Abstract
Aminopeptidase-A (APA) is a metalloprotease that cleaves N-terminal aspartyl and glutamyl residues from peptides. Its best-known substrate is angiotensin II (Ang II), the most active compound of the renin-angiotensin system (RAS). The RAS is involved in renal development. Most components of the RAS system are expressed in the developing kidney. Thus far, APA has not been studied in detail. In the present study we have evaluated the expression of APA at the protein, mRNA, and enzyme activity (EA) level in the kidney during nephrogenesis. Furthermore, we have studied the effect of inhibiting APA EA by injection of anti-APA antibodies into 1-day-old mice. APA expression was observed from the comma stage onwards, predominantly in the developing podocytes and brush borders of proximal tubular cells. Notably, APA was absent in the medulla or the renal arterioles. Inhibition of APA EA caused temporary podocyte foot-process effacement, suggesting a minimum role for APA during nephrogenesis.
Keywords
A
APA (EC 3.4.11.7) is a membrane-bound metalloprotease that cleaves N-terminal aspartyl and glutamyl residues from peptides. Its best-known substrate is Ang II (Wolf et al. 1997). APA has a widespread organ distribution, suggesting that this enzyme has a role in many diverse biological processes (Mentzel et al. 1996b).
Investigators have suggested that APA may be involved in blood pressure regulation and the pathogenesis of preeclampsia (Mitsui et al. 2004). In the adult mouse kidney, APA is expressed on podocytes and brush borders (BBs) of proximal tubular epithelial cells and, to a lesser degree, on juxtaglomerular (JG) cells, endothelial cells of peritubular capillaries (PTC), and in pars media of arteries. In our studies on membranous nephropathy, we have developed a panel of antibodies directed against APA (Mentzel et al. 1996a). Injection of a combination of antibodies that inhibit APA enzyme activity (EA) increased renal Ang II levels and induced acute and profound albuminuria (Gerlofs-Nijland et al. 2001). Monoclonal antibodies (MAbs) are highly specific for APA because we observed no binding upon injection into APA-deficient mice (Gerlofs-Nijland et al. 2001). These MAbs have provided us with tools to study the tissue distribution of APA (Mentzel et al. 1996b). In the present study we have evaluated the expression of APA at the mRNA, protein, and EA level during nephrogenesis in mice. In addition, we examined the effect of APA inhibition during nephrogenesis.
Materials and Methods
Animals
Female BALB/c mice in late pregnancy, aged 2–3 months, were purchased from Charles River Laboratories (Sulzfeld, Germany). BALB/c mouse embryos, embryonic days 18–19 (E18–19) postcoitum or 1-day-old mice were used to study nephrogenesis of the mouse kidney. In mice, nephrogenesis starts at E11–12 (forming metanephros) and continues for 7–10 days postnatally (P1–2). In mice, 80% of the glomeruli form after birth. Microscopic examination of kidneys from E18 embryos allows studying glomeruli at various developmental stages, ranging from the earliest developmental stage (vesicle formation) in the outer cortex to mature glomeruli in the inner cortex. One-day-old mice were used to study the effect of inhibiting APA EA during nephrogenesis. All procedures involving mice were approved by the Animal Care Committee of the University of Nijmegen and conformed to the Dutch Council for Animal Care and to National Institutes of Health guidelines.
Animal Experiments
For the distribution study, kidneys from E18 embryos were removed from BALB/c mice, snap frozen in liquid nitrogen for immunofluorescence (IF), enzyme histochemistry, and mRNA in situ hybridization, or immersion fixed for light microscopy (LM) and immunoelectron microscopy (IEM).
To study the effect of APA inhibition during nephrogenesis, we injected combinations of anti-APA antibodies in 1-day-old mice. Characteristics of the three rat MAbs used in this study (ASD-3, ASD-37, and ASD-41) were previously described by Mentzel et al. (1996a). Anti-APA MAbs were propagated in vitro by hollow fiber culture (Nematology Department, Agriculture University, Wageningen, The Netherlands). Injection of the combination ASD-37/41 inhibited the APA EA completely and induced massive acute albuminuria at day 1. In contrast, the combination ASD-3/41, which did partially inhibit APA activity on the podocyte and completely inhibited the EA of the BB, did not induce proteinuria. We used a single intravenous injection of a total dose of 0.8 mg of the nephritogenic combination ASD-37/41 or the non-nephritogenic combination ASD-3/41 (both with 1:1 weight ratio). Groups of mice (n=5) were studied 9 days, 21 days, and 3 months after injection. Urine samples were collected via spontaneously voiding at day 1 and via bladder puncture at days 9 and 21 and at 3 months, after which mice were killed and the kidneys processed for LM, IF, enzyme histochemistry, and electron microscopy (EM).
Morphological Studies
To study the expression of APA by IF, we have used the MAb ASD 3, an anti-APA MAb of subclass IgG1. All three rat MAbs used in this study (ASD-3, ASD-37, and ASD-41) gave the same staining pattern of APA. Kidneys from E18 embryos were snap frozen in liquid nitrogen. Two-μm-thick, acetone-fixed sections were incubated for 60 min at room temperature. Binding of the MAb was visualized with FITC-labeled rabbit anti-rat IgG containing 4% normal mouse serum (Dako; Glostrup, Denmark) as described previously (Assmann et al. 1992; Mentzel et al. 1996a).
Enzyme activity of APA was visualized by enzyme histochemistry according to Lojda and Gossrau (1980) as previously described, with the APA-specific substrate
APA mRNA was detected by RNA in situ hybridization using both a sense and an antisense 344-bp digoxigenin-labeled cRNA probe, as described previously (Mentzel et al. 1996a).
For LM and EM, the kidneys were immersion fixed in 2.5% glutaraldehyde dissolved in 0.1 M sodium cacodylate buffer, pH 7.4, overnight at 4C and washed in the same buffer. Tissue fragments were postfixed in palade-buffered 2% OsO4 for 1 hr, dehydrated, and embedded in Epon 812, Luft's procedure (Merck; Darmstadt, Germany). Semithin (1 μm) and ultrathin sections were cut on an ultratome (Reichert Ultracut S; Leica Microsystems, Vienna, Austria). The semithin slices were stained with toluidine blue and examined using LM. Ultrathin sections were stained with 4% uranyl acetate for 45 min and subsequently with lead citrate for 4 min at room temperature. Sections were examined in a JEOL 1200 EX2 electron microscope (JEOL; Tokyo, Japan).
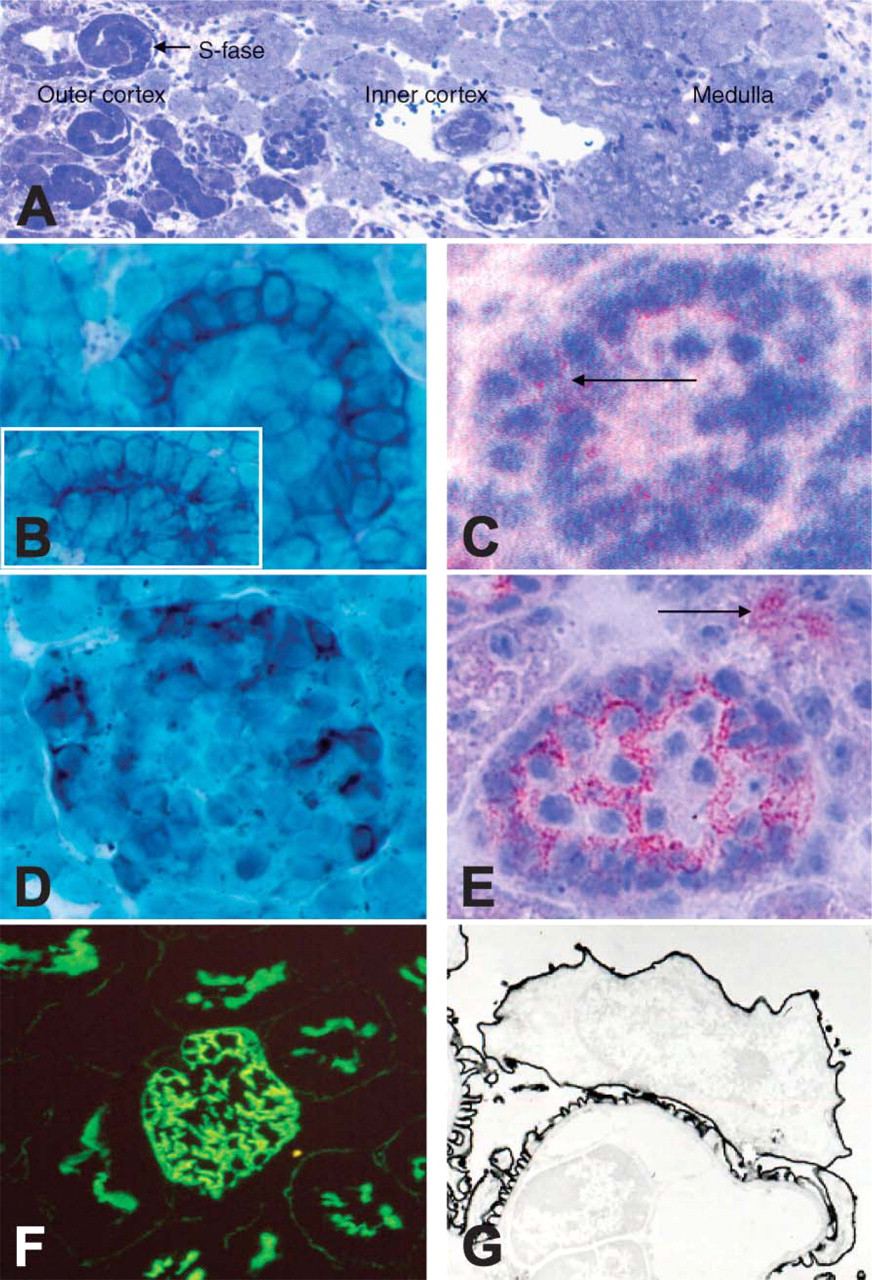
Developmental stages: kidneys from embryonic day 18 (E18) embryos show glomeruli at various developmental stages ranging from the earliest developmental stage (vesicle formation) in the outer cortex to glomerular maturation in the inner cortex (overview toluidine blue staining,
For IEM, the kidneys were immersion fixed in a mixture of 10 mM periodate, 75 mM lysine, and 2% paraformaldehyde, pH 6.2, for 3 hr. After rinsing several times in PBS, the embryonic kidneys were cryoprotected by immersion in 2.3 M sucrose, pH 7.2, for 1 hr and then frozen in liquid nitrogen. Twenty-μm-thick sections were incubated with Assman-Son-Dijkman (ASD) 3 for 18 hr at 4C. Binding of the MAb was visualized with peroxidase-labeled rabbit anti-rat IgG containing 4% normal mouse serum (Seralab/Sanbio; Uden, The Netherlands) as previously described. For IEM, ultrathin sections were cut on an ultratome (Reichert Ultracuts; Leica). Sections were examined in a JEOL 1200 EX2 electron microscope.
Results
In mice, nephrogenesis starts at E11–12 (metanephros formation) and continues for 7–10 days after birth. Formation of the kidney epithelium involves reciprocal inductive interactions between two mesoderm-derived structures, the uretic bud, an outgrowth of the Wolffian duct, and the adjacent metanephric mesenchyme. The bud branches into collecting ducts, and the mesenchyme differentiates into nephrons (Woolf and Loughna 1998; Piscione and Rosenblum 1999) In mice, 80% of the glomeruli form after birth. Microscopic examination of kidneys from E18 embryos reveals glomeruli at various developmental stages ranging from the earliest developmental stage (vesicle formation) in the outer cortex to mature glomeruli in the inner cortex. A low magnification overview shows several stages (Figure 1). The various stages of glomerular development can be easily distinguished by transmission electron microscopy. Figure 2 illustrates a typical example of a developing glomerulus in the comma stage (Figure 2), the S-stage (Figure 2), and the early capillary stage (Figure 2).
Histology and Immunofluorescence
In the earliest histologically definable glomerular stage, the comma stage (Figure 3), there was only trace staining for APA by immunofluorescence (Figure 3). Controls for background were completely negative. Its morphological shape and the presence of the earliest ingrowth of capillaries recognized by using our antibody ASD-13, which is specific for glomerular endothelium, identified the comma stage.
In the next stage, the S-stage (Figure 3), there was staining for APA of the cells lining the vascular cleft destined to become podocytes and with lower intensity also of the epithelial cells destined to become parietal epithelial cells. In addition, cells that will develop into the proximal tubule stained faintly positive for APA without a clear polarity (Figure 3D, arrow). In the more advanced early capillary stage (Figure 3E), staining of cells destined to become podocytes is more intense (Figure 3F). Staining is membranous and strongest closer to the capillary tuft. At this stage there is luminal staining of the proximal tubules, restricted to the BB (Figure 3F, arrow). In addition, there is still weak staining of the developing parietal epithelial cells. Finally, in the mature glomerulus (Figure 3G), APA staining shows a homogeneous podocytic pattern. In addition, the BBs of the proximal tubules stain for APA (Figure 3H). In the mature kidney, faint APA expression can also be observed in the granular JG cells and on the endothelial cells of the PTC (Figure 1F), whereas no expression is observed in the efferent and afferent arterioles, the distal tubules, the loops of Henle, or the collecting ducts.
To obtain more insight into the exact subcellular localization of APA during nephrogenesis, IEM on E18 embryos was carried out (Figure 4). In the comma stage there was only a faint APA expression (Figure 4). In the S-stage there was strong membrane staining of both developing podocytes and parietal epithelial cells. Staining was predominantly seen on surfaces lining Bowman's space (Figure 4). In the S-stage, proximal tubular epithelial cells showed some expression of the apical/lateral membranes (Figure 4B, arrows) and strong staining of cytoplasmic vesicles close to the luminal surface of the epithelial cells (Figure 4D). In the early capillary stage, membranes lining Bowman's capsule were still strongly positive, probably apical BB staining. In addition, early podocytes showed staining of apical, lateral, and basal membranes and sometimes carried cytoplasmic vesicles (Figure 4E). At this later stage the epithelial cells of the proximal tubules showed merely BB staining and no further staining of cytoplasmic vesicles (Figure 4F). In the mature kidney, IEM confirmed the membranous staining pattern of the podocytes (Figure 1G).
Enzyme histochemistry with a specific APA substrate on frozen sections of E18 embryonic kidneys showed that APA EA could be demonstrated in early stages (Figure 1) and further increased in glomeruli in a more advanced developmental stage, early capillary stage (Figure 1E). In the proximal tubules, APA EA could be demonstrated in the BB (Figure 1E, arrow).
We also determined APA mRNA expression during nephrogenesis by non-radioactive RNA in situ hybridization. Glomerular APA mRNA could be demonstrated in all developmental stages. Expression is seen clearly in cells destined to become podocytes (Figure 1B, S-stage). In addition, we could demonstrate APA mRNA in developing proximal tubules (Figure 1B, inset). In the early capillary stage, expression was restricted to the podocytes (Figure 1D). In the mature kidney, APA mRNA expression was restricted to the perinuclear zone of podocytes. Expression in other compartments could not be detected or was very low (Mentzel et al. 1996a). Table 1 summarizes our findings.
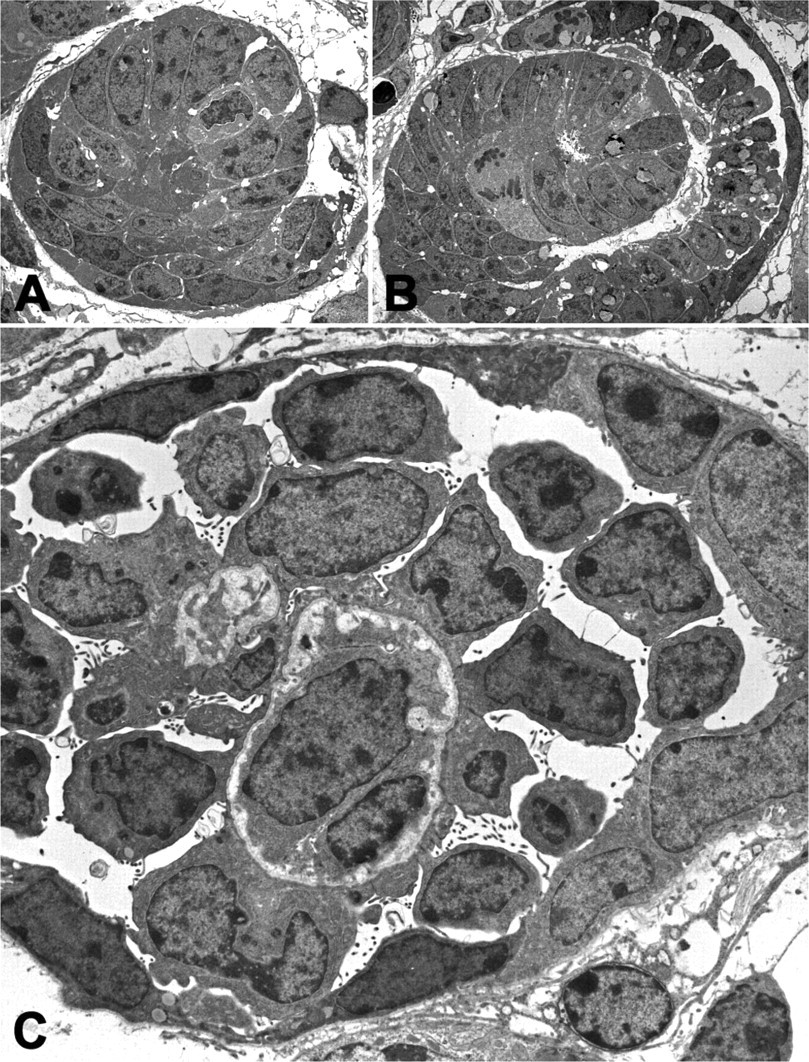
At the electron microscopical level the different stages and the developing podocytes could be easily distinguished: the comma-stage (
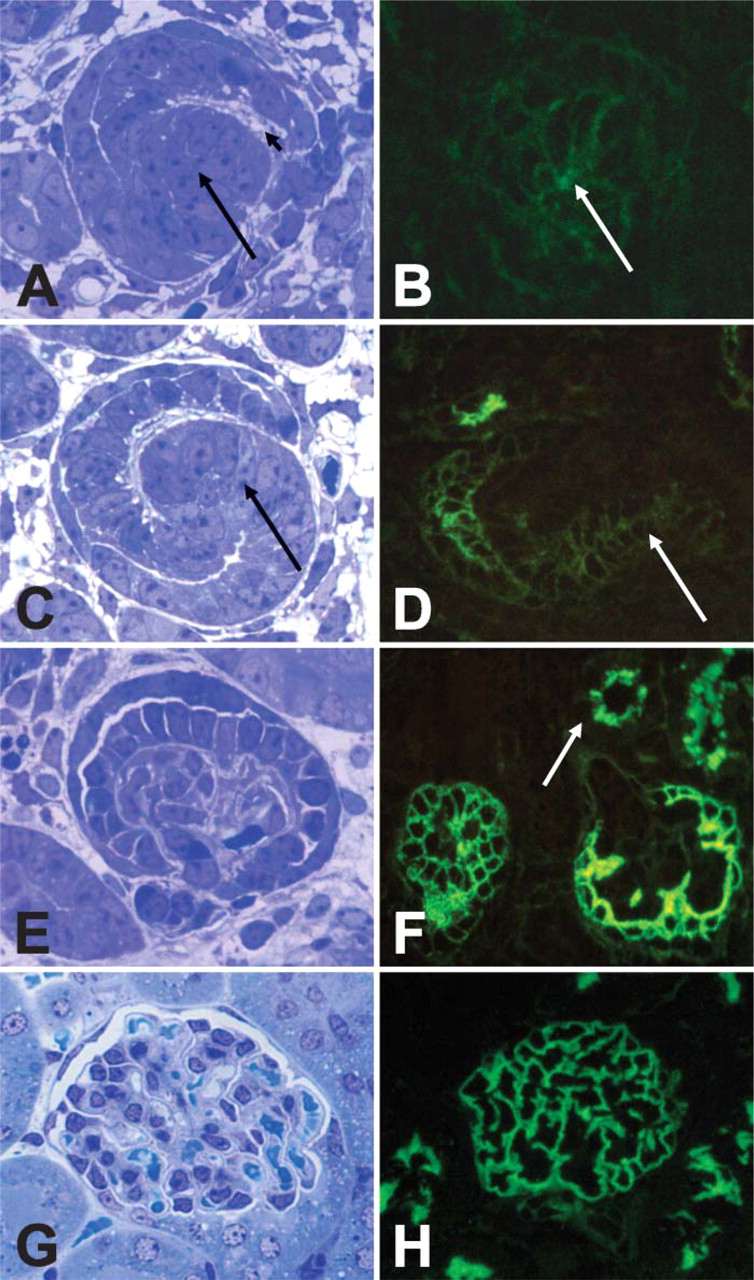
Expression of APA in the developing glomeruli studied by immunofluorescence: APA (demonstrated with the MAb ASD-3) could already be detected in the earliest histologically definable glomerular stage, the comma stage, and proximal tubular stages in the cortex but not in the medulla. In the comma stage the vascular cleft (arrowhead,
Injection of the nephritogenic APA antibody combination ASD-37/41 in 1-day-old mice completely blocked EA (Figure 5) and caused albuminuria at day 1 (60,000 ± 4000 μg/ml), whereas the combination ASD-3/41 albuminuria only partially blocked EA (Figure 5) and did not induce albuminuria (140 ± 21 μg/ml). APA EA was still absent at day 9 after injection of ASD-37/41 and was normalized at day 21 (Figure 5). Partial foot-process effacement was present at day 1 and persisted at days 9 and 21 (Figure 5E). These abnormalities were not accompanied by albuminuria at day 9 ASD-37/41 (240 ± 47 μg/ml); ASD-3/41 (120 ± 20 μg/ml); day 21 ASD-37/41 (180 ± 25 μg/ml); ASD-3/41 (120 ± 20 μg/ml); mice were followed for 3 months after injection of antibodies. At 3 months there were no abnormalities either by LM (Figure 5D), EM (Figure 5F), or enzyme histochemistry.
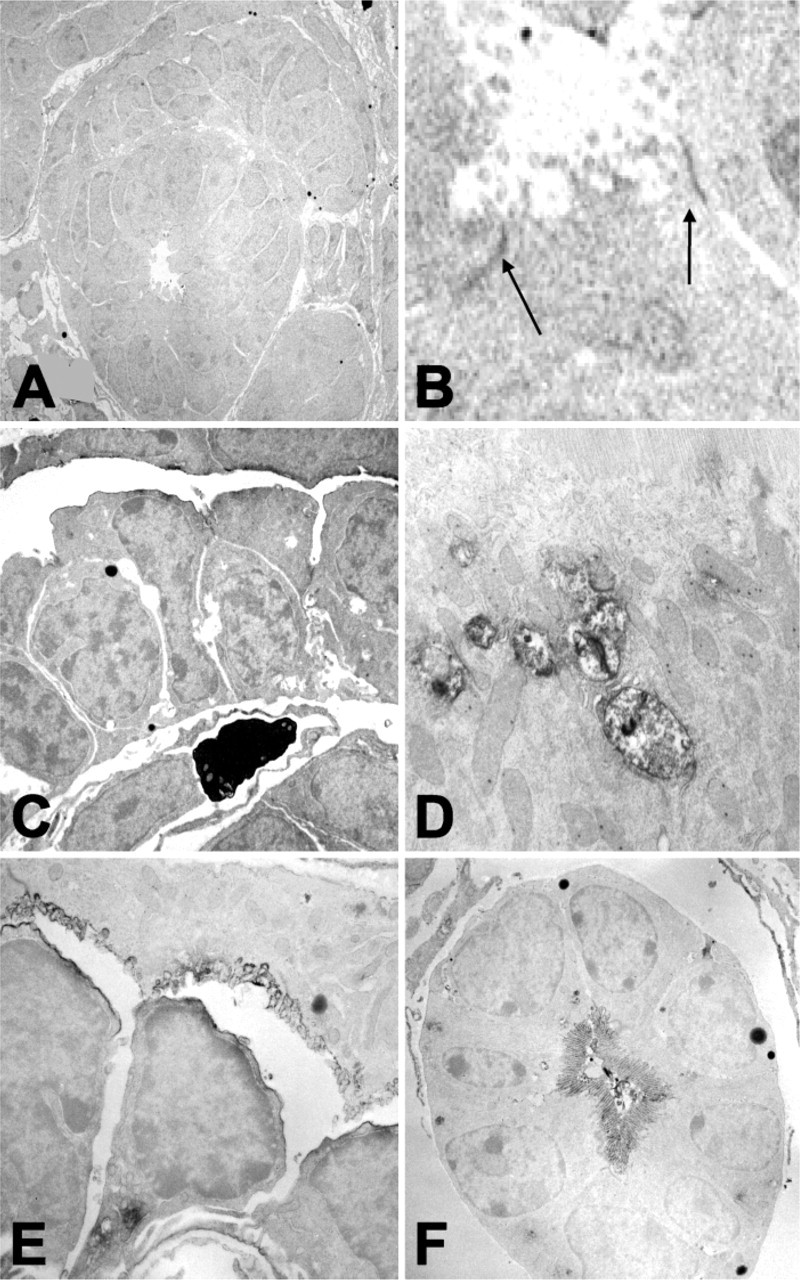
IEM on day E18 embryos: in the comma stage APA was faintly observed on the membranes of the early epithelial cells. APA was seen only on those epithelial cells that first came in contact with the in-growing endothelial cells of the vascular cleft (
Expression of aminopeptidase-A (APA) during kidney development

aE18–P1, embryonic day 18-postnatal week 1.
bPostnatal week 3.
cAPA expression was observed on endothelial cells of peritubular capillaries and in the pars media of arteries.
dScale from negative (−) to maximum (++++).
IF, immunofluorescence; IEM, immunoelectronmicroscopy; EA, enzyme activity; BB, brush border.
Discussion
From our study, it is evident that APA is expressed in the kidney during nephrogenesis. APA was detectable from the earliest stage of glomerular development, the comma stage, onwards. In later stages, APA expression was confined to the podocytes and the BB membranes of the proximal tubular epithelial cells. The subcellular localization of APA was confirmed by IEM (Mentzel et al. 1996b). We have confirmed the activity of APA by enzyme histochemistry. Of note, APA was not expressed in the medulla or in the endothelium of the renal arteries or arterioles. In the mature glomerulus only, faint expression was observed in the JG cells, the PTC, and pars media of arteries.
We were interested in APA because this enzyme is involved in the degradation of Ang II, the most active compound of the RAS system. Ang II is a cytokine that participates in renal damage and has vasoactive and profibrotic properties and contributes to kidney injury by causing hypertension and glomerulosclerosis. Impairment of Ang II degradation may influence Ang II action and, indeed, animal studies have suggested a role for APA in blood pressure regulation (Mitsui et al. 2003, 2004). All components of the RAS are highly expressed in the developing kidney. A role for Ang II in renal development has been suggested. The cellular distribution of AT(1) and AT(2) receptor mRNA in mouse kidneys at several embryonic stages and up to 3 weeks after birth by in situ hybridization is widespread. Expression is extra high at E18–P1 and is localized in tubular, epithelial, and endothelial cells and in the papilla. In support of this notion, pharmacological interruption of AT(1) receptor signaling in animals with ongoing nephrogenesis produces specific renal abnormalities characterized by papillary atrophy, abnormal wall thickening of intrarenal arterioles, tubular atrophy associated with expansion of the interstitium, and a marked impairment in urinary concentrating ability, suggesting a role for Ang II in renal development. Similar changes in renal morphology and function develop also in mice with targeted inactivation of genes encoding renin, angiotensinogen, ACE, or both AT(1) receptor isoforms simultaneously. Together these results clearly indicate that an intact signaling through AT(1) receptors is a prerequisite for normal renal development (Chen et al. 2004). An intact RAS system is also important for normal kidney development. However, in our experiments, blockade of APA EA for 9 days led only to temporary effacement of foot processes with no morphological abnormalities present after 3 months. Our findings are in accordance with studies demonstrating normal renal development in APA knockout mice (Lin et al. 1998) or in rats with an increased expression of the AT1a receptor on podocytes (Hoffmann et al. 2004). In view of the dependence of normal kidney development on Ang II, adverse effects of impaired Ang II degradation by blockade of APA might not be expected. Of note, we observed no APA expression in the areas of the kidney that are most dependent on intact Ang II for development i.e., the papilla, the collecting ducts, and the intrarenal arterioles. We observed APA expression in the podocytes and the BBs of the proximal tubules. The expression partially overlaps with the expression of the AT1 receptor. In the fetal kidney, high circulating levels of Ang II are present. It has been demonstrated that the expression of APA increases upon stimulation by Ang II (Mitsui et al. 2004). Upregulation of APA is directed by the AT1a receptor (Ino et al. 2003).
Therefore, it is possible that the expression of APA in the developing kidney follows, and is dependent on, the local Ang II levels. We cannot exclude that APA is becoming more important after birth when Ang II exerts negative effects. These negative effects are shown via short-term infusion of Ang II that causes renal injury in rats, leading to the development of salt-dependent hypertension in these rats in later life (Lombardi et al. 1999). Furthermore, Ang II is involved in the development of hypertension in the spontaneously hypertensive rat because ACE inhibitors administered at a young age prevented hypertension. Development of hypertension has been attributed to an increased expression of the AT1a receptor in the proximal tubules (Cheng et al. 1998). Thus, future studies should evaluate the effects of APA inhibition in young mice on long-term blood pressure regulation.
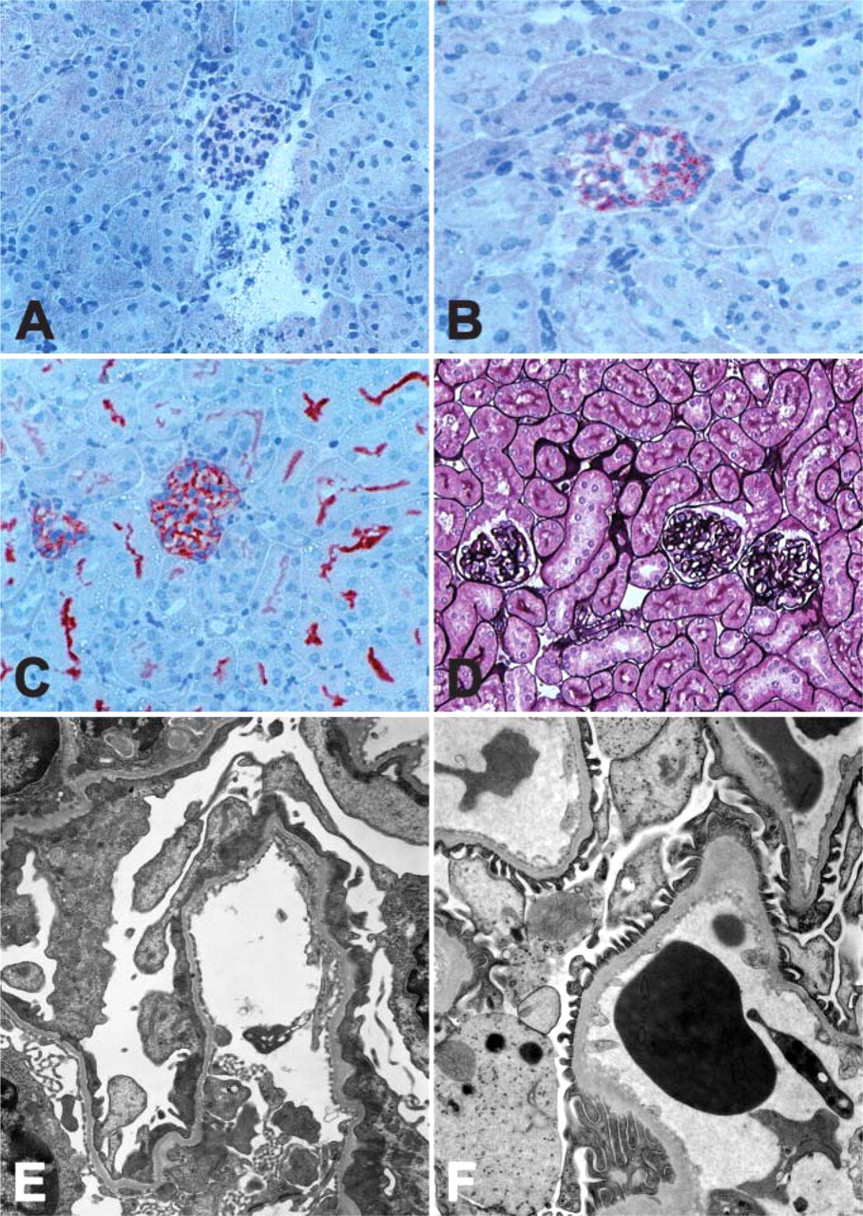
Blocking APA enzyme activity: injection of the nephritogenic APA antibody combination ASD-37/41 in 1-day-old mice blocked EA completely for 9 days (
In conclusion, APA is highly expressed in the developing kidney. Complete inhibition of APA EA during nephrogenesis for 9 days after birth led only to podocyte effacement at days 9 and 21 that disappeared after 3 months, suggesting a minimum role for APA in embryonal development.
