Abstract
System x− c, one of the main transporters responsible for central nervous system cystine transport, is comprised of two subunits, xCT and 4F2hc. The transport of cystine into cells is rate limiting for glutathione synthesis, the major antioxidant and redox cofactor in the brain. Alterations in glutathione status are prevalent in numerous neurodegenerative diseases, emphasizing the importance of proper cystine homeostasis. However, the distribution of xCT and 4F2hc within the brain and other areas has not been described. Using specific antibodies, both xCT and 4F2hc were localized predominantly to neurons in the mouse and human brain, but some glial cells were labeled as well. Border areas between the brain proper and periphery including the vascular endothelial cells, ependymal cells, choroid plexus, and leptomeninges were also highly positive for the system x− c components. xCT and 4F2hc are also present at the brush border membranes in the kidney and duodenum. These results indicate that system x− c is likely to play a role in cellular health throughout many areas of the brain as well as other organs by maintaining intracellular cystine levels, thereby resulting in low levels of oxidative stress. (J Histochem Cytochem 54: 549–557, 2006)
G
Availability of intracellular cysteine regulates the production of GSH (Orrenius et al. 1983) as well as the production of taurine (Awapara and Wingo 1953) and the iron-sulfur clusters critical for many cellular reactions [reviewed in Sticht and Rosch (1998)]. Cysteine transport is present in many cell types throughout the body [reviewed in Bannai (1984)]. In the brain, however, the transporter responsible for the majority of cysteine transport, ASC-1, is localized predominantly to neurons (Helboe et al. 2003). Another method for uptake of cysteine or its redox partner cystine into cells of the brain must be present, particularly in glial cells with a high capacity for GSH production. Cysteine autooxidizes to its disulfide form, cystine, under aerobic conditions (Wang and Cynader 2000), and cystine is present in the blood at levels 5- to 10-fold higher than cysteine (Droge et al. 1991; Wang and Cynader 2000). Cystine transport has been demonstrated in numerous cultured cell types including macrophages (Piani and Fontana 1994), C-6 rat glioma cells and primary rat astrocytes (Cho and Bannai 1990), PC12 cells, (Froissard et al. 1997), primary fetal rat cells (Sagara et al. 1993), and primary rat cortical neurons (Murphy et al. 1990). Once within the reducing environment of the cell, cystine is readily reduced to cysteine (Cooper 1983). Transport of cystine into cells thus becomes rate limiting for GSH production and therefore plays a major role in cell function and survival.
System x− c is the sodium-independent antiporter responsible for the uptake of cystine via 1:1 exchange with glutamate across the cell membrane (Bannai and Kitamura 1980; Bannai et al. 1986; Watanabe and Bannai 1987). This exchange function is mediated by a heterodimer of a widely used amino acid transporter heavy chain, 4F2hc, and a specific light chain, xCT. xCT mRNA has been detected through Northern blot analysis in macrophages (Piani and Fontana 1994) and mouse brain (Sato et al. 1999) and also by in situ hybridization in mouse brain (Sato et al. 2002). The critical role of cystine uptake in GSH synthesis coupled with the fact that GSH status is altered in several neurodegenerative diseases including Alzheimer's (Adams et al. 1991) and Parkinson's (Sian et al. 1994) diseases makes understanding the distribution of system x− c in the brain and other tissues essential.
In this study we examined for the first time the distribution of xCT and 4F2hc in the mouse and human brain as well as in the monkey intestine and kidney, the two organs most critical for maintenance of cystine levels in the body. Our results show that the 4F2hc antiserum recognizes a single band at 80 kDa on reducing gels, whereas the xCT antiserum recognizes two bands of 40 and 80 kDa. Further analyses including RNAi and in vitro transcription/translation confirmed that although different from the predicted size of 52 kDa, the 40- and 80-kDa bands do indeed represent xCT, most likely a monomer and homodimer of the protein. The two components of system x− c are predominantly present in neurons in the mouse and human brain but are also found in glial cells and in those cells that separate the brain proper from the periphery, including ependymal cells, vascular endothelial cells, choroid plexus, and leptomeninges. xCT and 4F2hc are both also present in brush border epithelial cells of the monkey duodenum and kidney, two areas important for amino acid uptake. These results indicate that system x− c is positioned to play a significant role in cystine and glutamate transport into and/or out of the brain as well as within the brain itself and at sites that are important in nutrient transport.
Materials and Methods
Materials
HT22 cells are immortalized mouse hippocampal neurons used extensively in the field of central nervous system (CNS) oxidative stress (Davis and Maher 1994; Maher and Davis 1996; Li et al. 1997a,b; Sagara and Schubert 1998). Cells were grown in Dulbecco's modified Eagle's medium containing 10% fetal bovine serum and maintained at no greater than 50% confluence. For xCT induction, cells were exposed to 100 μM diethyl maleate (DEM) for 24 hr.
The xCT antibody was raised against the sequence QTHHFKDAFSGRDTSL at position 323-338 in the xCT protein. A pBAD/ThioTOPO (Invitrogen; San Diego, CA) fusion protein using amino acids 316-382 of the xCT protein was generated and expressed in Escherichia coli by arabinose induction, extracted by B-PER reagent and inclusion body solubilization reagent (Pierce; Rockford, IL), and dialyzed. The soluble fusion protein was then coupled to an Affi-Gel 10 (Bio-Rad Laboratories; Hercules, CA) column, followed by addition of antiserum for affinity purification. The 4F2hc antibody was raised against the sequence PYEGLLLQFPFVA at position 514-526 in the 4F2hc protein. The 4F2hc peptide was used for affinity purification of the antibody in a similar manner as described above using the xCT fusion protein.
We have attempted to replicate our results using the lone commercial xCT antibody available (Kim et al. 2001; Tomi et al. 2003; Lim et al. 2005) with poor results. In our hands the antiserum produces many nonspecific bands on immunoblots and therefore is unusable for immunohistochemical procedures. This is in contrast to our antiserum, which produces well-defined specific bands at a molecular weight common to similar light-chain transporters.
Fixation
C57/B6 mice were maintained on Purina 5001 diet. At 12 weeks of age, mice were overanesthetized with ketamine/rompun (120 and 30 mg/kg, respectively). The animals were first perfused with normal saline until their livers were pale and the atrial efflux was clear, then with 4% paraformaldehyde in isotonic phosphate buffer (pH 7.3). The brains were subsequently removed and placed in 4% paraformaldehyde at 4C for 16 hr, followed by 10% sucrose for 24 hr, 30% sucrose for 24 hr, and finally 30% glycol until sectioning took place. Human brain tissue was kindly provided by Dr. Carol Ann Miller of the University of Southern California Alzheimer's Disease Research Center and monkey tissue by Dr. Howard Fox of the Scripps Research Institute. The tissue was thawed in 4% paraformaldehyde for 16 hr at 4C and then cryoprotected as described above for the mouse tissue. All procedures involving animals were reviewed and approved by the Animal Care and use Committee of the Salk Institute for Biological Studies.
Immunohistochemistry and Immunofluorescence
Adult mouse and human brain sections were cut at 40 μm thickness on a freezing microtome. Sections were immunostained with antiserum to xCT (1:100) or 4F2hc (1:100) and visualized using the Vectastain ABC immunoperoxidase system (Vector Laboratories; Burlingame, CA). The blue/gray reaction product was obtained by using the Nova SG chromagen and the red reaction product using the Nova Red chromagen (Vector Laboratories). Negative controls for nonspecific staining were performed using the xCT or 4F2hc antibody preincubated for 24 hr at 4C with 100 × of the respective peptide used for antibody generation. For xCT immunofluorescence, 3T3 cells were transfected for 24 hr with a pcDNA (+) vector (Invitrogen) containing the xCT coding sequence inserted downstream from a T7 promoter site, fixed with 4% paraformaldehyde, blocked with 1% BSA/PBS, and incubated in 1:100 xCT antibody/PBS solution. Alexa 488 goat anti-rabbit IgG secondary antibody conjugate (Invitrogen) was used as the fluorophore at 1:1000.
Immunoblotting
HT22 cells were harvested in RIPA buffer (150 mM NaCl, 1% NP-40, 0.1% SDS, 50 mM Tris, and 1X protease inhibitor (Roche; Indianapolis, IN), and protein content was quantified using the bicinchoninic acid assay (BioRad). In vitro transcription/translation was performed using the STP3 kit (EMD Biosciences; San Diego, CA) and the pcDNA/xCT vector described above. The translated protein and HT22 extracts were treated with 2.5% β-mercaptoethanol, 100 mM dithiothreitol, and 20 mM iodoacetamide and boiled for 5 min. Equal amounts of protein (10 μg) were resolved using 4-15% reducing SDS-PAGE (Bio-Rad), electroblotted onto Immobilon P membrane (Millipore; Bedford, MA), and blocked with 1% milk and 3% BSA in Tris-buffered saline. Blots were hybridized with antibodies against xCT or 4F2hc at a concentration of 1 μg/ml overnight and, following washing, were further hybridized with appropriate horseradish peroxidase-conjugated secondary antibodies (Bio-Rad) at a concentration of 1:15,000. Detection was performed using ECL Western blotting detection reagents (Amersham Biosciences; Piscataway, NJ).
RNA Interference
HT22 cells were grown to ∼30% confluence and incubated with 100 nM xCT dsRNA (Ambion; Austin, TX) generated from position 1736-1758 of the published xCT mRNA sequence or scrambled xCT dsRNA as a control. After 24 hr of incubation, the medium was replaced with fresh medium, and the cells were grown for a further 24 hr and then either solublized and prepared for immunoblotting as described above or used for amino acid uptake studies. Cystine (35S) and leucine (3H) uptake experiments were performed on control or RNAi cells in 12-well culture plates. The cells were rinsed with sodium-free Hanks' Balanced Salt Solution (Na-HBSS) and incubated with either 1 μM cystine or 1 μM leucine for 45 min at 37C. Cells were then rinsed three times with ice-cold Na-HBSS and harvested for scintillation counting.
Results
The distribution of system x− c in the CNS is not well characterized due to the lack of an effective anti-xCT antibody. Therefore, antibodies against the two subunits of system x− c were developed that work well in both immunohistochemical and immunoblotting experiments. The anti-4F2hc antiserum recognizes a single band of 80 kDa on gels of HT22 cell extracts run under disulfide bond-reducing conditions (Figure 1A), consistent with the predicted molecular weight of 4F2hc. The anti-xCT antiserum recognizes two bands of 40 and 80 kDa; the 40-kDa protein band is increased substantially by treating the cells with the electrophilic agent DEM, 100 μM, for 24 hr. To determine if these two xCT bands are indeed specific for the xCT protein, we treated HT22 cells with 100 nM xCT siRNA for 24 hr and assayed both xCT protein and sodium-independent cystine uptake. Figure 1B demonstrates that RNAi leads to a 100% decrease of the 80-kDa bands and a 37% decrease of the 40-kDa band for a total xCT immunoreactivity decrease of56%. The decrease in xCT protein correlates with a 68% decrease in sodium-independent radiolabeled cystine uptake, whereas there was no change in radiolabeled leucine uptake in the dsRNA-treated cells, confirming xCT specificity. The sodium-independent radiolabeled cystine uptake in HT22 cells is inhibitable by unlabeled glutamate or cystine, consistent with known xCT activity (data not shown). Levels of the control protein GAPDH did not change with xCT dsRNA treatment, and control cells were incubated with scrambled xCT siRNA to rule out nonspecific changes in expression and activity. To further confirm the specificity of the xCT antiserum, an in vitro transcription/translation assay was performed that shows that the 40- and 80-kDa bands detected by our antiserum are indeed specific for xCT (Figure 1C). using this same expression vector to overexpress xCT in 3T3 cells leads to a dose-dependent increase in xCT expression, which is detectable by our antibody (Figure 1D). Together these results indicate that our 4F2hc and xCT antisera are highly specific for their respective proteins.
To determine the in vivo expression of system x− c components, we first examined the distribution of xCT in the mouse and human brain. Negative controls omitting the respective primary antibody were performed on all immunohistochemical sections described below and were devoid of reactivity. Figure 2A shows that in the mouse cerebral cortex, xCT (gray) is found in cells that express the neuronal marker MAP2 (red). xCT (gray) is also seen in cells that contain the glial marker glial fibrillary acidic protein (GFAP, red) in the mouse cerebral cortex, as well as in the leptomeningeal cells of the pia mater, the pia vessels, and their subarachnoid coating (Figure 2B). xCT is present in cells that line the mouse brain ventricles, both the choroid plexus epithelial cells (Figure 2C) and the ependymal cells (Figure 2D). Similar to the mouse brain, xCT (blue) in the human brain is expressed in cerebral cortical neurons along with MAP2 (red) (Figure 2E). xCT (blue) is also expressed in GFAP (red) positive glial cells, as well as in vascular endothelial cells and the leptomeninges (Figure 2F). Negative controls of the mouse cortex (Figure 2G) or human cortex (Figure 2H) are devoid of xCT (gray) background reactivity and display only a very low level of background reactivity in the MAP2 chromogenic channel (red).
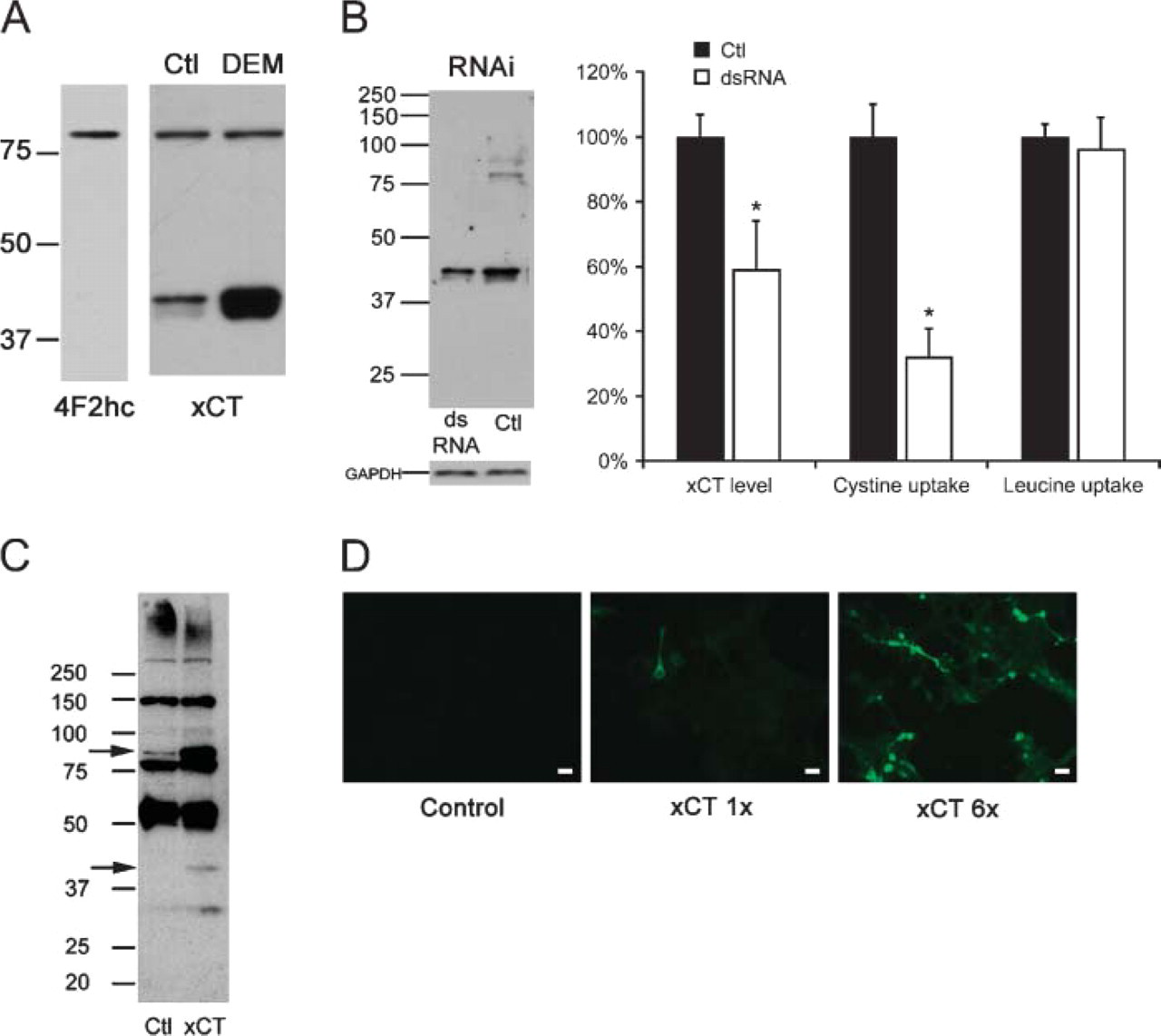
4F2hc and xCT antibody specificity. (
We next investigated the distribution of 4F2hc in the mouse and human brain. Because this protein is a required component of system x− c but is also shared with other amino acid transport systems, we expected the distribution to be broader than, but also overlap, that of xCT. Similar to xCT, 4F2hc is present in neurons (Figure 3A) and glia (Figure 3B) of the mouse cerebral cortex, as well as in the leptomeninges (Figure 3B) and the ependymal cells that line the ventricles (Figure 3C). However, noticeably different from the distribution of xCT, 4F2hc expression is also high in oligodendrocytes of the mouse subcortical white matter (Figure 3D). 4F2hc is present in human brain cerebral cortical neurons, vascular endothelial cells (Figure 3E), and glial cells (Figure 3F), again similar to the distribution of xCT in the human. Negative controls of the mouse cortex (Figure 3G) or human cortex (Figure 3H) are nearly devoid of background reactivity. These results demonstrate that the two individual components of system x− c have a very similar distribution in the mouse and human brain.
Dietary cystine uptake in the intestine and resorption in the kidney are important for maintaining required levels of this amino acid within the body. We examined the distribution of system x− c components in these organs to determine if they are positioned to mediate cystine transport. xCT (Figure 4A) and 4F2hc (Figure 4C) are present in the brush border membrane of the monkey renal tubules, the anatomical location for amino acid transport in this organ. In the intestine, both xCT (Figure 4B) and 4F2hc (Figure 4D) are located at the enterocyte brush border membrane, the site of dietary amino acid entry into the body. Figures 4A′-4D′ show the respective negative controls. These results show that system x− c is indeed positioned to mediate cystine and/or glutamate transport in the duodenum and kidney.
Discussion
System x− c is responsible for the import of cystine into cells, where it is reduced and used for the synthesis of both proteins and the tripeptide antioxidant GSH. Glutamate blocks the import of cystine through system x− c, and non-ionotropic glutamate-induced cell death is directly proportional to the ability of glutamate to block cystine uptake into the cell. Low levels of cystine or elevated levels of glutamate in the culture media have similar effects on cytotoxicity. This phenomenon has been described in neuroblastoma-primary retina hybrid cells and primary rat hippocampal neurons (Murphy et al. 1989), primary rat embryonic cortical neurons (Murphy and Baraban 1990; Murphy et al. 1990), astrocytes (Chen et al. 2000), and oligodendrocytes (Oka et al. 1993) and has been extensively studied in HT22 cells (Tan et al. 1998; Satoh et al. 2004). The glutamate-mediated inhibition of cystine uptake into cells leads to a decrease in GSH, which results in oxidative stress and a form of programmed cell death called oxytosis (Tan et al. 2001). Therefore, system x− c plays a critical role in modulating the ability of cells to deal with oxidative stress. Data presented here for the first time describe the distribution for this antiporter in the brain, intestine, and kidney, demonstrating that system x− c likely plays a significant role in the modulation of cellular oxidative stress by mediating cystine uptake into both neurons and glia, as well as across membranes involved in dietary amino acid uptake.
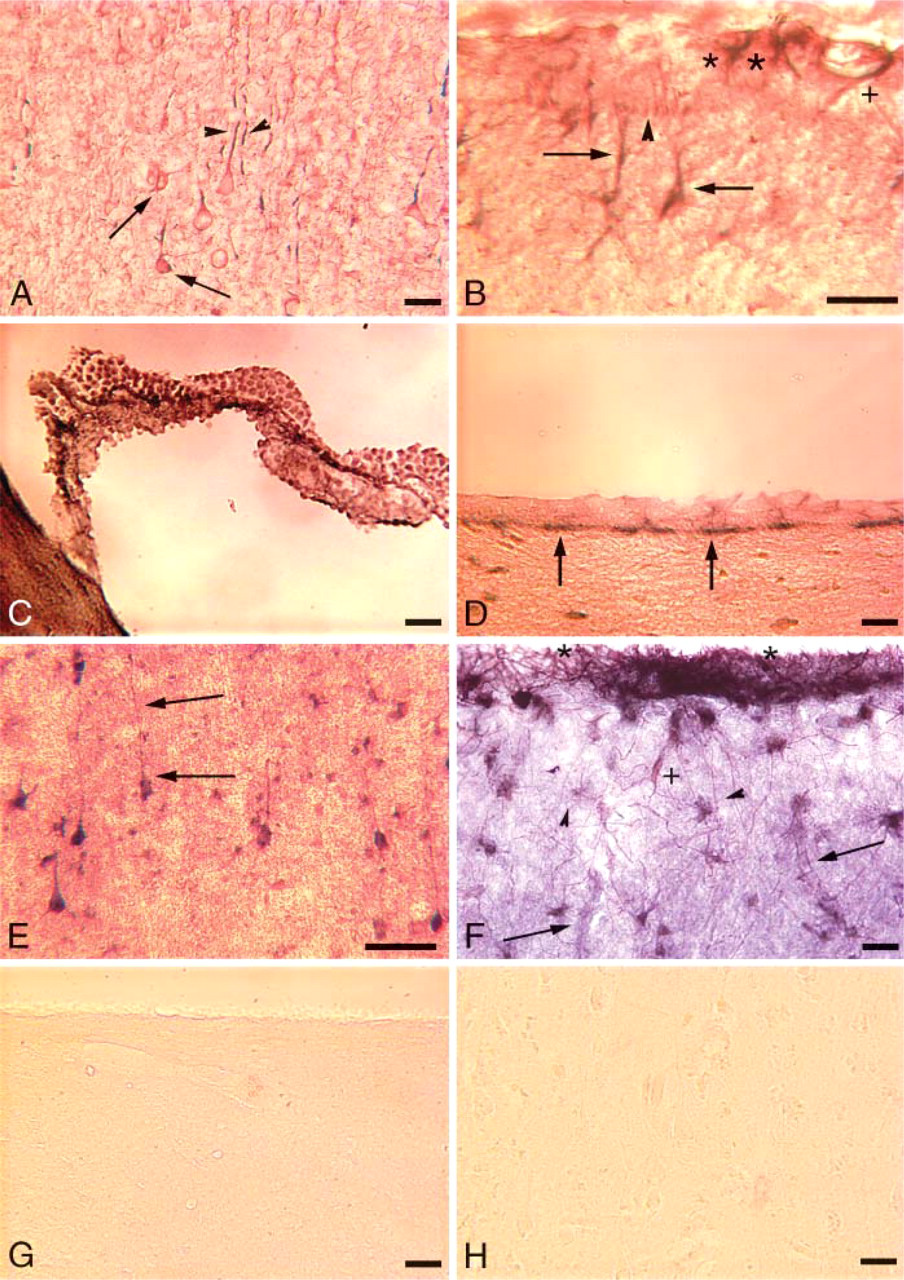
xCT expression in the mouse and human brain. Overlapping expression of xCT with MAP2 or glial fibrillary acidic protein (GFAP) produces a chromogenic product nearly black in color in the micrographs. (
Our results show that the xCT monomer migrates faster than would be predicted based on its estimated molecular weight of 52 kDa (Kim et al. 2001). Previous studies have shown that other light chains with a predicted molecular weight similar to xCT that associate with the 4F2hc heavy chain also migrate faster than expected at ∼40-45 kDa (Hemler and Strominger 1982; Kolesnikova et al. 2001; Nawashiro et al. 2002), so the observation that xCT migrates at this size is clearly not unusual for amino acid transporter light chains. The slight separation of the 40- and 80-kDa xCT bands into two bands seen on some of our blots seems to be a minor artifact of our gel system, as this anomaly is not always apparent. The 80-kDa band most likely represents a non-disulfide-linked homodimer of xCT, present even under the harsh reducing conditions used in this study. The intensity ratio of the 80- to 40-kDa bands is variable as seen in Figures 1A and 1B. The reason for this variability is currently unknown but may be related to the oxidative stress status of the cells or to a reserve pool of xCT light chain. Other nutrient transporters (Overvoorde et al. 1997; Blasco et al. 2003) and anion exchangers (Zolotarev et al. 1999) are known to form homodimers in a similar manner. Our RNAi and in vitro transcription/translation experiments (Figure 1) demonstrate that both the 40- and 80-kDa bands are indeed specific for xCT. There is a greater decrease in sodium-independent cystine uptake than there is for xCT protein in the RNAi experiments, but this difference is not statistically significant. Our xCT antiserum also recognizes the DEM-mediated increase in protein (Figure 1A) demonstrated previously for xCT RNA (Hosoya et al. 2002; Tomi et al. 2002), and it recognizes in a dose-dependent manner the overexpression of xCT in 3T3 cells (Figure 1D). These results indicate that our xCT antiserum is indeed specific for xCT protein and is suitable for use in immunohistochemical studies. Further studies that define the structure of the xCT homodimer and investigate the relative proportion of each xCT band are currently underway.
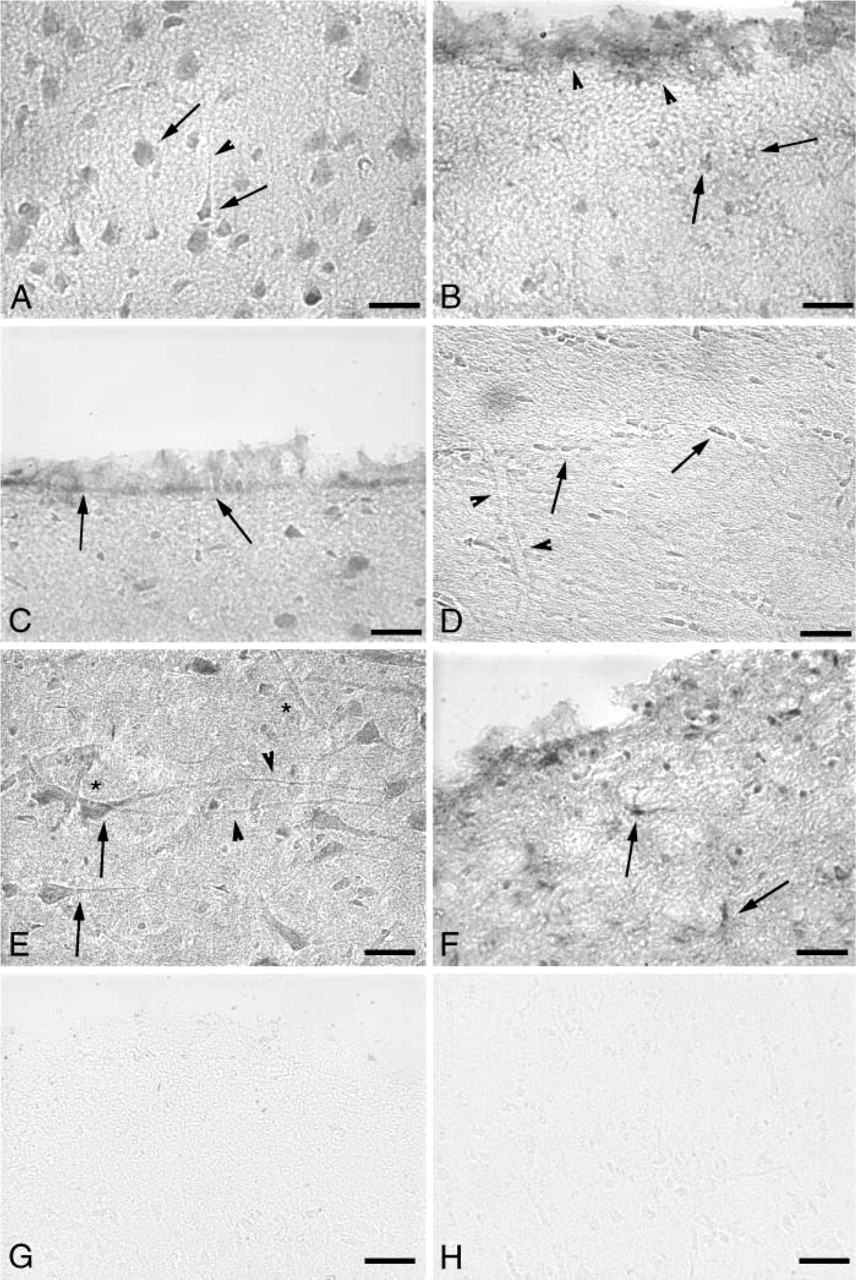
4F2hc expression in the mouse and human brain. (
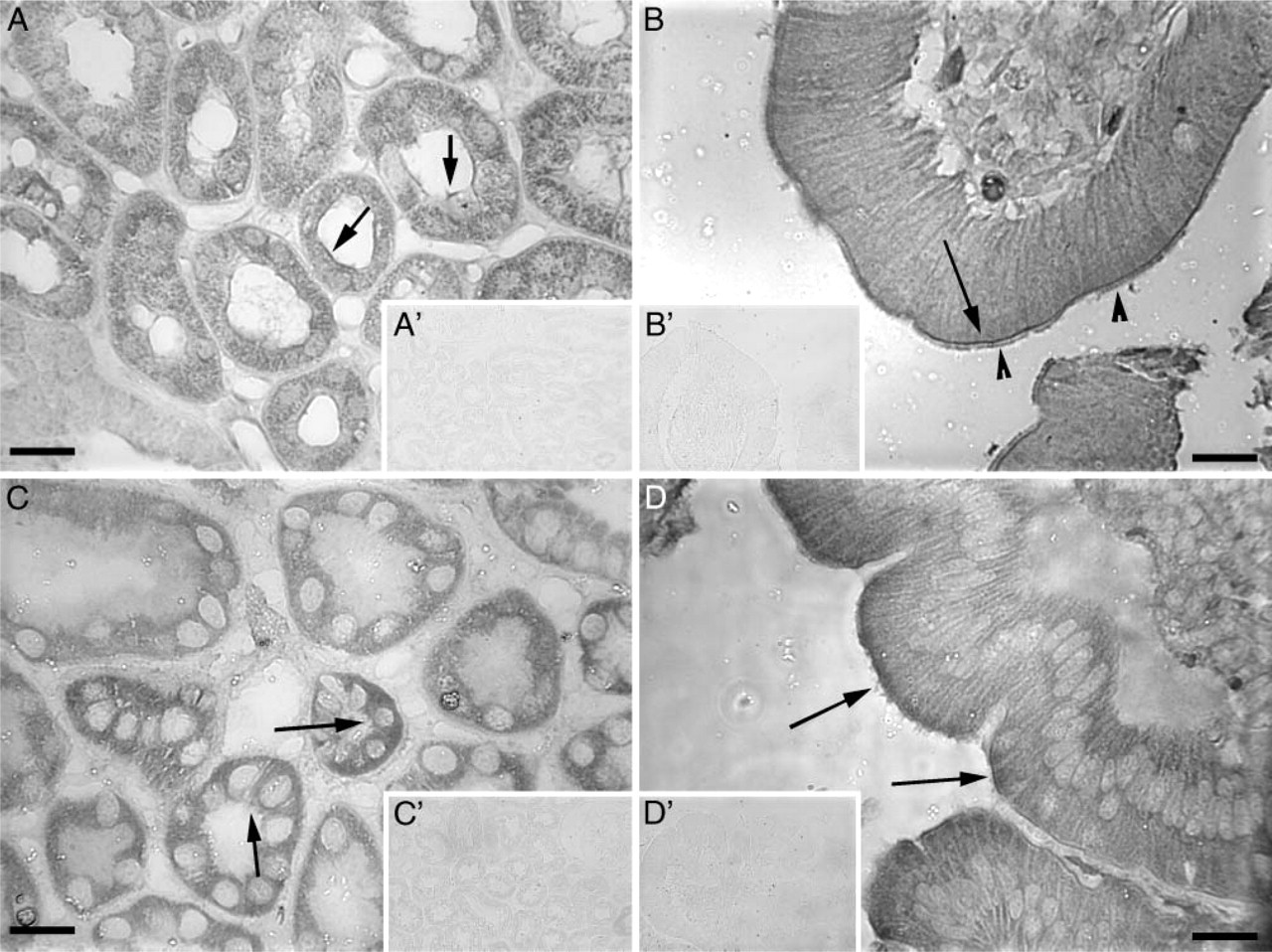
xCT and 4F2hc in the monkey kidney and duodenum. (
The components of system x− c are expressed in neurons and glia of the mouse and human brain and in areas that mediate transport of nutrients into and out of the brain parenchyma, namely, the vasculature, choroid plexus, leptomeninges, and ependymal cells. Double labeling of cells with xCT antibodies and MAP2 or GFAP antibodies helped to clarify the cellular specificity of the xCT antiserum, as the cellular identity of the plasma membrane and apical process immunoreactivity for xCT alone was less clear (data not shown). 4F2hc immunohistochemistry was limited to single labeling as the identity of 4F2hc-positive cells is less ambiguous; therefore, grayscale micrographs of 4F2hc immunoreactivity in the brain are presented herein. Our results indicate that system x− c plays a critical role in maintaining the cystine/glutamate balance between the brain and the periphery. Our ddta are consistent with previous reports that detected xCT in the mouse leptomeninges and ependymal cells by in situ hybridization (Sato et al. 2002), in human brain by Northern blot (Bassi et al. 2001), in mouse kidney and intestine by RT-PCR (Bassi et al. 2001), in primary neuronal cultures (Shih et al. 2003), immortalized (Tetsuka et al. 2001) and primary (Gochenauer and Robinson 2001) astrocyte cultures, and in the HT22 neuronal cell line (Lewerenz et al. 2003). One would expect that 4F2hc would be more widely distributed than xCT, because it is paired with numerous other amino acid light chain transporters and, in general, that is what we found. Although we did not include choroid plexus in our 4F2hc immunostaining panel, system x− c activity has been demonstrated in a conditionally immortalized mouse choroid plexus epithelial cell line (Tetsuka et al. 2001). 4F2hc staining is somewhat weaker in the human brain leptomeninges than that for xCT (Figure 2F vs. Figure 3F), although the qualitative nature of immunohistochemistry makes it difficult to make value comparisons. Additionally, it is possible that the heavy chain rBAT plays a role along with xCT in the leptomeninges to transport cystine and glutamate. rBAT has significant homology to 4F2hc and has been shown to functionally pair with xCT to form a system x− c-like activity (Wang et al. 2003). The cell body 4F2hc immunoreactivity in the neuronal-sparse plexiform layer of the cerebral cortex (Figures 3B and 3F) is most likely glial in nature, as it is similar in appearance to the xCT staining in this same area (Figures 2B and 2F). Although the glial cell bodies in these figures are xCT and 4F2hc positive, many of the glial processes are xCT negative as demonstrated clearly in the xCT/GFAP double-staining panel (Figure 2F). Although we did detect xCT in human brain vasculature by immunohistochemistry, we did not detect it in the vasculature of the mouse brain, which also agrees with previous work that failed to demonstrate system x− c activity at the rodent blood-brain barrier (BBB) (Benrabh and Lefauconnier 1996). Our results leave open the possibility that there is a functional cystine/glutamate antiporter in the human BBB, particularly because 4F2hc is also present in this location. These findings are important as they indicate that there are different pathways in the rodent and human BBB for the transport of cystine and glutamate, which may alter the kinetics and function of the intracellular redox pathway.
System x− c components are also present in enterocytes at the duodenal brush border and in the brush border of the renal tubules. This is the first demonstration of xCT and 4F2hc in these areas that are critical for cystine uptake and resorption. Whereas cystine uptake in duodenal brush border membrane vesicles has weak to zero sodium dependence (Furlong and Stiel 1993), system bo,+, formed by rBAT and bo,+ AT, is thought to play the major role for cystine transport in the duodenum. Indeed, the recessive disorder of intestinal and kidney cystine absorption, cystinuria, is due at least in part to mutations in rBAT (type I) and bo,+ AT (non-type I). Although a previous study has localized 4F2hc to the basolateral aspect of the kidney and intestinal brush border (Quackenbush et al. 1986), our results show that it is located in a proximal brush border location. It is possible that species differences are in part responsible for this discrepancy, as the previous study used human tissue whereas we used monkey tissue. This localization of 4F2hc and xCT to the absorptive sites for cystine in the intestine and kidney indicates that system x− c may play a significant and heretofore unknown role in gut and kidney amino acid transport. Functional studies of system x− c at the isolated duodenal and kidney brush border are necessary to further clarify this issue.
In conclusion, our results define the distribution of system x− c components xCT and 4F2hc in the rodent and human brain, as well as in the monkey kidney and duodenum. This is the first report of system x− c protein localization in these areas. The results confirm some aspects of previous studies involving system x− c activity and RNA distribution and also raise new questions about cystine uptake in areas vulnerable to the disease cystinuria, in which there is a defect in cystine transport. The results also indicate that system x− c is important for maintaining redox homeostasis in both neuronal and glial cells in the CNS by importing cystine for GSH synthesis. The brain is particularly vulnerable to oxidative injury due to its high rate of energy metabolism, and maintenance of GSH levels is critical for proper brain function.
Footnotes
Acknowledgments
This work was supported by grants from the National Institutes of Health and The Bundy Foundation.
