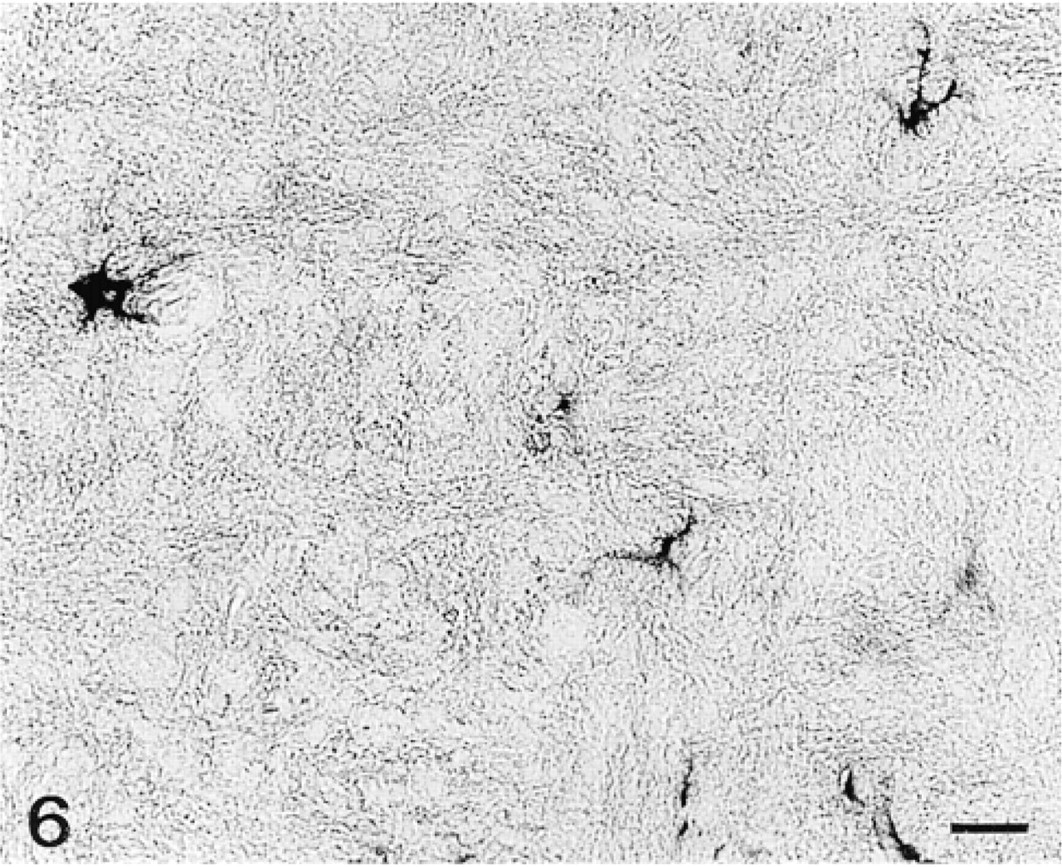
Rat brain section stained with anti-GFAP PAb. The antibody stains exclusively some astrocytes. Fixation in 10% formalin; embedding in paraffin. Bar = 20 μm.
Semithin section of an islet of Langerhans (
The exact role of GFAP in glucagon-secreting cells is not clear at the moment. GFAP expression has been linked to the stellate morphology of some cell types including astrocytes, periacinar stellate cells, pituitary folliculostellate cells, and perisinusoidal stellate cells (Velasco et al. 1982; Gard et al. 1985; Weinstein et al. 1991; Apte et al. 1998), but recent studies with GFAP-deficient astrocytes have demonstrated that, even without GFAP, such astrocytes can extend their processes (Pekny et al. 1998), and pancreatic glucagon-secreting cells, as well as chondrocytes and lens epithelial cells, display a very regular shape even though expressing GFAP. It has also been suggested that GFAP may be implicated in a presumptive regulatory role of capillary permeability (Buniatian et al. 1996, 1998). This hypothesis has been formulated because of its expression in astrocytes and Leydig cells, both types of cells being involved in the formation of biological barriers between blood and tissue (blood-brain barrier and blood-testis barrier). Speculating in this direction, in spite of the absence of an analogous barrier between sinusoids and hepatocytes, perisinusoidal stellate cells have also been proposed to be implicated in such a regulatory role (Buniatian et al. 1996, 1998). However, the presence of GFAP cannot be linked to this type of function in cells located in avascular tissues, such as chondrocytes and lens epithelial cells, nor can it be linked to this type of function in pancreatic A-cells, even though they face blood capillaries, because they are not interposed between blood vessels and other parenchymal cells.
A variety of evidence suggests that IFs confer on cells higher resistance to mechanical stress. This has been clearly demonstrated for CKs in epidermal cells and for desmin in muscle cells (Houseweart and Cleveland 1998). However, it is difficult to envision the same role for GFAP in glucagon-secreting cells, which do not appear to be subjected to particular mechanical stresses and which are already provided with a network of CKs. Certainly, it is difficult to predict exactly which types of external forces may act on a single cell type. Therefore, we might even suppose that glucagon-secreting cells may require structural reinforcement by a GFAP network, but it is difficult to hypothesize a reinforcement role that is not required by somatostatin-secreting cells as well, which are GFAP-negative and are apparently in the same situation.
To date, therefore, there is no hypothesis on GFAP function that can explain its presence in all GFAP-immunoreactive cells. It is possible, however, that GFAP may serve several different functions according to the cell type. In this regard, the finding that GFAP immunoreactivity appears to be restricted to the secretory pole of A-cells raises the possibility that in this type of cell GFAP is involved in the secretory machinery.
Footnotes
Acknowledgment
This work is dedicated to the memory of Prof Leonetto Comparini.
