Abstract
The communication between the intestinal epithelium and the enteric nervous system has been considered indirect. Mechanical or chemical stimuli activate enteroendocrine cells inducing hormone secretion, which act on sub-epithelial nerve ends, activating the enteric nervous system. However, we identified an epithelial cell that expresses NKAIN4, a neuronal protein associated with the β-subunit of Na+/K+-ATPase. This cell overexpresses Na+/K+-ATPase and ouabain-insensitive Na+-ATPase, enzymes involved in active sodium transport. NKAIN4-positive cells also express neuronal markers as NeuN, acetylcholine-esterase, acetylcholine-transferase, α3- and α7-subunits of ACh receptors, glutamic-decarboxylase, and serotonin-receptor-7, suggesting they are neurons. NKAIN4-positive cells show a polarized shape with an oval body, an apical process finished in a knob-like terminal in contact with the lumen, a basal cilia body at the base of the apical extension, and basal axon-like soma projections connecting sub-epithelial nerve terminals, lymphoid nodules, glial cells, and enterochromaffin cells, forming a network that reaches the epithelial surface. We also showed, using retrograde labeling and immunofluorescence, that these cells receive afferent signals from the enteric nervous system. Finally, we demonstrated that acetylcholine activates NKAIN4-positive cells inducing Ca2+ mobilization and probably serotonin secretion in enterochromaffin cells. NKAIN4-positive cells are neurons that would form a part of a duodenal sensory network for physiological or noxious luminal stimuli.
Introduction
The enteric nervous system (ENS) receives and sends projections to the brain and spinal cord, guaranteeing an enormous information exchange between the gut and the central nervous system (CNS).1–3 The ENS, located in the wall of the gastrointestinal tract, has enteric ganglia (a cluster of neurons and glia interconnected by bundles of nerve fibers), where their neurons innervate other ganglia, muscle layers, and the mucosa. This structural network supports multiple reflex circuits that modulate the digestive tract function. In addition, the ENS depends on the innervation of the autonomic nervous system through the vagus nerve and sympathetic ganglia; however, the system can function even with the severed vagus nerve. 4
Most neurons are in the myenteric and sub-mucosal plexi. In the myenteric plexus (Auerbach’s plexus), located between the longitudinal and circular gastrointestinal tract muscle layers, there is a continuous net around the circumference and along the intestine, with single neurons occasionally located outside this plexus. In the sub-mucosal plexus (Meissner’s plexus), there are ganglia containing secretomotor but not motor neurons, although, in larger mammals (pigs, monkeys, and man), some motor neurons have their cell bodies in the sub-mucosal ganglia. 5 Some small groups of neurons lie in the lamina propria of the mucosa, almost always close to the muscularis mucosae. At the mucosa level, a dense network of interconnecting nerve bundles is found throughout the connective tissue of the lamina propria. This complex net has a sub-glandular, a periglandular, a villous sub-epithelial, and a villous-core plexus. Nerve ends that innervate the mucosa do not penetrate the epithelium.
In the intestine, at least 15 types of neurons have been identified and grouped into four functional categories: intrinsic primary afferent neurons (IPANs), interneurons, motor neurons, and intestinofugal neurons. Intestinal IPANs have Dogiel type II morphology. 6 These cells project endings into the mucosa that respond to physiological stimuli. Moreover, IPANs make synapses with interneurons, motor neurons, and other IPANs. The IPANs in the sub-mucosal and myenteric plexi respond to different luminal stimuli. 7
In general, the CNS receives sensory signals from neural circuits located at the epithelia of different organs (skin, gustative, and olfactory systems). Nevertheless, in the intestine, the evidence indicates that the epithelial sensors do not make synapsis with nerve terminals of the ENS. The synaptic connection would imply that the nervous system could immediately modulate processes like motility and nutrient absorption and allow instantaneous response to the passage of nutrients from one region to another of the intestine. 8 The communication between the epithelium and the ENS has been considered indirect through hormone secretion, not through direct cell-nerve contact. Thus, the luminal stimuli activate the enteroendocrine cells (EECs), causing secretion of serotonin (5-hydroxitriptamine [5-HT]) and gastro intestinal peptides (e.g. cholecystokinin, gastrin, somatostatin, ghrelin). At the same time, these stimuli induce prostaglandin synthesis, which is released from enterocytes. These mediators act on the IPAN’s endings. The IPANs make synapsis with interneurons in the myenteric plexus, which project to vasointestinal peptide (VIP)-containing secretomotor/vasodilator neurons. These neurons release VIP and pituitary adenylate cyclase-activating polypeptide, which interact with enterocytes at the intestinal crypts, increasing the cyclic adenosine 5′-monophosphate and inducing the opening of luminal Cl– channels and intestinal secretion. 7
To the best of our knowledge, the mammalian intestinal epithelium does not have a neuronal cell that could serve as a sensor for luminal stimuli. However, during our immunohistochemical studies on molecular modulation of intestinal Na+ pumps in the guinea pig intestine, we identified a unique epithelial cell type located at the duodenum, which expresses NKAIN4, a neuronal protein associated with the β-subunit of Na+/K+-ATPase. 9 These NKAIN4-positive cells also overexpress the Na+/K+-ATPase and the K+-independent, ouabain-insensitive Na+-ATPase, enzymes involved in the active transepithelial Na+ transport at the intestine level. 10 Our results suggested a possible neuronal origin of the NKAIN4-expressing cells. To confirm this hypothesis, we studied the expression of different neuronal markers and some morphological and functional characteristics of these duodenal cells.
Materials and Methods
Animals
Albino male guinea pigs (300–350 g, n=18) from Instituto Venezolano de Investigaciones Científicas (IVIC) animal facilities were held in polypropylene cages (1.7 square meters and 4–5 animals per cage), with a metal grid cover with specific space for food and water dispensers and hard-wood beds (sterilized by gamma radiation, cleaned daily, and changed weekly), and placed in ventilated rooms (10–12 fresh air changes per hour per room) with controlled environmental temperature (at 22C), light/dark alternating periods of 12 hr and free access to food and filtered potable water. We only use healthy animals fed ad libitum from the weaning with the same commercial guinea pig food (Kaytee Timothy Complete for Guinea Pigs, supplemented with minerals and vitamins, including vitamin C, Kaytee Products Inc., Chilton, Wisconsin), trying to keep the intestinal microbiota constant. In the experiments reported in this study, we did not find differences between male and female guinea pigs. However, for local logistic reasons, we preferentially used male guinea pigs.
Animal procedures followed the protocols approved by the IVIC Bioethical Committee and the Guidelines for the Care and Use of Laboratory Animals (8th Edition) of the US National Research Council, 2011.
Guinea pigs, anesthetized by intraperitoneal injections of sodium pentobarbital (50 mg/Kg), were perfused through the heart with 4% paraformaldehyde in 0.06 M phosphate buffer at pH 7.6. Fixation was completed by immersion of the chosen anatomical piece in the same fixative overnight at 4C. In addition, after the desired intestinal segment extraction, we macroscopically examined the rest of the intestine, spleen, liver, kidneys, heart, and lungs. Any anatomical alteration made these animals unsuitable for experimentation. Subsequently, samples of the affected organs, if any, were subjected to histopathological examination.
Materials
Primary Antibodies
Goat-polyclonal immunoglobulin G (IgG) anti-sodium/potassium-transporting ATPase β-1 subunit-interacting protein-40 (NKAIN4, cat# sc-86183, lot# A2010a, RRID: AB_2282693), blocking peptide for anti-NKAIN4 antibody (cat# sc-86183P, lot # I1014), mouse monoclonal IgG anti-Na+/K+-ATPase α1-subunit (cat# sc-21712, lot# C87113, RRID: AB_62671), goat-polyclonal IgG anti-acetylcholinesterase (AChE, cat# sc-6431, lot# I2606, RRID:AB_2289247), rabbit-polyclonal IgG anti-glutamate decarboxylase-65Kd (GAD65, cat# sc-5601, lot# L1103, RRID:AB_2107921), rabbit-polyclonal IgG anti-acetylcholine receptor α3-subunit (α3-AChR, cat# sc-5590, lot# L2204, RRID:AB_2080519), rabbit-polyclonal IgG anti-AChR α7-subunit (AChR-α7, cat# sc-5544, lot# B0106, RRID:AB_2229517), and goat-polyclonal IgG anti-serotonin-receptor-7 (5-HTR7, cat# sc-31474, lot # D1509, RRID: AB_2122697) were obtained from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA); mouse monoclonal IgG anti-acetylcholine transferase (cat# MAB305, lot# 21010663, RRID: AB_94647) was from EMD Millipore (Darmstadt, Germany); rabbit monoclonal IgG anti-vimentin (cat# D21H3, lot# 5741, RRID: AB_1069545) was from Cell Signaling Technology Inc.(Danvers, MA); and rabbit-polyclonal IgG anti-NeuN (cat# ab-128886, RRID: AB_2744676), rabbit-polyclonal IgG anti-AChE (cat# ab-97299, lot# GR376521-3, RRID:AB_10679986), and monoclonal IgG anti-COX-1-Alexa-Fluor-488 (cat# ab-199027, lot # GR217795-1) were from Abcam (Carlsbad, CA). We also use two chicken immunoglobulin Y (IgY) polyclonal antibodies raised in our laboratory employing an EGGstract IgY purification system (cat# G26610, lot# 223185) acquired from Promega Co. (Madison, WI): An anti-γ-tubulin, a marker for the neuronal basal body.11,12 The γ-tubulin was purified from the guinea pig brain by cycles of microtubule–protein assembly and disassembly at 30C/0C and diethylaminoethyl (DEAE)-Sepharose chromatography (gently ceded by Dr. L. E. Thomas). In addition, we used an anti-K+-independent, ouabain-insensitive Na+-ATPase purified and tested as indicated by Rocafull et al.10,13
Secondary Antibodies
Sheep anti-mouse IgG-Fluorescein isothiocyanate (FITC) (cat# F6252-2ML, lot# 18020801, RRID: AB_259692) were from Sigma-Aldrich Co (St Louis, MO); cow polyclonal anti-rabbit IgG-FITC (cat# sc-2365, lot# K2907, RRID: AB_634836), cow polyclonal anti-goat IgG-FITC (cat# sc-2348, lot# C2709, RRID:AB_634810), donkey polyclonal anti-goat IgG labeled with rhodamine (cat# sc-2094, lot# I0303, RRID:AB_641161), and goat anti-chicken IgY coupled to phycoerythrin (cat# sc-3730, lot# A0406, RRID:AB_649168) were from Santa Cruz Biotechnology Inc.; and goat anti-chicken IgY-FITC (cat# G2691, lot# 18020801, RRID:AB_430852) was from Promega Co.
Dyes, Chemicals, and Other Products
DAPI (4′, 6′-diamino-2-phenylindole) (D9542-10MG), acetylcholine chloride (A6625), xylene histological grade (534050-4L), Paraplast Plus (P3683-1KG), sodic Penthotal (P-3661), Triton X-100 (T-6878), Fast Garnet GBC, C.I.:37210 (F8761), pararosaniline, CI: 42500 m(P-1528), hematoxylin, (A3136), serotonin hydrochloride (5-HT, H-5755), nickel chloride hexahydrate (N-5756), ammonium heptamolybdate (A-7302), dimethyl-sulfoxide (grade molecular biology) (D8418-100ML), poly-L-lysine (P-8920), poly-L-lysine hydro-bromide (P-1524), digitonin (D-1407), tetra-methyl-rhodamine-labeled α-bungarotoxin (Rd-α-Bgtx) (T0195-.5MG), ethylenediaminetetraacetic acid (EDTA; ED4SS) Tris-HCl (T-3253), Trizma (T-4253), glutaraldehyde grade I, for electron microscopy (G5882-100ML), DL-dithiothreitol (DTT) (D-0632), malachite green oxalate salt, C.I.:42000:1 (M9015-25G), albumin bovine serum (B4287-25G), HEPES (N-[2-hydroxyethyl]piperazine-N′-[etha-nesulfonic acid]) (H3375-100G), formamide (grade molecular biology) (F9037-100ML), 3, 3′-diaminobenzidine (D-5637), D(+)-glucose (G5767-500G), polyethylene glycol (PEG; grade molecular biology) (P-5413), and paraformaldehyde (P6148-500G) were obtained from Sigma-Aldrich Co and DiI (1,1′-Dioctadecyl-3,3,3,′3′-tetramethyl-indocarbocyanine perchlorate) (D282), neutral red (3-amino-7-dimethylamino-2methylphenazine) (N3246), pluronic F-127 (P-1572), and Fluo-4 AM (F14201) were obtained from Invitrogen (Carlsbad, CA). Normal goat (sc-2043) and horse (product number 11829220) sera were procured from Santa Cruz Biotechnology, Inc. and optimal cutting temperature embedding medium (OCT) (Cryomatrix cat. 6769006) and Immu-Mount (cat. 9990402) were from Thermo Scientific (Waltham, MA). Hydrogen peroxide (cat. 31642) and hydroquinone (cat. 15613) were from Riedel-de Haën (Seelze, Germany), and Permount (SP15-500) and Crystal-Mount (cat. Mø2) were obtained from Fisher (Waltham, MA) and Biømeda Corp. (Foster City, CA), respectively. Nicotine (art. 820877), acetone GR (art. 510), ethanol GR (art. 1.00983.2500), methanol GR (art. 2500), sodium bicarbonate (art. 1329), disodium sulfate (K2SO4) (art. 6649), di-potassium hydrogen phosphate (art. 5101), sodium chloride (art. 6404), potassium chloride (art. 4936), tert-butanol (GR) (art. 9629), D(+)saccharose, (art. 7651), sodium sulfite (art. 6657), and phosphomolybdic acid (art. 532) were from Merck (Rahway, NJ), and bovine pancreas trypsin (cat 6502) was from Calbiochem (San Diego, CA).
Methods
Microscopic Observation and Integrated Optical Density Measurements
We use a light microscope Nikon Eclipse E600 (Nippon Kogahu KK, Tokyo, Japan) equipped with epifluorescence illumination, a 100W mercury lamp light source, and a 60× differential interference contrast (DIC) plan-apochromatic objective with 1.40 numerical aperture and oil immersion to observe fluorescent samples. We used the appropriate cube, UV-2A (Ex. 330–380 nm), B-2A (Ex. 450–490 nm), and G-2A (Ex. 510–560 nm) to visualize DAPI, FITC, and rhodamine/phycoerythrin, respectively. When necessary, we used an RGB cube to register all emissions, including autofluorescence.
Images (2048 × 2048 pixels), obtained as 24-bit color TIFF files with a SPOT Flex FX1520 camera (Diagnostic Instruments Inc. Sterling Heights, MI), were analyzed using ImageJ version 1.52a (Free Software, National Institutes of Health, Bethesda, test; RRID: SCR_018743).
To quantify fluorescence in positive cells or to distinguish them from negative control or autofluorescence (particularly in critical cases), we measured the integrated optical density (IOD) on 50 differentially stained cells for each tested antigen; to determine the IOD, a region on the labeled cell was selected (circle of 50 × 50 pixels = 2500 pixel 2 = 20 µm2, using the Elliptical selection in the software), and then the fluorescence intensity measured; at that moment, we moved the same circle to the external boundary of the tissue, and the IOD was quantified there. We also determined IOD in negative control sections. Finally, the linearity of our determinations was verified by measuring the IOD of objects excited at different UV intensities through a neutral density filter, as reported by Iwamoto and Allen. 14
The thick sections and whole mounts were also analyzed with a confocal laser scanning microscope (Nikon C1; Nippon Kogahu KK), coupled to an inverted microscope Nikon Eclipse TE2000-U (Nippon Kogahu KK), with sapphire (488 nm); HeNe (543 nm and 633 nm) lasers; and 535/50, 590/50 (BP), and 650 (LP) emission filters, using the Nikon EZ-C1 3.4 and Nikon EZ-C1 Free-Viewer 3.9 software (Nippon Kogahu KK) for image acquisition and processing, respectively. In addition, we used the ImageJ 1.52a software (Free Software, National Institutes of Health) for processing. In these experiments, we obtained a series of confocal laser scanning microscopy sections (0.5–1 µm thickness) to evaluate structures of interest. Images were generated in two fluorescence channels using 488- and 543-nm excitation wavelengths. The transmitted image was recorded at the 633-nm excitation wavelength. Images were analyzed in 3D projections using ImageJ plugins.
The thick sections and whole mounts stained with silver and methylene blue were analyzed using DIC with a Nikon Eclipse 600 microscope (Nippon Kogahu KK) at different focal depths (1 µm). Stacks were analyzed using ImageJ, as suggested by Zimmermann et al. 15
Immunohistochemistry
Fixed duodenum, jejunum, ileum, proximal, and distal colon were dehydrated and embedded in Paraplast-Plus. Sections (5–10 μm) were deparaffinized in xylene and rehydrated with phosphate-buffered saline (PBS). Autofluorescence was quenched using 0.25% NH3 in 70% ethanol. 16 We used proteolyte-induced epitope retrieval with trypsin. 17 Sections were incubated with 0.1% trypsin and 0.1% calcium chloride in PBS at 37C for 15 min and then extensively washed with PBS. Slices were incubated with PBST (PBS plus 0.1% Triton X-100 [unless otherwise indicated]) for 10 min at room temperature to permeabilize the tissue and re-incubated with a blocking solution (5% bovine serum albumin [BSA] in PBST) for 1 hr. The primary antibody diluted (1:1000, in the blocking solution) was added and incubated at 4C overnight. Samples were washed with PBS and incubated with secondary antibodies (1:1000) coupled to FITC or rhodamine for 60 min at room temperature in the dark. Nuclei were stained with DAPI (1 μg/mL in PBS) for 10 min. Sections were mounted using an Immu-Mount.
In immunohistochemical assays, negative controls used non-immune serum, as recommended by Hewitt et al. 18 IOD measurement was employed to quantify the fluorescence of positive cells, binding of unspecific antibodies, and background for each primary antibody. Nonspecific antibody binding was blocked by incubating the tissue slices with 5% normal horse serum in PBST for 60 min at room temperature. In addition, for the NKAIN4 primary antibody, we used pre-absorbed negative controls. The antibody was incubated with its blocking peptide (1:2 ratios, as recommended by the manufacturer) and then used at a 1:1000 antibody dilution, replacing the primary antibody alone. Positive controls for primary antibodies were performed in guinea pig anatomical samples, where the presence of the biomarker is known. In particular, we used immunofluorescence in guinea pig encephalon as positive controls for anti-NKAIN4 and neuronal markers antibodies.
Enterochromaffin Cell Quantification
Serotonin-producing cells were identified by fluorescence in paraffin-embedded tissue sections following the method described by Kaneko et al. 19 During the treatment with formaldehyde, the serotonin contained in enterochromaffin cells (ECs) is converted to strongly fluorescent substances in situ. In biological samples, the fluorescence intensity of formed compounds is proportional to the amine concentrations.20,21
Microscopic observation was performed with a 40× plan-apochromatic objective (0.75 numerical aperture) and the UV-2A, B2A, and G-2A filters and digitally photographed. We determined the parenchymal area occupied by the mucosae and the percentage and density of serotonin-containing and NKAIN4-positive cells in the sample.22,23 The brightness and contrast of fluorescent images, previously converted from 24 to 8 bits, were increased to differentiate cells, nuclei, and parenchyma from the background, and the color range threshold was set to encompass these structures. Under these conditions, the ImageJ (RRID: SCR_003070) provided the number and the fractional area occupied by cell nuclei or nuclear clusters in the pictures. This procedure allowed us to estimate the parenchyma area occupied by the mucosa, villi, and crypts and to determine the number and ratio of the serotonin-producing and NKAIN-4 positive cells in the evaluated area. In addition, this method allowed for determining the nucleus/cytoplasm ratio, diameter, and height of NKAIN-4 positive cells in cryosections employed for retrograde labeling.
Retrograde Labeling
The retrograde labeling with a lipophilic carbocyanine dye (DiI) was performed using the procedure described by Berthoud et al. 24 and Tassicker et al. 25 with modifications. Animals were anesthetized, perfused, and fixed as indicated earlier; a section of a central portion of the duodenum, 1- to 2-cm long, was excised with its mesentery, the nerve trunks dissected under a stereomicroscope up to 1 cm away from the wall of the duodenum, the end pinned firmly, and a DiI crystal placed on the mesenteric para-vascular nerve and finally covered with cyanoacrylate adhesive. The preparation was maintained in the fixative solution at 4C in the dark for 30 days. Small pieces were embedded in Cryomatrix, frozen in liquid nitrogen, and cut into 12- and 50-µm sections. Tissue sections were treated with digitonin, as suggested by Matsubayashi et al. 26 NKAIN-positive cells were visualized using immunoreactivity for anti-NKAIN4 polyclonal antibody, and the anti-goat IgG conjugated to FITC. Samples were observed with the epifluorescence microscope using the excitation cubes G-2A (510–560 nm) and B-2A (460–490 nm) for viewing fluorescence.
Isolation of Villous-Crypt Units of the Intestinal Mucosa
Villous-crypt units of guinea pig duodenal mucosa were isolated using a modification of the method described by del Castillo 27 that employs a hyperosmotic intracellular-like solution to isolate small intestinal cells. Briefly, the duodenum was excised from the pyloric sphincter to the end of the duodenal flexure, rinsed with NaCl 0.9% at 4C, and washed with an intracellular-like solution (7-mM K2SO4, 44-mM K2HPO4, 9-mM NaHCO3, 10-mM HEPES, and 180-mM glucose, pH 7.4 and 340 mosmol/l] at 4C. The intestinal segment was filled with the intracellular-like solution containing 0.036-mM DTT and 3-mM EDTA at 4C and incubated for 30 min in the same medium. Arterial forceps were used to close both intestinal ends. The segment was gently palpated for 2 min, and the mucosal material was transferred to 10-ml Dulbecco’s medium at 4C. The isolated villous-crypt units were collected by centrifugation at 100 × g for 5 min and re-suspended in 1 ml of the same medium. This procedure allowed us isolation of highly viable villous-crypt units (more than 99% viability) without contamination with other intestinal structures.
Histochemical Methods
Demonstration of Serotonin in NKAIN-4-Positive Cells
Paraffin-embedded sections (5 μm) were stained for NKAIN4 using an anti-goat secondary antibody coupled to rhodamine. Slices were photographed and registered. Next, the samples were treated with PBS-Tween and PBS to remove the immunostaining, and then Schmorl’s reaction was performed as described by Stagaard et al. 28 Sections were incubated with ferric ferricyanide for 15 min, rinsed with running tap water for 10 min, dehydrated, mounted in synthetic resin, and newly photographed and compared. We did not use counterstaining. As a control, other sections were stained using the diazonium reaction, with Fast Garnet GBC as the substrate, for localization of ECs. 29
Bungarotoxin Binding
Native expression of a functional neuronal acetylcholine receptor subunit (nAChR-α7) was analyzed using rhodamine-labeled α-bungarotoxin (Rd-α-Bgtx) following the method described by Rogozhin et al.
30
and de Lucas-Cerrillo et al.
31
with some modifications. The isolated villous-crypt units were adhered to slides using poly-
Silver Impregnation
The complete guinea pig duodenum was excised and fixed in 4% paraformaldehyde in phosphate buffer. A segment was dehydrated in 30% saccharose in PBS, embedded in OCT, and sectioned in slices of 150-µm thicknesses. Sections were impregnated with 10% AgNO3 for 1 min at room temperature, rinsed in distilled water for 5 min with agitation, and then reduced with Bodian’s developer (5% sodium sulfite and 1% hydroquinone in distilled H2O) for 1 min. Finally, sections were incubated in xylene and mounted in Permount. We counterstained some samples with 0.001% w/v malachite green or pararosaniline. A second segment was directly impregnated with 10% (w/v) AgNO3 and cleared in ClearT2, a 50% formamide/20% PEG solution described by Kuwajima et al., 32 for at least 24 hr. Finally, the mucosa was isolated under a microscope, mounted in the same medium, and digitally registered as indicated earlier. The incubation of the intestinal tissue impregnated with AgNO3 in the ClearT2 medium allows mucosal dissection since it remains soft, in contrast with the not-cleared tissue that breaks very easily.
Methylene Blue Staining
To study the innervation in the duodenal epithelium, we use a modification of the technique of methylene blue staining described by Müller. 33 Briefly, segments of the duodenum, approximately 9 mm2, were stained with methylene blue (0.25%). The stained tissue was then fixed by immersion in 2% paraformaldehyde and 1.75% glutaraldehyde in 0.1-M phosphate buffer (pH 7.4) containing 1.8% phosphomolybdic acid and 0.1% hydrogen peroxide (final pH 5.8) for 2.5 hr at 4C, rinsed with distilled H2O, and fixed again in 5% aqueous ammonium heptamolybdate for 5 hr to stabilize the resultant dye precipitate. The tissue was dehydrated with tert-butanol and cleared with the ClearT2 medium 32 for 24 hr. Then, the mucosa was isolated under the microscope, mounted in the same medium, and observed by DIC microscopy. To stain isolated villous-crypt units, we used a variant of this technique. The tissue was fixed with 70% ethanol at 4C for 24 hr, washed in PBS, stained with methylene blue, and cleared with the ClearT2 medium.
Detection of Neural Activity
Neuronal activity of NKAIN4-positive cells was monitored utilizing Okada’s method,34,35 with modifications, using neutral red as a membrane potential fluorescent probe and measuring the intracellular calcium ion dynamics with Fluo-4 AM.36,37
Isolated villous-crypt units were adhered to poly-
After recording the neural activity (for neutral red and Ca2+ imaging), tissues were fixed using cold acetone (−20C) for 10 min and extensively washed with PBS. Endogenous peroxidase activity was blocked with 3% H2O2 for 30 min in methanol and exhaustively washed with PBS. Unspecific binding sites were blocked using PBS containing 5% BSA and 0.1% triton X-100 (blocking solution) for 1 hr at room temperature before incubation with the primary antibody. The samples were incubated overnight at 4C with goat-polyclonal anti-NKAIN4 IgG diluted in the blocking solution (1:500). Then, preparations were incubated with peroxidase-complexed mouse anti-goat IgG (1:500 in PBS) for 1 hr at room temperature, followed by extensive washing with PBS. Finally, sections were incubated with 8% NiCl2, 0.05% 3-diaminobenzidine tetrahydrochloride, 0.02% H2O2, and 50-mM Tris-HCI, pH 7.6 (all components mixed just before use) for 10 min and washed with 50-mM Tris-HCl buffer, pH 7.6. Sections were counterstained with Mayer’s hematoxylin, mounted in Crystal-Mount, and made permanent with Permount. This method results in a dark brown color of immune-stained antigens.
Furthermore, we evaluated the activity of the NKAIN4-positive cells using Ca2+ imaging. This technique allows the visualization of individual neurons and their circuits using the fluorescent Ca2+-sensor Fluo-4 AM (prepared and used as indicated by the manufacturer). Changes in fluorescence reflect fluctuations in intracellular calcium (in[Ca2+]), an indirect indicator of neural activity. 38 Action potentials increase in[Ca2+], causing the fluorescent probes to bind more calcium, resulting in augmented fluorescence emission. 39
Isolated villous-crypt units were seeded on poly-
Statistical Analysis
Values are mean ± SD. Differences between means were evaluated by non-parametric analysis of Kruskal–Wallis and considered significant at p≤0.05 (GraphPad Prism, RRID: SCR_002798). The experiments reported in this study were repeated at least three times, with samples coming from at least three different animals.
Results
NKAIN4 Coexpress With the Sodium Pumps at the Duodenal Epithelium
In the duodenal mucosa, we identified a unique population of epithelial cells that expresses NKAIN4, a neuronal transmembrane protein associated with the β-subunit of Na+/K+-ATPase (Fig. 1). NKAIN4 labeling (in red) is cytoplasmic and perinuclear and shows a lower intensity toward the apical pole of the cell. The NKAIN4-positive cells also overexpress the α1-subunit of the Na+/K+-ATPase (Fig. 1A) and the K+-independent, ouabain-insensitive Na+-ATPase (Fig. 1B), both in green, compared to the neighbor enterocytes. In addition, we recognized buttons and bands located in the lamina propria, near the NKAIN4-positive cells, which also express NKAIN4 and overexpress the Na+ pumps, probably corresponding to endings of the sub-epithelial nervous plexuses.
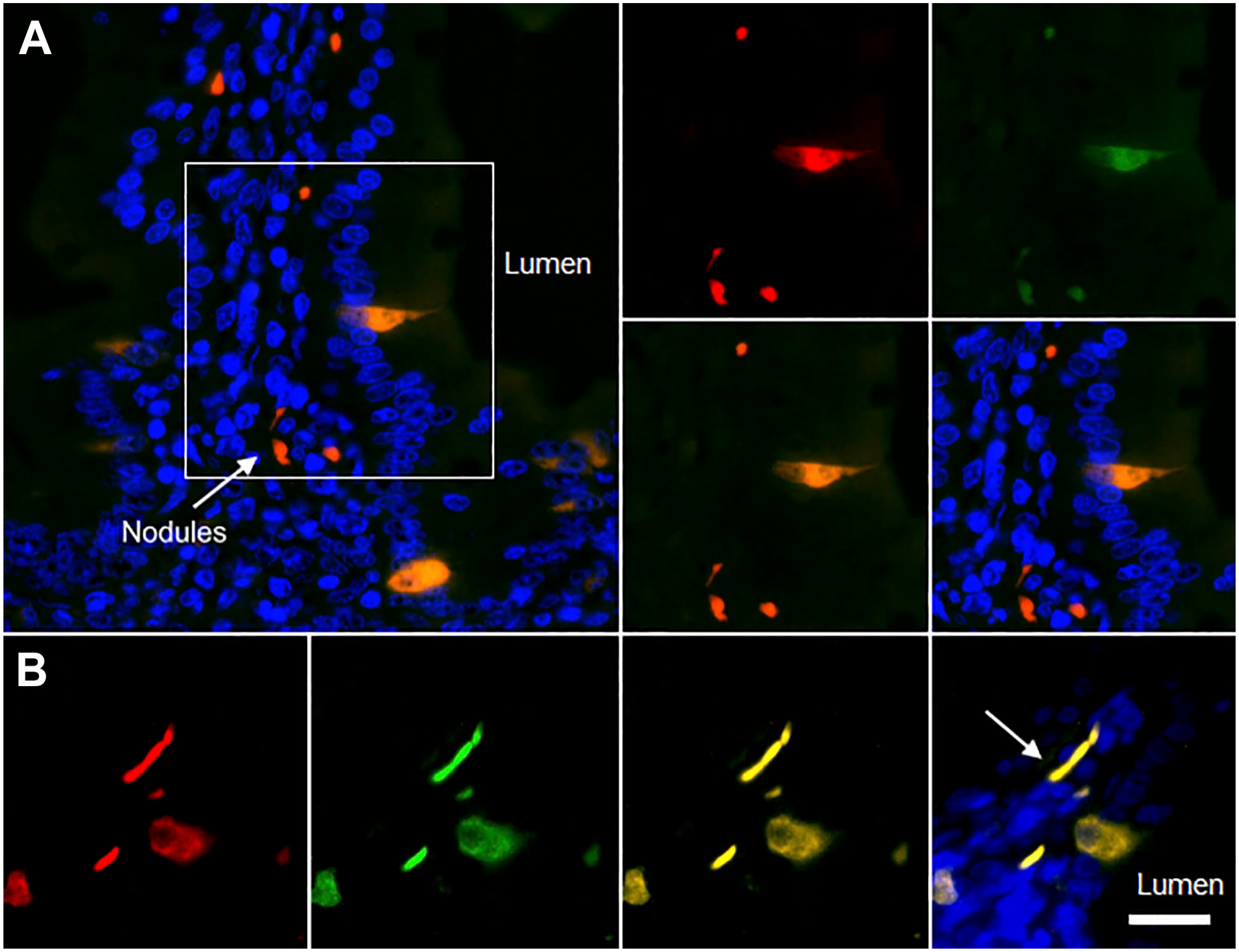
Coexpression of the NKAIN4 with the sodium transporting pumps in guinea pig duodenum. Pictures present the NKAIN-4 stained with rhodamine (red), the α1-subunit of the Na+/K+-ATPase (A), and the K+-independent, ouabain-insensitive Na+-ATPase (B) with fluorescein (green) and nuclei with DAPI (blue). Arrows indicate nerve terminals. Bar corresponds to 20 µm. Abbreviation: DAPI, 4′, 6′-diamino-2-phenylindole.
NKAIN4-Positive Cells Coexpress NeuN, AChE, ChAT, AChR-α3, AChR-α7, GAD65, and 5-HTR7 (Neuronal Markers)
The presence of NKAIN4 and the overexpression of the Na+-transporting enzymes in these cells suggest its possible neuronal origin; to test this hypothesis, we evaluated the expression of different neuronal markers in NKAIN4-positive cells. Figure 2 shows the coexpression of NKAIN4 and NeuN (A), AChE (B), ChAT (C), AChR-α3 (D), AChR-α7 (E), GAD65 (F), and 5-HTR7 (G). The coexpression of NKAIN4 with these neuronal markers strongly suggests the neural origin of the NKAIN4-positive cells and indicates that they could be cholinergic neurons. In addition, the buttons and bands detected in the mucosal stroma coexpress NKAIN4 and all evaluated neuronal markers, confirming they correspond to sub-epithelial nerve endings of the ENS.
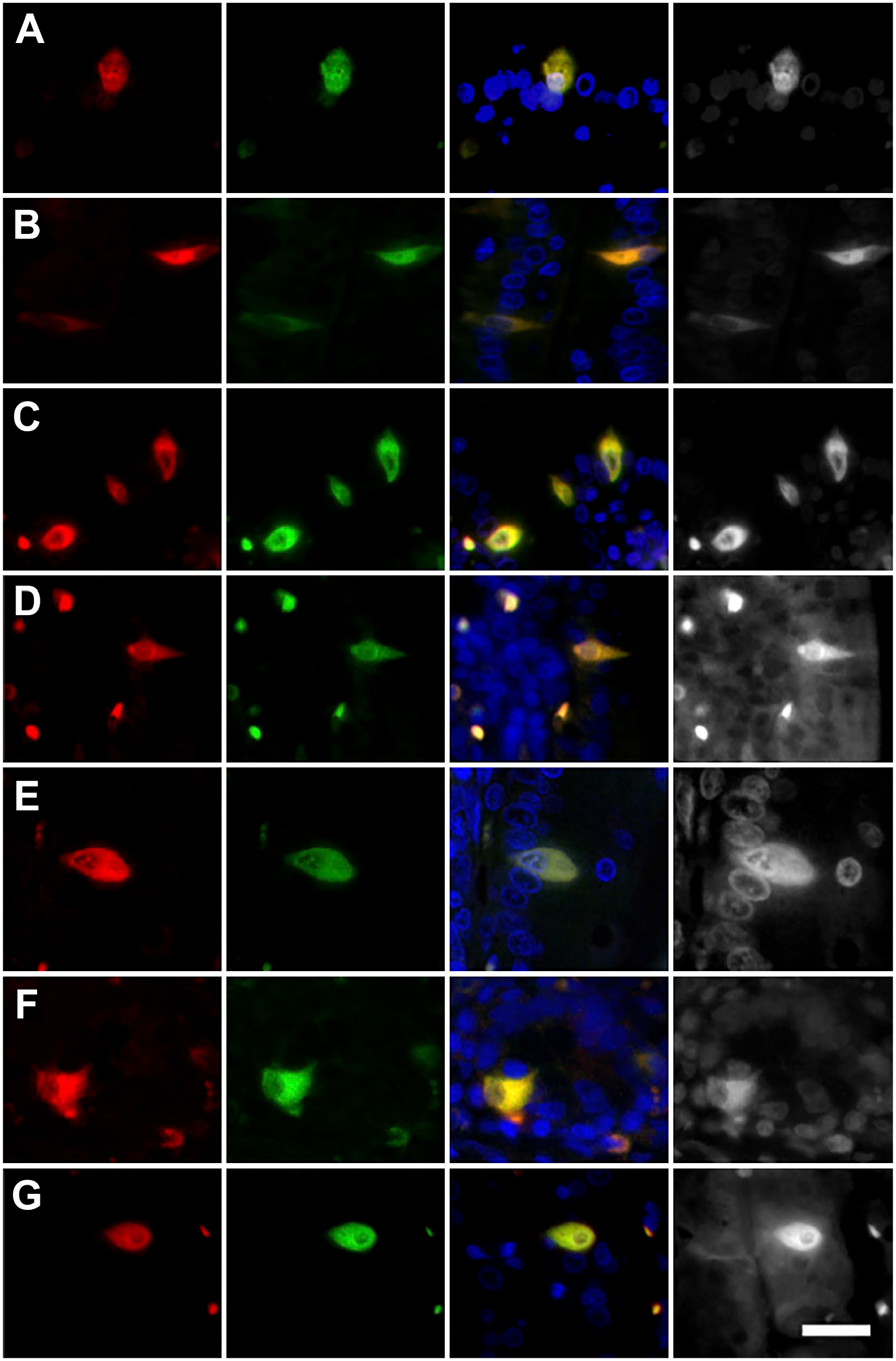
Coexpression of the NKAIN4 with different neuronal markers in guinea pig duodenum. Images present the coexpression of NKAIN4 and NeuN (A), AChE (B), ChAT (C), AChR-α3 (D), AChR-α7 (E), GAD-65 (F), and 5-HTR7 (G). NKAIN4-positive cells are in red, all other markers in green, and nuclei in blue. Bars correspond to 20 µm.
The NKAIN4-positive cells express the α3- and α7-subunits of ACh receptors. The α7-subunit type is the main contributor to the α-bungarotoxin-binding sites in either brain or the peripheral nervous system. Accordingly, we evaluated the binding of bungarotoxin in isolated villous-crypt units of duodenal epithelium. Figure 3 shows the α-bungarotoxin binding, labeled with rhodamine, in villous-crypt units. Bungarotoxin-positive cells (arrows) are in the middle and base of the duodenal villus. The apical and basal extensions of the NKAIN4-positive cells (arrowhead) also bind the toxin.
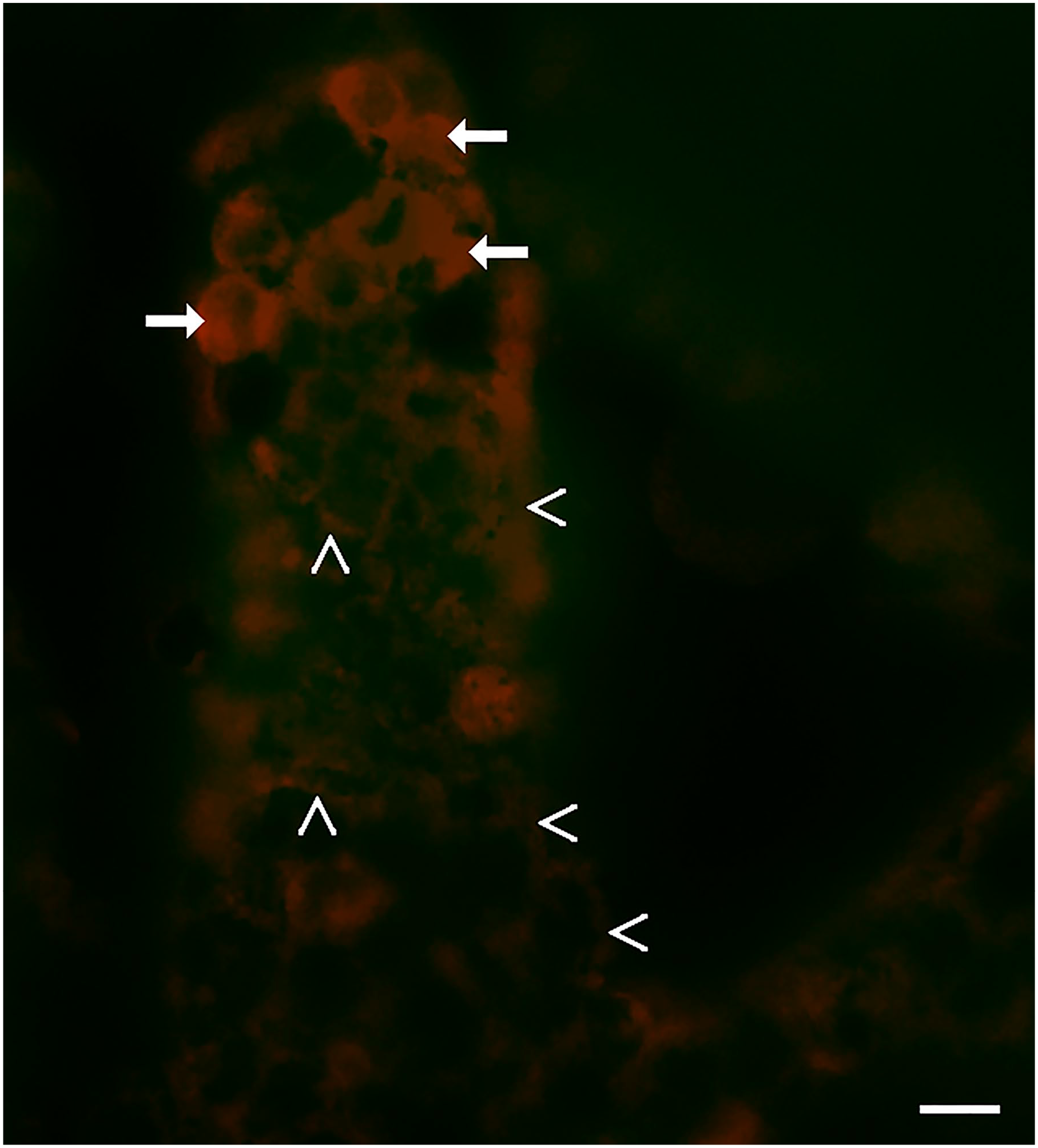
Bungarotoxin binding to the duodenal epithelium. The picture shows the binding of tetramethylrhodamine-α-bungarotoxin in isolated villous-crypt units from guinea pig duodenum. Arrows indicate α-bungarotoxin-positive cells, and arrowheads show the network generated by them. Bars correspond to 20 µm.
NKAIN-Positive Cells Also Express COX-1 and Vimentin
Next, we evaluated if NKAIN4-positive cells are related to other intestinal epithelial cells as the tuft or cup cells. Tuft cells are COX-1 positive and vimentin negative, while cup cells are negative for COX-1 but positive for vimentin.40,41 Thus, we evaluated NKAIN4/COX-1 and NKAIN4/vimentin coexpression in the duodenum. NKAIN4-positive cells were positive for both markers (Fig. 4A and B), indicating that they do not belong to the mentioned cell types. Furthermore, the NKAIN4-positive cells also express γ-tubulin (Fig. 4C).
Coexpression of the NKAIN4 with COX-1, vimentin, and γ-tubulin in guinea pig duodenum. Images present the coexpression of NKAIN-4 and COX-1 (A), vimentin (B), and γ-tubulin (C). The NKAIN4-positive cells are in red, all other markers in green, and nuclei in blue. Bar corresponds to 20 µm.
NKAIN4-Positive Cells Have Soma Projections, a Luminal Knob-Like Terminal, Cilia Basal Body, and Connections With Sub-epithelial Nerve Endings
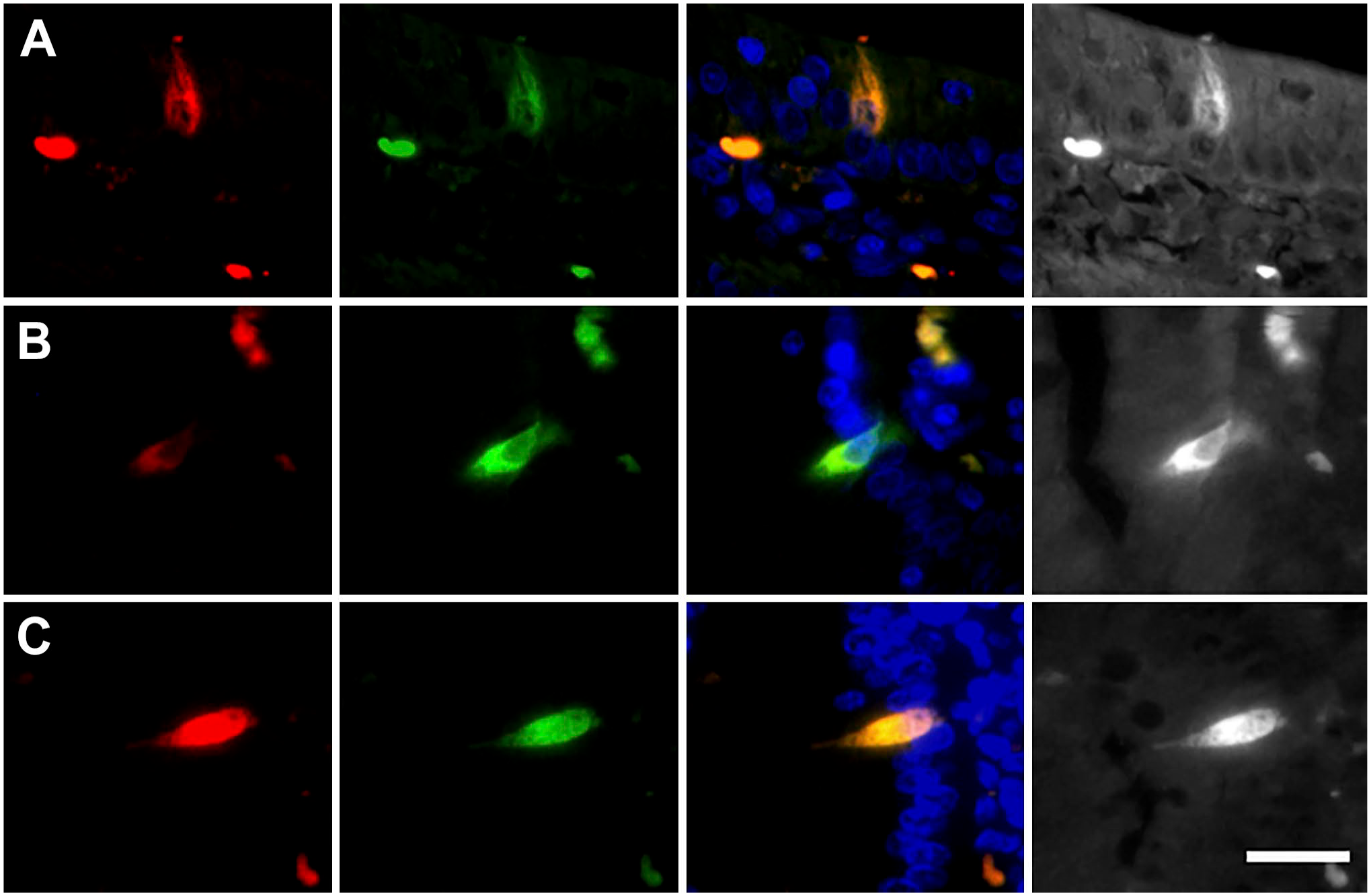
The morphology and epithelial position of the NKAIN4-positive cells permit their characterization with other markers. Thus, we evaluated the coexpression of the α1-subunit of Na+/K+-ATPase (green) and the AChE (red) in the duodenal mucosa. The double-labeling allowed observing a direct connection between the NKAIN4-positive cells and nerve endings at the mucosal stroma (Fig. 5A). Confirming these findings, the double-labeling with NKAIN4 and COX-1 (presented in Fig. 5B) clearly shows cytoplasm projections of the NKAIN4-positive cell toward nerve terminals at the mucosal stroma. Furthermore, it was possible to identify a knob-like terminal at the cell apical pole (Fig. 5C), similar to the dendritic knob present in neurons of some sensory epithelia. This cellular element reaches the luminal space and could be a part of a hypothetical sensory apparatus in this epithelial cell. We also observe at the basal region of the same cell a varicose nerve termination (NKAIN4 positive and COX-1 negative, arrow) that reaches the NKAIN4-positive cells (Fig. 5C). In addition, AChE/5-HTR7 and AChE/NeuN labeling of these cells permitted observing apical and basal projections (Fig. 5D and E, respectively). Evaluating the coexpression of NKAIN4 and γ-tubulin (Fig. 5F), we identified in the supra-nuclear region of the NKAIN4-positive cell, just at the base of the apical process, intracellular nodules with a high concentration of γ-tubulin. Similar elements have been identified in sensory neurons,42–44 including myenteric neurons, 45 called basal bodies.
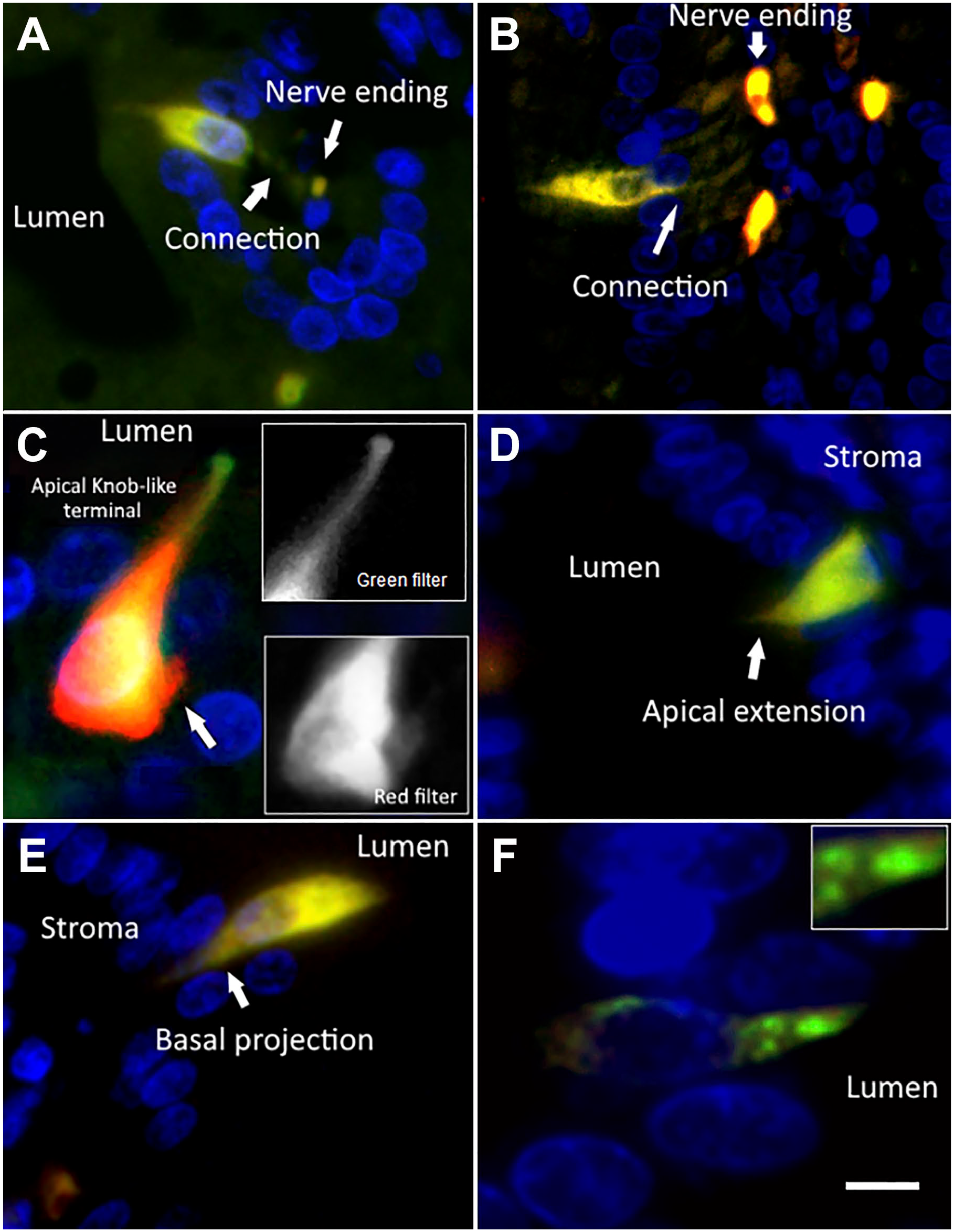
The NKAIN4-positive cells have soma projections, a luminal knob-like terminal, cilia basal body, and direct contact with sub-epithelial nerve endings. Combinations of working antibodies allowed the identification of particular cell structures and connections in NKAIN4-positive cells. The grayscale, lower magnification photographs show the epithelial location of the cells presented in the colored pictures. Image (A) shows the coexpression of AChE and the α1-subunit of the Na+/K+-ATPase. Cell projections connect the epithelial cell and nerve terminal buttons. The coexpression of the NKAIN-4 and COX-1 allows the identification of cell soma projections toward neural terminal buttons (B) and a knob-like structure at the apical pole of the epithelial cell, as well as a nerve tab reaching the base of epithelial cell (C). Coexpression of 5-HTR7 and AChE permits observation of the apical extension of the epithelial cell (D). Image (E) presents the coexpression of NeuN and AChE and allows the identification of epithelial cell basal extensions. Image (F) shows the coexpression of NKAIN4 and γ-tubulin, evidencing tubulin-rich nodules at the base of the apical knob-like structure, probably as a part of the cilia basal body. Bars correspond to 8 µm (A, E); 10 µm (B); 6 µm (C, D, F), and 4 µm (insert in F).
Retrograde Labeling of NKAIN-Positive Cells With Dil
To confirm if the epithelial NKAIN4-positive cells have connections with the ENS, we use retrograde labeling with DiI, an orange-red-fluorescent dye often used as a long-term tracer for neuronal circuits.
Figure 6A presents a thin section (12 µm) of the preparation. The Dil, placed on the mesenteric paravascular nerve, reaches NKAIN-positive cells (green) at the duodenal epithelium. Some white-blue fluorescent granules are observed at the base of the neuroepithelial cell, suggesting the presence of biogenic amines (see following sections). Figure 6B shows, in a thick section (50 µm), the direct contact between the NKAIN4-positive cell projections and nerve terminals (Dil-positive) at the mucosal stroma, confirming the connection of the epithelial cells with the ENS. Figures 6C and D show Dil-positive bands reaching the NKAIN4-positive cells at the epithelium. Finally, Fig. 6E presents the contact between an NKAIN4-positive cell and a highly granulated EC with multiple pseudopods. Moreover, extensions of the NKAIN4-positive cell wrap around sub-epithelial nerve endings, particularly at the base of the crypts. Supplementary video 1 shows additional images of Dil-labeling reaching epithelial NKAIN4-positive cells, the basal projections of these cells contacting nerve terminals at the stroma, and ECs adjacent to nerve endings and NKAIN4-positive cells.
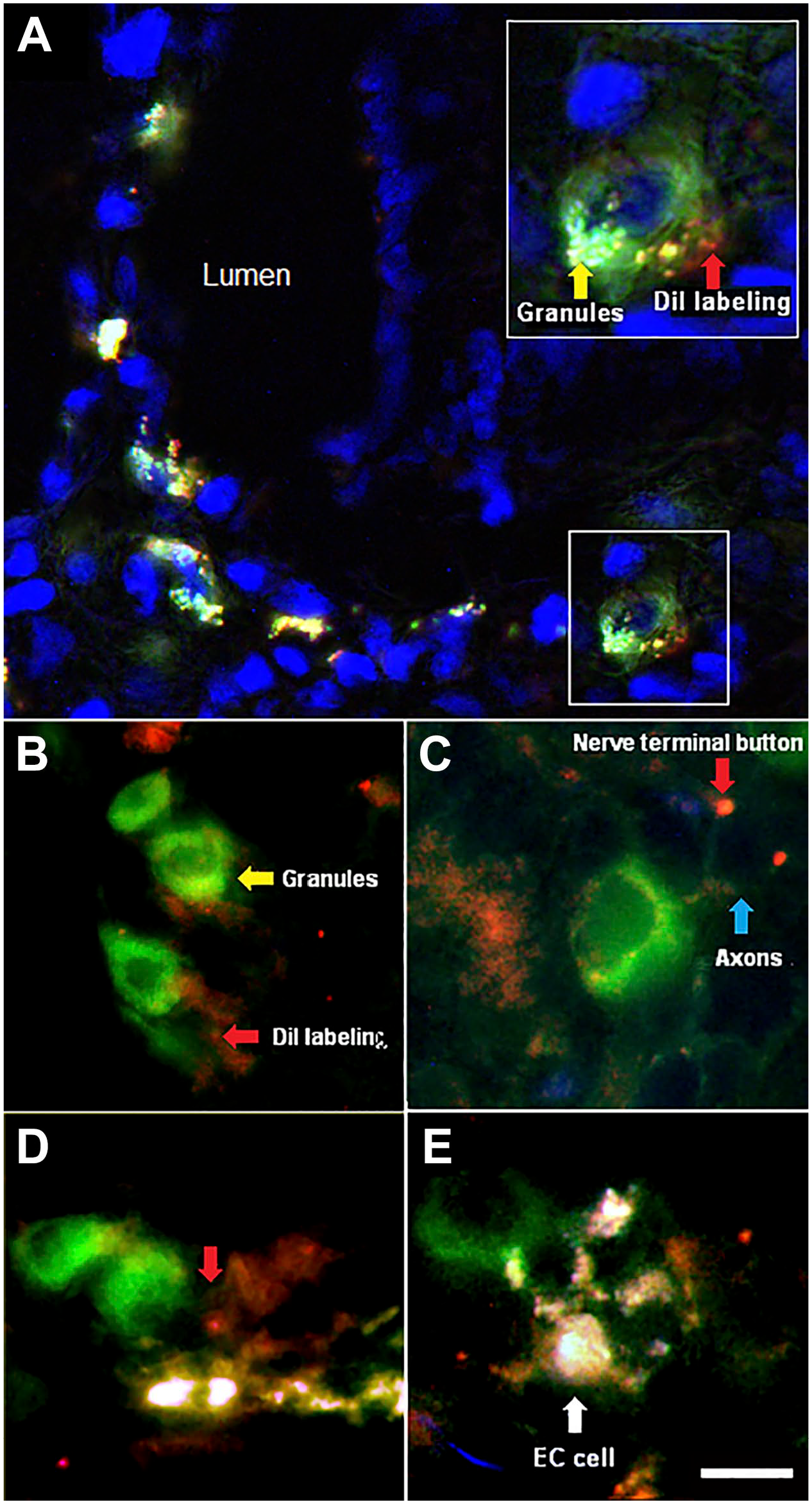
DiI retrograde labeling and NKAIN4 immunohistochemistry. The dye (red), placed on the para-arterial mesenteric nerve, reaches NKAIN4-positive cells (green) at the duodenal epithelium, demonstrating the direct connection between the enteric nervous plexuses and these epithelial cells. Image A shows a 25-µm-thick slice of the duodenal mucosa; the insert is a close-up of an NKAIN4-positive cell. Image B shows the contact between the NKAIN4-positive cell and sub-epithelial nerve endings. Image C presents a 50-µm-thick slice of the duodenal mucosa. NKAIN4-positive cells, containing small fluorescent granules, are reached by DiI-labeling. Image D displays the link between NKAIN4-positive cell and nerve endings through cell projections (red arrow). Image E demonstrates the direct contact between an NKAIN4-positive cell and an enterochromaffin cell, the highly granulate cell with pseudopods. Bars correspond to 16 µm (A, D), 1 µm (B), 10 µm (C), 8 µm (E), and 10 µm (insert in A). Abbreviation: DiI, 1,1′-Dioctadecyl-3,3,3,′3′-tetramethyl-indocarbocyanine perchlorate.
The confocal observation of the duodenal epithelium labeled with anti-NKAIN4 and anti-AChE antibodies showed a nerve terminal, intensively labeled by the anti-AChE antibody, contacting the NKAIN4-positive cell (Fig. 7), confirming that these epithelial cells receive projections from the sub-epithelial neural system.
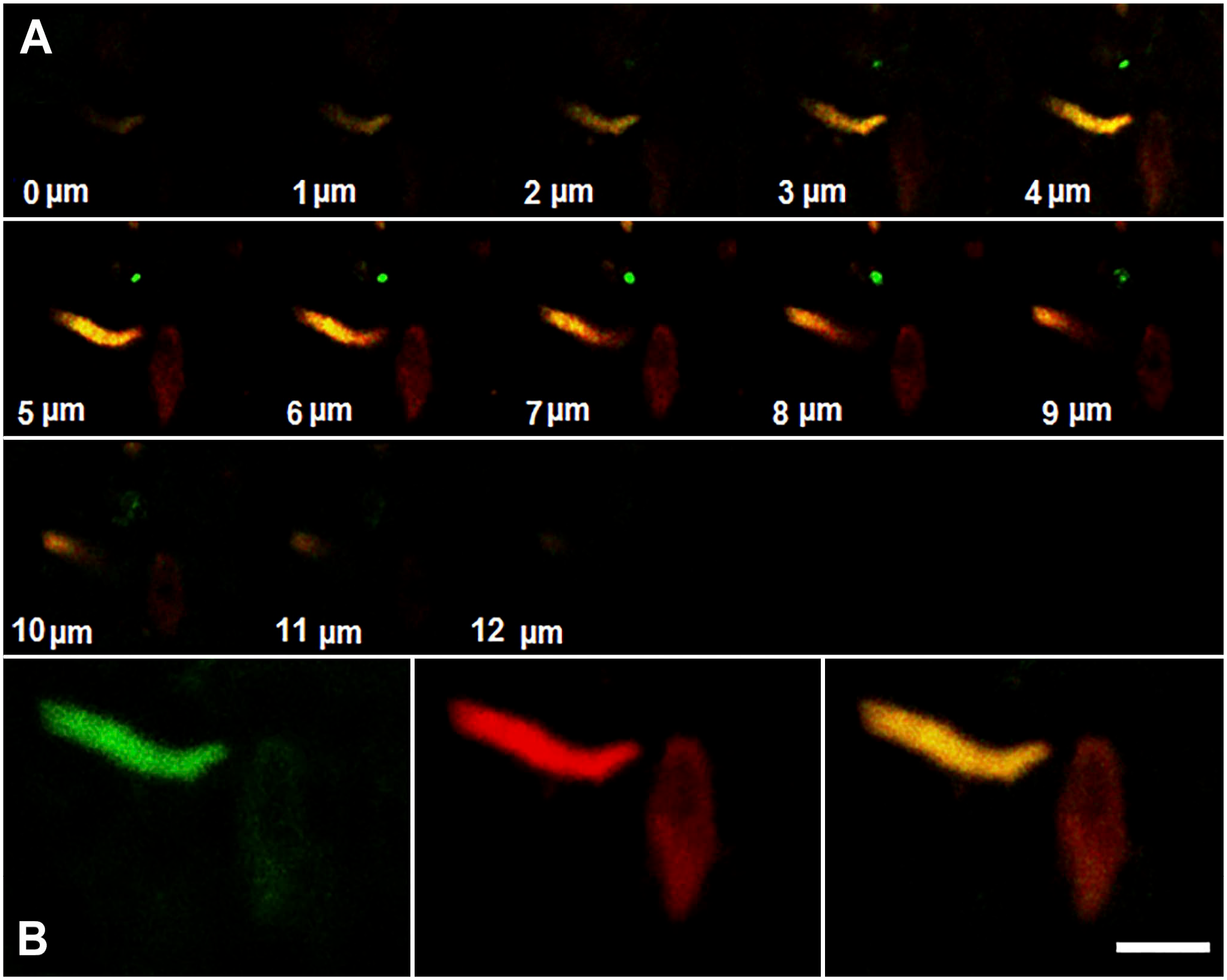
Enteric nerve terminals reach the NKAIN4-positive cell. A duodenal tissue section (12 µm) was labeled with anti-NKAIN4 (red) and anti-COX1 (green) antibodies and observed with a confocal microscope as indicated in the methods. The figure shows the confocal spatial stack (A) and their mounting (B). Bars correspond to 20 µm (A) and 10 µm (B).
NKAIN4-Positive Cells Contain Small Serotonin Granules Different From Those Present in Enterochromaffin Cells
Formaldehyde fixation induces fluorescence in red, green, and blue channels in biogenic amine-containing cells. Detergents (such as Triton X-100) eliminate this effect. In the present study, we used digitonin instead of Triton X-100 during the immunohistochemical procedure associated with DiI-labeling to preserve the structure of the chromaffin granules and to identify the NKAIN4-positive cells by immunofluorescence and 5-HT-containing granules by formaldehyde-induced fluorescence without interference.
In Fig. 8 (Panel I), image A shows an RGB image (corresponding to the simultaneous fluorescent emission in the red, green, and blue channels) of the preparation, which includes immunofluorescence for NKAIN4; image B presents a DIC microscopy of the evaluated duodenal mucosa, where some highly granulated cells were identified (cyan arrows); image C is a composite of DIC and RGB images. The highly granulated cells were negative for NKAIN4 but had a high white-blue fluorescence, indicating elevated biogenic amine content. The NKAIN4-positive cells (in green, red arrows) have smaller white-blue fluorescent granules suggesting a lower content of 5-HT. DiI-labeling is indicated by yellow arrows.
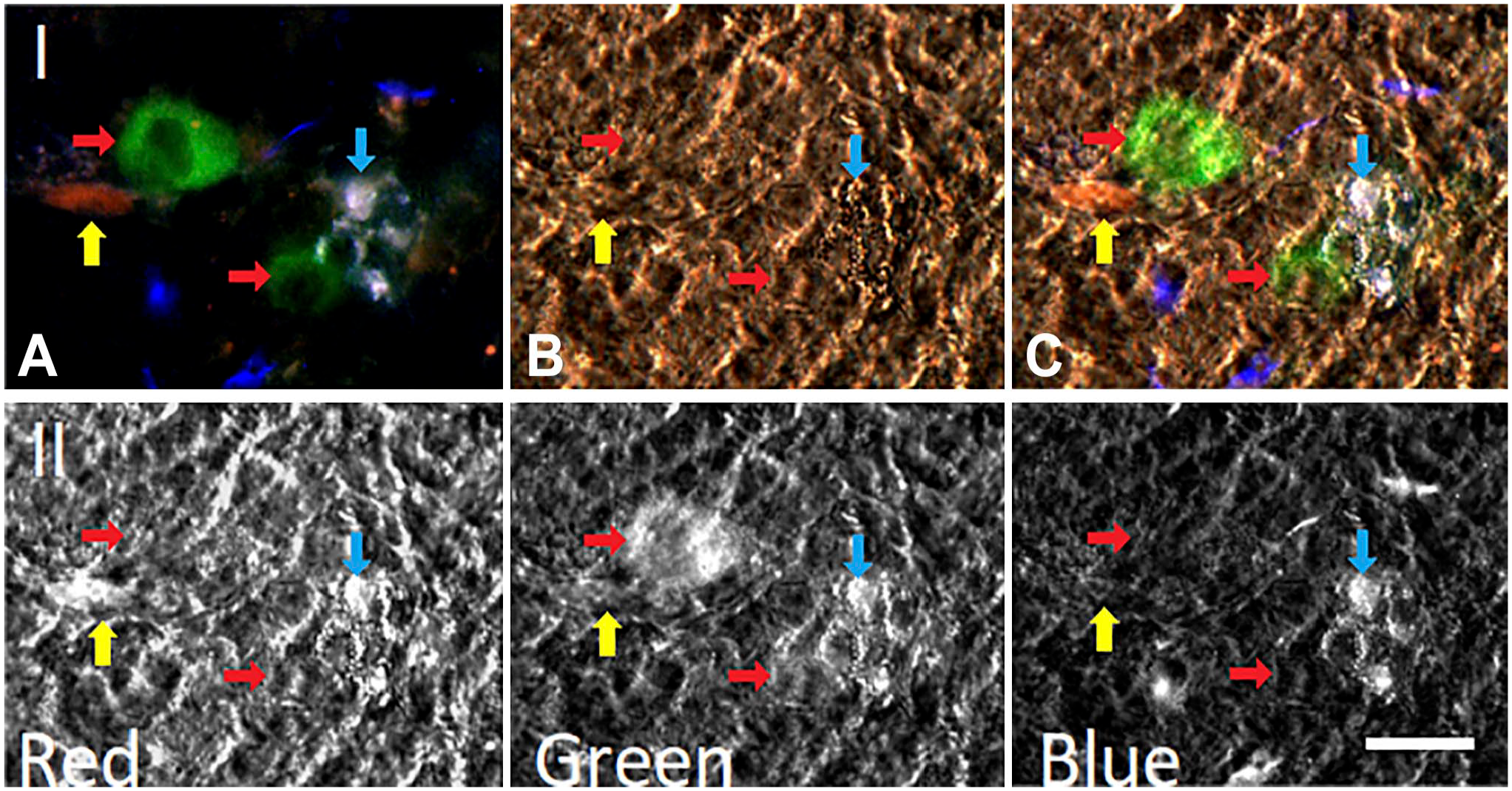
NKAIN4-positive cells contain small serotonin granules different from those present in enterochromaffin cells. Panel I. Image (A) shows a total fluorescence RGB picture of a 50-µm-thick section of the duodenal mucosa. The fluorescence of the highly granulate cells is white/blue (cyan arrows), while the NKAIN4-positive cells (green, red arrows) contain small white granules. The DiI-labeling is red and indicated by yellow arrows. Image (B) presents a differential interference contrast (DIC) register of (A). (C) This image is a composite of (A) and (B) images. Panel II. Formaldehyde-induced fluorescence of biogenic amines using red, green, and blue channels on the composite image (Panel I, C). Cells with pseudopods and large fluorescent granules are enterochromaffin (cyan arrows), and rounded cells with small fluorescent granules correspond to NKAIN4-positive cells (red arrows). Bar: 20 µm. Abbreviation: DiI, 1,1′-Dioctadecyl-3,3,3,′3′-tetramethyl-indocarbocyanine perchlorate.
In Panel II, we show the formaldehyde-induced fluorescence of biogenic amines using red, green, and blue filters on image C of Panel I in Fig. 8. Cells with pseudopods and large fluorescent granules are ECs (cyan arrow), while the rounded cells with small fluorescent granules are NKAIN4-positive cells (red arrow).
Finally, NKAIN4-positive cells (Fig. 9A) contain serotonin (Fig. 9B), as determined using the alkaline diazonium reaction for biogenic amines, mainly 5-HT.
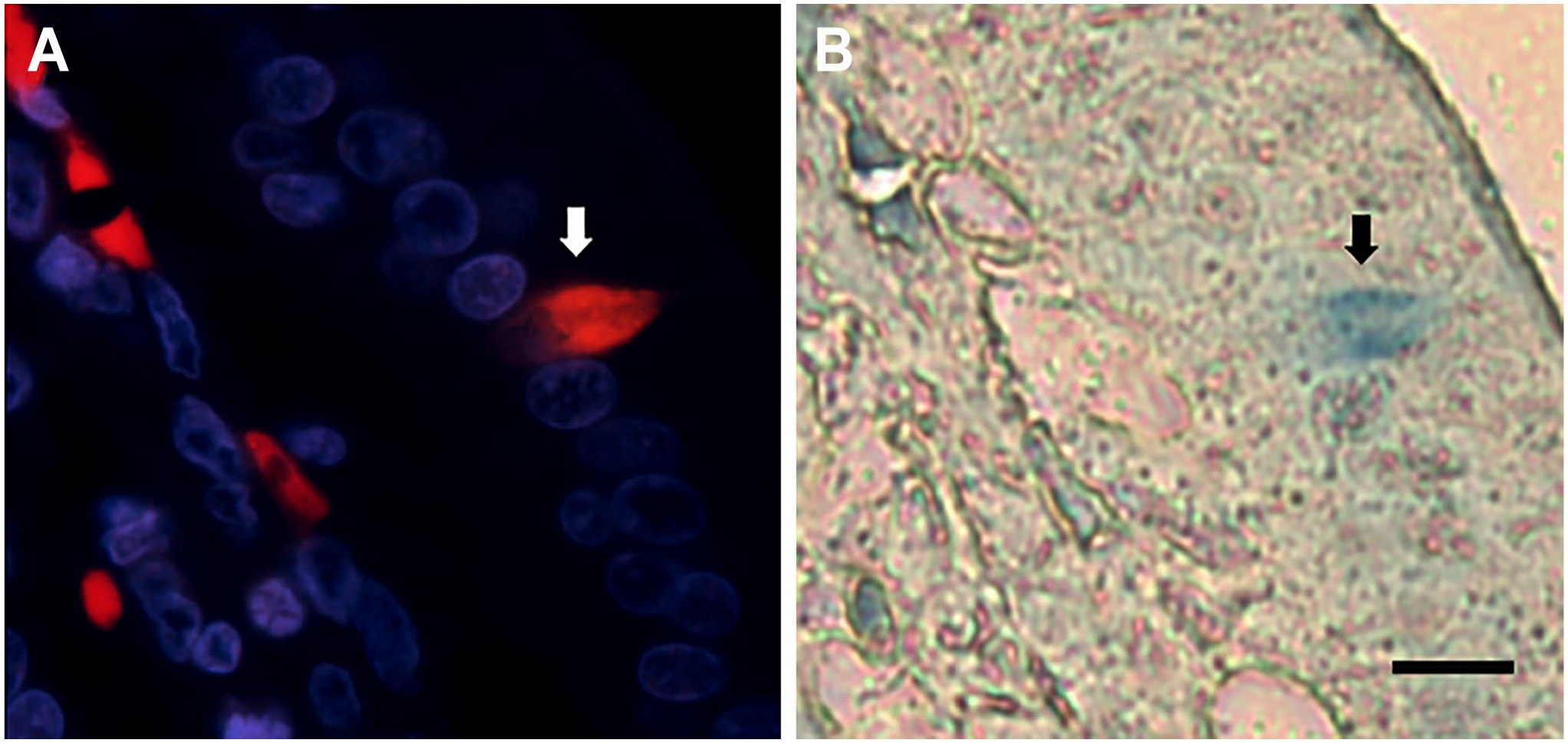
NKAIN4-positive cells contain serotonin. Image (A) shows a NKAIN4-positive cell (white arrow). Image (B) presents the same cell containing 5-HT, determined in the same preparation using the alkaline diazonium reaction for biogenic amines (black arrow). Bar: 10 µm.
The NKAIN4-Positive Cells and Their Neural Circuit (Glia and Fibers) Are Identified by Using Silver Impregnation and Methylene Blue Staining
We used additional strategies to characterize the NKAIN4-positive cells and determine their projections and connections with epithelial and sub-epithelial cells and structures. In this sense, we used dissected mucosa, thick sections (50–100 µm), and isolated villous-crypt units of the duodenal tissue stained either by silver impregnation techniques or a modified methylene blue staining, which permit the identification of neurons and their extensions. 33
Figure 10A shows isolated duodenal mucosa impregnated with silver nitrate, as described in the methods. We identified a large cell situated very close to the surface of the epithelium, with basal extensions into the sub-epithelial region, creating an intricate network of fibers contacting glial cells and sub-epithelial lymphoid nodules (Fig. 10B, arrows). According to its epithelial localization and morphology, this cell corresponds to the NKAIN4-positive cells described here. The fiber network extends to the villous surface, where it contacts glial cells. It was also possible to identify Shawn’s cells at the origin of the basal extension (Fig. 10C, arrow). Supplementary video 2 shows additional images of isolated duodenal mucosa impregnated with silver nitrate.
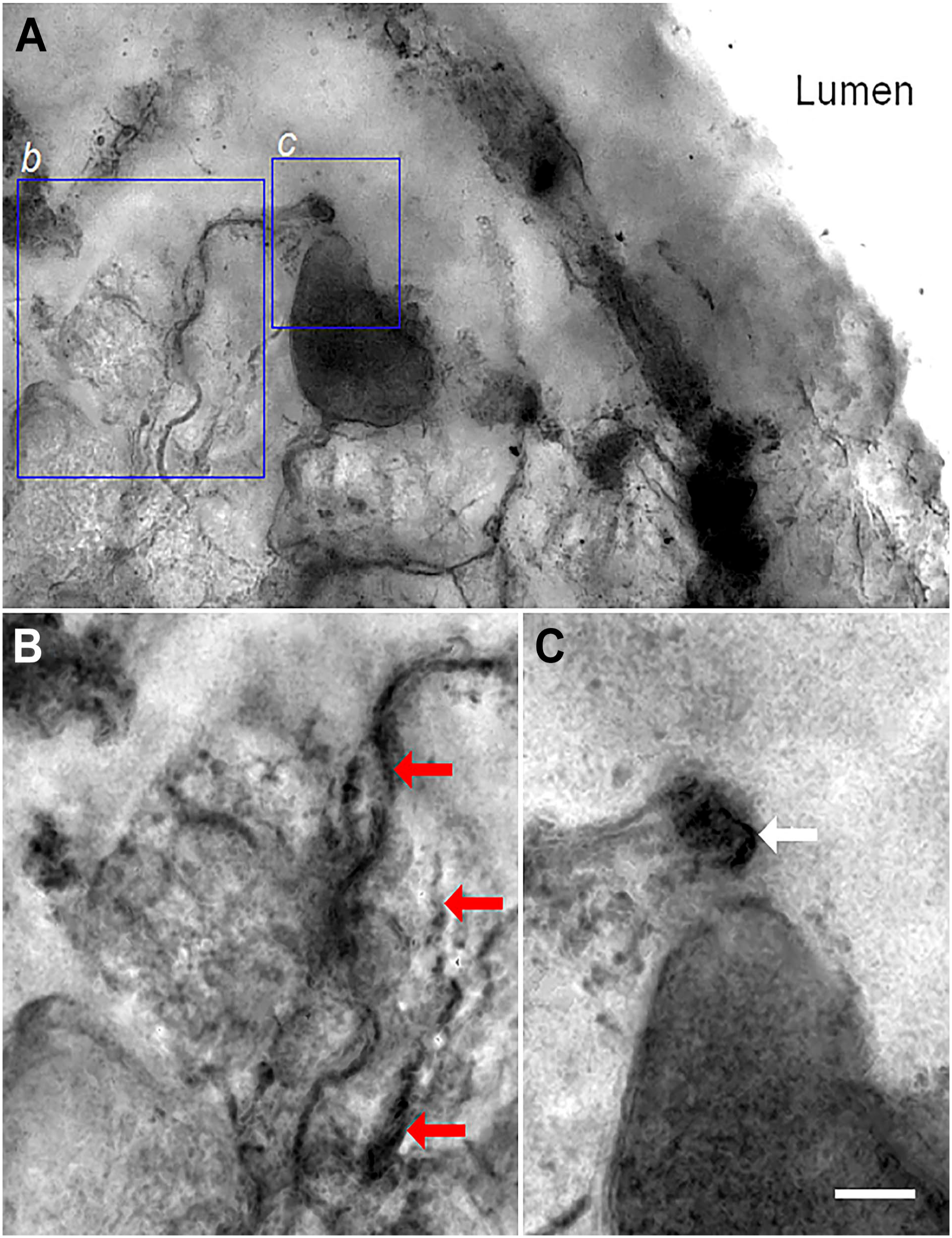
Silver nitrate impregnation of the isolated duodenal mucosa. Image (A) shows a complete image of the preparation, where we can observe the neuron body, its basal projections, and connections with neuroglia and sub-epithelial lymphoid nodules. Image (B) shows a close-up of the cell projections contacting a sub-epithelial lymphoid nodule, partially covered by nerve fibers and glial cells (red arrows). Image C presents oligodendrocytes at the base of a neuron cell basal extension (white arrow). Bars: 15 µm (A); 10 µm (B, C).
Figure 11A shows the immunofluorescence evaluation of NKAIN4 (in red) and glial fibrillar acid protein (GFAP) (in green), a specific glial marker. Glial cells (arrows) are close to the neuroepithelial cell in the duodenal mucosa. Figure 11B shows the immunofluorescent labeling of duodenal epithelium with 5-HTR7 (red), AChE (green), and DAPI (blue). These cells extend their projections toward the intestinal lumen forming with glial cells and fibers, a neural network on the epithelial surface.
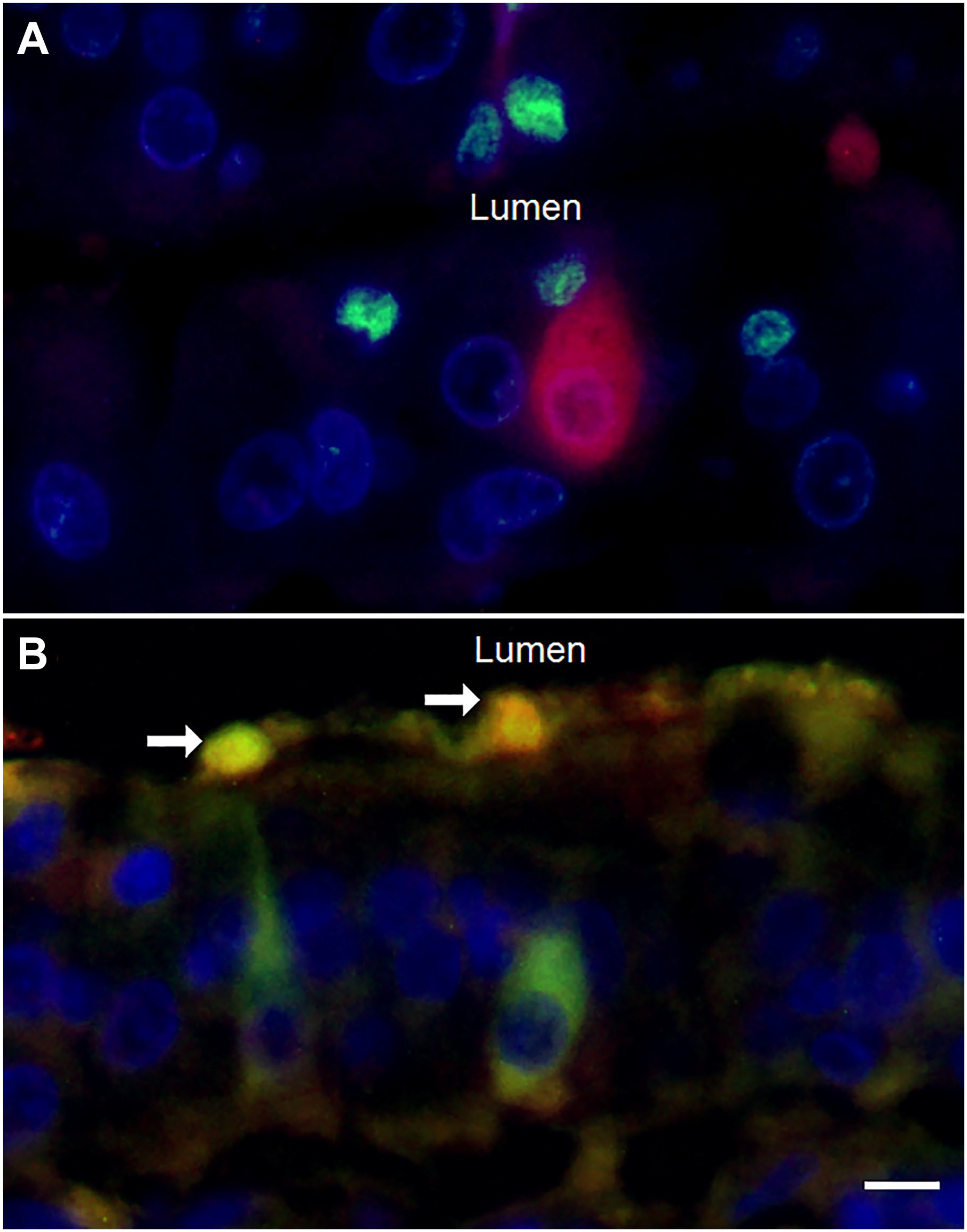
Neural fibers and neuroglia are on the epithelium surface. Image A shows glial cells labeled anti-GFAP (green) and NKAIN4-positive cells (red). Image B presents duodenal tissue labeled using anti-5-HTR7 (red) and anti-AChR-α3 (green) antibodies. The labeling identified the neurons (green) and two smaller cells (white arrows), probably neuroglia, located on the epithelial surface. Bar: 6 µm.
Results presented in Fig. 12 confirm these observations. The duodenal epithelium was labeled by immunofluorescence with the anti-NKAIN4 antibody (Fig. 12A) and simultaneously analyzed with DIC (Fig. 12B). The NKAIN4-positive cells (arrows) touch the epithelial surface through their apical extensions (arrowheads), reaching epithelial microvilli (Fig. 12C) and surrounding an EC (asterisk) on the epithelial surface (Fig. 12D).
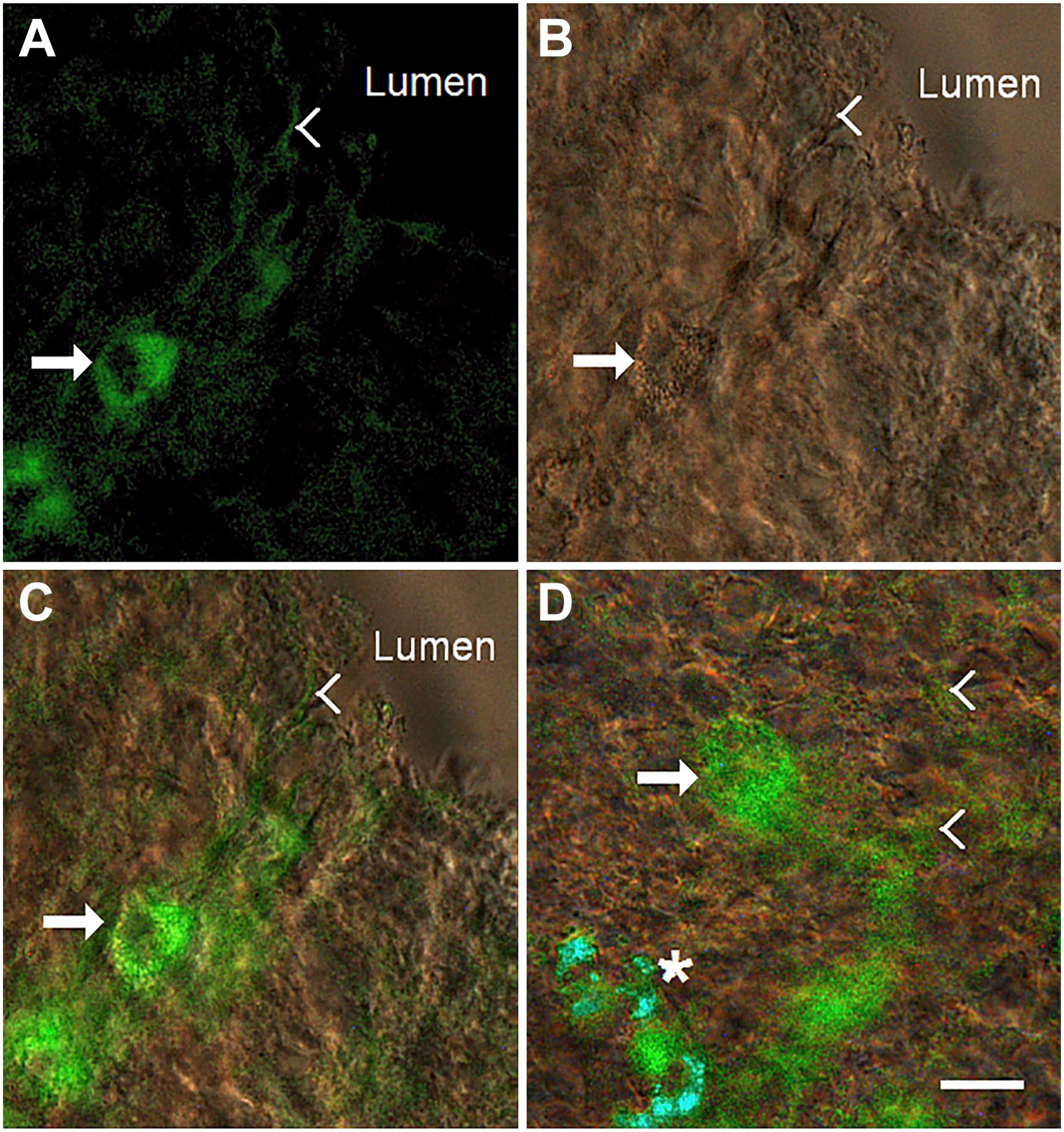
NKAIN4-positive cells and their network. The isolated duodenal epithelium was labeled with an anti-NKAIN4 antibody (image A) and observed with differential interference contrast (DIC, image B). Image C presents a composite of (A) and (B). Image D shows an area where NKAIN4-positive cell extensions surround an enterochromaffin cell (asterisk). In all images, arrows indicate the NKAIN4-positive cell bodies, and arrowheads, their projections. Bar: 10 µm.
The methylene blue staining and DIC allowed the observation of this neural network at the epithelial surface. This neural network forms patches in the duodenal epithelium (Fig. 13A). Nerve fibers elapse between epithelial cells, presenting small nodes and contacting glial cells on the epithelium (Fig. 13B). Image C shows an NKAIN4-positive cell and its web on the epithelial surface. Image D is a close-up of image C.
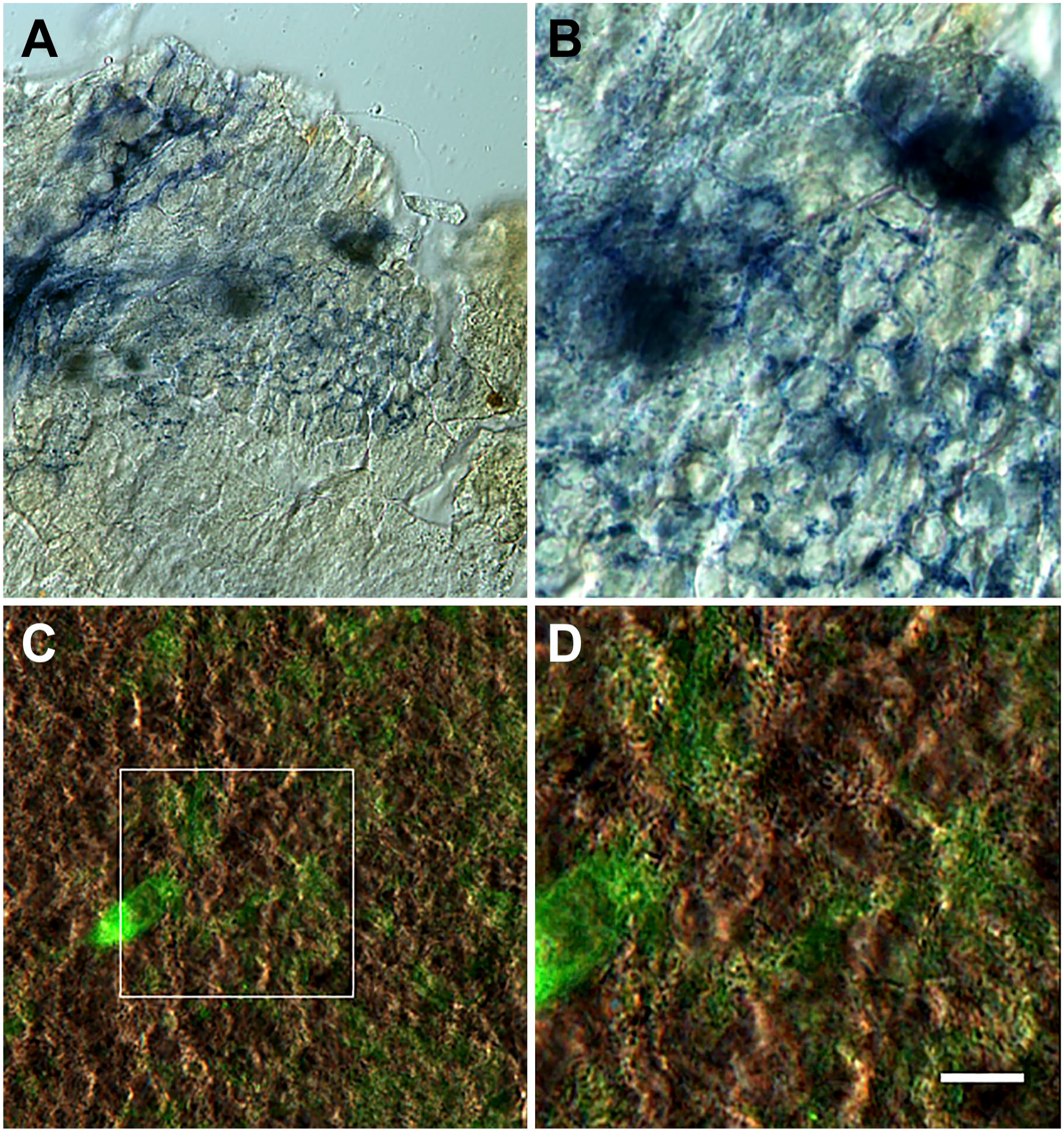
Neural network on the duodenal epithelium. The duodenal epithelium was stained with methylene blue and observed with differential interference contrast (DIC). The neuron, glia, and neural fibers constitute a system organized in patches in the mucosa (A). This system extends toward the epithelium surface (amplified in B). Image C shows NKAIN4-positive neurons, and their extensions form a network that elapses between the epithelial cells. Image (D) is an amplification of (C). Bars correspond to 90 µm (A), 15 µm (B, C), and 6 µm (D).
Morphometry
There are some morphometric aspects that we would like to highlight. NKAIN4-positive cells have a rounded cell body with a thin projection toward the intestinal lumen, which ends in a knob-like structure. They are much wider at the base and present basal extensions toward nerve endings and other sub-mucosal elements. The NKAIN4-positive cells measure 21.1 ± 2.90 µm in height × 13.7 ± 2.96 µm in width and have a nuclear area of 38.7 ± 9.76 µm2, a cytoplasm area of 186.6 ± 36.50 µm2, and a nucleus/cytoplasm ratio of 0.21 ± 0.018. On the other hand, enterocytes measure 32.7 ± 6.29 µm in height × 6.8 ± 0.86 µm in width and have a nuclear area of 42.4 ± 4.30 µm2, a cytoplasm area of 110.3 ± 14.36 µm2, and a nucleus/cytoplasm ratio of 0.39 ± 0.059. Differences in height, width, cytoplasm area, and nucleus/cytoplasm ratio were significant (p<0.001, n=44) compared with enterocytes.
The concurrent utilization of the method proposed by Kaneko et al. 19 to identify amine-containing cells (including the NKAIN4-positive cells) and immunofluorescence for NKAIN4 allowed the quantification of both cell types in the same sample. Thus, we identified 51 ± 3.4 ECs per 104 µm2 of epithelial area, of which 20 ± 1.4 were NKAIN4-positive cells (39%). Intestinal ECs are 1.98 ± 0.43% (mean ± SD, n=22 different slices) of epithelial cells in the duodenum, while NKAIN4-positive cells represent 0.77 ± 0.17%.
Ach Activates the NKAIN4-Positive Cell
As previously demonstrated, the NKAIN4-positive epithelial cells expressed neuronal markers such as NeuN, ChAT, AChE, GAD-65, AChR-α3, and AChR-α7, suggesting that they are cholinergic neurons, so they must respond to Ach. To evaluate this possibility, we designed an experimental protocol that included duodenal villous-crypt unit isolation, cell attach, neutral red loading, perfusion in an inverted chamber, and monitoring epifluorescence. Under these conditions, we evaluated the effect of ACh on neutral red fluorescent emission. Figure 14 shows the results as pseudo-colored pictures. Original images rely on an eight-bit grayscale. ACh induced a propagating increase of fluorescence intensity from the center to the extremes of the cell during 300 msec (Fig. 14A). We also identified the activation of NKAIN4-positive cells by using Ca2+-dynamic imaging. Figure 14B presents the effect of ACh on in[Ca2+] in isolated villous-crypt units. Most epithelial cells have low in[Ca2+]. The ACh (100 µM) transiently increases in[Ca2+] in the NKAIN4-positive cell and its extensions for 1–6 sec.
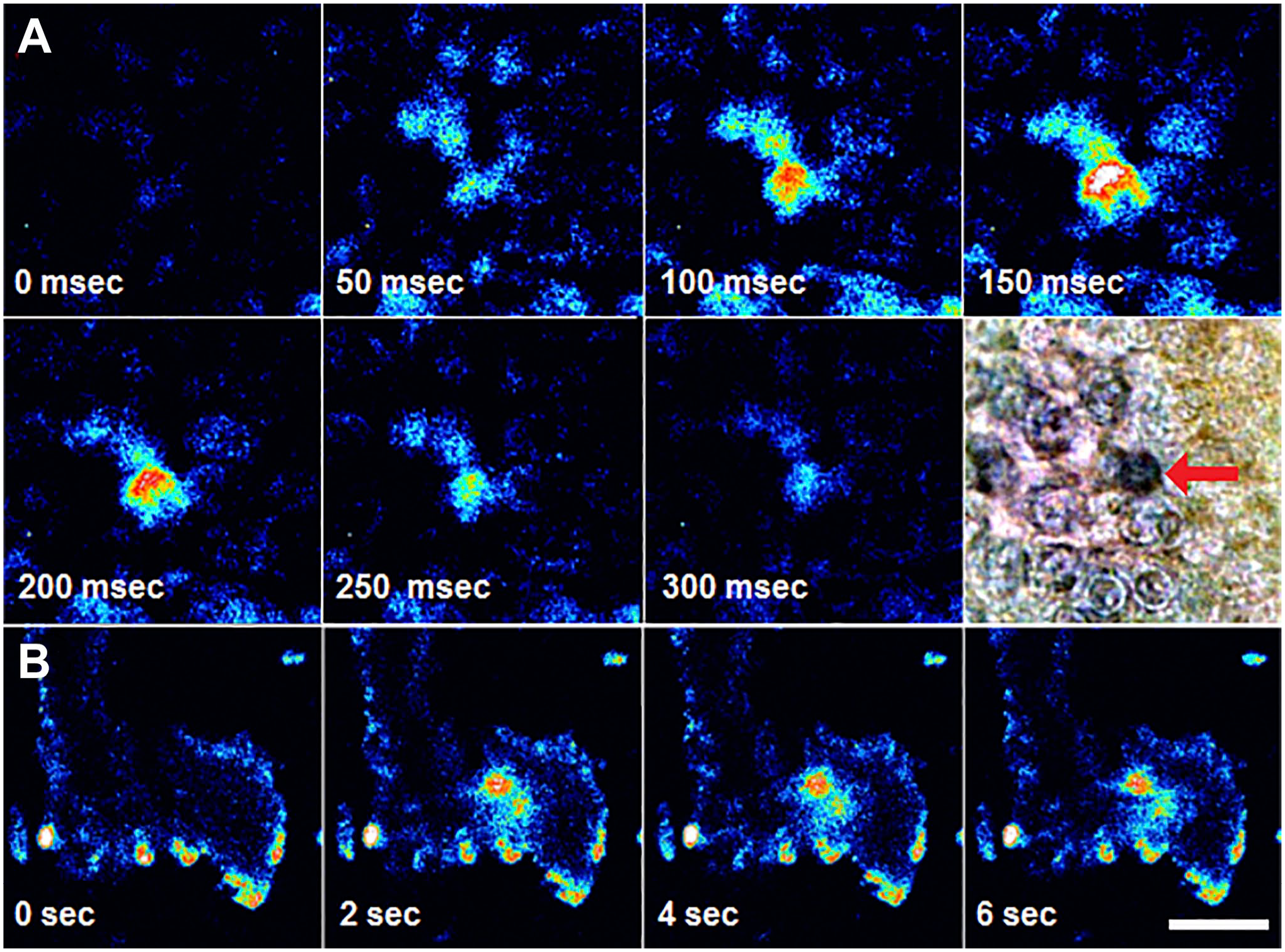
Acetylcholine activates the NKAIN4-positive cells. (A) Isolated villous-crypt units of duodenal mucosa loaded with neutral red (10 mM) were mounted on an epifluorescence microscope and continuously perfused. Once fluorescent emission stabilized, we added acetylcholine (100 µM) to the perfusion medium, and the signal followed for 300 msec. Images collected every 50 msec were analyzed and processed to generate pseudo-colors. At the end of the activation experiments, the sample was fixed with cold acetone and processed for immunostaining using anti-NKAIN4 antibody and peroxidase (results shown in the last image of this panel). The activated cell corresponds to the NKAIN4-positive cell (red arrow). (B) Isolated villous-crypt units of duodenal mucosa, loaded with Fluo-4 AM (10 µM), were mounted on a perfusion chamber (300 µl volume), continuously perfused, and observed in a confocal microscope. Once fluorescent emission stabilized, we added acetylcholine (100 µM) to the perfusion medium, and the signal was registered every 2 sec and processed to generate pseudo-colors pictures. Bar: for A and B, 60 µm.
Figure 15 presents isolated villous-crypt units loaded with Fluo-4 AM at a lower magnification. As indicated, most epithelial cells have low in[Ca2+]. Some cells located toward the center and edges of the preparation showed a slightly higher intracellular calcium level, and a few others, placed at the villous-crypt unit borders, presented a high in[Ca2+]. These last cells were identified as enterochromaffin by fluorescence induced by paraformaldehyde. ACh (100 µM) significantly increase in[Ca2+] in the NKAIN4-positive cells (white arrow) and their extensions (yellow arrow), outlining the conductive pathways and glial cells (green arrow) that carry the signal to ECs (red arrow). The in[Ca2+] returns to their initial levels in approximately 30 sec. By contrast, ACh did not modify the fluorescence intensity in ECs during the first 1–5 sec but induced intracellular calcium redistribution, and then an in[Ca2+] decrease was observed in these cells (30–60 sec). This effect is probably due to intracellular Ca2+ mobilization associated with serotonin release, induced by ACh and mediated by NKAIN4-positive cells.
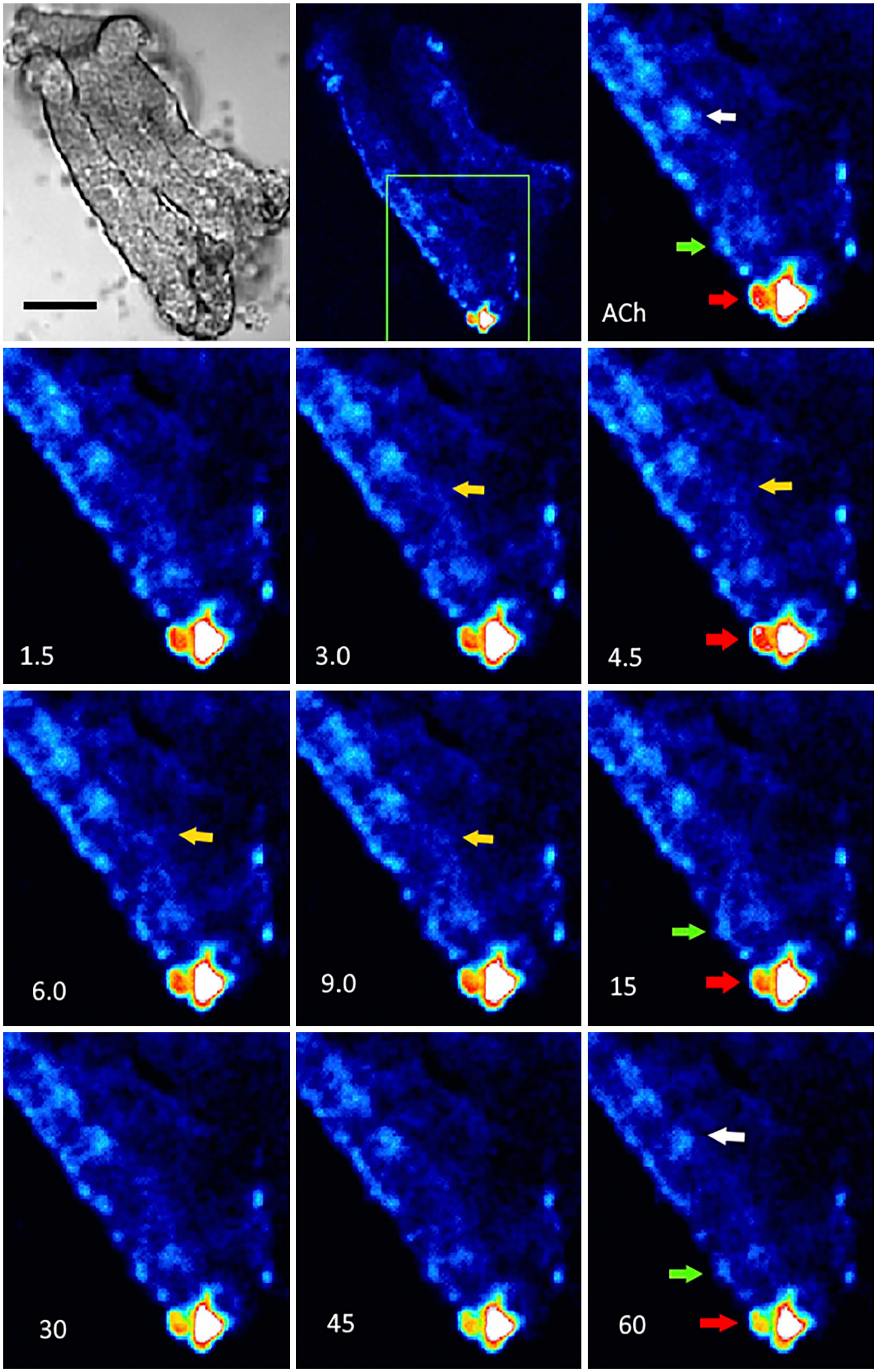
Confocal imaging of intracellular calcium increase induced by acetylcholine. Villous-crypt units were loaded with Fluo-4 AM, mounted on a perfusion chamber, and observed with a confocal microscope under continuous perfusion. Series, obtained every 1.5 sec at 488 nm, were analyzed and processed to generate pseudo-color images. ACh (100 µM) transiently activated the NKAIN4-positive cells (white arrows), their extensions (yellow arrows), and glial cells (green arrows) during the first seconds. In enterochromaffin cells (red arrows), the fluorescence signal was intracellularly re-distributed during the first few seconds and then continually decreased after ACh addition. The first image shows a transmission picture of the villous-crypt preparation. Bar: transmission and first fluorescence figures, 50 µm, and for the rest, 40 µm.
We have shown that NKAIN4-positive cells express serotonin receptors (5-HTR7), which could regulate the activity of these cells. Thus, we tested the effect of serotonin on in[Ca2+] in villous-crypt units (Fig. 16). Serotonin (50–500 µM) does not affect in[Ca2+] in NKAIN4-positive nor ECs. ACh (100 µM) did not change intracellular calcium in cells previously exposed to serotonin. Afterward, the preparation was extensively washed with Dulbecco medium to eliminate both compounds. Then, we re-evaluated the ACh effect. As expected, the neurotransmitter increased the in[Ca2+] in an NKAIN4-positive cell and induced Ca2+, and probably serotonin, release from ECs. These results suggest a potential negative feedback mechanism between ACh and serotonin mediated by the NKAIN4-positive cell.
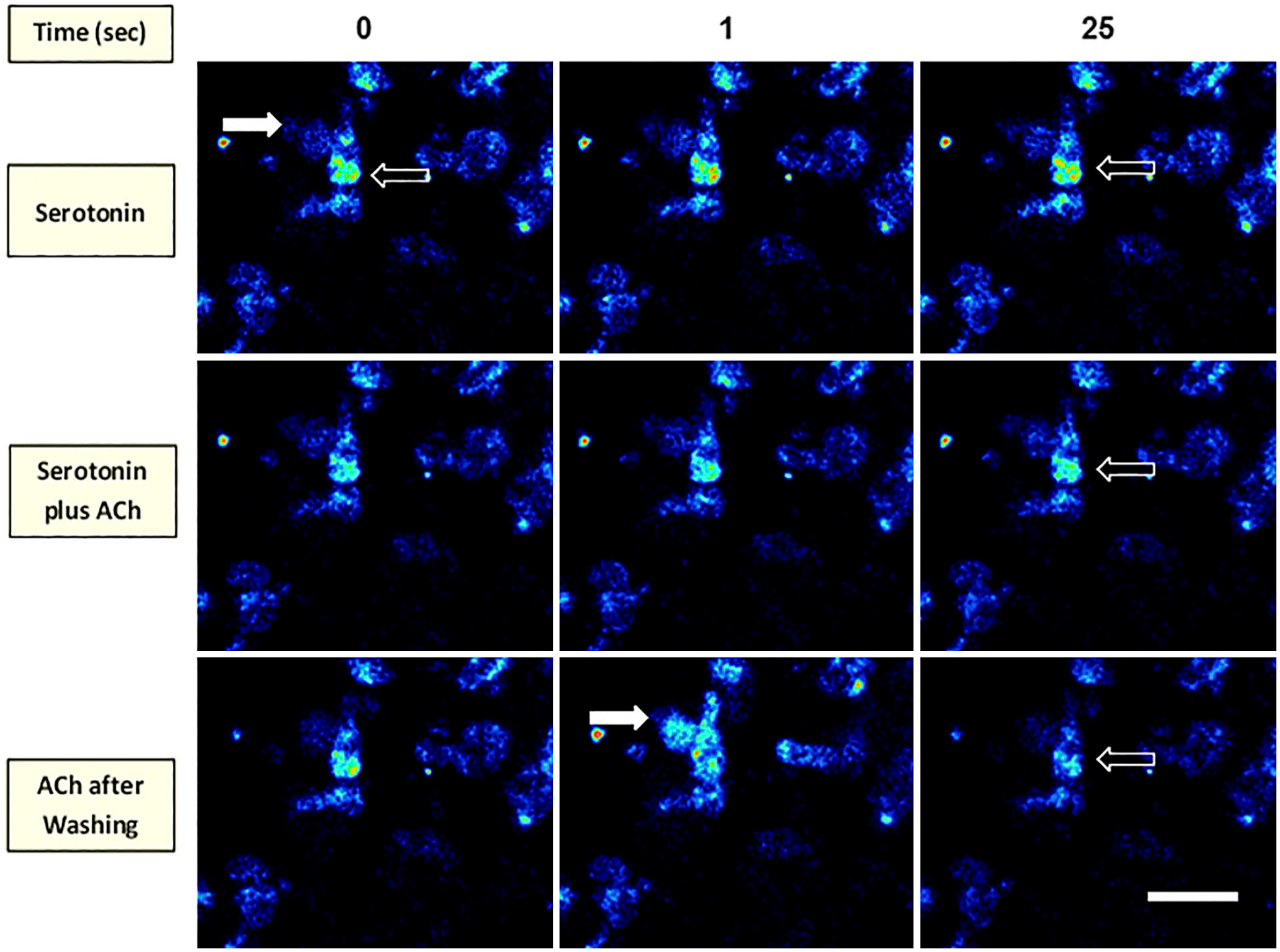
Serotonin inhibits the acetylcholine effect. The figure shows confocal series of isolated villous-crypt units loaded with Fluo-4 AM and sequentially incubated with serotonin (100 µM) or serotonin (100 µM) plus ACh (100 µM) at 0, 1, and 25 sec after additions. Finally, after extensive washing with Dulbecco medium, we added only ACh (100 µM) for 25 additional seconds. Open arrows show the NKAIN4-positive cell body, and white arrows indicate the neuronal network. Bar corresponds to 60 µm.
Discussion
The intestinal epithelium is a barrier between the luminal milieu and nerve endings of the lamina propria. These nerve terminals do not penetrate the epithelium. In addition, there is no evidence of neuron within it. Therefore, luminal substances can only influence the terminal nerve endings if they induce a signal from epithelial cells that affect the nerve terminals or if the luminal compounds cross the epithelial barrier and directly stimulate these endings.
Intestinal secretomotor and other reflexes induced by mechanical or chemical luminal stimuli activate the ENS. Thus, mucosal chemical or mechanical stimuli induce IPANs’ response.46,47 The communication between the epithelium and the ENS has been considered indirect through mediator secretion and not by direct cell–nerve contact. Mediators would act on endings of sub-mucosal IPANs initiating action potentials that activate the enteric nervous circuit. There is evidence that 5-HT release, induced by changes in the chemical contents of the intestinal lumen or by mucosal stretch, is a potent stimulant of IPAN cell endings.46,48–50
How the intestine transmits sensory information from the epithelial barrier to the brain has been intriguing for decades. An essential step in this mechanism is the communication between the luminal space and the ENS. It is generally accepted that the gut and the brain exchange sensory information only through hormones that act on the sub-epithelial nerve terminals. 8 However, an efficient communication network through nervous structures, including sensory and amplification elements, is necessary for rapid information exchange between the gut and the CNS. Neuron and nerve endings identification in the intestinal epithelium has been elusive. Newson et al. 51 showed sensory neurons in the intestinal mucosae although the evidence was non-conclusive; Lundberg et al. 52 showed innervation of ECs in the intestine of guinea pigs.Recently Bohórquez et al. 53 identified synaptic links between epithelial sensory cells and nerves in the colon, using co-culture of cholecystokinin-green fluorescent protein (CCK-GFP) cells purified by cell sorting and sensory neurons dissociated from sensory ganglia by enzymatic digestion. Furthermore, they reported intestinal EECs with cytoplasmic processes that they named neuropods, providing a putative direct connection between EECs and neurons innervating the small intestine and colon.8,54
The NKAIN is a protein family of unknown function whose members are in the neurons of mouse encephalon, 9 including the cerebral cortex, thalamus, hippocampus, olfactory bulb, brainstem, and cerebellum, with high expression in the granular cell layer. The family has four splicing variants (NKAIN 1–4). The heart, lung, spleen, and liver also express NKAIN4, as determined by RT-PCR. There is evidence that the NKAIN1 interacts with the β1-subunit of the Na+/K+-ATPase. Because only the splicing variant-4 was expressed in non-brain tissues, we decided to evaluate its occurrence and functional relationship with the Na+/K+-ATPase in the gastrointestinal tract. During these studies, we identified an epithelial cell in the middle segment and the base of the duodenal villi, which expresses NKAIN4. These cells also overexpress the α1-subunit of the Na+/K+-ATPase and the K+-independent, ouabain-insensitive Na+-ATPase, suggesting their high capacity to mobilize sodium (Fig. 1). They represent 0.77% of the epithelial cells. The expression of the neuronal protein NKAIN4 and the overexpression of the Na+-transporting enzymes in this epithelial cell suggest its possible neuronal origin.
Enteric neurons exhibit a remarkable diversity at molecular, morphological, physiological, connectional, and functional levels. The precise definition and classification of a cell as a neuron needs to consider and integrate these different characteristics. In the guinea pig, the profile of functionally identified neurons has been defined by their shape, protein markers, the relationship with the structures they innervate, the chemical properties of the transmitter that the neuron utilizes, and their projections to different targets. 2 We used these criteria to identify the NKAIN4-positive cells as enteric neurons.
Evaluation of the Presence of Neuronal Markers in the NKAIN4-Positive Cells
We determined, by immunohistochemistry, the expression of several neuronal markers (NeuN, ChAT, AChE, AChR-α3, AChR-α7, GAD-65, and 5-HTR7) in the NKAIN4-positive cells (Fig. 2).
NeuN (neuronal nuclear protein) is used as a marker of post-mitotic neurons and is involved in mRNA splicing regulation. This marker is not in tissues other than nervous ones. Moreover, NeuN has never been identified in glial cells, which suggests it is a specific neuronal marker. 55 The neuronal nuclear protein occurs in both the nucleus and cytoplasm and is specific to IPANs, labeling the entire population of these neurons in the guinea pig ileum. 56 In addition, Nassauw et al. 57 demonstrate that cytoplasmic expression of NeuN is an exclusive feature of IPANs in the guinea pig gastrointestinal tract.
Choline-acetyl-transferase (ChAT) catalyzes the biosynthesis of the neurotransmitter acetylcholine. The vast majority of cholinergic neurons express choline acetyltransferase. There are at least two splicing variants for ChAT; cChAT, mainly expressed in CNS, and pChAT, predominantly expressed in the peripheral nervous system. 58 Using immunohistochemistry, cChAT has been widely used to identify cholinergic neurons of the CNS; however, it can recognize the choline-acetyl transferase of the PNS in guinea pigs and humans.59,60
Acetylcholine, released by cholinergic neurons, is the principal excitatory neurotransmitter in the ENS, regulating motility and mucosal function in the guinea pig’s small bowel. It activates two groups of AChRs; muscarinic AChRs (mAChRs) relate to the superfamily of G-protein-coupled receptors and nicotinic AChRs (nAChRs) part of the ligand-gated ion channel superfamily. The nAChRs consist of α-subunits (1–10 subtypes), which are responsible for the binding of ligands, and other subunits (β 1–4 subtypes, γ, δ, and ε). An active nAChR is generally a heteropentamer of these various subunits organized around a central pore. The nAChR receptors conduct Na+, K+, and Ca2+ when activated. 61
Neuronal cells have all α-subunit types except the α1-subunit, specifically expressed in muscles. Nicotinic receptors are prominent in the peripheral autonomic nervous system and mediate fast synaptic transmission in all peripheral autonomic ganglia.62,63 Moreover, a subpopulation of serotonin-containing cells in the upper guinea pig small intestine expresses nAChRs. 64 In the peripheral nervous system, the AChR-α3 mediates excitatory neurotransmission at virtually all autonomic ganglia, the adrenal gland, and the sensory ganglia. 65 The α7-subtype is the principal contributor to the α-bungarotoxin-binding sites in either brain or the peripheral nervous system. ACh receptors that contain the α7-subunit, as a homomeric or heteromeric receptor, most commonly bind α-bungarotoxin and have relatively low affinity for nicotine and fast kinetics.66–68 The NKAIN4-positive cell binds α-bungarotoxin, as shown in Fig. 3, confirming ACh-receptor’s presence in these cells.
AChE hydrolyzes acetylcholine at neuromuscular junctions and brain cholinergic synapses, thus terminating the signal transmission. AChE’s expression is necessary to recognize a cholinergic neuron. AChE histochemistry is used to localize cholinergic structures in nervous systems. Double staining of ChAT and AChE indicated that every ChAT immunoreactive neuron was also positive for AChE. However, not all AChE-positive ganglionic neurons express ChAT in the ENS of rats and guinea pigs. 69 Thullbery et al. 70 studied acetylcholinesterase expression in neuronal and non-neuronal cell lines at the transcriptional and translational levels. The presence of AChE protein does not correlate with catalytic activity, but the diffuse cytoplasmic and plasma membrane localization of AChE, such as in the NKAIN4-positive cells, is a property of neuronal cell types.
Another neuronal marker of particular interest is glutamic acid decarboxylase (GAD). This neuronal enzyme catalyzes the synthesis of γ-aminobutyric acid (GABA, an inhibitory neurotransmitter) by decarboxylation of glutamic acid. There are two homologous isoforms named GAD65 and GAD67, by their molecular mass (65 and 67 kDa, respectively). GAD67 is a cytosolic protein, while GAD65 is a membrane-associated protein. 71 GABA and GABA receptors have been identified along the gastrointestinal tract 72 and implicated in immune-mediated inflammatory processes.73,74
The 5-HTR7 (serotonin receptor-7), from the 5-HT receptor family, is coupled to adenylyl cyclase. The central and peripheral nervous system and the gastrointestinal tract expressed 5-HTR7 mRNA. Also, neurons of the sub-mucosal plexus of the guinea pig ileum have 5-HTR7. 75
The coexpression of NeuN, ChAT, AChE, AChR-α3, AChR-α7, GAD-65, and 5-HTR7 with NKAIN4, besides the binding of α-bungarotoxin (Fig. 3), defines the neural origin of the epithelial NKAIN4-positive cells. The presence of receptors for Ach (AChR-α3 and AChR-α7) and 5-HT (5-HTR7) could be part of an auto-regulatory mechanism for these neuronal mediators.
Shape and Other Structural Characteristics of the NKAIN4-Positive Cells
Recently, Heiman 76 reviewed some morphological and functional characteristics of sensory neurons that exhibit hallmarks of epithelial cells. These cells use tight junctions to form a continuous diffusion barrier within the epithelia and exhibit apical-basal polarity, with an outward-facing apical surface morphologically, biochemically, and functionally distinct from their inward-facing basolateral surface. They are neurons with axon-dendrite polarity (such as the olfactory receptor neurons and photoreceptors).
The duodenal NKAIN-positive cell has morphological characteristics of sensory neurons. It is a polarized cell with a thin and long apical process that finishes in a knob-like terminal protrusion in contact with the luminal space (Fig. 5C). The receptor neuron of the olfactory epithelium possesses an analogous structure. 77 In addition, we identified at the base of the apical extension of the NKAIN4-positive cell, in the supra-nuclear region, nodular elements rich in γ-tubulin (Fig. 5E). Similar structures have been identified as primary cilium and basal bodies in some sensory neurons. Primary cilia are non-motile organelles based on a microtubule structure present in neurons of many sensory systems.42–45 These cellular elements could be part of a sensory apparatus in this epithelial cell. Furthermore, the NKAIN4-positive cell presents, at the basal region, projections toward the lamina propria that contact nerve terminals of the ENS (Fig. 5A, B and E).
Silver impregnation and methylene blue staining are used in neuroscience to describe neurons, glia, and neuronal connections in the central and peripheral nervous systems. Evaluation of villous-crypt units, thick tissue sections, and isolated mucosa impregnated with silver or stained with methylene blue allowed us to identify some morphological characteristics of the NKAIN4-positive cell and its extensions in the duodenal epithelium. These cells send projections toward the epithelial surface and in the direction of the lamina propria. The apical and basal extensions connect with the neuroglia, forming a neural network in particular areas of the epithelial surface (Fig. 13) and the sub-epithelial region of the mucosa (Figs. 10 and 11). There, it contacts lymphoid nodules and ECs. The network distribution formed by the neuron cells, glial cells, and neural fibers is non-aleatory. The complex forms patches in the epithelium, frequently surrounded by ECs (Figs. 12 and 13). Enteric neuroglia is an essential homeostatic cell in the intestine. Active signaling mechanisms between enteric glia and neurons modulate gastrointestinal functions (enteric reflexes and communication with intrinsic and extrinsic neurons). Moreover, the intestinal glial cells interact with immune cells to maintain intestinal immune homeostasis. 78
Connection With the ENS
The lipophilic, long-chain carbocyanine dyes (e.g., Dil) have been used in ex vivo studies to identify neural pathways.79,80 Upon application to fixed tissues, the tracer diffuses through the plasma membranes of the neurons, 81 allowing us to follow the anterograde (away from the cell body), retrograde (toward the cell body), or bidirectional pathways. In the plasma membrane, the tracer moves bidirectionally and even to adjacent cells through the aqueous medium, although much more slowly due to its lipophilic nature. The fluorescent carbocyanine dyes permit visualization of the morphology and connectivity along a desired axonal pathway.
The retrograde labeling demonstrated that the NKAIN4-positive cells directly connect with the ENS. DiI placed on the para-arterial mesenteric nerve reached the NKAIN4-positive cells at the duodenal epithelium (Fig. 6). Furthermore, we observed the contact between NKAIN4-positive cell projections and nerve terminals (Dil-positive) at the mucosal stroma and nerve fiber bundles (Dil-positive) reaching the NKAIN4-positive cells. These results confirm the direct connection between the ENS and these epithelial cells. In addition, we showed (Fig. 7), using immunofluorescent labeling for NKAIN4, AChE, and confocal microscopy, a nerve terminal reaching the NKAIN4-positive cell, confirming the connection of these cells with ENS plexuses.
Comparison With Other Epithelial Cells
The frequency and shape of the NKAIN4-positive cells within the duodenal epithelium are similar to those of other epithelial cells described in the intestinal mucosa, such as tuft, cup, or ECs. The tuft cells, identified by doublecortin-like kinase-1 as a marker, 82 represent approximately 0.5% of epithelial cells in the murine small intestine. 83 Intestinal tuft cells have an oval body that narrows at the apical and basal ends.84,85 They have large and dense apical brush borders and lateral membrane projections. 86 These cells have been identified in the gastrointestinal tract from the stomach to the rectum, while the NKAIN4-positive cells are only in the duodenal mucosa. The tuft cells have broad heterogeneity, including serotonin-containing cells. 87 These cells would be a new secretory cell type. 40 Tuft cells contain COX-1 but not vimentin,40,41 whereas the NKAIN4-positive cells have both antigens. Besides, the cup cells are mainly located at the ileum, 88 while the NKAIN4-positive cells are only present at the duodenum. Moreover, cup cells are positive to vimentin and negative to COX-1, 41 while the NKAIN4-containing cells express both markers (Fig. 4). The NKAIN4-positive cells are unique and do not belong to these cell types.
ECs are the most abundant type among EECs along the gastrointestinal tract. Their main secretory product is the biogenic amine serotonin, although they can also store peptide hormones such as secretin and substance P. The ECs accumulate serotonin in large dense-core vesicles in complex with acidic proteins such as chromogranin A and B. 89 These cells provide approximately 95% of the total body 5-HT. 90 Serotonin produced in the intestine has diverse endocrine roles; however, in the lamina propria, 5-HT acts as a paracrine mediator modulating epithelial growth, enterocyte secretion, intestinal barrier function, immune response, and enteric nerve activation. The ECs are considered sensors.48,51,91–94 The pleomorphic ECs have been identified along the gastrointestinal tract, while the NKAIN4 cells are only present in the duodenal mucosa. In addition, we demonstrated that the NKAIN4-positive cells store serotonin in small granules similar to neurosecretory vesicles, contrasting with the large dense vesicles in the ECs. Using the formaldehyde-induced fluorescence in 5-HT-containing cells and NKAIN4 immunolabeling, we determined that 39% of the total 5-HT-containing cells in the duodenal epithelium are NKAIN4-positive.
Acetylcholine Activates the NKAIN4-Positive Cells
Ach activates NKAIN4-positive cells, confirming these cells are cholinergic neurons. ACh activation was demonstrated, in ex vivo experiments, using neutral red to detect changes in membrane potential and Fluo-4 AM to evaluate in[Ca2+] dynamic in isolated villous-crypt units (Figs. 14–16).
Neutral red is a highly lipophilic colorant (uncharged at neutral pH) that readily enters the cells. On entry, it is protonated, trapped, and concentrated in the cytoplasm, with little dye associated with the cell plasma membrane. Neutral red changes its absorbance inversely with changing pH. Therefore, the intracellular acidification of neurons resulting from depolarizing/excitatory stimulation increases the fluorescence. Furthermore, its hydrophobic properties permit us to follow rapid changes in membrane potential. Depolarization alters the association of the neutral red with the plasma membrane, inducing changes in fluorescence. The pH-independent sensitivity is moderate and unlikely to account for the fluorescent changes reported for the neutral red. 35
In addition, Ca2+ imaging allows the identification of neurons and their circuits. Using these techniques, we have identified the activation of the NKAIN4-positive cells by ACh and its associated neuronal circuit that remotely triggers ECs. Serotonin secretion and the intracellular Ca2+ dynamic are intimately linked. EC cells release 5-HT from large dense-core vesicles in a Ca2+-dependent manner, presenting kinetics similar to the serotonin release from synaptic vesicles. 93 Dense chromaffin granules have a high luminal content of proteins, neuropeptides, and calcium. 95 In neuroendocrine cells, secretory vesicles constitute an active calcium reservoir. The presence of ryanodine receptors on secretory vesicles suggests that local Ca2+-induced calcium release from vesicles docked at the plasma membrane could participate in triggering exocytosis. 96 In addition, Jaffe et al. 97 reported, in peritoneal mast cells, ryanodine activated serotonin secretion and caffeine restored exocytic function to control levels in a calcium-free medium, demonstrating the role of intracellular Ca2+ stores in the process.
Finally, we showed that serotonin blocks, in a reversible way, the activation of the NKAIN4-positive cell by ACh (Fig. 16). These results suggest the potential existence of a negative feedback mechanism to control serotonin secretion by the ECs, modulated by the activity of the NKAIN4-positive cell.
In summary, we obtained immunohistochemical and functional evidence indicating that the NKAIN-positive cells from the duodenal epithelium are cholinergic and probably GABAergic neurons. These cells, representing 0.7% of the mucosal cell population, have a luminal knob-like terminal structure associated with an apparent cilia basal body, strongly suggesting that they are sensory neurons. At the basal pole, the epithelial neuron projects axonal extensions to the lamina propria, contacting glial cells, lymphoid nodules, and ECs. The basal projections reach the epithelial surface, forming a neural network in patches on the epithelium. Their connections with sub-epithelial nerve terminals, lymphoid nodules, and ECs would define an epithelial sensory system integrated by the epithelial neuron, glia, and neural fibers that reach the epithelial surface and immune and endocrine sub-epithelial structures.
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors contributed to the conception, design, acquisition, analysis, interpretation of the data, and critical revision of the manuscript for relevant intellectual content.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly supported by “Mision Ciencia” Grant 20070015856, Caracas, Venezuela.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
