Abstract
A single primary cilium is found in chondrocytes and other connective tissue cells. We have previously shown that extracellular matrix (ECM) macromolecules such as collagen fibers closely associate with chondrocyte primary cilia, and their points of contact are characterized by electron-opaque plaques suggesting a direct link between the ECM and the cilium. This study examines the expression of receptors for ECM molecules on chondrocyte primary cilia. Embryonic chick sterna were fluorescently labeled with antibodies against α and β integrins, NG2, CD44, and annexin V. Primary cilia were labeled using acetylated α-tubulin antibody. Expression of ECM receptors was examined on chondrocyte plasma membranes and their primary cilia using immunofluorescence and confocal microscopy. All receptors examined showed a punctate distribution on the plasma membrane. α2, α3, and β1 integrins and NG2 were also present on primary cilia, whereas annexin V and CD44 were excluded. The number of receptor-positive cilia varied from 8/50 for NG2 to 43/50 for β1 integrin. This is the first study to demonstrate the expression of integrins and NG2 on chondrocyte primary cilia. The data strongly suggest that chondrocyte primary cilia have the necessary machinery to act as mechanosensors, linking the ECM to cytoplasmic organelles responsible for matrix production and secretion.
T
Intracellular calcium signaling is also initiated in chondrocytes subjected to dynamic mechanical load, but the transductory mechanisms are not fully understood (Roberts et al. 2001; Pingguan-Murphy et al. 2005). The interaction between chondrocytes and the extracellular matrix (ECM) that surrounds them is fundamental for normal development and for the maintenance of tissue architecture and function. Mechanical loading of the ECM results in the activation of mechanotransduction signaling pathways which, in turn, affects the metabolic activity of the chondrocytes (Muir et al. 1970; Hardingham and Fosang 1992; Lee et al. 1998,2000; Salter et al. 2001; Deschner et al. 2003; Chowdhury et al. 2004; Millward-Sadler and Salter 2004). In embryonic chick hyaline cartilage in situ, the chondrocyte ciliary axoneme projects into the ECM in a range of bent configurations, in contrast to the straight ciliary configuration observed in isolated chondrocytes (Poole et al. 1985,2001; Jensen et al. 2004). Our previous studies have led us to propose that deflection of the primary cilium by forces transmitted through the ECM allows it to act as a mechanosensor, detecting mechanical and physicochemical changes within its pericellular environment and transducing signals to the cell (Poole et al. 1997,2001; Jensen et al. 2004). It is well established that the activation of mechanotransduction signaling pathways is transmitted via cell surface receptors (Gray et al. 1988; Roberts et al. 2001; Chowdhury et al. 2004; Lucchinetti et al. 2004; Millward-Sadler and Salter 2004). Using electron tomography, we have demonstrated close associations between the ciliary membrane and the ECM (Jensen et al. 2004). These points of contact between the fibers of the ECM and the membrane are characterized by electron-opaque plaques, which may represent receptors for the collagens and proteoglycan aggregates of the ECM.
Chondrocytes express several classes of transmembrane ECM receptors. The most well-characterized of these are the integrins, which are heterodimeric transmembrane receptors that bind to many cartilage ECM molecules, including collagens, fibronectin, and laminin (Hynes 1992; Loeser 1993,2000; Loeser et al. 1995). Intracellularly, these receptors complex with the cortical actin cytoskeleton, activating intracellular signaling cascades and regulating gene expression of matrix molecules (Hynes 1992). NG2 is a transmembrane proteoglycan that contains chondroitin sulfate side chains, is expressed in developing and adult cartilage (Nishiyama et al. 1991; Midwood and Salter 1998,2001), and is a putative ligand for collagen types V and VI (Burg et al. 1996; Tillet et al. 1997,2002). This receptor plays a role in signal transduction, regulation of cell proliferation, adhesion, and cell spreading (Burg et al. 1996; Fukushi et al. 2004). Annexin V (anchorin CII) is a member of the large annexin family of proteins defined by their ability to bind phospholipids in a calcium-dependent manner and has been identified on the surface of chondrocytes (Mollenhauer et al. 1984,1999). A suggested role of annexin V within cartilage is binding of chondrocytes to type II collagen (von der Mark and Mollenhauer 1997; Lucic et al. 2003). CD44 is a cell surface glycoprotein that binds hyaluronan and is expressed in a variety of cell types including hyaline chondrocytes (Knudson 1993; Knudson et al. 1996; Jiang et al. 2002). Hyaluronan-CD44 binding retains proteoglycan aggregates in the chondrocyte pericellular matrix, and CD44 also functions to regulate matrix assembly and retention (Chow et al. 1995; Jiang et al. 2002).
In the current study we have examined the specific expression of ECM receptors and their ligands in relation to the primary cilia of embryonic chick hyaline chondrocytes in situ, using immunofluorescent labeling and confocal microscopy. We present novel data showing the expression of receptors for ECM molecules on the chondrocyte primary cilium, strongly suggesting that the primary cilium has the molecular machinery necessary to act as a mechanosensory organelle.
Materials and Methods
Tissue Preparation
Fertile Shaver Brown eggs (Bromley Hatcheries; Tuakau, New Zealand) were sacrificed under approved ethical conditions between day 16 and day 17 following incubation at 39C. Fifty sterna were dissected free, the perichondrium was removed, sterna were divided into caudal and cephalic regions, and the cephalic regions were discarded. The caudal regions of sterna were either snap frozen in liquid nitrogen or fixed for 1 hr in 4% paraformaldehyde in 0.1 M PBS maintained at 37C.
Immunoblotting
Frozen sterna were rendered to a powder using a mortar and pestle in liquid nitrogen. The powder was extracted using RIPA buffer (1% NP-40, 0.5% sodium deoxycholate, 0.1% SDS in 50 mM Tris-HCl buffer, pH 8) containing protease inhibitors (10 mg/ml leupetin, 1 mM phenylmethylsulfonyl fluoride, and 2 mM sodium orthovanadate; Roche, Auckland, New Zealand) for 3 hr at 4C. Protein extract was centrifuged at 12,000 × g for 20 min at 4C. The supernatant was removed, and total protein was measured using the BioRad DC Protein Assay (BioRad; Auckland, New Zealand). Supernatants were heated to 70C for 10 min, and equal amounts of protein (50 μg) were separated on 4-12% SDS polyacrylamide gels (Invitrogen; Auckland, New Zealand) at 200 V for 35 min. Proteins were subsequently transferred onto PVDF membranes (Hybond-P; Amersham Biosciences, Auckland, New Zealand) at 30 V for up to 90 min. Membranes were blocked with 5% non-fat milk powder in Tris-buffered saline containing 1% Tween-20 (TBS-T) for 1 hr at room temperature (RT) and incubated overnight at 4C with the appropriate primary antibody. Membranes were washed in TBS-T and incubated with species-specific horseradish peroxidase secondary antibody (1:2000; Chemicon, Auckland, New Zealand) for 2 hr at RT. Peroxidase activity was visualized using enhanced chemiluminescence (ECL; Amersham Biosciences) according to the manufacturer's instructions. Prestained protein standards and MagicMarker molecular mass standards (Invitrogen) were used to determine the apparent molecular masses of the blotted proteins.
Immunohistochemical Labeling
Sterna were embedded in optimal cutting temperature compound and frozen in the presence of methyl-butane, and 16-μm-thick cryosections were cut and air dried onto Superfrost Plus slides (Menzel Glazer; Braunschweig, Germany). Sections were predigested with testicular hyaluronidase (300 U/ml in 0.1 M Tris-buffered saline; pH 5.5) for 2 hr at RT to remove matrix proteoglycans and allow antibody penetration. Sections were subsequently permeabilized with 0.5% (v/v) Triton X-100 (Serva; Heidelberg, Germany) for 5 min at RT. Nonspecific binding was prevented by incubation with 2% (v/v) goat serum (Life Technologies; Auckland, New Zealand) for 30 min at RT. Primary antibodies (see Table 1) were diluted in PBS + 0.1% (w/v) BSA (Life Technologies) and added to the sections that were left overnight at 4C. Sections were incubated with the appropriate fluorescently conjugated secondary antibody—either goat anti-mouse or goat anti-rabbit FITC (1:200; Sigma-Aldrich, Auckland, New Zealand), diluted in PBS + 0.1% BSA, incubated at RT for 2 hr, and rinsed and mounted in Citifluor (Citifluor Ltd.; London, UK). For double-labeling experiments, sections were fixed in 4% paraformaldehyde for 5 min and nonspecific binding was blocked using 2% goat serum. The second primary antibody was added to the sections and incubated overnight at 4C. Sections were subsequently incubated with the appropriate secondary antibody—either goat anti-mouse or goat anti-rabbit Cy3 (1:500; Jackson Immuno-Research Laboratories, West Grove, PA) for 2 hr at RT, rinsed, and mounted in Citifluor. In some experiments, the nucleus was also labeled with Hoechst 33258 (100 nM; Sigma-Aldrich) for 15 min at RT. For the labeling of ECM molecules, sections were not permeabilized with Triton X-100 prior to the addition of the primary antibody. For negative controls, sections were incubated with PBS + 0.1% BSA instead of the primary antibody. In addition, to test the specificity of integrin labeling, integrin antibodies were preadsorbed with active RGD-peptide (GRGDSP; 500 mg/ml, Calbiochem, Nottingham, UK) overnight at 4C prior to immunolabeling.
Summary of antibodies used in this study
Confocal Microscopy and Image Processing
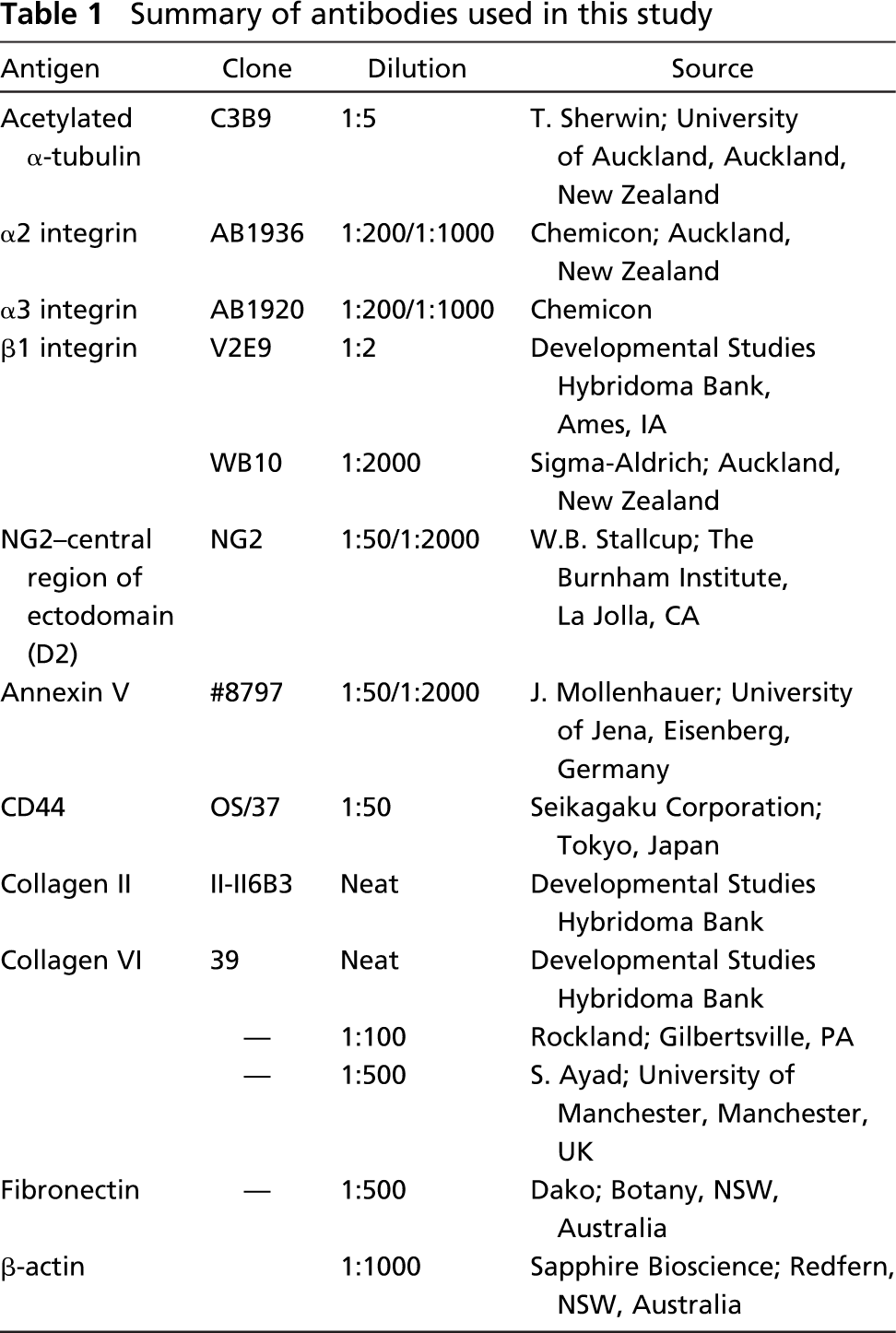
Sections were imaged using a Leica SP2 confocal laser scanning microscope (Leica; Heidelberg, Germany) and a X100 oil-immersion lens. At least 10 random fields of view per sternum from 5 to 10 chicks were examined for each labeling (see Figure 1). Single confocal images and z-axis series (z-section ~0.5-μm thick) were collected at a range of electronic zooms up to 6X. Negative control sections were imaged with identical settings to those used to image experimental sections. Gain and offset settings were often higher for imaging of receptor expression on the primary cilium compared with expression on the cell membrane. This was due to the relatively small diameter of the cilium and the maximum signal that could be obtained from its small volume. Because optimal imaging of cilia receptor expression often resulted in saturated images of plasma membrane receptor expression, some images of cilia receptor expression are presented separately. Emissions from multiple-labeled sections were captured separately and overlaid using software associated with the microscope (AnalySiS, Leica). All images were processed using Adobe Photoshop 7.0 (Adobe; San Jose, CA).
To examine ECM receptor expression on the primary cilium, only cilia that were oriented at right angles (i.e., orthogonal) to the incident light path (i.e., within the focal plane) were selected for imaging (Figure 1). This was to ensure that the full length of the cilium was imaged and that receptor-labeling signal with the ciliary membrane was not superimposed from the signal from the plasma membrane. Therefore, although cilia could be detected in at least 50% of cells in our preparations, our strict criteria resulted in selection of only a few chondrocytes per field of view that were suitable for quantification (see Figure 1).
Results
Integrins and NG2 Are Expressed on the Chondrocyte Primary Cilium
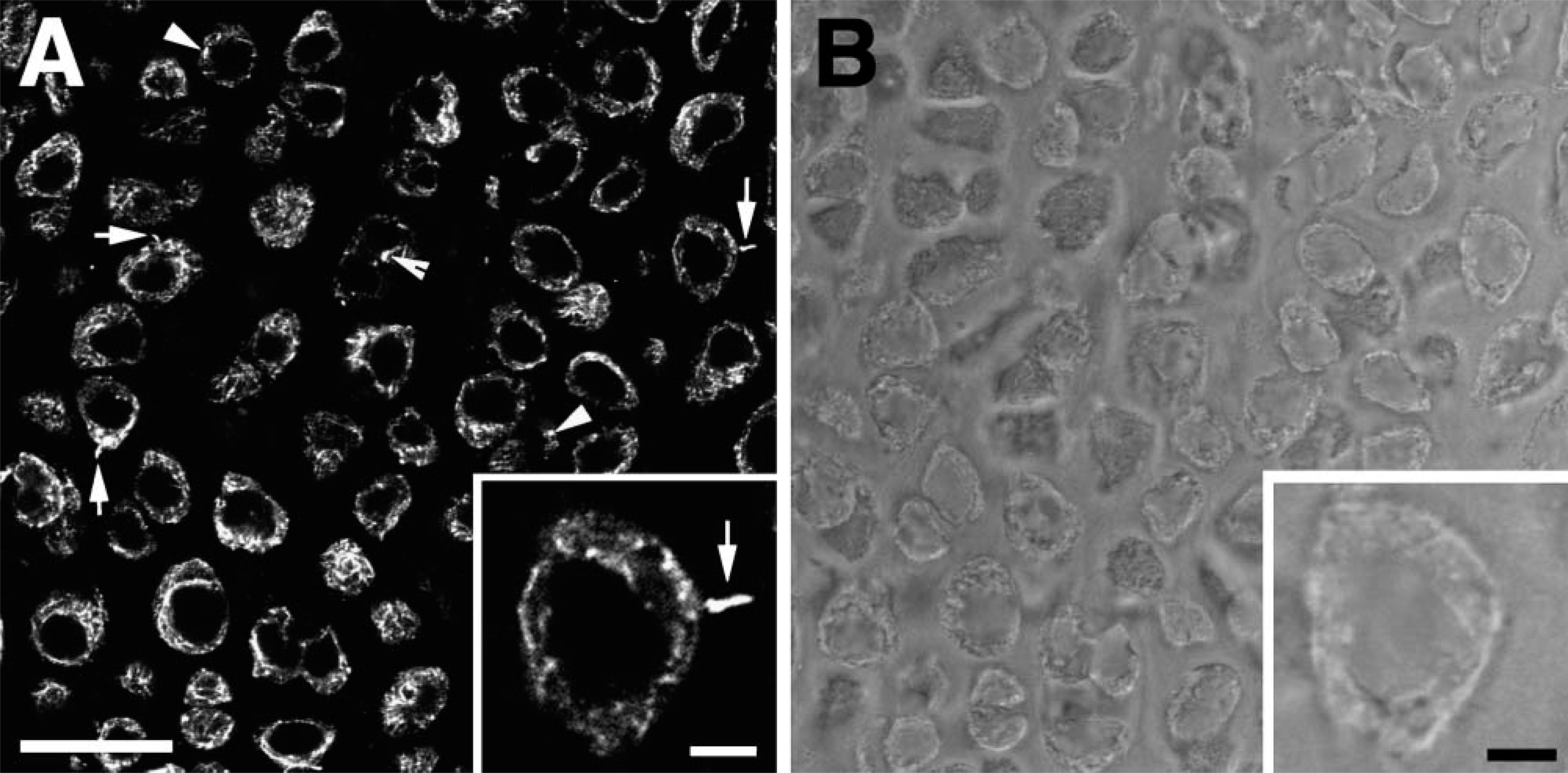
Integrins α2, α3, and β1 were present on the cell membrane as small plaques and showed a punctate distribution within the cytoplasm (Figures 2A-2C). There was no evidence of nuclear labeling with any of the three integrin antibodies. All three integrins localized as distinct puncta along the length of the primary cilium (see Figures 2D-2F). However, not all cilia examined were positive for integrin labeling. Fifty cilia were examined for each receptor protein, and the number of integrin-positive cilia ranged from 17 for α2 to 43 for β1 (see Table 2). Fibronectin, a ligand for several integrins, was present in the ECM but showed no specific labeling on the cilium or within the cell (Figure 2A). Following incubation of the anti-integrin antibodies with active RGD peptides, there was no positive staining of integrins on or within either the cell or the cilium (Figure 2G). Negative controls for each antibody showed no positive labeling (Figure 2H).
Antibody specificity was tested by Western blotting. Anti-integrin antibodies recognized proteins of the expected size for α2 (160 kDa) and β1 integrin (120 kDa) (Figure 2I). Anti-α3 integrin antibody has been raised against the cytoplasmic domain of the receptor and has reported specificity in chicken (Chemicon; Auckland, New Zealand). Western blotting showed two specific bands at ~55 and 70kDa, which are likely to represent glycosylated and unglycosylated protein (de Melker et al. 1997; DiPersio et al. 2001). A faint band was also present at 25 kDa, which represents the light chain of the reduced α3A subunit, a splice variant of the cytoplasmic domain reported in chicken (DiPersio et al. 2001). No band was present at the predicted mass (under reducing conditions) of 130 kDa.
Distribution of primary cilia in chick hyaline cartilage. (
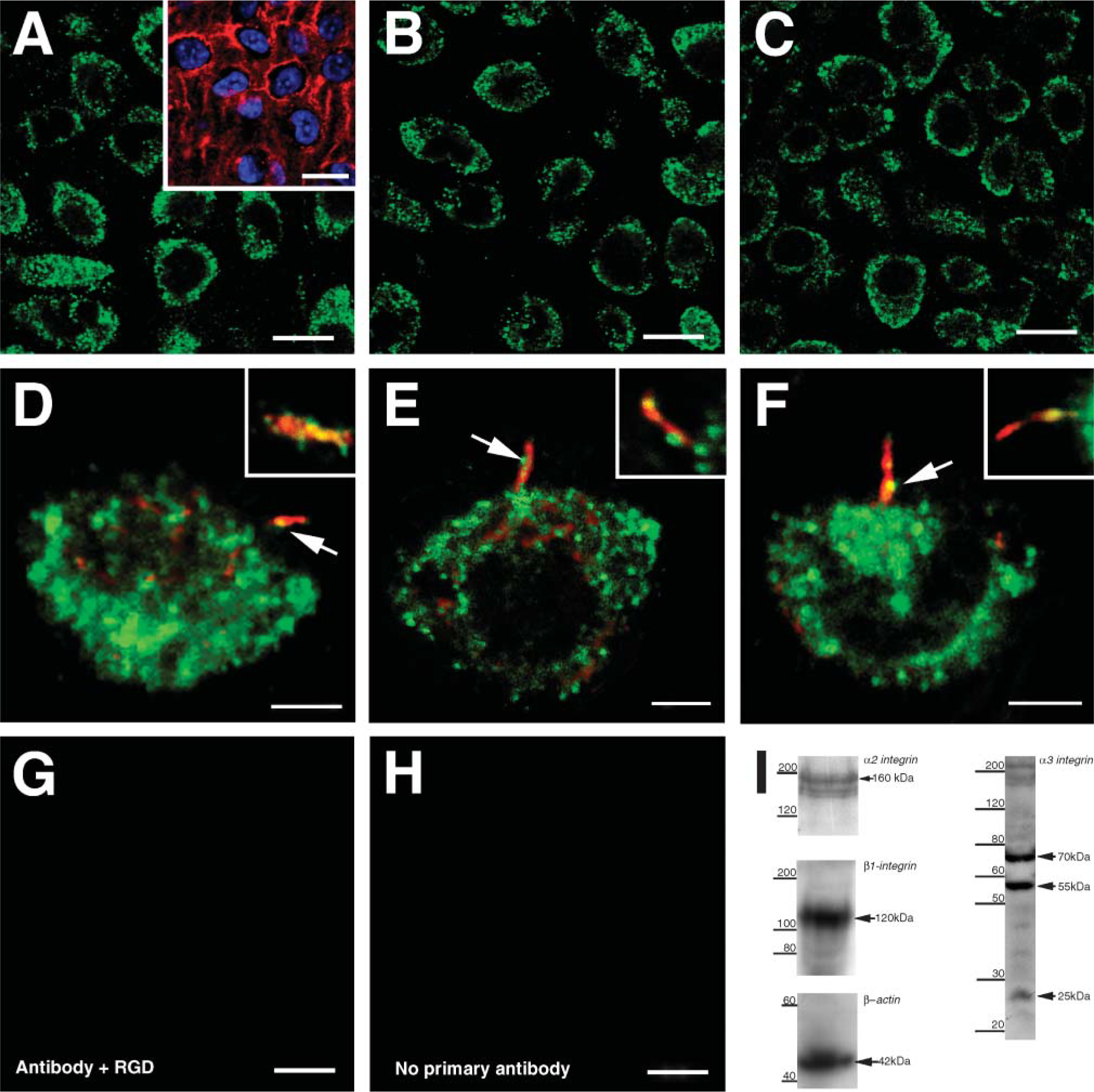
NG2 antibody showed a punctate distribution on the plasma membrane and occasionally in the cytoplasm (Figure 3A). In the majority of cells examined, NG2 was not detected along the primary cilium (Figure 3B). However, in a small number of cells (8/50) (Table 2), NG2 was present on the cilium, appearing as puncta (Figures 3C and 3D). No labeling was observed in negative control sections (Figure 3E). Specificity of the NG2 antibody was tested by immunoblotting and a specific band was observed at 270 kDa (Figure 3F). Although several antibodies against mammalian type VI collagen, a putative ligand for NG2, were tested on both sternal and articular cartilage from chick (see Table 1), none showed consistently positive labeling. No antibodies against chick type VI collagen were available.
Annexin V and CD44 Are Excluded from the Chondrocyte Primary Cilium
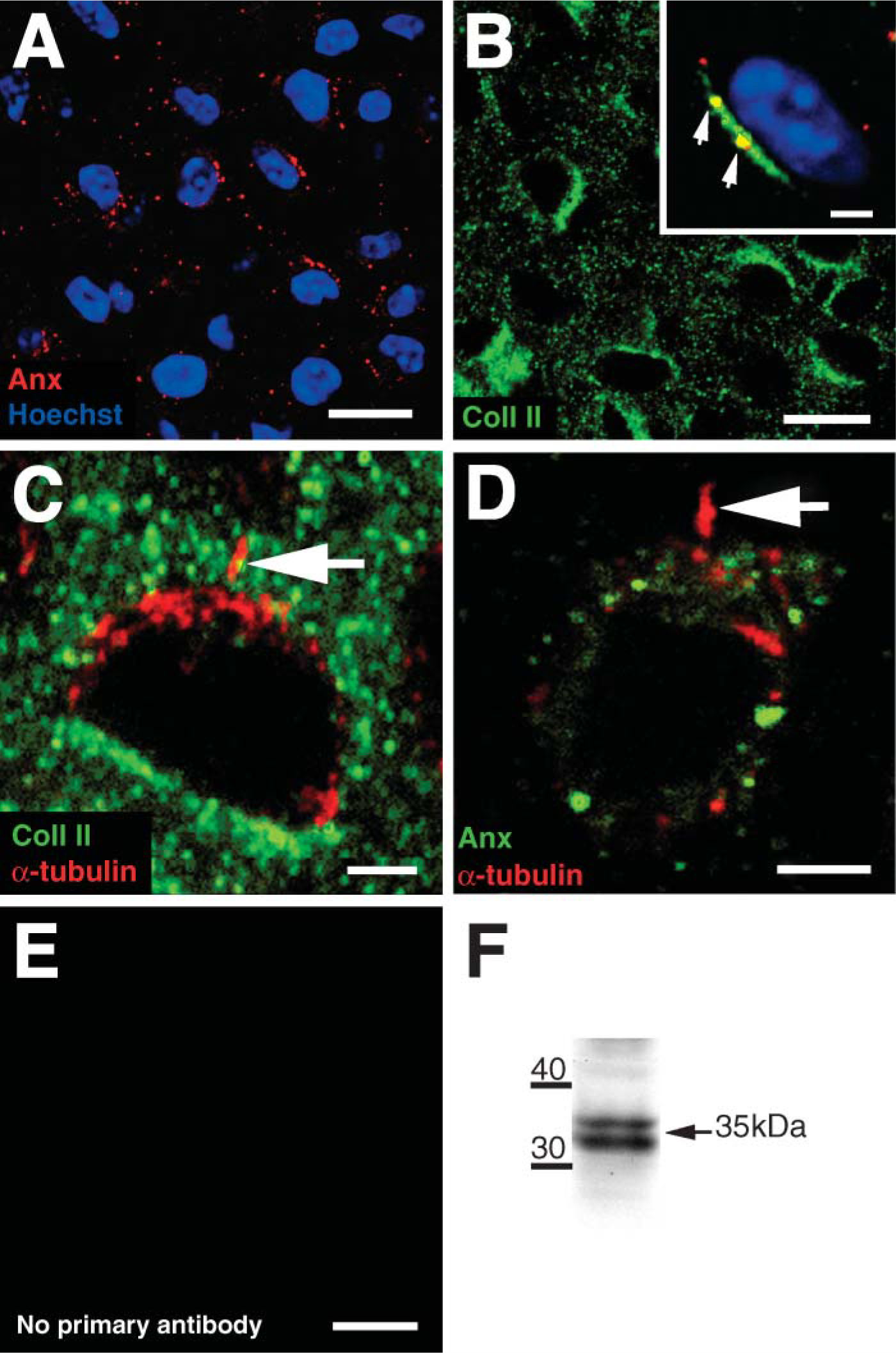
Annexin V was expressed on the surface of all chondrocytes as large plaques, with occasional labeling also present within the ECM (Figure 4A). Type II collagen, a putative ligand of annexin V, was present throughout the ECM (Figure 4B) and colocalized with annexin V puncta (Figure 4B). Type II collagen was also concentrated pericellularly and completely surrounded the primary cilium (Figure 4C). However, there was no evidence of annexin V expression on the primary cilium itself (Figure 4D; Table 2). No labeling was observed in negative control sections (Figure 4E). Western blotting using anti-annexin V antibody revealed a specific band at 35 kDa (Figure 4F).
CD44 was expressed as large plaques on the plasma membrane and in the cytoplasm of all chondrocytes, but there was no evidence of its expression on the primary cilium (data not shown). No labeling was observed in negative control sections.
Discussion
This study demonstrates for the first time that α2, α3, and β1 integrins and NG2 are present on the chondrocyte primary cilium in situ. We have also shown that two matrix receptors, CD44 and annexin V, are excluded from the primary cilium. These results strengthen our previous findings that ECM macromolecules make contact with the ciliary membrane (Jensen et al. 2004) and indicate that integrins and NG2 are the likely receptors mediating this attachment.
Chick embryo sternal cartilage has proved an ideal model to investigate the structural relationship between the ECM and the primary cilium (Poole et al. 1985,2001). The majority of hyaline chondrocytes possess a primary cilium, which protrudes up to 5 μm into the ECM (Wilsman 1978; Wilsman et al. 1980). Although the cilia are unable to be identified with conventional light microscopy because of optical interference from the ECM, fluorescent immunohistochemical labeling has allowed us to visualize primary cilia and their interactions with the ECM (see Figure 1). Significantly, chick sternal cartilage, like articular cartilage, is influenced by mechanical load. It expresses cartilage-specific molecules such as type II collagen (see Figures 4B and 4C) and aggrecan to provide a mechanically functional load bearing ECM (Liu et al. 1999). Recent in ovo studies have shown that physical movement is necessary for successful development of the skeleton and that immobilization of chick embryos results in altered matrix composition and compromised material properties of cartilage (Mikic et al. 2004).
Distribution of integrins in chick chondrocytes. Field views of (
Number of primary cilia expressing extracellular matrix receptors a

Only cilia projecting at right angles to the light path (focal plane) were included for analysis. Fifty cilia were examined for each receptor.
Integrins are heterodimeric transmembrane glycoproteins that are composed of an α and β subunit (Hynes 1992,2002). The β1 subunit is a common feature of most matrix-binding integrins, whereas the type of α subunit varies and determines the ligand specificity of the receptor. The punctate pattern of staining on the plasma membrane of all three subunits examined in the present study (α2, α3, and β1) was consistent with previous studies on the cellular distribution of integrins in both human articular and chick sternal cartilage (Durr et al. 1993; Hirsch et al. 1996). However, the number of cilia expressing these subunits varied from 17/50 for α2 integrin up to 43/50 for β1. This variability in expression is not unique to cartilage. For example, Praetorius et al. (2004) have shown that β1 integrin is present on the majority of, but not all, primary cilia in the collecting ducts of the kidney, and we have observed somatostatin receptor-3 expression targeted to ~50% of kidney epithelial primary cilia (unpublished observations). It is unclear from the results of the current study whether this variation is a reflection of the temporal state of the primary cilium of a particular cell or variability among cilia, providing a potential mechanism for differentiating among cell subpopulations.
(
(
The two main functions of integrins are (a) to mediate cell adhesion to the ECM via their large extracellular globular domain and (b) to act as receptors that transduce signals from the cell surface into the cytoplasm (Hynes 1992,2002). β1 integrins bind to many ligands present in cartilage, including collagen types II and VI, fibronectin, and laminin (Wurster and Lust 1984; Durr et al. 1993,1996; Loeser 1993,1997). We have also confirmed the expression in chick sterna of two β1 integrin ligands: type II collagen (Figures 4B and 4C) and fibronectin (Figure 2A). Our present findings, coupled with our previous studies, strongly suggest that the integrins associated with the chondrocyte primary cilium play a role in anchoring the cilium to the mechanically functional collagen fibers within the ECM (Poole et al. 1985; Jensen et al. 2004).
Intracellularly, integrins possess a short cytoplasmic tail that interacts with adaptor proteins, signaling molecules, and the actin cytoskeleton (Humphries et al. 2004). To our knowledge, there is no evidence to suggest that an actin cytoskeleton or any of the adaptor proteins such as paxillin, talin, and α-actinin are present within the cilium, although this has not been specifically examined. It is also unknown whether α or β integrins interact with polycystin 1, polycystin 2, fibrocystin, or other signaling and adaptor proteins (e.g., STAT6) that have recently been identified within primary cilia, although integrins and similar proteins are often associated at the cell-matrix interface of focal adhesions in epithelial cells (Wilson 2004). Therefore, we are currently unable to suggest what, if any, interactions are associated with the cytoplasmic tail of ciliary integrins.
The present study has shown for the first time that NG2 is expressed in chick sternal chondrocytes. More importantly, NG2 was also detected in a small proportion (8/50) of primary cilia (Figures 3C and 3D; Table 2). Interestingly, the NG2 molecule contains a chondroitin sulfate moiety and the sites for binding type V and VI collagen (Burg et al. 1996; Tillet et al. 1997). Although several different antibodies against mammalian collagen type VI were tested unsuccessfully in this study, the lack of species-specific antibodies meant that we were not able to identify this ligand in chick sterna. Because it is well established that collagen type VI is present in the pericellular microenvironment of hyaline chondrocytes from a number of species and is also present in developing epiphyseal cartilage (Poole et al. 1992; Sherwin et al. 1999), it is likely that type VI is also present in the developing chick sternum. We therefore suggest that NG2 in both the plasma membrane and the primary cilium is likely to interact with pericellular type VI collagen in chick sternal cartilage.
Type II collagen was abundantly expressed throughout the ECM, including the pericellular microenvironment (Figure 4B) where it completely surrounded the primary cilium (Figure 4C). These results strongly suggest that type II collagen is one of the fibrillar collagens that we have previously observed ultrastructurally in contact with the plasma and ciliary membranes (Jensen et al. 2004). One of the putative receptors for type II collagen is annexin V, which is located on the outer surface of chondrocytes (Koopman et al. 1994; Mollenhauer et al. 1999). The present study is the first to describe its localization in chick sternal cartilage in situ. Annexin V was distributed as large plaques on the plasma membrane (Figures 4A and 4C) and colocalized with pericellular type II collagen (Figure 4B), confirming reports of its role as a putative receptor for type II collagen (Lucic et al. 2003). However, annexin V was not present on the primary cilium. Therefore, although the current study shows an intimate relationship between type II collagen and the primary cilium (Figure 4C), this association is likely to be mediated via integrins rather than annexin V.
CD44 is a well-characterized plasma membrane glycoprotein and the principal hyaluronan receptor in chondrocytes (Chow et al. 1995; Knudson et al. 1996; Jiang et al. 2002). Hyaluronan complexes with aggrecan and link protein to form large proteoglycan aggregates with a substantial swelling potential (Knudson and Knudson 2001). Our own studies using ruthenium red staining have shown that aggrecan complexes are closely associated with the ciliary membrane (Jensen et al. 2004). However, although our results showed a punctate distribution of CD44 on the cell surface, this receptor was not present on the chondrocyte primary cilium. These results therefore suggest that there is no CD44-hyaluronan binding at the ciliary membrane.
It is well established that, although the membrane of the primary cilium is continuous with the plasma membrane of the cell, it is a separate membrane domain with a unique complement of proteins (Bloodgood 1992). Numerous previous studies have shown the localization of specific receptors to the primary cilium in a variety of different cell types. These receptors include somatostatin receptor 3 and serotonin receptor 5-HT6 on neuronal cilia (Handel et al. 1999; Brailov et al. 2000; Pan et al. 2005) and polycystins 1 and 2 on kidney epithelial cilia (Pazour and Rosenbaum 2002; Pazour et al. 2002; Yoder et al. 2002; Nauli et al. 2003). In the current study, the presence of NG2 and integrins on the chondrocyte cilium and the complete absence of ciliary annexin V and CD44 confirm that certain proteins are targeted to the ciliary membrane whereas others are excluded. Because primary cilia of chick sternal chondrocytes extend away from the cell surface, it is possible that receptors expressed in the plasma membrane are involved in cell attachment to the ECM and cell surface signaling, whereas receptor expression on the ciliary membrane may represent a mechanism for sensing changes in the wider pericellular microenvironment, several microns away from the immediate cell surface (Poole et al. 1985; Jensen et al. 2004).
Our finding that several matrix receptors are present on the chondrocyte primary cilium suggests that primary cilia are involved in the signaling processes related to the synthesis and maintenance of the ECM. Integrins have been directly implicated in mechanotransduction pathways in chondrocytes, and mechanical stimulation of chondrocytes results in an increase in intracellular calcium (reviewed in Loeser 2000; Roberts et al. 2001; Lee et al. 2002; Chowdhury et al. 2004; Millward-Sadler and Salter 2004). We have also reported ciliary bending in a wide range of connective tissue cells including chondrocytes, all of which are subjected to a variety of mechanical forces (Poole et al. 1985; Jensen et al. 2004). Previous studies of kidney epithelia have shown that the apical primary cilium acts as a mechanosensor, whereby flow-induced bending of the primary cilium results in an increase in intracellular calcium mediated through mechanosensitive ion channels localized along the cilium or at its base (Praetorius and Spring 2001; Praetorius et al. 2004). In addition, many recent studies in several connective tissue cell types including limb bud cells and fibroblasts also provide strong evidence supporting the hypothesis that primary cilia act as sensory organelles via hedgehog, Wnt, and PDGFαα signaling pathways during development and growth (Corbit et al. 2005; Germino 2005; Haycraft et al. 2005; May et al. 2005; Schneider et al. 2005; Teilmann et al. 2005; Low et al. 2006; Siroky et al. 2006). Therefore, we suggest that integrins on the chondrocyte primary cilium anchor mechanically functional collagen fibers to the ciliary membrane and, following ciliary bending induced by mechanical loading, transduce these signals via intracellular signaling cascades.
In conclusion, this is the first report that α2, α3, and β1 integrins and NG2 are present on the chondrocyte primary cilium and that annexin V and CD44 are excluded from the cilium. We also demonstrate that type II collagen and fibronectin are expressed in the developing sternum, and that type II collagen is closely associated with the cilium. This relationship between the ECM, the receptors, and the primary cilium supports our hypothesis that a direct link exists between the ECM and the primary cilium and implicates integrins as the potential signal transduction molecules utilized by primary cilia of all connective tissue cells.
Footnotes
Acknowledgements
This work was supported by The Royal Society of New Zealand Marsden Fund.
We thank Drs. Bill Stallcup, Jurgen Mollenhauer, Shirley Ayad, and Trevor Sherwin for the generous gifts of antibodies to NG2, annexin V, collagen type VI, and α-acetylated tubulin, respectively. We acknowledge Drs. A.F. Horwitz, T.F. Linsenmayer, and D.M. Famborough for the use of V2E9, II-II6B3, and 39 antibodies supplied through the Developmental Studies Hybridoma Bank, University of Iowa, Ames, IA. We thank the Biomedical Imaging Research Unit, Department of Anatomy with Radiology, University of Auckland for technical support and advice, Dr. Sam S. Bowser for helpful discussions, and S. Swain for help with tissue preparation.
